Abstract
The phosphoinositide 3-kinase (PI 3-K) signal relay pathway represents arguably one of the most intensely studied mechanisms by which extracellular signals elicit cellular responses through the generation of second messengers that are associated with cell growth and transformation. This chapter reviews the many landmark discoveries in the PI 3-K signaling pathway in biology and disease, from the identification of a novel phosphoinositide kinase activity associated with transforming oncogenes in the 1980s, to the identification of oncogenic mutations in the catalytic subunit of PI 3-K in the mid 2000s. Two and a half decades of intense research have provided clear evidence that the PI 3-K pathway controls virtually all aspects of normal cellular physiology, and that deregulation of one or more proteins that regulate or transduce the PI 3-K signal ultimately leads to human pathology. The most recent efforts have focused on the development of specific PI 3-K inhibitors that are currently being evaluated in clinical trials for a range of disease states.
This chapter is devoted to a historical review of the landmark findings in the PI 3-K from its relatively humble beginnings in the early to mid 1980s up until the present day. When considering the key findings in the history of PI 3-K, it is essential to recognize the landmark studies by Lowell and Mabel Hokin in the 1950s who were the first to describe that extracellular agonists such as acetylcholine could stimulate the incorporation of radiolabeled phosphate into phospholipids (Hokin and Hokin 1953). Their work initiated an entirely new field of lipid signaling, and subsequent studies in the 1970s by Michell and Lapetina who linked phosphoinositide turnover to membrane-associated receptors that initiate intracellular calcium mobilization (Lapetina and Michell 1973). Later studies revealed that the phospholipase-mediated breakdown of the same minor membrane phospholipids such as PtdIns-4,5-P2 (phosphatidylinositol-4,5-bisphosphate) is responsible for the release of two additional key second messengers, diacylglycerol (DG) and IP3 (inositol-1,4,5-trisphosphate) (Kirk et al. 1981; Berridge 1983; Berridge et al. 1983). Berridge, Irvine and Schulz then revealed that one of the byproducts of this lipid signal relay pathway is the release of calcium from intracellular stores such as the endoplasmic reticulum (Streb et al. 1983). Finally, pioneering studies by Nishizuka in the late 1970s identified PKC (protein kinase C) as a phospholipid and diacylglycerol-activated serine/threonine protein kinase (Inoue et al. 1977; Takai et al. 1977). At this point, it probably seemed to most at the time that the story was complete, such that hydrolysis of phosphoinositides such as PtdIns-4,5-P2 and PtdIns-4-P would account for the major mechanisms of agonist-stimulated lipid signaling leading to physiological responses. On the contrary, the story was far from complete and was about to become a lot more complex.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
4.1 The Discovery of PtdIns 3-OH Phosphorylation and PI 3-Kinases
The discovery of an enzymatic activity that could phosphorylate the 3-OH position of the inositol head ring of phosphoinositides has its roots in initial work by Macara and Balduzzi who showed that a phosphatidylinositol kinase activity was found associated with an avian sarcoma virus when expressed in cells (Macara et al. 1984). Simultaneously, Erickson and Cantley, working on the recently-discovered oncogenic tyrosine kinase Src, showed that it could phosphorylate phosphatidylinositol and phosphatidylglycerol in immunoprecipitates (Sugimoto et al. 1984). This suggested to them that Src itself has lipid kinase activity, yet as it turned out later it was the Src-associated PI 3-K activity that was responsible for this reaction. This result prompted Cantley to extend these findings, and in collaboration with Roberts, Schaffhausen and Kaplan they showed that the phosphatidylinositol kinase activity was due to an 85 kDa phosphoprotein bound to the middle T antigen of the polyoma tumor DNA virus (Kaplan et al. 1987). Despite these exciting new findings demonstrating an interaction between lipid-derived signals and transforming oncoproteins, PI 3-K had not yet formally been discovered. It was Whitman, a graduate student in the laboratory of Cantley who realized that the product of the phosphatidylinositol activity in Src immunoprecipitates migrated slightly differently on thin layer chromatography plates from PtdIns-4-P, which was the presumed product of the reaction. A collaboration with Downes, an expert in inositol lipids, proved beyond doubt that the lipid product of the reaction was in fact PtdIns-3-P, the first new phosphoinositide identified in some 30 years (Whitman et al. 1988). Downes, Hawkins and Stephens working independently also provided evidence of the existence of PtdIns-3-P in astrocytoma cells (Stephens et al. 1989). As became evident later, that PtdIns-3-P transduces the PI 3-K signal in response to growth factor was somewhat of a red herring, since the primary activity of class I PI 3-Ks that are activated by both receptor tyrosine kinases (RTK) and G protein-coupled receptors (GPCR) is to interconvert PtdIns-4,5-P2 into PtdIns-3,4,5-P3. PtdIns-3-P, as it turned out later, is generated by class III PI 3-Ks, the prototype being the yeast ortholog vps34p. In this context, it is often overlooked that PtdIns-3,4,5-P3 was actually discovered by Traynor-Kaplan and Sklar, who identified an inositol tetrakisphosphate (IP4)-containing phospholipid in stimulated neutrophils (Traynor-Kaplan et al. 1988). They showed that PtdIns-3,4,5-P3 is a new phosphoinositide that contains four phosphates, making it the most negatively charged lipid in the cell. Both the Cantley, Stephens and Sklar papers were published within a few months of each other in a period between 1988 and 1989, representing the birth of the PI 3-K field.
There then followed a flurry of activity by the laboratories highlighted above, though arguably the rest of the signaling world did not take much notice at first. At the time most were heavily invested in understanding the molecular mechanisms by which oncogenes such as Src and Ras transduce mitogenic signals, and delineating the mechanisms of activation of ERK (extracellular-regulated kinase). In the PI 3-K world, it was shown that exposure of a variety of cells with mitogenic growth factors such as PDGF (platelet-derived growth factor) (Auger et al. 1989) and GPCR (G protein coupled receptor) agonists (such as fMLP, formylmethionyl leucyl phenylalanine) (Stephens et al. 1991) stimulated the rapid accumulation of PtdIns-3,4,5-P3, and also PtdIns-3,4-P2, the latter primarily due to the activity of class II PI 3-Ks and/or breakdown of PtdIns-3,4,5-P3 by lipid phosphatases. In contrast, the levels of PtdIns-3-P remained relatively constant in most cells studied. The conclusion was therefore that PtdIns-3,4-P2, and primarily PtdIns-3,4,5-P3, are true second messengers that transduce the PI 3-K signal. The initial identification of the 85 kDa phosphoprotein associated with Src and polyoma middle T led to its biochemical purification and subsequent cloning by several laboratories (Escobedo et al. 1991; Otsu et al. 1991; Skolnik et al. 1991). During these efforts, Carpenter and Cantley recognized that p85, that we now know represents one of the regulatory subunits of class I PI 3-Ks, was found tightly associated with a p110 protein (Carpenter et al. 1990). Protein microsequencing of purified p110 allowed for the design of degenerate oligonucleotide probes, and using this information Waterfield’s laboratory was the first to clone and characterize the catalytic subunit of PI 3-K, in their case p110a (Hiles et al. 1992). Analysis of the p110 sequence revealed significant homology with the yeast gene vps34p, which in 1990 Herman and Emr showed is required for vacuolar protein sorting and segregation in budding yeast (Herman and Emr 1990). Subsequent pioneering studies by Emr’s laboratory showed that yeast vps34p and its mammalian ortholog are class III PI 3-Ks with a unique substrate specificity, incapable of generating PtdIns-3,4-P2 or PtdIns-3,4,5-P3, and exclusively responsible for PtdIns-3-P synthesis (Schu et al. 1993; Brown et al. 1995; De Camilli et al. 1996).
