Abstract
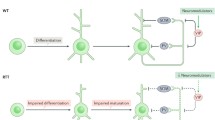
Rett syndrome (RTT) is a devastating neurodevelopmental disorder with autistic features caused by loss-of-function mutations in the gene encoding methyl-CpG-binding protein 2 (MECP2), a transcriptional regulatory protein. RTT has attracted widespread attention not only because of the urgent need for treatments, but also because it has become a window into basic mechanisms underlying epigenetic regulation of neuronal genes, including BDNF. In addition, work in mouse models of the disease has demonstrated the possibility of symptom reversal upon restoration of normal gene function. This latter finding has resulted in a paradigm shift in RTT research and, indeed, in the field of neurodevelopmental disorders as a whole, and spurred the search for potential therapies for RTT and related syndromes. In this context, the discovery that expression of BDNF is dysregulated in RTT and mouse models of the disease has taken on particular importance. This chapter reviews the still evolving story of how MeCP2 might regulate expression of BDNF, the functional consequences of BDNF deficits in Mecp2 mutant mice, and progress in developing BDNF-targeted therapies for the treatment of RTT.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Rett syndrome (RTT) is a complex neurodevelopmental disorder that affects approximately 1 in 10,000 live female births worldwide (Chahrour and Zoghbi 2007). RTT is characterized by apparently normal early postnatal development with neurological symptoms appearing around 6–18 months of age. The subsequent course of the disorder is variable and patients exhibit a diverse array of symptoms that generally includes loss of acquired speech, head growth deceleration, autistic features such as emotional withdrawal and diminished eye contact, motor stereotypies, early hypotonia followed by rigidity, epileptiform seizures, exaggerated responses to stress, and severe respiratory and autonomic (cardiac and gastrointestinal) dysfunction (Chahrour and Zoghbi 2007; Hagberg et al. 1983; Katz et al. 2009; Shahbazian and Zoghbi 2002; Vorsanova et al. 2004; Weese-Mayer et al. 2006, 2008). Up to a quarter of RTT patients may die prematurely of cardiorespiratory failure (Kerr et al. 1997).
The vast majority of typical RTT cases result from loss-of-function mutations in the gene encoding methyl-CpG-binding protein 2 (MeCP2; Amir et al. 1999; Chahrour and Zoghbi 2007), a transcriptional regulatory protein (Klose and Bird 2006). Over 250 different MECP2 mutations have been identified in RTT patients, most of which tend to cluster either within the methyl-binding or transcription repression domains. The MECP2 gene is X-linked, and affected females are heterozygotes and somatic mosaics for MeCP2, i.e., cells in which the mutated allele occurs on the inactive X are phenotypically normal for MeCP2 expression, whereas cells in which the mutated allele occurs on the active X are mutant. Disease phenotype is therefore affected not only by the specific MECP2 mutation but also by the skewing of X chromosome inactivation; individuals in which inactivation is skewed towards the mutant allele are less severely affected, and vice versa. Hemizygosity in males is usually fatal, and the chances of homozygosity in females are exceedingly small, given that most disease-causing mutations arise in the paternal germ line and child-bearing by affected females is extremely rare.
The full scope of MeCP2 function in neurons remains a subject of some controversy. Although it is clear that MeCP2 binds methylated DNA and can potently silence transcription (Klose and Bird 2006), additional functions, including transcriptional activation (Chahrour et al. 2008), regulation of RNA processing (Young et al. 2005), and control of higher order chromatin structure (Georgel et al. 2003), have been proposed. Moreover, it is unclear whether or not MeCP2 selectively regulates transcription of specific genes or, alternatively, acts globally to regulate chromatin state across the genome. A recent study by Skene et al. (2010) demonstrated that MeCP2 protein is abundantly expressed in neurons at levels comparable to histone octamers, i.e., sufficient to blanket the genome at methylated CpG dinucleotides. Therefore, these authors have suggested that the primary function of MeCP2 is to globally repress spurious transcription, e.g., of nucleotide repeats across the genome rather than to dynamically regulate expression of specific genes. However, Skene et al. (2010) showed that, in addition to its widespread binding across genome, MeCP2 also shows peaks of even higher binding at specific sites within promoter regions. Whether or not this is evidence for a more specific role in dynamic regulation of particular genes remains unclear. Nonetheless, what is clear is that expression of many genes is disrupted, either directly or indirectly, by loss-of-function mutations in MECP2 and that the complexity of RTT is related to the diversity of affected gene targets.
2 Regulation of BDNF Expression, Trafficking, and Secretion by MeCP2
The debate concerning the role of MeCP2 in gene regulation is particularly relevant to understanding the evolution of current thinking regarding BDNF and the pathogenesis of RTT. The initial suggestion that dysregulation of BDNF expression might play a role in RTT came from in vitro evidence that BDNF is a transcriptional target of MeCP2 and repressed by MeCP2 binding to BDNF promoter regions. Specifically, Chen et al. (2003) and Martinowich et al. (2003) used chromatin immunoprecipitation to demonstrate binding of MeCP2 protein to BDNF promoter IV (referred to at the time as promoter III), one of nine BDNF promoters and one that is particularly important for activity-dependent regulation of BDNF expression. Moreover, MeCP2 binding appears to recruit a transcriptional repressor complex that includes HDAC1 and Sin3A to the BDNF locus (Martinowich et al. 2003). Chen et al. (2003) and Martinowich et al. (2003) further showed that MeCP2 binding to the BDNF gene was dynamic and subject to regulation in cultured neurons by exposure to depolarizing stimuli, such as elevated potassium chloride (KCl). Specifically, strong chemical depolarization reduces MeCP2 binding to BDNF promoter IV (Martinowich et al. 2003) in association with a change in the phosphorylation state of MeCP2 (Chen et al. 2003), reduces methylation of promoter IV (Martinowich et al. 2003), and increases BDNF expression (see also Ballas et al. 2005). Subsequently, Zhou and colleagues (Zhou et al. 2006) demonstrated that phosphorylation of MeCP2 at serine 421 is particularly important for activity-dependent increases in BDNF expression in cultured hippocampal neurons. Consistent with this repression model, Chen et al. (2003) showed that Mecp2 null embryonic cortical neurons cultured in the presence of blockers of neuronal activity exhibited higher levels of BDNF exon IV mRNA than wild-type cells. However, in the presence of a depolarizing concentration of KCl, wild-type and mutant cells exhibited similar levels of BDNF expression, which the authors interpreted as consistent with BDNF already being derepressed in the mutant cells to levels similar to those achieved in wild-type cells upon stimulation. More recently, evidence has emerged that BDNF expression can also be regulated by the acetylation state of MeCP2 in a manner consistent with the repression model. Specifically, mice lacking functional SIRT1, a nicotinamide-adenine dinucleotide-dependent histone deacetylase, exhibit increased MeCP2 binding to the BDNF exon IV promoter and decreased levels of BDNF mRNA and protein (Zocchi and Sassone-Corsi 2012).
