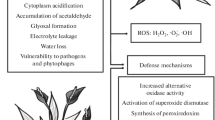
Abstract
In their ecosystem, plants have to cope with a plethora of potentially unfavourable conditions. Stress factors affecting plant’s fitness not only derive from natural sources, such as adverse temperature fluctuations (heating, chilling and freezing), high irradiance (photoinhibition, photooxidation), osmotic imbalance (salinity and drought), hypoxia/anoxia (flooding), mineral (macro- and micronutrient) deficiency, wounding, phytophagy and pathogen attack, but also from anthropogenic activities. The latter include xenobiotics employed in agriculture (herbicide, pesticides and fungicides), environmental (air, soil and water) pollutants and increased UV radiations. Particularly, many atmospheric pollutants, belonging to greenhouse gases, may increase the greenhouse effect, a natural warming process that prevents heat from diffusing to the outer atmosphere, thus balancing Earth cooling processes. Without the natural greenhouse effect, temperature on Earth would be much lower than it is now, and the existence of life would have not been possible. However, the rising emissions of greenhouse gases due to anthropogenic activities, namely carbon dioxide (CO2), chlorofluorocarbons (CFCs), nitrous oxide (N2O), tropospheric ozone (O3) and water vapour, may cause a short-term increase of the mean global temperature on the planet surface with consequent changes in precipitation patterns (Krupa and Kickert 1989). In this scenario, life on the earth depended from the co-evolution between atmosphere and biosphere, because the gradual and long-term climate changes enabled living organism adaptation to the new temperatures, precipitation patterns and other climate conditions (Voronin and Black 2007).
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Cinnamyl Alcohol Dehydrogenase
- Ozone Exposure
- Chem Ecol
- Biogenic Volatile Organic Compound
- Plant Secondary Metabolism
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Abe I, Rohmer M, Prestwich GD (1993) Enzymatic cyclization of squalene and oxidosqualene to sterols and triterpens. Chem Rev 93:2189–2206
Alstad DN, Edmunds GF, Weinstein LH (1982) Effects of air pollution on insect populations. Annu Rev Entomol 27:369–384
Aycock MK (1975) Phenotypic correlation of weather fleck with certain agronomic and chemical traits in Maryland tobacco. Tobacco Sci 19:96–97
Bach TJ (1987) Synthesis and metabolism of mevalonic acid in plants. Plant Physiol Biochem 25:163–178
Bagni T, Tassoni A (2001) Biosynthesisn oxidation and conjugation of aliphatic polyamines in higher plants. Amino Acids 20:301–317
Baumbach G, Vogt U (2003) Influence of inversion layers on the distribution of air pollutants in urban areas. Water Air Soil Pollut Focus 3:67–87
Bors W, Langebartels C, Michel C, Sandermann H (1989) Polyamines as radical scavengers and protectans agaist ozone damages. Phytochemistry 28:1589–1595
Bouchereau A, Aziz A, Larher F, Martin-Tanguy J (1999) Polyamines and environmental challenges: recent development. Plant Sci 140:103–125
Cabané M, Pireaux J-C, Léger E, Weber E, Dizengremel P, Pollet B, Lapierre C (2004) Condensed lignins are synthesized in poplar leaves exposed to ozone. Plant Physiol 134:586–594
Cane DE (1990) Enzymatic formation of sesquiterpenes. Chem Rev 90:1089–1103
Chappell J (1995) The biochemistry and molecular biology of isoprenoid metabolism. Plant Physiol 107:6
