Abstract
The concepts of damage control surgery and resuscitation have been known for many years, as have data affirming the advantages for adult patients. There has been no doubt that the application of these damage control resuscitation principles have led to improved success of non-operative management of solid organ injury, decreased multi-system organ failure, and improved mortality [1]. As of this writing, there has been no prospective randomized trial proving the benefit of damage control resuscitation in the pediatric patient. There are several challenges with such trials; primarily, the most common injuries are traumatic brain injury, and overall mortality is low. Death from hemorrhage is rare and precludes effective trials without broad multicenter participation over an extended time frame. It should be said, however, that when a therapy or intervention is proven to have benefit in adult patients, it should be considered applicable to the pediatric population unless there is compelling reason to believe that there are differences in the pediatric anatomy or physiology that would change the efficacy of the therapy or intervention. Such is the case with damage control surgery and resuscitation. The concept of limited crystalloid resuscitation; early, balanced blood component replacement; rapid correction of physiological derangements, including coagulopathy and inflammation; and source control surgery for bleeding and contamination should all be applicable in the pediatric patient based on the developing biology of the child.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Damage Control Resuscitation (DCR)
- Solid Organ Injury
- Pediatric Vascular Injuries
- Trauma-induced Coagulopathy (TIC)
- Conventional Coagulation Tests
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
14.1 Background
The concepts of damage control surgery and resuscitation have been known for many years, as have data affirming the advantages for adult patients. There has been no doubt that the application of these damage control resuscitation principles have led to improved success of non-operative management of solid organ injury, decreased multi-system organ failure, and improved mortality [1]. As of this writing, there has been no prospective randomized trial proving the benefit of damage control resuscitation in the pediatric patient. There are several challenges with such trials; primarily, the most common injuries are traumatic brain injury, and overall mortality is low. Death from hemorrhage is rare and precludes effective trials without broad multicenter participation over an extended time frame. It should be said, however, that when a therapy or intervention is proven to have benefit in adult patients, it should be considered applicable to the pediatric population unless there is compelling reason to believe that there are differences in the pediatric anatomy or physiology that would change the efficacy of the therapy or intervention. Such is the case with damage control surgery and resuscitation. The concept of limited crystalloid resuscitation; early, balanced blood component replacement; rapid correction of physiological derangements, including coagulopathy and inflammation; and source control surgery for bleeding and contamination should all be applicable in the pediatric patient based on the developing biology of the child.
14.2 Differences of the Pediatric Patient
There obviously are differences in the mode of injury, the anatomical injuries sustained, and the physiological and psychological response to injury on an age-based spectrum. The infant has important differences in anatomy and physiology, while the adolescent suffers injuries and responds to them very similarly to an adult.
14.3 Mode of Injury
The young child’s most common injury is fall until early adolescence where motor vehicle-related injuries dominate. Penetrating injuries are uncommon, typically unintentional and less likely to be firearm related, making the likelihood of vascular injury substantially less. Intentional injury is a leading cause of injury-related death in the young child when child abuse dominates the mode of injury (Fig. 14.1) [2]. Injury patterns, therefore, are primarily head injury in the young child, transitioning to more adult patterns by early adolescence.
14.4 Anatomic Differences
The young child has a relatively large head-to-body ratio and a cartilaginous skeleton, making CNS injuries and multi-system injuries common. Death is most likely from the traumatic brain injury and less likely than adults to be from hemorrhage. Specifically, the cartilaginous skeleton and lax ligaments of the pelvis make it unlikely that a young child will suffer significant pelvic injury with retroperitoneal bleeding. The growth plates are closing by the early teen years in girls and the mid-teen years in boys. This anatomic change, combined with driving motor vehicles, makes the adult pattern pelvic injuries much more common in the late teen years. These are important distinctions to make because hypotension in the younger child is less likely to be from a pelvic and retroperitoneal source. When children do suffer the rare posterior pelvic disruption from a crush mechanism, the finding is not subtle and typically easily demonstrable on physical exam in the trauma bay.
Another important difference is the mobility of the mediastinum in the young child. Small volumes of hemothorax and pneumothorax can cause significant shift, decreasing venous return (Fig. 14.2).