The hypothesis that MeCP2 normally represses BDNF transcription predicted that loss of MeCP2 function in RTT, or mouse models of the disease, would be associated with elevated BDNF expression. However, this prediction has not been borne out, as Mecp2 null or heterozygous mice exhibit reduced levels of BDNF mRNA and protein in vivo (Chang et al. 2006; Ogier et al. 2007; Wang et al. 2006). Similarly, two studies of postmortem brain samples from RTT patients have demonstrated reduced levels of BDNF mRNA (Abuhatzira et al. 2007; Deng et al. 2007). The BDNF mRNA and protein deficits observed in the brain and peripheral nervous system of Mecp2 mutant mice are progressive (Chang et al. 2006; Ogier et al. 2007; Wang et al. 2006), being virtually undetectable at birth and declining to as much as 50 % of wild-type levels in some tissues by 5 weeks of age in male nulls (Wang et al. 2006). Moreover, the postnatal decline in BDNF levels occurs with a slower time course in heterozygous females compared to male nulls (Schmid et al. 2012). Clearly, these in vivo data are inconsistent with a model in which MeCP2 simply represses expression of BDNF.
Various hypotheses have been offered to explain how loss of MeCP2 function could lead to deficits in BDNF expression. One idea, already introduced above, is that MeCP2 activates rather than represses gene expression. In support of this hypothesis, Chahrour et al. (2008) showed that global overexpression of MeCP2 in mice is associated with increased expression of BDNF mRNA in the hypothalamus, whereas MeCP2 loss is associated with decreased BDNF. Similarly, selective deletion of Mecp2 from Sim-1-positive neurons also causes a reduction in BDNF in the hypothalamus (Fyffe et al. 2008). The activator hypothesis is also supported by a recent report by Li et al. (2013) demonstrating global reductions in transcription and Akt/mTOR-dependent protein translation—including BDNF—in human iPSC-derived neurons in which the Mecp2 gene was deleted using TALEN-mediated DNA editing. One caveat to these findings is that the possible contribution of decreased BDNF mRNA and/or protein stability, rather than decreased gene transcription per se, has not been ruled out. Further support for the activator model comes from studies showing that derepression of microRNA (miRNA)-mediated inhibition of MeCP2 translation in cultured neurons increases expression not only of MeCP2 but BDNF as well (Klein et al. 2007).
A recent approach to resolving the repressor versus activator debate is the “dual operation model” (Li and Pozzo-Miller 2013). This model is motivated by data from one study showing that either knockdown or overexpression of MeCP2 in cultured neurons leads to increased expression of BDNF (Larimore et al. 2009), as well as evidence that MeCP2 can undergo diverse posttranslational modifications, including phosphorylation, acetylation, and ubiquitylation, leading to unique associations with either co-repressors or co-activators (Gonzales et al. 2012).
A second hypothesis that has been proposed to explain decreased BDNF expression in the absence of MeCP2 function is that MeCP2 normally represses the activity of repressors of BDNF expression, i.e., the RE1 silencing transcription factor (REST)/CoREST complex (Abuhatzira et al. 2007). This model is based on data from mice and humans demonstrating elevated levels of REST/CoREST in RTT patients and Mecp2-deficient mice, presumably leading to reduced BDNF expression through repressive interactions with the RE1 element in BDNF promoter regions. A third hypothesis is that reduced BDNF expression in Mecp2 null neurons is a consequence of reduced neuronal activity (Sun and Wu 2006). This idea was based on the finding that cortical neurons from Mecp2 null mice exhibit reduced firing rates associated with a loss of excitatory synaptic drive (Dani et al. 2005). However, we found that even after exposure to strongly depolarizing stimuli in vitro, Mecp2 null cells express less BDNF protein than wild-type, indicating that differences in activity alone are unlikely to account for BDNF deficits in the absence of MeCP2 (Ogier et al. 2007). Thus, at present, the normal role of MeCP2 in regulating BDNF expression, as well as the mechanism(s) responsible for reduced BDNF levels in the RTT brain, remain to be clarified. One possibility is that, although loss of MeCP2 may result in derepression of BDNF gene expression, translation and/or stability of the protein may also be adversely affected, resulting in a net decrease in BDNF levels in the RTT brain. In support of this possibility, Wu et al. (2010) recently demonstrated that MeCP2 controls transcription of several microRNAs (miRNAs) that target the 3′ UTR of Bdnf mRNA, some of which are upregulated in the absence of MeCP2 function and negatively regulate Bdnf mRNA levels. Conversely, inhibition of two such miRNAs, miR-381 and miR-495, in both wild-type and Mecp2 null neurons in vitro, increased levels of Bdnf mRNA and BDNF protein. Thus, Wu et al. (2010) proposed that, in the absence of MeCP2 function, the net effect of direct derepression of Bdnf mRNA, combined with depression of miRNAs that negatively regulate Bdnf mRNA, is reduced BDNF levels. This hypothesis requires further testing, as Wu et al. (2010) also identified miRNAs that target Bdnf mRNA and are downregulated in the absence of MeCP2 function. In particular, it will be critical to define the stoichiometry of these positive and negative influences on Bdnf transcription, translation, and stability in vivo in order to fully understand the role of miRNAs in BDNF protein deficits in RTT.