Chappelka AH, Kraemer ME, Mebrahtu T, Rangappa M, Benepal PS (1988) Effects of ozone on soybean resistance to Mexican bean beetle (Epilachna varivestis Mulsant). Environ Exp Bot 28:53–60
Cheng A-X, Lou Y-G, Mao Y-B, Lu S, Wang L-J, Chen X-Y (2007) Plant terpenoids: biosynthesis and ecological functions. J Integr Plant Biol 49:179–186
Cordell GA, Quinn-Beattie ML, Farsnsworth NR (2001) The potential of alkaloids in drug discovery. Phytot Res 15:183–205
Criegee R (1957) The course of ozonation of unsaturated compounds. Record Chem Progr 18:110–120
Croteau R (1987) Biosynthesis and catabolism of monoterpenoids. Chem Rev 87:929–954
Crutzen PJ, Lelieveld J (2001) Human impacts on atmospheric chemistry. Ann Rev Earth Planet Sci 29:17–45
Dercks W, Trumble JT, Winter C (1990) Impact of atmospheric pollution on linear furanocoumarin in celery. J Chem Ecol 16:443–454
Dicke M (2000) Chemical ecology of host-plant selection by herbivorous arthropods: a multitrophic perspective. Biochem. Syst Ecol 28:601–617
Dogbo A, Camara B (1987) Purification of isopentenyl pyrophosphate isomerase and geranylgeranyl pyrophosphate synthase from Caspicum chromoplasts by affinity chromatography. Biochim Biophys Acta 92:140–148
Dutsch HV (1978) Vertical ozone distribution on a global scale. Pure Appl Geophys 116:511–529
Eckey-Kaltenbach HE, Ernst D, Heller W, Sandermann H (1994) Biochemical plant response to ozone IV. Cross-induction of defensive pathways in parsley (Petroselinum crispum L.) plants. Plant Physiol 104:67–74
Endress AG, Post SC (1985) Altered feeding preferences for Mexican bean beetle Epilachna varivestis for ozonated soybean foliage. Environ Pollut 39:9–16
Evans LS, Ting IP (1973) Ozone-induced membrane permeability changes. Am J Bot 60:155–162
Facchini PJ (2001) Alkaloid biosynthesis in plants: biochemistry, cell biology, molecular regulation and metabolic engineering. Ann Rev Plant Physiol Plant Mol Biol 52:29–66
Fowler D, Cape JN,Coyle M, Smith RI, Hjellbrekke A-G, Simpson D, Derwent RG, Johnson CE (1999) Modelling photochemical oxidant formation, transport, deposition and exposure of terrestrial ecosystems. Environ Pollut 100:43–55
Frank SA (1993) Coevolutionary genetics of plant and pathogens. Evolut Ecol 7:45–75
Freeman BA, Mudd JB (1981) Reaction of ozone with sulfhydryls of human erythrocytes. Arch Biochem Biophys 208:212–220
Foy D, Lee EH, Rowland R, Devine TE, Buzzel RI (1995) Ozone tolerance related to flavonol glycoside genes in soybean. J Plant Nutr 18:637–647
Fuhrer J (2003) Agroecosystem responses to combinations of elevated CO2, ozone and global climate change. Agric Ecosys Environ 97:1–20
Galliano H, Heller W, Sandermann H (1993) Ozone induction and purification of spruce cinnamyl alcohol dehydrogenase. Phytochemistry 32:557–563
Goudrian J, Zadoks JC (1995) Global climate change: modelling the potential response of agro-ecosystems with special reference to crop protection. Environ Pollut 87:215–224
Grunwald C (1971) Effects of free sterols, steryl ester and steryl glycoside in membrane permeability. Plant Physiol 48:653–655
Grunwald C (1974) Effects of free sterols, steryl ester and steryl glycoside in membrane permeability. Plant Physiol 54:624–628
Grunwald C, Endress AG (1985) Foliar sterols in soybeans exposed to chronic levels of ozone. Plant Physiol 77:245–247