4.2 Expansion of the Family of PI 3-Kinases and Their Regulators
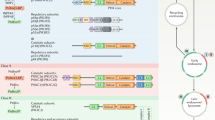
In the years that followed the cloning of p85a and p110a, several laboratories described additional PI 3-K isoforms in both mammals, flies, nematodes, yeasts and even plants, and this required reclassification of the PI 3-K family into distinct classes. We now know there are eight mammalian PI 3-kinases: class Ia enzymes (p110a, p110b, p110d) and class Ib (p110g); class II enzymes (PI 3-K-C2a, PI 3-K-C2b, PI 3-K-Cg); and class III enzymes (vps34p). Note that the nomenclature refers to the catalytic subunits, yet distinct regulatory subunits for each class exist: (class Ia, p85a, p55a, p50a, p85b, p55g); class Ib, p101 and p84; a p150 regulatory subunit for vpv34p has also been identified. For more detail, see the following reviews (Vanhaesebroeck et al. 2010; Engelman et al. 2006; Hawkins et al. 2006) and Vol. I, Chap. 5 by Wymann.
The protein sequencing and cloning of the p85a regulatory subunit coincided with the discovery of SH2 (Src homology 2) domains in pioneering studies by the Hanafusa and Pawson laboratories (Moran et al. 1990; DeClue et al. 1987; O’Brien et al. 1990; Matsuda et al. 1990). Recognizing that p85 subunits have two copies of SH2 domains provided the mechanism by which PI 3-K is relocalized to sites of activated, phosphorylated receptor tyrosine kinases. Zhou and Cantley then used this information to decode the specificity of SH2 domains, including that of p85a, towards phosphotyrosine motifs in RTKs (Zhou et al. 1993). At the same time, Kazlauskas and Cooper developed an approach in which individual tyrosines in the PDGF-receptor are individually mutated to prevent binding to SH2 domains in proteins including PI 3-K (Kazlauskas and Cooper 1989). This showed that both PI 3-K and PLCg-1 (phospholipase C-g) are required to transduce the PDGF signal to DNA synthesis and cell cycle progression (Valius and Kazlauskas 1993). These PDGF-R mutants were then subsequently used by many other laboratories to probe the mechanisms by which PI 3-K engages the cell cycle, and also activates secondary effectors such as S6K1 (p70 ribosomal protein S6 kinase-1) (Chung et al. 1994). Importantly, Kazlauskas later went to show that receptors such as PDGF-R stimulate two waves of PI 3-K activity, one immediately after ligand stimulation lasting about an hour, and a second much larger wave typically 4–8 h post stimulation (Jones et al. 1999). Using PDGF-R mutants as well as PI 3-K inhibitors, his laboratory showed that the first wave is actually dispensable for cell cycle progression to S phase, instead it is the second wave that is responsible.
The next major discovery in the field was wortmannin. In 1993, two laboratories, Wymann and Arcaro as well as Matsuda’s group showed that the furanosteroid metabolite isolated from the fungus Penicillium funiculosum, known as wortmannin, potently inhibits fMLP-stimulated PtdIns-3,4,5-P3 synthesis in neutrophils at low nanomolar doses (Arcaro and Wymann 1993; Yano et al. 1993). Other laboratories subsequently confirmed the inhibitory activity of wortmannin on PI 3-K. A year later, Vlahos at Ely Lilly published a structurally distinct compound termed LY294002, that also directly inhibits PI 3-K (Vlahos et al. 1994). Remarkably, both compounds are still widely used in the field, considering that they have since been shown to have off-target effects on other kinases. Indeed much more specific PI 3-K inhibitors have since been described, to the extent that isoform-specific compounds are now available, many of them in phase I and II clinical trials. Regardless, the availability of wortmannin and LY294002 stimulated much progress in the still nascent PI 3-K signaling field.
The discovery that PI 3-K functions a direct effector of GTP-loaded Ras by Rodriguez-Viciana and Downward added further credence to the model that the PI 3-K pathway contributes to cancer etiology, considering the numerous human solid tumors that harbor oncogenic activating mutations in the Ras GTPase (Rodriguez et al. 1994). Although largely overlooked, it was initially Lapetina who first showed that p21 Ras can associate with PI 3-K in cells (Sjolander et al. 1991). This was later confirmed in the seminal paper by Downward’s group who showed that GTP-Ras is required for efficient PI 3-K activation by growth factors, and they further identified a direct PI 3-K binding site on Ras (Rodriguez et al. 1994). Thus PI 3-K was added to the list of Ras effectors which now encompass the Ser/Thr kinase Raf, the nucleotide dissociation factor RalGDS and the phospholipase PLCe. It is also important to note that as first recognized by Hawkins and Stephens (Wennstrom et al. 1994; Welch et al. 2003), PI 3-K and PtdIns-3,4,5-P3 also contribute to the regulation of other GTPases, most notably Rac, and this in turn has profound effects on the regulation of the actin cytoskeleton and cell motility in virtually all cells. This is best illustrated in both neutrophils and Dictyostelium amoeba where a gradient of PtdIns-3,4,5-P3 and Rac activation is rate-limiting for actin assembly and directed cell migration (Funamoto et al. 2001) (also see Vol. II, Chap. 6 by Yin and Vol. II, Chap. 7 by Parent).