In addition to dysregulation of BDNF expression, loss of MeCP2 also appears to disrupt regulated secretion and transport of BDNF. Although mature sensory neurons lacking MeCP2 express lower levels of BDNF protein, they actually secrete a larger proportion of their total BDNF content than wild-type cells, at least in cell culture (Ladas et al. 2009). However, this enhanced secretion is not sufficient to completely compensate for reduced levels of BDNF expression, and the absolute amount of BDNF released by mutant cells is nonetheless lower than wild-type. This is also seen at mossy fiber inputs onto CA3 pyramidal neurons in Mecp2 null mice, in which activity-dependent BDNF release is reduced compared to wild type, resulting in reduced activation of TrkB and reduced signaling through TRPC3 channels (Li et al. 2012). On the other hand, in newborn Mecp2 null neurons, which do not yet exhibit a significant deficit in BDNF expression, the absolute amount of BDNF released is actually greater than wild-type (Wang et al. 2006). These data raise the possibility that during early development, enhanced secretion of BDNF from Mecp2 null cells could derange developmental processes that depend on tight coupling between neuronal activity and BDNF release, such as activity-dependent refinement of synaptic connections (Lein and Shatz 2000). Enhanced BDNF release appears to be just one manifestation of a more widespread dense core vesicle phenotype in Mecp2 null mice. Studies of catecholamine release in Mecp2 null adrenal chromaffin cells demonstrated that the readily releasable pool of dense core vesicles is significantly larger and individual vesicles are more fusigenic than in wild-type cells, resulting in hypersecretion of epinephrine (Ladas et al. 2009; Wang et al. 2006). Given that BDNF is also a dense core vesicle cargo (Decker et al. 2010; Farhadi et al. 2000; Luo et al. 2001; Salio et al. 2007; Wu et al. 2004), similar mechanisms may underlie the BDNF secretory phenotype in Mecp2 null mice.
Recent studies indicate that BDNF signaling in Mecp2 mutants is also impacted by deficits in axonal transport, resulting from dysregulation of huntingtin (Htt)- and huntingtin-associated protein (Hap1)-dependent vesicle trafficking (Roux et al. 2012). Specifically, the velocity of vesicular BDNF transport in cortico-striatal projection neurons is impaired by loss of MeCP2. Given the importance of cortically derived BDNF for the maintenance of striatal medium-spiny neurons (Baquet et al. 2004), these data raise the possibility that deficits in BDNF transport from the cortex contribute to striatal pathology in RTT (cf., Stearns et al. 2007).
3 Topography of BDNF Deficits in Mouse Models of RTT
The time course and distribution of BDNF deficits resulting from loss of MeCP2 have been studied in some detail in Mecp2 null and heterozygous mice (Chang et al. 2006; Ogier et al. 2007; Wang et al. 2006; Deogracias et al. 2012). The earliest and most dramatic deficits in BDNF mRNA and protein occur in the vagal sensory nodose ganglion (NG), followed by the brainstem, cerebellum, and cortex (Chang et al. 2006; Ogier et al. 2007; Wang et al. 2006). In NG sensory neurons, for example, BDNF mRNA and protein levels fall to approximately 50 % wild-type values within 5 weeks after birth (Ogier et al. 2007), leading to synaptic dysfunction in vagal afferent inputs to the brainstem (see below). Within the brain, the effect of MeCP2 loss on BDNF levels is not uniform across cell groups. For example, although Mecp2 null mutants exhibit marked decreases in BDNF immunostaining in the neuropil of some brainstem nuclei, such as the nucleus tractus solitarius (nTS), nucleus ambiguus, and nucleus locus coeruleus (LC), others, such as the gracile and principal sensory trigeminal nuclei, are only mildly affected or unchanged (Kline et al. 2010). Mechanisms that underlie the differential temporal and spatial patterns of BDNF decline in the Mecp2 mutant brain have not been defined. Recent data indicate that regional BDNF deficits in Mecp2 null mutants are accompanied by reduced levels of TrkB phosphorylation without a change in total TrkB expression (Schmid et al. 2012).
4 BDNF Deficits in Mouse Models of RTT: Functional Consequences
With a few exceptions, relatively little is known about the specific functional consequences of reduced BDNF expression in Mecp2 mutants and RTT patients. Morphologic and synaptic phenotypes observed in the brains of RTT patients and/or Mecp2 null mutants, including decreased brain weight and neuronal size, reduced dendritic arborizations and impaired hippocampal long-term potentiation (reviewed in Chahrour and Zoghbi 2007), overlap with deficits seen in Bdnf loss-of-function mutants (Chang et al. 2006; Huang and Reichardt 2001). In addition, at least some of the behavioral features of Mecp2 mutant mice, including irregular breathing and impaired locomotion, overlap to some degree with deficits observed in Bdnf mutants (Conover et al. 1995; Erickson et al. 1996). Moreover, genetic overexpression of BDNF in Mecp2 null mutants can improve survival and locomotor function, whereas BDNF deletion hastens the onset of symptoms (Chang et al. 2006). However, few studies have examined how reduced BDNF availability in identified neural circuits is linked to specific functional deficits in RTT. What is clear is that because BDNF declines postnatally in Mecp2 mutants, the size of neuronal populations that depend on BDNF for survival before birth is unaffected (Wang et al. 2006). Therefore, increasing attention has focused on the role of BDNF deficits in the maturation and function of the RTT brain after birth.
4.1 MeCP2 and Stimulation of Dendritic Growth by BDNF
MeCP2 plays a key role in mediating the effects of environmental stimuli, such as neuronal depolarization, on expression of genes required for neuronal maturation, including BDNF (Cohen et al. 2011; Ebert et al. 2013). For example, Zhou et al. (2006) demonstrated that phosphorylation of MeCP2 at serine 421 (ser421) is required for activity-dependent expression of BDNF in postnatal hippocampal neurons. BDNF, in turn, can stimulate ser421 phosphorylation of MeCP2, indicating that BDNF functions both upstream and downstream of MeCP2. MeCP2 phosphorylation at ser421 is also required for expression of mature dendritic morphologies in hippocampal neurons (Chapleau et al. 2009; Zhou et al. 2006), possibly by activating this BDNF signaling loop. In support of this possibility, overexpression of BDNF can reverse dendritic atrophy in hippocampal neurons that are null for Mecp2 (Larimore et al. 2009).