Guidi L, Degl’Innocenti E, Genovesi S, Soldatini GF (2005) Photosynthetic process and activities of enzymes involved in the phenylpropanoid pathway in resistant and sensitive genotypes of Lycopersicon esculentum L. exposed to ozone. Plant Sci 168:153–160
Hahlbrock K, Scheel D (1989) Physiology and molecular biology of phenylpropanoid metabolism. Ann Rev Plant Physiol Plant Mol Biol 40:347–369
Halliwell B (2006) Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 141:312–322
Heagle AS, Branderburg RL, Burns JC, Miller JE (1994) Ozone and carbon dioxide effects on spider mites in white clover and peanut. J Environ Qual 23:1168–1176
Heagle AS, Miller JE, Pusley WA (1998) Influence of ozone stress on soybean response to carbon dioxide enrichment III. Yield and seed quality. Crop Sci 38:128–134
Heagle AS, Miller JE, Booker FL, Pusley WA (1999) Ozone stress, carbon dioxide enrichment and nitrogen fertility interactions in cotton. Crop Sci 39:731–741
Heagle AS, Miller JE, Pusley WA (2000) Effects of ozone and carbon dioxide interactions on growth and yield of winther wheat. Crop Sci 40:1656–1664
Heiden AC, Hoffmann T, Kahl J, Kley D, Klockow D, Langebartels C, Mehlhorn H, Sandermann H, Schraudner M, Schuh G, Wildt J (1999) Emission of volatile organic compounds from ozone-exposed plants. Ecolog Appl 9:1160–1167
Heiser I, Elstner EF (1998) The biochemistry of plant stress and disease. Ann NY Acad Sci 851:224–232
Holstein SA, Hohl RJ (2004) Isoprenoids: remarkable diversity of form and function. Lipids 34:293–309
Hughes EH, Shanks JV (2002) Metabolic engineering of plants for alkaloid production. Metab Engin 4:41–48
Hussain MZ, Mustafa MG, Ghani QP, Bhatnagar RS (1985) Stimulation of poly(ADP-ribose) synthetase activity in the lungs of mice exposed to a low level of ozone. Archiv Biochem Biophys 241:477–485
Ignatenko AV, Cherenkevich SN, Komyak AI (1984) Chromatographic and spectroscopic investigation of the products of oxidation of tyrosine with ozone. J Appl Spectrosc 41:159–164
Iriti M, Faoro F (2004) Plant defense and human nutrition: the phenylpropanoids on the menu. Curr Top Nutrac Res 2:47–95
Iriti M, Faoro F (2006) Lipid biosynthesis in Spermatophyta. In: Teixeira da Silva JA (ed) Floriculture, Ornamental and Plant Biotechnology Vol. I Global Science Books, UK, pp 359–372
Iriti M., Faoro F (2007) Review on innate and specific immunity in plants and animals. Mycopathol 164:57–64
Jackson DM, Rufty TW, Heagle AS, Severson RF, Eckel RVW (2000) Survival and development of tobacco hornworm larvae on tobacco plants grown under elevated levels of ozone. J Chem Ecol 26:1–19
Jeandet P, Sbaghi M, Bessis R, Meunier P (1995) The potential relationship of stilbene (resveratrol) synthesis to anthocyanin content in grape berry skins. Vitis 34:91–94
Jeffords MR, Endress AG (1984) Possible role of ozone in tree defoliation by the gypsy moth (Lepidoptera: Lymantriidae). Environ Entomol 13:1249–1252
Karonen M, Ossipov V, Ossopova S, Kapari L, Loponen J, Matsumura H, Kohno T, Mikami C, Sakai Y, Izuta T, Pihlaja K (2006) Effects of elevated carbon dioxide and ozone on foliar proanthocyanidins in Betula platyphylla, Betula ermanii and Fagus crenata seedlings. J Chem Ecol 32:1445–1458
Keen NT, Taylor OC (1975) Ozone injury in soybeans: isoflavonoid accumulation is related to necrosis. Plant Physiol 55:731–733
Kelly FJ, Mudway I, Krishna MT, Holgate ST (1995) The free radical basis of air pollution: focus on ozone. Respiratory Med 89:647–656
