Abstract
Nuclear cardiology imaging with SPECT or PET is used widely in North America for the diagnosis and management of patients with coronary artery disease. Conventional myocardial perfusion imaging (MPI) can identify areas of reversible ischemia as suitable targets for coronary artery revascularization by angioplasty or bypass surgery. However, the accuracy of this technique is limited in patients with advanced disease in multiple coronary arteries, where there is no normal reference territory against which to assess the “relative” perfusion defects. We have developed methods for the routine quantification of absolute myocardial blood flow (MBF mL/min/g) and coronary flow reserve (stress/rest MBF) using rubidium-82 dynamic PET imaging. The incremental diagnostic and prognostic value of absolute flow quantification over conventional MPI has been demonstrated in several recent studies. Clinical use of this added information for patient management to direct optimal therapy and the potential to improve cardiac outcomes remains unclear, but may be informed by recent progress and widespread clinical adoption of invasive fractional flow reserve(FFR)-directed revascularization. This paper presents recent progress in this field, toward noninvasive CFR image-guided therapy with cardiac PET and SPECT.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
- Noninvasive cardiac imaging
- Myocardial ischemia
- Myocardial blood flow
- Coronary flow reserve
- Positron emission tomography
1 Introduction
Improvements in diagnostic imaging and therapeutic methods have helped to reduce the cardiac death rate in Canada and other developed nations over the past decade [16]. However, cardiovascular disease is still the number one cause of death in most industrialized countries [3]. Noninvasive diagnostic imaging is used increasingly as a “gatekeeper” to help optimize the most effective use of higher-risk invasive (and costly) diagnostic and interventional procedures, such as coronary angiography and revascularization.
This work is motivated in part by the recent FAME trials [7, 38] showing that impaired flow reserve, when used to identify “flow-limiting” epicardial stenoses for revascularization, improved clinical outcomes (reduced cardiac death and myocardial infarction rates) and lowered the total cost of treatment. The FAME trials used invasive angiography measurements of fractional flow reserve (FFR), but with associated risks of embolic stroke and other complications of coronary artery catheterization that may be avoided with the use of noninvasive imaging methods.
Myocardial perfusion reserve (MPR) imaging using positron emission tomography (PET) may enable diagnosis of patients with microvascular disease (uVD) or nonobstructive diffuse epicardial disease, who should not be recommended for coronary revascularization, sparing them the unnecessary risks of invasive angiography for diagnosis alone. Some enhancements to the conventional methods of PET flow reserve imaging are proposed for accurate noninvasive imaging of ischemia, to improve identification of hemodynamically and physiologically significant “flow-limiting” lesions that are optimal targets for invasive revascularization. According to recent AHA/NIH publications [28], “Standard tests used to diagnose CAD are not designed to detect coronary uVD. More research is needed to find the best diagnostic tests and treatments for the disease.” The flow reserve concepts used in this study are illustrated in Fig. 12.1, and the specific terms are defined in Table 12.1.
Epicardial (EFR, FFR) ± microvascular (MPR, CFR) flow reserve measurements using PET imaging and ICA. Normal values of MPR and CFR are approximately 3–5 (average 4.0) in young healthy adults without microvascular disease. Normal epicardial vessels have FFR = 1.0, whereas “flow-limiting” stenoses with FFR < 0.75–0.80 can produce myocardial ischemia. See Table 12.1 for definitions
Current international practice guidelines [1, 2] recommend the use of treadmill exercise-ECG testing and stress perfusion imaging for the diagnosis of ischemia (benefit class I, evidence levels A,B) and the use of invasive flow reserve (FFR) measurements to direct invasive revascularization (benefit class I, IIa, evidence level A) for the treatment of symptoms in patients with suspected ischemic heart disease. Despite a wealth of observational data, stress MPI is still not a class 1(A) indication to direct revascularization in patients with stable ischemic heart disease because there remains insufficient evidence that ischemia-directed therapy reduces the risk of death and/or myocardial infarction.
