Abstract
The conceptual framework for antibody drug conjugates (ADC’s) emerged contemporaneously with the discovery of antibodies, with Paul Ehrlich proposing in the early 1900’s the concept of a “magic bullet”, an ideal therapeutic that would specifically target a disease-causing agent without causing harm to the body. This concept still underpins the overarching goal of biopharmaceutical development today: to produce drugs that have a broad therapeutic index by effectively targeting the disease while causing minimal damage to normal tissue. Although the concept of ADC’s is simple, achieving the ideal combination of properties has proven challenging, as reflected by the limited number of ADC’s that have demonstrated success in the clinic to date. Recent years have witnessed a burgeoning field, with the number of clinical stage ADC’s more than doubling in just the last two years to more than 70 candidates currently in clinical development. Despite the successes to date and the prospect of new ADC’s reaching patients in the coming years, many challenges remain and there is substantial room for improvement, most notably in improving the therapeutic index. The key challenge in developing an ADC is balancing its efficacy and safety. This review will focus on ways to capitalize on bispecific antibody technology to improve the therapeutic index of ADC’s, in pursuit of the magic bullet ideal. The nature of bispecific antibodies allows for fine tuning of the interactions between each target to impact the overall properties of the molecule. Here, we discuss some of the cutting edge bispecific antibody strategies that are currently under investigation to address both the efficacy and safety aspects of ADC’s.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
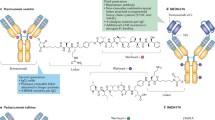
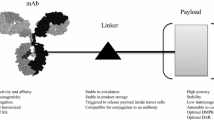
There are four key elements comprising an antibody drug conjugate (ADC) strategy: the target, the antibody, the cytotoxic warhead, and the linker connecting the warhead to the antibody. Consideration of all of these parameters is crucial for the successful development of an ADC [1, 2]. There has been a great deal of progress made in understanding the relationship between these various components, however several challenges remain, most notably improving the therapeutic index [3, 4]. Over the past few years numerous improvements have been made in the chemical properties of the warheads, the linkers, and the means of conjugation to the antibody. These essential advancements, which have greatly expanded the ADC toolbox, have been reviewed elsewhere in this volume. This chapter will focus on the antibody and the target, with a specific emphasis on how to capitalize on bispecific technology to optimize ADC’s. To date, the majority of the bispecific antibody approaches to treat cancer have fallen into one of two broad functional categories: (i) simultaneous blockade of two cancer associated targets (e.g., oncogenic receptors, growth factor ligands, or cytokines) or (ii) redirection of a therapeutic effector (e.g., engaging immune effector cells or molecules, pre-targeting of therapeutic toxin or radionuclide) [5,6,7,8]. While many of these prior approaches do not translate directly to ADC’s, recent efforts have sought to exploit the unique features of bispecific antibodies to produce ADC’s that are more efficacious and better tolerated. Ultimately, the key challenge in developing any ADC is balancing its efficacy and safety [1, 2, 4]. The ability of bispecific antibodies to simultaneously engage two targets affords some creative possibilities to address both the efficacy and safety aspects of ADC’s. Several strategies currently in development employ bispecific targeting to enhance ADC internalization and lysosomal delivery, with the goal of improving efficacy [9,10,11]. Another emerging area of research seeks to use the dual targeting capability of bispecific antibodies to improve selectivity toward the tumor relative to normal tissue, an approach that could impact both the safety and efficacy of ADC’s [12,13,14].
With the approval of catumaxomab (Removab) in 2009 and Blinatumomab in 2015, pharmaceutical companies have started to use bispecific antibodies (BsAbs) more frequently for therapeutic applications. The proof of concept for bispecific antibodies was first demonstrated more than half a century ago, initially by chemical conjugation of two antibodies to form bispecific F(ab’)2 molecules [15] and later by fusing two different hybridoma cells [16] which was enabled by the hybridoma technology established in 1975 [17]. The hybridoma approach to produce bispecific antibodies is time consuming, requires multiple purification steps, suffers from low purification yields, and faces potential immunogenicity issues. Advances in protein engineering technologies have enabled the generation of recombinant bispecific antibodies with defined architecture and the desired biochemical, functional, and pharmacological properties. The ability to select among different bispecific formats to tailor these properties for the specific application provides opportunities to extend the potential of therapeutic antibodies. The enhanced capacity for fine tuning of bispecific antibodies is particularly relevant for ADC approaches to improve both their efficacy and safety.