4.3 How Do PI 3-Kinase Lipid Products Affect Cell Function?
In the mid 1990s, the field of PI 3-K was firmly established, but was missing a critical piece of the puzzle. How do the PI 3-K lipid products, particularly PtdIns-3,4,5-P3, transduce the signal to immediate effectors and then onto cellular responses? The first clue came from Fesik and Harlan, who showed that PH (pleckstrin homology) domains bind with high affinity to phosphoinositides, in their case, PtdIns-4,5-P2 (Harlan et al. 1994). One year later, Tsichlis working with Kaplan and Franke published a seminal finding identifying the first bona-fide effector of PI 3-K, the Ser/Thr kinase Akt (Franke et al. 1995). Akt had been originally identified by Tsichlis as the cellular homolog of the transforming retroviral oncogene v-Akt (Bellacosa et al. 1991). It was independently identified by Woodgett and termed PKB (protein kinase B), as it was sequence-related to PKA (protein kinase A) and PKC (protein kinase C) (Coffer and Woodgett 1991), and also by Hemmings as Rac (related to A and C kinases (Jones et al. 1991)). Akt contains a PH domain, and this prompted Tsichlis and colleagues to investigate whether Akt is PI 3-K target, and indeed they showed that PDGF stimulates Akt activation in a PI 3-K-dependent manner (Franke et al. 1995). Burgering and Coffer came to the same conclusion independently (Burgering and Coffer 1995). Initially suggested to be mediated by PtdIns-3-P, several groups subsequently showed that the PH domain of Akt has high affinity for PtdIns-3,4-P2 and PtdIns-3,4,5-P3 (Frech et al. 1997; Andjelkovic et al. 1997; Franke et al. 1997). Although several groups showed that PI 3-K lipids, particularly PtdIns-3,4-P2, could weakly activate Akt in vitro, it was not until two groups independently discovered PDK-1 (phosphoinositide-dependent kinase-1), the enzyme that is responsible for full Akt activation. Working with Hemmings and Cohen, Alessi had originally shown that Akt is phosphorylated at two key residues, Thr308 in the T loop segment of the kinase domain, and Ser473 in a hydrophobic region at the carboxyl-terminus (Alessi et al. 1996). Later, working in the lab of Cohen, Alessi set about to purify the enzymatic activity capable of phosphorylating and activating Akt. This led to the identification of PDK-1 as the Thr308 upstream kinase (Alessi et al. 1997). Once again, Hawkins and Stephens working with Stokoe independently identified the same activity, and they further showed that PDK-1 phosphorylates Akt at Thr308 in a manner that depends on PtdIns-3,4,5-P3 binding to the Akt PH domain (Stokoe et al. 1997). Subsequent studies by Alessi and other laboratories demonstrated that PDK-1 is the master upstream T loop kinase for many AGC kinases, such as S6K, PKCs, SGKs and several others (Alessi 2001; Mora et al. 2004). As for Ser473, whose phosphorylation contributes to full Akt activation, several upstream kinases were proposed over the years, and while some of these may function in specific settings, it wasn’t until 2005 when Sabatini’s group showed that the mTORC2 (mammalian target of rapamycin complex 2) complex represents the physiologically-relevant Ser473 kinase, at least in most cells and tissues (Sarbassov et al. 2005). It is also important to note that PH domains with very discrete phosphoinositide-binding specifies are now known to exist (Lemmon 2008). Moreover, since the discovery of PH domains, other domains in signaling proteins have been described that bind other 3 phosphoinositides, such as the PX and FYVE domains, providing the mechanistic basis for the regulation of specific cellular responses to individual PI 3-K lipid products.
Once Akt was identified as a direct effector of PI 3-K and PtdIns-3,4,5-P3, the field moved quickly to identify the immediate substrates of Akt that would transduce the signal to a specific response. Cohen identified the first Akt substrate, GSK-3b, a rate-limiting enzyme in the regulation of glycogen synthase in insulin-responsive cells and tissues (Cross et al. 1995). Because Cooper initially showed that PI 3-K comprises a key cellular survival pathway (Yao and Cooper 1995), this prompted Greenberg and colleagues to subsequently show that indeed Akt is one of the PI 3-K effectors in cellular survival mechanisms (Dudek et al. 1997). His laboratory also showed that the pro-apoptotic protein BAD is an Akt substrate whereby phosphorylation attenuates its apoptotic function (Datta et al. 1997). Secondly, Brunet also working in Greenberg’s lab showed that the pro-apoptotic FOXO3a transcription factor is phosphorylated by Akt, and in turn this blocks its apoptotic function (Brunet et al. 1999). Burgering’s lab published the same finding for AFX (Kops et al. 1999; Morris et al. 1996). It is important to note that much of the work on the importance of PI 3-K and Akt signaling in cellular survival was pioneered by Ruvkun and colleagues, who provided irrefutable genetic evidence for a linear pathway in Caenorhabditis elegans comprising PI 3-K (age-1) (Morris et al. 1996), PDK-1 (pdk-1) (Paradis et al. 1999), Akt (akt-1 and akt-2) (Paradis and Ruvkun 1998), and FOXO (daf-16) (Ogg et al. 1997) that controls survival and lifespan in the nematode worm. Similar findings using Drosophila genetics also corroborated the importance of PI 3-K signaling to cellular growth and survival (Leevers et al. 1996).
At the start of the new millennium, the PI 3-K field was firmly established as one of the central mechanisms by which extracellular stimuli initiate a lipid signaling pathway that culminates in the synthesis of PtdIns-3,4,5-P3, activation of downstream effectors such as Akt, and in turn alterations in cell growth, proliferation, survival, migration and gene transcription. Much of this had been achieved using biochemistry and cell-based assays with overexpression, and inhibitors such as wortmannin. What was missing was a genetic approach to begin to probe the contributions of individual regulatory p85 and catalytic p110 subunits in normal physiology and disease. In 1999, Kadowaki’s group published the first p85a (PIK3R1) knockout mouse which displayed increased insulin sensitivity and hypoglycemia (Terauchi et al. 1999). Shortly thereafter, Fruman and Cantley reported that ablation of all three splice variants encoded by p85a (p85a, p55a and p50a) results in perinatal lethality due to hepatocyte necrosis and chylous ascites (Fruman et al. 2000). Similar to Kadawaki’s finding, they also reported that loss of p85a alone results in hypoglycemia and increased insulin sensitivity. Knockout of p85b has also been reported, with phenotypes similar to p85a null mice (Ueki et al. 2002). At about the same time reports began to emerge on the knockouts of the p110 catalytic subunits. Knockout of p110a and p110b results in early embryonic lethality, and this has precluded a detailed analysis of the role of these isoforms in adult tissues (Bi et al. 1999, 2002). On the other hand, considerable information on p110 isoform-specific signaling has been gained by analysis of the p110g and p110d isoforms, which are preferentially, although by no means exclusively, expressed in the immune system. Hirsch and Wymann showed that p110g knockout mice are viable, yet their neutrophils show an impaired respiratory burst and migration phenotype (Hirsch et al. 2000). Macrophages from these mice reveal impaired motility, and defective accumulation in a sepsis model. Interestingly, both Hirsch, Wymann and Penninger also reported effects on cardiomyocyte contractility that at least in one case was attributed to a non-kinase scaffolding function of p110g that functions through a cyclic AMP (cAMP) and phosphodiesterase (PDE3B) pathway (Crackower et al. 2002; Patrucco et al. 2004). Two groups also independently deleted p110d, and also generated ‘knock-in’ mice, and although the mice are viable, they reveal impaired immune cell development and function, much like the p110g null mice (Clayton et al. 2002; Jou et al. 2002). In combination, these studies have provided compelling evidence that p110g and p110d are viable targets for immunity and cardiac dysfunctions. To this end, specific p110g and p110d inhibitors are currently undergoing clinical trials for a range of immunological pathologies (see below and Vol. I, Chap. 6 by Hirsch for more details).
4.4 Linking PtdIns-3,4,5-P3 to Cancer
A major leap forward in our understanding of the importance of PI 3-K signaling in pathophysiology was made by the discovery of the PtdIns-3,4,5-P3 phosphatase PTEN. Parsons group had been studying a region on chromosome 10q23 known to be frequently subjected to loss of heterozygosity (LOH), particularly in prostate cancer. This led to speculation that a yet-to-be-discovered tumor suppressor gene resides in this area whose loss would predispose to malignancy in several cancer types. Li, Parsons and colleagues used positional cloning to identify an open reading frame that encoded a protein with homologies to protein tyrosine phosphatases, chicken tensin and bovine auxilin (Li et al. 1997). They therefore named the gene PTEN, for phosphatase and tensin homolog deleted on chromosome 10. The same year Steck and colleagues also independently cloned the same gene and named it MMAC1, for mutated in multiple advanced cancers (Steck et al. 1997). Both groups recognized the striking homology to the catalytic domain of protein tyrosine phosphatases. However, the discovery that PTEN is a PtdIns-3,4,5-P3 phosphatase is attributed to Dixon and Maehama (Maehama and Dixon 1998). Dixon, an expert in protein tyrosine phosphatases, realized that in vitro PTEN has a much higher specific activity towards acidic and thus negatively-charged phosphopeptides compared to uncharged peptides. He then recognized that PTEN might utilize lipid rather than proteinaceous substrates, and recalled that PtdIns-3,4,5-P3 is the most negatively charged lipid in the cell. Sure enough, when PtdIns-3,4,5-P3 was used as substrate, PTEN efficiently dephosphorylated it back to PtdIns-4,5-P2, providing evidence that PTEN is the first identified 3 phosphoinositide phosphatase (Maehama and Dixon 1998). Given that PTEN LOH is a frequent event in various human solid tumors, this once again bolstered the link between PI 3-K, PtdIns-3,4,5-P3 and tumorigenesis. We now know that inactivating mutations, deletions and LOH are frequent events in the PTEN locus, to the extent that over 30% of glioblastoma patients have PTEN loss leading to hyperactivation of PI 3-K and Akt signaling (Keniry and Parsons 2008).
