Abstract
Human use of land has reduced the amount of carbon (C) in terrestrial ecosystems, probably since the first use of fire as a tool for clearing land thousands of years ago. Because variations in climate have also affected C storage over this period, it is difficult to attribute long-term changes in terrestrial C to direct human activity. Over the last 150–300 years, however, reconstructions of land use and land-use change suggest that between ∼100 and ∼200 Pg (1 Pg = 1015 g) C were lost from land, largely from the conversion of forests to agricultural lands. This loss of C over the past century or so is greater than the loss attributable to human activity for all of time before 1850. Most of the loss since 1850 has been from forest biomass, while the loss of C from soil organic matter (SOM) as a result of cultivation is estimated to have contributed ∼25% of the net loss. The restoration of forests on cleared lands could, in theory, re-carbonize the biosphere with 100–200 Pg C; but most of these lands are currently in use and unlikely to be returned to forests. Management practices would have to reverse the centuries-long loss of C.
For most of the last 300 years, the net annual loss of C from land use seems to explain (i.e., is roughly equivalent to) the net terrestrial flux of C to the atmosphere. Starting near the middle of the twentieth century, however, the annual net emissions of C from land use appear to have been offset by a terrestrial C sink not directly related to land use. The explanations for this residual terrestrial sink include carbon dioxide (CO2) fertilization, nitrogen (N) deposition, variations in climate, and, possibly, a centuries-long reduction of natural disturbances. Much of the offsetting C sink is thought to be in forests. The residual C sink indicates that terrestrial ecosystems, despite land use, have removed C from the atmosphere over the last decades. The magnitude of this sink is large relative to the effect human management could have, but recent evidence suggests that the sink may be beginning to saturate. If the residual terrestrial sink were to disappear or become an additional source of C as a result of climate change, managing the global C cycle would be much more difficult than envisioned today.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Carbon management
- Deforestation
- Global carbon budget
- Land use
- Residual carbon sink
- Terrestrial carbon pool
- Biotic carbon pool
- Soil carbon pool
- Residual C sink
- Missing C
- Historic C loss
- Deforestation
- Soil degradation
- Global C budget
- Direct human effects
- Croplands
- Pastures
- Settled lands
- Wood harvest
- Reforestation
- Fire management
4.1 Introduction
The global carbon (C) cycle is not in equilibrium. The C accumulated in fossil reservoirs over millions of years is being mobilized into the atmosphere over a few centuries and is being redistributed into the oceans and land (Fig. 4.1). Human management of land has offset to a large extent the terrestrial uptake of fossil C, but the net terrestrial sink of recent decades may be changing. This chapter reviews historical changes in the global C cycle, with an emphasis on changes in terrestrial C storage 1850–2005.
The three major drivers of changes in terrestrial C storage are: (1) natural processes, (2) direct effects of human management, and (3) indirect effects of human activity. Regarding the natural processes of de- and re-carbonization, the highest C densities on land appear in forests, wetlands, tundra, and prairies, as a result of natural processes. Two aspects of these processes are important here. First, most of these high C densities accumulated over centuries or millennia, and the accumulation rates today are generally low. Nevertheless, large areas of low accumulation rates can add up to be globally significant.
Second, these C-rich ecosystems have been sinks in the past and may be sinks today where they still exist (Luyssaert et al. 2008; Dommain et al. 2011; Donato et al. 2011). However, the direct effects of management, such as draining and mining of wetlands for crops, forestry, and fuel, have released to the atmosphere much of the C built-up in these ecosystems over centuries. More than that, the loss of these ecosystems has eliminated the potential C sink that might otherwise have persisted (Gitz and Ciais 2003; Bridgham et al. 2006; Strassmann et al. 2008).
The third driver of changes in terrestrial C storage is the indirect effect of human activity, such as increasing concentrations of carbon dioxide (CO2) in the atmosphere, increased rates of deposition of reactive nitrogen (N), and changes in climate. These environmental factors may affect the rates of photosynthesis and respiration, thereby potentially changing the storage of C on land. The factors are the indirect effects of human activity, in contrast to the direct effects of land management.
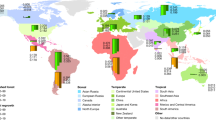
This chapter considers all three effects, but focuses on the direct effects of human management; i.e., land use and land-use change. In contrast to most of the following chapters, which consider the potential for re-carbonization of the biosphere, this chapter deals largely with de-carbonization because the net effect of land management over millennia has been to reduce the amount of C in terrestrial ecosystems (Kaplan et al. 2009; Ellis 2011; Pinter et al. 2011). Furthermore, the rate of loss has accelerated over the last ∼150 years, although not equally in all regions (Fig. 4.2). On the contrary, the gradual loss, globally, is composed of recent, accelerated losses in tropical countries and little change or re-carbonization in developed countries of the temperate zone and boreal regions.
4.1.1 The Global Carbon Budget 1850–2005
With the start of the industrial revolution in the eighteenth century, fossil C began being increasingly added to the atmosphere. Carbon was already being emitted from the expansion of agricultural lands, and not until half way through the twentieth century did the annual emissions of C from fossil fuels exceed the annual net emissions from land-use change (Fig. 4.1). Between 1850 and 2005 the emissions from fossil fuels (and cement production) were 320 Pg C, while the emissions from land-use change were 156 Pg C. Total anthropogenic emissions of C were, thus, 476 Pg C. The amount of C in the atmosphere increased by 198 Pg C (∼33%) during this 155-year period, from ∼280 ppm (595 Pg C) to ∼374 ppm (793 Pg C) (Table 4.1). The accumulation of C in the atmosphere was ∼42% of total emissions; land and oceans accumulated 28% and 30%, respectively. The fraction remaining in the atmosphere has increased slightly, averaging 37% between 1850 and 1955, and 45% between 1956 and 2005.
One interesting feature of Fig. 4.1 is that the terrestrial component of the budget appears twice. It appears as a net source of C from land use and land-use change. Before ∼1950 the net emissions from land management were larger than global emissions of C from fossil fuels. Obviously, the proportion of emissions from fossil fuels has grown dramatically since then, and the proportion of total emissions from land use has declined from ∼30% over the period 1850–2005 to 20% for the 1990s to 15% in 2008.
But the land has also been a net sink for C, at least for the last several decades, for reasons that are not well understood. This ‘residual terrestrial sink’, unlike the other terms, is not independently determined. Rather, it is determined from the other terms in the global C budget and the requirement that sources and sinks must balance (Table 4.1 and Fig. 4.1). The residual terrestrial sink does not include those C sinks that result from management; e.g., regrowth following wood harvest or agricultural abandonment. The latter sinks are the result of direct human activity and are included in the net land use term. The residual terrestrial sink is the result of indirect and/or natural effects. It is worth noting that before ∼1900 the annual residual terrestrial sink was close to zero. This implies that the net loss of C from direct human management was nearly equivalent to the net terrestrial flux. A natural or indirect effect is not apparent until after 1900.
Possible explanations for the residual terrestrial C sink include errors or omissions in the estimated net flux from land-use change or errors in the other terms of the global C balance (fossil fuels, atmosphere, oceans).
