Abstract
Liver transplantation and resection for hilar cholangiocarcinoma are mutually exclusive therapeutic pathways, without possibility of cross over. Interpretation of relevant imaging is essential for accurate preoperative diagnosis, operative planning, management of complications during neoadjuvant therapy, and post-transplant surveillance of patients with hilar cholangiocarcinoma. Preoperatively, careful and dynamic interpretation of both cross-sectional imaging and cholangiography is needed to appreciate tumor presence, location, and vascular involvement. While many post-transplant vascular complications can be managed with percutaneous endovascular techniques, a number of specific vascular complications are preferentially treated operatively. An experienced multidisciplinary team is required for successful treatment of hilar cholangiocarcinoma with liver transplantation.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Hepatic Artery
- Primary Sclerosing Cholangitis
- Deceased Donor
- Hilar Cholangiocarcinoma
- Hepatic Artery Thrombosis
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Hilar cholangiocarcinoma requires accurate imaging for both diagnosis and choice of treatment. The goals of imaging are to (1) confirm clinical suspicion of cancer and its location; (2) assess local extent of disease including vascular and biliary involvement; and (3) detect regional and distant metastatic disease that may affect decisions regarding treatment. The Bismuth–Corlette classification (Fig. 1), is the most widely adopted system used to describe tumor location and biliary involvement [1]. Tumor location and biliary involvement are assessed by cholangiography with endoscopic (ERC), percutaneous (PTC), and/or cross-sectional imaging studies such as MRCP and CT cholangiography.
Bismuth–Corlette classification of perihilar cholangiocarcinoma: I below the bifurcation of right and left hepatic ducts; II at the bifurcation of right and left hepatic ducts; IIIa at the bifurcation with extension into the right hepatic duct; IIIb at the bifurcation with extension into the right hepatic duct; and IV extension into both right and left hepatic ducts or multicentric disease
Appreciation of the tumor location within the biliary system is critical for operative management of patients with hilar cholangiocarcinoma. Understanding of biliary anatomy as well as appreciation of nomenclature is also paramount: Biliary anatomy can be viewed proximal to distal based on either embryologic development or bile drainage. For the purpose of this chapter, we will refer to biliary bifurcation based on embryologic development and proximal to distal bile ducts based on bile drainage (i.e., proximal ducts in the liver and distal duct draining bile into the duodenum). Patients with hilar cholangiocarcinoma below the bifurcation of the hepatic ducts (Bismuth–Corlette I) or just at the bifurcation (Bismuth–Corlette II) are best treated with resection of the extrahepatic bile duct and biliary reconstruction. Intraoperative pathological examination of the specimen is necessary to confirm a tumor-free margin. Types I and II hilar cholangiocarcinoma are rare; most patients prove to have extension of tumor into the biliary bifurcation and the right or left hepatic ducts or both. Cholangiocarcinoma arising in the common bile duct also often extends into the head of the pancreas.
Bismuth–Corlette classification of hilar cholangiocarcinoma involving one or both hepatic ducts is IIIa for right duct involvement, IIIb for left duct involvement, and IV for bilateral duct involvement or diffuse multifocal disease. In general, resection is possible for IIIa and IIIb tumors by right or left hepatectomy provided that the vasculature to the contralateral side (residual liver) is free from involvement.
Liver vasculature must be assessed to ensure that the remnant liver will have both arterial and portal venous inflow. Vascular involvement is best assessed by CT or MRI. Limited vascular involvement to the remnant liver can occasionally be overcome by vascular reconstruction of the hepatic artery and/or portal vein, more commonly for IIIa tumors involving the right duct than for IIIb tumors involving the left duct since the left portal vein is more amenable to reconstruction.
