Abstract
Periprosthetic joint infection is the most severe complication in terms of mortality, morbidity and functional disability. Diagnosis is challenging and it is a frequent cause of repeated surgeries. Treatment depends on the general status of the patient, the bone status (stock) receiving the new implant, and the microbiology analysis. Diagnosis of infection include the type of pain; some signs as drainage and dehiscence of the wound; blood analysis; imaging assessment; articular aspiration, biopsy and cultures; sonication of retrieval implants; pathology and cultures of intraoperative samples. International Consensus has postulated different infection criteria. Once the infection is diagnosed and the surgery is decided, a multidisciplinary approach with the Microbiology and Infectious Disease Department is recommended to properly manage the antibiotherapy. The reconstruction of the acetabular bone defect in the infected hip include filling with antibiotic-loaded cement only for small cavitary defects and low-demand patients. Cementless implants are more widely used for small or moderate acetabular bone defects. The most serious cases with large bone acetabular defects will be managed with similar techniques in infected cases and aseptic cases, such as impacting bone grafting technique associated with a cemented cup, porous augments or metallic rings.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Periprosthetic joint infection (PJI) is the most severe complication in terms of mortality, morbidity and functional disability. The cost of a septic revision surgery is 3–4 times greater than primary total hip arthroplasty , and double that of an aseptic revision surgery [1, 2]. Frequency varies depending on different etiopathogenic factors such as local contamination, hematogenous contamination and well as patient-associated factors. Local contamination is related with the type of surgical theater (laminar air flow and space suits, less than 1%), the existance of previous surgery [3], dislocations, surgical wound secretion [4], skin coverage defect [5], and antibiotic prophylactic non-use that can multiply the infection risk by seven [6]. Hematogenous contamination is related with the existance of distant septic foci (urinary, dental), and multiplies the risk of infection by three [6]. In regard to patient-associated risk factors, the existance of associated diseases, such as diabetes, rheumatoid arthritis, tuberculosis etc.; the abuse of some drugs and toxic-substances, such as corticotherapy, alcoholism, or drug addiction can also increase the risk of infection [7]. Older ages can also produce immunodepresion associated to progressive thymus gland atrophy (the site of T cell maduration), delayed hypersensibility and weaker lymphocyte response [8]. Nutritional alterations including obesity or deficiencies are also related with higher infection rates.
Diagnosis is challenging and it is a frequent cause of repeated surgeries that affect not only soft-tissues around a joint but also bone. Prosthetic joint infection is less frequent in the hip than in the knee, management is significantly different. Systemic antibiotic therapy after positive intraoperative cultures subsequent to a revision procedure, single- or two-stage procedures depending on the general status of the patient, the bone status (stock) receiving the new implant, and the microbiology analysis, all affect outcome. Controversial issues like temporary spacer use, have been reported. The low number of cases and the differences in patient’s characteristics make the analysis of clinical studies the surgeon’s decision very difficult.
Diagnosis of Infection
Risk Factors and Clinical Suspicion
During the last decades, many studies have reported some clinical and laboratory issues to help to identify risk factors in individual patients and enable post-operative evaluation of failed arthroplasty, determining the probability of infection in high versus low risk patients or situations; this information should help the surgeon decide on a more or less aggressive response to of possible infection [9].
Symptoms: Pain
When the surgeon evaluates a patient with a painful hip arthroplasty all important questions should be replanted including whether the pain had presented recently arises, if it is different than before surgery, or whether it could be related to other sites of infection. Pain in infection is not always “inflammatory”, many times “mechanical” pain occurs, when walking and standing, particularly if there is loosening, or a continuous pain that is not alleviated by rest. At this point, it is important to identify “over-demanding” patients (those expecting complete absence of pain and unlimited walking/activity) through the quantity and quality of analgesics, life style, their actual functional limitations, by the use of internationally-accepted patient reported outcomes.
Signs
During physical examination most International Scientific Societies and Consensus Systems include some signs as major criteria of infection like persistent drainage and a chronics in us with or without the abscence of associated issues such as edge necrosis or wound dehiscence [10, 11]. Most authors agree that these signs are critical even a case shows a negative culture. To date, some controversial studies have reported that simply culturing fistula samples is inadequate while others supports the 80% agreement between cultures of fistula and deep tissue infection [12]. Other inflammatory changes (redness, swelling, and/or increase of local temperature) are less specific and require confirmation. When inflammatory changes appear the clinician will have doubts; these signs need confirmation further study.
Blood Analysis
The most commonly-used laboratory markers are increases in of white cells blood counts, erithrocyte sedimentation rate (ESR) and C-reactive protein (CRP). Erithrocyte sedimentation rate is usually higher than 30 in theses patients, but ESR sensitivity is between 66% and 82%, and the specificity between 85% and 90%. A CRP above one has with a sensitivity between 77% and 96%, and specificity between 84% and 92% for infection. However, sensitivity and specificity increase when are combined showing a high positive (83%) and negative (92–100%) predictive value [13,14,15].