Kerr JB, McElroy CT (1993). Evidence for large trend of ultraviolet-B radiation linked to ozone depletion. Science 262:1032–1034
Kesselmeier J, Staudt M (1999) Biogenic volatile organic compounds (VOC): an overview on emission, physiology and ecology. J Atmos Chem 33:23–88
Kley D, Kleinmann M, Sandermann H, Krupa S (1999) Photochemical oxidants: state of the science. Environ Pollut 100:19–42
Krupa SV, Kickert RN (1989) The greenhouse effect: impacts of ultraviolet-B (UV-B) radiation, carbon dioxide (CO2) and ozone (O3) on vegetation. Environ Pollut 50:263–293
Langebartels C, Kerner K, Leonardi S, Schraudner M, Trost M, Heller W, Sandermann H (1991) Biochemical plant resonses to ozone I: differential induction of polyamine and ethylene biosynthesis in tobacco. Plant Physiol 95:882–889
Langebartels C, Wohlgemuth H, Kschieschan S, Grü n S, Sandermann H (2002) Oxidative burst and cell death in ozone-exposed plants. Plant Physiol Biochem 40:567–575
Lichtenthaler HK (1998) The stress concept in plants: an introduction. Ann NY Acad Sci 851:187–198
Liu J-H, Kitashiba H, Wang J, Ban Y, Moriguchi T (2007) Polyamines and their ability to provide environmental stress tolerance to plants. Plant Biotechn 24:117–126
Llusià J, Peñuelas J, Gimeno BS (2002) Seasonal and species-specific response of VOC emissions by Mediterranean woody plant to elevated ozone concentrations. Atmos Environ 36:3931–3938
Logan JA (1985) Tropospheric ozone: seasonal behaviour, trends and anthropogenic influences. J Geophys Res 90:10463–10482
Lorenzini G, Medeghini-Bonatti, Nali C, Baroni Fornasiero R (1994) The protective ffect of rust infection against ozone, sulphur dioxide and paraquat toxicity symptoms in broad bean. Physiol Mol Plant Pathol 45:263–279
Loreto F, Velikova V (2001) Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products and reduces lipid peroxidation of cellular membrane. Plant Physiol 127:1781–1787
Loreto F, Ferranti F, Mannozzi M, Maris C, Nascetti C, Pasqualini S (2001) Ozone quenching properties of isoprene and its antioxidant role in plants. Plant Physiol 126:993–1000
Loreto F, Pinelli P, Manes F, Kollist K (2004) Impact of ozone on monoterpene emission and evidence for an isoprene-like antioxidant action of monoterpenes emitted by Quercus ilex leaves. Tree Physiol 24:361–367
Manning WG, von Tiedemann A (1995) Climate change: potential effects of increased atmospheric carbon dioxide (CO2), ozone (O3) and ultraviolet-B (UV-B) radiation on plant diseases. Environ Pollut 88:219–245
Martin-Tanguy J (2001) Metabolism and function of polyamines in plants: recentdevelopment (new approaches). Plant Growth Regul 34:135–148
McKee IF, Farage PK, Long SP (1995) The interactive effects of elevated CO2 and O3 concentration on photosynthesis spring wheat. Photosynth Res 45:111–119
McKee IF, Eiblmeier M, Polle A (1997) Enhanced ozone-tolerance in wheat grown at an elevated CO2 concentration: ozone exclusion and detoxification. New Phtytol 137:275–284
Menser HA, Chaplin JF (1969) Air pollution: effects of phenol and alkaloid content of cured tobacco leaves. Tobacco Sci 13: 69–170
Menser HA, Chaplin JF (1975) Effects of ozone air pollution on nitrogen constituents, pH and water soluble ash of air-cured tobacco leaves. Tobacco Sci 19:108–110
Menser HA, Chaplin JF, Cheng ALS, Sorokin T (1977) Polyphenols, phytosterols and reduced sugar in air-cured tobacco leaves injured by ozone air pollution. Tobacco Sci 21:35–38
