Abstract
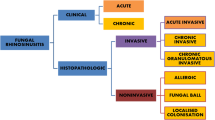
Several cases can mimic invasive fungal rhinosinusitis, either clinically or radiologically or both. This causes confusion and controversy among clinicians with misdirection of treatment. A few such cases are discussed here.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Several cases can mimic invasive fungal rhinosinusitis, either clinically or radiologically or both. This causes confusion and controversy among clinicians with misdirection of treatment. A few such cases are discussed here.
Case 1
A 26-year-old immunocompetent lady came with complaints of recurrent rhinitis, headaches, and right diplopia since 4 days. On nasal endoscopy, there was grossly deviated septum to the right with multiple polyps (arrow) in the left nasal cavity (Fig. 1). CT scan of paranasal sinuses was obtained (Figs. 2 and 3).
The patient underwent endoscopic nasal clearance of the sinuses. Intraoperatively, there was inspissated pus and debris (arrow) (Fig. 4) in both the sphenoid sinuses, but the lining mucosa of the sinuses appeared normal.
Histopathology showed typical features of allergic fungal sinusitis (Fig. 4).
The patient was started on oral and topical nasal steroids with nasal douching following surgery. Three months postoperatively, the patient’s diplopia recovered and the appearance of the sphenoid sinus is shown in Fig. 5.
Discussion: Allergic fungal sinusitis (AFS) is a noninvasive form of fungal rhinosinusitis. It contributes to approximately 6–9 % of all rhinosinusitis requiring surgery [1]. Patients of AFS usually present with chronic rhinosinusitis with nasal polyps, atopy, and elevated total serum IgE, and their sinuses are filled with characteristic “peanut butter”-like eosinophil-rich “allergic mucin” containing sparse fungal hyphae. CT scan of the sinuses (Figs. 2 and 3) is always abnormal, usually showing bilateral and multiple sinus involvement with near total opacification and expansion of the sinuses. Expansion, remodeling, or thinning/erosion of the adjacent bony sinus wall is frequently seen, due to the expansile nature of the accumulating mucin. Bony erosions and extension into adjacent vital cavities may be seen in about 20 % cases [1]. AFS allergic mucin, on culture, is typically positive for either dematiaceous fungi like Bipolaris spicifera or Curvularia lunata or Aspergillus species like A. fumigatus, A. flavus, or A. niger [2]. Cases of allergic fungal sinusitis with loss of bony architecture on imaging studies may be misinterpreted as evidence of bony invasion [3] and labeled as invasive fungal sinusitis. But histopathology showing absence of tissue invasion should enable the clinician to diagnose AFS and differentiate it from other forms of both noninvasive and invasive fungal sinusitis. The patient of AFS can be managed with surgical debridement, systemic and local steroids, and nasal douching without systemic antifungals.
Patients with allergic fungal sinusitis showing tissue invasion on histopathology are being encountered and are being termed as “mixed” infection. Such a progression may be due to change in host defenses [4]. These cases may require systemic antifungals, in addition to surgical debridement, nasal douching, and regular follow-up with nasal endoscopy and radiological imaging. Reports suggest oral itraconazole therapy, especially in recalcitrant cases, may avoid or delay revision surgery [5, 6] and even enable patients to reduce or even stop oral steroid therapy [6].
Case 2
A 55-year-old male patient came with history of foul smelling breath with left purulent rhinorrhea. On examination, his left nasal cavity showed purulent discharge from the left middle meatus region (Fig. 6). A CT scan of the paranasal sinuses was done (Fig. 7).
The patient underwent endoscopic debridement of the left maxillary sinus. Intra-op endoscopic appearance of the fungal ball is shown in Fig. 8.
Histopathological appearance is shown in Fig. 9.
Subsequently he was started on saline nasal douches and completely recovered.