On the other hand, the residual terrestrial C sink may be explained by terrestrial processes not included in analyses of land use and land-use change. There are three general processes potentially responsible. First, many management practices are not included in analyses of land use and land-use change, although the major practices are described in the following chapters. Second, natural disturbance regimes (including recovery) may act independently of land use and land-use change, and may themselves be changing, causing C to accumulate (Marlon et al. 2008; Wang et al. 2010). It must be noted, however, that in many regions the effects of climate change (droughts and fires) appear over the last decades to be causing additional C to be lost rather than accumulated (Gillett et al. 2004; Westerling et al. 2006; Kurz et al. 2008). Finally, C stocks may be influenced by environmental changes in climate, CO2, or biologically available N. For the last few decades the net effects of these three processes has been to sequester C on land, but their relative contributions are unknown.
The important questions for the residual terrestrial sink, globally or locally, are:
-
When did it become significant in the net balance?
-
How large is it now?
-
Where is it?
-
What are its causes?
-
Will it persist?
A recent analysis of forest inventories suggests that at least a portion of the residual sink is in forests, worldwide (Pan et al. 2011). Regarding the sink’s persistence, the thawing of permafrost in response to the current and committed global warming is likely to expose labile organic C to decomposition and may increase the loss of C from land. This positive feedback to the warming is an example of a process that would reduce the residual sink.
The interactions of direct human effects, indirect effects, and natural effects on terrestrial C emissions are complex. The component measured most directly seems to be the direct effects; i.e., the changes in C due to land use and land-use change. Estimates of this change are important for both political and scientific reasons. The political reasons relate to assigning C credits and debits as incentives to reduce emissions of C through management (e.g., REDD, Reduced Emissions from Deforestation and forest Degradation). The scientific reasons relate to understanding the global C cycle and, in particular, the rate and extent of feedbacks between climate change and the C cycle. Arguably the most important feedback is the effect of climate change on terrestrial and oceanic C sinks. Over the last several decades, those sinks have been responsible for removing about ∼55% of anthropogenic C emissions (Le Quéré et al. 2009) (Table 4.1). Remarkably, these sinks have increased in proportion to emissions. The single best indicator of whether the sinks are continuing to grow is the airborne fraction, the ratio of growth in atmospheric C to total emissions (land use and fossil fuel). Of the three terms needed to evaluate the airborne fraction, the net emissions from land use and land-use change are the most uncertain. The trend of annual emissions over the last three decades is known barely well enough to suggest that the airborne fraction is increasing (Canadell et al. 2007; Le Quéré et al. 2009), but that conclusion has been challenged (Knorr 2009; Gloor et al. 2010). Reducing the uncertainty in the land-use flux would enable a more precise measure of trend in the airborne fraction.
The calculation of a residual terrestrial sink for the globe suggests that residual sinks (and sources) exist at other scales as well; that is, the changes in C contributed by land use and land-use change in individual regions or sites may be different from the sources and sinks actually measured with forest inventories or fluxes of CO2. Measured changes include natural or indirect effects as well as the direct effects of land management. For example, the net sink of C in Europe’s terrestrial ecosystems (Janssens et al. 2003) is not necessarily attributable to management.
Most ecosystems where C is accumulating are recovering from an earlier disturbance, either short term (e.g., fire) or long term (climatic variations). “Disturbance” is perhaps not the appropriate term to describe climatic variations, but terrestrial C storage varies significantly over glacial cycles. The warming that followed the last glacial maximum increased terrestrial C storage by 550–694 Pg C (Prentice et al. 2011). This increased storage associated with warming is larger than total anthropogenic emissions (fossil and land use) since ∼1850 (476 Pg C) (Table 4.1). The magnitude of this change, attributable to natural processes, illustrates the difficulty of separating climatic from land-use effects in pre-historic times (Pinter et al. 2011; Reick et al. 2011). It also reminds us that management effects may be overwhelmed by natural processes. Today natural processes on land and in the oceans are removing about 55% of anthropogenic emissions, but in past warming periods land and ocean have been additional sources of C.
4.2 Direct Human Effects on De- and Re-carbonization
Direct human effects on terrestrial C storage from land use, land-use change, or management, are divided here into changes before 1850 and changes since 1850.
4.2.1 Losses Before 1850
Estimates of the amount of terrestrial C lost before 1850 (Table 4.2) are based on a combination of three sets of data: estimates of natural vegetation cover, the associated C densities (in vegetation and soil), and the conversion of those natural covers to croplands and pastures, the latter based on a combination of agricultural maps and population density. Four estimates of pre-historic C loss range between 45 and 114 Pg C (Table 4.2).
4.2.2 Losses Between 1850 and 2005
Two approaches have been used to calculate the more recent losses of C from land use and land-use change. One approach uses process-based ecosystem models to estimate biomass density (e.g., Le Quéré et al. 2009 for some contemporary models). These models simulate spatial and temporal variations in ecosystem structure and physiology. They differ in detail with respect to number of plant functional types and number of C pools. To simulate the correct biomass density, however, the models account for disturbances and recovery, whether natural or anthropogenic.
A second approach uses empirically-based response curves to define annual per hectare changes in C density (Mg C ha−1 year−1) as a result of management (Houghton 1999, 2003). Carbon density declines with management as a result of burning and decay; it increases as a result of forest growth (recovery) and some forms of land management. Rates of decay and regrowth vary by geographic region, ecosystem type, and type of land use. Changes are defined for living vegetation (above- and belowground), coarse woody debris, wood products, and soil organic matter (SOM). The per hectare changes are initiated each year by the areas affected by land use or land-use change; that is, by the areas logged or the areas converted from one ecosystem to another. A C-tracking model sums the changes per hectare over all of the hectares affected by management. Hectares unaffected by management are assumed to be unchanged with respect to C density.
This second approach was used in the analyses reported by Canadell et al. (2007) and Le Quéré et al. (2009), which extended Houghton’s (2003) earlier work to 2005 using rates of deforestation from the U.N. Food and Agriculture Organization (FAO) (FAO 2006) and rates of wood harvest from FAOSTAT (FAO 2009). Many of the results presented here have not previously been published and are cited here as Houghton, this study. Preliminary estimates of C emissions based on the most recent Forest Resources Assessment (FRA) from FAO extended Houghton’s analysis to 2010 (Friedlingstein et al. 2010), but those preliminary estimates are not included here. They have only a small effect on the long-term fluxes (1850–2005).
The total net loss of C from land as a result of land use and land-use change over the period 1850–2005 is estimated, using both methods, to have been 108–188 Pg C (Table 4.2). Annual rates of net C loss increased from ∼0.5 Pg C year−1 in 1850 to ∼1.5 Pg C year−1 in 2005 (Fig. 4.3), but the regional contributions to this global trend have varied. Until the mid-twentieth century, the major losses of C were from non-tropical regions (North America, Europe, Russia, Australia) as a result of wood harvests and agricultural expansion. After ∼1950 deforestation for agricultural expansion was predominantly in tropical regions. The emphasis in this chapter on global, tropical, and non-tropical regions hides the fact that many sub-regions may have been sinks for C as a result of forest growth (Kauppi et al. 2006; Pan et al. 2011). In fact, most regions include both sources and sinks of C as a result of past and present management practices. Figure 4.4 shows the net source/sink history of individual regions.
4.2.2.1 Deforestation
The amount of C lost from land or sequestered as a result of land-use change and management practices depends on two factors: the total areas affected by any particular land use or land-use change and the per hectare changes in C density associated with a management practice. These two factors are described below for the human activities believed to be responsible for the largest changes in C storage: deforestation, degradation, reforestation, and other management activities.
