Abstract
Geometric morphometric analysis was applied to the skull (separately in lateral and dorsal view) of sauropodomorph dinosaurs from the Mesozoic. These analyses were applied to quantify the macroevolutionary trends in sauropodomorph craniofacial form. As the early craniofacial evolution of Sauropodomorpha has been considered to be “conservative”, with all major changes to skull shape occurring within Sauropoda itself, geometric techniques are herein employed to quantify this statement. The landmark-based approach (relative warps analysis) was able to confirm this statement, albeit within the limits of the sample of this study. The morphospaces in both views demonstrate that sauropodomorphs became more divergent in terms of craniofacial form, with the sauropods occupying a distinct region of, and a greater volume of, morphospace than the “prosauropods”. Although the concordance between craniofacial form and phylogeny is statistically significant, the correlations themselves are not significant. As such, craniofacial evolution within Sauropodomorpha is “shaped” by more than just historical contingency.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Idea and Aims
Geometric morphometric analysis was applied to the skull (separately in lateral and dorsal view) of sauropodomorph dinosaurs from the Mesozoic. These analyses were applied to quantify the macroevolutionary trends in sauropodomorph craniofacial form. As the early craniofacial evolution of Sauropodomorpha has been considered to be “conservative”, with all major changes to skull shape occurring within Sauropoda itself, geometric techniques are herein employed to quantify this statement. The landmark-based approach (relative warps analysis) was able to confirm this statement, albeit within the limits of the sample of this study. The morphospaces in both views demonstrate that sauropodomorphs became more divergent in terms of craniofacial form, with the sauropods occupying a distinct region of, and a greater volume of, morphospace than the “prosauropods”. Although the concordance between craniofacial form and phylogeny is statistically significant, the correlations themselves are not significant. As such, craniofacial evolution within Sauropodomorpha is “shaped” by more than just historical contingency.
2 Introduction
Throughout most of the Mesozoic era (250–65 Mya) sauropodomorph dinosaurs were the dominant terrestrial herbivores. They reached the biomechanical limits of terrestrial gigantism, with many species of taxa achieving sizes an order of magnitude larger than other extinct or living terrestrial animals (Sander 2000). Therefore, the question arises, how did these multi-tonne giants fuel their colossal bodies? How was it possible for there to be six or more contemporaneous species of giant sauropods in Late Jurassic (155–145 Mya) ecosystems? As with examples of multiple contemporaneous large carnivorous dinosaurs (e.g. Henderson 1998), resource (niche) partitioning via morphological differentiation has been posited as maintaining high biodiversity. In the case of sauropods, differentiation is concentrated on craniofacial form (although tooth crown and neck morphology are also important) (e.g. Barrett and Upchurch 1994; Calvo 1994; Upchurch and Barrett 2000; Barrett and Upchurch 2007; Upchurch et al. 2007).
Sauropodomorpha is a clade consisting of two well-known groups, Sauropoda and Prosauropoda. Recent phylogenetic analyses have convincingly demonstrating the paraphyly of Prosauropoda (e.g. Upchurch et al. 2007; Yates 2003, 2007; Yates and Kitching 2003), therefore we hereafter refer to all non-sauropod sauropodomorphs as “prosauropods” in the knowledge they do not constitute a natural group. Although the topology of the sauropodomorph evolutionary tree is still disputed in these analyses, the taxa selected in this study remain stable throughout (Fig. 11.1).
Although recent work has demonstrated the unrealised craniofacial diversity within Sauropoda (e.g. Nigersaurus; Sereno et al. 2007), “prosauropods” have long been considered conservative in cranial morphology, while their feeding mechanisms are still considered to be conservative (e.g. Upchurch and Barrett 2005). As recent phylogenetic analyses include numerous “prosauropods” and sauropods, we use geometric morphometrics and multivariate statistics to examine the macroevolutionary history of craniofacial form within this clade, assessing whether broad statements concerning craniofacial diversity are defensible.