Generally accepted criteria for unresectability are as follows: portal and/or arterial involvement not amenable to reconstruction (all types); unilateral biliary involvement with contralateral vascular involvement not amenable to reconstruction (types IIIa and IIIb); bilateral biliary involvement of secondary ducts (type IV); and an inadequate future liver remnant. Underlying chronic liver disease—especially primary sclerosing cholangitis (PSC)—usually precludes resection [2, 3]. These patients are best treated by neoadjuvant therapy and liver transplantation.
The aim of this chapter is to discuss (1) preoperative imaging of patients with hilar cholangiocarcinoma to determine resectability, (2) evaluation of unresectable patients to determine candidacy for transplantation, and (3) utilization of post-transplant imaging to follow patients transplanted for hilar cholangiocarcinoma since they are prone to develop late vascular complications related to neoadjuvant radiotherapy.
1 Determination of Resectability
1.1 Cross-Sectional Imaging
Both CT and MRI are useful imaging techniques for evaluation of patients with hilar cholangiocarcinoma [4–6]. The choice between CT and MRI varies due to clinician preference and institutional experience. MRI combined with MRCP is the preferred preoperative test at many institutions since it provides both cholangiography and cross-sectional imaging [7–9]. MRI allows for accurate assessment of both longitudinal and radial tumor extent in a single study. MRCP highlights biliary anatomy, delineates the level of biliary obstruction, and allows for visualization of the proximal and distal biliary ducts. Contrast MRI can detect a hilar mass (if present) and, most importantly, demonstrates vascular (arterial and venous) involvement.
Contrast multiphase CT has excellent diagnostic accuracy. CT can also demonstrate the location of the mass (if present) and determine vascular anatomy [6, 10]. Advances in CT imaging, particularly CT cholangiography and 3-dimensional reconstruction, have allowed for vast improvements in the assessment of anatomic relationships between the hepatic vasculature and biliary pathology [11, 12]. CT is somewhat better than MRI for detection of intra-abdominal and chest metastases.
ERCP is done to assess biliary anatomy, obtain an intraluminal specimen for histology and/or cytology, and to provide preoperative biliary decompression. Patients with suspected hilar cholangiocarcinoma require an experienced endoscopist. They require precise imaging, intraluminal biopsy and brush cytology for diagnosis, and biliary intubation for decompression. PTC is reserved for patients who have an inadequate ERCP (usually inadequate imaging to determine the extent of left or right duct involvement) or who are not amenable to biliary decompression with ERCP. We avoid PTC whenever possible. PTC and PTC-directed biopsy of cholangiocarcinoma can lead to tumor seeding and have been associated with higher rates of postoperative recurrence [13, 14].
Cross-sectional imaging studies should be carefully interpreted by a multidisciplinary team. Images cannot be interpreted in isolation, and a combination of cross-sectional imaging and dynamic biliary reconstructions is needed to appreciate tumor presence, location, and vascular involvement. As an example, the lesion in (Fig. 2a) can be misinterpreted as Bismuth–Corlette I, if the MRCP is not rotated to unmask the cystic duct overlap and appreciate Bismuth–Corlette II extension (Fig. 2b). Similarly, Fig. 3 demonstrates a Bismuth–Corlette III lesion, but additional imaging interpretation is necessary to distinguish IIIA from IIIB—a critical step for operative preparation.
Both MRI and CT imaging modalities frequently fail to identify a discrete tumor mass. If visible, the mass is usually hypovascular compared to adjacent liver parenchyma and increases in intensity with delayed MRI imaging. MRCP and CT cholangiography can identify ductal irregularities and intraluminal infiltration in the absence of a discrete mass [8, 11]. Biliary stricture and corresponding proximal dilatation help delineate longitudinal extension of tumor along the duct. Viewing of both axial and coronal reconstructions helps to visualize the anatomical relationships between the tumor and vascular structures. Extension of a stricture to secondary biliary ducts in a contralateral lobe is a contraindication to resection.
