Abstract
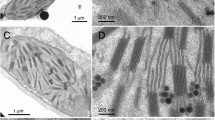
Most chloroplasts in higher plants have a differentiated thylakoidal system into zones with a potential for membrane adhesion forming grana stacks and zones of single unappressed membranes which lack this adhesion potential and form end-membranes, margins of grana and stroma lamellae. This structural differentiation is also accompanied by differences in function: Photosystem 2 (PS 2) together with most of the light-harvesting complex (LHC) is contained mainly in the appressed grana while Photosystem 1 (PS 1) is predominantly in the non-appressed membranes (Andersson, Haehnel 1982) together with the ATP synthetase (CF0 and CF1) (Faludi-Dániel et al. 1983). Although this differentiation is a very common ultrastructural feature, the nature and the control of membrane adherence is not understood completely. A well established hypothesis developed by Barber (1980) points to the importance of the low surface charge density as a prerequisite for membrane assembly. In the granum region negative charges originating from the occasionally occurring PS 1 particles are screened by the abundant LHC plus PS 2 particles and/or neutralized by cations. In the stroma thylakoids, however, negative surface density could not be sufficiently reduced by cation treatment to achieve a complete assembly (Barber et al. 1980). Here we show that EDTA washing of stroma thylakoids can unmask such regions of low surface charge density and facilitate the adherence of single membranes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Andersson B and Haehnel W (1982) Location of photosystem I and photosystem II reaction centers in different thylakoid regions of stacked chloroplasts, PEBS Lett. 146, 13–17.
Avron M (1960) Photophosphorylation by Swiss-chard chloroplasts, Biochim. Biophys. Acta 40, 257–272.
Barber J (1980) An explanation for the relationship between salt-induced thylakoid stacking and the chlorophyll fluorescence changes associated with changes in spillover of energy from photosystem II to photosystem I, PEBS Lett. 118, 1–10.
Barber J, Chow WS, Scoufflaire C and Lannoye R (1980) The relationship between thylakoid stacking and salt induced chlorophyll fluorescence changes, Biochim. Biophys. Acta 591, 92–193.
Brangeon J (1974) Structural modifications in the lamellar system of isolated Zea mays chloroplasts under different ionic conditions, J. Microscopie 21, 75–84.
Faludi-Dániel Á, Schmidt O, Szczepaniak A and Machold O (1983) Distribution of characteristic membrane proteins in granum and stroma thylakoids, Eur. J. Biochem. 131, 567–570.
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde-fixative of high osmolarity for use in electron microscopy, J. Cell Biol. 27, 137A–138A.
Nakatani HY, Barber J and Forrester JA (1978) Surface changes on chloroplast membranes as studied by particle electrophoresis, Biochim. Biophys. Acta 504, 215–225.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1984 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Mustárdy, L.A. (1984). Increased Stacking Capacity by Modified Thylakoid Surfaces. In: Sybesma, C. (eds) Advances in Photosynthesis Research. Advances in Agricultural Biotechnology, vol 3. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-4973-2_17
Download citation
DOI: https://doi.org/10.1007/978-94-017-4973-2_17
Publisher Name: Springer, Dordrecht
Print ISBN: 978-90-247-2944-9
Online ISBN: 978-94-017-4973-2
eBook Packages: Springer Book Archive