Croplands. Globally, the conversion of lands to croplands has been responsible for the greatest loss of C from land-use change (91 Pg C over the period 1850–2005) (Houghton, this study). The emissions are large because the global area of croplands has grown substantially in the last one and a half centuries and because the changes in C stocks per hectare are large when lands, especially forests, are converted to croplands. The estimated net release may be an overestimate, however, because it is based on an estimate of change that is larger than other estimates (Table 4.3).
Permanent croplands in 2000 are estimated to have covered 1,414–1,750 × 106 ha (Table 4.3), or between 10% and 12% of the Earth’s total land surface (12–15% of the productive land surface – rock, sand, snow, ice, and water bodies excluded). Estimates of the increase over the last 150 years (1850–2000) vary from 875 to 1,035 × 106 ha, a doubling or tripling, depending on the estimates. The three different estimates of global cropland area are based on data initially published by Ramankutty and Foley (1999) (SAGE), Klein Goldewijk (2001) (HYDE), and Houghton (this study). Ramankutty and Foley (1999) estimated changes in croplands from 1700 to 1992 from historical cropland inventories. Klein Goldewijk (2001) used population density to estimate the spatial and temporal distribution of croplands from 1700 to 1990. The original SAGE and HYDE data sets have been compared (Klein Goldewijk and Ramankutty 2004), revised (Klein Goldewijk and van Drecht 2006), and used by others (Hurtt et al. 2006; Pongratz et al. 2008; Strassmann et al. 2008).
The changes in vegetation and soil, at least in the top meter, that result from clearing and cultivation are among the changes in terrestrial C stocks best documented. Essentially all of the initial vegetation is replaced by crops, so if the initial vegetation and its biomass are known, it is, in principle, straightforward to calculate the net loss of C associated with clearing. Because forests hold so much more C per unit area than grasslands, the loss of C associated with cropland expansion depends primarily on whether the croplands were claimed from forests or open lands. The variation in C stocks of different crop types is relatively small as long as tree (permanent) crops are differentiated from herbaceous crops. Some uncertainty results from estimating the time it takes for the release or uptake of C to occur. How much of the biomass is burned at the time of clearing? How much woody material is removed from site (wood products) and not decayed immediately? Answers vary across regions and through time (e.g., Morton et al. 2008). Estimates of annual sources and sinks depend on the answers, yet site-specific data are generally lacking. A few case studies usually provide the values used in calculation of C emissions and uptake over large regions.
On average, soil organic carbon (SOC) in the upper meter of soil is reduced by 25–30% as a result of cultivation, and this average has been documented in a large number of reviews (Mann 1985, 1986; Detwiler 1986; Schlesinger 1986; Johnson 1992; Davidson and Ackerman 1993; Post and Kwon 2000; Guo and Gifford 2002; Murty et al. 2002). There is some variation about this average, but the loss is broadly robust across all ecosystems, despite the variety of soil types, cultivation practices, and decomposition processes. Below a meter SOC seems to be more dynamic than previously thought (Schipper et al. 2007), and these dynamics add uncertainty to the estimated change.
The remaining uncertainty with respect to changes in SOC in response to cultivation concerns the fate of C lost from soil. Is all of it, in fact, released to the atmosphere, as most analyses assume, or is some of it eroded and moved to a different location, perhaps buried in anoxic environments and thereby sequestered? Comparison of erosion rates with the amount of organic C in freshwater sediments suggests that some of the C lost through erosion may accumulate in riverbeds, lakes, and reservoirs (Stallard 1998; Smith et al. 2001; Berhe et al. 2007). To the extent that this is so, the calculated loss of C from cropland expansion are overestimated.
When croplands are abandoned, C re-accumulates in vegetation as the land reverts to the natural ecosystem. The greater the biomass of the returning ecosystem, the greater the long-term C sink associated with recovery. In the short term, however, the magnitude of the annual sink for a particular parcel of land will vary with rate of recovery, which may be affected by the intensity of previous land use or by biophysical factors, such as distance from seed source, herbivory, soil fertility, or climatology (Uhl et al. 1988; Kozlowski 2002). The rate of recovery of vegetation can also depend on both climate conditions (growing season length) and soil type (Johnson et al. 2000). Soil organic C may also re-accumulate after abandonment of cultivation, although the rates of C accumulation in mineral soil are generally modest, albeit less well known (Post and Kwon 2000), especially when compared to the much faster rates of C accumulation in vegetation, surface litter, or woody debris (e.g., Harrison et al. 1995; Huntington 1995; Barford et al. 2001; Hooker and Compton 2003). Globally, C accumulation in mineral soils recovering from past cultivation is likely to amount to less than 0.1 Pg C year−1 (Post and Kwon 2000), although this estimate could change as deeper soil profiles are investigated further.
Pastures. The global expansion of pastures over the last 150 years is estimated to have caused a de-carbonization of 24 Pg C, the second largest loss from land-use change (Houghton, this study). The estimate is probably low because it is based on a low estimate of pasture expansion (67% and 80% lower than the estimates from the HYDE and SAGE data sets). The original SAGE data set (Ramankutty and Foley 1999) did not consider pastures. Changes in pastures attributed to SAGE were generated by merging the HYDE data on pastures with the cropland data from Ramankutty and Foley (1999) (Hurtt et al. 2006; Shevliakova et al. 2009).
In the year 2000, pastures covered between 2,650 and 3,378 × 106 ha, or 18–23% of the Earth’s land surface (Table 4.3). Pastures and croplands, together, covered 4,040–4,775 × 106 ha, or 28–33% of total land area.
The net loss of C from changes in pasture area has been less than the loss from cropland expansion, despite the larger growth in pasture area, because many pastures expanded into natural grasslands rather than forests (thus changing aboveground C stocks little) and because pastures are generally not cultivated, and thus lose little C from soils. The primary exception to this grassland origin of pastures has been in Latin America where cattle pasture is still the main driver of deforestation.
The changes in SOC resulting from the conversion of forests to pastures are highly variable, however, with both increases and decreases observed (Post and Kwon 2000; Guo and Gifford 2002; Osher et al. 2003; Parfitt et al. 2003). For example, pasture soils cleared from forests in the Brazilian Amazon have been shown to lose C in some cases and gain it in others (Neill and Davidson 2000). The direction of change may be related to rainfall, site fertility, fertilizer practices, species of grass planted, or other factors that govern site productivity. In a meta-analysis of 170 studies, Guo and Gifford (2002) observed a modest mean increase in SOC (about 10%) in upper soil layers (<100 cm) when forests were converted to pastures; however, some sites had large C gains and others had large losses. When pastures are converted to croplands, SOC is lost as a result of cultivation. It should also be noted that overgrazing, particularly in dry lands, often leads to a loss of C from soil (Lal 2001).
Shifting cultivation. Shifting cultivation is a rotational form of agriculture, where crops alternate with periods of forest recovery (fallow). The initial clearing for shifting cultivation requires land conversion, most often forest or savanna. In terms of historical de-carbonization, shifting cultivation is fifth in importance, releasing ∼9 Pg C over the last 150 years.
The areas in shifting cultivation and fallow are not well documented. The FAO includes the cropping portion of shifting cultivation in “arable and permanent crops” and excludes the fallow areas if they are older than 5 years, but these definitions may not be applied consistently by all countries reporting.