1.2 Bismuth–Corlette Classification
Patients with Bismuth–Corlette IIIa and IIIb cholangiocarcinoma are best treated with right or left hepatectomy (respectively), radical common bile duct resection, hepatoduodenal ligament lymphadenectomy, and bilio-enteric reconstruction. Preoperative cross-sectional imaging is necessary to confirm patency of the main and contralateral branch of the hepatic artery and patency of the main and contralateral branch of the portal vein. Ipsilateral portal vein impingement or occlusion, or involvement of ipsilateral hepatic artery does not preclude resection. A hallmark feature of locally advanced resectable hilar cholangiocarcinoma is lobar atrophy. It is associated with ipsilateral portal vein involvement and/or complete biliary obstruction [15, 16] and usually develops over time (Fig. 4a, b). While lobar atrophy has been associated with worse overall survival among patients with resectable hilar cholangiocarcinoma, it does not preclude a margin-negative resection [15, 17].
Patients with Bismuth–Corlette IV cholangiocarcinoma (Fig. 5) and those with contralateral vascular involvement are unresectable and should be evaluated for liver transplantation. Vascular encasement of the hepatic artery and/or portal vein and their branches is not a contraindication to liver transplantation. Liver transplantation is also a primary treatment option for cholangiocarcinoma in patients with PSC (Fig. 6a, b). PSC is an idiopathic chronic cholestatic liver disease with manifestations of progressive inflammatory destruction and biliary fibrosis of the entire bile duct system [18, 19]. These patients often have parenchymal disease precluding resection. Seven to 15 % of patients with PSC develop cholangiocarcinoma during their lifetime. The diagnoses of cholangiocarcinoma and PSC may be established at the same time, or cholangiocarcinoma may develop at a later time in patients with PSC. These patients are considered to have a “field defect” and are probably best treated by neoadjuvant therapy and liver transplantation rather than resection, even if they have otherwise potentially resectable tumors.
2 Evaluation for Transplantation
2.1 Inclusion/Exclusion Criteria for Liver Transplantation
Patients with unresectable hilar cholangiocarcinoma or hilar cholangiocarcinoma arising in the setting of PSC should be evaluated for liver transplantation. Transplantation and resection are mutually exclusive therapeutic pathways, without possibility for cross over. Patients found to have unresectable disease during exploration for resection do not do well with subsequent neoadjuvant therapy and liver transplantation. In our experience, operative exploration and subsequent neoadjuvant therapy increases the technical difficulty with transplantation and increased the likelihood for recurrence after transplantation.
Patients who undergo pathologic confirmation of tumor by transperitoneal tumor biopsy or fine-needle aspiration (including endoscopic ultrasound (EUS)-directed aspiration of the tumor) should also be excluded from transplantation [20].
Conversely, patients who fall out of the neoadjuvant therapy transplantation protocol cannot undergo liver resection even if they were thought to have potentially resectable disease. Neoadjuvant therapy causes widespread hilar biliary necrosis that would make resection and subsequent biliary reconstruction hazardous.
The United Network for Organ Sharing/Organ Procurement and Transplantation Network (UNOS/OPTN) approved a model for end-stage liver disease (MELD) exception score in 2009 for patients enrolled in an approved neoadjuvant therapy protocol. The MELD score exception is similar to the exception model for patients with transplantable hepatocellular carcinoma [21, 22]. Neoadjuvant therapy and staging are critical to success [22–24]. Since histological and/or cytological confirmation of diagnosis is not always possible, diagnosis is often dependent on imaging. Definitive diagnosis for treatment requires presence of a malignant-appearing stricture and at least one of the following: (1) endoluminal biopsy or cytology positive for cholangiocarcinoma; (2) polysomy by fluorescent in situ hybridization (FISH); (3) mass lesion on cross-sectional imaging at the location of the malignant-appearing stricture; or (4) CA 19-9 >100.