Discussion: A fungal ball or mycetoma or aspergilloma or chronic noninvasive granuloma is described as the presence of noninvasive accumulation of dense conglomeration of fungal hyphae in one sinus cavity. It usually affects a single sinus, commonly the maxillary sinus and rarely the sphenoid sinus [7]. It is more common in middle-aged and elderly females, in contrast to invasive sinusitis and aspergillosis which are common in males [8]. The disease is identified due to its characteristic radiologic appearance (Fig. 7) – single sinus opacification with or without radiographic heterogeneity; mucopurulent, cheesy, or clay-like material within the sinus, a dense conglomeration of hyphae separate from the sinus mucosa; nonspecific chronic inflammation of the mucosa; lack of predominance of eosinophils or granuloma or allergic mucin; and lack of evidence of mucosal invasion on histopathology [9]. The fungal ball appears like a hyperattenuating mass on non-contrast CT scan due to dense matted hyphae. Occasionally there may be punctate calcifications. The bony walls of the sinus may be sclerotic and thickened or expanded and thinned with focal areas of erosion due to pressure necrosis. On MRI, the fungal ball appears hypointense on T1- and T2-weighted images due to the absence of free water. Signal void is also generated on T2-weighted images due to calcifications and paramagnetic metals such as iron, magnesium, and manganese within the mycetoma.
Such cases could be clinically and radiologically mistaken as invasive fungal sinusitis. But histopathology is diagnostic (Fig. 9) as it does not show tissue invasion. The fungi remain noninvasive in the context of the fungal ball, but could rarely become invasive after substantial immunosuppression, such as in renal transplantation [10]. Identification of Aspergillus species as the causative agent may be aided by the use of galactomannan detection in the sinus material [11].
Surgical debridement, as in this patient, is the only treatment required.
Case 3
A 59-year-old lady came with complaints of severe pain along the right cheek with diplopia and photophobia. She had history of recurrent rhinitis and had undergone a CT scan of the paranasal sinuses elsewhere. It had shown (Figs. 10 and 11) right maxillary sinusitis with hyperdense foci with sclerosis and erosion of the maxillary sinus and inferior orbital wall with suspicion of fungal sinusitis and local invasion. Based on the scan, she had undergone nasal endoscopy elsewhere, and the tissue from the right maxillary sinus had shown Aspergillus species on KOH mount. So she had been started on oral voriconazole. A few weeks after voriconazole, due to increasing severity of pain, she reported to us for a second opinion. On examination, nasal endoscopy showed only scarring from previous surgery.
Case 3: Axial CT contrast-enhanced scan shows the same lesion in the right maxillary sinus as in Fig. 10. Erosion is seen along the anterior wall of the right maxillary sinus (arrows)
Since no histopathological confirmation of the diagnosis of invasive fungal sinusitis was available from the first surgery, it was decided to obtain tissue for histopathology. MRI scan of the orbit and paranasal sinuses done at this time showed (Fig. 12) decrease in the mucosal thickening and enhancement within the right maxillary sinus and along the floor of the right orbit as compared to the previous CT scan. The ill-defined soft tissue and enhancement within the retro-maxillary fat extending into the infratemporal and pterygopalatine fossa were unchanged.
An endoscopic biopsy from the right medial maxillary wall as well as a subconjunctival biopsy from the infraorbital region was performed. On histopathology, the lesion showed moderately dense fibrosclerotic tissue with nodular and diffuse lymphoplasmacytic infiltrate. The lesion was seen eroding the bone, entrapping the vessels, nerves, and mucous glands. The entrapped glands were atrophic. There were no granulomas, no fungal infection, and no lymphoproliferative disorder. The lesion was diagnosed as inflammatory pseudotumor (Fig. 13) on histopathology. The patient was started on oral steroids and voriconazole was discontinued. Microbiology of the tissue was negative for fungus and bacteria. Voriconazole was discontinued, and patient was started on 40 mg oral steroids in divided doses. The patient was relieved of pain within a week of starting steroids. She has been symptom free for the past 5 months.
Discussion: Inflammatory pseudotumor (IPT) is a chronic, inflammatory, nonmalignant lesion of unknown origin [12]. It is a space-occupying lesion produced by chronic inflammation and tissue fibrosis and is steroid responsive [13]. IPT in the head and neck region can be locally aggressive, with bony erosion and severe neuralgia like infraorbital neuralgia in this patient. Diagnosis is difficult as clinically and radiologically IPT is indistinguishable from invasive neoplasms [14–16] and invasive fungal lesions [17]. In this patient, it is debatable whether the initial treatment with oral voriconazole made it difficult to isolate Aspergillus spp. during the subsequent tissue sampling or whether the patient was a case of pseudotumor primarily who had been misdiagnosed as invasive Aspergillus sinusitis.