Houghton and Hackler (2006) estimated the areas annually deforested for shifting cultivation in Africa when the loss of forests reported by the FAO (2006) was greater than the increase in permanent croplands and pastures (FAOSTAT 2009). The assumption that the difference was explained by shifting cultivation seemed consistent with the definitions used by the two sources within the FAO. Subsequent revisions to FAOSTAT (2009) suggest the differences were more likely due to uncertainties than to real changes in agricultural area. However, errors in assigning deforestation to shifting cultivation, as opposed to permanent cropland, affect the attribution of C fluxes from one land use to another, but they do not have a large effect on the calculated net flux of C, which is determined more by the rate of deforestation than by the end land use.
According to the data and assumptions used to characterize the areas and changes in shifting cultivation, the practice is estimated to have occupied 411 × 106 ha in 2000, about 3% of the Earth’s land surface. The increase in shifting cultivation (1850–2005) was 161 × 106 ha, only 10% of the increase in croplands or pastures. Neither Ramankutty and Foley (1999) nor Klein Goldewijk (2001) included fallow areas in their estimates of croplands and pastures, but shifting cultivation is included in the data set assembled by Hurtt et al. (2006). Fallow periods can be long or short, and generally the stocks of C in fallow forests re-cleared for cultivation are less than the stocks in undisturbed forests. Because the cultivation does not involve tillage, the loss of C from soil is less than the loss under cultivation of “permanent” croplands. Thus, the net per hectare changes in C stocks (both biomass and soil) are smaller under shifting cultivation than under permanent cultivation.
In many areas of tropical Asia and Africa, the fallow periods are being reduced as land becomes scarce (Myers 1980; Uhlig et al. 1994). Often the shortened fallow does not allow the recovery of nutrients necessary for crop production, and this intensification may lead to an increase in degraded lands that support neither crops nor forests, and a gradual reduction in C stocks. Some of these degraded lands gradually return to forest, but the changes are not systematically documented either on the ground or with satellites (Grainger 2008, 2009).
Settled lands. Because the area of urban ecosystems is small, globally, <0.5% (Schneider et al. 2009) to 2.4% (Potere and Schneider 2007) of the land surface, urban areas have been ignored in most estimates of C emissions from land use and land-use change. However, exurban areas were nearly 15 times greater than urban areas in the U.S. in 2000 (Brown et al. 2005). Furthermore, much of the deforestation in developed countries and China is currently for residential, industrial, and commercial use rather than for agriculture (Jeon 2011). The magnitude of net C emissions from the expansion of settled lands is uncertain. Newly established areas may be net sources initially but may become net sinks as trees are re-established. In arid areas settlements may become sinks if irrigation is used to expand the areas vegetated or to increase the C density of existing vegetation and soil.
4.2.2.2 Degradation
Degradation is defined here as a reduction in C density within a land cover.
Wood harvest. The harvest of wood from forests can increase the amount of C on land if the wood products have a long residence time and, thereby, accumulate enough mass to offset the reductions in forest biomass. In practice, however, little of the C initially held in the forest ends up in long-term products, especially because half of global harvests are for fuelwood (FAOSTAT 2009). Furthermore, although a constant rate of logging would eventually yield a net flux nearly zero, as decay and regrowth offset each other, as long as rates of harvest are increasing, the losses in biomass will be greater than the accumulations in wood products, yielding a net overall loss of C from land. Such has been the case globally over the last 150 years or more.
Estimates of the effects of harvest on vegetation C stocks are based on information on preharvest biomass and the fractions of this biomass harvested, damaged, and left living. Wood removed from the forest enters the forest products stream, whereas wood left behind enters the harvest residue pool. Woody debris provides a large source of C to the atmosphere as the dead wood decomposes, with the rate and duration of this source dependent on the amount and condition of wood left on-site. The amount of C in the dead wood pool is large during the years after harvest, decreases as the slash pool decomposes, and then increases again later in succession as dead wood accumulates (e.g., Harmon et al. 1990; Idol et al. 2001). The rate of C accumulation in vegetation during forest recovery after harvest, as after other disturbances, varies with climate and soil conditions (Johnson et al. 2000).
At the global level, approximately the same volumes of wood are harvested for industrial wood (timber, pulp) and fuelwood, although most of the wood harvested in developed countries is for industrial wood, while most harvests in developing countries are for fuelwood (FAOSTAT 2009). Fuelwood use is probably minor in affecting C stocks except in those regions where the supply of wood is less than the demand. Particularly around urban centers, demand often exceeds supply, and C stocks are reduced (Ahrends et al. 2010). Such degradation, and its effects on C stocks, are not well documented, globally.
Illegal logging makes estimates of industrial logging uncertain, and fuelwood use is also difficult to assess. Nevertheless, the cumulative area harvested for industrial wood (∼1,500 × 106 ha according to the analysis by Houghton (this study)) is larger than the cumulative area of forest cleared for croplands (875–1,035 × 106 ha) (Table 4.3). The total area harvested is also nearly twice the area computed to be in secondary forest, indicating that many forests were harvested more than once or converted to other uses. Despite the large area logged, even the gross emissions of C from wood harvest are smaller than from deforestation for crops because harvests may have little effect on the C content of mineral soil (Johnson 1992; Johnson and Curtis 2001; Nave et al. 2010) (e.g., Zummo and Friedland (2011) for an exception). The forest floor often loses C after harvest, due largely to reduced C inputs and to the mechanical transfer of forest floor material to deeper soil layers (Currie et al. 2002; Yanai et al. 2003; Nave et al. 2010). Because forest harvest usually causes little loss of SOM, little additional accumulation of SOC occurs with forest recovery.
The net loss of C from industrial wood harvest (including losses from wood products and gains in regrowing forests) over the last 155 years was 17 Pg C according to the analysis by Houghton (this study). At the same time, 19 Pg C accumulated in wood products (Table 4.4). That accumulation accounts for ∼40% of the industrial wood harvested over the period. The fraction is consistent with efficiencies reported for harvests in the U.S., taking into account that the fraction of harvested products going to long-term storage (>5 years) has increased over recent decades from ∼20% to 30–40% (Harmon et al. 1990; Smith et al. 2006). On the other hand, the fraction of the original forest biomass held in wood products 100 years after harvest may be only 1%, with another 13% in landfills (Ingerson 2010).
Other. Most forms of management other than wood harvest have received little attention in global estimates of C flux from land use and land-use change. An exception is the net release of C estimated to have occurred in China between 1900 and 1980 (Houghton and Hackler 2003). During this interval, the net loss of forest area was more than three times greater than the net increase in croplands and pastures. The loss may have resulted from unsustainable harvests, from deliberate removal of forest cover (for protection from tigers or bandits), and from the deleterious effects of long-term intensive agriculture on soil fertility. Unlike croplands, pastures, and forests, the area in degraded lands is rarely enumerated (Oldeman 1994), yet the losses of C may be equivalent to the losses resulting from cultivation, especially if the degradation results from worn-out cultivated lands, abandoned but not returning to forest.
4.2.2.3 Reforestation and Management
Reforestation, afforestation, and other types of land management have run counter to the global trend of de-carbonization. Some of these activities are described below.
