Summary
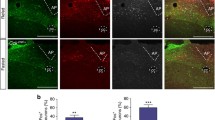
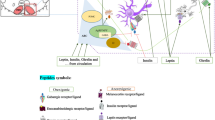
Orphan G protein-coupled receptors (GPCRs) as targets to identify new transmitters have led over the last decade to the discovery of at least twelve novel neuropeptide families. Interestingly, several of these novel neuropeptides have physiological effects that include the modulation of food intake and energy expenditure. Neuropeptide W (NPW) is a novel neuropeptide which was recently isolated from the porcine hypothalamus and shown to be an endogenous ligand for the GPR7 and GPR8 orphan G protein-coupled receptors. NPW is widely distributed in the brain. Infusion of NPW is known to increase food intake in the light phase but inhibit intake in the dark phase. In spite of numerous morphological studies on NPW, its function has yet to be fully elucidated. Moreover, as the distribution of NPW-positive cell bodies in the hypothalamus has not been described, a detailed examination of NPW’s distribution and localization in this brain region is required. The expression of NPW mRNA was demonstrated in the hypothalamic paraventricular nucleus (PVN), arcuate nucleus (ARC), ventromedial nucleus (VMH) and lateral hypothalamus (LH). NPW-like immunoreactivity (LI) was dramatically enhanced in animals pretreated with colchicine. At the light microscopic level, NPW-LI cell bodies have been identified in the preoptic areas (POA), PVN, ARC, VMH, LH, periaqueductal gray (PAG), lateral parabrachial nucleus (LPB) and prepositus nucleus. NPW-LI axon terminals have also been observed in the POA, bed nucleus of the stria terminals, amygdala, PVN, ARC, VMH, LH by electron microscopy. In addition, at the electron microscopic level, NPW-LI cell bodies and dendritic processes were often observed receiving inputs from other unknown neurons in the ARC, PVN, VMH and amygdala. Moreover, double immunostaining experiments showed that NPW-LI axon terminals were in close apposition to orexin-, MCH-, and NPY-containing neurons in the hypothalamus. These morphological and physiological findings strongly suggest that NPW participates in the regulation of feeding behavior in harmony with other feeding-regulating neurons in the hypothalamus.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Bockaert J, Pin JP (1999) Molecular tinkering of G protein-coupled receptors: an evolutionary success. EMBO J 18:1723–1729
Brezillon S, Lannoy V, Franssen JD, Le Poul E, Dupriez V, Lucchetti J, Detheux M, Parmentier M (2003) Identification of natural ligands for the orphan G protein-coupled receptors GPR7 and GPR8. J Biol Chem 278:776–783
Dun, SL, Brailoiu GC, Yang J, Chang JK, Dun NJ (2003) Neuropeptide W-immunoreactivity in the hypothalamus and pituitary of the rat. Neurosci Lett 349:71–74
Fujii R, Yoshida H, Fukusumi S, Habata Y, Hosoya M, Kawamata Y, Yano T, Hinuma S, Kitada C, Asami T, Mori M, Fujisawa Y, Fujino M (2002) Identification of a neuropeptide modified with bromine as an endogenous ligand for GPR7. J Biol Chem 277:34010–34016
Hondo M, Ishii M, Sakurai T (2008) The NPB/NPW neuropeptide system and its role in regulating energy homeostasis, pain, and emotion. Results Probl Cell Differ 46:239–256
Howard AD, McAllister G, Feighner SD, Liu Q, Nargund RP, Van der Ploeg LH, Patchett, AA (2001) Orphan G-protein-coupled receptors and natural ligand discovery. Trends Pharmacol Sci 22: 132–140
Ishii M, Fei H, Friedman JM (2003) Targeted disruption of GPR7, the endogenous receptor for neuropeptides B and W, leads to metabolic defects and adult-onset obesity. Proc Natl Acad Sci U S A 100:10540–10545
Jackson VR, Lin SH, Wang Z, Nothacker HP, Civelli O (2006) A study of the rat neuropeptide B/neuropeptide W system using in situ techniques. J Comp Neurol 497:367–383