It took almost 20 years since the discovery of PI 3-K for researchers to identify oncogenic activating mutations in PIK3CA, the gene that encodes p110a. In spite of the findings that PI 3-K associated with transforming oncogenes, that it mediates growth factor signaling in response to mitogens, and the fact that the PtdIns-3,4,5-P3 phosphatase PTEN is a tumor suppressor, the discovery in 2004 that PIK3CA is an oncogene frequently mutated in human solid tumors was the first real direct evidence linking this pathway to cancer (Samuels et al. 2004; Campbell et al. 2004). In certain tumors, for example estrogen receptor positive breast cancer, PIK3CA is the most frequently mutated oncogene, leading to hyperactivation of Akt and other PI 3-K effectors to drive cellular proliferation, survival and metastasis (reviewed by (Engelman 2009)). Surprisingly, no oncogenic activating mutations have been detected in other p110 isoforms, particularly PIK3CB, yet studies have clearly demonstrated that p110b can promote tumorigenesis, particularly in settings on PTEN loss (Zhao et al. 2005; Jiang et al. 2010). On the other hand, mutations in PI 3-K regulatory subunits, for example PIK3R1 are found in human tumors, albeit at relatively low frequencies, and thus may activate p110b and p110d indirectly (Jaiswal et al. 2009). Similarly, activating oncogenic mutations in Akt isoforms have also been described in various human tumors, again at low frequency (Carpten et al. 2007). The frequency and spectrum of PTEN LOH and mutations, PIK3CA amplifications and mutations, PIK3R1 and Akt oncogenic mutations render PI 3-K one of the most frequently mutated and druggable pathways in human cancer. It is not surprising therefore that numerous clinical trials for novel drugs are targeting one or more of these enzymes for therapeutic benefit. Presently the compound in the most advanced trials is the dual mTOR and PI 3-K inhibitor NVP-BEZ235 from Novartis (Maira et al. 2008). Other dual TOR/PI 3-K inhibitors are also being evaluated for targeted therapy, and this is because the TORC1 pathway effects a negative feedback loop on PI 3-K, such that inhibition of TORC1 with rapamycin can actually enhance PI 3-K/Akt activity in vivo (O’Reilly et al. 2006). As already discussed, specific p110b inhibitors are also in trials for treatment of thrombosis, and a p110d inhibitor is being evaluated for hematological malignancies, in addition to a range of next generation p110a and p110b inhibitors (reviewed by (Vanhaesebroeck et al. 2010)). While the outlook for the use of these inhibitors is promising, it is likely that they will be used in dual targeted therapies with drugs that block activated or amplified receptor tyrosine kinases (such as EGF-R or Met) or drugs that interfere with ERK activity. Moreover, while some or possibly even many of the PI 3-K inhibitors currently in trials will fail, this will be of significant benefit to the research community who will use these much more specific inhibitors for basic research. It is also reasonable to argue that the use of these inhibitors, coupled with global RNAi screening approaches in tumor cells as well as model organisms such as flies will result in the identification of previously unappreciated mechanisms by which PI 3-K promotes disease states.
In this context, much of the activity and function of PI 3-K in both normal physiology and human disease has been attributed to Akt, most likely because of the availability of numerous reagents for studying Akt biology. Yet it is clear that a significant proportion of PI 3-K and PtdIns-3,4,5-P3 and PtdIns-3,4-P2 signaling is mediated by non-Akt effectors. Indeed a recent RNAi screening experiment revealed that in certain breast cancer cells harboring oncogenic PIK3CA, Akt is actually dispensable for growth and proliferation, yet both PDK-1 and SGK3 (serum and glucocorticoid-regulated kinase-3) are required (Vasudevan et al. 2009). Because SGK3 has a PX domain that interacts primarily with PtdIns-3-P in endosomes, the suggestion is that PIK3CA may mediate oncogenic signaling in certain human tumors through PtdIns-3-P at the endosome. Moreover, other targets of PI 3-K likely function independently of Akt, for example the tyrosine kinase Btk which binds with high affinity to PtdIns-3,4,5-P3 and PtdIns-3,4-P2 (Isakoff et al. 1998; Fruman et al. 2002). A large family of PtdIns-3,4,5-P3 binding proteins include guanine nucleotide exchange factors (GEFs) and GTPase activating proteins (GAPs) for members of the Rho superfamily of small GTPases. Examples are the proteins pREX2a and P-REX1 which function as GEFs for Rac and integrate PtdIns-3,4,5-P3 and heterotrimeric G protein signaling in cancer (Fine et al. 2009; Sosa et al. 2010).
4.5 Challenges Ahead
Due to the enormous advances in PI 3-K signaling and biology during the past two decades, we are presently at the stage where this pathway has entered the clinical arena and there is the realistic expectation that current drugs, or their derivatives, that target enzymes in the PI 3-K pathway will ultimately prove to have therapeutic value. This should not be taken as meaning that we now have learned the major mechanisms by which the PI 3-K pathway controls human biology and disease. There are numerous gaps in our understanding as to how PI 3-K and its lipid products mediate signal relay, and how deregulation of these mechanisms results in pathology, and yet new discoveries are constantly being made. Even though we knew back in the 1990s that PtdIns-3,4-P2 has a signaling role per se, for example in the binding and regulation of Akt, it wasn’t until the polyphosphates 4 phosphatase INPP4B was shown to function as a tumor suppressor that the exclusive role of this phosphoinositide was directly demonstrated (Gewinner et al. 2009). The implication is that in addition to Akt there likely exist other direct PtdIns-3,4-P2 effectors. Another major unexplored area of signaling in the field is the specific roles played by p110 isoforms. Much information has been garnered from studies of individual p110 knockout mice, and also cell-based studies using RNAi. What is lacking is a more compete understanding how individual p110 isoforms, perhaps in specific cellular compartments, elicit pools of PtdIns-3,4-P2 and PtdIns-3,4,5-P3 at discrete locations, and in turn how this affects downstream signaling. Compartmentalized PI 3-K signaling is likely to have a profound impact on certain cellular responses, for example remodeling of the actin cytoskeleton which requires specific temporal as well as kinetic regulation during directed cell migration. Genetically-encoded FRET-based sensors to monitor both PI 3-K effectors, as well as PtdIns-3,4,5-P3/PtdIns-3,4-P2 themselves will likely aid in this pursuit. This issue also applies to the nuclear phosphoinositide cycle, first recognized and pioneered by Irvine, Divecha and Cocco (Cocco et al. 1987; Divecha et al. 1991). Even before the discovery of PI 3-K we knew there exists a nuclear phosphoinositide cycle, and nuclear phosphoinositides had been detected (Cocco et al. 1987). Since then, both PI 3-K and its lipid products have been detected inside the nucleus, consistent with the model that certain targets of Akt, for example FOXO3a, are actually phosphorylated in the nucleus, and subsequently exported to the cytoplasm thus terminating transcriptional activity (Brunet et al. 2002). Yet there remain many gaps in our knowledge of the regulation and function of nuclear PI 3-K signaling. Many of the effectors of PI 3-K also exist as multi-gene families, a good example is Akt (Akt1, Akt2 and Akt3 in mammals). While it was originally assumed that they would function redundantly, we now know that there are specific functions and even specific substrates of Akt isoforms. For example, in the context of cancer progression, Akt2 promotes tumor invasion and metastasis, whereas Akt1 either does not or even attenuates this phenotype (Yoeli-Lerner et al. 2005; Irie et al. 2005; Maroulakou et al. 2007). These findings of opposing or non-redundant function for PI 3-K effectors have profound implications for drugs that are being developed for therapeutic intervention in the PI 3-K/Akt pathway. Whether similar opposing phenotypes exist for other PI 3-K effectors remains to be determined, but is likely to be the case. It is also very likely that additional feedback loops exist in the pathway, analogous to the mTOR/S6K1 negative feedback loop. If this is the case, this will make the use of PI 3-K and Akt inhibitors somewhat of a challenge. Moreover, although powerful genetic models exist, such as p110 isoform knockout as well as transgenes, the next generation models will likely be considerably more complex, and recapitulate the multiple genetic lesions in the PI 3-K pathway that we now know exist in human cancer patients. These can then be used in combination with the latest PI 3-K inhibitors to more accurately model human cancer therapy in preclinical models.
