Abstract
Recreating natural pink esthetics around single or multiple recession defects require proper surgical planning and a careful choice of the grafting approach. The transplantation of free autogenous soft tissue grafts in combination with state-of-the-art surgical techniques for recession coverage still represents the gold standard in terms of long-term tissue stability. Donor site morbidity has to be considered prior to surgery; graft harvesting procedures should be well planned and executed to minimize postoperative patient complaints.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
8.1 Introduction
One of the main goals in the field of periodontal plastic surgery is to recreate natural pink esthetics. The treatment of gingival recessions with autogenous soft tissue grafts is a clear example of the thorough search for satisfactory and predictable methods aiming at optimized root coverage and tissue blending. Laterally positioned flap procedures maintaining tissue integrity with the donor site after harvesting were already used in the 1950s and 1960s for treating single gingival recessions [1, 2] and are still used today [3]. These surgical interventions resulted in natural tissue color and texture at recipient sites, however with remarkable postoperative pain and a high risk for developing secondary recessions as well as bone resorption at the denuded donor area. Free autogenous soft tissue grafts have been introduced for gingival recession coverage during the early 1960s [4, 5], as an alternative for laterally positioned flap procedures. The aim was to increase the width and thickness of the keratinized gingiva, which is necessary to preserve gingival health [6, 7]. According to recent literature data, transplantation of free soft tissue grafts for recession coverage results in increased long-term tissue stability compared to root coverage techniques alone [8,9,10]. Free soft tissue grafts are completely detached from their donor area and can be used in combination with apically or coronally repositioned pedicle flaps, as well as envelope and tunneling techniques. By harvesting a free soft tissue graft from a remote, esthetically irrelevant area of the oral mucosa, donor site complications at adjacent teeth following the elevation of laterally positioned flaps can be avoided. As a result, there is a low risk for root hypersensitivity and impaired esthetics due to secondary intention wound healing at adjacent sites; nevertheless, tissue blending depends on the composition of transplanted grafts.
8.2 Autogenous Soft Tissue Grafts
8.2.1 Free Gingival Graft
Gingival graft transplantation has been used since the 1960s for gingival recession coverage. The first case was published by Björn in 1963 [4], reporting on the harvesting and transplantation of a gingival graft containing connective tissue and overlaying epithelium. Nabers, who utilized full-thickness gingival transplants removed during gingivectomy, was the first to use the term free gingival graft (FGG) [5]. The palate became only later the main donor site of harvesting autogenous grafts [11] (Fig. 8.1). Despite some of the literature data suggesting that the width of keratinized tissues around teeth has low correlation with long-term tissue health and stability [12, 13], the use of free gingival grafts (FGG) became a frequently applied clinical approach to treat gingival recessions [14,15,16,17].
Application of FGGs shows high predictability in terms of graft survival and postsurgical tissue stability. It is of high clinical importance that palatal soft tissue grafts with epithelial coverage will maintain their original characteristics after transplantation to the recipient site. This delivers favorable results in terms of induced keratinization, nevertheless may result in graft hyperplasia and color mismatch due to the fact that FGG carries the genetic determination of the donor site, resulting in a gingival phenotype different from the recipient sites [18, 19]. For this reason, in esthetically demanding cases, the application of alternative autogenous, allogeneic, or xenogeneic grafts might represent a valid treatment alternative.
8.2.2 Subepithelial Connective Tissue Graft
As reported in literature, FGGs have two major limitations: overaugmented tissue contours and impaired color blending with the recipient site [20]. During the 1980s, the clinical benefits of the subepithelial connective tissue graft (SCTG) became evident as reported in literature [21, 22]. Considering these well documented advantages of the SCTG in comparison to FGGs due to lack of epithelium (Fig. 8.1), it has become the first grafting approach of choice during the last three decades. SCTG transplantation is one of the most versatile and esthetically predictable grafting procedures in periodontal plastic surgery. The application of an SCTG in combination of split-thickness pedicle-, envelope-, or tunnel-type flaps aims at the bilaminar reconstruction of lost gingival tissues using both free and recipient connective tissue layers to preserve graft viability and to cover denuded root surfaces. As a result of the dual graft blood supply (from the underlying periosteum and the overlying mucosal flap), the SCTG treatment results in improved root coverage [23].
It has been suggested that the underlying connective tissue is decisive in determining epithelial keratinization [18, 19] in the overlying flap. Nevertheless, in the case of SCTG transplantation, this phenomenon is significantly less pronounced compared to FGGs, and the induced limited keratinization is associated with more favorable tissue blending [20]. Therefore, the result is an enhanced color match and more esthetic results due to the surface characteristics of the overlying flap being similar to the adjacent recipient gingiva. In addition, if SCTG is harvested via partial-thickness flap preparation, wound healing in both the donor and recipient sites occurs mostly by primary intention. This may enhance tissue maturation and may also reduce postoperative discomfort [24].