Therefore, early decompression of the hemithorax is prudent in the hypotensive child. Additionally, because the skeleton and specifically the ribs are cartilaginous, force may be transmitted more generally than in the adult patient, making multi-system injuries more common. The presence of a broken rib in the young child with cartilaginous ribs connotes a significant force that has been applied and is independently predictive of solid organ injury and mortality [3, 4].
14.5 Physiological Differences
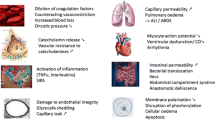
Christiaans’ review article in Shock outlines the age-dependent differences in coagulation factors. Mean values of coagulation proteins (II, V, VII, IX, X, XI, XII) are all significantly lower in the pediatric patient than the adult. There are decreased levels in the pediatric patient of tissue plasminogen, intracellular calcium, tissue plasminogen activator (t-PA), and alpha antiplasmin activity and increases in fibrinolytic activity [5,6,7,8]. These give plausible expectation that the response to injury and the induction of trauma-induced coagulopathy (TIC) may differ in the young child. The next major difference comes from the fact that the primary tissue injury is CNS. The biological differences in the mechanism of trauma-induced coagulopathy between CNS injury and hemorrhagic injury are not well delineated, but these differences may well account for much of the differences seen between pediatric and adult patients in how they manifest TIC. The third difference is in the manifestation of shock. In response to injury, the pediatric patient increases sympathetic output and increases systemic vascular resistance by increasing tone in the medium-sized arteries. This maintains mean arterial pressure and perfusion pressure to vital organs [9]. This response lessens with increasing age and is not as prevalent in the adult patient [10]. The practical point is that the child who is hypotensive from blood loss is far down the volume curve, potentially as much as 40% of total circulating blood volume has been lost.
14.6 Detection of Trauma-Induced Coagulopathy in Children
Conventional coagulation testing such as INR, aPTT, PT, and fibrinogen levels is commonly used for the evaluation of the injured pediatric patient. There are several limitations to the use of these conventional coagulation tests in the pediatric patient, including the fact that these measurements do not measure the overall coagulation system, specifically missing those components derived from injury to the endothelium and cellular components. The conventional coagulation tests also do not describe the hypercoagulable state associated with trauma. Excellent data from both Pittsburg and Houston show that trauma-induced hypercoagulable states exist more commonly in the pediatric patient than the adults [11, 12]. Tests measuring viscoelastic properties of whole blood and platelet function are available and have been used successfully in pediatric trauma patients to elucidate the hemostatic state following injury [13]. Multiple studies, both from combat and civilian practices, demonstrate a high incidence of TIC in the pediatric patient and a significant correlation to injury-related mortality [10]. Age-specific norms have been established for thromboelastography, and it has proven an extremely useful tool in characterizing the coagulation state in children. First used in the hematologic patients, its use now is common in spine, cardiac, and pediatric trauma surgery.
14.7 Treatment Options for Pediatric Patient with Trauma-Induced Coagulopathy
Little has been written to guide our approach to damage control resuscitation in the pediatric patient. A practical approach would dictate that the principles applied to the adult are a reasonable place to start a pediatric resuscitation. The challenge is that almost 80% of pediatric patients receive their initial care at a non-pediatric center and frequently receive excessive crystalloid and packed red cells prior to arrival in definitive care. Few places employ a strategy of damage control resuscitation for pediatrics prior to transfer to a pediatric trauma center.
Once in the pediatric center, a goal-directed approach for reversal of TIC is a more practical approach than an empiric method of resuscitation and blood administration [14]. Evaluation of the TIC with biological testing and replacement of specific factors to replace the deficits is a logical way to approach these transferred patients. Results following replacement of many of the specific factors are only anecdotal in the pediatric population. A replacement ratio of 0.9 (g) of fibrinogen concentrate to 1(u) pRBCs has been proposed by Schochl [10] for fibrinogen levels less than 1.5 g/dL and bleeding. Recombinant factor VIIa (90 μg/kg as a bolus and 25 μg/kg/h as a continuous infusion until bleeding has stopped) has been used successfully in children to stop bleeding but has been associated with significant thrombosis [10]. Prothrombin complexes (PCC) derived from pooled plasma have a high concentration of the proteins but significant blood exposure. Only case reports exist for the use of PCCs in pediatric care [15].