The normal elevation of CRP usually decreases after three weeks and ESR after 6 or even 12 weeks. Other comorbidity problems like another infection affecting the urinary or teeth or rheumatological conditions may also elevate these parameters, so the clinician needs to be aware. Other serum markers like procalcitonin or IL-6 have been investigated but further evidence and cost-efectiveness studies are recommended to confirm their usefulnes. Bottner et al. report a not high sensitivity for these markers: 87% for IL6, 33% for Procalcitonin, and 43% for TNF [16]. Recently, D-dimer seems to be another promising and easy to evaluate marker when compared to combined ESR and PCR values [17].
Imaging assessment
Imaging assessment is also controversial, since simple radiographs can show signs of prosthetic radiographic loosening, such as migration of the components, subsidence of the stem, radiolucent lines wider 2 mm around all the contour of the socket and the stem. Suspicion of infection arises when these signs appear early, present a fast progression, and/or there is an absence of mechanical explanation. The appearance of bone cysts and osteolysis unrelated to radiographic polyethylene wear strongely suggest infection when they are focal, rapidly progressive and massive. Early periosteal reaction (periostitis) is also frequent in these patients [18, 19] (Fig. 12.1).
Anteroposterior radiographs of hip. Early signs of prosthetic radiographic loosening, such as component migration, stem subsidence, radiolucent lines wider than 2 mm all around the contour of the socket and the stem strongely suggest infection. Bone cysts and osteolysis are unrelated to significant radiographic polyethylene wear
The use of Computed tomography(CT) associated with radiographs can be useful in helping to confirm acetabular and femoral bone loss particularly when combined with other signs like secuestrum and cortical defects. Periprosthetic fluid collections can also be diagnosed using ultrasound, CT, or MRI [10, 19].
An even more controversial topic is the use of conventional scintigraphy [20, 21]. Tecnecium and Galium scintigraphy become positive when bone turnover is increased and remain high during first year after any arthroplasty due to secondary implant fixation. Labeled leukocytes [21, 22] have shown high specificity but low sensitivity. Galium scintigraphy is positive when inflammation is present, so it is always positive during secondary fixation of THA. It has high sensitivity (95%) but very poor specificity (20%). This makes it useful for negative diagnosis (to confirm no infection is present). CT images of bone marrow with labeled leukocytes shows increased in sensitivity and specificity (accuracy: 89–98%).
The accuracy of fluorodeoxyglucose positron emission tomography (PET) in detecting differences between aseptic and septic failure has lead to an increased use of this technique, although it is still difficult to access and expensive [10, 23]. There also are doubts about its accuracy, which may be less than that of CT combined with labeled leukocytes.
Articular aspiration
A preoperative analysis of articular liquid has been also recommended. Some technical issues make this management somewhat difficult, requiring an aseptic preparation (operation room), and these are some imaging techniques like radiographs, ultrasonography or CT that can help determine the best sample [14]. A liquid sample in a blood-culture bottle is much better than a swab [24]. Another controversial issue with articular aspiration is that a needle can only obtain planctonical bacteria, and the biofilm is not analyzed.
The leukocyte level diagnostic ranges are quite controversial and many authors have proposed very different thresholds: >1800 [10, 11], >2700, >3000 in chronic cases [25], or >4350. The sensitivity of this parameter is 90% (>1800) and the specificity: 99% (>1800). The proportion of polymorphonuclear(PMN) cells is also very controversial because many authors again have proposed quite different thresholds: >73% [10, 11], >77%, >80%in chronic cases [25], and >85%. PMN level sensitivity is 83% (>73%), and its specificity 93% (>73%). The use of leukocyte esterase reagent strips, offers a good sensitivity (93%) with low specificity (77%), but 33% of the samples are unuseful because of blood or debris [26]. Alpha-defensin determination has a sensitivity of 97–100% and specificity of 95–97% [27, 28]. These new markers are not significantly better than the more traditional ESR, CRP, leukocyte count and/or PMN [27).
Gram staining has a very low sensitivity (50–75%). Aspirate cultures only show a sensitivity of 56–92%. The specificity is 95%, but very low with skin contaminants (S epidermidis, Propionibacterium sp) [14, 29,30,31,32,33,34].
Biopsy. Culture and Diagnosis of Infection
So, biopsy appears to be a better option than aspiration, offering a sensitivity and specificity ranging between 82% and 98% [35], which increases with the use of arthroscopic basketsup to 88–100% [36].
Intraoperative Diagnosis of Infection
Macroscopical Observations. Are They Useful?
An intraoperative diagnosis will usually confirm the preoperative diagnoses. Subjective, macroscopic observations based on the surgical teams experience and sensorial perceptions (eyes and noses) are important. Photographs ought to be taken. The Musculoskeletal Infection Diseases Society criteria includes gross purulence, great amount of liquid, necrotic and devitalized tissues or smell characteristics as signs of infection [10, 25]. To date, some types of bacteria can be suspected based on the appearance of the purulence. For example, a creamy, yellowish-white, abundant pus, suggests S. Aureus; scarce purulence, dirty interfaces, no smell, and undefinite color suggest S. Epidermidis; abundant exudation and “urine-like” smell suggest E Coli, Proteus spp.; a fecal appearance and/or smell suggest Enterobacter, Enterococcus, Klebsiella and Serratia; asewer smell, Pseudomonas spp.; and strong acid/sour smell: Streptococcus spp.
