Abstract
Antifungal activity of tomato leaf volatiles (TLV) against three types of plant pathogens of Botryotinia fuckeliana, Glomerella cingulata, and Fusarium oxysporum was investigated. Growths of B. fuckeliana and G. cingulata were completely inhibited by 700 and 1,000 μL of TLV extract, respectively. These suggest that TLV are proven to be efficacious as a biological control agent and that there is the defense response against plant pathogens in tomato plant. In another study, repellent and insecticidal effects of eight (peppermint, cherry sage, dokudami, sweet pepper, eucalyptus, lavender, chives, and tansy) and seven (rosemary, spearmint, eucalyptus, chives, sage, tansy, and sweet pepper) extracts were against Myzus persicae and Pieris rapae crucivora Boisduval, respectively, in laboratory and field conditions. In the laboratory, sweet pepper and tansy extracts proved to be a potent insecticide against M. persicae. In the field test, dokudami extract was a highly effective repellent against M. persicae. These findings suggest that dokudami extract can be used to control wingless green peach aphids in the field. Moreover, neem cake showed a strong repellent effect against green peach aphid. Among the seven plants, the tested herbs, rosemary and spearmint volatile extracts have a notable feeding repellent effect against P. rapae larvae.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Recently, there are growing concerns on the use of chemical pesticides and fertilizers due to environmental pollution, adverse effects on human health, disruption of natural biological control, and evolution of pesticide-resistant pest populations (Flint 2012). As consumer interest in the safety of food and agricultural products has risen in recent years, producers are increasingly focused on pesticide-free and organic farming that take human health and the environment into account. Without the use of chemicals, however, farmers risk disease and insect damage to crops (Perry et al. 1998; Isman 2006; Mulumba et al. 2012).
Many plant species are known to possess insecticidal properties, and the compounds from these plants have a number of useful activities such as toxicity, repellency, feeding, oviposition deterrence, and insect growth regulation properties (Sharma and Gupta 2009; Mann and Kaufman 2012). In mixed vegetation, odors released by some plants can mask the effect of odors released by other plants, thereby disorientating insects such that they spend less time on plants suitable for egg laying and feeding. Consequently, plant and herb extracts and neem materials are attracting increased attention as a basis for botanical insecticides (Isman 2006; Tripathi et al. 2009; Schrader et al. 2010). Botanical insecticides are an environmentally friendly alternative to hazardous chemicals as they either are plant-derived insecticides which occur naturally or are extracts of such plants (Gupta et al. 2005).
In Japan, the amount of tomato (Solanum lycopersicum L.) fruit is produced at about 760,000 t year−1, although the same amount of the leaf and stem is wasted. However, the leaf and stem may become valuable organic resources. The future challenge is to make use of them, since social structure of resources recycling is currently desired. There are food preservative and biological control agents as the usage of functional constituents in plant. In addition, it was reported that herbal aroma was different by picking season and by site and that the antibacterial activity varied greatly (Chorianopoulos et al. 2006; Kiralan et al. 2012). Thus, it can be expected that functional constituents in tomato leaf and stem are used as antibacterial agents. It is well known that there is characteristic odor in tomato leaves (Buttery et al. 1987).
The neem tree (Azadirachta indica L.) is well known in India and neighboring countries where it has been one of the most versatile medicinal plants for 2,000 years with a wide spectrum of biological activity (Alves et al. 2009; Atawodi and Atawodi 2009; Deng et al. 2012; Gangwar 2012). There are two types of harvestable neem materials. The first is neem oil which is compressed from neem seeds, concentrated, and purified. Neem oil functions as a pest repellent and is diluted with water and sprayed onto leaves. The second material is neem seed kernel oil cake, simply called neem cake, which is divided into fruitcake, seed cake, and neem seed kernel oil cake and is used primarily as an amendment and growth-promoting agent.