The use of the lysine analog tranexamic acid (TXA) to inhibit plasminogen activation and decrease the activity of plasmin, deceasing fibrinolysis, has been shown in the 2010 CRASH-2 trial to decrease all-cause mortality for injured patients. Anecdotal reports and Cochrane meta-analysis reviews demonstrate a benefit of the use of TXA for pediatric patients during elective cases such as craniofacial, cardiac, and scoliosis surgery. The 2014 paper from Eckert and colleagues in The Journal of Trauma was the first to show a decrease in injury-related mortality when TXA was used in the pediatric population in a conflict setting. Pre-teen patients with bleeding and acidosis had a reduced mortality (OR, 0.27) with the use of TXA. Further, there were no thrombotic or seizure episodes noted in the cohort in this retrospective analysis. The recommendation was, therefore, that the standard dosing protocol be utilized for pediatric patients [16].
In the United Kingdom, the Royal College of Paediatrics and Child Health and the Neonatal and Paediatric Pharmacists Group issued evidence-based statements recommending dosing TXA for injured children at 15 mg/kg loading (max 1 g) over 10 min and 2 mg/kg/h until bleeding stops [17]. Trauma care providers have three choices when considering TXA administration: (1) provide it for all patients with potential significant bleeding, (2) provide it for patients known to have significant bleeding and confirmed acidosis, and (3) provide it for patients known to have hyperfibrinolysis based on biological testing. While definitive data is lacking, at our institution, we begin TXA if there is significant bleeding prior to thromboelastography confirmation of hyperfibrinolysis [18].
14.8 Massive Transfusion in Children
The use of a massive transfusion protocol (MTP) is rare in the young child because of the infrequent incidence of hemorrhagic shock. The use of MTP in pediatric trauma centers is variable and historically has been based on practitioner preference rather than evidence. Neff retrospectively looked at the threshold of blood products given in a combat setting and determined that one-half of a pediatric patient’s circulating blood volume or 40 cm3/kg of all products given over 24 h defined a patient at risk for mortality from bleeding with TIC [19]. Others have used proven coagulopathy by conventional coagulation testing methods along with bleeding and acidosis to define the need for a MTP [20]. There have been no studies that have definitively defined those patients who need an MTP prospectively, and little evidence guides our decision to initiate the MTP in the trauma bay [21].
The physiological difference in children where they maintain mean arterial pressure would suggest that the child who is already hypotensive is significantly volume depleted and may benefit from earlier institution of an MTP. The American College of Surgeons Committee on Trauma’s Resources for the Optimal Care of the Injured Patient (2014) requires a weight-based MTP in pediatric trauma centers [22]. Most centers divide the MTP protocol into distinct weight groups. Table 14.1 is an example of a weight-based transfusion threshold for initiation of MTP. Table 14.2 is an example of the weight-based transfusion protocol.
14.9 Summary of Damage Control Resuscitation
While little evidence exists that a damage control resuscitation (DCR) strategy decreases mortality in the pediatric trauma population, there are enough circumstantial evidence and evidence of success of the various components to recommend its use. An “expert opinion” recommendation would be:
-
(1)
Minimize crystalloid use to maintain minimally acceptable, age-specific, systolic pressures in the prehospital and ED setting.
-
(2)
Initiate TXA for confirmed hypotension from hemorrhage.
-
(3)
Initiate a weight-based MTP for confirmed hypotension from hemorrhage.
-
(4)
Initiate viscoelastic biological testing as soon as feasible in the resuscitation.
-
(5)
Transition to a goal-directed replacement strategy based on the results from testing.
14.10 Damage Control in the Neonatal Period
The neonatal patient suffers from few surgical catastrophes, but among them are those resulting from congenital anomalies such as malrotation with midgut volvulus. In these cases, the rapid laparotomy, reduction of the volvulus, and rapid resection, if required, of frankly dead intestine, followed by resuscitation and reoperation, are the preferred methods (Fig. 14.3). If a second look operation is planned, then temporary closure with a silo of material from Gore-Tex to Silastic is an option (Figs. 14.4 and 14.5).