Molecular Formats of Bispecific Antibodies
Bispecific antibodies (BsAbs) provide the ability to recognize two different antigens or two distinct epitopes (a subset of bispecifc antibodies designated biparatopic antibodies) simultaneously as a single molecule and offer the potential to maximize the benefits of therapeutic antibodies by a number of mechanisms, including, but not limited to: (1) simultaneously blocking two different targets or mediators that have a primary role in the disease pathogenesis; (2) retargeting to mediate effector functions, such as antibody-dependent cell-mediated cytotoxicity (ADCC); (3) avoiding or delaying the development of resistance; (4) inducing more potent anti-proliferative effects, and (5) activating cytotoxic T and NK cells to induce tumor lysis (e.g., bispecific T-cell engagers (BiTE) and bispecific killer cell engagers (BiKE)). There are now more than 100 different bispecific formats [18] enabling researchers to select the ideal parameters (e.g., size, half-life, stability, flexibility, orientation, and developability) to achieve the desired therapeutic outcome. Bispecific antibody formats can be classified into five distinct structural groups [18, 19]: (1) monovalent bispecific IgG (IgG-like architecture with a single binding moiety for each specificity); (2) appended IgG (IgG backbone with a second antigen targeting domain fused at specific locations); (3) BsAb fragments; (4) bispecific fusion proteins, and (5) BsAbs generated by chemical conjugations. In this chapter we will focus on recombinant approaches to generate bispecific antibodies and will highlight their potential in ADC applications.
While conventional IgG antibodies are bivalent and monospecific, bispecific IgG-like antibodies that are monovalent for each antigen are most often used (referred to as monovalent bispecific IgG herein). These monovalent bispecific IgG’s typically contain an asymmetric Fc region for heterodimerization to avoid heavy chain (HC) mispairing [18,19,20,21,22]. Some approaches for IgG-based bispecifics use a wild-type homodimeric Fc regions. Examples of this approach include two-in-one antibody [23], κλ-body with a common heavy chain [24] and iMab with all four different chains tethered by flexible linkers [25]. However, two-in-one antibodies and κλ-body technologies require extensive antibody engineering and screening and cannot be generated with preexisting mAbs without reengineering the binding sites. To address the HC mispairing problem, heterodimeric Fc technologies have been developed to allow the correct assembly of two different HCs [26,27,28,29,30,31,32,33,34,35,36,37,38] into BsAbs (Fig. 1a ). The most common Fc heterodimeric technologies are (1) knobs-into-holes (KIH) [36]; (2) electrostatic steering [33, 34]; (3) Fab-arm exchange (Duobody) [37]; and (4) SEED body [30]. Monovalent bispecific IgG’s with heterodimeric Fc have been generated using a number of strategies to avoid light and heavy chain mispairing including combination with a common light chain (LC) approach [24] or with two distinct LCs, using the CrossMab [39], DuetMab [40] with a reengineered HC-LC disulfide bond, electrostatic steering [41], and κλ-body with a common heavy chain [24].
In contrast to the monovalent bispecific IgG format, bivalent bispecific antibodies can also be generated by engineering additional antigen binding units into different locations on IgG’s [18, 19], including appended IgG fused to the heavy chain [42, 43]. Besides the N- and C-terminal fusion of scFvs to heavy chain, scFvs have been successfully inserted into the heavy chain hinge [43, 44] (Some examples are listed in Fig. 1b) and Fc regions (manuscript in preparation) to generate fully functional BsAbs.
Selection of Bispecific Formats and Binding Modalities for ADC’s
Identification of bispecific antibody formats with the desired functionality is critical to develop bispecific antibody drug conjugates. Selection of the correct bispecific format for drug development is a challenge, with many different bispecific formats to choose from [18]. The choice of molecular format can impart key features, such as binding modality (ex. monovalent or bivalent binding to each target, biparatopic binding, etc.). Typically, the bispecific format is chosen to match the proposed mechanisms of action and the specific clinical application. Ideally, several alternative bispecific formats are constructed and the final lead candidate is chosen after in vitro and in vivo functional characterization. The conventional IgG-like monovalent bispecific format is usually selected for good developability properties, prolonged in vivo half-life, and desired antibody effector functions such as ADCC and CDC. Although the mechanisms of ADC toxicity are complex, target expression in normal tissue can lead to on-target toxicity [45, 46]. Thus, strategies for increasing tumor selectivity, and thus the therapeutic index, of ADC’s are needed to limit toxicity resulting from target engagement in normal tissue. Monovalent bispecific IgG’s are the preferred format for increasing target selectivity by altering antibody affinity to maximize killing of cancer cells while sparing normal cells [12, 47]. One potential major advantage of appended IgG’s is that they preserve the natural antibody avidity to cell surface receptors and can enable the simultaneous binding of antigen to all variable domains and hence provide a higher specific binding capacity [48]. This may be useful in targeting cells with low abundance receptors for enhanced potency. Biparatopic antibodies (a subset of bispecific antibodies in which each antigen binding domain recognizes unique, non-overlapping epitopes on the same target antigen) have demonstrated the superior ability to promote receptor clustering for improved receptor internalization, lysosomal trafficking, and receptor down regulation, therefore increasing drug potency [11, 49]. Capitalizing on this ability of biparatopic antibodies to increase lysosomal trafficking is a promising strategy to enhance delivery of ADC’s to target cells and is discussed in detail below. Ultimately, the selection of the appropriate bispecific antibody format will be dictated by the specific biology of the targets, the clinical need, and the features offered by a given format to address those requirements.