The neem tree synthesizes compounds for chemical defense to protect against herbivorous insects (Atawodi and Atawodi 2009). These compounds function by interfering with insect hormones (Mordue and Blackwell 1993; Anibal and Condor 2007). Nine limonoid compounds with pest control properties have been extracted from neem seeds and shown to inhibited pest growth, and the most effective of the main compounds is azadirachtin (Schmutterer 1990; Alves et al. 2009). Azadirachtin induces a physiological effect on insects by interfering with the synthesis and release of ecdysteroids which disrupts larval molting in hemimetabolous and holometabolous insects and interferes with pupation and/or eclosion of adults and with reproduction (Mordue and Blackwell 1993). Neem-based pesticides are known for their pesticidal activity against more than 400 species of insects (Siddiqui et al. 2003). However, they are not toxic to humans and many beneficial arthropods and target pests which are unlikely to develop resistance. Therefore, these pesticides have been advocated to replace synthetic pesticides as it is regarded as more sensible to use natural pesticides in most pest management programs (Schmutterer 1990; Mordue and Blackwell 1993; Isman 2006; Irigaray et al. 2010; Mulumba et al. 2012). Thus, neem oil-based pesticides are available for use against many pests and have been evaluated as an alternative to synthetic pesticides (Anibal and Condor 2007).
On the other hand, the wingless green peach aphid (Myzus persicae Sulzer, Hemiptera: Aphididae) is a major serious polyphagous pest carrying a multitude of viral diseases and infecting about 100 plant species worldwide, including those of Solanaceae, Fabaceae, and Brassicaceae (Feng and Isman 1995; Hori 1998). The aphid is difficult to eradicate as it multiplies in greenhouses and fields (Hori and Harada 1995). To date, azadirachtin and neem-based pesticides have been shown to be effective in controlling M. persicae and other aphid species (Lowery and Isman 1993; Lowery et al. 1993). However, no studies have been done on the potential of neem seed cake to repel M. persicae. In addition, there are only a few studies on the repellent and insecticidal activity of herb extracts on aphids (Hori and Harada 1995; Hori 1999a, b; Salari et al. 2012).
The cabbage white butterfly, Pieris rapae crucivora Boisduval, is a serious pest of Brassicaceae plants such as cabbage, cauliflower, and broccoli, and its larvae damage the host plant leaves when feeding (Ikeura et al. 2012). Gao et al. (2004) showed that deoxypodophyllotoxin, identified in Juniperus sabina L., has insecticidal activity against P. rapae larvae. In addition, Zhong et al. (2006) showed that rhodojaponin-III isolated from Rhododendron molle G. Don flowers is an antifeedant, stomach poison, contact toxicant, and growth inhibitor against P. rapae. However, the compounds identified in their study were nonvolatile. Only a few reports have been published on the feeding repellent effect of volatile extracts against P. rapae larvae.
In this chapter, we aimed to develop a biological pesticide alternative to chemical insecticide for future use. Therefore, we investigated as follows: I. antifungal activity of tomato leaf volatiles against Botryotinia fuckeliana, Glomerella cingulata, and Fusarium oxysporum, which were plant pathogens, II. the repellent effects of eight kinds of herb extracts from companion plants against wingless green peach aphids, III. repellent effect of neem materials against the green peach aphid and the effects of different concentrations of neem and repellent tests with the volatile extracts of neem materials, and IV. the feeding repellent effect of seven kinds of herb extracts to develop a botanical insecticide against P. rapae larvae. In this chapter, we introduce that plant volatile extracts have repellent effects and antifungal activity against insects and fungi.
2 Methodology Followed
2.1 Plants
Tomato cultivar ‘Reiyo’(S. lycopersicum L.), from Daiichi Engei Co., Ltd., Tokyo, Japan, was cultivated in a greenhouse at Meiji University from August to December 2007. The leaves from fourth to seventh flower cluster were sampled at the anthesis stage of seventh flower cluster, frozen with liquid nitrogen, crushed, and stored at −40 °C until the time of extraction of volatile compounds.
Radish ‘Comet’ (Raphanus sativus var. sativus) was purchased from Takii Seed Co. (Kyoto, Japan), seeded in black polyethylene pods, and grown for 15 days. Peppermint (Mentha piperita L.), cherry sage (Salvia microphylla L.), dokudami (Houttuynia cordata L.), sweet pepper (Capsicum annuum L.), eucalyptus (Eucalyptus globulus L.), lavender (Lavandula intermedia L.), chives (Allium schoenoprasum L.), and tansy (Tanacetum vulgare L.) were used.