Application of SCTGs provides excellent esthetics and predictability. On the other hand, SCTGs are not the first grafts of choice in cases where the surface characteristics of gingival tissues ought to be changed, or a substantial increase of the width and thickness of keratinized gingiva is necessary. Moreover, in patients with thin palatal masticatory mucosa presenting limited amount of donor tissues, instead of the hard palate, alternate donor sites (e.g., maxillary tuberosity or mandibular alveolar tuberculum) or the application of allogeneic and xenogeneic grafts should be considered [25, 26].
8.2.3 Partly Epithelialized Soft Tissue Grafts
Covering exposed roots in sites with thinned keratinized gingiva and shallow vestibule presents a challenge for clinicians. In case of high frenal attachment or muscle pull, in particular in the anterior mandible, transplantation SCTGs in combination with pedicle or tunneled flaps might deliver impaired graft stability and thus lead to treatment failure. On the other hand, placement of FGGs combined with apically repositioned flaps ensures graft stability, however, associated with unfavorable esthetic outcomes.
Therefore, as an alternative combining the benefits of FGGs and SCTGs, an epithelialized-subepithelial connective tissue graft (ESCTG) (Fig. 8.1) has been proposed to treat such cases [27] combined with an envelope type flap. When utilizing this approach, the epithelialized graft portions are placed to cover the denuded root surfaces. A similar grafting procedure, the partly epithelialized free gingival graft (PE-FGG), has been suggested to treat gingival recessions of the anterior mandible in combination with an apically repositioned flap [28]. Both approaches deliver increased resistance against the tension of the muscular-mucosal environment, lowering the risk for displacement of the mucogingival junction (MGJ) or flattening of the vestibule. Furthermore, increased amount of keratinized tissues and color blending similar to application of SCTGs have been reported by both authors following complete tissue maturation.
8.3 Anatomical Considerations of Choosing Free Autogenous Soft Tissue Graft Donor Sites
Free autogenous soft tissue grafts require a second surgical area as a donor site. As with all periodontal surgical procedures, harvesting of autogenous soft tissue grafts is highly technique sensitive. To avoid surgical complications, thorough knowledge of the anatomy of the donor area is essential.
In daily clinical practice, the area of choice is usually the hard palate that eventually might increase postoperative patient morbidity. Furthermore, autogenous soft tissue grafts can be obtained from the maxillary tuberosity, from edentulous ridges (e.g., from the mandibular alveolar tuberculum), or on some occasions from gingival donor sites. When treating patients, who refuse graft harvesting from the previously mentioned donor areas, allogeneic or xenogeneic grafts might be utilized instead of autogenous soft tissue grafts [25].
8.3.1 Anterior Part of the Hard Palate
The hard palate is the most common soft tissue graft donor site; however, the dimensions of the masticatory mucosa may influence the amount of soft tissue that can be harvested. Histological analysis of the hard palate shows three tissue layers: epithelium (0.3–0.6 mm), lamina propria (1–1.5 mm), submucosa, and periosteum. The submucosa contains adipose tissues and small salivary glands [29].
The thickness of the masticatory mucosa shows significant individual variance and is also dependent on the donor site location within the hard palate. The thickest tissues are found from the distal aspect of the canine toward the mesial aspect of the palatal root of the first molar, where the mucosa usually thins out significantly due to root prominence. The second anatomical aspect that should always be taken into account is the greater palatine artery (GPA). The palatal neurovascular bundle runs through a bony groove anterior to the greater palatal foramen from which it emerges. The anastomosis with the end branches of the nasopalatine artery is located in the premaxilla, anterior from the canine (Fig. 8.2). The main course of the GPA is located at an average of 12 mm distance from the gingival margin at the canine region; this is increased to 14 mm in the molar region [30]. According to other authors, the GPA runs at approximately 76% of the global palatal height, measured from the cemento-enamel junction (CEJ) of maxillary molars [31]. To analyze the thickness at palatal donor sites, an endodontic needle with a silicon stop might be used. The transition between the lamina propria and the adipose submucosa might be recognized by this approach [32].
8.3.2 Maxillary Tuberosity and Posterior Part of the Hard Palate
The maxillary tuberosity and the masticatory mucosa of the hard palate at the level of the second and third molars demonstrate similar histological characteristics: both contain a high amount of dense collagen fibers without adipose and glandular tissues [33].
