Abstract
The operative mortality, graft patency, and long-term outcomes in OPCAB compared with conventional CABG are still controversial. The 30-day mortality after primary elective CABG with CPB was less than 1.0 % in Japan in 2011. Therefore, it is not surprising that the recent RCTs in western countries showed no difference in operative mortality between OPCAB and on-pump CABG. Prospective, randomized trials have been criticized because they lack adequate power to assess clinical outcomes because they had been performed in single centers and the surgeons were not accustomed to both OPCAB and standard CABG. Considering the high occlusion late of saphenous vein graft (SVG) in OPCAB because of hypercoagulability status after surgery, OPCAB may sacrifice graft patency and cause late unfavorable cardiac events.
Routine early postoperative angiography was performed in 3,532 patients between January 2002 and December 2005 in 12 centers within 3 weeks after CABG in Japan. OPCAB was performed in 85 % of all cases. The occlusion rate of SVG (7.3 %) was significantly higher than left internal thoracic artery (LITA) (1.7 %), right internal thoracic artery (RITA) (1.7 %), radial artery (RA) (3.4 %), and right gastroepiploic artery (GEA) (3.8 %) in OPCAB. There was no difference in the bypass occlusion and stenosis rate between OPCAB and standard CABG cases. The occlusion rate of SVG (7.3 %) in OPCAB was significantly (p = 0.032) higher than that (2.8 %) in standard CABG. Bypass occlusion and stenosis rate of RITA to left anterior descending artery (LAD) (6.1 %) was significantly (p = 0.013) higher than that of LITA to LAD (3.8 %). In the circumflex area bypass occlusion rate of SVG (7.1 %) was significantly higher than that of all arterial grafts of LITA (4.2 %, p = 0.030), RITA (1.7 %, p < 0.001), RA (2.2 %, p < 0.001), and GEA (3.7 %, p = 0.029). Bypass occlusion rate in the right coronary territory was not related to graft material. The LITA was the best graft for LAD, and the RA was as good as the RITA and better than SVG in the circumflex area. SVG should be avoided in OPCAB, and multiple arterial OPCAB in Japan is the optimal strategy.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Different Situation of OPCAB in Japan
The number of coronary artery bypass grafting (CABG) reached more than 22,000 in 2003 in Japan [1]. However, the number declined up to 30 % in 2011 [2] owing to unrestricted percutaneous coronary intervention with drug-eluting stents regardless of coronary artery revascularization guidelines. During the last decade, the development of CABG without cardiopulmonary bypass (CPB) has significantly changed CABG in Japan. In 1967, Kolessov first grafted the left internal thoracic artery (LITA) to the anterior descending artery through a left thoracotomy without CPB [3]. Coronary artery revascularization with cardioplegic arrest became the standard CABG procedure with development of CPB technique and myocardial preservation methods for cardiac arrest during the early 1980s. After the pioneering efforts by Benetti [4] and Buffolo [5] in South America for OPCAB through a median sternotomy during the 1990s, suction-type mechanical stabilizer and apical suction device in addition to Trendelenburg positioning by the operating table allowed exposure of all coronary branches. According to the Japanese Association for Thoracic Surgery, the percentage of off-pump CABG (OPCAB) among the total CABG cases was 35 % in 2001, 46 % in 2002, 55 % in 2003, and 60 % in 2004. The frequency of OPCAB reached a plateau after 2005 according to the database of the Japanese Association for Coronary Artery Surgery. Therefore, OPCAB has become the standard method of CABG in Japan, whereas the percentage of OPCAB cases among all CABGs in North America was less than 20 % and in Germany 5 %. As some surgeons in the United States are performing 90 % or more of the CABGs without CPB, the frequency of OPCAB in standard hospitals might be less than 10 %.
2 Positive Impact of OPCAB
The indications of OPCAB are the high risk factors of CPB such as cerebrovascular disease, chronic renal failure, chronic obstructive pulmonary disease, atheromatous or calcified ascending aorta, older age, liver cirrhosis, and cancer. Many retrospective and cohort studies have been published comparing clinical outcomes after OPCAB with conventional CABG [6–11]. In moderate and high-risk patients, the operating time, intubation time, length of ICU stay, length of hospital stay, perioperative myocardial infarction, amount of bleeding and transfusion, inflammatory reaction, stroke, neurocognitive dysfunction, atrial fibrillation, and inotrope requirements were in favor of OPCAB [12].
A meta-analysis of 42 non-randomized trials of high-risk patients by Puskas and associates [13] demonstrated a significant reduction in mortality after OPCAB versus conventional CABG in various high-risk patient subsets (odds ratio (OR): 0.58). Mortality was reduced in patient subgroups with high risk factors of EuroSCORE >5 (OR: 0.39), LV dysfunction (OR: 0.55), atheromatous aorta (OR: 0.54), and multiple risk factors (OR: 0.60). Increased age, left main trunk lesion, diabetes mellitus, renal dysfunction, and chronic obstructive pulmonary disease were not related to the operative mortality irrespective of CPB.