Gram staining has a sensitivity below 17%, so, itis not useful at all [30]. The use of frozen sections can supply the number of PMN cells per high-power microscopic field [37,38,39,40,41,42,43,44,45]. A figure higher than 10 PMN suggest probable infection (implant extraction). From 5 to less than 10 PMN: possible infection. A figure below than 5 PMN: very low probability of infection. However, Gram staining has very low sensitivity in suspected aseptic loosening (50%) and reimplantation surgery (second stage of two-stage exchange) (29%) [38].
Postoperative Diagnosis of Infection
Conventional Cultures
Conventional recommendations for culturing include samples obtained from synovial fluid and tissues (synovial, devitalized soft-tissues, periprosthetic membranes, intramedullary bone content) [31]. Liquids only have planctonic bacteria, while tissuescan provideintracellular as well as sessile (from biofilm) bacteria [46, 47]. At least three periprosthetic tissue specimens, ideally five to six samples must be taken before irrigation is started [11, 14, 30, 31, 48].
Sonication of Retired Implants
Sonication of retired implants has been reported to be of great value due to its sensitivity, specificity and ability to supply material for quantitative cultures and culture-media for slow-growing microorganisms. It is always recommended and the resulting cultures show a sensitivity of 78%, and specificity of 99% [15]. Associated with quantitative cultures and culture-media for slow-growing microorganisms, sensitivity improves but specificity is worse [47]. There is no consensus if as yet on usually-non-pathogenic bacteria obtained by sonication are actually responsible for clinical syndromes? [49, 50].
Pathology of Intraoperative Samples
Pathologists can be very helpful since frozen sections are still probably the most accurate intraoperative test giving the number of PMN cells per high-power microscopic field [51]. Also quantification of sinovial CRP hace been reported to be evaluable as study of frozen sections [52].There are some doubts as to when the neutrophils are the immune response against plantonic bacteria, and macrophages are the immune response against biofilm. But, how do we differentiate between macrophages against wear particles and those against biofilm?. The pathological report on intraoperative samples is affected by the macrophage response to wear particles and biofilm.
Infection Criteria
Different infection criteria have been postulated based on an International Consensus in order to clearly define this complication [25]. Any of the following will indicate a deep infection: (1) There is a sinus tract communicating with the prosthesis; (2) A pathogen is isolated by culture from at least two separate tissue or fluid samples obtained from the affected prosthetic joint; or (3) Four of the following six criteria exist:
-
1.
Elevated serum erythrocyte sedimentation rate (ESR) and serum C-reactive protein (CRP) concentration,
-
2.
Elevated sinovial leukocyte count,
-
3.
Elevated sinovial neutrophil percentage (PMN%),
-
4.
Presence of purulence in the affected joint,
-
5.
Isolation of a microorganism in one culture of periprosthetic tissue or fluid, or
-
6.
More than five neutrophils per high-power field in five high-powe fields observed from histologic analysis of periprosthetic tissue at ×400 magnification.
The presence of a sinus tract, visible pus surrounding the joint without other explanation (e.g. no crystals), acute inflammation on histopathological examination (>5 neutrophils/high-powerfield), >4200 leukocyte per μl and/or >80% polymorphonuclear leukocytes in synovial fluid, growth of the same microorganism in at least two cultures of synovial fluid, peri-prosthetic issue and/or sonication fluid. Patients were classified into acute postoperative (≤1 month after implantation), acute hematogenous (≤3 weeks of infectious symptoms), and chronic PJI (all other situations).
Major criteria are two positive periprosthetic cultures with identical microroganisms, and sinus tract communication with the joint. Minor criteria are: elevated serum ESR and CRP, elevated synovial fluid white blood cell count or leucocyte esterase test strip, elevated PMN synovial fluid, positive histological analysis, a single positive culture.
This complex problem is critically important to making the right decision regarding management.
Treatment of the Infected Total Hip Arthroplasty
Once the infection is diagnosed, the surgeon must keep several considerations in mind. Particularly is important the time element (early or late), the severity of the infection according to the microbiological analysis and the comorbidity and medical situation of the patient. Once surgery is decided, the question of a single- or two-stage procedure, the local status of soft tissues, surgical and hospital environment must be taken into account in order to manage the most important aspect in hip revision reconstruction: bone loss.
A multidisciplinary approach with the Microbiology and Infectious Disease Department is recommended to properly manage the antibiotherapy. In a few cases a suppressive therapy (unexpected intraoperative cultures in an aseptic revision procedure or very fragile patients with previous repeated surgeries) may be indicated, but, in most cases surgery should be done.
According to the origin of infection and biofilm formation, the implant must be removed whenever possible. A single surgical debridement is often considered controversial due to its inefficacy because bacteria create biofilm in less than 24 h, and thus a single- or two-stage procedure may be indicated. Even more controversial, the surgeon must decide which components should be changed: First it was the polyethylene, then the femoral head, more recently all the uncemented and loose components (it is supposed that in the first 2–4 weeks they are not biologically fixed, but this may be not true with hydroxyapatite-coated pieces). The question arises immediately from a formal point of view: Is it a debridement, or is its a “one-stage” exchange?