4.2 BDNF and Synaptic Dysfunction in RTT
The potential synaptic consequences of BDNF loss have been studied in detail at primary afferent synapses between NG primary sensory neurons and second order neurons in the nTS. These synapses are the first site at which peripheral visceral sensory inputs impinge on central autonomic reflex pathways and thereby play a critical role in autonomic functions disrupted in RTT, such as respiratory, cardiovascular, and gastrointestinal homeostasis. Normally, BDNF plays a sensory gating function at these synapses by modulating postsynaptic responses to glutamate, the primary excitatory transmitter of visceral afferent neurons (Balkowiec et al. 2000). We hypothesized, therefore, that in Mecp2 null mice, decreased BDNF expression in NG sensory neurons would be associated with a deficit in modulation of fast glutamatergic transmission at primary afferent synapses in nTS. Indeed, the amplitudes of spontaneous miniature and evoked EPSCs in nTS neurons are significantly increased in Mecp2 null mice (Kline et al. 2010; Kron et al. 2012a), and accordingly, mutant cells are more likely than wild-type to fire action potentials in response to primary afferent stimulation (Kline et al. 2010). These changes occur without any increase in intrinsic neuronal excitability and are unaffected by blockade of inhibitory GABA currents. A prediction of these results is that autonomic reflexes mediated by primary afferent inputs to nTS would be disinhibited in the absence of MeCP2 function. This prediction has been borne out by studies demonstrating that the hypoxic ventilatory response, a reflex mediated by primary chemoafferent inputs to nTS, is markedly exaggerated in Mecp2 null mice compared to wild-type controls (Bissonnette and Knopp 2006; Roux et al. 2008; Voituron et al. 2009). Similarly, Mecp2 nulls exhibit a loss of habituation in the Breuer–Hering reflex, an nTS-mediated behavior that plays an essential role in regulating the post-inspiratory phase of the respiratory cycle (Stettner et al. 2007). More generally, these findings suggest that reduced sensory gating in nTS contributes to cardiorespiratory instability in RTT and that nTS is a site at which restoration of normal BDNF signaling could help to reestablish normal homeostatic controls. Indeed, exaggerated synaptic responses to primary afferent input in nTS are reversed by application of exogenous BDNF to brainstem slices in vitro (Kline et al. 2010). Moreover, respiratory function in vivo is improved by treatments that enhance BDNF/TrkB signaling in Mecp2 mutants (see below).
4.3 BDNF and Hypothalamic Dysfunction in RTT
Feeding behavior and energy homeostasis are strongly influenced by BDNF/TrkB signaling in the hypothalamus (Noble et al. 2011; Rios et al. 2001). Specifically, increased levels of BDNF are associated with cessation of feeding and increased energy expenditure. Although the specific circuitry underlying the role of BDNF in feeding has not been completely defined, BDNF has been identified as a downstream effector of melanocortin-4 receptor (MC4R) signaling in the ventromedial hypothalamus (Noble et al. 2011; Xu et al. 2003), a key site for regulating feeding and satiety. Fyffe et al. (2008) demonstrated that loss of Mecp2 by Cre-mediated deletion specifically within Sim-1 expressing neurons in the hypothalamus results in reduced BDNF levels in Mecp2 null neurons in the paraventricular nucleus, also a site of MC4R expression (Nicholson et al. 2007), as well as hyperphagia and obesity. Although the relevance of the obesity phenotype to RTT is unclear, these data provide further evidence that MeCP2 is required for maintaining normal levels of BDNF expression and metabolic homeostasis.
5 BDNF-Targeted Therapies for RTT
Recent studies in conditional Mecp2 null mice have demonstrated that reactivation of the Mecp2 gene, even in severely symptomatic animals, can rescue neurologic function to a remarkable degree (Guy et al. 2007). These findings indicate that deficits caused by loss of MeCP2 function are not due to irreversible changes in brain structure or function. In addition, as noted above, genetic overexpression of the BDNF gene in Mecp2 null mice improves somatomotor function and prolongs life span (Chang et al. 2006), and exogenous BDNF can reverse synaptic deficits caused by MeCP2 deficiency (Kline et al. 2010). Together, these findings raise the possibility of rescuing neurologic function in Mecp2 null mice and, eventually, RTT patients, by pharmacologic therapies that enhance BDNF/TrkB signaling. BDNF itself does not have good drug-like characteristics, i.e., limited half-life and poor blood–brain barrier penetration, thus motivating the search for alternative approaches to increasing BDNF/TrkB signaling in RTT. As discussed below, these approaches include enhancing expression of endogenous BDNF, increasing BDNF trafficking, and directly activating the TrkB receptor.
5.1 Increasing Expression or Delivery of Endogenous BDNF
In the first test of a BDNF-targeted therapeutic strategy, Ogier et al. (2007) examined whether or not pharmacologic elevation of endogenous BDNF expression with ampakine drugs could improve respiratory function in Mecp2 null mice. Ampakines are benzamide derivatives that facilitate the activity of glutamatergic AMPA receptors and thereby increase expression of activity-dependent genes, including BDNF (Lynch and Gall 2006). Repeated administration of ampakines in rats and mice increases expression of BDNF mRNA and protein in the forebrain for several days (Lauterborn et al. 2003; Rex et al. 2006) and augments BDNF-dependent synaptic function (Ingvar et al. 1997; Porrino et al. 2005; Rex et al. 2006). Indeed, treatment of Mecp2 null mutants with the ampakine CX546 for 3 days significantly increases BDNF levels in NG sensory neurons and reverses the respiratory tachypnea that is a prominent feature of breathing dysfunction in RTT (Ogier et al. 2007). Although additional studies are required to elucidate the mechanism of ampakine action in this model, these data are consistent with the hypothesis that BDNF deficits contribute to the respiratory phenotype of Mecp2 null mice and that BDNF signaling may be a pharmacological target for improving respiratory function in RTT. More recently, Deogracias et al. (2012) showed that fingolimod, a sphingosine-1 phosphate receptor agonist used to treat multiple sclerosis, increases BDNF in cultured neurons and protects against NMDA-induced neuronal death in a BDNF-dependent manner. In vivo, treatment of Mecp2 mutant mice partially reversed BDNF deficits and also increased striatal volume, an index of BDNF signaling. Treated mice also showed improvement in locomotor behavior, a clinically relevant outcome measure for RTT patients. Finally, it is well known that BDNF expression in the rodent forebrain can be increased by environmental enrichment and exercise (cf., Cotman and Berchtold 2002). Indeed, rearing Mecp2 mutant mice in an enriched environment, particularly at early stages of postnatal development, leads to improvements in motor and spatial learning, coordination, and anxiety, as well as hippocampal circuit function, that correlate well with increases in BDNF expression (Kondo et al. 2008; Lonetti et al. 2010).