In conjunction with, or following exercise-ECG testing, stress myocardial perfusion imaging (MPI) is used widely in North America for the noninvasive diagnosis of coronary artery disease. While single-photon emission computed tomography (SPECT) is used most commonly, rubidium-82 (82Rb) PET has been available in the USA since 1989 for the diagnosis of obstructive coronary artery disease (CAD). We recently completed enrolment of >15,000 patients in the Canadian multicenter trial [8] evaluating 82Rb PET as an alternative radiopharmaceutical for myocardial perfusion imaging (Rb-ARMI). Initial results confirmed the high accuracy (>90 %) of low-dose 82Rb PET-CT for diagnosis of obstructive coronary artery disease in patients with epicardial stenoses ≥ 50–70 % [20]. Recent meta-analyses also confirm that PET has higher accuracy for diagnosis of CAD compared to SPECT, even when using current cameras with attenuation correction and ECG-gating [24].
Stress perfusion imaging is also used for the assessment of myocardial ischemia, to identify patients that will benefit from invasive revascularization therapy procedures such as coronary angioplasty and bypass surgery [15] as shown in Fig. 12.2. The efficacy of this approach was suggested initially in the nuclear sub-study of the COURAGE trial [37] and confirmed recently in patients from the DUKE registry [12], showing survival and outcome benefits from invasive revascularization using angioplasty in addition to optimal medical (drug) therapy, in patients with at least 5 % ischemic myocardium improvement. The ISCHEMIA trial currently in progress [23] is intended to verify prospectively, in patients with ischemia by physiological testing (for MPI: at least 10 % ischemic left ventricular (LV) myocardium), whether or not revascularization compared to medical therapy will result in improved clinical outcomes. This is a pivotal trial intended to prove conclusively the value of ischemia detection by stress perfusion imaging. However, but it is important to recognize that conventional stress MPI (using SPECT or PET) will still underestimate the extent and severity of ischemia from diffuse or multivessel disease (patients with left-main coronary artery disease are excluded) and will neither identify - nor direct treatment of - high-risk patients with disease of the coronary microvasculature.
Patients with moderate-to-severe ischemia in the COURAGE nuclear sub-study (A) had a lower rate of death or MI when there was a 5 % improvement (reduction) in ischemic burden following revascularization. In the DUKE nuclear cardiology registry (B), patients with >5 % ischemia worsening had increased risk of death or MI. Retrospective analysis of ~14,000 SPECT-MPI patients (C) indicated that the percent ischemic myocardium (>10–15 %) predicted lower risk (log hazard ratio) of death following early revascularization. In patients with less than 10–15 % ischemic myocardium, medical therapy was the most effective treatment [Reproduced from (A) [37], (B) [12], and (C) [15].]
2 Myocardial Blood Flow (Perfusion) Imaging
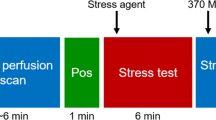
Some limitations of conventional (relative) MPI can be overcome by quantifying myocardial perfusion blood flow (MBF) in absolute units of mL/min/g. Dynamic imaging is required starting from the time of tracer injection, to capture the first-pass transit through the venous-arterial circulation as shown in Fig. 12.3. The concentration of tracer is measured over time in the arterial blood and myocardial tissues, and the rate of uptake or transfer from blood to tissue (influx rate K1 mL/min/g) is related to the absolute myocardial perfusion [21]. Flow quantification restores the true normal-to-diseased tissue contrast (Fig. 12.4), which is otherwise underestimated by measurement of the tracer retention (net uptake) alone. It also allows visualization of the stress/rest perfusion or flow reserve (MPR or MFR) as a measure of the total coronary vascular dilator capacity.
