Abstract
Epilepsy affects 0.5–1 % of the pediatric population, and a relevant proportion of these patients are refractory to medical therapy. In such cases, surgical resection of the epileptic focus is often the treatment of choice. In this setting, nuclear medicine methods can provide useful information for the precise presurgical localization of the lesion, in association with MRI findings.
PET imaging, in particular interictal imaging of glucose metabolism by 18F-FDG–PET, may allow identification of the dysfunctional cortex giving rise to the epileptogenic activity, usually encompassing the epileptic focus and the areas of seizure propagation. In pediatric patients with temporal lobe epilepsy, interictal FDG–PET imaging has a good sensitivity, identifying the affected hemisphere in up to 85 % of the cases, including a relevant proportion of patients with no visible lesions on MRI. In the investigation of extratemporal epilepsy, the sensitivity is usually lower and depends on the specific etiology. However, even in these cases, FDG–PET is a useful tool to guide invasive EEG measures and to support MRI and EEG findings.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Temporal Lobe Epilepsy
- Tuberous Sclerosis
- Epilepsy Patient
- Epileptic Focus
- Temporal Lobe Epilepsy Patient
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
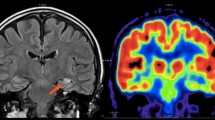
FDG–PET is a well-established functional imaging modality in the evaluation of pediatric patients with epilepsy [1, 12]. While ictal scans can be useful, the long duration required to reach steady-state glucose uptake (on the order of many minutes compared with partial seizures, which usually last <2 min) often leads to scans that contain a difficult-to-interpret mixture of interictal, ictal, and postictal states. In addition, the practical realization of an ictal FDG–PET study requires the coordination of radiotracer availability with the ictal event, which is difficult to assure. Consequently, ictal PET studies are rarely performed, and ictal SPECT perfusion studies are preferred instead. However, if ictal injection is feasible, ictal PET images may clearly depict the cortical area responsible for the epileptic event (Fig. 21.1).
Presurgical FDG–PET scans in epilepsy patients are typically obtained when the patient is in the interictal state, with the goal of detecting focal areas of decreased metabolism, i.e., relative hypometabolism, as these presumably reflect focal functional disturbances of cerebral activity associated with epileptogenic tissue. Nonetheless, the specific cause of hypometabolism in and near epileptogenic regions of the brain remains unclear [3].
A 5-year-old girl with Rasmussen’s encephalitis and partial seizures since the age of 3 years. (a) Axial PET image, (b) PET/MRI fusion. FDG was fortuitously administered during a seizure and shows, with very high spatial resolution and excellent image contrast, the involved cortex in the left hemisphere
2 FDG–PET in Temporal Lobe Epilepsy
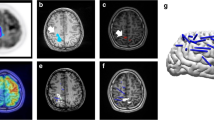
The value of FDG–PET has been best proven in the evaluation of medically refractory epilepsy patients who are candidates for surgery, specifically those with clinically suspected temporal lobe epilepsy. In this setting, the sensitivity of FDG–PET is between 80 and 90 % [1–4]. Only a few studies have addressed the sensitivity and specificity of FDG–PET in medial temporal lobe epilepsy patients with and without evidence of hippocampal sclerosis on MRI. However, FDG–PET is still considered to be reliable in lateralizing the epileptogenic temporal lobe even in MRI-negative patients, as shown in Fig. 21.2 [5]. Of course, MRI techniques are constantly evolving, and new sequences and new analytical approaches may one day allow the identification of focal alterations in cases previously considered to be “MRI negative.”
A 13-year-old boy with partial complex epileptic seizures. (a) Coronal PET image, (b) MRI image. MRI does not show any clear abnormality, while on the PET image, there is marked hypometabolism in the left temporal pole. A repeated MRI investigation identified an area of probable cortical dysplasia. A left anterior temporal lobectomy was planned
3 FDG–PET in Extratemporal Epilepsy
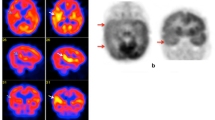
The clinical value of FDG–PET in neocortical epilepsy is less clear. The larger reported series consist of observational retrospective studies, and only a few were performed in the era of advanced MRI techniques [1, 6]. Most importantly, the FDG–PET findings in nonlesional neocortical epilepsy are often obtained from heterogeneous patients and patient groups. An example of the FDG pattern in a specific syndrome such as tuberous sclerosis is provided in Fig. 21.3 [7].
In tuberous sclerosis typically FDG-PET shows hypometabolism in and around tubers, believed to be due to decreased neuronal number and simplified dendritic pattern. A tuber with a disproportionately large area of hypometabolism compared with its size on MR images is most likely epileptogenic [8]. However, both epileptic and nonepileptic tubers show reduced uptake on FDG-PET, while promising results have been obtained using a tracer specific for the serotonergic system, the α-[11C]methyl-l-tryptophan (AMT). AMT-PET shows increased AMT uptake interictally in epileptic but not quiescent tubers in almost two-thirds of children with tuberous sclerosis and intractable epilepsy: all tubers with at least 10% AMT increase were found to be epileptogenic [11].