At the national level, forest area may be related to the country’s stage of development – the “forest transition” (Mather 1992). Frontier lands may have large forests areas, which are used to fuel economic growth. The result is a loss of forest area. With development, however, forest areas expand again. Examples include China, India, Europe, and the northeastern and mid-western regions of the U.S. The trends are broadly consistent with the emissions from tropical and non-tropical regions (Fig. 4.2). The developed countries lost C in the past, but many are now sequestering C. In contrast, the losses of C from tropical countries are still high. There are exceptions, however. Forests are being lost, again, in New England (US) – this time for residential and commercial uses rather than for agriculture (Jeon 2011). As many of these residences are second homes, they suggest a new wrinkle to the forest transition paradigm.
Plantations. Plantations, globally, have not been a large net sink for C, as might be expected, because plantations are often established on forest lands, and the accumulation of C in growing plantations is offset by the emissions of C from deforestation. Further, many plantations are timber or fuelwood plantations, periodically harvested and thus have an average biomass less than the forests from which they were derived. The conversion of native forests to plantations normally leads to a net reduction in C stocks.
Although the rate of plantation establishment has recently increased, especially in China, Russia, and Vietnam, the area annually planted globally was ∼25% of the area deforested (FAO 2006). This estimate does not include changes in the areas of orchards or oil palm plantations, which are “permanent crops” according to the FAO. The rate of accumulation of C aboveground is well documented for plantations, but the spatial heterogeneity (which types of plantation, planted where?) is not readily available for large regions. For example, plantations may be established for timber, shelter belts, or fuel, and the stocks of C in biomass vary considerably. Whether plantations are established on non-forest lands or on recently cleared forests also affects the net changes in biomass and SOC that result. Reviewing more than 100 observations, Guo and Gifford (2002) found that the establishment of plantations on forest lands or pastures generally decreased SOC stocks, while establishment on croplands increased them. This finding is consistent with the observations, above, that cultivation causes a 25–30% decline in the top meter, while pastures, often not cultivated, lose considerably less or even gain C. In another review Paul et al. (2002) found that plantations established on agricultural lands (both croplands and pastures) lost SOC during the first 5–10 years but gained it over periods longer than 30 years. The length of time land has been cultivated before being converted to a plantation may explain some of this variability. Overall, the changes in SOC were small relative to the gains in biomass.
Agricultural management. The changes in SOC that result from the conversion of natural ecosystems to croplands and their subsequent cultivation are addressed above (Sect. 4.2.1), but changes in C stocks result from cropland management, including cropping practices, irrigation, use of fertilizers, different types of tillage, changes in crop density, and changes in crop varieties. Many studies have addressed the potential for management to sequester C. Fewer studies have tried to estimate past or current C sinks. Recent analyses for the U.S. suggest a current sink of 0.015 Pg C year−1 in croplands (Eve et al. 2002), while a recent assessment for Europe suggests a net source of 0.300 Pg C year−1, perhaps because of reduced application of manure to cropland (Janssens et al. 2003). In Canada, the flux of C from cropland management is thought to be changing from a net source to a net sink, with a current flux near zero (Smith et al. 2000). Globally, the current flux is uncertain but probably not far from zero.
Aside from the losses of C resulting from cultivation of native soils and the re-accumulation of C in abandoned croplands soils, changes in agricultural management have not generally been included in global analyses of land use and land-use change although they have been included in regional analyses at high spatial resolution (e.g., Kutsch et al. 2010; West et al. 2010). The effects of erosion and redeposition of organic C, discussed above under croplands, pertain here as well.
Fire management. The emissions of C from fires associated with the conversion of forests to croplands and pastures are included in analyses of land-use change, but fire management has largely been ignored despite the fact that fire exclusion, fire suppression, and controlled burning are practiced in many parts of the world. In many regions, fire management may cause a terrestrial sink (Houghton et al. 1999; Marlon et al. 2008). In other regions it increases the net source. In particular, the draining and burning of peatlands in Southeast Asia are thought to add another 0.3 Pg C year−1 to the net emissions from land-use change (not included in the estimates reported here) (Hooijer et al. 2009).
Woody encroachment. The expansion of trees and woody shrubs into herbaceous lands, although it cannot be attributed definitively to natural, indirect (climate, CO2), or direct effects (fire suppression, grazing), is, nevertheless, re-carbonizing land in many regions. Scaling it up to a global estimate is problematical, however (Scholes and Archer 1997; Archer et al. 2001), in part because the areal extent of woody encroachment is unknown and difficult to measure (e.g., Asner et al. 2003). Also, the increase in vegetation C stocks observed with woody encroachment is in some cases offset by losses of soil C (Jackson et al. 2002). In other cases the soils may gain C (e.g., Hibbard et al. 2001) or show no discernable change (Smith and Johnson 2003). Finally, woody encroachment may be offset by its reverse process, woody elimination, an example of which is the fire-induced spread of cheatgrass (Bromus tectorum) into the native woody shrublands of the Great Basin in the western U.S. (Bradley et al. 2006).
The net effect of woody encroachment and woody elimination is, thus, uncertain, not only with respect to net change in C storage, but also with respect to attribution. It may be an unintended effect of management, or it may be a response to indirect or natural effects of environmental change.
4.3 Summary and Conclusions
4.3.1 The Past
The amount of C lost from terrestrial ecosystems over the last ∼150 years is 1–3 times greater than the losses before 1850 (Table 4.2). Since 1850, most of the loss has been from biomass; soils contributed only about 25% of this net release (Table 4.4), although the proportion varies by ecosystem type. The amount of C in slash and wood products is small, but the increases over 155 years are significant relative to the changes in living biomass and soil (Table 4.4)
More than one third of the total land surface of the Earth has been altered directly by human activity (nearly half of the productive land surface – rock, sand, snow, ice, and water bodies excluded). Croplands and pastures, together, occupy a quarter to a third of the land surface, and secondary forests recovering from some form of management account for ∼30% of the world’s forests. All of the lands that are known to have been managed in 2000 account for 37% of total land area, or 46% of the productive land surface. Hurtt et al. (2006) estimated that 42–68% of the land surface had been affected by land-use activities, but even those estimates are probably conservative. It seems likely that most of the productive land surface has been used in one way or another over the last 150 years, but many uses are never reported or recorded.
Direct human activity has caused a total net decarbonization of 170–260 Pg C from the world’s terrestrial ecosystems (Table 4.2). As discussed in Sect. 4.1, this decarbonization in recent years has been offset to a large extent by natural or indirect human effects that have led to an accumulation of C on land. Nevertheless, the net effect of management, to date, has been to decarbonize rather than recarbonize the land surface. Most (60–70%) of this long-term decarbonization occurred over the last 155 years (108–188 Pg C). According to the analysis reported here, about 90% of this more recent decarbonization has resulted from the clearing and management of forests; cultivation of prairie soils account for the other 10%. Estimates are that 30–40% of the world’s forests have been lost (Pongratz et al. 2008; World Commission on Forests and Sustainable Development 1999). The loss of ∼1,074 × 106 ha of forest over the last 155 years has had the greatest effect on decarbonization, ∼110 Pg C, but widespread harvest of wood and other processes degrading forests have contributed ∼40 Pg C (∼25%). This degradation has reduced the carbon density of forest biomass from a global average of 103 Mg C/ha to 101 Mg C/ha by 1850 to 91 Mg C/ha by 2005 (Fig. 4.5).
The rate of these transformations over the surface of the Earth has been accelerating, globally, although not equally in all regions. In the northern mid-latitudes (largely developed countries) the area in agriculture has been nearly constant over the last decades, while in the tropics agricultural lands have been expanding.