A systematic review and meta-analysis of propensity score analysis by Kuss and associates in 123,137 patients of 35 papers found that OPCAB was superior to conventional CABG [14]. The benefit was operative mortality (OR: 0.69), renal failure, transfusion, wound infection and prolonged ventilation, inotropic support, and intra-aortic balloon pump (IABP) support. However, myocardial infarction, atrial fibrillation, and reoperation for bleeding were similar.
In the early RCTs [15–20], postoperative various complications, ICU stay, hospital stay, bleeding, and transfusion were in favor of OPCAB. The early meta-analysis of previous RCTs by Cheng and associates [21] showed no significant differences in the 30-day mortality, IABP support, myocardial infarction, stroke, renal dysfunction, wound infection, re-exploration for bleeding, or re-intervention for mixed-risk patient population. However, OPCAB significantly decreased atrial fibrillation (OR: 0.58), transfusion (OR: 0.43), inotrope requirements (OR: 0.48), respiratory infection (OR: 0.41), ventilation time (−2.4 h), ICU stay (−0.3 day), and hospital stay (−1.0 day). Results of graft patency and neurocognitive function were inconclusive. In-hospital and 1-year medical costs were generally higher for standard CABG.
Møller also meta-analyzed 66 randomized trials [22]. There were no statistically significant differences regarding mortality (OR: 0.98), myocardial infarction (OR: 0.95), or renewed coronary revascularization (OR: 1.34). There was a significant reduced risk of atrial fibrillation (OR: 0.69) and stroke (OR: 0.53) in off-pump patients. However, when continuity correction for zero-event trials was included, the reduction in stroke became insignificant.
Puskas and associates reported the long-term outcomes of earlier Surgical Management of Arterial Revascularization Therapy (SMART) trial [19] involving 297 patients after isolated elective CABG. After 7.5 years of follow-up, there was no difference in mortality or late graft patency between OPCAB and on-pump CABG. Recurrent angina was more common in the OPCAB group though this did not reach statistical significance [23].
3 Negative Impact of OPCAB in RCT
Prospective, randomized trials have been criticized because they lack adequate power to assess clinical outcomes because they had been performed in single centers and the surgeons were not accustomed to both OPCAB and standard CABG. To overcome these problems, Randomized On/Off Bypass (ROOBY) study was performed in 2203 patients in 18 Veterans Affairs medical centers with 53 attending surgeons [24]. There was no difference in 30-day mortality or short-term major adverse cardiovascular events. OPCAB patients received significantly fewer grafts per patient. After 1 year, cardiac-related death (8.8 % versus 5.9 %, p = 0.01) and major adverse events (9.9 % versus 7.4 %, p = 0.04) were significantly higher in the OPCAB group. Furthermore, graft patency was significantly lower in the OPCAB group (82.6 % versus 87.8 %, p < 0.001). There was no difference in neuropsychological testing between the groups.
Recently, the results of the Coronary Artery Bypass Surgery Off or On Pump Revascularization Study (CORONARY) were reported [25, 26]. In the largest trial of OPCAB versus standard CABG, with 4752 patients from 79 centers in 19 countries, no significant difference was observed in the primary outcome of death, myocardial infarction, stroke, or new hemodialysis at 30 days (9.8 % vs. 10.3 %, p = 0.59). There was a lower rate of transfusion (50.7 % vs. 63.3 %, p < 0.001) and a higher rate of repeat revascularization (0.7 % vs. 0.2 %, p = 0.01) in OPCAB group. The 1-year clinical results were consistent with the 30-day findings. There was no significant difference in the composite outcome of death, myocardial infarction, stroke, or new hemodialysis (12.1 % vs. 13.3 %, p = 0.24). Repeat revascularization remained more common in the OPCAB group (1.4 % vs. 0.8 %, p = 0.07), and QOL and neurocognitive outcomes were similar in both groups.
Similar negative results of OPCAB were found in the most recent meta-analysis of 8961 patients with a mean age of 63.4 and 16 % females in 59 trials by Afilalo [27]. There was a significant 30 % reduction in the occurrence of postoperative stroke with OPCAB (OR: 0.70). However, there was no significant difference in mortality (OR: 0.90) or myocardial infarction (OR: 0.89).
RCTs were performed in high-risk patients in several studies. In a study involving high-risk patients with three-vessel disease and a EuroSCORE ≥5, Møller and associates could find no difference in morbidity or mortality between OPCAB and standard CABG [28]. The mean number of grafts per patient did not differ significantly between groups (3.22 in OPCAB group and 3.34 in standard CABG group, p = 0.11). Fewer grafts were performed to the lateral part of the left ventricle territory during OPCAB (0.97 in OPCAB and 1.14 in standard CABG, p = 0.01).
The German Off-Pump Coronary Artery Bypass Grafting in Elderly Patients (GOPCABE) trial randomly assigned 2539 patients 75 years of age or older from 12 German centers to OPCAB or standard CABG [29]. No significant difference between groups was observed in the primary outcome of death, myocardial infarction, stroke, repeat revascularization, or new renal-replacement therapy at 30 days (7.8 % vs. 8.2 %, p = 0.74) or at 1 year (13.1 % vs. 14.0 %, p = 0.48). There were fewer transfusions and more repeat revascularizations (1.3 % vs. 0.4 %, p = 0.04) with off-pump surgery.