Case 4
A 45-year-old diabetic male came with an ulcer along the right nasolabial crease (Fig. 14) and a palatal ulcer (Fig. 15) since 3 months. It had been progressively increasing in size. Biopsy elsewhere had been inconclusive. Clinically it could be mistaken for invasive fungal sinusitis.
The biopsy was repeated, taking tissue from the palate. Histopathology revealed NK/T-cell lymphoma, and the patient was referred for chemotherapy – radiation.
Discussion: Clinically, the lesion could be mistaken for invasive fungal sinusitis, but histopathology is diagnostic and immunohistochemistry enables further classification of the tumor. In most cases EBV genomes are detectable in the tumor cells, and immunophenotyping may show CD56 positivity. NK/T-cell lymphoma is a rare, aggressive subtype of lymphoma associated with extensive necrosis and angioinvasion. It presents in extra-nodal regions, especially, the nasal or paranasal sinus region. Historically, this tumor was considered part of lethal midline granuloma. It may be associated with hemophagocytic syndrome and has a highly aggressive course with poor prognosis. In view of the poor response of the tumor to standard chemotherapy and radiation, some oncologists recommend bone marrow or peripheral stem cell transplantation.
Case 5
Mr M aged 55 complained of broadening of nose, nasal block, and occasional epistaxis since 5–6 months. On examination, the nose appeared broad (Fig. 16) (arrow) with thickening of the skin over the nasal dorsum. Anterior rhinoscopy showed hypertrophied turbinates, severe nasal mucosal congestion with no visible airway. Subcutaneous biopsy was obtained from the external nasal swelling. The biopsied tissue was sent for potassium hydroxide preparation and fungal culture. Broad thin-walled nonseptate mycelia were found in the KOH preparation. In the Sabouraud’s dextrose agar medium are rapidly growing flat cream-colored colonies with conidia indicative of subcutaneous dermatomycoses or Conidiobolus coronatus. Patient was started on oral potassium iodide drops in tapering doses along with tab. fluconazole 200 mg daily. During treatment, the patient’s thyroid function tests, SGPT, and serum potassium were monitored. The treatment was continued over 6 months, and the patient responded well with reduction in external nasal swelling. The nasal block disappeared, and the patient was able to breathe comfortably.
Discussion: Patients with rhinofacial swelling and unilateral nasal obstruction presenting to the ENT surgeon can present a diagnostic dilemma. Rhinoentomophthoromycosis (conidiobolomycosis) is a rare, chronic, localized, subcutaneous zygomycosis characterized by painless, woody swelling of the rhinofacial region [18]. Usually the lesion is firmly attached to the skin without involvement of the underlying bone. Therefore, tissue for testing has to be obtained from the affected subcutaneous tissue. It is a slowly progressive, but rarely life-threatening condition. It is caused by Conidiobolus coronatus (Entomophthora coronata), a fungus belonging to the order
Entomophthorales. KOH preparation of the biopsy tissue from the lesion reveals broad, nonseptate, thin-walled mycelial filaments. In Sabouraud’s dextrose agar (SDA) medium, colonies of Conidiobolus coronatus grow rapidly. Histopathology shows fibroblastic proliferation, chronic granulomatous inflammatory reaction, and broad thin-walled hyphae [19].
Treatment of rhinoentomophthoromycosis is difficult because the diagnosis is delayed. Patients often respond to oral itraconazole (200–400 mg/day), ketoconazole (200–400 mg/day), fluconazole (100–200 mg/day), amphotericin B, and co-trimoxazole [20]. Prolonged treatment and follow-up is required.
Saturated potassium iodide solution (1 g/ml) is useful for patients because of its ease of administration and low cost.
References
Case 1
Schubert MS. Allergic fungal sinusitis: pathophysiology, diagnosis and management. Med Mycol. 2009;47(Suppl I):S324–30.
Stammberger H, Jaske R, Beaufort F. Aspergillosis of the paranasal sinus x-ray diagnosis, histopathology and clinical aspects. Ann Otol Rhinol Laryngol. 1984;93:251–6.
de Shazo RD, O’Brien M, Chapin K, Soto-Aguilar M, Gardner L, Swain R. A new classification and diagnostic criteria for invasive fungal sinusitis. Arch Otolaryngol Head Neck Surg. 1997;123:1181–8.