Bispecific ADC Strategies: Maximizing Internalization and Trafficking to Lysosomes
The first consideration in developing an ADC is identifying an appropriate target that will serve to deliver the cytotoxic drug into the tumor. Generally, ADC target selection has focused primarily on its expression pattern, with the ideal target showing high, uniform expression in the tumor and little to no expression in normal tissues. Such a clean expression profile affords the best opportunity to achieve a broad therapeutic index. Nevertheless, the expression profile is only one factor that contributes to the success of an ADC target. Virtually all of the ADC payloads to date require not only binding of the target at the tumor cell surface, but also uptake into the cell and subsequent delivery to the lysosome in order to effectively release the active cytotoxic warhead [50, 51]. Many potential ADC targets either internalize poorly or undergo a high rate of endocytic recycling, which causes the ADC to return to the cell surface intact without delivering the payload to the lysosome [50, 52]. Several bispecific ADC approaches have emerged recently that seek to enhance internalization and trafficking to the lysosome, thus maximizing the amount of drug that is effectively delivered to tumor cells at a given dose.
Early work suggested that targeting a single receptor with bispecific antibodies that recognize distinct epitopes could lead to increased avidity/overall affinity toward the target and greater potency [53]. Subsequent studies demonstrated that non-overlapping antibody pairs and biparatopic antibodies or non-antibody scaffolds could drive receptor clustering and cross-linking, which promotes enhanced internalization, trafficking to the lysosome and degradation of the target [54,55,56]. Importantly, not all non-overlapping antibody pairs are equally effective at promoting receptor down regulation, and there is evidence that the specific epitopes and spatial orientation induced by their combination has an impact on their ability to synergistically drive enhanced lysosomal trafficking [55]. Symphogen applied this principle to achieve targeted degradation of EGFR using a pair of monoclonal antibodies, termed Sym-004, which is currently in PhII clinical testing in multiple solid tumor indications [57, 58]. Similarly, Covagen developed a bispecific, biparatopic HER2 targeted Fynomab, COVA208, which recognizes two distinct epitopes and induces degradation of the receptor, as well as other HER family members, EGFR and HER3 [59]. These investigators noted that simply targeting two distinct epitopes does not ensure functional activity, and they proposed that the molecular architecture and spatial orientation of the different binding arms influences the ability to efficiently induce receptor clusters that are targeted for lysosomal degradation. While these examples demonstrated how biparatopic targeting can promote receptor trafficking to the lysosome and subsequent down modulation of signaling through receptor degradation, the MOA is also well suited to an ADC approach that seeks to maximize delivery of a cytotoxic drug to tumor cells.
As noted above, there are a number of targets that show promising tumor expression profiles, but poor lysosomal trafficking limits their full potential as effective ADC targets. HER2 is an example of a recycling receptor that exhibits a dynamic equilibrium between the cell surface and recycling endosomes [52]. As a result, when anti-HER2 antibodies, such as trastuzumab, bind to HER2 at the cell surface, the majority of the antibody-receptor complex is internalized and rapidly recycled back to the cell surface intact, with only a small fraction trafficking to lysosomes. Consequently, the Her2 targeted ADC, T-DM1 (Kadcyla®), which consists of trastuzumab conjugated to the microtubule toxin maytansinoid DM1, exhibits only limited delivery to lysosomes [52]. While it is currently the only ADC approved for solid tumors, and provides clinical benefit for a subset of HER2 positive breast cancer patients, its clinical utility is restricted to patients whose tumors express a high level of HER2. Disappointingly, T-DM1 failed to show a treatment benefit in gastric cancer (GATSBY trial, NCT01641939 [60]), an indication for which the unarmed trastuzumab is approved. Similarly, it has been slow to move up to earlier lines of therapy in breast cancer, as multiple Phase II and III clinical trials have been terminated or have failed to demonstrate superiority compared to other standard of care therapies, including trastuzumab plus taxane (e.g., MARIANNE trial, NCT01120184 [61]). Several investigators have suggested that the limited clinical benefit of T-DM1 can be attributed in part to its poor lysosomal