Raw neem leaves were obtained from Mizusaki Farm (Fukuoka, Japan). Neem oil (‘AZ green N’; 1.2 % azadirachtin and 2.8 % neem extract) and neem seed kernel oil cake (‘Rikunomegumi’; 0.3–0.5 % azadirachtin) were purchased from OM Science (Osaka, Japan). The following seven plants, well known for agriculturists and horticulturists and common in the literature, were selected as the basis for herbal extracts: rosemary (Rosmarinus officinalis L.), spearmint (M. spicata L.), eucalyptus (E. globulus L.), chives (A. schoenoprasum L.), sage (Salvia officinalis L.), tansy (T. vulgare L.), and sweet pepper (C. annuum L.).
2.2 Pests and Diseases
Botryotinia fuckeliana NBRC 9760, G. cingulata NBRC 5257, and F. oxysporum NBRC 6385 were purchased from the National Institute of Technology and Evaluation (Kisarazu, Japan). Wingless green peach aphids virginoparae (M. persicae Sulzer) were collected around the Meiji University and were reared on radish plants at 25 °C under a L16:D8 photoperiod in an incubator until reaching 1.8–2.0 mm in length after which they were used in the experiments.
Gravid female adults of P. rapae crucivora Boisduval were collected from cabbage patches in Kawasaki City. They were released in a cage (1.6 m × 1.6 m × 1.8 m) made from white shading net in a greenhouse of Meiji University, fed an approximately 10 % sucrose solution, and free to oviposit on potted cabbages. When eggs were laid, the cabbages were placed under a natural condition in the greenhouse until hatching. The hatched larvae, which were fed cabbage in a plastic case (10 cm × 20 cm × 7 cm), were reared at 25 °C under a L16: D8 regime until being at the fourth instar.
2.3 Plant Extracts
Volatile components were extracted from tomato leaves using the Porapak Q method (PQM) as described by Hayata et al. (2003). The eluate containing the volatile compounds was dried over hydrous sodium sulfate (Kanto Chemical Co., Ltd., Tokyo, Japan) overnight at room temperature and concentrated to 6 mL under a nitrogen stream.
Fresh herb leaves (10 g) were homogenized with 40 mL of 50 % EtOH for 24 h and filtered to obtain an herb extract. The control was 25 % EtOH solution. Each herb extract was added to a spreading agent (Dain®, Sumitomo Chemical Co. Ltd., Osaka, Japan). Neem seed cake (50 g) was added to 1,000 mL of diethyl ether and mixed for 4 h. It was then filtrated, and the filtrate was concentrated to 10 mL under nitrogen gas flow and used as a bioassay sample. Ten grams of fresh leaves were homogenized and mixed with 40 mL of diethyl ether at 4 °C for 4 h with a stirrer. Next, the sample was centrifuged at 12,000 rpm at 4 °C for 20 min and filtered, and the filtrate was used as the herb extract.
2.4 Antifungal Activity: Laboratory Conditions
One hundred microliter of each plant pathogens solution (106sporemL−1) was plated on the potato dextrose agar plate (Difco, USA). Paper disks containing ~1,000 μL of extract were put on the lid of the plates turned upside down and the extract was dried. The plates were incubated at 25 °C for 7 days after sealed with parafilms. Antifungal activity of extract was shown as inhibition ratio calculated by the following equation: inhibition ratio (%) = d/D × 100, where, d is diameter of inhibition circle (mm) and D is diameter of plate (85 mm). All experiments were performed in triplicate. The data presented are the means with standard errors.
2.5 Antifungal Activity: Field Test
Radishes for the field test were seeded in 12-cm diameter pots and were harvested. The dorsal and ventral leaf surfaces of radish seedlings (four leaves) were coated with 2 mL of herb extract diluted twofold with water and control solution (25 % EtOH) once every 3 days. Aphid source plants were propagated in other plant pots. Four pots of each herb extract were placed around each radish pot. The total number of aphids on each radish plant was calculated from day 1 to day 21. Experiments for each extract were performed in triplicate.