EUS with fine-needle aspiration (FNA) of suspicious nodes is useful to rule out patients with regional lymph node involvement destined to fall out at operative staging. Neoadjuvant therapy is associated with a multitude of side effects and potentially lethal complications and should not be administered unless the patient is a candidate for transplantation. It is important to make sure that aspiration is only done on the regional nodes and not on the primary tumor since that will preclude transplantation. Nodal metastases in any location—hepatoduodenal ligament (N1) or celiac, aortocaval, or retropancreatic (N2) lymph node basins—are a contraindication to neoadjuvant therapy and transplantation. Cross-sectional imaging of the abdomen and chest is also necessary to rule out extrahepatic metastases. Approximately 25–35 % of patients evaluated for neoadjuvant therapy are not amenable to treatment due to distant or nodal metastases.
Patients with intrahepatic cholangiocarcinoma, distal cholangiocarcinoma (below the level of the cystic duct), and gallbladder cancer are best treated by resection. Even if tumors in these locations are unresectable, neoadjuvant therapy and transplantation have not been shown to have any efficacy. Other exclusion criteria include the following: primary tumor greater than 3 cm in radial diameter (perpendicular to the duct); uncontrolled infection; prior treatment with radiotherapy and/or chemotherapy that would preclude full-dose neoadjuvant therapy; and history of other malignancy within 5 years [23, 24]. Patients must also be a suitable candidate for transplantation.
Operative staging is essential prior to transplantation. When possible, operative staging is done by hand-assisted laparoscopy. It is best done prior to the actual transplant procedure to rule out locally extensive disease and presence of nodal, peritoneal, and extrahepatic metastases. Operative staging involves a thorough intra-abdominal exploration and biopsy of any suspicious lesions, a common hepatic artery lymph node overlying the hepatic artery at the takeoff of the gastroduodenal artery, and a pericholedochal lymph node. Occasionally, patients are too sick to undergo a separate operation, and staging is done at the time a donor liver becomes available.
2.2 Pre-Transplant Imaging
Pre-transplant cross-sectional imaging is performed every 3 months prior to transplantation as required by UNOS/OPTN policy for continuous surveillance of patients with malignancy awaiting liver transplantation. Evidence of disease progression or metastases while on the neoadjuvant protocol precludes transplantation. The patient dropout rate after starting neoadjuvant therapy is approximately 11.5 % per 3 months. Recurrent cholangitis occurs in the majority of the patients during neoadjuvant therapy [22]. Select patients, particularly those with PSC, can develop hepatic abscesses (Fig. 7a, b) requiring preoperative percutaneous drainage Fig. (8) and prolonged antibiotic therapy. Active patient surveillance and supervision by a multidisciplinary transplant team throughout the course of therapy is critical for successful management of treatment complications and achieving success.
2.3 Liver Transplantation
Liver transplantation for hilar cholangiocarcinoma is done in a similar fashion to transplantation for other acute and chronic liver diseases with several technical considerations [24, 25]. Hilar dissection is avoided to prevent dissemination of tumor. The portal vein is transected as proximal to its emergence from retropancreatic groove as possible to excise majority of native vein present in the irradiated field. Similarly, the bile duct is transected as close to the pancreas as possible, and the margin is checked by frozen section. Marginal involvement has been limited to patients with underlying PSC and has been observed in approximately 10 % of PSC patients. Options include re-excision and pancreatoduodenectomy. Hepatic artery thrombosis (HAT) due to irradiation of the artery is avoided by reconstruction with a donor iliac artery jump graft from infrarenal aorta to the donor hepatic artery during deceased donor liver transplantation. This technique has not been successful with living donor liver transplantation due to the size mismatch between an iliac graft and a donor left or right hepatic artery. We now perform reconstruction with the irradiated recipient proper or common hepatic artery and monitor the recipient closely with Doppler ultrasound during the postoperative period for any change in hepatic arterial flow.
Portal vein reconstruction is done by a direct anastomosis between the donor and recipient portal veins during deceased donor transplantation. A direct anastomosis is not possible during living donor transplantation due to low division of the recipient portal vein. The gap is reconstructed with a segment of deceased donor iliac vein as an interposition graft.