Another potential strategy for enhancing BDNF/TrkB signaling in RTT is to increase the bioavailability of endogenous BDNF by promoting increased axonal transport and/or secretion. Recently, Roux et al. (2012) showed that cysteamine, a drug that increases vesicular trafficking of BDNF (Borrell-Pages et al. 2006), extends life span and improves motor function in Mecp2 mutant mice.
5.2 Targeting the BDNF Receptor, TrkB
One potential limitation of pharmacologic approaches that globally increase BDNF is that BDNF activates receptors other than TrkB, including p75. The properties of BDNF binding to p75 as well as functioning as a full agonist at TrkB could lead to unwanted pleiotropic effects of elevated BDNF levels. An alternative approach is to directly activate TrkB; potential strategies include TrkB activating antibodies (Qian et al. 2006) and small molecules that function as direct TrkB ligands (Jang et al. 2010; Massa et al. 2010; Xie and Longo 2000). Our laboratory has recently examined the ability of a small molecule, non-peptide BDNF loop 2 domain mimetic, LM22A-4, which functions as a direct and specific partial agonist of TrkB, but not p75 (Massa et al. 2010), to increase TrkB activation and improve breathing in Mecp2 mutant mice. LM22A-4 was developed by Longo, Massa, and colleagues by in silico screening for mimetics of BDNF loop domains that selectively activate TrkB and downstream signaling partners in vitro and in vivo (Han et al. 2012; Massa et al. 2010; Schmid et al. 2012). Recent studies in our laboratory have shown that LM22A-4 (1) reduces synaptic hyperexcitability in the brainstem respiratory network in brain slice preparations (Kron et al. 2012b), (2) reverses deficits in TrkB activation in the brainstem (Schmid et al. 2012), and (3) significantly improves respiratory function (Schmid et al. 2012), including the elimination of apneic breathing (Kron et al. 2012b), following systemic administration to symptomatic Mecp2 null and heterozygous mice. Together, these data provide direct evidence linking TrkB signaling to respiratory dysfunction in mouse models of RTT and further highlight the therapeutic potential of strategies aimed at enhancing BDNF/TrkB signaling for the treatment of RTT patients.
6 Summary
BDNF is only one of many genes whose expression is dysregulated in RTT (Chahrour et al. 2008). Nonetheless, given the multiplicity of roles played by BDNF signaling in brain maturation and neural circuit function across the life span, it is not surprising that deficits in BDNF protein levels have now been linked, either directly or indirectly, to diverse neurologic deficits in RTT, including reduced dendritic growth, breathing dysfunction, and impaired locomotion. Certainly, much more work is required to understand how BDNF deficits may contribute to the expression of specific RTT endophenotypes. It is encouraging, however, that the possibility of treating RTT using BDNF/TrkB-targeted therapies has already been established in principle in mouse models of the disease.
Abbreviations
- MECP2, BDNF :
-
Human gene
- Mecp2, Bdnf :
-
Mouse gene
- MeCP2, BDNF:
-
Protein
References
Abuhatzira L, Makedonski K, Kaufman Y, Razin A, Shemer R (2007) MeCP2 deficiency in the brain decreases BDNF levels by REST/CoREST-mediated repression and increases TRKB production. Epigenetics 2:214–222
Amir RE, Van den Veyver IB, Wan M, Tran CQ, Francke U, Zoghbi HY (1999) Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet 23:185–188
Balkowiec A, Kunze DL, Katz DM (2000) Brain-derived neurotrophic factor acutely inhibits AMPA-mediated currents in developing sensory relay neurons. J Neurosci 20:1904–1911
Ballas N, Grunseich C, Lu DD, Speh JC, Mandel G (2005) REST and its corepressors mediate plasticity of neuronal gene chromatin throughout neurogenesis. Cell 121:645–657
Baquet ZC, Gorski JA, Jones KR (2004) Early striatal dendrite deficits followed by neuron loss with advanced age in the absence of anterograde cortical brain-derived neurotrophic factor. J Neurosci 24:4250–4258
Bissonnette JM, Knopp SJ (2006) Separate respiratory phenotypes in methyl-CpG-binding protein 2 (Mecp2) deficient mice. Pediatr Res 59:513–518
Borrell-Pages M, Canals JM, Cordelieres FP, Parker JA, Pineda JR, Grange G, Bryson EA, Guillermier M, Hirsch E, Hantraye P, Cheetham ME, Neri C, Alberch J, Brouillet E, Saudou F, Humbert S (2006) Cystamine and cysteamine increase brain levels of BDNF in Huntington disease via HSJ1b and transglutaminase. J Clin Invest 116:1410–1424
Chahrour M, Zoghbi HY (2007) The story of Rett syndrome: from clinic to neurobiology. Neuron 56:422–437
Chahrour M, Jung SY, Shaw C, Zhou X, Wong ST, Qin J, Zoghbi HY (2008) MeCP2, a key contributor to neurological disease, activates and represses transcription. Science 320:1224–1229
Chang Q, Khare G, Dani V, Nelson S, Jaenisch R (2006) The disease progression of Mecp2 mutant mice is affected by the level of BDNF expression. Neuron 49:341–348