4 Multicenter Angiographic Analysis of Early Graft Patency in Relation to Graft Material and OPCAB in Japan
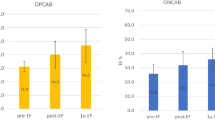
This study was a multicenter retrospective analysis of early elective postoperative coronary angiography performed within 3 weeks after CABG between January 2002 and December 2005 in 12 centers. Postoperative angiography was performed in 3,532 patients who underwent elective and emergent CABG. Mean age at operation was 65.6 ± 10.1 years. OPCAB was performed in 85 % of all cases. Average number of distal anastomoses was 3.4 ± 1.2 (OPCAB, 3.4 ± 1.2; standard CABG, 3.2 ± 1.2, p < 0.001) (Fig. 3.1). Average number of grafts was 2.4 ± 0.6 (OPCAB, 2.4 ± 0.6; standard CABG, 2.3 ± 0.7, p = 0.35). Average number of arterial graft anastomosis was 2.9 ± 2.1. Distribution of graft material was 36 % left internal thoracic artery (LITA), 18 % right internal thoracic artery (RITA), 10 % gastroepiploic artery (GEA), 19 % radial artery (RA), and 17 % saphenous vein graft (SVG) with regard to distal anastomosis. Exclusive use of LITA and SVG was applied in 30 % of patients. Both LITA and RITA were used in 45 % of patients. More than two arterial grafts were used in 84 % of patients. Total arterial revascularization was performed in 67 % of patients. Graft configuration was composed of 49 % in situ graft, 39 % composite graft, and 12 % of aortocoronary bypass.
Overall bypass occlusion rate was 3.1 % (366/11,968). Graft stenosis rate was 1.9 %. Overall bypass occlusion rate was 2.1 % in LITA, 1.9 % in RITA, 3.3 % in RA, 6.3 % in SVG, and 4.2 % in GEA. There were 297 patients with at least one occluded bypass. Univariate analysis showed that the risk factors of patients with occluded bypass grafting were female gender, weight, height, diabetes, blood creatinine level, liver dysfunction, non-left main trunk lesion, three-vessel disease, family history of ischemic heart disease, more than four distal anastomoses, and no use of preoperative β blocker and aspirin (Table 3.1). OPCAB or prior percutaneous coronary intervention was not a risk factor. In multivariate analysis, only female gender (HR, 1.53: 95 % CI, 1.13–2.07), liver dysfunction (HR, 2.09: 95 % CI, 1.18 to 3.70), non-left main trunk lesion (HR, 1.54: 95 % CI, 1.14–2.08), family history of ischemic heart disease (HR, 1.52: 95 % CI, 1.07 to 2.17), and more than four distal anastomoses (HR, 1.34: 95 % CI, 1.01–1.77) were risk factors (Table 3.2). Bypass occlusion was related to operative morbidity (p = 0.003) and all morbidity (p = 0.01). Bypass occlusion was not related to operative death or hospital death (Table 3.3).
Graft occlusion rate in relation to graft material and cardiopulmonary bypass was shown in Fig. 3.2. The occlusion rate of SVG (7.3 %) was significantly (p < 0.001) higher than LITA (1.7 %), RITA (1.7 %), RA (3.4 %), and GEA (3.8 %) in OPCAB. The occlusion rate of RA and GEA was also significantly (p < 0.001) higher than LITA and RITA in OPCAB. In standard CABG patients, bypass occlusion rate was not statistically different between graft materials. The occlusion rate of RITA (1.7 %) in OPCAB was significantly (p = 0.046) lower than that (3.5 %) in standard CABG. On the contrary, the occlusion rate of SVG (7.3 %) in OPCAB was significantly (p = 0.032) higher than that (2.8 %) in standard CABG. As a whole there was no difference in the bypass occlusion rate between OPCAB (3.1 %) and standard CABG (2.9 %).
Bypass occlusion and stenosis was examined in relation to graft material and bypass area. Bypass occlusion and stenosis rate of RITA to left anterior descending artery (LAD) (6.1 %) was significantly (p = 0.013) higher than that of LITA to LAD (3.8 %) though the difference of occlusion rate did not reach statistical significance (p = 0.053) (Fig. 3.3). In the circumflex area bypass occlusion rate of SVG (7.1 %) was significantly higher than that of all arterial grafts of LITA (4.2 %, p = 0.030), RITA (1.7 %, p < 0.001), RA (2.2 %, p < 0.001), and GEA (3.7 %, p = 0.029). The occlusion rate of RITA was significantly lower than that of LITA (p = 0.002) and GEA (p = 0.046) in this area (Fig. 3.4). The occlusion rate of RA was significantly lower than that of LITA (p = 0.024) in the circumflex area. Bypass occlusion rate in the right coronary territory was not related to graft material (Fig. 3.5). However, overall bypass occlusion rate in the right coronary area (4.8 %) was significantly (p < 0.001, p = 0.015) higher than that in LAD (1.8 %) and circumflex area (3.5 %).