Despite deciding on whether to do a single- or a two-stage procedure the management of bone loss is critical for the success of the problem. The surgeon must be aware that a bone defect is created after the implant’s removal so while preoperative planning may be helpful it is not definitive. Currently, aggressive debridement is recommended to remove all membranes from the bone as well as all necrotic and devitalized tissue so the infection can be healed before re-implanting the new prosthesis.
Acetabular Bone Defect in an Infected Total Hip Arthroplasty
As previously mentioned, it is critical to remove all necrotic soft-tissue and membranes. The bone defect will be determined after explanting the acetabular and femoral components, a point that will determine the re-implantation stage [53]. Another issue, when a two-stage procedure is performed is the use of spacer. Although widely used for their theoretical advantages in terms of soft tissue management and infection control [54,55,56,57,58,59,60], the high number of intrinsic complications can make considering the good results of not using attractive. The possibility of acetabular erosion when the spacer is left over long times is also a matter for concern [61, 62].
Girdlestone resection-arthoplasty (RA) can be a useful procedure to solve difficult cases such as severe infections with very severe bone defects, however, numerous studies have reported some unsatisfactory outcomes, including painful hips, the necessity of crutch use, severe limps, hip instability, leg length discrepancy (LLD) and increased oxygen consumption.
Considering the published results, the indications for RA of the hip must be strictly limited to non-ambulatory patients, intravenous drug abuse and impossible reimplantation because of new medical comorbidities or technical difficulties in medically compromised patients. Given these difficulties, some patients do not accept their condition as definitive and request conversion of their RA to a THA, hoping to alleviate symptoms and enhance their quality of life. The clinical outcome after conversion to a THA is unpredictable, so patients must be well informed regarding the expected results of this kind of complex procedure. It is uncommon, technically demanding and frequently associated with difficulties that can lead to complications resulting from some preoperative conditions like severe bone defects, osteoporotic bone, or LLD, which may make a shortening femoral osteotomy necessary, and abductor weakness.
Although the surgery may be technically demanding and the number of complications is not low, the clinical outcome for conversion from RA to THR is comparable to conventional revision surgery. Since the surgery is adapted to the bone defect in both groups, the defect may influence outcome more than the conversion procedure itself.
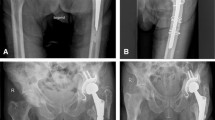
In fact, conversion of an RA, even after several years, can be done with an acceptable rate of complications (Fig. 12.2). Although dislocation is one of the concerns [63], this complication may be the result of inadequate cup positioning or soft tissue tension or both; at conversion, the soft tissues have invariably tightened, making proper testing of hip instability difficult and thus reconstruction of the hip rotation centre and proper management of the bone defect are more determinant of outcome rather than the use of constraint or dual mobility cups [64]. The number of peri-prosthetic fractures is usually low, and limited to intraoperative proximal cracks, which can be solved during the surgery. The circumstance that there is no need to remove a previous implant in an RA conversion may be one of the explanations for the relatively low number of complications, particularly for intraoperative fractures.
(a) Anteroposterior radiograph of a hip in a 72 year-old woman shows an infected cementless hip prosthesis. (b) Resection-arthroplasty. (c) Three months later, the hip was reconstructed using a cementless cup and impacting bone grafting with a cemented femoral long-stem showing a good outcome at 10 years
Functional outcome is commonly affected in many patients. Limp is frequent due to destruction of the soft tissue and poor active joint movement that may result in only fair improvement after re-implantation. When compared by bone defect and age the clinical outcome can be similar to that of an aseptic revision surgery, but nevertheless, pain, function, and motion are improved after conversion in most series [63, 65].
Age and bone defect, and/or physical status determine acetabular and femoral reconstruction [53, 66, 67]. RA in older low-demand patients may be adequate for some years, but muscular weakness and excessive oxygen consumption can be very disabling in this population [63]. Acetabular bone defect is the most important factor to affect postoperative clinical outcome, function, range of mobility and LLD so adequate reconstruction is critical to improve results in this complex situation. The low number of cases in most series makes it difficult to recommend the optimal interval between RA and conversion [68,69,70,71,72].
Despite THA after RA showing a clinical outcome and radiographic results similar to those obtained in aseptic revision surgery when the hips have similar bone defects, preoperative counseling must stress the influence of the intraoperative bone defect that will determine the surgical procedure and the risk of complications. Nevertheless, the fact that the functional level will be related to any resulting limp and LLD, mean a patients’ high expectations of improved hip function should be tempered to a realistic level.
Reconstruction of the Acetabular Bone Defect in the Infected Hip
Although an infected THA has been considered as a contraindication for bone graft due to the possibility of a potential sequestrum [73], many series have reported acceptable results and contributed to this option now being considered in some cases.
Filling with antibiotic-loaded cement should be recommended only for small cavitary defects and low-demand patients. Despite its efficacy in controlling infection, mechanically done filling can be less safe in a more active population.
Cementless implants are more widely used for small or moderate acetabular bone defects of less than 30% [67] since they provide better mechanical fixation in most cases. Infection healing has been reported to be similar to cemented fixation with contemporary cups, including “Jumbo” cups, and hemispherical tantalum cups (Fig. 12.3); tantalum supplements [19, 74,75,76,77,78]; and antiprotrusio cage plus tantalum augments [79]. Currently, It is not yet established how much healthy bone is necessary for integration using tantalum prosthesis [80].