Gungor A, Adusumilli V, Corey JP. Fungal sinusitis progression of disease in immunosuppression – a case report. Ear Nose Throat J. 1998;77:207–15.
Seiberling K, Wormald PJ. The role of itraconazole in recalcitrant fungal sinusitis. Am J Rhinol Allergy. 2009;23(3):303–6. doi:10.2500/ajra.2009.23.3315.
Rains 3rd BM, Mineck CW. Treatment of allergic fungal sinusitis with high-dose itraconazole. Am J Rhinol. 2003;17(1):1–8.
Case 2
Grosjean P, Weber R. Fungus balls of the paranasal sinuses: a review. Eur Arch Otorhinolaryngol. 2007;264:461–70.
Dufour X, Kauffmann-Lacroix C, Ferrie JC, Goujon JM, Rodier MH, Klossek JM. Paranasal sinus fungal ball epidemiology, clinical features and diagnosis. A retrospective analysis of 173 cases from a single center in France: 1989–2002. Med Mycol. 2006;44:61–7.
de Shazo RD, O’Brien M, Chapin K, Soto-Aguilar M, Gardner L, Swain R. A new classification and diagnostic criteria for invasive fungal sinusitis. Arch Otolaryngol Head Neck Surg. 1997;123:1181–8.
Gungor A, Adusumilli V, Corey JP. Fungal sinusitis progression of disease in immunosuppression – a case report. Ear Nose Throat J. 1998;77:207–15.
Chakrabarti A, Denning DW, Ferguson BJ, et al. Fungal rhinosinusitis: a categorization and definitional schema addressing current controversies. Laryngoscope. 2009;119:1809–18.
Case 3
Som PM, Brandwein MS, Maldjian C, Reino AJ, Lawson W. Inflammatory pseudotumour of the maxillary sinus: CT and MR findings in six cases. AJR Am J Roentgenol. 1994;163(3):689–92.
Batsakis JG, Luna MA, el-Naggar AK, Goepfert H. Pathology consultation: inflammatory pseudotumour” – what is it? How does it behave? Ann Otol Rhinol Laryngol. 1995;104:329–31.
Han MH, Chi JG, Kim MS, Chang KH, Kim KH, Yeon KM, et al. Fibrosing inflammatory pseudotumours involving the skull base: MR and CT manifestations with histopathologic comparison. AJNR Am J Neuroradiol. 1996;17:515–21.
McKinney AM, Short J, Lucato L, Santacruz K, McKinney Z, Kim Y. Inflammatory myofibroblastic tumour of the orbit with associated enhancement of the meninges and multiple cranial nerves. AJNR Am J Neuroradiol. 2006;27:2217–20.
Chen JM, Moll C, Schotton JC, Fisch U. Inflammatory pseudotumours of the skull base. Skull Base Surg. 1994;4:93–8.
de Shazo RD, O’Brien M, Chapin K, Soto-Aguilar M, Gardner L, Swain R. A new classification and diagnostic criteria for invasive fungal sinusitis. Arch Otolaryngol Head Neck Surg. 1997;123:1181–8.
Case 5
Richardson MD, Warnock DW. Entomophromycosis. In: Richardson MD, Warnock DW, editors. Fungal infection. Diagnosis and management. 3rd ed. Chichester: Blackwell Publishing; 2003. p. 293–7.
Richardson MD, Pirkko KK, Gillian SS, et al. Rhizopus. Rhizomucor, absidia and other agents of systemic and subcutaneous zygomycosis. In: Patrick RM, Ellen JB, editors. Manual of clinical microbiology, vol. 2. 8th ed. Washington, DC: ASM Press; 2003. p. 1761–80.
Restrepo A. Treatment of tropical mycoses. J Am Acad Dermatol. 1994;31(3 Pt 2):S91–102.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer India
About this chapter
Cite this chapter
Mankekar, G. (2014). Case Studies. In: Mankekar, G. (eds) Invasive Fungal Rhinosinusitis. Springer, New Delhi. https://doi.org/10.1007/978-81-322-1530-1_10
Download citation
DOI: https://doi.org/10.1007/978-81-322-1530-1_10
Published:
Publisher Name: Springer, New Delhi
Print ISBN: 978-81-322-1529-5
Online ISBN: 978-81-322-1530-1
eBook Packages: MedicineMedicine (R0)