Bypass occlusion and stenosis in relation to the site of the right coronary artery (RCA) was examined. Bypass occlusion and stenosis of the RITA to the right main coronary artery (11.1 %) was significantly higher than that of RA (0 %, p = 0.004) and SVG (2.0 %, p = 0.013) (Fig. 3.6). Bypass occlusion and stenosis of the GEA to the main RCA was significantly higher (13.3 %) than that of RA (p < 0.001) and SVG (p < 0.001). Bypass occlusion and stenosis rate of GEA to the main right coronary artery was significantly (p = 0.009) higher than that to the posterior descending artery (5.1 %). Bypass occlusion and stenosis rate of SVG to the main RCA was significantly lower than that to the posterior descending artery (7.4 %, p = 0.045) and atrioventricular branch (11.6 %, p < 0.001).
Bypass occlusion of the RA in relation to composite and aortocoronary bypass configuration was examined. The occlusion rate of RA as a composite graft was significantly (p = 0.0023) higher in the right coronary territory (5.4 %) than that in the circumflex territory (2.1 %) (Fig. 3.7). There was no significant difference in RA occlusion rate between composite and aortocoronary bypass both in the circumflex and the right coronary area.
Female gender, liver dysfunction, non-left main trunk lesion, family history of ischemic heart disease, and more than four distal anastomoses were independent risk factors in patients who had at least one bypass occlusion. Women have more severe coronary disease, higher age, higher comorbidity, lower body surface area, and lower percentage of LITA use. However, even after the risk matching of these factors, it was confirmed that female gender is an independent risk factor for adverse events and mortality after CABG [30]. Smaller body surface area and fragile coronary artery were the major causes of higher risk of perioperative mortality and morbidity in women [31]. Early occlusion of the bypass graft may explain the higher mortality and morbidity in women according to our results. There was no previous report regarding liver dysfunction and bypass graft patency to my knowledge. Liver dysfunction or cirrhosis may cause coagulation abnormality after CABG. Risk factors of non-left main trunk lesion, family history of ischemic heart disease, and more than four distal anastomoses may be related to the severity and diffuseness of the coronary lesion. The difficulty and many graft anastomosis sites may be attributed to bypass occlusion.
In the present study, there were several interesting results regarding the graft material and the method of its use both in OPCAB and standard CABG. First, LITA was the best graft for LAD. The RITA was not as good as the LITA as a graft for LAD. As the multivariate analysis showed that the female gender was an independent risk factor of graft occlusion, distal RITA in women might be not appropriate for LAD grafting. Second, in the circumflex area, graft patency of RITA and RA were better than LITA, GEA, and SVG. There was no difference in the graft patency of RA and RITA in this area. RITA was used as a composite graft with LITA rather than in situ graft. There was no difference in the RA graft patency in this area between the method of composite and aortocoronary bypass in the present study. RA could be used as a composite graft as well as aortocoronary bypass in some previous studies [32, 33]. However, Gaudino and associates found flow competition more frequently in the composite RA conduits than in the aortocoronary RA conduit [34]. RA is suitable for the second arterial graft next to LITA as composite graft at least in the circumflex area. Third, in the right coronary territory, the overall graft patency was worse than LAD and circumflex area. Bypass occlusion rate in the right coronary territory was not related to graft material. However, RITA and GEA were not suitable for the graft to the main RCA probably because the coronary arterial wall was much thicker than the wall of RITA and GEA, which might cause graft stenosis and occlusion. For the main RCA RA and SVG grafts were better than the RITA and the GEA as in situ graft. RA graft patency in this site was not better than SVG. Considering the higher occlusion rate of the composite RA in this area compared to aortocoronary RA grafting, the RA as aortocoronary bypass to the right coronary branches might be favorable when total arterial grafting is aimed.
Total arterial coronary revascularization could avoid the problems associated with vein graft failure [35, 36]. Bilateral ITAs are the conduits of first choice because of excellent short- and long-term patency and improved survival [37, 38]. However, bilateral ITA harvesting has shown a higher incidence of sternal wound infection in patients taking insulin or steroids, who are obese or have chronic obstructive lung disease [30]. RITA to LAD graft crossing the midline of the chest may obstruct future reoperation for aortic valve surgery, but to avoid this situation, the RITA is unable to reach the left ventricular posterolateral wall if it passes through the transverse sinus. As the second arterial graft in addition to LITA to LAD anastomosis, some studies comparing RITA and RA showed the same clinical and angiographic results [39, 40].