2.6 Feeding Repellent Assay
Y-Tube Olfactometer Bioassay: An olfactometer made of a silicon tube and glass rods was used. A Y-junction mixed treatment and control airflows. Aphids moved into one of the Y-tube branches down an apparent chemotaxis gradient. Air was drawn through the apparatus at a rate of 0.2 L min−1. Wingless virginoparae were placed at the base of the central tube. Tests were carried out in a darkroom at 22–24 °C to eliminate the potential influence of light. The number of aphids in each of the Y-tube branches was counted 5 min after the start of the test. Each experiment used 30 aphids and was replicated three times. In tests with liquid samples, a 2-mL sample on filter paper (No. 5B, 90 mm; Advantec, Tokyo, Japan) was placed in the treatment flask with radish as the host plant, while filter paper without radish was placed in the control flask. Aliquots of 2-mL diethyl ether solution of each sample were applied to filter paper, and their effects on the aphids were compared against a diethyl ether control. In tests with solid samples, 1, 10, 20, and 100 g of raw leaves or neem seed cake were placed in the treatment flask with radish, while radish only was placed in the control flask. Both treatment and control filter papers were allowed to dry to remove diethyl ether before being placed in the flask.
Leaf Disk Bioassay: A feeding repellent assay was conducted on two cabbage leaf disks (3 cm × 3 cm) on filter paper (5 cm × 5 cm) treated with herb extract or diethyl ether in a box with dimensions of 20 cm × 15 cm × 8 cm. A larva was placed between the disks, and the filter paper chosen by the larva was noted. Thirty larvae were used for each treatment. The repellent rate is expressed as follows: number of larvae choosing treated paper/(number of larvae choosing treated paper + number of larvae choosing control paper) × 100.
3 Observation and Discussion
3.1 Antifungal Activity of Volatiles from Tomato
Antifungal activity against all plant pathogens tested increased linearly with the increase of TLV extract volume (Fig. 9.1). Growths of B. fuckeliana and G. cingulata were completely inhibited by 700 (equivalent to about 12 g of tomato leaves) and 1,000 μL (equivalent to about 17 g of tomato leaves) of TLV extract, respectively. On the other hand, against F. oxysporum, inhibition ratio by 1,000 μL of TLV extract was about 55 %. Accordingly, antifungal effect of TLV varies with the type of plant pathogens. It was, also, recognized that there was no antifungal activity in diethyl ether of extract solvent (data not shown). It was reported that volatiles from thyme (Thymus vulgaris L.) showed strong antifungal activities against Aspergillus parasiticus and Cryptococcus neoformans while not against Candida albicans (Martos et al. 2007). Furthermore, in the present study, TLV exhibited strong antifungal activities, even though B. fuckeliana infects easily into tomato leaves and causes illness. The reason was probably due to the concentration of the TLV.
C6-alcohols and C6-aldehydes described as green leaf volatiles (GLV) contained fresh green flavor and were reported to be rapidly produced within the plant when the plant incurred insect damages and pathogens infected to the plant (Nakamura and Hatanaka 2002). Furthermore, it is noted that GLV shows antibacterial activities against many types of microorganisms (Virginie et al. 2009). In tomato plant, GLV was synthesized due to infect damage by Helicoverpa armigera larva and released from the leaves (Rose et al. 1996). In cotton plant, GLV production increased at the upper leaves when the feeding damages to the lower leaves by Spodoptera exigua larva occurred (Mann et al. 2000). Therefore, it is presumed that there is the resistant response system due to insect damage or infection of plant pathogens in tomato plant. In addition, it was reported that essential oil showed antifungal activity at three steps of conidial germination, growth of vegetative hyphae and conidial forming (Inouye et al. 1998, 2001). However, it is not clear whether TLV acted as antifungal activity at any life cycle stage of plant pathogens.
3.2 Herb Extracts on Myzus persicae Sulzer
The number of aphids on leaves treated with all herb extracts and the control was equal until day 13 but changed after day 16 (Table 9.1). Dokudami extract prevented an increase in aphids and completely repelled them by day 21. Cherry sage extract repelled aphids from day 16. Peppermint extract increased the number of aphids at day 16, but decreased the number from day 19. Lavender extract significantly prevented an increase in aphids from day 16. These results show that dokudami extract had the highest repellency followed by sage and lavender extracts.