The masticatory mucosa in the maxillary tuberosity can be thicker (over 4 mm) compared to the palate (not more than 3 mm) [34], which allows to harvest grafts of high volume in the absence of third molars, especially when retrieved 2–3 months following tooth removal. Therefore, along the distal portion of the hard palate, it is well indicated for recession coverage, in some cases allowing for sufficient graft removal for complete unilateral graft coverage. SCTGs obtained from these areas are firm compared to the anterior hard palate, thus showing less postoperative shrinkage, but on the other, they are more likely to develop graft hyperplasia and scar formation similar to FGGs [29].
8.3.3 Edentulous Ridges (Mandibular Alveolar Tuberculum)
In a minority of the cases, edentulous ridges, in particular the mandibular alveolar tuberculum in the absence of third molars, can be considered as an alternative, less traumatic donor site for autogenous soft tissue harvesting. The quality of harvested tissues resembles the characteristics of grafts from the posterior hard palate. This approach might be applied when treating mandibular recessions; since the donor site is located close to the recipient teeth, palatal harvesting approach can be avoided.
8.4 Soft Tissue Graft Harvesting Techniques
8.4.1 Free Gingival Graft Harvesting
Sullivan and Atkins were the first to use the hard palate as a donor site for FGG harvesting in 1968; this has become the standard and has not changed ever since. The optimal site for obtaining a FGG for root coverage is the area distally from the canines, starting at least 2 mm from the gingival margin in 5–8 mm width, in a desirable length to cover the whole recipient site. Graft preparation is first outlined by two parallel longitudinal incisions, interconnected with vertical incisions. Deliberation of the graft is initiated along the paramarginal incision in a split-thickness fashion, aiming at the removal of a 1.5–2-mm-thick FGG and leaving the periosteum untouched. The harvested tissue may serve as a ready-to-use FGG, or alternatively, it may be deepithelialized outside of the mouth to obtain an SCTG containing the lamina propria, which is rich in dense collagen fibers, especially when retrieved adjacent to the second molar, from the posterior part of the hard palate. The donor wound is a subject of secondary intention wound healing; therefore, several methods have been proposed to enhance epithelial ingrowth and thus shorten healing time. These include the placement of native collagen sponges or matrices (Fig. 8.3), fixed with horizontal or crossed mattress sutures, or alternatively delivering a prefabricated acrylic plate to cover the palate for increased blood clot stabilization. None of these methods has shown significantly more reduction in postoperative patient complaints, donor site pain, postoperative bleeding, and prolonged healing due to eventual tissue necrosis. The abovementioned complications are well-known side effects of palatal full-thickness graft harvesting [20].
8.4.2 Trap Door Technique
Edel was the first to report on the trap door technique in which epithelium is not removed from the palate for SCTG harvesting [15]. This technique utilizes mesial and distal vertical incisions according to the graft dimensions. A longitudinal incision from mesial to distal along the palate is used to connect the releasing incisions to elevate a partial-thickness trap door (Fig. 8.4). Vertical incisions should be extended 1 mm further over intended apicocoronal dimension of the graft, thus allowing for better access to the apical incision line, used for the removal of a SCTG from underneath the trap door by means of split-thickness sharp dissection as described for the single-incision technique in Sect. 8.4.4. After removing the connective graft, single interrupted or horizontal/modified crossed mattress sutures can be used to achieve wound closure. Donor site-related complications including secondary intention wound healing and postoperative bleeding might occur, mainly as a result of vertical incisions.
8.4.3 Parallel Incision Technique
The parallel incision technique was introduced by Langer and Calagna [21], followed by Harris in 1997 [35]. At 2 and 4 mm distance from the gingival margin, two longitudinal parallel split-thickness incisions are made (as described for the single-incision technique below) in 8–10 mm depth into the palate with vertical releases at the mesial and distal extent of the incision, similar to the trap door technique (Fig. 8.4). An incision at the base of the connective tissue between the vertical incisions deliberates the graft from the palatal bone. An SCTG is obtained by removing the epithelial collar determined by the first parallel incisions. In case a ESCTG/PE-FGG is needed, without deepithelialization, this harvesting technique represents a favorable approach. Donor site-related complications are comparable to the trapdoor approach, with more pronounced secondary intention wound healing.