The comparative performance of RA versus SVG is of considerable interest. Previous angiographic observational studies have shown that the RA achieved excellent short- (96–100 %), mid- (94–97 %), and long-term (84–96 %) patency [41]. Patency rates of the RA have exceeded those of the SVGs at all-time points. Only the Cleveland Clinic reported worse graft patency of the RA than the SVG [42]. Randomized comparison of midterm graft patency between the RA and the SVG at 5 years showed disappointing graft patency of RA (87 %) compared to SVG (94 %) in RAPCO trial [43]. However, these results were based on the small angiographic studies. The RSVP randomized trial was designed to compare 5-year patency rates of aortocoronary RA and SVG to the circumflex coronary artery. The graft patency of the RA (98.3 %) was significantly better than that of the SVG (86.4 %) [44]. In the RAPS trial, the RA was randomly assigned to bypass the major artery in either the right coronary territory or the circumflex coronary territory, with the SVG used for the opposite territory, which had proximal lesions at least 70 % diameter narrowing [45]. The graft occlusion of the RA (8.2 %) was significantly lower than that of the SVG (13.6 %) at 1 year. Diffuse narrowing of the graft was present in 7.0 % of the RA grafts and only 0.9 % of SVGs. The absence of severe native vessel stenosis was a risk of graft occlusion and diffuses narrowing of the RA. Patency of the RA grafts was similar in RCA and circumflex arteries. In our study graft occlusion rate of the RA (2.2 %) was better than that of the SVG (7.1 %) in the circumflex area, but the same in the right coronary territory (RA: 5.0 %, SVG: 5.6 %). In our early postoperative study, composite RA grafts showed competitive flow in the setting of <75 % proximal coronary stenosis especially in the right coronary branches [46].
Graft patency of RITA and GEA was lower in standard CABG than in OPCAB in this study. The reason could be explained as follows. In OPCAB cases grafting to the main RCA might be avoided because hypotension and bradycardia occur frequently when the native coronary stenosis is moderate. The main RCA is sometimes very thick and calcified compared to other territory of right coronary branches, which are not suitable for OPCAB. Bypass occlusion rate of RITA and GEA was higher when the target area was the main RCA compared to grafting to the right coronary branches.
In summary, the LITA was the best graft for LAD, and the RA was as good as the RITA and better than SVG in the circumflex area. SVG should be avoided in OPCAB due to postoperative hypercoagulability status. Graft patency in the RCA area was not related to graft material, and the RITA and GEA should be avoided as a graft to the main RCA.
5 Critical Appraisal of Recent RCT in Western Countries
The operative mortality, graft patency, and long-term outcomes in OPCAB compared with conventional CABG are still controversial. The 30-day mortality after primary elective CABG with CPB was less than 1.0 % in Japan in 2011 [2]. Therefore, it is not surprising that the recent RCTs in western countries showed no difference in operative mortality between OPCAB and on-pump CABG. Puskas and associates demonstrated that OPCAB using STS database disproportionally benefits high-risk patients and female gender [47, 48]. In elective CABG cases in Japan, the operative mortality of OPCAB and standard CABG has been similar since 2008 (Table 3.4). On the contrary, in all CABG cases, the operative mortality of standard CABG was twice as high as that of OPCAB last 10 years. In the real world OPCAB has decreased the mortality in high-risk patients at least in Japan. The operative mortality in conversion cases from OPCAB to CABG with CPB has been between 2.7 and 6.7 % even in primary elective CABG. Including all cases that were converted to CABG with CPB, the operative mortality was between 4.4 and 8.3 %. Cardiac center and cardiac surgeons should be accustomed to OPCAB as a daily operation from this data, although less than 10 % of patients who have CABG really need OPCAB. Otherwise unexpected conversion from OPCAB to CABG with CPB caused deleterious outcomes.
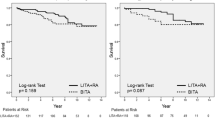
Poorer late outcomes of repeat revascularization in OPCAB must be related to fewer number of grafts especially to the lateral wall and poorer graft patency and quality of bypass. In the ROOBY study, the conversion rate from OPCAB to on-pump CABG was 12.4 % [24], which is significantly higher than the 2.2 % found in the STS database. The rate of crossover from the off-pump to the on-pump group was 7.9 % in the CORONARY trial [25]. The conversion rate from OPCAB to CABG with CPB was only 3 % in Japan in 2011 [2]; the inexperience of surgeons has biased these RCTs. In our RCT from Japan [49], the number of grafts performed per patient (3.5 for OPCAB and 3.6 for standard CABG) and the number of arterial grafts performed per patient (3.3 for OPCAB and 3.4 for standard CABG) were similar. No patients were converted from OPCAB to CPB.