trafficking and have developed bispecific ADC strategies to overcome this limitation ([11] Zymeworks ZW33). Li et al. recently demonstrated that a biparatopic antibody produced by combining two binding arms from each of two non-competing HER2 antibodies (for a total of four HER2 binding moieties per molecule) was capable of efficiently inducing large cross-linked antibody-receptor clusters, causing internalization and lysosomal degradation that resulted in greater than 90% depletion of HER2 from the cell surface within 1 h (Fig. 2). Over the same time period, trastuzumab induced a negligible degree of HER2 internalization and lysosomal degradation. The authors took advantage of this enhanced lysosomal trafficking to produce an ADC that could more effectively deliver a cytotoxic payload to tumor cells and direct it to lysosomes, where the toxin could be released. The resulting biparatopic ADC, conjugated to the microtubule toxin, tubulysin (AZD13599185) and designated MEDI4276, showed superior in vitro and in vivo activity compared to the trastuzumab based ADC, T-DM1. In patient derived tumor xenograft (PDX) models of human breast cancer representing both T-DM1 ineligible HER2 low tumors and T-DM1 relapsed/refractory HER2 positive tumors, the biparatopic HER2 ADC caused tumor stasis or regression in a large proportion of the models. While it may seem counterintuitive to invoke a mechanism that involves target downregulation, several considerations make this a tractable ADC strategy. First, the enhanced lysosomal trafficking results in more effective tumor cell killing in the first instance. Second, the authors demonstrate that the tubulysin warhead employed in the ADC possesses bystander killing activity, which means that, once liberated from target expressing tumor cells, the cytotoxic warhead can enter and kill nearby non-target expressing tumor cells [11]. Finally, downregulation of HER2 is in itself a potentially viable mechanism of shutting down oncogenic signaling in HER2 driven tumors [62]. MEDI4276 is currently under investigation in a Phase I clinical trial in patients with HER2 expressing solid tumors (NCT02576548). The enhanced lysosomal trafficking and superior preclinical activity of MEDI4276 represents an opportunity to fill an unmet medical need in patients that are T-DM1 resistant or are ineligible for T-DM1 due to low levels of HER2 tumor expression. More recently, Zymeworks has initiated development of ZW33, a biparatopic HER2 targeted ADC built on their IgG-like Azymetric™ platform [35]. The proposed MOA’s of ZW33 include cross-linked trans HER2 binding and clustering, enhanced toxin-mediated cytotoxicity due to increased HER2-mediated ADC internalization, HER2 downregulation, as well as signaling blockade and effector function-mediated cytotoxicity [10]. The U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation for ZW33 for the treatment of ovarian cancer and an IND filing is anticipated in the second half of 2017 for multiple indications, including breast and gastric cancer. These examples of HER2 targeting biparatopic ADC’s demonstrate that enhancing lysosomal trafficking is a viable strategy for improving the efficacy of ADC’s, and could be similarly used for other targets. Enhanced lysosomal trafficking has also been shown with multiple biparatopic molecule formats, including mAb combinations, bispecific antibodies, non-antibody scaffolds, and Ig domain formats. Accordingly, Crescendo Biologics has applied their Humabody® human VH domain platform to the biparatopic ADC approach [9]. They report that the small size of the molecular format combined with biparatopic targeting leads to an ADC with superior internalization, rapid tumor uptake and penetration, and potent in vivo tumor killing.
Proposed mechanisms of action of MEDI4276. Dual targeting of HER2 by biparatopic ADC maximizes toxin delivery into tumor cells by inducing receptor clustering, enhanced internalization, and trafficking to lysosomes. The released cytotoxin acts directly on target cells by inhibition of microtubule assembly, leading to mitotic arrest and apoptosis. In addition, the cytotoxin is membrane permeable, allowing it to passively diffuse out of target cells into the tumor microenvironment, where it can kill neighboring tumor cells. This enhances the ability of the ADC to kill tumor cells that exhibit heterogeneous expression of the target (reviewed in [69]). (From Li et al. [11]. Used with permission from Cell Press)