4.3.2 The Future
This short review has sketched broad global trends. It has not done justice to the numerous management practices that counter these trends in decarbonization. Indeed, the larger the decarbonization in the past, the greater the opportunity for recarbonization in the future. Most of the activities that have released C to the atmosphere might, with appropriate incentives, store it again on land. Examples include the management of forests and use of wood (Kohlmaier et al. 1998) and the management of agriculture (Smith et al. 2007).
Two activities have the potential to affect terrestrial C sinks at scales large and rapid enough to stabilize the concentration of CO2 quickly: massive reforestation and a halt to deforestation. On the order of 200–300 × 106 ha of new forest would be required to remove 1–1.5 Pg C year−1 from the atmosphere, and that uptake would decline after several decades. The area is large, but not in comparison to current areas in croplands and pastures. The magnitude of 1–1.5 Pg C year−1 assumes, optimistically, an average sequestration rate in wood and soils of 5 Mg (1 Mg = 106 g) C ha−1year−1, while the lands available for such afforestation are unlikely to be the most fertile and productive.
Deforestation is responsible at present for annual emissions of 1–1.5 Pg C. A halt to deforestation, combined with a massive program of reforestation, could thus reduce C emissions by 2.5–3 Pg C year−1. The reduction is more than half the rate at which C is accumulating in the atmosphere at present (∼4 Pg C year−1) (Table 4.1). Similar reductions of 1–1.5 Pg C year−1 in the emissions of C from fossil fuels, which are now nearly 9 Pg C year−1 (Friedlingstein et al. 2010), would stabilize the concentration of CO2 in the atmosphere immediately. Additional reductions would be required over time to bring the concentration of CO2 in the atmosphere back to 350 ppm, but the management of C on land offers a short-term solution to stabilization of atmospheric CO2 concentrations. It has the advantage of being technically achievable and cheap relative to other emissions reductions.
The emphasis in this chapter on net losses of C from land hides the much larger gross sources and sinks of C associated with land use and land-use change. The gross loss of C from land management, for example, was more than 4 Pg C year−1, while the net loss was only 1–1.5 Pg C year−1 (Richter and Houghton 2011). These gross fluxes indicate a much larger potential than the global net flux for reducing current losses and for re-carbonizing terrestrial ecosystems through management.
Stopping deforestation and establishing 200–300 × 106 ha of new forest would require a reversal of the global trends in land use and land-use change that have accelerated over the last 300+ years. Such reversals have already happened in many nations, as suggested by the forest transition (see Sect. 4.2.2.3). Can they become global? And can they become global when the demands for food are growing, as well as the demands for meat, and bioenergy? The competition for land looms as a particularly important issue over the next century.
Abbreviations
- C:
-
carbon
- SOC:
-
soil organic carbon
- SOM:
-
soil organic matter
References
Ahrends A et al (2010) Predicable waves of sequential forest degradation and biodiversity loss spreading from an African city. Proc Natl Acad Sci USA 107:14556–14561
Archer S, Boutton TW, Hibbard KA (2001) Trees in grasslands: biogeochemical consequences of woody plant expansion. In: Schulze E-D, Harrison SP, Heimann M, Holland EA, Lloyd J, Prentice IC, Schimel D (eds) Global biogeochemical cycles in the climate system. Academic, San Diego, pp 115–1337
Asner GP, Archer S, Hughes RF, James R, Wessman CA (2003) Net changes in regional woody vegetation cover and carbon storage in Texas drylands, 1937–1999. Glob Chang Biol 9:316–335
Barford CC et al (2001) Factors controlling long- and short-term sequestration of atmospheric CO2 in a mid-latitude forest. Science 294:1688–1691
Berhe AA, Harte J, Harden JW, Torn MS (2007) The significance of the erosion-induced terrestrial carbon sink. Bioscience 57:337–347
Bradley BA, Houghton RA, Mustard JF, Hamburg SP (2006) Invasive grass reduces aboveground carbon stocks in shrublands of the Western US. Glob Chang Biol 12:1815–1822
Bridgham SD, Megonigal JP, Keller JK, Bliss NB, Trettin C (2006) The carbon balance of North American wetlands. Wetlands 26:889–916
Brown DG, Johnson KM, Loveland TR, Theobald DM (2005) Rural land-use trends in the conterminous United States, 1950–2000. Ecol Appl 15:1851–1863
Canadell JG et al (2007) Contributions to accelerating atmospheric CO2 growth from economic activity, carbon intensity, and efficiency of natural sinks. Proc Natl Acad Sci USA 104:18866–18870
Currie WS, Yanai RD, Piatek KB, Prescott CE, Goodale CL et al (2002) Processes affecting carbon storage in the forest floor and in downed woody debris. In: Kimble JM et al (eds) The potential for U.S. forest soils to sequester carbon and mitigate the greenhouse effect. CRC Press, Boca Raton, pp 135–157
Davidson EA, Ackerman IL (1993) Changes in soil carbon inventories following cultivation of previously untilled soils. Biogeochemistry 20:161–193
DeFries RS, Field CB, Fung I, Collatz GJ, Bounoua L (1999) Combining satellite data and biogeochemical models to estimate global effects of human-induced land cover change on carbon emissions and primary productivity. Global Biogeochem Cycles 13:803–815
Detwiler RP (1986) Land use change and the global carbon cycle: the role of tropical soils. Biogeochemistry 2:67–93
Dommain R, Couwenberg J, Joosten H (2011) Development and carbon sequestration of tropical peat domes in south-east Asia: links to post-glacial sea-level changes and Holocene climate variability. Quaternary Sci Rev 30:999–1010
Donato DC, Kauffman JB, Murdiyarso D, Kurnianto S, Stidham M, Kanninen M (2011) Mangroves among the most carbon-rich forests in the tropics. Nat Geosci 4(5):293–297
Ellis EC (2011) Anthropogenic transformation of the terrestrial biosphere. Philos Trans A Math Phys Eng Sci 369:1010–1035
Eve MD, Sperow M, Paustian K, Follett RF (2002) National-scale estimation of changes in soil carbon stocks on agricultural lands. Environ Pollut 116:431–438
FAO (2006) Global forest resources assessment 2005. FAO forestry paper 147, Rome
FAO (2009) http://faostat.fao.org/site/377/default.aspx#ancor (11/09)
FAOSTAT (2009) http://faostat.fao.org/site/377/default.aspx#ancor (11/09)
Friedlingstein P, Houghton RA, Marland G et al (2010) Update on CO2 emissions. Nat Geosci 3:811–812
Gillett NP, Weaver AJ, Zwiers FW, Flannigan MD (2004) Detecting the effect of climate change on Canadian forest fires. Geophys Res Lett 31:L18211
Gitz V, Ciais P (2003) Amplifying effects of land-use change on future atmospheric CO2 levels. Global Biogeochem Cycles 17:1024. doi:10.1029/2002GB001963
GLCC2000 (2009). Global land cover characterization, version 1.2. http://edc2.usgs.gov/glcc/glcc_version1.php