A meta-analysis of graft patency in RCT by Takagi and associates showed a 27 % increase in overall graft occlusion with OPCAB, especially a 28 % increase in saphenous vein (SV) graft occlusion [50]. Table 3.5 showed the graft patency in several RCTs [17, 18, 24, 49, 51–55]. Khan and colleagues [18] showed that the graft patency of off-pump surgery (88 %) was worse than that of on-pump surgery (98 %). These poor results in OPCAB have been explained by the fact that only 13 % of total CABG cases were performed without CPB. This may indicate that this trial was performed within the learning curve phase. In the PRAGUE-4 study, graft patency of SV in OPCAB was also 10 % lower than that in standard CABG though statistically not significant [51]. Lingaas also reported that although patency of LITA was 94 % in the OPCAB group and 96 % in the on-pump group 12 months after RCT, graft patency of SV was 80 % and 87 %, respectively [53]. In the BHACAS 1 and BHACAS 2 study, graft occlusion rate was similar in the standard CABG and OPCAB groups, both overall (27/255 [10.6 %] and 26/237 [11.0 %], respectively) and for arterial and SVG separately (arterial grafts, 13/126 [10.3 %] vs 8/116 [6.9 %]; SVGs, 14/129 [10.9 %] vs 18/121 [14.9 %], respectively) [54]. In the study of Uva and associates, SV graft patency was 86 % in the OPCAB group and 93 % in the standard CABG group, which was not statistically different [55]. The high occlusion rate of grafts in OPCAB must be due to high occlusion rate of SV in addition to technical difficulty.
Kim and associates compared [56] the 1-year graft patency after OPCAB with CABG under CPB. The graft patency of SVG was 88 % in patients undergoing standard CABG, 87 % in patients undergoing on-pump beating CABG, and 68 % in patients undergoing OPCAB. Poor graft patency and poor long-term results in OPCAB were related to higher SVG occlusion rate because of the hypercoagulability status after OPCAB [57, 58]. Although they conducted the study during the learning time period, this paper could not be overlooked. Proper anticoagulation and antiplatelet regimens are mandatory. We recommended that heparin was reversed at OPCAB completion, but restarted (10 unit/kg/h) in the intensive care unit after hemostasis was secured until the next evening [59]. Aspirin was continued until the operation and restarted with 162 mg/day from the next morning. Without proper perioperative medical treatment, OPCAB may sacrifice graft patency and cause late unfavorable cardiac events. Considering the high occlusion rate of SVG in OPCAB, multiple arterial OPCAB in Japan is the optimal strategy.
References
Kazui T, Wada H, Fujita H (2005) Thoracic and cardiovascular surgery in Japan during 2003. Jpn J Thorac Cardiovasc Surg 53:517–536
Amano J, Kuwano H, Yokomise H (2013) Thoracic and cardiovascular surgery in Japan during 2011. Gen Thorac Cardiovasc Surg 61:578–607
Kolessov VI (1967) Mammary artery-coronary artery anastomosis as method of treatment for angina pectoris. J Thorac Cardiovasc Surg 54:535–544
Benetti FJ, Naselli G, Wood M et al (1991) Direct myocardial revascularization without extracorporeal circulation. Experience in 700 patients. Chest 100:312–316
Buffolo E, Andrade JCS, Branco JNR et al (1996) Coronary artery bypass grafting without cardiopulmonary bypass. Ann Thorac Surg 61:63–66
Cartier R, Brann S, Dagenais F et al (2000) Systematic off-pump coronary artery revascularization in multivessel disease: experience of three hundred cases. J Thorac Cardiovasc Surg 119:221–229
Zenati M, Cohen HA, Griffith BP (1999) Alternative approach to multivessel coronary disease with integrated coronary revascularization. J Thorac Cardiovasc Surg 117:439–446
Arom FV, Flavin TF, Emery RW et al (2000) Safety and efficacy of off-pump coronary artery bypass grafting. Ann Thorac Surg 69:704–710
Cleveland JC, Shroyer AL, Chen AY et al (2001) Off-pump coronary artery bypass grafting decreases risk-adjusted mortality and morbidity. Ann Thorac Surg 72:1282–1288
Plomondon ME, Cleveland JC Jr, Ludwig ST et al (2001) Off-pump coronary artery bypass is associated with improved risk adjusted outcomes. Ann Thorac Surg 72:114–119
Sabik JF, Gillinov AM, Blackstone EH et al (2002) Does off-pump coronary surgery reduce morbidity and mortality? J Thorac Cardiovasc Surg 124:698–707
Cheng W, Denton TA, Fontana GP et al (2002) Off-pump coronary surgery: effect on early mortality and stroke. J Thorac Cardiovasc Surg 124:313–320
Puskas J, Cheng D, Knight J et al (2005) Off-pump versus conventional coronary artery bypass grafting: a meta-analysis and consensus statement from the 2004 ISMICS consensus conference. Innovations 1:3–27
Kuss O, von Salviati B, Börgermann J (2010) Off-pump versus on-pump coronary artery bypass grafting: a systemic review and meta-analysis of propensity score analyses. J Thorac Cardiovasc Surg 140:829–835