Another catastrophe that benefits from a damage control procedure is fulminant necrotizing enterocolitis in the premature infant under 1 kg. Such infants often benefit from a temporizing abdominal drainage with or without lavage, further resuscitation, and later definitive operation if necessary (Fig. 14.6). Frequently, however, the temporizing drainage procedure is the only intervention required as the perforation will, at times, seal spontaneously or the baby will progress to a more fulminant total intestinal involvement for which operation is not curative.
The use of umbilical catheters in the neonatal intensive care unit has become ubiquitous. The use of these catheters is not without risk, including vascular and liver injuries. Rapid fluid bolus, particularly through an umbilical venous catheter, can have serious consequences, including opening of previously closed ductus arteriosus and rapid liver expansion with capsular rupture, the latter being a catastrophic consequence. Expanding liver capsular bleeding can occur intraoperatively with minimal contact with the neonatal liver and similarly can be catastrophic [23]. Packing the neonatal liver injury can be lifesaving and is often accomplished with hemostatic gauze alone [24]. The same principles apply, as they do in the adult patient—rapid control of the hemorrhage, correction of the acidosis and coagulopathy, and then reassessment of the hemorrhage.
One benefit of resuscitation of the neonatal patient is the availability of whole blood, which is often used in neonatal cardiac surgery and in many neonatal ICUs. Units that do not have whole blood still likely benefit from packed cells that have had a much shorter shelf life than those sent to non-neonatal units.
14.11 Damage Control for Neurological Injury
The incidence of coagulopathy in children suffering isolated traumatic brain injury is high and, in a paper from USC-LA County, was over 40% in the pediatric population. The release of tissue factor associated with parenchymal brain injury is frequently the inciting factor for the coagulopathy. Researchers have shown that this coagulopathy occurs later than the coagulopathy induced from bleeding but is still significantly associated with mortality [25, 26]. The term “damage control neurosurgery” was coined by JV Rosenfeld (Injury 2004) and referred to abbreviated operation to control bleeding, debride devitalized tissue, and replace dura with a temporary closure (Figs. 14.7 and 14.8) [27]. The Brain Trauma Foundation guidelines endorse early decompressive craniectomy for pediatric patients with intracranial hypertension [28, 29]. While there is no compelling data proving a benefit from this approach, it remains the expert opinion of the foundation and the modern standard of care.
14.12 Damage Control Orthopedic Surgery
The concept of damage control orthopedics was described initially by Scalea et al. in 2000 [30]. The concept involved temporarily stabilizing a fracture with external fixation to provide time for resuscitation and correction of coagulopathy and acidosis before proceeding to a definitive repair (Fig. 14.9). Since the early reports describing its use in adults, there has been very little written regarding children. External fixation was originally felt to be the temporizing method of choice but carried a higher complication rate than internal fixation. Complications, in both adults and children include non-union, joint stiffness, and pin site infections. More recent case series and reports are of submuscular bridge plating and antibiotic spacers that have been described with fewer complications.
The triage of patients to a damage control procedure is often predicated on the resuscitation status and other injuries present [31]. Adult literature demonstrates that the use of serum lactate levels or other biological markers of adequacy of resuscitation is valuable in determining the need for a damage control procedure versus an early definitive operation. Adult patients with high injury severity scores and preoperative lactate levels >3.7 had need for longer ventilation postoperatively than did those with lower lactate levels [32]. Other publications have demonstrated that preoperative lactate levels above 2.5 mmol/L had increased complication rates [33]. There are no such studies in the pediatric population, but there is no reason to believe that if the child is under-resuscitated, as determined by any valid method, delaying definitive repair would not be prudent. In such cases, a temporizing damage control procedure may be warranted.
The optimal timing of long bone fractures in the pediatric patient is also unknown. Again, adult data demonstrates a clear benefit in fixation of femur fractures within 24 h of injury. The primary benefit seems to be a decrease in pulmonary complications, particularly ARDS. The physiological differences in children make the overall incidence of ARDS much less; therefore, if studied, the outcomes may not be similar in children. The one injury pattern that is most bothersome in children is the concomitant long bone and traumatic brain injury because fixation should be accomplished as soon as possible before the cerebral edema precludes.