Quantification of MBF using dynamic PET imaging. Dynamic images are acquired starting at the time of tracer injection, then activity in the LV cavity and myocardium is measured over time and fit to a one-tissue-compartment model of the tracer kinetics. The influx rate of tracer uptake or transfer from blood to tissue (K1 mL/min/g) is related to MBF, according to a tracer extraction function E(MBF)
Polar-maps of MBF (flow), 82Rb uptake (K1 influx rate), and retention (net influx − efflux) demonstrating the effects of nonlinear tracer extraction and washout. MBF estimation restores the true disease-to-normal tissue contrast and increases the sensitivity to detect focal disease relative to areas of maximal flow
A one-tissue-compartment model is often used to describe the early kinetics (e.g., 0–5 min) of tracer exchange between the arterial blood supply and myocardial target tissues. This model has been validated for rubidium-82 (82Rb) imaging in humans using nitrogen-13 (13N)-ammonia PET as the reference standard [22] and has been demonstrated to give very reproducible results using several investigational and commercial implementations as shown in Fig. 12.5a, b [9, 27, 31]. Test-retest repeatability of the method (Fig. 12.5c) is approximately 10–12 % CV (coefficient of variation) at rest and 6–7 % during pharmacologic stress [11, 30], comparable to the theoretical expected values verified recently using Monte-Carlo and analytic simulations [25].
MBF values measured using the one-tissue-compartment model are highly reproducible between several investigational (A) and commercial (B) software implementations. Rest and stress flow values are generally within 15–20 %, allowing multicenter data to be pooled or combined between vendors. Test–retest repeatability is 7–10 % at stress and rest (C), for single-session back-to-back scans
Resting MBF is known to correlate highly with the metabolic demands of normal cardiac work [4] as shown in Fig. 12.6; therefore, it is common to adjust the rest of the MBF values to an average reference standard value (e.g., 8500 in typical patients). There a normal age-related increase in RPP, which also contributes to a progressive decline in MPR [10]. The adjusted values at rest represent the expected MBF under conditions of normal controlled systolic blood pressure and heart rate, which are often elevated in patients undergoing stress MPI. The RPP-adjusted MPR represents the flow reserve that would be expected in a patient with normal resting hemodynamics, which may be used to evaluate impairments in coronary vasodilator function associated with atherosclerosis that are unbiased by the effects of resting hypertension.
Because of the wide physiological variability in rest MBF values between patients, interpretation of absolute PET flow studies is recommended to include both the stress MBF and the stress/rest perfusion reserve in combination [18] as illustrated in Fig. 12.7. Abnormalities in both flow reserve < 1.5–2 and stress MBF < 1 mL/min/g have been suggested to represent ischemic tissues that should be considered for revascularization [17]. The absolute flow increase (stress–rest MBF delta) has also been proposed as an alternative method to evaluate the vasodilator response in some studies of vascular endothelial function [36] but has not been as widely applied in practice.
The diagnostic utility of PET MPR assessment has been confirmed in patients with multivessel disease [40]. As shown in Fig. 12.8a, there is a 50 % likelihood of three-vessel disease in patients with a global LV flow reserve that is severely impaired (MPR = 1), whereas the balance of patients presumably have severe microvascular disease limiting their ability to increase myocardial perfusion from rest to peak stress. Measurements of absolute MPR also have prognostic value that is incremental and independent of the standard assessments of relative MPI [41], as shown in Fig. 12.8b. Patients with normal MPI (SSS < 4) but abnormal flow reserve (MFR < 2) are at increased risk of cardiac events. In the case of abnormal MPI, if flow reserve is also impaired, then these patients have the highest rate of cardiac events within the following year. Similar findings have reported in a separate cohort of ischemic heart disease patients [26]; those with the lowest values of MPR had the highest cardiac event rates. Despite these observational studies, there is a lack of evidence proving that revascularization of ischemic myocardium as identified by absolute flow imaging will result in a lower risk of cardiac death or myocardial infarction.
Diagnostic utility of MPR (MFR) in multivessel disease is shown on the left [40]. Patients with global flow reserve < 1 have > 50 % probability of three-vessel disease; the remaining patients have severe microvascular disease. Patients with reductions in flow reserve (MFR < 2) have lower event-free survival, regardless of whether their relative perfusion (SSS) is normal or abnormal [41]
3 Fractional Flow Reserve Assessment
Invasive coronary angiography methods have been developed over the past two decades to quantify the functional or hemodynamic significance of epicardial coronary artery disease, using proximal-distal pressure measurements of the fractional flow reserve ratio (FFR) [13], as illustrated in Fig. 12.9. FFR is defined as the fractional pressure drop measured across one or more stenoses in an individual coronary artery. Interestingly, invasive measurements of FFR were originally validated against 15O-water PET measurements of relative MPR [5]. As shown in Fig. 12.10, coronary FFR values were similar to the relative MPR on average, whereas the myocardial FFR shows a small bias of approximately +10 % vs. the PET analogous values.