The most serious cases with large bone acetabular defects will be managed with similar techniques in infected cases and aseptic cases (Fig. 12.4). Reconstruction of large bone defects with impacting bone grafting technique associated with a cemented cup has obtained very good results and low recurrence figures (from 0% to 8%) (Fig. 12.5) [47, 75, 81]. Antibiotic-loaded cements and vancomycin-loaded graft chips are recommended by the original and other centres [82, 83]. As in aseptic cases, the biological restoration of bone stock makes this technique very attractive, particularly in the most active and young patients. Other options like the use of reinforcement rings, trabecular metal augments or custom-made implants along with the allograft have also provided acceptable early results in patients with infection [84].
(a) Anteroposterior radiograph of the pelvis of a 83 year-old woman shows a resection-arthroplasty done 28 years earlier due to infection of a total hip arthroplasty. (b) The hip was reconstructed using a tantalum augment associated to bone impacting grafting technique with a cemented cup and bone impacting technique in the femur
References
Herbert CK, Williams RE, Levy RS, Barrack RL. Cost of treating of a total knee replacement. Clin Orthop Relat Res. 1996;331:140–5.
Bozic KJ, Ries MD. The impact of infection after total hip arthroplasty on hospital and surgeon resource utilization. J Bone Joint Surg Am. 2005;87:1746–51.
Tsukayama DT, Estrada R, Gustilo RB. Infection after total hip arthroplasty. A study of the treatment of one hundred and six infections. J Bone Joint Surg Am. 1996;78-A:512–23.
Ure KJ, Amstutz HC, Nasser S, Schmalzried TP. Direct exchange arthroplasty for the treatment of of infection after total hip replacement. An average tenýear follow-up. J Bone Joint Surg Am. 1998;80:961–8.
Lieberman JR, Callaway GH, Salvati EA, Pellicci PM, Brause BDI. Treatment of the infected total hip arthroplasty with a two-stage reimplantation protocol. Clin Orthop Relat Res. 1994;301:205–12.
Surin VV, Sundholm K, Bäckman L. Infection after total hip replacement. With special reference to a discharge from the wound. J Bone Joint Surg (Br). 1983 Aug;65(4):412–8.
Canner GC, Stenberg ME, Heppenstall RB, Balderstorn R. The infection hip after total hip arthroplasty. J Bone Joint Surg Am. 1984;66:1393–9.
Garvin KL, Evans BG, Salvati EA, Brause BD. Palacos gentamicin for the treatment of deep periprosthetic hip infections. Clin Orthop Relat Res. 1994;298:97–105.
Cordero-Ampuero J, de Dios M. What are the risk factors for infection in hemiarthroplasties and total hip arthroplasties? Clin Orthop. 2010;468:3268–77.
Parvizi J, Zmistowski B, Berbari EF, Bauer TW, Springer BD, Della Valle CJ, Garvin KL, Mont MA, Wongworawat MD, Zalavras CG. New definition for periprosthetic joint infection: from the workgroup of the musculoskeletal infection society. Clin Orthop Relat Res. 2011;469:2992–4. https://doi.org/10.1007/s11999-011-2102-9.
Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med. 2004;351:1645–54.
Cuñé J, Soriano A, Martínez JC, García S, Mensa J. A superficial swab culture is useful for microbiologic diagnosis in acute prosthetic joint infections. Clin Orthop Relat Res. 2009;467(2):531–5.
Sanzén L, Carlsson AS, Josefsson G, Lindberg LT. Revision operations on infected total hip arthroplasties. Clin Orthop Relat Res. 1988;229:165–72.
Spangehl J, Masri BA, O’Connell JX, Duncan CP. Prospective analysis of preoperative and intraoperative investigations for the diagnosis of infection of the sites of two hindred and teo revision total hip arthroplasties. J Bone Joint Surg Am. 1999;81:672–83.
Trampuz A, Hanssen AD, Osmon DR, Mandrekar JR, Steckelberg JM, Patel R. Synovial fluid leukocyte count and differential for the diagnosis of prosthetic knee infection. Am J Med. 2004;117:556–62.
Bottner E, Wegner A, Winkelmann W, Becker K, Erren M, Götze C. Interleukin-6, procalcitonin and TNF-alpha. Markers of peri-prosthetic infection following total hip replacement. J Bone Joint Surg (Br). 2007;89:94–9.
Shahi A, Kheir MM, Tarabichi M, Hosseinzadeh HRS, Tan TL, Parvizi J. Serum D-Dimer test is promising for the diagnosis of periprosthetic joint Infection and timing of reimplantation. J Bone Joint Surg Am. 2017;99(17):1419–27.
Della Valle C, Zuckerman JD, Di Cesare PE. Periprosthetic sepsis. Clin Orthop Relat Res. 2004;420:26–31.
Toms AD, Davidson D, Masri BA, Duncan CP. The management of peri-prosthetic infection in total joint arthroplasty. JBJS-Br. 2006;88-B:149–55.