3 Materials and Methods
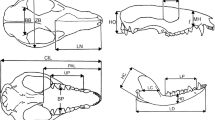
The sauropodomorph cranial specimens analysed herein are all based upon photographs or reconstructions from the literature (see Larvan 2007 for the specimen list). The landmarks (Fig. 11.2, Table 11.1) were digitised using Image J vers. 1.36b (Rasband 2007).
The program tpsRelw vers. 1.42 (Rohlf 2005) was used to perform the relative warps analyses (RWA; Rohlf 1993). The first step was to perform a generalised Procrustes analysis on the landmark configurations taken from the digital images, to remove differences in location, orientation and scale (see Stayton and Ruta 2006). Once the landmarks were in Procrustes superimposed space a consensus plot (= the mean shape) was calculated. This consensus plot is the mean landmark configuration from all species in the sample. A principle components analysis was done on shape co-ordinates (see Cavalcanti 2004 for the full methodology in detail).
Going from Procrustes superimposed alignment to an ordination plot involves projecting the data points (the taxa) from a curved surface on to a flat one. This can be thought of as taking one side of a globe and projecting the towns and cities onto a map. The process of projection can lead to significant distortion of the relative positions between taxa. To test for this, the Procrustes distances (distances between specimens in Procrustes alignment) were regressed against the Euclidean distances (distances between specimens on the morphospace plot) using the program tpsSmall vers. 1.20 (Rohlf 2003).
Four disparity metrics were calculated using all the RW scores from all axes, for “prosauropods” and sauropods: the sum and product of the ranges and variances (Wills et al. 1994). Each metric gives an indication of volume of morphospace occupied. However, range measures quantify the entire spread of morphological variation, or the “absolute extent of bodyplan variety” (Wills 1998), whereas variance measures indicate average dissimilarity among forms. The former are more sensitive to sample size, whereas the latter are more sensitive to taxonomic practice but robustly insensitive to sample size (Wills et al. 1994). All metrics were calculated using the software program Rare (Wills 1998), and multiplicative measures were normalised by taking the 13th root. Statistical significance between the disparity of “prosauropods” and sauropods were assessed in two ways: by the overlap or non-overlap of 95% bootstrap confidence intervals for each disparity metric (calculated by Rare with 1,000 replications) and NPMANOVA (non-parametric multivariate analysis of variance), which tests for significant differences in the distribution of groups in morphospace (Anderson 2001). NPMANOVA, the multivariate (and non-parametric) equivalent of ANOVA, was calculated in PAST (vers. 1.78; Hammer et al. 2001). One of the strengths of NPMANOVA is that as a non-parametric statistical test, it does not assume or require normality from the multivariate data.
In order to assess the fit between RW skull morphspace and phylogeny, Mantel’s test was used. From the phylogeny a simple symmetrical node-based difference matrix was collated, while Procrustes distances were calculated using tpsSmall (v1.20, Rohlf 2003) and then collated to a similar symmetrical matrix. The resulting matrices were imported into PAST (v1.78) and a Mantel’s test carried out.
4 Analyses
Regression of Procrustes distance against Euclidean distance for each pair of landmarks gave a high correlation coefficient with r > 0.99 for both cranium lateral and cranium dorsal aspect views. As such, distortion did not prevent interpretation of results from the Relative Warp morphospaces.
The first two RW axes in lateral view were deemed significant as they account for 97.7 percent cumulatively of the variance about the mean shape (Fig. 11.3). Moving across RW1, negative to positive, all landmarks move rostrally; 5 (inflexion point of quadratojugal at the mandibular joint), 6 (premaxilla-maxilla suture along dorsal margin) and 7 (dorsal-most point of the premaxilla) only slightly; whereas 8 (jugal-postorbital suture along the orbit rim) and 10 (prefrontal-frontal suture along the orbit rim) “move” considerably. In addition, landmarks 1 (anterior-most point along the premaxilla ventral margin), 2 (premaxilla-maxilla suture along ventral margin) and 12 (centroid of orbit) exhibit a downwards movement, while landmark 2 converges on landmark 1. Landmark 4 (anterior-most point along quadratojugal ventral margin) is the only one to show an elevation in position. Based on the landmark position changes, progressively positive RW1 values have a lengthened premaxilla and the quadratojugal elongating and advancing rostrally (rostral movement of the jaw joint). The second RW axis subsumes three shape variations: 1) snout elongation, 2) tooth-row shortening and 3) orbit moving up and backwards. Moving negative to positive across this axis, landmarks 1–3 (posterior-most point along maxilla ventral margin) and 8–12 are elevated, 4–7 and 13 (posterior end of tooth row) are depressed, while landmarks 1, 4, 5 and 8–12 “move” caudally and 2, 3, 6, 7 and 13 “move” rostrally.