Fractional flow reserve is measured as the ratio of intracoronary pressure distal to a stenosis and relative to the (proximal normal) aortic pressure during peak adenosine pharmacologic stress. Comparison of the FFR measurements to coronary angiography allows identification of flow-limiting stenoses that are optimal targets for revascularization
Invasive measurements of FFR were originally validated against 15O-water PET MBF studies in a group of N = 22 patients with single-vessel coronary artery disease. Coronary (epicardial) FFR produced values that were ~10 % higher than the relative flow reserve (relative MPR) values. The myocardial (epicardial + microvascular) FFR values corrected for atrial venous pressure were more accurate on average, but demonstrated an increasing trend versus PET. Adapted from [5]
Epicardial stenoses with abnormal FFR < 0.75 were initially shown to identify the presence of myocardial ischemia with high accuracy compared to a positive test on one or more of three noninvasive methods: exercise thallium planar imaging, or dobutamine stress echocardiography, or treadmill exercise ECG (Fig. 12.11) [33, 34]. This FFR threshold is therefore very sensitive for the detection of ischemia, because it correlates with ischemia on any of the reference standards above.
Fractional flow reserve (FFR) compared to ischemia testing in N = 45 patients, using exercise ECG, thallium imaging, and stress echo [34]. Abnormal FFR < 0.75 was reported to have 88 % sensitivity and 100 % specificity to identify ischemia according to stress echo or exercise thallium imaging or exercise-ECG tests combined(*), but specificity decreases dramatically in the “gray zone” between the FFR cutoff values of 0.75 to 0.80 and when FFR is compared individually to the ischemia standard tests. At the FFR cutoff value < 0.80 commonly used to direct revascularization, fewer than 50 % of subjects had exercise-ECG, stress echo, and thallium tests that were all positive for ischemia
Test-retest repeatability of FFR measurements has been reported in the range of 4–7 % CV [6, 29, 32], similar to the precision of PET stress MBF (Fig. 12.12). This has led to the adoption of a 5 % “gray zone” of uncertainty in FFR measurements considered to be hemodynamically significant or flow limiting.
Test-retest repeatability of back-to-back FFR measurements 10 min apart (4 % CV), reanalyzed from the DEFER study by [32]. The measurement (or classification) uncertainty is shown as the red-green colorbar, reflecting the probability that a revascularization decision would change with repeat measurement. The conventional diagnostic uncertainty or “gray zone” of 0.75–0.80 is shown as the grey-green colorbar
The pivotal FAME trial [38] showed that percutaneous coronary intervention (PCI) revascularization using coronary stenting of anatomically and hemodynamically significant lesions (stenosis ≥ 50 % and FFR ≤ 0.80) improved cardiac outcomes (Fig. 12.13) and reduced the total cost of treatment compared to the standard practice of revascularization for anatomically significant lesions only (stenosis > 50–70 %). However, there remains a significant cost (interventional pressure wires) and patient morbidity (risk of embolic strokes) associated with this invasive procedure. While FFR provides a useful physiological assessment of epicardial stenoses, it does not assess the severity of microvascular disease and actually underestimates the functional significance of epicardial lesions in the presence of microvascular disease [35]. Despite these limitations, FFR has recently been upgraded to a class I(A) indication in Europe and class IIa(A) in North America for use in directing revascularization therapy to improve clinical outcomes.
The FAME randomized controlled trial in N = 1005 patients showed that clinical outcome was improved (87 % vs. 82 % event-free survival; p = 0.02) using FFR-guided revascularization by PCI with drug-eluting stents in patients with intermediate-grade stenosis > 50 % and FFR < 0.80. The FFR-guided approach also resulted in 30 % fewer stents placed per patient (p < 0.001) and 11 % lower overall costs including the added FFR pressure wires [38]
4 Noninvasive PET (MPR) vs. Invasive Coronary Angiography (FFR)
Reductions in the supply of blood to the myocardium are caused by two separate consequences of disease: (1) epicardial coronary stenoses and (2) microvascular dysfunction. The “flow-limiting” epicardial stenoses should be identified ideally as targets for revascularization, whereas patients with diffuse or microvascular disease may be better treated with targeted aggressive medical therapies such as lipid-lowering statins or other novel drug treatments under development to improve endothelial function by increasing nitric-oxide bioavailability, for example.