8.4.4 Single-Incision Technique
Hürzeler and Weng introduced the single-incision technique in 1999 [24], confirmed by Lorenzana and Allen in 2000 [36]. First, a single full-thickness incision is performed with a Nr. 15 surgical blade 90 degrees to the palatal bone, after which the blade is angled from 135 to 180 degrees to undermine the palatal masticatory mucosa in split thickness toward the palatal midline (Fig. 8.4). This incision should not be extended deeper than 8 mm from the single incision. Therefore a No. 15 scalpel blade might be used as a reference, which has a cutting edge of the approximate length of 8 mm [29]. Subsequently, after opening a palatal envelope, a SCTG is obtained by incisions on mesial, distal, and apical sides of the connective tissue within the opened envelope. The most significant advantage of this technique is the less compromised blood supply and simplified closure of the wound due to the lack of vertical incisions; however, visibility is impaired [24].
Care has to be taken to avoid the greater and lesser palatine nerves and vessels. Incisions should be limited to the distal of the canine to avoid the greater palatine nerve and artery as they run closer to the CEJ in the anterior area. While harvesting a graft, the incision should be at least 2 mm apically from the gingival margin of the teeth to avoid necrosis of the marginal tissues during donor site healing due to impaired collateral blood supply [29].
The harvested SCTG may be obtained with or without periosteum; the latter might represent better mechanical properties but more negative postoperative consequences in case of a thin masticatory mucosa. The single-incision technique delivers the most favorable postoperative healing when achieving primary intention wound healing; nevertheless, donor site pain and necrosis might occur on certain occasions.
8.4.5 Distal Wedge Technique
The distal wedge approach was originally introduced to correct soft tissue excess at the distal aspects of maxillary second molars during resective periodontal pocket elimination procedures [37]. This procedure could be adapted to individual patient characteristics for esthetically intended graft harvesting indications. In case the most distal tooth is a first molar, the graft harvesting site can be extended; in the presence of the third molar, graft dimensions will be limited. This technique may also be applied to obtain SCTGs, FGGs, or partly epithelialized connective tissue grafts from the maxillary tuberosity as well as from the mandibular alveolar tuberculum.
The distal wedge procedure is carried out by placing two mesiodistal, apically diverging incisions outlining a graft area with a trapezoid cross section. Incisions should start from the distal surface of the last adjacent tooth and are extended as distal as possible within the masticatory mucosa. Incisions may be placed outlining a rectangular or a triangular graft shape from the occlusal view [29] (Fig. 8.4). This technique may also be performed with simultaneous wisdom tooth removal, nevertheless graft harvesting approximately 2 months following tooth extraction is even more preferable due to the increased amount of connective tissue at the donor site. In most cases the donor wound can be closed following slight buccal split-thickness flap mobilization; in case not, secondary intention wound healing is associated with very limited patient complaints. Therefore, the distal wedge approach is one of the most preferable autogenous graft harvesting procedures in terms of low patient morbidity besides the high quality of harvested tissues [29].
8.5 Healing of Autogenous Soft Tissue Grafts
During the integration of autogenous soft tissue grafts mainly reparative healing occurs, a long epithelial junction is formed. Real periodontal regeneration can only occur at the alveolar crest [38,39,40]. Wound healing after autogenous soft tissue transplantation takes place in three phases according to animal studies, performed in dogs and monkeys [41, 42].
-
(a)
Initial healing (0–3 days): Graft survival is ensured through avascular “plasmatic circulation” originating from the recipient bed. A thin residual exudate layer between graft and recipient tissues is formed following the pressure applied to remove most of it and following graft securing at the recipient site. This “plasmatic circulation” might be compromised in case thick exudate or a blood clot is left over, ultimately resulting in graft rejection. Epithelial layer of FGGs is prone to early necrosis with subsequent desquamation.
-
(b)
Revascularization (3–11 days): Within 2–3 days after surgery, anastomosis between graft and recipient site blood vessels reestablishes tissue circulation at the site of surgery. Thereafter, progressive vascular proliferation gradually results in the formation of a dense capillary network. Simultaneously, a fibrous attachment between the graft and recipient connective tissue is established. Reepithelialization of non-submerged grafts occurs mostly by adjacent tissue proliferation.
-
(c)
Tissue maturation (11–42 days). The vascular network of the graft regains normal structure and function. Furthermore, epithelium maturation gradually occurs along with the establishment of a keratin layer during this stage of healing.
8.6 State-of-the-Art Surgical Techniques in Combination with Free Autogenous Soft Tissue Grafts
Several techniques have been described in literature in combination with free autogenous soft tissue grafts to treat single and multiple gingival recessions. Among these, there are several other techniques, which proved to be less technique sensitive and deliver more predictable esthetic results compared to more individual surgical approaches with higher risk for complications, e.g., the laterally repositioned flap [1, 43] (Fig. 8.5) and the double papilla flap [44] (Fig. 8.6). Widely recognized and universally applicable surgical techniques for root coverage in single- and multiple recession-type defects [45] will be discussed in detail. The following surgical approaches are currently considered as state-of-the-art for the clinical application in conjunction with FGGs, SCTGs, and partly epithelialized autogenous soft tissue grafts.