Biliary reconstruction is performed with a Roux-en-Y choledochojejunostomy or hepaticojejunostomy. We prefer to use an external transjejunal cholangiocatheter to obtain a cholangiogram during the early postoperative period with deceased donor transplantation; biliary reconstruction in a living donor transplant recipient is performed with an internal biliary stent.
3 Post-Transplant Imaging
3.1 Routine Surveillance
Biliary complications occur with the same frequency after transplantation for cholangiocarcinoma as they do for other diseases. Vascular complications, however, are more common after transplantation for cholangiocarcinoma due to the preoperative neoadjuvant therapy. Patients are examined by Doppler ultrasound immediately after transplantation and the next day to confirm patency of all vessels with flows in the normal directions. In addition, Doppler ultrasonography is also routinely performed on days 7 and 21, at 4 months, and annually. Patients with external biliary cholangiocatheters undergo tube cholangiography on days 7 and 21 or when laboratory tests indicate a potential biliary problem. Follow-up specific for patients with cholangiocarcinoma includes chest, abdomen, and pelvis CT and CA 19-9 testing at 4 month intervals for the 1st year and yearly afterward. Approximately 20 % of patients develop recurrent cholangiocarcinoma, and local recurrence is a frequent cause of vascular (Fig. 9a, b), biliary, or enteric occlusion.
3.2 Vascular Complications
Vascular complications are more common after transplantation for cholangiocarcinoma than for other liver diseases due to the high-dose neoadjuvant radiotherapy. These complications primarily occur with the reconstructed portal vein in deceased and living donor recipients, and the reconstructed hepatic artery in living donor recipients [26]. Hepatic artery complications are avoided in deceased donor recipients by abandoning the irradiated artery and using an iliac artery jump graft. Vascular complications occurring late after transplantation can also be due to recurrent cancer. We have observed vascular complications in up to 40 % of our recipients attributable to neoadjuvant therapy: 20 % for the hepatic artery (living donor recipients) and 20 % for the portal vein (living and deceased donor recipients). Hepatic venous or caval outflow complications are rare and are comparable to non-cholangiocarcinoma transplant recipients [26]. Doppler ultrasound and contrast-enhanced ultrasound technology have formed the foundation of vascular evaluation after liver transplantation with very high sensitivity and specificity [27–29]. Suspicious but inconclusive findings can be confirmed by cross-sectional imaging.
3.3 Hepatic Artery Complications
An iliac artery jump graft from donor hepatic artery to recipient infrarenal aorta is routinely used in deceased donor liver recipients. This approach was used in our early living donor recipients, but high rates of early hepatic artery complications led to a change in our practice. Currently, we perform donor hepatic artery to recipient-irradiated hepatic artery reconstructions with acceptable morbidity. In contrast to non-transplant patients, compromise in the hepatic arterial flow must be promptly corrected to avoid biliary ischemia, cholangiopathy, and subsequent biliary and infectious complications.
Arterial complications are approximately twice as common (approximately 20 %) after transplantation for hilar cholangiocarcinoma compared to non-cholangiocarcinoma recipients; rates can be even higher among living donor recipients. Hepatic artery thrombosis (HAT) is more severe than hepatic artery stenosis (HAS) and is associated with higher morbidity and mortality. Early HAT is best managed with emergency operative thrombectomy and thrombolysis. Operative intervention for late HAT is largely futile. Retransplantation is necessary for up to 75 % of patients with early HAT, usually due to development of cholangiopathy and hepatic abscesses [30].
HAS may be managed with percutaneous transluminal angioplasty [31, 32]. Percutaneous stents have been used with increasing success for HAS, but they have also been associated with rare post-procedural thrombosis requiring retransplantation. Depending on the location of HAS, operative revision is also a possible treatment. We routinely use aspirin for HTA prophylaxis, and all cholangiocarcinoma patients receive short-term prophylactic low molecular weight heparin during the immediate postoperative period.