Myocardial and fractional flow reserve measurements represent different hemodynamic effects of microvascular and epicardial disease. The interrelated physiological interpretation of PET MFR vs. invasive FFR measurements has been the subject of several recent reviews [14, 19]. The discordance between FFR and MPR is attributed to the differences in epicardial vs microvascular disease (Fig. 12.14c) and is consistent with our PET data in over 3,000 patients (Fig. 12.14a, b).
Discordance between FFR and MPR is due to the physiological differences in focal epicardial vs. diffuse or microvascular disease. (A) Invasive [and (B) noninvasive] measures of CFR [and absolute MPR] vs. FFR [and relative MPR] measurements can be discordant in some patients, due to the different physiological consequences of focal vs. diffuse microvascular disease (C). Adapted from [19]
As illustrated in Fig. 12.1, noninvasive PET imaging of MPR measures the capacity to increase perfusion (and tracer delivery) in the downstream microvasculature within the myocardium, reflecting the combined “total” effects of microvascular and epicardial disease. Invasive FFR measures the pressure drop across a single epicardial stenosis during hyperemic stress, representing the peak flow compared to the (restored or expected) normal flow in the absence of stenosis. FFR determines whether a particular epicardial lesion is “flow limiting”; however, this measurement assumes that maximal peak-stress vasodilatation was achieved in the downstream microvasculature. Therefore, in the presence of microvascular dysfunction, FFR can be overestimated (i.e., the severity of disease underestimated) due to a submaximal stress flow response, resulting in underdiagnosis and potential undertreatment of the disease [35].
There is a wide variation in reported MFR values at a given lesion stenosis severity (Fig. 12.15a) confirming the influence of confounding variables such as peak-MFR and/or microvascular flow reserve (uVR). Measurements of total MFR alone cannot separate the fundamental difference in stress flow responses present in the epicardial conduit arteries vs. the microvascular resistance vessels. This effect is illustrated in Fig. 12.15b showing that a 70 % stenosis can appear to have normal or abnormal FFR depending on the peak hyperemic flow response (peak-MFR).
Total myocardial perfusion (flow) reserve (MPR) is a function of epicardial stenosis severity (0–100 %) and microvascular vasodilator response (uVR) as shown in (A, B). In patients with severe microvascular (endothelial) dysfunction (e.g. uVR = −0.5), invasive measurement of fractional flow reserve (FFR) may appear normal in coronary lesions up to 90 % stenosis (C), due to the absence of hyperemic flow response (peak-MFR = 1.0). Epicardial flow reserve (EFR = MPR–uVR) may be useful to identify flow-limiting lesions associated with myocardial ischemia (D)
We have proposed a simple model describing MPR as the sum total of uVR and epicardial CFR as shown in Fig. 12.15c. This model is consistent with previous observations that MFR decreases with increasing lesion stenosis%, but at different reference levels depending on the burden of microvascular disease. uVR is presumed to be independent of epicardial stenosis severity, also consistent with previous invasive measurements of microcirculatory resistance (IMR) [39]. The model predicts that a particular threshold value (EFR = MPR – uVR) for epicardial coronary revascularization will only improve symptoms of ischemia in patients without severe microvascular disease, e.g., with uVR > 0, as shown in Fig. 12.15d. Conversely, myocardial ischemia may be overestimated in young patients without uVD, where an “apparent ischemic” stress perfusion defect in a patient with very high peak-MFR may still be above the true ischemic threshold of stress MBF.