While induction of receptor clustering and cross linking has emerged as a general strategy for enhancing ADC internalization and trafficking to lysosomes, another strategy that several groups have begun to explore is a passive cargo, or “drag and degrade” mechanism. In this bispecific approach, a poorly internalizing target that provides tumor specificity is paired up with a target that efficiently internalizes and traffics to lysosomes. Lee, J.M., et al. demonstrated this concept in a non-ADC context by constructing bispecific antibodies consisting of a strongly internalizing anti-cMET antibody, SAIT301, paired with anti-HER2 or anti-EGFR antibodies [63]. The authors demonstrated that these bispecific antibodies induce efficient EGFR or HER2 internalization and degradation when cMet was present, a process that they termed “drag and degrade”. They further show that the bispecific antibodies cause HSP90, a chaperone that is known to protect proteins from lysosomal degradation, to dissociate from the target receptors. Although these authors did not use the approach to deliver a cytotoxic drug, their work demonstrates that it is possible to use bispecific antibodies to induce increased lysosomal trafficking of poorly internalizing or highly recycling targets by pairing them with a strongly internalizing target. More recently, de Goeij, et al. demonstrated that one could use a similar approach to deliver an ADC to target tumors [13]. They created a bispecific ADC in which one binding arm specifically targets CD63 (also known as LAMP-3), a protein that shuttles between the plasma membrane and endosomal compartments, including lysosomes, and combined it with a HER2 binding arm, which was selected to provide tumor specific binding. CD63 is a ubiquitously expressed protein in the tetraspanin superfamily. Although the bulk of the cellular pool exists intracellularly in late endosomes and lysosomes, a small fraction is present on the cell surface. While the functions of CD63 are not completely understood, it appears to regulate intracellular transport of interacting proteins via endocytosis, with lysosomal targeting as a major fate of the internalized cargo [64]. The authors selected a low affinity anti-CD63 arm so that efficient binding and internalization preferentially occurred only when the tumor associated HER2 target was present along with the CD63. They demonstrated that bispecific targeting of CD63 and HER2 resulted in enhanced internalization and co-localization with lysosomes on target expressing tumor cells, whereas the monospecific parental antibodies did not internalize appreciably. In contrast, the bispecific antibody showed minimal binding and intracellular accumulation in peripheral blood thrombocytes and granulocytes, which express CD63 but not HER2. Such a strategy has the potential to provide both enhanced lysosomal delivery, thereby improving efficacy, as well as enhanced tumor selectivity, thereby improving safety. In a similar fashion, investigators at Regeneron have recently generated a bispecific ADC targeting HER2 and prolactin receptor (PRLR) and tested its activity in vitro [14]. PRLR is a tumor associated target, but in contrast to HER2, it is constitutively internalized, trafficked to lysosomes, and degraded. The HER2xPRLR bispecific antibody dramatically enhanced the degradation of HER2 in vitro, and, when conjugated to the toxin, DM1, the resulting ADC kills double positive breast cancer cells more effectively than the corresponding monospecific HER2 ADC. Although these molecules have yet to be proven in the clinic, they show that it is possible to redirect a poorly internalizing target for lysosomal degradation by employing a bispecific antibody that targets a second antigen with enhanced lysosomal trafficking. This strategy has the potential to significantly expand the number of viable ADC targets to include those that do not readily traffic to lysosomes.
A key question going forward is whether these enhanced lysosomal targeting strategies will improve the therapeutic index. While these strategies could, in principle, introduce an increased risk for on-target toxicity, the majority of ADC toxicities observed in the clinic are target independent [45, 46]. Considerations such as proliferative index and regenerative potential of the target organ will also play a role in the toxicity profile of an ADC. For example, the mechanism of many ADC warheads are designed to differentially affect rapidly dividing cells (ex. disruption of the microtubule network required for cell division). Thus, if a normal tissue expresses the target antigen but proliferates slowly, it likely to be less sensitive to the ADC compared to a rapidly dividing tumor that expresses the target [45, 46]. Ongoing and pending clinical trials will provide the key proof of concept for biparatopic ADC’s, but preclinical evidence suggests that they represent a promising strategy to enhance lysosomal trafficking and delivery, thus turning poorly internalizing tumor associated antigens into tractable ADC targets.