In closing, this chapter has highlighted the major discoveries in the PI 3-K field in some two and a half decades. From the identification of the first novel phosphoinositide PtdIns-3-P in the mid 1980s, to the discovery of oncogenic PIK3CA mutations in the mid 2000s, to the present day with clinical trials for p110 inhibitors, the field has matured at a dizzying pace. Yet we have clearly much more to learn as we await the results from the PI 3-K inhibitor clinical trials. We are now at a point where biochemical studies, genome-wide screening approaches and clinical trials are all informing the basic and clinical arena, and the field is poised to capture this information and much more efficiently translate it into therapeutic benefit.
References
Alessi DR (2001) Discovery of PDK1, one of the missing links in insulin signal transduction. Colworth Medal Lecture. Biochem Soc Trans 29:1–14
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P, Hemmings BA (1996) Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J 15:6541–6551
Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB, Cohen P (1997) Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr Biol 7:261–269
Andjelkovic M, Alessi DR, Meier R, Fernandez A, Lamb NJ, Frech M, Cron P, Cohen P, Lucocq JM, Hemmings BA (1997) Role of translocation in the activation and function of protein kinase B. J Biol Chem 272:31515–31524
Arcaro A, Wymann MP (1993) Wortmannin is a potent phosphatidylinositol 3-kinase inhibitor: the role of phosphatidylinositol 3,4,5-trisphosphate in neutrophil responses. Biochem J 296(Pt 2):297–301
Auger KR, Serunian LA, Soltoff SP, Libby P, Cantley LC (1989) PDGF-dependent tyrosine phosphorylation stimulates production of novel polyphosphoinositides in intact cells. Cell 57:167–175
Bellacosa A, Testa JR, Staal SP, Tsichlis PN (1991) A retroviral oncogene, akt, encoding a serine-threonine kinase containing an SH2-like region. Science 254:274–277
Berridge MJ (1983) Rapid accumulation of inositol trisphosphate reveals that agonists hydrolyse polyphosphoinositides instead of phosphatidylinositol. Biochem J 212:849–858
Berridge MJ, Dawson RM, Downes CP, Heslop JP, Irvine RF (1983) Changes in the levels of inositol phosphates after agonist-dependent hydrolysis of membrane phosphoinositides. Biochem J 212:473–482
Bi L, Okabe I, Bernard DJ, Wynshaw-BORIS A, Nussbaum RL (1999) Proliferative defect and embryonic lethality in mice homozygous for a deletion in the p110alpha subunit of phosphoinositide 3-kinase. J Biol Chem 274:10963–10968
Bi L, Okabe I, Bernard DJ, Nussbaum RL (2002) Early embryonic lethality in mice deficient in the p110beta catalytic subunit of PI 3-kinase. Mamm Genome 13:169–172
Brown WJ, Dewald DB, Emr SD, Plutner H, Balch WE (1995) Role for phosphatidylinositol 3-kinase in the sorting and transport of newly synthesized lysosomal enzymes in mammalian cells. J Cell Biol 130:781–796
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, Anderson MJ, Arden KC, Blenis J, Greenberg ME (1999) Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96:857–868
Brunet A, Kanai F, Stehn J, Xu J, Sarbassova D, Frangioni JV, Dalal SN, Decaprio JA, Greenberg ME, Yaffe MB (2002) 14–3-3 transits to the nucleus and participates in dynamic nucleocytoplasmic transport. J Cell Biol 156:817–828
Burgering BM, Coffer PJ (1995) Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature 376:599–602
Campbell IG, Russell SE, Choong DY, Montgomery KG, Ciavarella ML, Hooi CS, Cristiano BE, Pearson RB, Phillips WA (2004) Mutation of the PIK3CA gene in ovarian and breast cancer. Cancer Res 64:7678–7681
Carpenter CL, Duckworth BC, Auger KR, Cohen B, Schaffhausen BS, Cantley LC (1990) Purification and characterization of phosphoinositide 3-kinase from rat liver. J Biol Chem 265:19704–19711
Carpten JD, Faber AL, Horn C, Donoho GP, Briggs SL, Robbins CM, Hostetter G, Boguslawski S, Moses TY, Savage S, Uhlik M, Lin A, Du J, Qian YW, Zeckner DJ, Tucker-KELLOGG G, Touchman J, Patel K, Mousses S, Bittner M, Schevitz R, Lai MH, Blanchard KL, Thomas JE (2007) A transforming mutation in the pleckstrin homology domain of AKT1 in cancer. Nature 448:439–444
Chung J, Grammer TC, Lemon KP, Kazlauskas A, Blenis J (1994) PDGF- and insulin-dependent pp70S6k activation mediated by phosphatidylinositol-3-OH kinase. Nature 370:71–75
Clayton E, Bardi G, Bell SE, Chantry D, Downes CP, Gray A, Humphries LA, Rawlings D, Reynolds H, Vigorito E, Turner M (2002) A crucial role for the p110delta subunit of phosphatidylinositol 3-kinase in B cell development and activation. J Exp Med 196:753–763
Cocco L, Gilmour RS, Ognibene A, Letcher AJ, Manzoli FA, Irvine RF (1987) Synthesis of polyphosphoinositides in nuclei of Friend cells. Evidence for polyphosphoinositide metabolism inside the nucleus which changes with cell differentiation. Biochem J 248:765–770
Coffer PJ, Woodgett JR (1991) Molecular cloning and characterisation of a novel putative protein- serine kinase related to the cAMP-dependent and protein kinase C families [published erratum appears in Eur J Biochem 1992 May 1;205(3):1217]. Eur J Biochem 201:475–481
Crackower MA, Oudit GY, Kozieradzki I, Sarao R, Sun H, Sasaki T, Hirsch E, Suzuki A, Shioi T, Irie-SASAKI J, Sah R, Cheng HY, Rybin VO, Lembo G, Fratta L, Oliveira-DOS-SANTOS AJ, Benovic JL, Kahn CR, Izumo S, Steinberg SF, Wymann MP, Backx PH, Penninger JM (2002) Regulation of myocardial contractility and cell size by distinct PI3K-PTEN signaling pathways. Cell 110:737–749
Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA (1995) Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378:785–789
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y, Greenberg ME (1997) Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91:231–241
De Camilli P, Emr SD, McPherson PS, Novick P (1996) Phosphoinositides as regulators in membrane traffic. Science 271:1533–1539