5 Conclusion
Noninvasive nuclear imaging of myocardial blood flow (MBF) and coronary flow reserve (CFR) is now feasible as part of the clinical routine using positron emission tomography (PET) imaging. PET measurements of absolute MBF are reliable and reproducible between imaging centers and software methods, with test–retest repeatability below 10 % coefficient of variation. Ischemic thresholds have been proposed for stress MBF and coronary flow reserve in the range of 1.5 [mL/min/g] and 1.0 [stress/rest MBF], respectively. Prospective trials are needed to determine whether patient outcomes can be improved using these ischemic thresholds to direct appropriate revascularization vs. optimal medical therapies.
References
ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention. J Am Coll Cardiol. 2011;58(24):e44–e122.
ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the Diagnosis and Management of Patients with Stable Ischemic Heart Disease. J Am Coll Cardiol. 2012;60(24):e44–e164.
Canadian Institutes of Health Research Annual Report 2010–11. Moving Forward CIHR Performance across the Spectrum: From Research Investments to Knowledge Translation.
Czernin J, Porenta G, Brunken R, Krivokapich J, Chen K, Bennett R, Hage A, Fung C, Tillisch J, Phelps ME. Regional blood flow, oxidative metabolism, and glucose utilization in patients with recent myocardial infarction. Circulation. 1993;88(3):884–95.
De Bruyne B, Baudhuin T, Melin JA, et al. Coronary flow reserve calculated from pressure measurements in humans. validation with positron emission tomography. Circulation. 1994;89:1013–22.
de Bruyne B, Bartunek J, Sys SU, Pijls NH, Heyndrickx GR, Wijns W. Simultaneous coronary pressure and flow velocity measurements in humans. Feasibility, reproducibility, and hemodynamic dependence of coronary flow velocity reserve, hyperemic flow versus pressure slope index, and fractional flow reserve. Circulation. 1996;94(8):1842–9.
De Bruyne B, Pijls NH, Kalesan B, Barbato E, Tonino PA, Piroth Z, Jagic N, Möbius-Winkler S, Rioufol G, Witt N, Kala P, MacCarthy P, Engström T, Oldroyd KG, Mavromatis K, Manoharan G, Verlee P, Frobert O, Curzen N, Johnson JB, Jüni P. Fearon WF; FAME 2 Trial Investigators. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med. 2012;367(11):991–1001.
deKemp RA, Wells GA, Beanlands RSB. Rubidium-82 – An Alternative Radiopharmaceutical for Myocardial Imaging (Rb-ARMI) 2009–2015. http://clinicaltrials.gov/ct2/show/NCT01128023
deKemp RA, DeClerck J, Klein R, Pan X-B, Nakazato R, Tonge C, Arumugam P, Berman DS, Germano G, Beanlands RS, Slomka PJ. Multi-software reproducibility study of stress and rest myocardial blood flow assessed with 3D dynamic PET-CT and a one-tissue-compartment Model of 82Rb Kinetics. J Nucl Med. 2013;54:571–577.
deKemp RA, Klein R, Renaud J, Garrard L, Wells GA, Beanlands R. Patient age, gender and hemodynamics are independent predictors of myocardial flow reserve as measured with dipyridamole stress PET perfusion imaging. J Nucl Med. 2014;55(S1):79.
Efseaff M, Klein R, Ziadi MC, Beanlands RS, deKemp RA. Short-term repeatability of resting myocardial blood flow measurements using rubidium-82 PET imaging. J Nucl Cardiol. 2012;19(5):997–1006.
Farzaneh-Far A, Phillips HR, Shaw LK, Starr AZ, Fiuzat M, O'Connor CM, Sastry A, Shaw LJ, Borges-Neto S. Ischemia change in stable coronary artery disease is an independent predictor of death and myocardial infarction. JACC Cardiovasc Imaging. 2012;5(7):715–24.
Gould KL, Kirkeeide RL, Buchi M. Coronary flow reserve as a physiologic measure of stenosis severity. J Am Coll Cardiol. 1990;15(2):459–74.
Gould KL. Does coronary flow trump coronary anatomy? JACC Cardiovasc Imaging. 2009;2:1009–23.