Myzus persicae aphids use visual cues such as plant size, shape, and color to search for host plants, with nonhost plant odor serving only as an avoidance measure (Hori 1999c). There was no significant difference between each herb extract and the control until day 13, because it was assumed that aphids entered the larval stage when they colonized the radish after moving from the source plant. From day 13, dokudami, cherry sage and lavender extracts continued to prevent an aphid increase, indicating their potential to protect against aphid damage. Peppermint extract had an immediate effect on reproductive inhibition of aphids, since although peppermint extract increased the number of aphids at day 16, it significantly decreased the number thereafter. Hori (1996) described that mint inhibited the sucking ability of aphids but did not repel them. The mint extract treatment inhibited the sucking activity of aphids causing them to move on to other host plants, likely because the host plant odor was masked by the mint extract.
The chives extract had a slow reproductive inhibiting effect since the number of aphids increased rapidly at day 16 and dramatically decreased from day 19 to day 21. Hori and Harada (1995) suggested that compounds in Allium genus plants play a role in repelling aphids. However, the reason that their data are not concomitant with ours can likely be attributed to the use of a solvent which could not extract the repellent from the chives in our study.
Of all the herbs, dokudami extract resulted in the lowest number of aphids throughout the field test period and proved to be a strong repellent against M. persicae. This result supports the finding that aphids did not settle on dokudami plants (Hori and Harada 1995). Harvey and Fortuna (2012) propose that components in essential oil have either a repellent or attractant effect on insects and that these compounds have various functions such as masking host plant odors.
3.3 Repellency Against Myzus persicae Sulzer
The average number of M. persicae for 10 and 20 g of raw leaves was 12.33 and 11.00, respectively, and the preference to 20 g of raw leaves was lower than that of the control (Fig. 9.2). This shows that raw neem leave have a slight repellent effect on M. persicae. The average number of M. persicae for neem oil diluted 100-fold, 500-fold, and 1,000-fold was 14.00, 14.67, and 14.67, respectively, indicating a nonsignificant effect (Fig. 9.3). The average number of M. persicae for 1, 10, and 100 g of neem seed cake was 15.00, 10.00, and 6.00, respectively; neem seed cake of >10 g had a significant repelling effect, which increased with treatment amount (Fig. 9.4). The average number of M. persicae for neem seed cake extract and control was 10.33 and 19.67, respectively (Fig. 9.5). Neem seed cake extract showed a repellent effect similar to that of 10 g of neem seed cake. This result revealed that neem seed cake extract contains repellent compounds.
In a previous study, Lowry et al. (1993) demonstrated that neem seed oil sprayed onto intact plants in the laboratory resulted in a significant reduction in the number of M. persicae; however, they also described that the effectiveness of neem appears to be influenced by the host plant, specific aphid species, and weather conditions. On the other hand, Hori (1996, 1999a) reported that mint, thyme, garlic, and onion oil strongly repel M. persicae and that the volatile constituents of essential oils of garlic and onion may inhibit M. persicae from settling on plants. In addition, rosemary and ginger oil repelled M. persicae in an olfactometer when linalool, camphor, and α-terpineol essential oils were used (Hori 1999b, c). Hori (1999c) suggests that polyphagous aphids such as M. persicae have a tendency to locate their host plants mainly by visual cue, avoiding the odors of some nonhost plants. Thus, the olfactory behaviors of aphids against host plant odors are related to host proximity. In the present study, aphids detected volatile compounds more in the neem seed cake than in the host plant so preferred the host plant significantly more. There is no report on the repellency of neem seed cake on M. persicae. The present study is the first to show that neem seed cake has a repellent effect on these aphids. The present findings suggest that volatile compounds in neem seed cake have a repellent effect against M. persicae.
3.4 Volatile Extracts on Pieris rapae crucivora
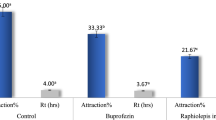
Only rosemary (75 %; chi-test, χ 2 = 14.75, p < 0.001) and spearmint (72 %; χ 2 = 11.36, p < 0.001) extracts had a significant feeding repellent effect against the larvae (Fig. 9.6). Plants have wide action spectra, whereas a particular plant can affect insects though different mechanisms of action. In this study, rosemary and spearmint affected P. rapae larvae by inhibiting their feeding, suggesting the presence of feeding repellent compounds in the volatile extracts. To date, there have been many studies on repellency and the insecticidal effects of herbs against pests (Dover 1985; Sharma and Gupta 2009; Salari et al. 2012). Rosemary is known to have a repellent effect on various pests (Salari et al. 2012), and Sampson et al. (2005) reported that rosemary oil repelled Lipaphis pseudobrassicae. Waliwitiya et al.(2009) assessed the essential oil of rosemary against larval stages of Aedes aegypti and found that the larvicidal activity of 15 terpenoids, pulegone, thymol, eugenol, trans-anethole, and citronellal exhibited high larvicidal activity against A. aegypti. Dover (1985) reported that the alcohol extract of rosemary has a repellent effect on P. brassicae. Moreover, mint oil is known to repel various pests and insects (Sampson et al. 2005; Abbaszadeh et al. 2009). However, there is no data on the antifeeding effects of rosemary on P. rapae larvae; nevertheless, rosemary and spearmint are known to repel various pests.