Bispecific ADC Strategies: Enhancing Selectivity
The examples above demonstrate that bispecific antibodies can improve the efficacy of ADC’s by enhancing targeting to lysosomes. Recent work has sought to further capitalize on bispecific technology to improve ADC targeting and selectivity. In its simplest form, bispecific antibodies can employ dual targeting to extend the reach of an ADC, namely to create a two in one ADC. In this scenario, either target is sufficient to deliver the ADC into the tumor cell, which can be useful to broaden the therapeutic benefit when the targets are heterogeneously expressed within the tumor. Waldron, et al. demonstrated the feasibility of such an approach with a bispecific EpCAM-CD133 toxin conjugate [65]. Other more recent efforts are aimed at fine tuning the properties of each binding arm to suit the particular targets and to improve tumor selectivity. As noted above, the HER2-CD63 used a reduced affinity anti-CD63 arm to favor selective binding and ADC delivery to tumor cells expressing both the tumor associated target, HER2, and the lysosomal associated protein, CD63 [13]. This same principle could be applied to two tumor associated targets in order to achieve improved tumor selectivity. That said, an important lesson can be taken from the experience with some immunocytokines, as Tzeng, et al. exemplified with IL2-IgG bispecific fusions [66]. These investigators found that fusion of IL-2 to an antibody against a tumor associated antigen (TAA) caused a dramatic redistribution away from the tumor and toward IL-2 receptor expressing immune cells. These results show that simply creating a bispecific molecule does not ensure that both targets will contribute equally to the behavior of the final molecule. One arm can dominate, and care must be taken to select complementary targets. Likewise, Mazor, et al. have demonstrated that dual targeting alone is not sufficient to achieve tumor selectivity, and that the affinity of the individual arms, the density of the target, the overall avidity and the valency of the bispecific format all play significant roles in producing a bispecific mAb that can discriminate between tumors that express both targets from normal tissue/non-transformed cells that express only one of the targets [12, 47]. The authors systematically evaluated a series of bispecific HER2/EGFR variants with different EGFR affinities and showed that a reduced affinity monovalent bispecific could discriminate dual target expressing tumors, in vivo, from those expressing a single target, while the higher affinity variants lacked this selectivity. They further show that incorporating the same antibody arms into a bivalent bispecific format abolished the gains in selectivity, suggesting that achieving optimal tumor selectivity requires a delicate balance of multiple factors, including both affinity and valency. Similarly, Sellmann, et al. generated EGFRxcMET bispecific ADC’s with different EGFR affinities and showed that an affinity attenuated variant had greater selectivity in vitro for tumor cells overexpressing both antigens [67]. The authors showed that reducing the EGFR affinity led to decreased cytotoxicity toward human keratinocytes, which express moderate levels of EGFR and low cMET. They propose that selecting the appropriate combination of affinity optimized bispecific ADC variants could lead to higher selectivity for tumor versus normal tissue, which could broaden the therapeutic index.
The strategies presented here represent sophisticated applications of bispecific technology that are designed to derive the maximal potential of dual targeting with a single molecular entity, which goes well beyond simply binding and neutralizing two targets. The principle of avidity, defined as the accumulated strength of multiple individual interactions, is likely key to the success of these strategies [68]. Bispecific antibodies may tolerate low affinity interactions toward each individual target because dual targeting drives the overall strength of binding through avidity effects. This property can be advantageous when one or both of the targets has some expression in normal tissues, but are only substantially co-expressed in tumors. The ability to independently fine tune each arm of a bispecific ADC to suit the expression and safety profiles of each target may enable mitigation of potential toxicities in normal tissue while maintaining potency against tumors.
As we move forward, the types of bispecific and biparatopic technologies described in this chapter will likely start to be used more frequently for poorly internalizing tumor antigens where optimization of cytotoxic warhead delivery requires greater tumor selectivity, increased ADC uptake and enhanced lysosomal trafficking. The strides made in antibody engineering technologies coupled with advances made in the development of ADC’s make this an ideal time to develop bispecific and biparatopic ADC’s with improved activity and a better therapeutic index.
References
Beck A et al (2017) Strategies and challenges for the next generation of antibody-drug conjugates. Nat Rev Drug Discov 16:315