Chapleau CA, Calfa GD, Lane MC, Albertson AJ, Larimore JL, Kudo S, Armstrong DL, Percy AK, Pozzo-Miller L (2009) Dendritic spine pathologies in hippocampal pyramidal neurons from Rett syndrome brain and after expression of Rett-associated MECP2 mutations. Neurobiol Dis 35:219–233
Chen WG, Chang Q, Lin Y, Meissner A, West AE, Griffith EC, Jaenisch R, Greenberg ME (2003) Derepression of BDNF transcription involves calcium-dependent phosphorylation of MeCP2. Science 302:885–889
Cohen S, Gabel HW, Hemberg M, Hutchinson AN, Sadacca LA, Ebert DH, Harmin DA, Greenberg RS, Verdine VK, Zhou Z, Wetsel WC, West AE, Greenberg ME (2011) Genome-wide activity-dependent MeCP2 phosphorylation regulates nervous system development and function. Neuron 72:72–85
Conover JC, Erickson JT, Katz DM, Bianchi LM, Poueymirou WT, McClain J, Pan L, Helgren M, Ip NY, Boland P et al (1995) Neuronal deficits, not involving motor neurons, in mice lacking BDNF and/or NT4. Nature 375:235–238
Cotman CW, Berchtold NC (2002) Exercise: a behavioral intervention to enhance brain health and plasticity. Trends Neurosci 25:295–301
Dani VS, Chang Q, Maffei A, Turrigiano GG, Jaenisch R, Nelson SB (2005) Reduced cortical activity due to a shift in the balance between excitation and inhibition in a mouse model of Rett syndrome. Proc Natl Acad Sci U S A 102:12560–12565
Decker H, Lo KY, Unger SM, Ferreira ST, Silverman MA (2010) Amyloid-beta peptide oligomers disrupt axonal transport through an NMDA receptor-dependent mechanism that is mediated by glycogen synthase kinase 3beta in primary cultured hippocampal neurons. J Neurosci 30:9166–9171
Deng V, Matagne V, Banine F, Frerking M, Ohliger P, Budden S, Pevsner J, Dissen GA, Sherman LS, Ojeda SR (2007) FXYD1 is an MeCP2 target gene overexpressed in the brains of Rett syndrome patients and Mecp2-null mice. Hum Mol Genet 16:640–650
Deogracias R, Yazdani M, Dekkers MP, Guy J, Ionescu MC, Vogt KE, Barde YA (2012) Fingolimod, a sphingosine-1 phosphate receptor modulator, increases BDNF levels and improves symptoms of a mouse model of Rett syndrome. Proc Natl Acad Sci U S A 109:14230–14235
Ebert DH, Gabel HW, Robinson ND, Kastan NR, Hu LS, Cohen S, Navarro AJ, Lyst MJ, Ekiert R, Bird AP, Greenberg ME (2013) Activity-dependent phosphorylation of MeCP2 threonine 308 regulates interaction with NCoR. Nature 499:341–345
Erickson JT, Conover JC, Borday V, Champagnat J, Barbacid M, Yancopoulos G, Katz DM (1996) Mice lacking brain-derived neurotrophic factor exhibit visceral sensory neuron losses distinct from mice lacking NT4 and display a severe developmental deficit in control of breathing. J Neurosci 16:5361–5371
Farhadi HF, Mowla SJ, Petrecca K, Morris SJ, Seidah NG, Murphy RA (2000) Neurotrophin-3 sorts to the constitutive secretory pathway of hippocampal neurons and is diverted to the regulated secretory pathway by coexpression with brain-derived neurotrophic factor. J Neurosci 20:4059–4068
Fyffe SL, Neul JL, Samaco RC, Chao HT, Ben-Shachar S, Moretti P, McGill BE, Goulding EH, Sullivan E, Tecott LH, Zoghbi HY (2008) Deletion of Mecp2 in Sim1-expressing neurons reveals a critical role for MeCP2 in feeding behavior, aggression, and the response to stress. Neuron 59:947–958
Georgel PT, Horowitz-Scherer RA, Adkins N, Woodcock CL, Wade PA, Hansen JC (2003) Chromatin compaction by human MeCP2. Assembly of novel secondary chromatin structures in the absence of DNA methylation. J Biol Chem 278:32181–32188
Gonzales ML, Adams S, Dunaway KW, LaSalle JM (2012) Phosphorylation of distinct sites in MeCP2 modifies cofactor associations and the dynamics of transcriptional regulation. Mol Cell Biol 32:2894–2903
Guy J, Gan J, Selfridge J, Cobb S, Bird A (2007) Reversal of neurological defects in a mouse model of Rett syndrome. Science 315:1143–1147
Hagberg B, Aicardi J, Dias K, Ramos O (1983) A progressive syndrome of autism, dementia, ataxia, and loss of purposeful hand use in girls: Rett’s syndrome: report of 35 cases. Ann Neurol 14:471–479
Han J, Pollak J, Yang T, Siddiqui MR, Doyle KP, Taravosh-Lahn K, Cekanaviciute E, Han A, Goodman JZ, Jones B, Jing D, Massa SM, Longo FM, Buckwalter MS (2012) Delayed administration of a small molecule tropomyosin-related kinase B ligand promotes recovery after hypoxic-ischemic stroke. Stroke 43:1918–1924
Huang EJ, Reichardt LF (2001) Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci 24:677–736
Ingvar M, Ambros-Ingerson J, Davis M, Granger R, Kessler M, Rogers GA, Schehr RS, Lynch G (1997) Enhancement by an ampakine of memory encoding in humans. Exp Neurol 146:553–559
Jang SW, Liu X, Yepes M, Shepherd KR, Miller GW, Liu Y, Wilson WD, Xiao G, Blanchi B, Sun YE, Ye K (2010) A selective TrkB agonist with potent neurotrophic activities by 7,8-dihydroxyflavone. Proc Natl Acad Sci U S A 107:2687–2692
Katz DM, Dutschmann M, Ramirez JM, Hilaire G (2009) Breathing disorders in Rett syndrome: progressive neurochemical dysfunction in the respiratory network after birth. Respir Physiol Neurobiol 168:101–108