Declue JE, Sadowski I, Martin GS, Pawson T (1987) A conserved domain regulates interactions of the v-fps protein-tyrosine kinase with the host cell. Proc Natl Acad Sci U S A 84:9064–9068
Divecha N, Banfic H, Irvine RF (1991) The polyphosphoinositide cycle exists in the nuclei of Swiss 3T3 cells under the control of a receptor (for IGF-I) in the plasma membrane, and stimulation of the cycle increases nuclear diacylglycerol and apparently induces translocation of protein kinase C to the nucleus. EMBO J 10:3207–3214
Dudek H, Datta SR, Franke TF, Birnbaum MJ, Yao R, Cooper GM, Segal RA, Kaplan DR, Greenberg ME (1997) Regulation of neuronal survival by the serine-threonine protein kinase Akt. Science 275:661–665
Engelman JA (2009) Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer 9:550–562
Engelman JA, Luo J, Cantley LC (2006) The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet 7:606–619
Escobedo JA, Kaplan DR, Kavanaugh WM, Turck CW, Williams LT (1991) A phosphatidylinositol-3 kinase binds to platelet-derived growth factor receptors through a specific receptor sequence containing phosphotyrosine. Mol Cell Biol 11:1125–1132
Fine B, Hodakoski C, Koujak S, Su T, Saal LH, Maurer M, Hopkins B, Keniry M, Sulis ML, Mense S, Hibshoosh H, Parsons R (2009) Activation of the PI3K pathway in cancer through inhibition of PTEN by exchange factor P-REX2a. Science 325:1261–1265
Franke TF, Yang SI, Chan TO, Datta K, Kazlauskas A, Morrison DK, Kaplan DR, Tsichlis PN (1995) The protein kinase encoded by the Akt proto-oncogene is a target of the PDGF-activated phosphatidylinositol 3-kinase. Cell 81:727–736
Franke TF, Kaplan DR, Cantley LC, Toker A (1997) Direct regulation of the Akt proto-oncogene product by phosphatidylinositol-3,4-bisphosphate. Science 275:665–668
Frech M, Andjelkovic M, Ingley E, Reddy KK, Falck JR, Hemmings BA (1997) High affinity binding of inositol phosphates and phosphoinositides to the pleckstrin homology domain of RAC/protein kinase B and their influence on kinase activity. J Biol Chem 272:8474–8481
Fruman DA, Mauvais-Jarvis F, Pollard DA, Yballe CM, Brazil D, Bronson RT, Kahn CR, Cantley LC (2000) Hypoglycaemia, liver necrosis and perinatal death in mice lacking all isoforms of phosphoinositide 3-kinase p85 alpha. Nat Genet 26:379–382
Fruman DA, Ferl GZ, An SS, Donahue AC, Satterthwaite AB, Witte ON (2002) Phosphoinositide 3-kinase and Bruton’s tyrosine kinase regulate overlapping sets of genes in B lymphocytes. Proc Natl Acad Sci U S A 99:359–364
Funamoto S, Milan K, Meili R, Firtel RA (2001) Role of phosphatidylinositol 3¢ kinase and a downstream pleckstrin homology domain-containing protein in controlling chemotaxis in dictyostelium. J Cell Biol 153:795–810
Gewinner C, Wang ZC, Richardson A, Teruya-FELDSTEIN J, Etemadmoghadam D, Bowtell D, Barretina J, Lin WM, Rameh L, Salmena L, Pandolfi PP, Cantley LC (2009) Evidence that inositol polyphosphate 4-phosphatase type II is a tumor suppressor that inhibits PI3K signaling. Cancer Cell 16:115–125
Harlan JE, Hajduk PJ, Yoon HS, Fesik SW (1994) Pleckstrin homology domains bind to phosphatidylinositol-4,5-bisphosphate. Nature 371:168–170
Hawkins PT, Anderson KE, Davidson K, Stephens LR (2006) Signalling through Class I PI3Ks in mammalian cells. Biochem Soc Trans 34:647–662
Herman PK, Emr SD (1990) Characterization of VPS34, a gene required for vacuolar protein sorting and vacuole segregation in Saccharomyces cerevisiae. Mol Cell Biol 10:6742–6754
Hiles ID, Otsu M, Volinia S, Fry MJ, Gout I, Dhand R, Panayotou G, Ruiz LF, Thompson A, Totty NF et al (1992) Phosphatidylinositol 3-kinase: structure and expression of the 110 kd catalytic subunit. Cell 70:419–429
Hirsch E, Katanaev VL, Garlanda C, Azzolino O, Pirola L, Silengo L, Sozzani S, Mantovani A, Altruda F, Wymann MP (2000) Central role for G protein-coupled phosphoinositide 3-kinase gamma in inflammation. Science 287:1049–1053
Hokin LE, Hokin MR (1953) The incorporation of 32P into the nucleotides of ribonucleic acid in pigeon pancreas slices. Biochim Biophys Acta 11:591–592
Inoue M, Kishimoto A, Takai Y, Nishizuka Y (1977) Studies on a cyclic nucleotide-independent protein kinase and its proenzyme in mammalian tissues. II. Proenzyme and its activation by calcium-dependent protease from rat brain. J Biol Chem 252:7610–7616
Irie HY, Pearline RV, Grueneberg D, Hsia M, Ravichandran P, Kothari N, Natesan S, Brugge JS (2005) Distinct roles of Akt1 and Akt2 in regulating cell migration and epithelial-mesenchymal transition. J Cell Biol 171:1023–1034
Isakoff SJ, Cardozo T, Andreev J, Li Z, Ferguson KM, Abagyan R, Lemmon MA, Aronheim A, Skolnik EY (1998) Identification and analysis of PH domain-containing targets of phosphatidylinositol 3-kinase using a novel in vivo assay in yeast. EMBO J 17:5374–5387
Jaiswal BS, Janakiraman V, Kljavin NM, Chaudhuri S, Stern HM, Wang W, Kan Z, Dbouk HA, Peters BA, Waring P, Dela VEGA T, Kenski DM, Bowman KK, Lorenzo M, Li H, Wu J, Modrusan Z, Stinson J, Eby M, Yue P, Kaminker JS, De Sauvage FJ, Backer JM, Seshagiri S (2009) Somatic mutations in p85alpha promote tumorigenesis through class IA PI3K activation. Cancer Cell 16:463–474
Jiang X, Chen S, Asara JM, Balk SP (2010) Phosphoinositide 3-kinase pathway activation in phosphate and tensin homolog (PTEN)-deficient prostate cancer cells is independent of receptor tyrosine kinases and mediated by the p110beta and p110delta catalytic subunits. J Biol Chem 285:14980–14989
Jones PF, Jakubowicz T, Pitossi FJ, Maurer F, Hemmings BA (1991) Molecular cloning and identification of a serine/threonine protein kinase of the second-messenger subfamily. Proc Natl Acad Sci U S A 88:4171–4175
Jones SM, Klinghoffer R, Prestwich GD, Toker A, Kazlauskas A (1999) PDGF induces an early and a late wave of PI 3-kinase activity, and only the late wave is required for progression through G1. Curr Biol 9:512–521