Pieris brassicae belongs to the same family as P. rapae and avoids feeding on leaves treated with hyssop, rosemary, sage, thyme, white clover, and eucalyptus (Sharma and Gupta 2009). On the other hand, P. rapae larvae avoid feeding on the wild mustard, Erysimum cheiranthoides (Sachdev-Gupta et al. 1993). Thus, Pieris spp. reject various plants, and these repellents are soluble. The findings of the present study suggest that compounds from rosemary may be volatile due to extract high volatile organic solvent. Furthermore, it seems desirable to use rosemary for companion plants.
4 Conclusions
In this chapter, we investigated that plant volatile extracts have repellent effects and antifungal activity against insects and fungi. First, it was clarified that the TLV showed strong antifungal activities against three types of plant pathogens of B. fuckeliana, G. cingulata, and F. oxysporum. Furthermore, it was presumed that there was the defense response against plant pathogens by TLV. Second, sweet pepper and tansy extracts proved to be a potent insecticide against aphids in the laboratory, and dokudami extract was a highly effective repellent against aphids in the field. Third, neem seed cake had the highest repellent effect on M. persicae, and effectiveness increased in line with increasing concentration. Finally, rosemary and spearmint volatile extracts have a notable feeding repellent effect against P. rapae larvae. Thus, we consider the repellent effect on insects and antifungal activity on fungi to be attributed to plant volatile compounds In the future, we need to identify these repellent and antifungal compounds.
References
Abbaszadeh B, Valadabadi SA, Farahani A, Darvishi HH (2009) Studying of essential oil variations in leaves of Mentha species. African J Plant Sci 3:217–221
Alves PD, Brandӑo MGL, Nunan EA, Vianna-Soares CD (2009) Chromatographic evaluation and antimicrobial activity of neem (Azadirachta indica A. Juss., Meliaceae) leaves hydroalcoholic extracts. Revista Brasileira de Farmacognosia/Brazilian J Pharma 19:510–515
Anibal F, Condor G (2007) Effect of neem (Azadirachta indica A. Juss) insecticides on parasitoids. Revista Peruana de Biología 14:69–74
Atawodi SE, Atawodi JC (2009) Azadirachta indica (neem): a plant of multiple biological and pharmacological activities. Phytochem Rev 8:601–620
Buttery RG, Ling LC, Light DM (1987) Tomato leaf volatile aroma components. J Agric Food Chem 35:1039–1042
Chorianopoulos N, Evergetis E, Mallouchos A, Kalpoutzakis E, Nychas GJ, Haroutounian SA (2006) Characterization of the essential oil volatiles of Satureja thymbra and Satureja parnassica: influence of harvesting time and antimicrobial activity. J Agric Food Chem 54:3139–3145
Deng Y, Shi D, Yin Z, Guo J, Jia R, Xu J, Song X, Lv C, Fan Q, Liang X, Shi F, Ye G, Zhang W (2012) Acaricidal activity of petroleum ether extract of neem (Azadirachta indica) oil and its four fractions separated by column chromatography against Sarcoptes scabiei var. cuniculi larvae in vitro. Exp Parasitol 130:475–477
Dover JW (1985) The responses of some Lepidoptera to labiate herb and white extracts. Entomol Exp Appl 39:177–182
Feng R, Isman MB (1995) Selection for resistance to azadirachtin in the green peach aphid, Myzus persicae. Experientia 51:831–833
Flint ML (2012) IPM in practice: principles and methods of integrated pest management. University of California Agriculture and Natural Resources, California
Gangwar SK (2012) Experimental study to find the effect of different neem (Azadirachta indica) based products against moringa hairy caterpillar (Eupterote mollifera Walker.). Bull Envir Pharma Life Sci 1:35–38