8.6.1 Surgical Techniques in Combination with FGGs and PE-FGGs
8.6.1.1 One-Stage FGG/PE-FGG Technique
Björn in 1963 and Nabers in 1966 were the first to suggest the apically repositioned flap in combination with FGGs for widening the zone of the attached gingiva [4, 5]. Single or multiple recession defects may be treated by this approach for root coverage and for the augmentation of the keratinized gingiva, preferably in the lateral zone, due to the expected color blending heterogeneity between graft and recipient tissues. According to the standard apically repositioned flap procedure, after root planing, a split-thickness flap is prepared, and the superficial layer is removed in a 3–5-mm-wide zone, preparing a recipient periosteal bed. The donor site is usually the hard palate or the maxillary tuberosity. The harvested FGG is adapted to the recipient periosteum and adjacent gingiva by either resorbable or nonresorbable 6/0 monofilament sutures. Pressure is exerted for 1–2 min to prevent graft necrosis caused by blood clot between the two layers. Application of a gingival dressing was suggested for wound protection, nevertheless this procedure may lead to possible postoperative suture loosening caused by the dressing.
In the first few days of healing, the graft receives nutrition from the periosteum via diffusion; revascularization takes place as described in Sect. 8.5. Sutures are removed after 14 days. Tissue maturation and connective tissue formation needs 6–8 weeks to complete.
Partial recession or graft necrosis may occur during healing in case of adverse events causing infection or graft loosening. If there is no inflammation and wound healing is uneventful, graft surface desquamation and subsequent reepithelialization from adjacent sites takes place. Graft overgrowth due to hyperplasia may be observed as excessive creeping attachment between 6 and 12 months after surgery [46, 47]. This may result in frequently occurring color and texture mismatch between graft and adjacent sites, which is the ultimate drawback of this approach, limiting its use in the esthetic zone nowadays (Fig. 8.7).
As an alternative, placement of a PE-FGG with the same surgical approach was proposed [28], delivering similar tissue stability with more favorable color blending.
8.6.1.2 Two-Stage FGG Technique
The two-stage FGG technique was published by Bernimoulin et al. in 1975 [14,15,16,]. It is based on a first surgery for gingival augmentation and a second surgery for coronal repositioning of the integrated graft. The first stage of surgery is similar as described in Sect. 8.6.1.1. The main difference to the classical apically repositioned flap-FGG approach is that the existing keratinized gingiva is preserved; following an incision at the MGJ, an FGG is adapted to a periosteal bed apically to widen the residual keratinized gingiva around recession sites. Following at least 2 months of graft integration, a coronally advanced flap (CAF) is raised to reposition the previously enlarged zone of keratinized tissues for root coverage. The second stage of surgery is carried out as described in Sect. 8.6.2.1.
8.6.2 Surgical Techniques Used in Combination with SCTGs and ESCTGs for Single Recession Coverage
8.6.2.1 Coronally Advanced Flap Technique
The coronally advanced flap (CAF) was first described by Brustein as a cosmetic periodontics-coronally repositioned pedicle graft [48] and modified by others [49, 50]. The CAF is recognized as the most predictable technique to cover single gingival recessions according to recent systematic reviews [51].
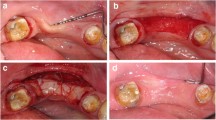
The surgical site is outlined by a bilateral trapezoid incision on the mesial and distal aspects of the exposed root surface as follows: bilateral horizontal split-thickness incisions are placed by 15C or microsurgical blades at a distance from the tip of the anatomical papilla exceeding the depth of the recession by 1 mm. These are followed by diverging vertical releasing incisions. A split-thickness flap is prepared at the papillary zone, followed by full-thickness elevation of the attached gingiva by blunt elevators from the level of the gingival zenith to the MGJ. Subsequently, flap elevation is continued in a split-thickness manner by sharp dissection from the MGJ deep apically into the vestibule, detaching the loose and flexible mucosal-submucosal layers from the underlying muscles and periosteum. Following complete flap mobilization, a previously harvested SCTG may be placed at the level of the CEJ. The graft can be fixed with resorbable sutures to the recipient periosteal bed or alternatively via mattress sutures to the adjacent mucosa. Thereafter, anatomical papillae are fully deepithelialized, and the flap is advanced coronally to achieve full coverage of the deepithelialized anatomical papillae by the surgical papillae. The flap is secured via double-sling nonresorbable 6/0 monofilament sutures to secure the flap margin 1 mm coronally from the CEJ. Lastly, the vertical incisions are closed by diagonally placed single interrupted sutures, starting from the most apical aspect. Pressure is exerted for 1–2 min to prevent graft necrosis caused by blood clot between the layers. Sutures are removed after 14 days (Fig. 8.8).