van Dijk D, Nierich AP, Jansen EW et al (2001) Early outcome after off-pump versus on-pump coronary artery bypass surgery. Results from a randomized study. Circulation 104:1761–1766
Angelini GD, Taylor FC, Reeves BC et al (2002) Early and midterm outcome after off-pump and on-pump surgery in beating heart against Cardioplegic Arrest Studies (BHACAS 1 and 2): a pooled analysis of two randomized controlled trials. Lancet 359:1194–1199
Nathoe HM, van Dijk D, Jansen EW et al (2003) A comparison of off-pump and on-pump coronary bypass surgery in low-risk patients. N Engl J Med 348:394–402
Khan NE, De Souza A, Mister R et al (2004) A randomized comparison of off-pump and on-pump multivessel coronary-artery bypass surgery. N Engl J Med 350:21–28
Puskas JD, Williams WH, Duke PG et al (2003) Off-pump coronary artery bypass grafting provides complete revascularization with reduced myocardial injury, transfusion requirements, and length of stay: a prospective randomized comparison of 200 unselected patients undergoing off-pump versus conventional coronary artery bypass grafting. J Thorac Cardiovasc Surg 125:797–808
Légaré JF, Buth KJ, King S et al (2004) Coronary bypass surgery performed off pump does not result in lower in-hospital morbidity than coronary artery bypass grafting performed on pump. Circulation 109:887–892
Chen DC, Bainbridge D, Martin JE et al (2005) Does off-pump coronary artery bypass reduce mortality, morbidity, and resource utilization when compared with conventional coronary artery bypass? A meta-analysis of randomized trials. Anesthesiology 102:188–203
Møller CH, Penninga L, Wetterslev J et al (2008) Clinical outcomes in randomized trials of off- vs. on-pump coronary artery bypass surgery: systematic review with meta-analyses and trial sequential analyses. Eur Heart J 29:2601–2616
Puskas JD, Williams WH, O’Donnell R et al (2011) Off-pump and on-pump coronary artery bypass grafting are associated with similar graft patency, myocardial ischemia, and freedom from re-intervention: long-term follow-up of a randomized trial. Ann Thorac Surg 92:1836–1843
Shroyer AL, Grover FL, Hattler B et al (2009) On-pump versus off-pump coronary-artery bypass surgery. N Engl J Med 361:1827–1837
Lamy A, Devereaux PJ, Prabhakaran D et al (2012) Off-pump or on-pump coronary-artery bypass grafting at 30 days. N Engl J Med 366:1489–1497
Lamy A, Devereaux PJ, Prabhakaran D et al (2013) Off-pump or on-pump coronary-artery bypass grafting at 1 year. N Engl J Med 368:1179–1188
Afilalo J, Rasti M, Ohayon SM et al (2012) Off-pump vs. on-pump coronary artery bypass surgery: an updated meta-analysis and meta-regression of randomized trials. Eur Heart J 33:1257–1267
Møller CH, Perko MJ, Lund JT et al (2010) No major differences in 30-day outcomes in high-risk patients randomized to off-pump versus on-pump coronary bypass surgery. The Best Bypass Surgery Trial Circ 121:498–504
Diegeler A, Börgermann P, Kappert U et al (2013) Off-pump versus on-pump coronary-artery bypass grafting in elderly patients. N Engl J Med 368:1189–1198
Hills LD, Smith PK, Anderson JL et al (2011) ACCF/AHA guideline for coronary artery bypass graft surgery: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 124:e652–e735
Blankstein R, Ward RP, Arnsdorf M et al (2005) Female gender is an independent predictor of operative mortality after coronary artery bypass graft surgery: contemporary analysis of 31 Midwestern hospitals. Circulation 112:1323–1327
Maniar HS, Barner HB, Bailey MS et al (2003) Radial artery patency: are aortocoronary conduits superior to composite grafting? Ann Thorac Surg 76:1498–1504
Caputo M, Dphil BR, Marchetto G et al (2003) Radial versus right internal thoracic artery as a second arterial conduit for coronary surgery: early and midterm outcomes. J Thorac Cardiovasc Surg 126:39–47
Gaudino M, Alessandrini F, Pragliola C et al (2004) Effect of target artery location and severity of stenosis on mid-term patency of aorta-anastomosed versus internal-artery anastomosed radial artery grafts. Eur J Cardio Thorac Surg 25:424–428
Loop FD, Lytle BW, Cosgrove DM, Stewart RW, Goormastic M, William GW, Golding LA, Gill CC, Taylor PC, Sheldon WC (1986) Influence of the internal mammary artery graft on 10-year survival and other cardiac events. N Engl J Med 314:1–6
Lytle BW, Loop FD, Cosgrove DM, Ratliff NB, Easley K, Taylor PC (1985) Long-term (5 to 12 years) serial studies of internal mammary artery and saphenous vein coronary artery bypass grafts. J Thorac Cardiovasc Surg 89:248–258
Pick AW, Orszulak TA, Anderson BJ, Schaff HV (1997) Single versus bilateral internal mammary artery grafts: 10-year outcome analysis. Ann Thorac Surg 64:599–605
Lytle BW, Blackstone EH, Loop FD, Houghtaling PL, Arnold JH, Akhrass R, McCarthy PM, Cosgrove DM (1999) Two internal thoracic arteries are better than one. J Thorac Cardiovasc Surg 117:855–872