The morphospace delimited by the first two RW axes in dorsal view (Fig. 11.3) displays separation between “prosauropods” and sauropods across RW1, with Shunosaurus intermediate between the two. The diplodocoids (Diplodocus and Nigersaurus) are distinguished from all other sauropodomorphs, with an elongate snout and a retracted external nares (bony hole for the soft tissue of the nostrils and olfactory senses).
The first two RW axes in dorsal view were deemed significant as they accounted for 72.6 percent cumulatively of the variation about the mean shape (Fig. 11.4). Moving across RW axis one, from negative to positive, landmark 3 (premaxilla-maxilla suture along lateral margin) advances rostrally. Landmarks 2 (posterior-most point of premaxilla along the midline), 4 (nasal-frontal suture along the midline) 6 (prefrontal-nasal-frontal suture), 8 (anterior-most point of the nasal along the midline) and 9 (prefrontal-frontal suture) all “move” caudally. The second RW axis subsumes skull narrowing and lengthening, in addition to narrowing of the premaxillae and the shortening of the nasals. Moving from negative to positive across the axis, landmarks 1 (anterior-most point of the premaxilla along the midline), 4, 5 (frontal-parietal suture along the midline), 6 and 9 are in a rostral position; whereas landmarks 2 and 8 are in a caudal position, and landmarks 3 and 9 “move” in to a medial position. Landmark 7 (posterior-most point of the parietal along the midline) does not alter position in either RW axes.
The morphospace delimited by the first two RW axes in dorsal view (Fig. 11.4) displays separation between “prosauropods” and sauropods across RW1 and RW2, with Shunosaurus intermediate between the two. Unlike the lateral view morphospace, the diplodocoids (Diplodocus and Nigersaurus) are not united as Nigersaurus has a laterally expanded snout. Europasaurus (the only known case of insular dwarfism within Sauropoda) is the only sauropod with negative RW1 values.
Comparing the distributions of “prosauropods” and sauropods morphospace occupation in both lateral and dorsal view achieved statistical significance (Table 11.2), meaning they occupied significantly distinct regions of morphospace.
All disparity metrics show that sauropods are more disparate in terms of volume of morphospace occupation; however the very small sample size (“prosauropod” n = 5, sauropod n = 8) of this analysis severally limits the rarefaction profiles (Fig. 11.5). As the 95% confidence intervals consistently overlapped, statistically significance could not be achieved.
The fit between craniofacial form and phylogeny was found to be statistically significant for both dorsal and lateral view (Table 11.3). However, the correlations themselves were not significant for either view (the highest R2being 0.55).
5 Discussion
This study was primarily concerned with using a measure of morphological variation in order to quantify craniofacial macroevolutionary trends. Relative Warp Analysis was used to measure the variation in skull geometry as the deviation from a mean shape of the landmark positions. Herein, geometric morphometric techniques delineated “prosauropods” and sauropods in morphospace, although small sample size meant that quantifying volume of morphospace occupation (disparity metrics) failed.
Relative warps morphospaces for both lateral and dorsal view of the cranium could distinguish clades within Sauropodomorpha. The separation between “prosauropods” and sauropods can be distinguished in their cranial morphology. The prosauropods have lower, more tapered snouts, narrowing towards the tip, whereas sauropods have taller skulls with more robust snouts. The dorsal view shows this varied design, with a clear distinction of “prosauropods”, but a large degree of variation within Sauropoda. The basal sauropods (Shunosaurus and Mamenchisaurus) have a similarly slender appearance, whereas the neosauropods (all taxa more derived than those two, see Fig. 11.1) exhibit a broadening in the snout, resulting in a generally even width from posterior to anterior end of the skull.