Maynard in 1977 [52] outlined the following requirements as criteria for success when utilizing CAF as part of the two-stage FGG approach: presence of shallow crevicular depths on proximal surfaces, anatomical interproximal bone heights, tissue height within 1 mm of the cemento-enamel junction of adjacent teeth, 6-week healing of a FGG prior to coronal positioning, reduction of any root prominence, and adequate flap release during surgery to prevent retraction during healing.
With the evolution of the technique and subsequent modifications, these anatomical limitations have been revised and partly extended. According to today’s standards, complete root coverage is possible in Miller class I–II cases by using the CAF approach; partial coverage can be expected in Miller class III cases. Nevertheless, the lack of keratinized gingiva is still a major limitation of the CAF, requiring combination therapy with SCTG or the choice of a different surgical approach. More favorable long-term results may be achieved in combination with simultaneous SCTG or previous FGG placement, compared to CAF alone; this may prevent the recurrence of recessions after 5 years [9].
8.6.2.2 Semilunar Coronally Advanced Flap Technique
The semilunar coronally advanced flap (SCAF) procedure [54] is an alternative to the CAF to treat single or multiple recessions with at least 3-mm-wide and 1-mm-thick keratinized gingiva apically from the defect. The SCAF is applicable for limited indications, without additional free soft tissue grafts (Fig. 8.9).
Following local anesthesia, a semilunar incision is placed at the level of the MGJ. Then a split-thickness flap is raised starting from the sulcus. The mobilized keratinized gingival collar is secured at the level of the CEJ by 6/0 nonresorbable monofilament sutures, which are removed after 14 days. SCAF delivers excellent esthetic results as well as long-term tissue stability; nevertheless it is only applicable in cases with thick gingival biotype and favorable baseline mucogingival conditions.
8.6.2.3 Envelope Technique
The envelope technique (ET) is used to cover Miller class I, II, and III gingival recessions in combination with SCTGs or ESCTGs. A split-thickness envelope flap (gingival pouch) is prepared by sharp dissection via 15C or microsurgical blades, starting from the sulcus toward the adjacent papillae, in a depth determined by the previously harvested graft size. Subsequently, an SCTG [55] or an ESCTG [27] is placed into the envelope at the level of the cemento-enamel junction. Graft and flap fixation is carried out by 5/0 or 6/0 nonresorbable monofilament single interrupted, mattress or sling sutures. Sutures are removed after 14 days (Fig. 8.10).
The uncovered part of the graft shows desquamation and reepithelization similar to FGGs during the healing period; however, this does not result in visible color blending heterogeneity compared to recipient tissues. Graft shrinkage and enlargement of the keratinized zone is more pronounced with the ESCTG approach due to the protective function of the preserved epithelial collar. A major advantage of this technique is the lack of coronal flap advancement, which makes the ET feasible to treat sites with a shallow vestibular fold, especially the anterior mandible.
8.6.3 Surgical Techniques Used in Combination with SCTGS and Partly Epithelialized Soft Tissue Grafts for Multiple Recession Coverage
8.6.3.1 Modified Coronally Advanced Flap Technique
The modified coronally advanced flap technique was published by Zucchelli and de Sanctis in 2000 [56]. This approach is the redesigned version of the original CAF for multiple recession coverage.
MCAF is similar to CAF in terms of split-thickness preparation of interdental papilla, full-thickness preparation of the keratinized gingiva between gingival zenith and MGJ, and split-thickness preparation of the mucosal flap beyond MGJ. Main differences can be found in the releasing incisions outlining the surgical papillae. These are similarly designed to the Zucchelli modification of the original CAF, with the oblique incisions always directed toward the center of the flap, which in most cases is either a canine or a midline papilla, which is tunneled. Subsequently graft insertion and suturing is carried out in the same fashion as described in Sect. 8.6.2.1 for CAF. Sutures are removed after 14 days (Fig. 8.11).
8.6.3.2 Subperiosteal Envelope Technique
The subperiosteal envelope technique (SET) is the adaptation of the original ET to treat multiple adjacent gingival recessions ([57, 58].