14.13 Vascular Injuries
Vascular injuries in civilian trauma are not common and even more infrequent in the pediatric population. It is estimated that less than 1% of all injuries are vascular in nature. As more trauma care is delivered in freestanding children’s hospitals, the experience of surgeons in dealing with significant and complex vascular injuries is diminishing. Most pediatric vascular injuries are due to an accidental penetrating mechanism (Fig. 14.10), occur in males, and are in the extremities. Many are not isolated and are seen concurrently with other significant injuries. There are physiological differences in the child that must be considered when planning a reconstruction. Pediatric vessels are prone to vasospasm. This fact can be protective preoperatively but can be clinically confusing intraoperatively. The use of a vasodilator around the repair is often necessary. There is also the consideration of growth in the pediatric limb and the need for growth in the vessel. Because of this concern, interrupted sutures are often used instead of a “running” suture. The benefit of this, however, has never been proven.
The last consideration is that of the use of synthetic graft material in the reconstruction [34]. The recommendation is that vascular reconstructions utilize native vessels as a conduit rather than synthetic graft material. The recommendation is based on the belief that autologous material will have a longer patency rate than a synthetic material. This however, must be balanced against the additional time that may be needed to obtain autologous conduit in a damage control situation.
Given the lack of pediatric-specific vascular expertise, the need for autologous conduit, and the likelihood of concomitant injuries, damage control for pediatric vascular injuries with temporary vascular shunts is an attractive tool to have in the armamentarium, since definitive repair of the pediatric vascular injury may not be feasible [35]. The use of such shunts is well-documented in the military and urban civilian trauma literature but is limited to anecdotal reports in the pediatric patient population.
Argyle™ carotid shunts are ideal conduits for temporary vascular shunts in children. They range in size from 8 to 14 French. The straight shunts are 6 in. long and the looped shunts are 11 in. (Fig. 14.11). The ends are rounded and less likely to cause intimal injury for dissection with passage, and they are sufficiently stiff to tie in place with a suture to prevent dislodgement. The shunts can be used for both arterial and high flow venous injuries as a temporizing means to definitive repair.
Systemic heparinization is often used when temporizing shunts are deployed, but there are case reports of long-term use in the arterial position without heparinization with no untoward consequence.
14.14 Other Adjuncts
Blunt tracheobronchial injuries are rare in the pediatric population, but in the adolescent population, when motorized vehicles are involved, the force and complexity increase significantly. Injuries seen are typically managed with gentle ventilation and chest drainage, but the occasional high force injury sheers the bronchus at or near the carina creating a bronchopleural fistula, where hypoxia precludes successful thoracotomy and repair. The mortality reported in the adult literature is between 8 and 16%. There are no case series large enough to define a reliable mortality rate in children. In these rare incidences, extracorporeal membrane oxygenation (ECMO) has been used successfully with differential lung ventilation to manage these children through resuscitation and even into the OR for repair [36]. ECMO is a valuable tool for the stabilization of patients with respiratory or cardiac compromise; its use is limited to situations where systemic anticoagulation is feasible [37].
14.15 Summary
The changes in anatomy and physiology that occur as a child matures to adulthood influence the pattern and response to injury. While the specific response to damage control resuscitation may vary in an age-dependent way, the principles appear to be applicable to the pediatric patient. The use of damage control surgery has broad applicability in both trauma and non-trauma settings. Rapidly controlling the source of bleeding or infection, relief of intracranial pressure, and stabilization of the bony skeleton followed by continued, targeted resuscitation are undoubtedly equally beneficial in the child as it is in the adult. The opportunities presently facing us in the care of injured children are to adapt technology to aid in the damage control procedures and to elucidate further the changing physiologic response to injury and resuscitation. Only through this understanding will we take full advantage of these concepts.
References
Shrestha B, Holcomb JB, Camp EA, Del Junco DJ, Cotton BA, Albarado R, Gill BS, Kozar RA, Kao LS, McNutt MK, Moore LJ, Love JD, Tyson GH 3rd, Adams PR, Khan S, Wade CE. Damage-control resuscitation increases successful nonoperative management rates and survival after severe blunt liver injury. J Trauma Acute Care Surg. 2015;78(2):336–41.
Centers for Disease Control. Web-based Injury Statistics Query and Reporting System (WISQARS). Atlanta, GA: US Department of Health and Human Services, 10 Leading Causes of Nonfatal Injury, United States; 2014. https://www.cdc.gov/injury/wisqars. Accessed 1 Dec 2016.