Mudd JB, Leavitt R, Ongun A, McManus TT (1969) Reaction of ozone with amino acids and proteins. Atmos Environ 3:669–682
Mustafa MG (1990) Biochemical basis of ozone toxicity. Free Radicals in Biol and Med 9:245–265
Navakoudis E, Lütz C, Langebartels C, Lütz-Meindl, Kotzabasis K (2003) Ozone impact on the photosynthetic apparatus and theprotective role of polyamines. Biochim Biophys Acta 1621:160–169
Ormrod DP, Beckerson DW (1986) Polyamines as antiozonant for tomato. HortScience 21:1070–1071
Pääkkönen E, Seppänen, S, Holpainen T, Kokko H, Kärenlampi S, Kärenlampi L, Kangasjärvi (1998) Induction of genes for stress proteins PR-10 and PAL in relation to growth, visible injury and stomatal conductance in birch (Betula pendula) clones exposed to ozone and/or drought. New Phytol 138:295–305
Paiva N (2000) An introduction to the biosynthesis of chemicals used in plant-microbe communication. J Plant Growth Regul 19:131–143
Paolacci AR, D’Ovidio R, Marabottini R, Nali C, Lorenzini G, Abenavoli MR, Badiani M (2001) Ozone induces a differential accumulation of phenylalanine ammonia-lyase, chalcone synthase and chalcone isomerase RNA transcripts in sensitive and resistant bean cultivars. Astr J Plant Physiol 28:425–428
Peñuelas J, Llusià J (2001) The complexity of factors driving volatile organic compound emissions by plants. Biol Plant 44:481–487
Platt U, Hönninger G (2003) The role of halogen species in the troposphere. Chemosphere 52:325–338
Poulter CD, Rilling HC (1978) The prenyl transferase reaction, enzymatic and mechanistic studies of the 1′-4 coupling reaction in the terpene biosynthetic pathway. Acc Chem Res 1:307–313
Pryor WA, Das B, Church DF (1991) The ozonation of unsaturated fatty acids: aldehydes and hydrogen peroxide as products and possible mediators of ozone toxicity. Chem Res Toxicol 4:341–348
Pryor WA (1992) How far does ozone penetrate into the pulmonary air/tissue boundary before it reacts? Free Rad Biol Medic 12:83–88
Pryor WA (1993) Ozone in all reactive splendour. J Laborat Clin Medic 122:483–486
Pryor WA (1994) Mechanisms of radical formation from reactions of ozone with target molecules in the lung. Free Rad Biol Medicine 17:451–465
Rao MV, Davis KR (2001) The physiology of ozone-induced cell death. Planta 213:682–690
Roselle SJ (1994) effects of biogenic emission uncertainties on regional photochemical modelling of control strategies. Atmos Environ 28:1757–1772
Rowland-Bamford AJ, Borland AM, Lea PJ, Mansfield TA (1989) The role of arginine decarboxylase in modulating the sensitivity of barley to ozone. Environ Pollut 61:95–106
Sacchettini JC, Poulter CD (1997) Creating isoprenoid diversity. Science 277:1788–1789
Saleem A, Loponen Y, Pihlaja K, Oksanen E (2001) Effects of long-term open-field ozone exposure on leaf phenolics of european silver birch (Betula pendula Roth). J Chem Ecol 27:1049–1062
Schraudner M, Ernst D, Langebartels C, Sandermann H 1992 Biochemical plant responses to ozone III. Activation of the defence-related proteins β-1,3-glucanase and chitinase in tobacco leaves. Plant Physiol 99:1321–1328
Schubert R, Fischer R, Hain R, Schreier PH, Bahnweg D, Ernst H, Sandermann H (1997) An ozone-responsive region of the grapevine resveratrol synthase promoter differs from the basal pathogen-responsive sequence. Plant Mol Biol 34:417–436