Following local anesthesia, planing of the exposed root surfaces is carried out. Intrasulcular incisions around involved teeth are performed using 15C or microsurgical blades. Mucoperiosteal envelope flap elevation is performed by blunt preparation via tunneling knives up to the level of the MGJ at each individual recession site, leaving the tip of the interdental papillae untouched. The separate mucoperiosteal envelopes are subsequently interconnected, resulting in a confluent tunnel preparation over the adjacent exposed root surfaces. Starting from here the MGJ preparation continues in split thickness in a depth of 3–5 mm. A subsequently harvested SCTG [58] or ESCTG [57] can be adapted to this supraperiosteal envelope by horizontal mattress sutures. The graft might be further secured by sling sutures. Sutures are removed 14 days after surgery (Fig. 8.12).
The main advantage of the SET is that the blood circulation of the papillae is not compromised; thus healing is usually uneventful, and postoperative complaints are minimized. Furthermore, due to the secondary epithelialization of the inserted grafts, enlargement of the keratinized zone can be observed, without color difference between the graft and the recipient site. Nevertheless, in certain cases, scar lines can occur due to epithelial invagination and cicatrization at the border of the exposed graft surfaces.
8.6.3.3 Modified Coronally Advanced Tunnel Technique
The modified coronally advanced tunnel technique (MCAT) is a modification of the original SET, delivering predictable root coverage even in Miller class III recessions. The main difference is that more excessive split-thickness flap mobilization is performed, attaching muscles and inserting collagen fibers are separated and released from the inner aspect of the alveolar mucosa by means of tunneling knifes and Gracey curettes. As a result, the tunneled flap can be mobilized and coronally advanced without tension. To achieve complete mobilization of the flap, interdental papillae are gently undermined using microsurgical elevators. Special attention is paid not to disrupt the interdental papillae. With the coronal margin positioned at the level of the CEJ, an SCTG is fixed to the mucosal flap via horizontal mattress sutures. Moreover, after securing the graft in the tunnel, the flap is advanced coronally by suspended or sling sutures. To enhance this, preoperatively resin bonding of adjacent contact points at the operation site may be performed to enable suspended suturing. In cases when complete graft coverage cannot be obtained with the suspended sutures, additional sling sutures are placed interdentally to enable coronal displacement of the tunnel 1 mm over the CEJ. Sutures are removed after 14 days [59] (Fig. 8.13).
The main indication for MCAT approach, when used in combination with SCTG, is the perfect color match and complete lack of any scar line at the treated sites. Furthermore, substantial root coverage can be achieved with this approach even in Miller class III multiple recessions. Nevertheless, increase in the width of keratinized tissues is lower compared to the original SET.
Conclusions
Establishing optimal pink esthetics by the reconstruction of single or multiple gingival recessions is the ultimate goal of periodontal plastic surgery. One of the most important considerations in surgical planning is the selection of the most feasible grafting approach to augment the gingival biotype and to fulfill patient expectations for long-term esthetics while keeping patient morbidity at the lowest possible level. To achieve complete root coverage and long-term tissue stability, increasing the width and thickness of the keratinized gingiva by the application of autogenous soft tissue grafts was shown to be the most effective grafting modality in a most recent systematic review [8]. Applying free autogenous soft tissue grafts requires a second surgical site with varying risks for possible complications (e.g., pain, swelling, infection, necrosis), which cannot be fully eliminated even by careful planning and high surgical skills. This may give rise to utilizing different valuable treatment alternatives, considering the application of novel allogeneic and xenogeneic grafting alternatives [8, 9]. Nevertheless, according to the 2015 consensus report of the AAP, “Predictable root coverage is possible for single-tooth and multiple-tooth recession defects, with SCTG procedures providing the best root coverage outcomes.”
In compromised clinical settings (e.g., recessions in the anterior mandible, compromised hard tissue environment in Miller class III defects, lack of keratinized gingiva, shallow vestibule, high muscular attachments and frenula, etc.), autogenous soft tissue grafts are recognized as gold standard treatment modalities in terms of predictable long-term esthetic outcomes.
References
Grupe J, Warren R. Repair of gingival defects by a sliding flap operation. J Periodontol. 1956;27:290–5.
Pennel BM, Higgason JD, Towner JD, Ko K, Fritz BD, Salder JF. Oblique rotated flap. J Periodontol. 1965;36:305–9.
Carnio J. Modified apically repositioned flap technique: a surgical approach to enhance donor sites prior to employing a laterally positioned flap. Int J Periodontics Restorative Dent. 2014;34(3):423–9.
Björn H. Free transplantation of gingiva propria (abstract) IN: Symposium in periodontology in Malmö. Odontologisk Revy. 1963;14:321–3.
Nabers JM. Extension of the vestibular fornix utilizing a gingival graft—a case report. Periodontics. 1966;4:77–9.
Lang NP, Löe H. The relationship between the width of the keratinized gingiva and the gingival health. J Periodontol. 1972;43:623–7.