Gao R, Gao C, Tian X, Yu X, Di X, Xiao H, Zhang X (2004) Insecticidal activity of deoxypodophyllotoxin, isolated from Juniperus sabina L., and related lignans against larvae of Pieris rapae L. Pest Manag Sci 60:1131–1136
Gupta S, Sharma AK, Sirohi A (2005) Neem: a botanical pesticides. Indian Farmers Digest 32:35–36
Harvey JA, Fortuna TM (2012) Chemical and structural effects of invasive plants on herbivore–parasitoid/predator interactions in native communities. Entomol Exp Appl 144:14–26
Hayata Y, Sakamoto T, Chamorn M, Kozuka H, Sakamoto K (2003) Application of the porapak Q column extraction method for tomato flavor volatile analysis. J Agric Food Chem 50:3401–3404
Hori M (1996) Settling inhibition and insecticidal activity of garlic and onion oils against Myzus persicae (Sluzer) (Homoptera: Aphididae). Appl Entomol Zoo 31:605–612
Hori M (1998) Repellency of rosemary oil against Myzus persicae in a laboratory and in a screenhouse. J Chem Eco 24:1425–1432
Hori M (1999a) Antifeeding, settling plants resistant to green peach aphid, Myzus persicae (Sulzer) (Homoptera: Aphididae). Appl Entomol Zoo 34:113–118
Hori M (1999b) The effects of rosemary and ginger oils on the alighting behavior of Myzus persicae (Sulzer) (Homoptera: Aphididae) and on the incidence of yellow spotted streak. Appl Entomol Zoo 34:351–358
Hori M (1999c) Role of host plant odors in the host findings behaviors of aphids. Appl Entomol Zoo 34:293–298
Hori M, Harada H (1995) Screening plants resistant to green peach aphid, Myzus persicae (Sulzer) (Homoptera: Aphididae). Appl Entomol Zoo 30:246–249
Ikeura H, Kobayashi F, Hayata Y (2012) Optimum extraction method for volatile attractant compounds in cabbage to Pieris rapae. Biochem Syst Ecol 40:201–207
Inouye S, Watanabe M, Nishiyama Y, Takeo K, Akao M, Yamaguchi H (1998) Antisporulating and respiratory-inhibitory effects of essential oils on filamentous fungi. Mycoses 41:403–410
Inouye S, Uchida K, Yamaguchi H (2001) In-vitro and in-vivo anti-Trichophyton activity of essential oils by vapour contact. Mycoses 44:99–107
Irigaray F, Javier SDC, Fernando MG, Vicente M, Ignacio PM (2010) Acute and reproductive effects of Align®, an insecticide containing azadirachtin, on the grape berry moth, Lobesia botrana. J Insect Sci 10:1–11
Isman MB (2006) Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu Rev Entomol 51:45–66
Kiralan M, Bayrak A, Abdulaziz OF, Özbucak T (2012) Essential oil composition and antiradical activity of the oil of Iraq plants. Nat Prod Res (Formerly Natural Product Letters) 26:132–139
Lowery DT, Isman M (1993) Antifeedant activity of extracts from neem, Azadirachta indica, to strawberry aphids, Chaetosiphon fragaefolii. J Chem Ecol 19:1761–1773
Lowery DT, Isman MB, Brard NL (1993) Laboratory and field evaluation of neem for the control of aphid (Homoptera: Aphididae). J Econ Entomol 86:864–870
Mann SR, Kaufman EP (2012) Natural product pesticides: their development, delivery and use against insect vectors. Mini-Rev Org Chem 9:185–202
Mann CM, Cox SD, Markham JL (2000) The outer membrane of Pseudomonas aeruginosa NCTC 6749 contributes to its tolerance to the essential oil of Melaleuca alternifolia (tea tree oil). Lett Appl Microbiol 30:294–297
Martos MV, Navajas YR, Lopez F, Perez-Alvarez JA (2007) Antifungal activities of thyme, clove and oregano essential oils. J Food Safety 27:91–101
Mordue AJ, Blackwell A (1993) Azadirachtin: an update. J Insect Physiol 39:903–924