Hachamovitch R, Rozanski A, Shaw LJ, Stone GW, Thomson LE, Friedman JD, Hayes SW, Cohen I, Germano G, Berman DS. Impact of ischemia and scar on the therapeutic benefit derived from myocardial revascularization vs. medical therapy among patients undergoing stress-rest myocardial perfusion scintigraphy. Eur Heart J. 2011;32(8):1012–24.
Heart and Stroke Foundation Annual report on Canadians Health. A Perfect Storm of Heart Disease Looming on Our Horizon, January 25, 2010.
Johnson NP, Gould KL. Physiological basis for angina and ST-segment change: PET-verified thresholds of quantitative stress myocardial perfusion and coronary flow reserve. JACC Cardiovasc Imaging. 2011;4:990–8.
Johnson NP, Gould KL. Integrating noninvasive absolute flow, coronary flow reserve, and ischemic thresholds into a comprehensive map of physiological severity. JACC Cardiovasc Imaging. 2012;5:430–40.
Johnson NP, Kirkeeide RL, Gould KL. Is discordance of coronary flow reserve and fractional flow reserve due to methodology or clinically relevant coronary pathophysiology? JACC Cardiovasc Imaging. 2012;5:194–202.
Kaster T, Mylonas I, Renaud JM, Wells GA, Beanlands RSB, deKemp RA. Accuracy of low-dose rubidium-82 myocardial perfusion imaging for detection of coronary artery disease using 3D PET and normal database interpretation. J Nucl Cardiol. 2012.
Klein R, Renaud JM, Ziadi MC, Beanlands RSB, deKemp RA. Intra- and inter-operator repeatability of myocardial blood flow and myocardial flow reserve measurements using rubidium-82 pet and a highly automated analysis program. J Nucl Cardiol. 2010;17:600–16.
Lortie M, Beanlands RS, Yoshinaga K, Klein R, Dasilva JN, deKemp RA. Quantification of myocardial blood flow with 82Rb dynamic PET imaging. Eur J Nucl Med Mol Imaging. 2007;34:1765–74.
Maron D, Boden W, Ferguson B, Harrington R, Stone G, Williams D. International study of comparative health effectiveness with medical and invasive approaches (ISCHEMIA). https://clinicaltrials.gov/ct2/show/NCT01471522
Mc Ardle BA, Dowsley TF, deKemp RA, Wells GA, Beanlands RS. Does rubidium-82 PET have superior accuracy to SPECT perfusion imaging for the diagnosis of obstructive coronary disease? A systematic review and meta-analysis. J Am Coll Cardiol. 2012;60(18):1828–37.
Moody J, Murthy V, Lee B, Corbett J, Ficaro E. Variance Estimation for Myocardial Blood Flow by Dynamic PET. IEEE Trans Med Imaging. 2015 May 13. [Epub ahead of print]
Murthy VL, Naya M, Foster CR, Hainer J, Gaber M, Di Carli G, Blankstein R, Dorbala S, Sitek A, Pencina MJ, Di Carli MF. Improved cardiac risk assessment with noninvasive measures of coronary flow reserve. Circulation. 2011;124(20):2215–24.
Nesterov SV, Deshayes E, Sciagrà R, Settimo L, Declerck JM, Pan XB, Yoshinaga K, Katoh C, Slomka PJ, Germano G, Han C, Aalto V, Alessio AM, Ficaro EP, Lee BC, Nekolla SG, Gwet KL, deKemp RA, Klein R, Dickson J, Case JA, Bateman T, Prior JO, Knuuti JM. Quantification of myocardial blood flow in absolute terms using (82)Rb PET imaging: the RUBY-10 Study. JACC Cardiovasc Imaging. 2014;7(11):1119–27.
NIH-NHLBI website. What is Coronary Microvascular Disease? http://www.nhlbi.nih.gov/health/health-topics/topics/cmd. Accessed September 2015.
Ntalianis A, Sels JW, Davidavicius G, Tanaka N, Muller O, Trana C, Barbato E, Hamilos M, Mangiacapra F, Heyndrickx GR, Wijns W, Pijls NH, De Bruyne B. Fractional flow reserve for the assessment of nonculprit coronary artery stenoses in patients with acute myocardial infarction. JACC Cardiovasc Interv. 2010;3(12):1274–81.