Calafiore AM, Di Mauro M, D’Alessandro S, Teodori G, Vitolla G, Contini M, Iacò AL, Spira G (2002) Revascularization of the lateral wall: long-term angiographic and clinical results of radial artery versus right internal thoracic artery grafting. J Thorac Cardiovasc Surg 123:225–231
Lemma M, Gelpi G, Mangini A, Vanelli P, Carro C, Condemi A, Antona C (2001) Myocardial revascularization with multiple arterial grafts: comparison between the radial artery and the right internal thoracic artery. Ann Thorac Surg 71:1969–1973
Kobayashi J (2009) Radial artery as a graft for coronary artery bypass grafting. Circ J 73:1178–1183
Khot UN, Friedman DT, Pettersson G, Smedira NG, Li J, Ellis SG (2004) Radial artery bypass grafts have an increased occurrence of angiographically severe stenosis and occlusion compared with left internal mammary arteries and saphenous vein grafts. Circulation 109:2086–2091
Buxton BF, Raman JS, Ruengsakulrach P, Gordon I, Rosalion A, Bellomo R, Horrigan M, Hare DL (2003) Radial artery patency and clinical outcomes: five-year interim results of a randomized trial. J Thorac Cardiovasc Surg 125:1363–1371
Collins P, Webb CM, Chong CF, Moat NE (2008) Radial artery versus saphenous vein patency randomized trial. Five-year angiographic follow-up. Circulation 117:2859–2864
Desai ND, Naylor CD, Kiss A, Cohen EA, Feder-Elituv R, Miwa S, Radhakrishnan S, Dubbin J, Schwartz L, Fremes SE (2007) Impact of patient and target-vessel characteristics on arterial and venous bypass graft patency. Insight from a randomized trial. Circulation 115:684–691
Nakajima H, Kobayashi J, Funatsu T, Shimahara Y, Kawamura M, Kawamura A, Yagihara T, Kitamura S (2007) Predictive factors for the intermediate-term patency of arterial grafts in aorta no-touch off-pump coronary revascularization. Eur J Cardio Thorac Surg 32:711–717
Puskas JD, Thourani VH, Kilgo P et al (2009) Off-pump coronary artery bypass disproportionately benefits high-risk patients. Ann Thorac Surg 88:1142–1147
Puskas JD, Kilgo PD, Kutner M et al (2007) Off-pump techniques disproportionately benefit women and narrow the gender disparity in outcomes after coronary artery bypass surgery. Circulation 116(Suppl I):I-192–I-199
Kobayashi J, Tashiro T, Ochi M et al (2005) Early outcome of a randomized comparison of off-pump and on-pump multiple arterial coronary revascularization. Circulation 112(Suppl I):I-338–I-343
Takagi H, Tanabashi T, Kawai N, Kato T, Umemoto T (2007) Off-pump coronary artery bypass sacrifices graft patency: meta-analysis of randomized trials. J Thorac Cardiovasc Surg 133:e2–e3
Widimsky P, Straka Z, Stros P et al (2004) One-year coronary bypass graft patency: a randomized comparison between off-pump and on-pump surgery. angiographic results of the PRAGUE-4 trial. Circulation 110:3418–3423
Al-Ruzzeh S, George S, Bustami M et al (2006) Effect of off-pump coronary artery bypass surgery on clinical, angiographic, neurocognitive, and quality of life outcomes: randomised controlled trial. BMJ 332:1365–1368
Lingaas PS, Hol PK, Lundblad R et al (2006) Clinical and Radiologic Outcome of Off-Pump Coronary Surgery at 12 Months Follow-Up: A Prospective Randomized Trial. Ann Thorac Surg 81:2089–2096
Angelini GD, Culliford L, Smith DK et al (2009) Effects of on- and off-pump coronary artery surgery on graft patency, survival, and health-related quality of life: long-term follow-up of 2 randomized controlled trials. J Thorac Cardiovasc Surg 137:295–303
Uva MS, Cavaco S, Oliveira AG et al (2010) Early graft patency after off-pump and on-pump coronary bypass surgery: a prospective randomized study. Eur Heart J 31:2492–2499
Kim KB, Lim C, Lee C et al (2001) Off-pump coronary artery bypass may decrease the patency of saphenous vein grafts. Ann Thorac Surg 72:S1033–S1037
Kurlansky PA (2003) Is there a hypercoagulable state after off-pump coronary artery bypass surgery? What do we know and what can we do? J Thorac Cardiovasc Surg 126:7–10
Vedin J, Antovic A, Ericsson A, Vaage J (2005) Hemostasis in off-pump compared to on-pump coronary artery bypass grafting: a prospective randomized study. Ann Thorac Surg 80:586–593
Kobayashi J, Sasako Y, Bando K et al (2002) Multiple off-pump coronary revascularization with “Aorta No-Touch” technique using composite and sequential methods. Heart Surg Forum 5:114–118
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Kobayashi, J. (2016). Evidence in OPCAB: Critical Appraisal of Recent RCT Based on CABG in Japan. In: Asai, T., Ochi, M., Yokoyama, H. (eds) Off-Pump Coronary Artery Bypass. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54986-4_3
Download citation
DOI: https://doi.org/10.1007/978-4-431-54986-4_3
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-54985-7
Online ISBN: 978-4-431-54986-4
eBook Packages: MedicineMedicine (R0)