Kerr AM, Armstrong DD, Prescott RJ, Doyle D, Kearney DL (1997) Rett syndrome: analysis of deaths in the British survey. Eur Child Adolesc Psychiatry 6(Suppl 1):71–74
Klein ME, Lioy DT, Ma L, Impey S, Mandel G, Goodman RH (2007) Homeostatic regulation of MeCP2 expression by a CREB-induced microRNA. Nat Neurosci 10:1513–1514
Kline DD, Ogier M, Kunze DL, Katz DM (2010) Exogenous brain-derived neurotrophic factor rescues synaptic dysfunction in Mecp2-null mice. J Neurosci 30:5303–5310
Klose RJ, Bird AP (2006) Genomic DNA methylation: the mark and its mediators. Trends Biochem Sci 31:89–97
Kondo M, Gray LJ, Pelka GJ, Christodoulou J, Tam PP, Hannan AJ (2008) Environmental enrichment ameliorates a motor coordination deficit in a mouse model of Rett syndrome – Mecp2 gene dosage effects and BDNF expression. Eur J Neurosci 27:3342–3350
Kron M, Howell CJ, Adams IT, Ransbottom M, Christian D, Ogier M, Katz DM (2012a) Brain activity mapping in Mecp2 mutant mice reveals functional deficits in forebrain circuits, including key nodes in the default mode network, that are reversed with ketamine treatment. J Neurosci 32:13860–13872
Kron M, Adams IT, Longo FM, Katz DM (2012b) A novel TrkB agonist eliminates apneic breathing and decreases synaptic hyperexcitability in brainstem respiratory nuclei in Mecp2 mutant mice. In: Society for Neuroscience Abstracts, 246.14
Ladas T, Chan SA, Ogier M, Smith C, Katz DM (2009) Enhanced dense core granule function and adrenal hypersecretion in a mouse model of Rett syndrome. Eur J Neurosci 30:602–610
Larimore JL, Chapleau CA, Kudo S, Theibert A, Percy AK, Pozzo-Miller L (2009) Bdnf overexpression in hippocampal neurons prevents dendritic atrophy caused by Rett-associated MECP2 mutations. Neurobiol Dis 34:199–211
Lauterborn JC, Truong GS, Baudry M, Bi X, Lynch G, Gall CM (2003) Chronic elevation of brain-derived neurotrophic factor by ampakines. J Pharmacol Exp Ther 307:297–305
Lein ES, Shatz CJ (2000) Rapid regulation of brain-derived neurotrophic factor mRNA within eye-specific circuits during ocular dominance column formation. J Neurosci 20:1470–1483
Li W, Pozzo-Miller L (2013) BDNF deregulation in Rett syndrome. Neuropharmacology. doi:pii: S0028-3908(13)00120-2.10.1016 [Epub ahead of print]
Li W, Calfa G, Larimore J, Pozzo-Miller L (2012) Activity-dependent BDNF release and TRPC signaling is impaired in hippocampal neurons of Mecp2 mutant mice. Proc Natl Acad Sci U S A 109:17087–17092
Li Y, Wang H, Muffat J, Cheng AW, Orlando DA, Loven J, Kwok SM, Feldman DA, Bateup HS, Gao Q, Hockemeyer D, Mitalipova M, Lewis CA, Vander Heiden MG, Sur M, Young RA, Jaenisch R (2013) Global transcriptional and translational repression in human-embryonic-stem-cell-derived Rett syndrome neurons. Cell Stem Cell 13:446–458
Lonetti G, Angelucci A, Morando L, Boggio EM, Giustetto M, Pizzorusso T (2010) Early environmental enrichment moderates the behavioral and synaptic phenotype of MeCP2 null mice. Biol Psychiatry 67:657–665
Luo XG, Rush RA, Zhou XF (2001) Ultrastructural localization of brain-derived neurotrophic factor in rat primary sensory neurons. Neurosci Res 39:377–384
Lynch G, Gall CM (2006) Ampakines and the threefold path to cognitive enhancement. Trends Neurosci 29:554–562
Martinowich K, Hattori D, Wu H, Fouse S, He F, Hu Y, Fan G, Sun YE (2003) DNA methylation-related chromatin remodeling in activity-dependent BDNF gene regulation. Science 302:890–893
Massa SM, Yang T, Xie Y, Shi J, Bilgen M, Joyce JN, Nehama D, Rajadas J, Longo FM (2010) Small molecule BDNF mimetics activate TrkB signaling and prevent neuronal degeneration in rodents. J Clin Invest 120:1774–1785
Nicholson JR, Peter JC, Lecourt AC, Barde YA, Hofbauer KG (2007) Melanocortin-4 receptor activation stimulates hypothalamic brain-derived neurotrophic factor release to regulate food intake, body temperature and cardiovascular function. J Neuroendocrinol 19:974–982
Noble EE, Billington C, Kotz CM, Wang C (2011) The lighter side of BDNF. Am J Physiol Regul Integr Comp Physiol 300(5):R1053–R1069
Ogier M, Wang H, Hong E, Wang Q, Greenberg ME, Katz DM (2007) Brain-derived neurotrophic factor expression and respiratory function improve after ampakine treatment in a mouse model of Rett syndrome. J Neurosci 27:10912–10917
Porrino LJ, Daunais JB, Rogers GA, Hampson RE, Deadwyler SA (2005) Facilitation of task performance and removal of the effects of sleep deprivation by an ampakine (CX717) in nonhuman primates. PLoS Biol 3:e299
Qian MD, Zhang J, Tan XY, Wood A, Gill D, Cho S (2006) Novel agonist monoclonal antibodies activate TrkB receptors and demonstrate potent neurotrophic activities. J Neurosci 26:9394–9403
Rex CS, Lauterborn JC, Lin CY, Kramar EA, Rogers GA, Gall CM, Lynch G (2006) Restoration of long-term potentiation in middle-aged hippocampus after induction of brain-derived neurotrophic factor. J Neurophysiol 96:677–685
Rios M, Fan G, Fekete C, Kelly J, Bates B, Kuehn R, Lechan RM, Jaenisch R (2001) Conditional deletion of brain-derived neurotrophic factor in the postnatal brain leads to obesity and hyperactivity. Mol Endocrinol 15:1748–1757