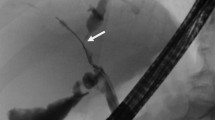
Mycotic pseudoaneurysms from either HA anastomosis or gastroduodenal artery stump (Fig. 10) are rare (approximately 1 %), but formidable complications of liver transplantation. The most frequent cause is an intra-abdominal infection or bile leak. We have also observed them after a leak from the pancreatic anastomosis for patients that required pancreatoduodenectomy. Unlike pancreatoduodenectomy only patients, these complications in liver transplant recipients require hepatic artery revision and occasionally retransplantation. Percutaneous embolization and/or intraluminal stent placement has been described, but both can lead to thrombosis, dissection, or arterial rupture with subsequent need for retransplantation [33].
3.4 Portal Vein Complications
Portal vein reconstruction is performed with primary end-to-end anastomosis during deceased donor transplantation and with an iliac vein jump graft during living donor transplantation. Regardless of donor type, the native portal vein is divided as close as possible to the pancreas to achieve better tumor clearance and avoid the irradiated vein. Portal vein thrombosis (PVT) and portal vein stenosis (PVS) are significantly more common (approximately 20 %) after transplantation for hilar cholangiocarcinoma than for other indications [26]. Early PVT is managed by operative thrombectomy and anastomotic revision. Late PVT is rare, and recurrent cholangiocarcinoma must be excluded as a cause. PVS due to neoadjuvant therapy is most often detected by surveillance ultrasound and CT 4 months after transplantation. We have had excellent success and very few complications with percutaneous transhepatic portal angioplasty and stent placement (Fig. 11a–d) [34, 35]. When detected late, we have observed progressive portal stenosis and thrombosis with observation, and we now prefer to intervene with endovascular angioplasty and stent placement for most patients who develop PVS [26].
3.5 Vascular Outflow Complications
Hepatic vein and inferior vena cava complications after liver transplantation for hilar cholangiocarcinoma are rare and do not differ in prevalence compared to non-cholangiocarcinoma liver recipients. Outflow complications have been more commonly described after living donor graft implantation, but we have not noticed a difference in our experience [26]. The hepatic veins are not affected by neoadjuvant radiotherapy. Outflow complications are thus purely technical and require intervention with either an endovascular approach or reoperation. Angioplasty and stent placement is the preferred strategy for management of venous outflow complications [36, 37]. Reoperation is possible only during the immediate postoperative period.
3.6 Biliary Complications
Biliary complications after transplantation for hilar cholangiocarcinoma occur in approximately 10 % of deceased donor liver recipients and 30 % of living donor liver recipients. Leaks with Roux-en-Y choledochojejunostomy after deceased donor transplantation are best managed by reoperation and revision. Leaks with Roux-en-Y hepaticojejunostomy after living donor transplantation are best managed by biliary intubation and drainage. Biliary strictures and cholangiopathy occur in approximately 15–20 % of liver recipients. Strictures can develop after resolution of a bile leak. Cholangiopathy can result from HAT (even with prompt revascularization) and donation after cardiac death (Fig. 12a–d) [22]. Biliary strictures are predominantly managed by endoscopic intervention and may require multiple procedures over a prolonged period of time. Cholangiopathy usually requires prolonged intervention and may eventually require retransplantation.
4 Summary
Interpretation of relevant imaging is essential for accurate preoperative diagnosis, operative planning, management of complications during neoadjuvant therapy, and post-transplant surveillance of patients with hilar cholangiocarcinoma. Treatment decisions and management of complications warrant a multidisciplinary team approach to patient care in order to achieve success.