Thomas A, Teicher BA, Hassan R (2016) Antibody–drug conjugates for cancer therapy. Lancet Oncol 17(6):e254-e262
Lambert JM, Morris CQ (2017) Antibody-drug conjugates (ADCs) for personalized treatment of solid tumors: a review. Adv Ther 34:1015
Tolcher AW (2016) Antibody drug conjugates: lessons from 20 years of clinical experience. Ann Oncol 27(12):2168–2172
Kontermann RE (2012) Dual targeting strategies with bispecific antibodies. MAbs 4(2):182–197
May C, Sapra P, Gerber H-P (2012) Advances in bispecific biotherapeutics for the treatment of cancer. Biochem Pharmacol 84(9):1105–1112
Yang F, Wen W, Qin W (2016) Bispecific antibodies as a development platform for new concepts and treatment strategies. Int J Mol Sci 18(1)
Fan GW et al (2015) Bispecific antibodies and their applications. J Hematol Oncol:8
Boku N (2014) HER2-positive gastric cancer. Gastric Cancer 17(1):1–12
ZW33, Anti-HER2 x HER2 ADC Overview, Zymeworks Company Website. July 2017.; Available from: https://www.zymeworks.com/our-pipeline/zw33
Li JY et al (2016) A Biparatopic HER2-targeting antibody-drug conjugate induces tumor regression in primary models refractory to or ineligible for HER2-targeted therapy. Cancer Cell 29(1):117–129
Mazor Y et al (2017) Enhanced tumor-targeting selectivity by modulating bispecific antibody binding affinity and format valence. Sci Rep 7:40098
de Goeij BE et al (2016) Efficient payload delivery by a bispecific antibody-drug conjugate targeting HER2 and CD63. Mol Cancer Ther 15(11):2688–2697
Andreev J et al (2017) Bispecific antibodies and antibody-drug conjugates (ADCs) bridging HER2 and prolactin receptor improve efficacy of HER2 ADCs. Mol Cancer Ther 16:681
Nisonoff A, Rivers MM (1961) Recombination of a mixture of univalent antibody fragments of different specificity. Arch Biochem Biophys 93(2):460
Brennan M, Davison PF, Paulus H (1985) Preparation of bispecific antibodies by chemical recombination of monoclonal immunoglobulin G1 fragments. Science 229(4708):81–83
Kohler G, Milstein C (1975) Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256(5517):495–497
Brinkmann U, Kontermann RE (2017) The making of bispecific antibodies. MAbs 9(2):182–212
Spiess C, Zhai Q, Carter PJ (2015) Alternative molecular formats and therapeutic applications for bispecific antibodies. Therapeutic antibodies: discovery, design and deployment. Mol Immunol 67(2, Part A):95–106
Ha JH, Kim JE, Kim YS (2016) Immunoglobulin Fc heterodimer platform technology: from design to applications in therapeutic antibodies and proteins. Front Immunol 7:394
Liu H et al (2017) Fc engineering for developing therapeutic bispecific antibodies and novel scaffolds. Front Immunol 8:38
Krah S et al (2017) Engineering bispecific antibodies with defined chain pairing. New Biotechnol 39:167
Bostrom J et al (2009) Variants of the antibody herceptin that interact with HER2 and VEGF at the antigen binding site. Science 323(5921):1610–1614
Fischer N et al (2015) Exploiting light chains for the scalable generation and platform purification of native human bispecific IgG. Nat Commun 6:6113
Dimasi N et al (2017) Guiding bispecific monovalent antibody formation through proteolysis of IgG1 single-chain. MAbs 9(3):438–454
Choi HJ et al (2015) Engineering of immunoglobulin fc heterodimers using yeast surface-displayed combinatorial fc library screening. PLoS One 10(12):e0145349
Wranik BJ et al (2012) LUZ-Y, a novel platform for the mammalian cell production of full-length IgG-bispecific antibodies. J Biol Chem 287(52):43331–43339
Leaver-Fay A et al (2016) Computationally designed bispecific antibodies using negative state repertoires. Structure 24(4):641–651
Moretti P et al (2013) BEAT® the bispecific challenge: a novel and efficient platform for the expression of bispecific IgGs. BMC Proc 7(6):O9
Davis JH et al (2010) SEEDbodies: fusion proteins based on strand-exchange engineered domain (SEED) CH3 heterodimers in an Fc analogue platform for asymmetric binders or immunofusions and bispecific antibodies. Protein Eng Des Sel 23(4):195–202
de Kruif C.A., Hendriks L.J.A., Logtenberg T. (2016) Methods and means for the production of Ig-like molecules. Google Patents
Choi HJ et al (2013) A heterodimeric Fc-based bispecific antibody simultaneously targeting VEGFR-2 and met exhibits potent antitumor activity. Mol Cancer Ther 12(12):2748–2759
Strop P et al (2012) Generating bispecific human IgG1 and IgG2 antibodies from any antibody pair. J Mol Biol 420(3):204–219
Gunasekaran K et al (2010) Enhancing antibody fc heterodimer formation through electrostatic steering effects applications to bispecific molecules and monovalent IgG. J Biol Chem 285(25):19637–19646
Von Kreudenstein TS et al (2013) Improving biophysical properties of a bispecific antibody scaffold to aid developability: quality by molecular design. MAbs 5(5):646–654
Merchant AM et al (1998) An efficient route to human bispecific IgG. Nat Biotechnol 16(7):677–681
Labrijn AF et al (2013) Efficient generation of stable bispecific IgG1 by controlled Fab-arm exchange. Proc Natl Acad Sci 110(13):5145–5150
Moore GL et al (2011) A novel bispecific antibody format enables simultaneous bivalent and monovalent co-engagement of distinct target antigens. MAbs 3(6):546–557