Al-Hassani A, Abdulrahaman H, Afifi I, Almadani A, Al-Den A, Al-Kuwari A, Recicar J, Nabir S, Maull KI. Rib fracture patterns predict thoracic chest wall and abdominal solid organ injury. Am Surg. 2010;76(8):888–91.
Rosenberg G, Bryant AK, Davis KA, Schuster KM. No breakpoint for mortality in pediatric rib fractures. J Trauma Acute Care Surg. 2016;80(3):427–32.
Andrew M, Paes B, Johnston M. Development of the hemostatic system in the neonate and young infant. Am J Pediatr Hematol Oncol. 1990;12(1):95–104.
Andrew M, Milner R, Johnston M, Mitchell L, Tollefsen DM, Castle V, Powers P. Development of the human coagulation system in the healthy premature infant. Blood. 1988;72(5):1651–7.
Andrew M, Paes B, Milner R, Johnston M, Mitchell L, Tollefsen DM, Powers P. Development of the human coagulation system in the full-term infant. Blood. 1987;70(1):165–72.
Andrew M, Vegh P, Johnston M, Bowker J, Ofosu F, Mitchell L. Maturation of the hemostatic system during childhood. Blood. 1992;80(8):1998–2005.
Advanced Trauma Life Support (ATLS). American College of Surgeons’ Committee on Trauma, and International ATLS Working Group. 9th ed. Chicago, IL: The ATLS Subcommittee. 2012.
Christiaans SC, Duhachek-Stapelman AL, Russell RT, Lisco SJ, Kerby JD, Pittet JF. Coagulopathy after severe pediatric trauma. Shock. 2014;41(6):476–90.
Liras IN, Cotton BA, Cardenas JC, Harting MT. Prevalence and impact of admission hyperfibrinolysis in severely injured pediatric patients. Surgery. 2015;158(3):812–8.
Leeper CM, Neal MD, McKenna C, Sperry J, Gaines BA. Abnormalities in fibrinolysis at the time of admission are associated with DVT, mortality and disability in a pediatric trauma population. J Trauma Acute Care Surg. 2017;82(1):27–34.
Vogel AM, Radwanss ZA, Cox CS Jr, Cotton BA. Admission rapid thromboelastography delivers real-time “actionable” data in pediatric trauma. J Pediatr Surg. 2013;48(6):1371–6.
Hendrickson JE, Shaz BH, Pereira G, Atkins E, Johnson KK, Bao G, Easley KA, Josephson CD. Coagulopathy is prevalent and associated with adverse outcomes in transfused pediatric trauma patients. J Pediatr. 2012;160(2):204–9.
Navaratnam M, Ng A, Williams GD, Maeda K, Mendoza JM, Concepcion W, Hollander SA, Ramamoorthy C. Perioperative management of pediatric en-bloc combined heart-liver transplants: a case series review. Paediatr Anaesth. 2016;26(10):976–86.
Eckert MJ, Wertin TM, Tyner SD, Nelson DW, Izenberg S, Martin MJ. Tranexamic acid administration to pediatric trauma patients in a combat setting: the pediatric trauma and tranexamic acid study (PED-TRAX). J Trauma Acute Care Surg. 2014;77(6):852–8.
Nishijima DK, Monuteaux MC, Faraoni D, Goobie SM, Lee L, Galante J, Holmes JF, Kuppermann N. Tranexamic acid use in United States Children’s Hospitals. J Emerg Med. 2016;50(6):868–74.
Arkansas Children’s Hospital Clinical Laboratory: Massive Transfusions. ACH Policy and Procedures, November 2016.
Neff LP, Cannon JW, Morrison JJ, Edwards MJ, Spinella PC, Borgman MA. Clearly defining pediatric massive transfusion: cutting through the fog and friction with combat data. J Trauma Acute Care Surg. 2015;78(1):22–8.
Chidester SJ, Williams N, Wang W, Groner JI. A pediatric massive transfusion protocol. J Trauma Acute Care Surg. 2012;73(5):1273–7.