Gloor M, Sarmiento JL, Gruber N (2010) What can be learned about the carbon cycle climate feedbacks from the CO2 airborne fraction? Atmos Chem Phys 10:7739–7751
Grainger A (2008) Difficulties in tracking the long-term global trend in tropical forest area. Proc Natl Acad Sci USA 105:818–823
Grainger A (2009) Measuring the planet to fill terrestrial data gaps. Proc Natl Acad Sci USA 106:20557–20558
Guo LB, Gifford RM (2002) Soil carbon stocks and land use change: a meta analysis. Glob Chang Biol 8:345–360
Harmon ME, Ferrell WK, Franklin JF (1990) Effects on carbon storage of conversion of old-growth forests to young forests. Science 247:699–702
Harrison KG, Post WM, Richter DD (1995) Soil carbon turnover in a recovering temperate forest. Global Biogeochem Cycles 9:449–454
Hibbard KA, Archer S, Schimel DS, Valentine DW (2001) Biogeochemical changes accompanying woody plant encroachment in a subtropical savanna. Ecology 82:1999–2011
Hooijer A et al (2009) Current and future CO2 emissions from drained peatlands in Southeast Asia. Biogeosci Discuss 6:7207–7230
Hooker TD, Compton JE (2003) Forest ecosystem carbon and nitrogen accumulation during the first century after agricultural abandonment. Ecol Appl 13:299–313
Houghton RA (1999) The annual net flux of carbon to the atmosphere from changes in land use 1850–1990. Tellus 51B:298–313
Houghton RA (2003) Revised estimates of the annual net flux of carbon to the atmosphere from changes in land use and land management 1850–2000. Tellus 55B:378–390
Houghton RA (2007) Balancing the global carbon budget. Annu Rev Earth Planet Sci 35:313–347
Houghton RA (2010) How well do we know the flux of CO2 from land-use change? Tellus B62:337–351. doi:10.1111/j.1600-0889.2010.00473.x
Houghton RA, Hackler JL (2003) Sources and sinks of carbon from land-use change in China. Global Biogeochem Cycles 17:1034. doi:10.1029/2002GB001970
Houghton RA, Hackler JL (2006) Emissions of carbon from land use change in sub-Saharan Africa. J Geophys Res 111:G02003. doi:10.1029/2005JG000076
Houghton RA, Hackler JL, Lawrence KT (1999) The U.S. carbon budget: contributions from land-use change. Science 285:574–578
Huntington TG (1995) Carbon sequestration in an aggrading forest ecosystem in the southeastern USA. Soil Sci Soc Am J 59:1459–1467
Hurtt GC et al (2006) The underpinnings of land-use history: three centuries of global gridded land-use transitions, wood-harvest activity, and resulting secondary lands. Glob Chang Biol 12:1–22
Idol TW, Filder RA, Pope PE, Ponder F (2001) Characterization of coarse woody debris across a 100 year chronosequence of upland oak-hickory forests. For Ecol Manage 149:153–161
Ingerson A (2010) Carbon storage potential of harvested wood: summary and policy implications. Mitig Adapt Strat Glob Chang. doi:10.1007/s11027-010-9267-5
Jackson RB, Banner JL, Jobbágy EG, Pockman WT, Wall DH (2002) Ecosystem carbon loss with woody plant invasion of grasslands. Nature 418:623–626
Janssens IA et al (2003) Europe’s terrestrial biosphere absorbs 7–12% of European anthropogenic CO2 emissions. Science 300:1538–1542
Jeon SB (2011) The effect of land-use change on the terrestrial carbon budget of New England. PhD dissertation, Boston University
Johnson DW (1992) Effects of forest management on soil carbon storage. Water Air Soil Pollut 64:83–120
Johnson DW, Curtis PS (2001) Effects of forest management on soil C and N storage: meta analysis. For Ecol Manage 140:227–238
Johnson CM, Zarin DJ, Johnson AH (2000) Post-disturbance above-ground biomass accumulation in global secondary forests. Ecology 81:1395–1401
Kaplan JO, Krumhardt KM, Zimmermann N (2009) The prehistoric and preindustrial deforestation of Europe. Quaternary Sci Rev 28:3016–3034
Kauppi PE, Ausubel JH, Fang J, Mather AS, Sedjo RA, Waggoner PE (2006) Returning forests analyzed with the forest identity. Proc Natl Acad Sci USA 103:17574–17579
Klein Goldewijk K (2001) Estimating global land use change over the past 300 years: the HYDE database. Global Biogeochem Cycles 15:417–433
Klein Goldewijk K, Ramankutty N (2004) Land cover change over the last three centuries due to human activities: the availability of new global data sets. GeoJournal 61:335–344
Klein Goldewijk K, van Drecht G (2006) HYDE3: current and historical population and land cover. In: Bouwman AF, Kram T, Klein Goldewijk K (eds) Integrated modeling of global environmental change. An overview of IMAGE 2.4. Netherlands Environmental Assessment Agency (MNP), Bilthoven, The Netherlands
Knorr W (2009) Is the airborne fraction of anthropogenic CO2 emissions increasing? Geophys Res Lett 36:L21710. doi:10.1029/2009GL040613
Kohlmaier GH, Weber M, Houghton RA (eds) (1998) Carbon dioxide mitigation in forestry and wood industry. Springer, Berlin
Kozlowski TT (2002) Physiological ecology of natural regeneration of harvested and disturbed forest stands: implications for forest management. For Ecol Manage 158:195–221
Kurz WA et al (2008) Mountain pine beetle and forest carbon feedback to climate change. Nature 452:987–990
Kutsch WL et al (2010) The net biome production of full crop rotations in Europe. Agric Ecosyst Environ 139:336–345
Lal R (2001) Potential of desertification control to sequester carbon and mitigate the greenhouse effect. Clim Chang 51:35–72
Le Quéré C, Raupach MR, Canadell JG et al (2009) Trends in the sources and sinks of carbon dioxide. Nat Geosci 2:831–836
Luyssaert S, Schulze E-D, Börner A et al (2008) Old-growth forests as global carbon sinks. Nature 455:213–215. doi:10.1038/nature07276
Mann LK (1985) A regional comparison of carbon in cultivated and uncultivated alfisols and mollisols in the central United States. Geoderma 36:241–253
Mann LK (1986) Changes in soil carbon storage after cultivation. Soil Sci 142:279–288
Marlon JR, Bartlein PJ, Carcaillet C et al (2008) Climate and human influences on global biomass burning over the past two millennia. Nat Geosci 1:697–701
Mather AS (1992) The forest transition. Area 24:367–379
Morton DC, DeFries RS, Randerson JT et al (2008) Agricultural intensification increases deforestation fire activity in Amazonia. Glob Chang Biol 14:2262–2275
Murty D, Kirschbaum MF, McMurtrie RE, McGilvray H (2002) Does conversion of forest to agricultural land change soil carbon and nitrogen? A review of the literature. Glob Chang Biol 8:105–123
Myers N (1980) Conversion of tropical moist forests. National Academy of Sciences Press, Washington, DC
Nave LE, Vance ED, Swanston CW, Curtis PS (2010) Harvest impacts on soil carbon storage in temperate forests. For Ecol Manage 259:857–866
Neill C, Davidson EA (2000) Soil carbon accumulation or loss following deforestation for pasture in the Brazilian Amazon. In: Lal R, Kimble JM, Stewart BA (eds) Global climate change and tropical ecosystems. CRC Press, Boca Raton, pp 197–211
Oldeman LR (1994) The global extent of soil degradation. In: Greenland DJ, Szaboles I (eds) Soil resilience and sustainable land Use. CAB International, New York, pp 99–118