Roux JC, Dura E, Villard L (2008) Tyrosine hydroxylase deficit in the chemoafferent and the sympathoadrenergic pathways of the Mecp2 deficient mouse. Neurosci Lett 447:82–86
Roux JC, Zala D, Panayotis N, Borges-Correia A, Saudou F, Villard L (2012) Modification of Mecp2 dosage alters axonal transport through the Huntingtin/Hap1 pathway. Neurobiol Dis 45:786–795
Salio C, Averill S, Priestley JV, Merighi A (2007) Costorage of BDNF and neuropeptides within individual dense-core vesicles in central and peripheral neurons. Dev Neurobiol 67:326–338
Schmid DA, Yang T, Ogier M, Adams I, Mirakhur Y, Wang Q, Massa SM, Longo FM, Katz DM (2012) A TrkB small molecule partial agonist rescues TrkB phosphorylation deficits and improves respiratory function in a mouse model of Rett syndrome. J Neurosci 32:1803–1810
Shahbazian MD, Zoghbi HY (2002) Rett syndrome and MeCP2: linking epigenetics and neuronal function. Am J Hum Genet 71:1259–1272
Skene PJ, Illingworth RS, Webb S, Kerr AR, James KD, Turner DJ, Andrews R, Bird AP (2010) Neuronal MeCP2 is expressed at near histone-octamer levels and globally alters the chromatin state. Mol Cell 37:457–468
Stearns NA, Schaevitz LR, Bowling H, Nag N, Berger UV, Berger-Sweeney J (2007) Behavioral and anatomical abnormalities in Mecp2 mutant mice: a model for Rett syndrome. Neuroscience 146:907–921
Stettner GM, Huppke P, Brendel C, Richter DW, Gartner J, Dutschmann M (2007) Breathing dysfunctions associated with impaired control of postinspiratory activity in Mecp2-/y knockout mice. J Physiol 579:863–876
Sun YE, Wu H (2006) The ups and downs of BDNF in Rett syndrome. Neuron 49:321–323
Voituron N, Zanella S, Menuet C, Dutschmann M, Hilaire G (2009) Early breathing defects after moderate hypoxia or hypercapnia in a mouse model of Rett syndrome. Respir Physiol Neurobiol 168:109–118
Vorsanova SG, Iourov IY, Yurov YB (2004) Neurological, genetic and epigenetic features of Rett syndrome. J Pediatr Neurol 2:179–190
Wang H, Chan SA, Ogier M, Hellard D, Wang Q, Smith C, Katz DM (2006) Dysregulation of brain-derived neurotrophic factor expression and neurosecretory function in Mecp2 null mice. J Neurosci 26:10911–10915
Weese-Mayer DE, Lieske SP, Boothby CM, Kenny AS, Bennett HL, Silvestri JM, Ramirez JM (2006) Autonomic nervous system dysregulation: breathing and heart rate perturbation during wakefulness in young girls with Rett syndrome. Pediatr Res 60:443–449
Weese-Mayer DE, Lieske SP, Boothby CM, Kenny AS, Bennett HL, Ramirez JM (2008) Autonomic dysregulation in young girls with Rett Syndrome during nighttime in-home recordings. Pediatr Pulmonol 43:1045–1060
Wu YJ, Kruttgen A, Moller JC, Shine D, Chan JR, Shooter EM, Cosgaya JM (2004) Nerve growth factor, brain-derived neurotrophic factor, and neurotrophin-3 are sorted to dense-core vesicles and released via the regulated pathway in primary rat cortical neurons. J Neurosci Res 75:825–834
Wu H, Tao J, Chen PJ, Shahab A, Ge W, Hart RP, Ruan X, Ruan Y, Sun YE (2010) Genome-wide analysis reveals methyl-CpG-binding protein 2-dependent regulation of microRNAs in a mouse model of Rett syndrome. Proc Natl Acad Sci U S A 107:18161–18166
Xie Y, Longo FM (2000) Neurotrophin small-molecule mimetics. Prog Brain Res 128:333–347
Xu B, Goulding EH, Zang K, Cepoi D, Cone RD, Jones KR, Tecott LH, Reichardt LF (2003) Brain-derived neurotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat Neurosci 6:736–742
Young JI, Hong EP, Castle JC, Crespo-Barreto J, Bowman AB, Rose MF, Kang D, Richman R, Johnson JM, Berget S, Zoghbi HY (2005) Regulation of RNA splicing by the methylation-dependent transcriptional repressor methyl-CpG binding protein 2. Proc Natl Acad Sci U S A 102:17551–17558
Zhou Z, Hong EJ, Cohen S, Zhao WN, Ho HY, Schmidt L, Chen WG, Lin Y, Savner E, Griffith EC, Hu L, Steen JA, Weitz CJ, Greenberg ME (2006) Brain-specific phosphorylation of MeCP2 regulates activity-dependent Bdnf transcription, dendritic growth, and spine maturation. Neuron 52:255–269
Zocchi L, Sassone-Corsi P (2012) SIRT1-mediated deacetylation of MeCP2 contributes to BDNF expression. Epigenetics 7:695–700
Acknowledgments
The author thanks Min Lang and James Cody Howell for help in preparing and reviewing this manuscript and gratefully acknowledges funding support from the National Institutes of Health/National Institute of Neurological Diseases and Stroke, Rett Syndrome Research Foundation, and the International Rett Syndrome Foundation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg 2014
About this chapter
Cite this chapter
Katz, D.M. (2014). Brain-Derived Neurotrophic Factor and Rett Syndrome. In: Lewin, G., Carter, B. (eds) Neurotrophic Factors. Handbook of Experimental Pharmacology, vol 220. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-45106-5_18
Download citation
DOI: https://doi.org/10.1007/978-3-642-45106-5_18
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-45105-8
Online ISBN: 978-3-642-45106-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)