Kawauchi H, Kawazoe I, Tsubokawa M, Kishida M, Baker BI (1983) Characterization of melanin-concentrating hormone in chum salmon pituitaries. Nature 305:321–323
Kitamura Y, Tanaka H, Motoike T, Ishii M, Williams SC, Yanagisawa M, Sakurai T (2006) Distribution of neuropeptide W immunoreactivity and mRNA in adult rat brain. Brain Res 1093: 123–134
Lee DK, Nguyen T, Porter CA, Cheng R, George SR, O’Dowd BF (1999) Two related G protein-coupled receptors: the distribution of GPR7 in rat brain and the absence of GPR8 in rodents. Brain Res Mol Brain Res 71:96–103
Levine AS, Winsky-Sommerer R, Huitron-Resendiz S, Grace MK, de Lecea L (2005) Injection of neuropeptide W into paraventricular nucleus of hypothalamus increases food intake. Am J Physiol Regul Integr Comp Physiol 288:1727–1732
Mondai MS, Yamaguchi H, Date Y, Shimbara T, Toshinai K, Shimomura Y,Mori M, Nakazato M (2003) A role for neuropeptide W in the regulation of feeding behavior. Endocrinology 144:4729–4733
Mondai MS, Yamaguchi H, Date Y, Toshinai K, Kawagoe T, Tsuruta T, Kageyama H, Kawamura Y, Shioda S, Shimomura Y, Mori M, Nakazato M (2006) Neuropeptide W is present in antral G cells of rat, mouse, and human stomach. J Endocrinol 188:49–57
O’Dowd BF, Scheideler MA, Nguyen T, Cheng R, Rasmussen JS, Marchese A, Zastawny R, Heng HH, Tsui LC, Shi X, et al (1995) The cloning and chromosomal mapping of two novel human opioid-somatostatin-like receptor genes, GPR7 and GPR8, expressed in discrete areas of the brain. Genomics 28:84–91
Rucinski M, Nowak KW, Chmielewska J, Ziolkowska A, Malendowicz LK (2007) Neuropeptide W exerts a potent suppressive effect on blood leptin and insulin concentrations in the rat. Int J Mol Med 19:401–405
Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, Williams SC, Richardson JA, Kozlowski GP, Wilson S, Arch, JR, Buckingham, RE, Haynes AC, Carr SA, Annan RS, McNulty DE, Liu WS, Terrett JA, Elshourbagy NA, Bergsma DJ, Yanagisawa M (1998) Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92:573–585
Shimomura Y, Harada M, Goto M, Sugo T, Matsumoto Y, Abe M, Watanabe T, Asami T, Kitada C, Mori M, Onda H, Fujino M (2002) Identification of neuropeptide W as the endogenous ligand for orphan G-protein-coupled receptors GPR7 and GPR8. J Biol Chem 277:35826–35832
Singh G, Maguire JJ, Kuc RE, Fidock M, Davenport AP (2004) Identification and cellular localisation of NPW1 (GPR7) receptors for the novel neuropeptide W-23 by [125I]-NPW radioligand binding and immunocytochemistry. Brain Res 1017:222–226
Takenoya F, Kitamura S, Kageyama H, Nonaka N, Seki M, Itabashi K, Date Y, Nakazato M, Shioda S (2008) Neuronal interactions between neuropeptide W-and orexin-or melanin-concentrating hormone-containing neurons in the rat hypothalamus. Regul Pept 145:159–164
Tanaka H, Yoshida T, Miyamoto N, Motoike T, Kurosu H, Shibata K, Yamanaka A, Williams SC, Richardson JA, Tsujino N, Garry MG.,Lerner MR, King DS, O’Dowd BF, Sakurai T, Yanagisawa M, (2003) Characterization of a family of endogenous neuropeptide ligands for the G protein-coupled receptors GPR7 and GPR8. Proc Natl Acad Sci U S A 100:6251–6256
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer
About this paper
Cite this paper
Takenoya, F., Haruaki, K., Date, Y., Nakazato, M., Shioda, S. (2009). Feeding regulation in the brain: Involvement of Neuropeptide W. In: Shioda, S., Homma, I., Kato, N. (eds) Transmitters and Modulators in Health and Disease. Springer, Tokyo. https://doi.org/10.1007/978-4-431-99039-0_3
Download citation
DOI: https://doi.org/10.1007/978-4-431-99039-0_3
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-99038-3
Online ISBN: 978-4-431-99039-0
eBook Packages: MedicineMedicine (R0)