Schaefer W et al (2011) Immunoglobulin domain crossover as a generic approach for the production of bispecific IgG antibodies. Proc Natl Acad Sci U S A 108(27):11187–11192
Mazor Y et al (2015) Improving target cell specificity using a novel monovalent bispecific IgG design. MAbs 7(2):377–389
Liu Z et al (2015) A novel antibody engineering strategy for making monovalent bispecific heterodimeric IgG antibodies by electrostatic steering mechanism. J Biol Chem 290(12):7535–7562
Coloma MJ, Morrison SL (1997) Design and production of novel tetravalent bispecific antibodies. Nat Biotechnol 15(2):159–163
DiGiandomenico A et al (2014) A multifunctional bispecific antibody protects against Pseudomonas aeruginosa. Sci Transl Med 6(262):262ra155
Bezabeh B et al (2017) Insertion of scFv into the hinge domain of full-length IgG1 monoclonal antibody results in tetravalent bispecific molecule with robust properties. MAbs 9(2):240–256
Hinrichs MJ, Dixit R (2015) Antibody drug conjugates: nonclinical safety considerations. AAPS J 17(5):1055–1064
Donaghy H (2016) Effects of antibody, drug and linker on the preclinical and clinical toxicities of antibody-drug conjugates. MAbs 8(4):659–671
Mazor Y et al (2015) Insights into the molecular basis of a bispecific antibody's target selectivity. MAbs 7(3):461–469
Jakob CG et al (2013) Structure reveals function of the dual variable domain immunoglobulin (DVD-Ig (TM)) molecule. MAbs 5(3):358–363
Godar M et al (2016) Dual anti-idiotypic purification of a novel, native-format biparatopic anti-MET antibody with improved in vitro and in vivo efficacy. Sci Rep 6
Ritchie M, Tchistiakova L, Scott N (2013) Implications of receptor-mediated endocytosis and intracellular trafficking dynamics in the development of antibody drug conjugates. MAbs 5(1):13–21
Xu S (2015) Internalization, trafficking, intracellular processing and actions of antibody-drug conjugates. Pharm Res 32(11):3577–3583
Austin CD et al (2004) Endocytosis and sorting of ErbB2 and the site of action of cancer therapeutics trastuzumab and geldanamycin. Mol Biol Cell 15(12):5268–5282
Robert B et al (1999) Tumor targeting with newly designed biparatopic antibodies directed against two different epitopes of the carcinoembryonic antigen (CEA). Int J Cancer 81(2):285–291
Friedman LM et al (2005) Synergistic down-regulation of receptor tyrosine kinases by combinations of mAbs: implications for cancer immunotherapy. Proc Natl Acad Sci U S A 102(6):1915–1920
Spangler JB et al (2010) Combination antibody treatment down-regulates epidermal growth factor receptor by inhibiting endosomal recycling. Proc Natl Acad Sci U S A 107(30):13252–13257
Hackel BJ et al (2012) Epidermal growth factor receptor downregulation by small heterodimeric binding proteins. Protein Eng Des Sel 25(2):47–57
Dienstmann R et al (2015) Safety and activity of the first-in-class Sym004 anti-EGFR antibody mixture in patients with refractory colorectal cancer. Cancer Discov 5(6):598–609
Pedersen MW et al (2010) Sym004: a novel synergistic anti-epidermal growth factor receptor antibody mixture with superior anticancer efficacy. Cancer Res 70(2):588–597
Brack S et al (2014) A bispecific HER2-targeting FynomAb with superior antitumor activity and novel mode of action. Mol Cancer Ther 13(8):2030–2039
Thuss-Patience PC et al (2017) Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): an international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol 18:640
Perez EA et al (2017) Trastuzumab emtansine with or without pertuzumab versus trastuzumab plus taxane for human epidermal growth factor receptor 2-positive, advanced breast cancer: primary results from the phase III MARIANNE study. J Clin Oncol 35(2):141–148
Burstein HJ (2005) The distinctive nature of HER2-positive breast cancers. N Engl J Med 353(16):1652–1654
Lee JM et al (2016) Novel strategy for a bispecific antibody: induction of dual target internalization and degradation. Oncogene 35:4437
Pols MS, Klumperman J (2009) Trafficking and function of the tetraspanin CD63. Exp Cell Res 315(9):1584–1592
Waldron NN et al (2014) A bispecific EpCAM/CD133-targeted toxin is effective against carcinoma. Target Oncol 9(3):239–249
Tzeng A et al (2015) Antigen specificity can be irrelevant to immunocytokine efficacy and biodistribution. Proc Natl Acad Sci 112(11):3320–3325
Sellmann C et al (2016) Balancing selectivity and efficacy of bispecific EGFR x c-MET antibodies and antibody-drug conjugates. J Biol Chem 291:25106
Rudnick SI, Adams GP (2009) Affinity and avidity in antibody-based tumor targeting. Cancer Biother Radiopharm 24(2):155–161
Kovtun YV, Goldmacher VS (2007) Cell killing by antibody-drug conjugates. Cancer Lett 255(2):232–240
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Comer, F., Gao, C., Coats, S. (2018). Bispecific and Biparatopic Antibody Drug Conjugates. In: Damelin, M. (eds) Innovations for Next-Generation Antibody-Drug Conjugates. Cancer Drug Discovery and Development. Humana Press, Cham. https://doi.org/10.1007/978-3-319-78154-9_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-78154-9_11
Published:
Publisher Name: Humana Press, Cham
Print ISBN: 978-3-319-78153-2
Online ISBN: 978-3-319-78154-9
eBook Packages: MedicineMedicine (R0)