References
Bismuth H, Corlette MB (1975) Intrahepatic cholangioenteric anastomosis in carcinoma of the hilus of the liver. Surg Gynecol Obstet 140:170–178
Rosen CB, Nagorney DM, Wiesner RH, Coffey RJ Jr, LaRusso NF (1991) Cholangiocarcinoma complicating primary sclerosing cholangitis. Ann Surg 213:21–25
Rosen CB, Heimbach JK, Gores GJ (2008) Surgery for cholangiocarcinoma: the role of liver transplantation. HPB (Oxford) 10:186–189
Ruys AT, van Beem BE, Engelbrecht MR, Bipat S, Stoker J, van Gulik TM (2012) Radiological staging in patients with hilar cholangiocarcinoma: a systematic review and meta-analysis. Br J Radiol 85:1255–1262
Hyodo T, Kumano S, Kushihata F et al (2012) CT and MR cholangiography: advantages and pitfalls in perioperative evaluation of biliary tree. Br J Radiol 85:887–896
Khan SA, Davidson BR, Goldin RD et al (2012) Guidelines for the diagnosis and treatment of cholangiocarcinoma: an update. Gut 61:1657–1669
Maccioni F, Martinelli M, Al Ansari N et al (2010) Magnetic resonance cholangiography: past, present and future: a review. Eur Rev Med Pharmacol Sci 14:721–725
Manfredi R, Masselli G, Maresca G, Brizi MG, Vecchioli A, Marano P (2003) MR imaging and MRCP of hilar cholangiocarcinoma. Abdom Imaging 28:319–325
Masselli G, Gualdi G (2008) Hilar cholangiocarcinoma: MRI/MRCP in staging and treatment planning. Abdom Imaging 33:444–451
Ariff B, Lloyd CR, Khan S et al (2009) Imaging of liver cancer. World J Gastroenterol 15:1289–1300
Kim HJ, Kim AY, Hong SS et al (2006) Biliary ductal evaluation of hilar cholangiocarcinoma: three-dimensional direct multi-detector row CT cholangiographic findings versus surgical and pathologic results–feasibility study. Radiology 238:300–308
Endo I, Shimada H, Sugita M et al (2007) Role of three-dimensional imaging in operative planning for hilar cholangiocarcinoma. Surgery 142:666–675
Chapman WC, Sharp KW, Weaver F, Sawyers JL (1989) Tumor seeding from percutaneous biliary catheters. Ann Surg 209:708–713
Sakata J, Shirai Y, Wakai T, Nomura T, Sakata E, Hatakeyama K (2005) Catheter tract implantation metastases associated with percutaneous biliary drainage for extrahepatic cholangiocarcinoma. World J Gastroenterol 11:7024–7027
Matsuo K, Rocha FG, Ito K et al (2012) The Blumgart preoperative staging system for hilar cholangiocarcinoma: analysis of resectability and outcomes in 380 patients. J Am Coll Surg 215:343–355
Friesen BR, Gibson RN, Speer T, Vincent JM, Stella D, Collier NA (2011) Lobar and segmental liver atrophy associated with hilar cholangiocarcinoma and the impact of hilar biliary anatomical variants: a pictorial essay. Insights Imaging 2:525–531
Zaydfudim VM, Clark CJ, Kendrick ML et al (2013) Correlation of staging systems to survival in patients with resected hilar cholangiocarcinoma. Am J Surg 206:159–165
Burak K, Angulo P, Pasha TM, Egan K, Petz J, Lindor KD (2004) Incidence and risk factors for cholangiocarcinoma in primary sclerosing cholangitis. Am J Gastroenterol 99:523–526
Lazaridis KN, Gores GJ (2006) Primary sclerosing cholangitis and cholangiocarcinoma. Semin Liver Dis 26:42–51
Heimbach JK, Sanchez W, Rosen CB, Gores GJ (2011) Trans-peritoneal fine needle aspiration biopsy of hilar cholangiocarcinoma is associated with disease dissemination. HPB (Oxford) 13:356–360
Gores GJ, Gish RG, Sudan D, Rosen CB (2006) Model for end-stage liver disease (MELD) exception for cholangiocarcinoma or biliary dysplasia. Liver Transpl 12:S95–S97