Levitsky KA, Hozack WJ, Balderston RA, Rothman RH, Gluckman SJ, Maslack MM, Booth RE Jr. Evaluation of the painful prosthetic joint. Relation value of bone scan, sedimentation rate, and joint aspiration. J Arthroplast. 1991;6:237–44.
Stumpe KD, Nötzli HP, Zanetti M, Kamel EM, Hany TF, Görres GW, von Schulthess GK, Hodler J. FDG PET for differentiation of infection and aseptic loosening in total hip replacements: comparison with conventional radiography and three-phase bone scintigraphy. Radiology. 2004;231(2):333–41.
Fuster D, Duch J, Soriano A, García S, Setoain X, Bori G, Rubí S, Rodríguez D, Doménech B, Piera C, Mensa J, Pons F. Potential use of bone marrow scintigraphy in suspected prosthetic hip infection evaluated with 99mTc-HMPAO-leukocytes. Rev Esp Med Nucl. 2008;27(6):430–5.
Chryssikos T, Parvizi J, Ghanem E, Newberg A, Zhuang H, Alavi A. FDG-PET imaging can diagnose periprosthetic infection of the hip. Clin Orthop Relat Res. 2008;466(6):1338–42.
Geller JA, MacCallum KP, Murtaugh TS, Patrick DA Jr, Liabaud B, Jonna VK. Prospective comparison of blood culture bottles and conventional swabs for microbial identification of suspected periprosthetic joint infection. J Arthroplast. 2016;31(8):1779–83.
Parvizi J, Gehrke T, Chen AF. Proceedings of the international consensus on periprosthetic joint infection. Bone Joint J. 2013;95-B(11):1450–2.
Wetters NG, Berend KR, Lombardi AV, Morris MJ, Tucker TL, Della Valle CJ. Leukocyte esterase reagent strips for the rapid diagnosis of periprosthetic joint infection. J Arthroplast. 2012;27(Suppl 1):8–11.
Bingham J, Clarke H, Spangehl M, Schwartz A, Beauchamp C, Goldberg B. The alpha defensin-1 biomarker assay can be used to evaluate the potentially infected total joint arthroplasty. Clin Orthop Relat Res. 2014;472(12):4006–9.
Bonanzinga T, Zahar A, Dütsch M, Lausmann C, Kendoff D, Gehrke T. How reliable is the alpha-defensin immunoassay test for diagnosing periprosthetic joint infection? A prospective study. Clin Orthop Relat Res. 2017;475(2):408–15.
Lachiewicz PF, Rogers GD, Thomason HC. Aspiration of the hip joint before revision total hip arthroplasty. Clinical and laboratory factors influencing attainment of a positive culture. J Bone Joint Surg Am. 1996;78(5):749–54.
Atkins BL, Athanasou N, Deeks JJ, Crook DW, Simpson H, Peto TE, McLardy-Smith P, Berend AR. Prospective evaluation of criteria for microbiological diagnosis of prosthetic joint infection at revision arthroplasty. The OSIRIS collaborative study group. J Clin Microbiol. 1998;36:2932–9.
Patel R. Biofilms and antimicrobial resistance. Clin Orthop Relat Res. 2005;437:41–7.
Deirmengian C, Hallab N, Tarabishy A, Della Valle C, Jacobs JJ, Lonner J, Booth RE Jr. Synovial fluid biomarkers for periprosthetic infection. Clin Orthop Relat Res. 2010;468:2017–23.
McArthur BA, Abdel MP, Taunton MJ, Osmon DR, Hanssen AD. Seronegative infections in hip and knee arthroplasty: periprosthetic infections with normal erythrocyte sedimentation rate and C-reactive protein level. Bone Joint J. 2015;97-B(7):939–44.
ShanmugasundaramS RBF, Briggs TW, Sussmann PS, Bostrom MP. Evaluation and management of periprosthetic joint infection-an international, multicenter study. HSS J. 2014;10(1):36–44.
Fink B, GrossmanA FM, Schäfer P, Frommelt L. Two-stage cementless revision of infected hip endoprostheses. Clin Orthop. 2009;467:1848–58.
Corona P, Gil E, Guerra E, Soldado F, Amat C, Flores X, Pigrau C. Percutaneous interface biopsy in dry-aspiration cases of chronic periprosthetic joint infections: a technique for preoperative isolation of the infecting organism. Int Orthop. 2012;36(6):1281–6.
Bori G, Soriano A, Garcia S, Gallart X, Casanova L, Mallofre C, Almela M, Martinez JA, Riba J, Mensa J. Low sensitivity of histology to predict the presence of microorganisms in suspected aseptic loosening of a joint prosthesis. Mod Pathol. 2006;19:874–7.
Bori G, Soriano A, Garcia S, Mallofré C, Riba J, Mensa J. Usefulness of histological analysis for predicting the presence of microorganisms at the time of reimplantation after hip resection arthroplasty for the treatment of infection. J Bone Joint Surg Am. 2007;89:1232–7.
Mirra JR, Amstutz HC, Matos M, Gold R. The pathology of the joint tissues and its clinical relevance in prostheses failure. Clin Orthop Relat Res. 1976;117:221–40.
Fehring TK, McAlister JRJA. Frozen histologic section as a guide to sepsis in revision joint arthroplasty. Clin Orthop Relat Res. 1994;304:229–37.