Nosanov L, Inaba K, Okoye O, Resnick S, Upperman J, Shulman I, Rhee P, Demetriades D. The impact of blood product ratios in massively transfused pediatric trauma patients. Am J Surg. 2013;206(5):655–60.
Resources for Optimal Care of the Injured Patient. Chicago, IL: American College of Surgeons, Committee on Trauma. 2014. Print.
Pumberger W, Kohlhauser C, Mayr M, Pomberger G. Severe liver haemorrhage during laparotomy in very low birthweight infants. Acta Paediatr. 2002;91(11):1260–2.
Villalona GA, Mckee MA, Diefenbach KA. Damage control surgery in a <1 kg neonate: a brief report. Yale J Biol Med. 2013;86(3):385–7.
Talving P, Lustenberger T, Lam L, Inaba K, Mohseni S, Plurad D, Green DJ, Demetriades D. Coagulopathy after isolated severe traumatic brain injury in children. J Trauma. 2011;71(5):1205–10.
Harhangi BS, Kompanje EJ, Leebeek FW, Maas AI. Coagulation disorders after traumatic brain injury. Acta Neurochir. 2008;150(2):165–75. discussion 175
Rosenfeld JV. Damage control neurosurgery. Injury. 2004;35(7):655–0.
The 4th Edition of Guidelines for Management of Severe Traumatic Brain Injury. Campbell, CA: Brain Trauma Foundation; 2016.
Kochanek PM, Carney N, Adelson PD, Ashwal S, Bell MJ, Bratton S, Carson S, Chesnut RM, Ghajar J, Goldstein B, Grant GA, Kissoon N, Peterson K, Selden NR, Tasker RC, Tong KA, Vavilala MS, Wainwright MS, Warden CR. Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents—second edition. Pediatr Crit Care Med. 2012;13(Suppl 1):S1–82, Erratum in: Pediatr Crit Care Med. 2012 Mar;13(2):252.
Mooney JF. The use of ‘damage control orthopedics’ techniques in children with segmental open fractures. J Pediatr Orthop B. 2012;21(5):400–3.
Flynn JM, Schwend RM. Management of pediatric femoral shaft fractures. J Am Acad Orthop Surg. 2004;12(5):347–59.
Richards JE, Matuszewski PE, Grififin SM, Koehler DM, Guillamondegui OD, O'Toole RV, Bosse MJ, Obremskey WT, Evans JM. The role of elevated lactate as a risk factor for pulmonary mortality after early fixation of femoral shaft fractures. J Orthop Trauma. 2016;30(6):312–8.
Crowl AC, Young JS, Kahler DM, Claridge JA, Chrzanowski DS, Pomphrey M. Occult hypoperfusion is associated with increased morbidity in patients undergoing early femur fracture fixation. J Trauma. 2000;48(2):260–7.
Kirkilas M, Notrica DM, Langlais CS, Muenzer JT, Zoldos J, Graziano K. Outcomes of arterial vascular extremity trauma in pediatric patients. J Pediatr Surg. 2016;51(11):1885–90.
Inaba K, Aksoy H, Seamon MJ, Marks JA, Duchesne J, Schroll R, Fox CJ, Pieracci FM, Moore EE, Joseph B, Haider AA, Harvin JA, Lawless RA, Cannon J, Holland SR, Demetriades D, Multicenter Shunt Study Group. Multicenter evaluation of temporary intravascular shunt use in vascular trauma. J Trauma Acute Care Surg. 2016;80(3):359–64; discussion 364–5.
Garlick J, Maxson T, Imamura M, Green J, Prodhan P. Differential lung ventilation and venovenous extracorporeal membrane oxygenation for traumatic bronchopleural fistula. Ann Thorac Surg. 2013;96(5):1859–60.
Cornfield DN. Acute respiratory distress syndrome in children: physiology and management. Curr Opin Pediatr. 2013;25(3):338–43.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Todd Maxson, R. (2018). Damage Control in Pediatric Patients. In: Duchesne, J., Inaba, K., Khan, M. (eds) Damage Control in Trauma Care. Springer, Cham. https://doi.org/10.1007/978-3-319-72607-6_14
Download citation
DOI: https://doi.org/10.1007/978-3-319-72607-6_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-72606-9
Online ISBN: 978-3-319-72607-6
eBook Packages: MedicineMedicine (R0)