Jou ST, Carpino N, Takahashi Y, Piekorz R, Chao JR, Wang D, Ihle JN (2002) Essential, nonredundant role for the phosphoinositide 3-kinase p110delta in signaling by the B-cell receptor complex. Mol Cell Biol 22:8580–8591
Kaplan DR, Whitman M, Schaffhausen B, Pallas DC, White M, Cantley L, Roberts TM (1987) Common elements in growth factor stimulation and oncogenic transformation: 85 kd phosphoprotein and phosphatidylinositol kinase activity. Cell 50:1021–1029
Kazlauskas A, Cooper JA (1989) Autophosphorylation of the PDGF receptor in the kinase insert region regulates interactions with cell proteins. Cell 58:1121–1133
Keniry M, Parsons R (2008) The role of PTEN signaling perturbations in cancer and in targeted therapy. Oncogene 27:5477–5485
Kirk CJ, Creba JA, Downes CP, Michell RH (1981) Hormone-stimulated metabolism of inositol lipids and its relationship to hepatic receptor function. Biochem Soc Trans 9:377–379
Kops GJ, De Ruiter ND, De Vries-Smits AM, Powell DR, Bos JL, Burgering BM (1999) Direct control of the Forkhead transcription factor AFX by protein kinase B. Nature 398:630–634
Lapetina EG, Michell RH (1973) A membrane-bound activity catalysing phosphatidylinositol breakdown to 1,2-diacylglycerol, D-myoinositol 1:2-cyclic phosphate an D-myoinositol 1-phosphate. Properties and subcellular distribution in rat cerebral cortex. Biochem J 131:433–442
Leevers SJ, Weinkove D, Macdougall LK, Hafen E, Waterfield MD (1996) The Drosophila phosphoinositide 3-kinase Dp110 promotes cell growth. EMBO J 15:6584–6594
Lemmon MA (2008) Membrane recognition by phospholipid-binding domains. Nat Rev Mol Cell Biol 9:99–111
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI, Puc J, Miliaresis C, Rodgers L, McCombie R, Bigner SH, Giovanella BC, Ittmann M, Tycko B, Hibshoosh H, Wigler MH, Parsons R (1997) PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer [see comments]. Science 275:1943–1947
Macara IG, Marinetti GV, Balduzzi PC (1984) Transforming protein of avian sarcoma virus UR2 is associated with phosphatidylinositol kinase activity: possible role in tumorigenesis. Proc Natl Acad Sci U S A 81:2728–2732
Maehama T, Dixon JE (1998) The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem 273:13375–13378
Maira SM, Stauffer F, Brueggen J, Furet P, Schnell C, Fritsch C, Brachmann S, Chene P, De Pover A, Schoemaker K, Fabbro D, Gabriel D, Simonen M, Murphy L, Finan P, Sellers W, Garcia-Echeverria C (2008) Identification and characterization of NVP-BEZ235, a new orally available dual phosphatidylinositol 3-kinase/mammalian target of rapamycin inhibitor with potent in vivo antitumor activity. Mol Cancer Ther 7:1851–1863
Maroulakou IG, Oemler W, Naber SP, Tsichlis PN (2007) Akt1 ablation inhibits, whereas Akt2 ablation accelerates, the development of mammary adenocarcinomas in mouse mammary tumor virus (MMTV)-ErbB2/neu and MMTV-polyoma middle T transgenic mice. Cancer Res 67:167–177
Matsuda M, Mayer B, Fukui Y, Hanafusa H (1990) Binding of transforming protein p47gag-crk to a broad range of phosphotyrosine-containing proteins. Science 248:1537–1539
Mora A, Komander D, Van Aalten DM, Alessi DR (2004) PDK1, the master regulator of AGC kinase signal transduction. Semin Cell Dev Biol 15:161–170
Moran MF, Koch CA, Anderson D, Ellis C, England L, Martin GS, Pawson T (1990) Src homology region 2 domains direct protein-protein interactions in signal transduction. Proc Natl Acad Sci U S A 87:8622–8626
Morris JZ, Tissenbaum HA, Ruvkun G (1996) A phosphatidylinositol-3-OH kinase family member regulating longevity and diapause in Caenorhabditis elegans. Nature 382:536–539
O’Brien MC, Fukui Y, Hanafusa H (1990) Activation of the proto-oncogene p60c-src by point mutations in the SH2 domain. Mol Cell Biol 10:2855–2862
Ogg S, Paradis S, Gottlieb S, Patterson GI, Lee L, Tissenbaum HA, Ruvkun G (1997) The Fork head transcription factor DAF-16 transduces insulin-like metabolic and longevity signals in C. elegans. Nature 389:994–999
O’Reilly KE, Rojo F, She QB, Solit D, Mills GB, Smith D, Lane H, Hofmann F, Hicklin DJ, Ludwig DL, Baselga J, Rosen N (2006) mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res 66:1500–1508
Otsu M, Hiles I, Gout I, Fry MJ, Ruiz LF, Panayotou G, Thompson A, Dhand R, Hsuan J, Totty N, et al (1991) Characterization of two 85 kd proteins that associate with receptor tyrosine kinases, middle-T/pp60c-src complexes, and PI3-kinase. Cell 65:91–104
Paradis S, Ailion M, Toker A, Thomas JH, Ruvkun G (1999) A PDK1 homolog is necessary and sufficient to transduce AGE-1 PI3 kinase signals that regulate diapause in Caenorhabditis elegans. Genes Dev 13:1438–1452
Paradis S, Ruvkun G (1998) Caenorhabditis elegans Akt/PKB transduces insulin receptor-like signals from AGE-1 PI3 kinase to the DAF-16 transcription factor. Genes Dev 12:2488–2498
Patrucco E, Notte A, Barberis L, Selvetella G, Maffei A, Brancaccio M, Marengo S, Russo G, Azzolino O, Rybalkin SD, Silengo L, Altruda F, Wetzker R, Wymann MP, Lembo G, Hirsch E (2004) PI3Kgamma modulates the cardiac response to chronic pressure overload by distinct kinase-dependent and -independent effects. Cell 118:375–387
Rodriguez VP, Warne PH, Dhand R, Vanhaesebroeck B, Gout I, Fry MJ, Waterfield MD, Downward J (1994) Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 370:527–532
Samuels Y, Wang Z, Bardelli A, Silliman N, Ptak J, Szabo S, Yan H, Gazdar A, Powell SM, Riggins GJ, Willson JK, Markowitz S, Kinzler KW, Vogelstein B, Velculescu VE (2004) High frequency of mutations of the PIK3CA gene in human cancers. Science 304:554
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307:1098–1101
Schu PV, Takegawa K, Fry MJ, Stack JH, Waterfield MD, Emr SD (1993) Phosphatidylinositol 3-kinase encoded by yeast VPS34 gene essential for protein sorting. Science 260:88–91
Sjolander A, Yamamoto K, Huber BE, Lapetina EG (1991) Association of p21ras with phosphatidylinositol 3-kinase. Proc Natl Acad Sci U S A 88:7908–7912