Feldman DS, Lonner JH, Desai P, Zuckerman JD. The role of intraoperative frozen section in revision total hip arthroplasty. J Bone Joint Surg Am. 1995;77:1807–13.
Athanasou NA, Pandey R, de Steiger R, Crook D, Smith PM. Diagnosis of infection by frozen section during revision arthroplasty. J Bone Joint Surg Br. 1995;77:28–33.
Lonner JH, Desai P, Di Cesare PE, Steiner G, Zuckerman JD. The reliability of analysis of intraoperative frozensections for identifiying active infection during revision hip or knee arthroplasty. J Bone Joint Surg Am. 1996;78:1553–8.
Della Valle CJ, Bogner E, Desai P, Lonner JH, Adler E, Zuckerman JD, Di Cesare PE. Analysisof frozen sections of intraoperative specimens obtained at the time of reoperation after hip or knee resection arthroplasty for the treatment of infection. J Bone Joint Surg Am. 1999;81:684–9.
Pandey R, Drakoulakis E, Athanasou NA. An assessment of the histological criteria used to diagnose infection in hip revision arthroplasty tissues. J Clin Pathol. 1999;52:118–23.
Costerton JW. Biofilm theory can guide the treatment of device-related orthopaedic infections. Clin Orthop Relat Res. 2005;437:7–11.
Esteban J, Molina-Manso D, Spiliopoulou I, Cordero-Ampuero J, Fernández-Roblas R, Foka A, Gómez-Barrena E. Biofilm development by clinical isolates of Staphylococcus spp. from retrieved orthopaedic prosthesis. Acta Orthop. 2010;81:674–9.
Kamme C, Lindberg L. Aerobic and anaerobic bacteria in deep infections after total hip arthroplasty: differential diagnosis between infectious and non-infectious loosening. Clin Orthop Relat Res. 1981;154:201–7.
Nelson CL, McLarec AC, McLaren SG, Johnson JW, Smeltzer MS. Is aseptic loosening truly aseptic. Cllin Orthop Rel Res. 2005;437:25–30.
Esteban J, Gómez-Barrena E, Cordero J, Zamora N, Kinnari TJ, Fernández-Roblas R. Evaluation of quantitative analysis of cultures from sonicated retrieved orthopaedic implants in diagnosis of orthopaedic infection. J Clin Microbiol. 2008;46:488–92.
Kwiecien G, George J, Klika AK, Zhang Y, Bauer TW, Rueda CA. Intraoperative frozen section histology: matched for musculo skeletal infection society criteria. J Arthroplast. 2017;32(1):223–7.
Buttaro MA, Martorell G, Quinteros M, Comba F, Zanotti G, Piccaluga F. Intraoperative synovial C-reactive protein is as useful as frozen section to detect periprosthetic hip infection. Clin Orthop Relat Res. 2015;473(12):3876–81.
García-Cimbrelo E, García-Rey E. Bone defect determines acetabular revision surgery. Hip Int. 2014;24(Suppl 10):S33–6. https://doi.org/10.5301/hipint.5000162.
Gustilo RB, Tsukayama D. Treatment of infected cemented total hip arthroplasty with tobramycin beads and delayed revision with a cementless prosthesis and bone grafting. Orthop Trans. 1988;12:739.
Duncan CP, Beauchamp C. A temporary antibiotic-loaded joint replacement system for management for complex infection involving the hip. Orthop Clin North Am. 1993;24:751–9.
Masri BA, Duncan CP, Beauchamp CP. Long-term elution of antibiotics from bone cement an in vitro study using the prosthesis of antibiotic-loaded acrylic cement (PROSTALAC) system. J Arthroplast. 1998;13:331–8.
Hsieh PH, Shih CH, Chang YH, Lee MS, Yang WE, Shih HN. Treatment of deep infection of the hip associated with massive bone loss: two-stage revision with an antibiotic-loaded interim cement prosthesis followed by reconstruction with allograft. J Bone Joint Surg (Br). 2005;87B:770–5.
Degen RM, Davey JR, Davey JR, Howard JL, McCalden RW, Naudie DD. Does a prefabricated gentamicin-impregnated, load-bearing spacer control periprosthetic hip infection? Clin Orthop Relat Res. 2012 Oct;470(10):2724–9.
Neumann DR, Hofstaedter T, List C, Dorn U. Two-stage cementless revision of late total hip arthroplasty infection using a premanufactured spacer. J Arthroplast. 2012;27(7):1397–401.
Romanò CL, Romanò D, Albisetti A, Meani E. Preformed antibiotic-loaded cement spacers for two-stage revision of infected total hip arthroplasty. Long-term results Hip Int. 2012;22(S8):46–53. https://doi.org/10.5301/HIP.2012.9566.
Macheras GA, Koutsostathis S, Kateros K, Papadakis S, Anastasopoulos P. A two stage re-implantation protocol for the treatment of deep periprosthetic hip infection. Mid to long-term results. Hip Int. 2012;22(S8):54–61. https://doi.org/10.5301/HIP.2012.9571.
Cordero-Ampuero J, Esteban J, García-Cimbrelo E. Oral antibiotics are effective for highly resistant hip arthroplasty infections. Clin Orthop. 2009;467:2335–42.