Ocneanu A, Adler A, Renaud J, Beanlands R, deKemp R, Klein R. Reproducible tracer injection profile improves the test-retest repeatability of myocardial blood flow quantification with 82Rb PET. J Nucl Med. 2015;56(S3):207.
Pan X-B, DeClerck J. Validation syngo PET Myocardial Blood Flow. Siemens Healthcare: White Paper 2012.
Petraco R, Sen S, Nijjer S, Echavarria-Pinto M, Escaned J, Francis DP, Davies JE. Fractional flow reserve-guided revascularization: practical implications of a diagnostic gray zone and measurement variability on clinical decisions. JACC Cardiovasc Interv. 2013;6(3):222–5. doi: 10.1016/j.jcin.2012.10.014. Erratum in: JACC Cardiovasc Interv. 2013;6(4):431.
Pijls NH, van Son JA, Kirkeeide RL, De Bruyne B, Gould KL. Experimental basis of determining maximum coronary, myocardial, and collateral blood flow by pressure measurements for assessing functional stenosis severity before and after percutaneous transluminal coronary angioplasty. Circulation. 1993;87:1354–67.
Pijls NH, De Bruyne B, Peels K, Van Der Voort PH, Bonnier HJ, Bartunek J, Koolen JJ, Koolen JJ. Measurement of fractional flow reserve to assess the functional severity of coronary-artery stenoses. NEJM. 1996;334(26):1703–8.
Pijls NH, Tonino PA. The CRUX of maximum hyperemia: the last remaining barrier for routine use of fractional flow reserve. JACC Cardiovasc Interv. 2011;4(10):1093–5.
Schindler TH, Zhang X-L, Prior JO, Cadenas J, Dahlbom M, Sayre J, Schelbert HR. Assessment of intra- and inter-observer reproducibility of rest and cold-pressor-test stimulated myocardial blood flow with 13N-ammonia and PET. Eur J Nucl Med Mol Imaging. 2007;34:1178–88.
Shaw LJ, Berman DS, Maron DJ, et al. Optimal medical therapy with or without percutaneous coronary intervention to reduce ischemic burden: Results from the clinical outcomes utilizing revascularization and aggressive drug evaluation (COURAGE) trial nuclear sub-study. Circulation. 2008;117:1283–91.
Tonino PA, De Bruyne B, Pijls NH, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360:213–24.
Yong ASC, Ho M, Shah MG, Ng MKC, Fearon WF. Coronary microcirculatory resistance is independent of epicardial stenosis. Circ Cardiovasc Interv. 2012;5:103–8.
Ziadi MC, deKemp RA, Williams K, Beanlands RSB. Does quantification of myocardial flow reserve using rubidium-82 positron emission tomography facilitate detection of multi-vessel coronary artery disease? J Nucl Cardiol. 2012;19:670–80.
Ziadi MC, deKemp RA, Williams K, Beanlands RSB. Impaired myocardial flow reserve on rubidium-82 positron emission tomography imaging predicts adverse outcomes in patients assessed for myocardial ischemia. J Am Coll Cardiol. 2011;58:740–8.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is distributed under the terms of the Creative Commons Attribution-Noncommercial 2.5 License (http://creativecommons.org/licenses/by-nc/2.5/) which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
The images or other third party material in this chapter are included in the work’s Creative Commons license, unless indicated otherwise in the credit line; if such material is not included in the work’s Creative Commons license and the respective action is not permitted by statutory regulation, users will need to obtain permission from the license holder to duplicate, adapt or reproduce the material.
Copyright information
© 2016 The Author(s)
About this paper
Cite this paper
deKemp, R.A., Beanlands, R.S. (2016). Noninvasive PET Flow Reserve Imaging to Direct Optimal Therapies for Myocardial Ischemia. In: Kuge, Y., Shiga, T., Tamaki, N. (eds) Perspectives on Nuclear Medicine for Molecular Diagnosis and Integrated Therapy. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55894-1_12
Download citation
DOI: https://doi.org/10.1007/978-4-431-55894-1_12
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55892-7
Online ISBN: 978-4-431-55894-1
eBook Packages: MedicineMedicine (R0)