The comparatively small morphospace occupied by the “prosauropods” provides very tentative support for the hypothesis that they were more conservative in craniofacial form (and presumably function) and that the sauropods were much more diverse, as shown by their greater morphospace occupation.
With the exception of the dorsal view, diplodocoids are significantly separated in morphospace from all other sauropodomorphs. This is due to their distinct craniofacial form (e.g. elongate snout, retracted external nares), which is possibly related to the feeding strategies hypothesised for this clade (branch stripping and precision biting; Barrett and Upchurch 1994, 2007; Calvo 1994).
Although broad trends within sauropodomorph craniofacial evolution can be discerned, the low correlation between form and phylogeny suggests that phylogenetic inertia has a limited impact upon craniofacial form. This refers to the notion that constraints imposed by phylogenetic history could potentially refrain the skull from reaching its functional optimal.
6 Conclusions
Geometric morphometrics analyses of skull landmark configurations (lateral and dorsal view) were able to delineate sauropods from “prosauropods”. Both views demonstrate that sauropodomorphs became more divergent in terms of craniofacial form throughout the Mesozoic, most especially the sauropods. A clear evolutionary trend towards cranial robustness in sauropods is supported by morphometrics, which also supports the general craniofacial conservativeness of the “prosauropods”. Evaluating statistical significance of the distribution and disparity for sauropods and “prosauropods” demonstrate that sauropods occupy a distinct region of morphospace, and have greater dispersal within morphospace. In addition, there is a statistically significant correlation between craniofacial form and phylogeny; however the correlations themselves are not highly significant.
Although this macroevolutionary study has begun the quantitative elucidation of sauropodomorph evolution, the key problem to our continuing understanding of sauropodomorph craniofacial evolution is the scarcity of well-preserved skulls. Recent studies have shown not only the unrealised cranial diversity of sauropodomorphs (e.g. Sereno et al. 2007), but tooth-crown gross morphology is also highly diverse (e.g. Upchurch and Barrett 2000) and that osteological correlates for structures like a fleshy cheek are known for “prosauropods” and early sauropods (Barrett and Upchurch 2007). Clearly, there are many more exciting discoveries to be made.
References
Anderson MJ. (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecology 26: 32–46.
Barrett PM, Upchurch P. (1994) Feeding mechanisms of Diplodocus. Gaia 10: 195–203.
Barrett PM, Upchurch P. (2007) The evolution of feeding mechanisms in early sauropodomorph dinosaurs. In: Barrett PM, Batten DJ (Eds.) Evolution and paleobiology of early sauropodomorph dinosaurs. Special Papers in Palaeontology 77: 91–112.
Calvo JO. (1994) Jaw mechanics in sauropod dinosaurs. Gaia 10: 183–193.
Cavalcanti MJ. (2004) Geometric morphometric analysis of head shape variation in four species of hammerhead sharks (Carcharhiniformes: Sphyrnidae). In: Elewa AMT. (Ed.) Morphometrics: applications in biology and paleontology. Springer-Verlag, Heidelberg: 97–114.
Hammer Ø, Harper DAT, Ryan PD. (2001) PAST: Palaeontological Statistics software package for education and data analysis. Palaeontologia Electronica 4(1): 9.
Henderson DM. (1998) Skull and tooth morphology as indicators of niche partitioning in sympatric Morrison Formation theropods. In: Perez-Moreno BP, Holtz T, Sanz JL, Moratalla J, (Eds.) Aspects of theropod palaeobiology. Gaia 15: 219–226.
Larvan MD. (2007) A combined geometric morphometrics and finite element analysis approach to comparative cranial mechanics of sauropodomorph dinosaurs. Unpublished MSc thesis, University of Bristol.