Skolnik EY, Margolis B, Mohammadi M, Lowenstein E, Fischer R, Drepps A, Ullrich A, Schlessinger J (1991) Cloning of PI3 kinase-associated p85 utilizing a novel method for expression/cloning of target proteins for receptor tyrosine kinases. Cell 65:83–90
Sosa MS, Lopez-Haber C, Yang C, Wang H, Lemmon MA, Busillo JM, Luo J, Benovic JL, Klein-Szanto A, Yagi H, Gutkind JS, Parsons RE, Kazanietz MG (2010) Identification of the Rac-GEF P-Rex1 as an essential mediator of ErbB signaling in breast cancer. Mol Cell 40:877–892
Steck PA, Pershouse MA, Jasser SA, Yung WK, Lin H, Ligon AH, Langford LA, Baumgard ML, Hattier T, Davis T, Frye C, Hu R, Swedlund B, Teng DH, Tavtigian SV (1997) Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet 15:356–362
Stephens L, Hawkins PT, Downes CP (1989) Metabolic and structural evidence for the existence of a third species of polyphosphoinositide in cells: D-phosphatidyl-myo-inositol 3-phosphate. Biochem J 259:267–276
Stephens LR, Hughes KT, Irvine RF (1991) Pathway of phosphatidylinositol(3,4,5)-trisphosphate synthesis in activated neutrophils. Nature 351:33–39
Stokoe D, Stephens LR, Copeland T, Gaffney PR, Reese CB, Painter GF, Holmes AB, McCormick F, Hawkins PT (1997) Dual role of phosphatidylinositol-3,4,5-trisphosphate in the activation of protein kinase B. Science 277:567–570
Streb H, Irvine RF, Berridge MJ, Schulz I (1983) Release of Ca2+ from a nonmitochondrial intracellular store in pancreatic acinar cells by inositol-1,4,5-trisphosphate. Nature 306:67–69
Sugimoto Y, Whitman M, Cantley LC, Erikson RL (1984) Evidence that the Rous sarcoma transforming gene product phosphorylates phosphatidylinositol and diacylglycerol. Proc Natl Acad Sci U S A 81:2117–2121
Takai Y, Yamamoto M, Inoue M, Kishimoto A, Nishizuka Y (1977) A proenzyme of cyclic nucleotide-independent protein kinase and its activation by calcium-dependent neutral protease from rat liver. Biochem Biophys Res Commun 77:542–550
Terauchi Y, Tsuji Y, Satoh S, Minoura H, Murakami K, Okuno A, Inukai K, Asano T, Kaburagi Y, Ueki K, Nakajima H, Hanafusa T, Matsuzawa Y, Sekihara H, Yin Y, Barrett JC, Oda H, Ishikawa T, Akanuma Y, Komuro I, Suzuki M, Yamamura K, Kodama T, Suzuki H, Koyasu S, Aizawa S, Tobe K, Fukui Y, Yazaki Y, Kadowaki T (1999) Increased insulin sensitivity and hypoglycaemia in mice lacking the p85 alpha subunit of phosphoinositide 3-kinase. Nat Genet 21:230–235
Traynor-Kaplan AE, Harris AL, Thompson BL, Taylor P, Sklar LA (1988) An inositol tetrakisphosphate-containing phospholipid in activated neutrophils. Nature 334:353–356
Ueki K, Yballe CM, Brachmann SM, Vicent D, Watt JM, Kahn CR, Cantley LC (2002) Increased insulin sensitivity in mice lacking p85beta subunit of phosphoinositide 3-kinase. Proc Natl Acad Sci U S A 99:419–424
Valius M, Kazlauskas A (1993) Phospholipase C-gamma 1 and phosphatidylinositol 3 kinase are the downstream mediators of the PDGF receptor’s mitogenic signal. Cell 73:321–334
Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B (2010) The emerging mechanisms of isoform-specific PI3K signalling. Nat Rev Mol Cell Biol 11:329–341
Vasudevan KM, Barbie DA, Davies MA, Rabinovsky R, McNear CJ, Kim JJ, Hennessy BT, Tseng H, Pochanard P, Kim SY, Dunn IF, Schinzel AC, Sandy P, Hoersch S, Sheng Q, Gupta PB, Boehm JS, Reiling JH, Silver S, Lu Y, Stemke-HALE K, Dutta B, Joy C, Sahin AA, Gonzalez-Angulo AM, Lluch A, Rameh LE, Jacks T, Root DE, Lander ES, Mills GB, Hahn WC, Sellers WR, Garraway LA (2009) AKT-independent signaling downstream of oncogenic PIK3CA mutations in human cancer. Cancer Cell 16:21–32
Vlahos CJ, Matter WF, Hui KY, Brown RF (1994) A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4- morpholinyl)-8-phenyl-4H-1-benzopyran-4-one (LY294002). J Biol Chem 269:5241–5248
Welch HC, Coadwell WJ, Stephens LR, Hawkins PT (2003) Phosphoinositide 3-kinase-dependent activation of Rac. FEBS Lett 546:93–97
Wennstrom S, Hawkins P, Cooke F, Hara K, Yonezawa K, Kasuga M, Jackson T, Claesson-WELSH L, Stephens L (1994) Activation of phosphoinositide 3-kinase is required for PDGF-stimulated membrane ruffling. Curr Biol 4:385–393
Whitman M, Downes CP, Keeler M, Keller T, Cantley L (1988) Type I phosphatidylinositol kinase makes a novel inositol phospholipid, phosphatidylinositol-3-phosphate. Nature 332:644–646
Yano H, Nakanishi S, Kimura K, Hanai N, Saitoh Y, Fukui Y, Nonomura Y, Matsuda Y (1993) Inhibition of histamine secretion by wortmannin through the blockade of phosphatidylinositol 3-kinase in RBL-2H3 cells. J Biol Chem 268:25846–25856
Yao R, Cooper GM (1995) Requirement for phosphatidylinositol-3 kinase in the prevention of apoptosis by nerve growth factor. Science 267:2003–2006
Yoeli-Lerner M, Yiu GK, Rabinovitz I, Erhardt P, Jauliac S, Toker A (2005) Akt blocks breast cancer cell motility and invasion through the transcription factor NFAT. Mol Cell 20:539–550
Zhao JJ, Liu Z, Wang L, Shin E, Loda MF, Roberts TM (2005) The oncogenic properties of mutant p110alpha and p110beta phosphatidylinositol 3-kinases in human mammary epithelial cells. Proc Natl Acad Sci U S A 102:18443–18448
Zhou S, Shoelson SE, Chaudhuri M, Gish G, Pawson T, Haser WG, King F, Roberts T, Ratnofsky S, Lechleider RJ et al (1993) SH2 domains recognize specific phosphopeptide sequences. Cell 72:767–778
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
Toker, A. (2012). Phosphoinositide 3-Kinases—A Historical Perspective. In: Balla, T., Wymann, M., York, J. (eds) Phosphoinositides I: Enzymes of Synthesis and Degradation. Subcellular Biochemistry, vol 58. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-3012-0_4
Download citation
DOI: https://doi.org/10.1007/978-94-007-3012-0_4
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-3011-3
Online ISBN: 978-94-007-3012-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)