Mulumba JW, Nankya R, Adokorach J, Kiwuka C, Fadda C, De Santis P, Jarvis DI (2012) A risk-minimizing argument for traditional crop varietal diversity use to reduce pest and disease damage in agricultural ecosystems of Uganda. Agr Ecosyst Environ 57:70–86
Nakamura S, Hatanaka A (2002) Green-leaf-derived C6-aroma compounds with potent antibacterial action that act both on gram-negative and gram-positive bacteria. J Agric Food Chem 50:7639–7644
Perry AS, Yamamoto I, Ishaaya I, Perry RY (1998) Insecticides in agriculture and environment: retrospects and prospects. Springer, Berlin
Rose U, Manukian A, Heath RR, Tumlinson JH (1996) Volatile semiochemicals released from undamaged cotton leaves-a systemic response of living plants to caterpillar damage. Plant Physiol 111:487–495
Sachdev-Gupta K, Radke CD, Renwick JAA, Dimock MB (1993) Cardenolides from Erysimum cheiranthoides: feeding deterrents to Pieris rapae larvae. J Chem Ecol 19:1355–1369
Salari E, Ahmadi K, Zamani RD, Purhematy A, Takalloozadeh HM (2012) Toxic and repellent effect of harmal (Peganum harmala L.) Acetonic extract on several aphids and Tribolium castaneum (HERBST). Chilean J Agri Res 72:147–151
Sampson BJ, Tabanca N, Kirimer N, Demirci B, Baser KHC, Khan IA, Spiers JM, Wedge DE (2005) Insecticidal activity of 23 essential oils and their major compounds against adult Lipaphis pseudobrassicae (Davis) (Aphididae: Homoptera). Pest Manag Sci 61:1122–1128
Schmutterer H (1990) Properties and potential of natural pesticides from the neem tree, Azadirachta indica. Ann Rev Entmol 35:271–297
Schrader KK, Andolfi A, Cantrell CL, Cimmino A, Duke SO, Osbrink W, Wedge DE, Evidente A (2010) A survey of phytotoxic microbial and plant metabolites as potential natural products for pest management. Chem Biodivers 7:2261–2280
Sharma A, Gupta R (2009) Biological activity of some plant extracts against Pieris brassicae (Linn.). J Biopesticide 2:26–31
Siddiqui BS, Afshan F, Gulzar T, Sultana R, Naqvi SN, Tariq RM (2003) Tetracyclic triterpenoids from the leaves of Azadirachta indica and their insecticidal activities. Chem Pharma Bull 51:415–417
Tripathi AK, Upadhyay S, Bhuiyan M, Bhattacharya RR (2009) A review on prospects of essential oils as biopesticide in insect-pest management. J Physiol Pathophysio 1:52–63
Virginie G, Nicolas H, Frederic F, Eric H, Paul WJ, Patrick DJ, Ivo F, Laure FM (2009) Attacks by a piercing-sucking insect (Myzus persicae Sulzer) or a chewing insect (Leptinotarsa decemlineata Say) on tomato plants (Solanum lycopersicum L.) induce differential changes in volatile compound release and oxylipin synthesis. J Exp Bot 60:1231–1240
Waliwitiya R, Kennedy CJ, Lowenberger CA (2009) Larvicidal and oviposition-altering activity of monoterpenoids, trans-anethole and rosemary oil to the yellow fever mosquito Aedes aegypti (Diptera: Culicidae). Pest Manag Sci 65:241–248
Zhong G, Liu J, Weng Q, Hu M, Luo J (2006) Laboratory and field evaluations of rhodojaponin-III against the imported cabbage worm Pieris rapae (L.) (Lepidoptera: Pieridae). Pest Manag Sci 62:976–981
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer India
About this chapter
Cite this chapter
Ikeura, H. (2014). Use of Plant Volatile for Plant Pathogens and Pests Management. In: Sahayaraj, K. (eds) Basic and Applied Aspects of Biopesticides. Springer, New Delhi. https://doi.org/10.1007/978-81-322-1877-7_9
Download citation
DOI: https://doi.org/10.1007/978-81-322-1877-7_9
Published:
Publisher Name: Springer, New Delhi
Print ISBN: 978-81-322-1876-0
Online ISBN: 978-81-322-1877-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)