Rasband W. (2007) Image J v. 1.36b, freeware available from http://rsb.info.nih.gov/ij/
Rohlf FJ. (1993) Relative warp analysis and an example of its application to mosquito wings. In: Marcus LF, Valdecasas A, Bello E. (Eds.) Contributions to morphometrics. Madrid Museum of Natural History, Madrid, Spain: 131–159.
Rohlf FJ. (2003) tpsSmall v.1.20, freeware available from http://life.bio.sunysb.edu/morph/
Rohlf FJ. (2005) tpsRelw v. 1.42, freeware available from http://life.bio.sunysb.edu/morph/
Sander PM. (2000) Longbone histology of the Tendaguru Sauropods: implications for growth and biology. Paleobiology 26: 466–488.
Sereno PC, Wilson JA, Witmer LM, Whitlock JA, Maga A, Ide O, Rowe TA. (2007) Structural extremes in a Cretaceous dinosaur. PLoS ONE 11: e1230.
Stayton CT, Ruta M. (2006) Geometric morphometrics of the skull roof of stereospondyls (Amphibia: Temnospondyli). Palaeontology 49(2): 307–337.
Upchurch P, Barrett PM. (2000) The evolution of sauropod feeding mechanisms. In: Sues H-D. (Eds.) Evolution of herbivory in terrestrial vertebrates: perspectives from the fossil record. Cambridge University Press, Cambridge: 79–122.
Upchurch P, Barrett PM. (2005) Phylogenetic and taxic perspectives on sauropod diversity. In: Rogers KC, Wilson JA (Eds.) The sauropods: evolution and paleobiology. University of California Press, Berkeley: 104–124.
Upchurch P, Barrett PM, Xijin Z, Xing X. (2007) A re-evaluation of Chinshakiangosaurus chunghoensis Ye vide Dong 1992 (Dinosauria, Sauropodomorpha): implications for cranial evolution in basal sauropod dinosaurs. Geological Magazine 144(2): 247–262.
Wills MA. (1998) Crustacean disparity through the Phanerozoic: comparing morphological and stratigraphic data. Biological Journal of the Linnean Society 65: 455–500.
Wills MA, Briggs DEG, Fortey RA. (1994) Disparity as an evolutionary index: a comparison of Cambrian and Recent arthropods. Paleobiology 20: 93–131.
Yates AM. (2003) A new species of the primitive dinosaur Thecodontosaurus (Saurischia: Sauropodomorpha) and its implications for the systematics of early dinosaurs. Journal of Systematic Palaeontology 1: 1–42.
Yates AM. (2007) The first complete skull of the Triassic dinosaur Melanorosaurus Haughton (Sauropodomorpha: Anchisauria). In: Barrett PM, Batten, DJ. (Eds.) Evolution and paleobiology of early sauropodomorph dinosaurs. Special Papers in Palaeontology 77: 9–55.
Yates AM, Kitching JW. (2003) The earliest known sauropod dinosaur and the first steps towards sauropod locomotion. Proceedings of the Royal Society of London B 270: 1753–1758.
Acknowledgments
We would like to thank N. Knötschke (Dino-Park, Germany) for providing generous access to the specimens in his care. For discussion etc we would like to thank P.M. Barrett (Natural History Museum, London); M. Ruta, E.J. Rayfield, M. Sakamoto and P.S.L. Anderson (University of Bristol); P. Upchurch (University College London); and M. Wills (University of Bath) for use of copies of his disparity programs. MTY is funded by the Natural Environment Research Council (grant NER/S/A/2006/14058) and the Natural History Museum, London. This analysis is derived from a study that was in partial fulfilment of an MSc at University of Bristol by MDL.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Young, M.T., Larvan, M.D. (2010). Macroevolutionary Trends in the Skull of Sauropodomorph Dinosaurs – The Largest Terrestrial Animals to Have Ever Lived. In: Elewa, A. (eds) Morphometrics for Nonmorphometricians. Lecture Notes in Earth Sciences, vol 124. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-95853-6_11
Download citation
DOI: https://doi.org/10.1007/978-3-540-95853-6_11
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-95852-9
Online ISBN: 978-3-540-95853-6
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)