Darwish Murad S, Kim WR, Harnois DM et al (2012) Efficacy of neoadjuvant chemoradiation, followed by liver transplantation, for perihilar cholangiocarcinoma at 12 US centers. Gastroenterology 143:88–98
Rosen CB, Heimbach JK, Gores GJ (2010) Liver transplantation for cholangiocarcinoma. Transpl Int 23:692–697
Rea DJ, Rosen CB, Nagorney DM, Heimbach JK, Gores GJ (2009) Transplantation for cholangiocarcinoma: when and for whom? Surg Oncol Clin N Am 18:325–337
Heimbach JK, Gores GJ, Haddock MG et al (2004) Liver transplantation for unresectable perihilar cholangiocarcinoma. Semin Liver Dis 24:201–207
Mantel HT, Rosen CB, Heimbach JK et al (2007) Vascular complications after orthotopic liver transplantation after neoadjuvant therapy for hilar cholangiocarcinoma. Liver Transpl 13:1372–1381
Dodd GD 3rd, Memel DS, Zajko AB, Baron RL, Santaguida LA (1994) Hepatic artery stenosis and thrombosis in transplant recipients: Doppler diagnosis with resistive index and systolic acceleration time. Radiology 192:657–661
Hom BK, Shrestha R, Palmer SL et al (2006) Prospective evaluation of vascular complications after liver transplantation: comparison of conventional and microbubble contrast-enhanced US. Radiology 241:267–274
Huang TL, Cheng YF, Chen TY et al (2010) Doppler ultrasound evaluation of postoperative portal vein stenosis in adult living donor liver transplantation. Transplant Proc 42:879–881
Duffy JP, Hong JC, Farmer DG et al (2009) Vascular complications of orthotopic liver transplantation: experience in more than 4,200 patients. J Am Coll Surg 208:896–903
Sommacale D, Aoyagi T, Dondero F et al (2013) Repeat endovascular treatment of recurring hepatic artery stenoses in orthotopic liver transplantation. Transpl Int 26:608–615
Rostambeigi N, Hunter D, Duval S, Chinnakotla S, Golzarian J (2013) Stent placement versus angioplasty for hepatic artery stenosis after liver transplant: a meta-analysis of case series. Eur Radiol 23:1323–1334
Saad WE, Dasgupta N, Lippert AJ et al (2013) Extrahepatic pseudoaneurysms and ruptures of the hepatic artery in liver transplant recipients: endovascular management and a new iatrogenic etiology. Cardiovasc Intervent Radiol 36:118–127
Ko GY, Sung KB, Yoon HK, Lee S (2007) Early posttransplantation portal vein stenosis following living donor liver transplantation: percutaneous transhepatic primary stent placement. Liver Transpl 13:530–536
Cheng YF, Ou HY, Tsang LL et al (2010) Vascular stents in the management of portal venous complications in living donor liver transplantation. Am J Transplant 10:1276–1283
Ko GY, Sung KB, Yoon HK et al (2002) Endovascular treatment of hepatic venous outflow obstruction after living-donor liver transplantation. J Vasc Interv Radiol 13:591–599
Darcy MD (2007) Management of venous outflow complications after liver transplantation. Tech Vasc Interv Radiol 10:240–245
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Zaydfudim, V.M., Nagorney, D.M., Rosen, C.B. (2014). Imaging of Hilar Cholangiocarcinoma for Liver Transplantation. In: Herman, J., Pawlik, T., Thomas, Jr., C. (eds) Biliary Tract and Gallbladder Cancer. Medical Radiology(). Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-40558-7_9
Download citation
DOI: https://doi.org/10.1007/978-3-642-40558-7_9
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-40557-0
Online ISBN: 978-3-642-40558-7
eBook Packages: MedicineMedicine (R0)