Co-registered multimodality imaging may provide other supportive localizing information confirming that a questionable PET metabolic abnormality is indeed a true disturbance reflective of the epileptogenic zone, thus emphasizing the added value of imaging fusion or hybrid imaging, when available [9, 13, 14].
A 5-year-old girl who at the age of 5 months was diagnosed with tuberous sclerosis and generalized epileptic seizures. (a) Axial MRI, (b) PET/MRI fusion, and (c) PET images. The PET/MRI fusion images show multiple cortical lesions in the two hemispheres, typically hypometabolic on FDG–PET imaging. However, these imaging modalities do not allow localization of the lesion generating the epileptic seizures. Instead, promising results were obtained using α-[11C]methyl-l-tryptophan (AMT), a tracer specific for the serotonergic system
4 Other PET Tracers
Many tracers have shown promising results in the molecular imaging of epilepsy, especially those targeting the GABAergic system (11C-flumazenil), the serotonergic system (11C-WAY and α-11C-methyl-l-tryptophan), the dopaminergic system, the glutamatergic system, nicotinic receptors, adenosine receptors, and opioid-based ligands. However, these tracers, mostly based on carbon-11 chemistry and on the availability of an on-site cyclotron, are still limited to dedicated research centers, and their description is beyond the scope of this chapter. The results obtained with these tracers were recently summarized in two reviews [10, 11, 12].
References
Kurian M, Spinelli L, Delavelle J et al (2007) Multimodality imaging for focus localization in pediatric pharmacoresistant epilepsy. Epileptic Disord 9:20–31
Ryvlin P, Bouvard S, Le Bars D et al (1998) Clinical utility of flumazenil-PET versus [18F]fluorodeoxyglucose-PET and MRI in refractory partial epilepsy. A prospective study in 100 patients. Brain 121:2067–2081
Knowlton RC (2006) The role of FDG-PET, ictal SPECT, and MEG in the epilepsy surgery evaluation. Epilepsy Behav 8:91–101
Henry TR, Engel J Jr, Mazziotta JC (1993) Clinical evaluation of interictal fluorine-18-fluorodeoxyglucose PET in partial epilepsy. J Nucl Med 34:1892–1898
Carne RP, O’Brien TJ, Kilpatrick CJ et al (2004) MRI-negative PET-positive temporal lobe epilepsy: a distinct surgically remediable syndrome. Brain 127:2276–2285
Juhasz C, Chugani DC, Muzik O et al (2001) Relationship of flumazenil and glucose PET abnormalities to neocortical epilepsy surgery outcome. Neurology 56:1650–1658
Luat AF, Makki M, Chugani HT (2007) Neuroimaging in tuberous sclerosis complex. Curr Opin Neurol 20:142–150
Chandra PS, Salamon N, Huang J, et al (2006) FDG-PET/MRI coregistration and diffusion-tensor imaging distinguish epileptogenic tubers and cortex in patients with tuberous sclerosis complex: a preliminary report. Epilepsia 47:1543–1549
Garibotto V, Heinzer S, Vulliemoz S et al (2013) Clinical applications of hybrid PET/MRI in neuroimaging. Clin Nucl Med 38:e13–e18
Garibotto V, Picard F (2013) Nuclear medicine imaging in epilepsy. Epileptologie 30:109–121
Chugani DC (2011) Alpha-methyl-L-tryptophan: mechanisms for tracer localization of epileptogenic brain regions. Biomark Med 5:567–575
Kumar A, Semah F, Chugani HT, et al (2012) Epilepsy diagnosis: positron emission tomography. Handb Clin Neurol 107:409–424
Salamon N, Kung J, Shaw SJ, et al (2008) FDG-PET/MRI coregistration improves detection of cortical dysplasia in patients with epilepsy. Neurology 71: 1594–1601
Rubi S, Setoain X, Donaire A, et al (2011) Validation of FDG-PET/MRI coregistration in nonlesional refractory childhood epilepsy. Epilepsia 52:2216–2224
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Italia
About this chapter
Cite this chapter
Garibotto, V., Vargas, M.I., Seeck, M., Picard, F. (2014). PET/CT in the Clinical Evaluation of Pediatric Epilepsy. In: Cistaro, A. (eds) Atlas of PET/CT in Pediatric Patients. Springer, Milano. https://doi.org/10.1007/978-88-470-5358-8_21
Download citation
DOI: https://doi.org/10.1007/978-88-470-5358-8_21
Published:
Publisher Name: Springer, Milano
Print ISBN: 978-88-470-5357-1
Online ISBN: 978-88-470-5358-8
eBook Packages: MedicineMedicine (R0)