Charlton WPH, Hozack WJ, Teloken MA, Rao R, Bissett GA. Complications associated with reimplantation after Girdlestone arthroplasty. Clin Orthop Relat Res. 2003;407:119–26.
Garcia-Rey E, Cruz-Pardos A, Madero R. Clinical outcome following conversion of Girdlestone’s resection arthroplasty to total hip replacement: a retrospective matched case-control study. Bone Joint J. 2014;96-B(11):1478–84. https://doi.org/10.1302/0301-620X.96B11.33889.
Dallari D, Fini M, Carubbi C, Giavaresi G, Rani N, Del Piccolo N, Sartori M, Masso A. Total hip arthroplasty after excision arthroplasty: indications and limits. Hip Int. 2011;21:436–40.
García-Cimbrelo E, García-Rey E, Cruz-Pardos A. The extent of the bone defect affects the outcome of femoral reconstruction in revision surgery. J Bone Joint Surg (Br). 2011;93-B:1457–64. 24
Garcia-Cimbrelo E. Porous-coated cementless acetabular cups in revision surgery: a 6- to 11-year follow-up study. J Arthroplast. 1999;14:397–406.
Castellanos J, Flores X, Llusà M, Chiriboga C, Navarro A. The Girdlestone pseudoartrosis in the treatment of infected hip replacements. Int Orthop. 1998;22:178–81.
Grauer JD, Amstutz HC, O’Carroll PF, Doray FG. Resection arthroplasty of the hip. J Bone Joint Surg Am. 1989;71-A:669–78.
McElwaine JP, Colville J. Excision arthroplasty or infected total hip replacement. J Bone Joint Surg (Br). 1984;66-B:168–71.
Schröder J, Saris D, Besselaar PP, Marti RK. Comparison of the results of the Girdlestone pseudoartrosis with reimplantation of a total hip replacement. Int Orthop. 1998;22:215–8.
Rittmeister ME, Manthei L, Hailer NP. Prosthetic replacement in secondary Girdlestone artthroplasty has an unpredictible outcome. Int Orthop. 2005;29:145–8.
Salvati EA, Chekofski KM, Brause BD, Wilson PD. Reimplantation in infection. A 12-year experience. Clin Orthop Relat Res. 1982;170:62–75.
Jeong M, Kim HJ, Lim SJ, Moon YW, Park YS. Revision total hip arthroplasty using tantalum augment in patients with Paprosky III or IV acetabular bone defect: a minimum 2-year follow-up study. Hip Pelvis. 2016;28:98–103.
Rowan FE, Gorenchtein M, Aslam S, Condon F, Masterson EL. A comparison of acetabular impaction grafting and trabecular metal for revision arthroplasty. Hip Int. 2016;26:350–4.
Kraay MJ, Goldberg VM, Fitzgerald SJ, Salata MJ. Cementless two-staged total hip arthroplasty for deep periprosthetic infection. Clin Orthop Relat Res. 2005;441:243–9.
Haddad FS, Muirhea-Allwood SK, Manktelow ARJ, Bacarese-Hamilton I. Two-stage uncemented revision hip arthroplasty for infection. J Bone Joint Surg (Br). 2000;82:689–94.
Fehring TK, Calton TF, Griffin WL. Cementless fixation in 2-stage reimplantation for periprosthetic sepsis. J Arthroplast. 1999;14(2):175–81.
Gunther KP, Wegner T, Kirschner S, Hartmann A. Modular reconstruction in acetabular revision with antiprotrusio cages and metal augments: the cage-and-augment system. Oper Orthop Traumatol. 2014;26:141–55.
Kim WY, Greidanus NV, Duncan CP, Masri BA, Garbuz DS. Porous tantalumuncemented acetabular shells in revision total hip replacement: two to four year clinical and radiographic results. Hip Int. 2008;18:17–22.
Petheram TG, Howell JR. The Exeter method-acetabular impaction grafting with cemented reimplantation. Oper Orthop Traumatol. 2014;26:114–25.
Witso E, Persen L, Loseth K, Bergh K. Cancellous bone as an antibiotic carrier. Acta Orthop Scand. 2000;71:80–4.
Buttaro MA, Pusso R, Piccaluga F. Vancomycin-supplemented impacted bone allografts in infected hip arthroplasty. Two-stage revision results. J Bone Joint Surg (Br). 2005;87-B:314–9.
Abolghasemian M, Tangsataporn S, Sternheim A, Backstein D, Safir O, Gross AE. Combined trabecular metal acetabular shell and augment for acetabular revision with substantial bone loss: a mid-term review. Bone Joint J. 2013;95:166–72.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Cordero-Ampuero, J., García-Rey, E. (2019). Acetabular Bone Defect in Infected Total Hip Arthroplasty. In: García-Rey, E., García-Cimbrelo, E. (eds) Acetabular Revision Surgery in Major Bone Defects. Springer, Cham. https://doi.org/10.1007/978-3-319-98596-1_12
Download citation
DOI: https://doi.org/10.1007/978-3-319-98596-1_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-98595-4
Online ISBN: 978-3-319-98596-1
eBook Packages: MedicineMedicine (R0)