Sgarbi E, Baroni Fornasiero R, Lins AP, Medeghini Bonatti P (2003) Phenol metabolism in differentially affected by ozone in two cell lines from grape (Vitis vinifera L.) leaf. Plant Sci 165:951–957
Shamay Y, Raskin VI, Brandis AS, Steinberger HE, Marder JB, Schwartz A. (2001) Ozone treatment affects pigment precursor metabolism in pine seedlings. Physiol Plant 112:285–292
Sharma YK, Davis KR (1994) Ozone-induced expression of stress-related genes in Arabidopsis thaliana. Plant Physiol 105:1089–1096
Sharma YK, Leon J, Raskin I, Davis KR (1996) Ozone-induced responses in Arabidopsis thaliana: the role of salicylic acid in the accumulation of defense-related transcripts and induced resistance. PNAS USA 93:5099–5104
Thalmair M, Bauw G, Thiel S, Dohring T, Langebartels C, Sandermann H (1996) Ozone and ultraviolet B effects on the defence-related proteins β-1-3-glucanase and chitinase in tobacco. J Plant Physiol 148:222–228
Tomlinson H, Rich S (1971) Effect of ozone on sterols and sterol derivatives in bean leaves. Phytopathology 61:1404–1405
Toumainen J, Pellinen R, Roy S, Kiiskinen M, Eloranta T, Karjalainen R, Kangasjärvi J (1996) Ozone affect birch (Betula pendula Roth) phenylpropanoid, polyamine and reactive oxygen detoxifying pathways at biochemical and gene expression levels. J Plant Physiol 148:179:188
Trevathan LE, Moore LD, Orcutt DM (1979) Symptom expression and free sterol and fatty acid composition of flue-cured tobacco plants exposed to ozone. Phytopathology 69:582–585
Trumble JT, Hare JD, Musselmann RC, McCool PM (1987. Ozone-induced changes in host-plant suitability: interactions of Keiferia lycopersicella and Lycopersicon esculentum. J Chem Ecol 13:2003–218
Velikova V, Pinelli P, Pasqualini S, Reale L, Ferranti F, Loreto F (2005) Isoprene decreases the concentration of nitric oxide in leaves exposed to elevated ozone. New Phytologist 166:419–426
Violini G (1995) Ozone and plant pathogens: an overview. Riv Pat Veg 5:113–130
Von Tiedemann A, Firsching KH (2000) Interactive effects of elevated ozone and carbon dioxide on growth and yield of leaf rust-infected versus non-infected wheat. Environ Pollut 72:205–224
Voronin PYu, Black CC (2007) Earth’s atmosphere as a result of coevolution of geo- and biospheres. Russ J Plant Physiol 54:132–136
Walters D (2000) Polyamines in plant-microbe interactions. Physiol Mol Plant Pathol 57:137–146
Wang W-C, Liang X-Z, Dudek MP, Pollard D, Thompson SL (1995) Atmospheric ozone as climate gas. Atmos Res 37:247–256
Weaver LM, Hermann KM (1997) Dynamics of the shikimate pathway in plants. Trends Plant Sci 2:346–351
Wegener A, Gimbel W, Werner T, Hani J, Ernst D, Sandermann H (1997) Molecular cloning of ozone-inducible protein from Pinus sylvestris L. with high sequence similarity to vertebrate 3-hydroxy-3-methylglutaryl-CoA-synthase. Biochim Biophys Acta 1350:247–252
Wendt KU, Schulz GE (1998) Isoprenoid biosynthesis: manifold chemistry catalysed by similar enzymes. Curr Biol 6:127–133
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Iriti, M., Faoro, F. (2009). Ozone-Induced Changes in Plant Secondary Metabolism. In: Singh, S.N. (eds) Climate Change and Crops. Environmental Science and Engineering. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-88246-6_11
Download citation
DOI: https://doi.org/10.1007/978-3-540-88246-6_11
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-88245-9
Online ISBN: 978-3-540-88246-6
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)