Olofsson J, Hickler T (2008) Effects of human land use on the global carbon cycle during the last 6,000 years. Veg Hist Archeobot 17:605–615. doi:10.1007/s00334-007-0126-6
Osher LJ, Matson PA, Amundson R (2003) Effect of land use change on soil carbon in Hawaii. Biogeochemistry 65:213–232
Pan Y, Birdsey RA, Fang J et al (2011) A large and persistent carbon sink in the world’s forests, 1990–2007. Science 333:988–993
Parfitt RL, Scott NA, Ross DJ, Salt GJ, Tate KR (2003) Landuse change effects on soil C and N transformations in soils of high N status: comparisons under indigenous forest, pasture and pine plantation. Biogeochemistry 66:203–221
Paul KI, Polglase PJ, Nyakuengama JG, Khanna PK (2002) Change in soil carbon following afforestation. For Ecol Manage 168:241–257
Piao S, Ciais P, Friedlingstein P et al (2009) Spatiotemporal patterns of terrestrial carbon cycle during the 20th century. Global Biogeochem Cycles 23:GB4026. doi:10.1029/2008GB003339
Pinter N, Fiedel S, Keeley JE (2011) Fire and vegetation shifts in the Americas at the vanguard of Paleoindian migration. Quaternary Sci Rev 30:269–272
Pongratz J, Reick C, Raddatz T, Claussen M (2008) A reconstruction of global agricultural areas and land cover for the last millennium. Global Biogeochem Cycles 22:GB3018. doi:10.1029/2007GB003153
Pongratz J, Reick CH, Raddatz T, Claussen M (2009) Effects of anthropogenic land cover change n the carbon cycle of the last millennium. Global Biogeochem Cycles 23:GB4001. doi:10.1029/2009GB003488
Post WM, Kwon KC (2000) Soil carbon sequestration and land-use change: processes and potential. Glob Chang Biol 6:317–327
Potere D, Schneider A (2007) A critical look at representations of urban areas in global maps. GeoJournal 69:55–80
Prentice IC, Harrison SP, Bartlein PJ (2011) Global vegetation and terrestrial carbon cycle changes after the last ice age. New Phytol 189:988–998
Ramankutty N, Foley JA (1999) Estimating historical changes in global land cover: croplands from 1700 to 1992. Global Biogeochem Cycles 13:997–1027
Ramankutty N, Evan AT, Monfreda C, Foley JA (2008) Farming the planet: 1. Geographic distribution of global agricultural lands in the year 2000. Global Biogeochem Cycles 22:GB1003. doi:10.1029/2007GB002952
Reick CH, Raddatz T, Pongratz JM, Claussen M (2011) Contribution of anthropogenic land cover change emissions to preindustrial atmospheric CO2. Tellus 62B:329–336
Richter DdeB, Houghton RA (2011) Gross CO2 fluxes from land-use change: implications for reducing global emissions and increasing sinks. Carbon Manag 2:41–47
Schipper LA, Baisden WT, Parfitt RL, Ross W, Claydon JJ, Arnold G (2007) Large losses of soil C and N from soil profiles under pasture in New Zealand during the past 20 years. Glob Chang Biol 13:1138–1144
Schlesinger WH (1986) Changes in soil carbon storage and associated properties with disturbance and recovery. In: Trabalka JR, Reichle DE (eds) The changing carbon cycle: a global analysis. Springer, New York, pp 194–220
Schneider A, Friedl MA, Potere D (2009) A new map of global urban extent from MODIS satellite data. Environ Res Lett 4. doi:1088/1748-9326/4/4/044003
Scholes RJ, Archer SR (1997) Tree-grass interactions in savannas. Annu Rev Ecol System 28:517–544
Shevliakova E et al (2009) Carbon cycling under 300 years of land use change: importance of the secondary vegetation sink. Global Biogeochem Cycles 23:GB2022. doi:10.1029/2007GB003176
Smith DL, Johnson LC (2003) Expansion of Juniperus virginiana L. in the Great Plains: changes in soil organic carbon dynamics. Global Biogeochem Cycles 17:1062. doi:10.1029/2002GB001990
Smith WN, Desjardins RL, Pattey E (2000) The net flux of carbon from agricultural soils in Canada 1970–2010. Glob Chang Biol 6:557–568
Smith SV, Renwick WH, Buddemeier RW, Crossland CJ (2001) Budgets of soil erosion and deposition for sediments and sedimentary organic carbon across the conterminous United States. Global Biogeochem Cycles 15:697–707
Smith JE, Heath LS, Skog KE, Birdsey RA (2006) Methods for calculating forest ecosystem and harvested carbon with standard estimates for forest types of the United States. Gen. Tech. Rep. NE-343. U.S. Department of Agriculture, Forest Service, Northeastern Research Station, Newtown Square, PA, 216 pp
Smith P, Martino D, Cai Z et al (2007) Agriculture. In: Metz B, Davidson OR, Bosch PR, Dave R, Meyer LA (eds) Climate change 2007: mitigation. Contribution of Working Group III to the fourth assessment report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge/New York
Stallard RF (1998) Terrestrial sedimentation and the carbon cycle: coupling weathering and erosion to carbon burial. Global Biogeochem Cycles 12:231–257
Strassmann KM, Joos F, Fischer G (2008) Simulating effects of land use changes on carbon fluxes: past contributions to atmospheric CO2 increases and future commitments due to losses of terrestrial sink capacity. Tellus 60B:583–603
Uhl C, Buschbacher R, Serrao EAS (1988) Abandoned pastures in eastern Amazonia. I. Patterns of plant succession. J Ecol 76:663–681
Uhlig J, Hall CAS, Nyo T (1994) Changing patterns of shifting cultivation in selected countries in Southeast Asia and their effect on the global carbon cycle. In: Dale V (ed) Effects of land-use change on atmospheric CO2 concentrations. South and Southeast Asia as a case study. Springer, New York, pp 145–200
Van Minnen JG, Klein Goldewijk K, Stehfest E et al (2009) The importance of three centuries of land-use change for the global and regional terrestrial carbon cycle. Clim Chang 97:123–144
Wang Z, Chappellaz J, Park K, Mak JE (2010) Large variations in southern hemisphere biomass burning during the last 650 years. Science 330:1663–1666
West TO et al (2010) Cropland carbon fluxes in the United States: increasing geospatial resolution of inventory-based carbon accounting. Ecol Appl 20:1074–1086
Westerling AL, Hidalgo HG, Cayan DR, Swetnam TW (2006) Warming and earlier spring increase western US forest wildfire activity. Science 313:940–943
World Commission on Forests and Sustainable Development (1999) Our forests, our future. Cambridge University Press, Cambridge
Yanai RD, Currie WS, Goodale CL (2003) Oil carbon dynamics after forest harvest: an ecosystem paradigm revisited. Ecosystems 6:97–212
Zummo LM, Friedland AJ (2011) Soil carbon release along a gradient of physical disturbance in a harvested northern hardwood forest. For Ecol Manage 261:1016–1026
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
Houghton, R.A. (2012). Historic Changes in Terrestrial Carbon Storage. In: Lal, R., Lorenz, K., Hüttl, R., Schneider, B., von Braun, J. (eds) Recarbonization of the Biosphere. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-4159-1_4
Download citation
DOI: https://doi.org/10.1007/978-94-007-4159-1_4
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-4158-4
Online ISBN: 978-94-007-4159-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)