Wennström JL, Lindhe J. Plaque induced gingival inflammation in the abscence of attached gingiva in dogs. J Clin Periodontol. 1983;10:266–76.
Cairo F, Pagliaro U, Buti J, Baccini M, Graziani F, Tonelli P, Pagavino G, Tonetti MS. Root coverage procedures improve patient aesthetics. A systematic review and Bayesian network meta-analysis. J Clin Periodontol. 2016;43(11):965–75.
Hofmänner P, Alessandri R, Laugisch O, Aroca S, Salvi GE, Stavropoulos A, Sculean A. Predictability of surgical techniques used for coverage of multiple adjacent gingival recessions—a systematic review. Quintessence Int. 2012;43(7):545–54.
Zucchelli G, Mounssif I, Mazzotti C, Stefanini M, Marzadori M, Petracci E, Montebugnoli L. Coronally advanced flap with and without connective tissue graft for the treatment of multiple gingival recessions: a comparative short- and long-term controlled randomized clinical trial. J Clin Periodontol. 2014;41(4):396–403.
Sullivan HC, Atkins JH. Freeutogenous gingival grafts. 1. Principles of successful grafting. Periodontics. 1968;6(1):5–13.
Bowers GM. A study of the width of attached gingiva. J Periodontol. 1963;34:201.
Kennedy JE, Bird WC, Dorfman HS. A longitudinal evaluation of varying widths of attached gingiva. J Clin Periodontol. 1985;12(8):667–75.
Bernimoulin JP, Lüscher B, Mühlemann HR. Coronally repositioned periodontal flap. Clinical evaluation after one year. J Clin Periodontol. 1975;2(1):1–13.
Edel A. Clinical evaluation of free connective tissue grafts used to increase the width of keratinised gingiva. Periodontal Clin Investig. 1974;20(1):12–20.
Haggerty PC. The use of a free gingival graft to create a healthy environment for full crown preparation. Case history. Periodontics. 1966;4(6):329–31.
Miller PD Jr. Root coverage using the free soft tissue autograft following citric acid application. III. A successful and predictable procedure in areas of deep-wide recession. Int J Periodontics Restorative Dent. 1985;5(2):14–37.
Karring T, Cumming BR, Oliver RC, Löe H. The origin of granulation tissue and its impact on postoperative results of mucogingival surgery. J Periodontol. 1975;46:577–85.
Karring T, Ostergaard E, Löe H. Conservation of tissue specificity after heterotopic transplantation of gingiva and alveolar mucosa. J Periodontal Res. 1971;6:282–93.
McGuire MK, Scheyer ET. Randomized, controlled clinical trial to evaluate a xenogeneic collagen matrix as an alternative to free gingival grafting for oral soft tissue augmentation. J Periodontol. 2014;85(10):1333–41.
Langer B, Calagna L. The subepithelial connective tissue graft. J Prosthet Dent. 1980;44(4):363–7.
Langer B, Langer L. Subepithelial connective tissue graft technique for root coverage. J Periodontol. 1985;56:715–20.
Tatakis DN, Chambrone L, Allen EP, Langer B, McGuire MK, Richardson CR, Zabalegui I, Zadeh HH. Periodontal soft tissue root coverage procedures: a consensus report from the AAP Regeneration Workshop. J Periodontol. 2015;86(2 Suppl):S52–5.
Hürzeler MB, Weng D. A single-incision technique to harvest subepithelial connective tissue grafts from the palate. Int J Periodontics Restorative Dent. 1999;19(3):279–87.
Aroca S, Molnár B, Windisch P, Gera I, Salvi GE, Nikolidakis D, Sculean A. Treatment of multiple adjacent Miller class I and II gingival recessions with a Modified Coronally Advanced Tunnel (MCAT) technique and a collagen matrix or palatal connective tissue graft: a randomized, controlled clinical trial. J Clin Periodontol. 2013;40(7):713–20.
Cummings LC, Kaldahl WB, Allen EP. Histologic evaluation of autogenous connective tissue and acellular dermal matrix grafts in humans. J Periodontol. 2005;76(2):178–86.
Stimmelmayr M, Allen EP, Gernet W, Edelhoff D, Beuer F, Schlee M, Iglhaut G. Treatment of gingival recession in the anterior mandible using the tunnel technique and a combination epithelialized-subepithelial connective tissue graft-a case series. Int J Periodontics Restorative Dent. 2011;31(2):165–73.
Cortellini P, Tonetti M, Prato GP. The partly epithelialized free gingival graft (pe-fgg) at lower incisors. A pilot study with implications for alignment of the mucogingival junction. J Clin Periodontol. 2012;39(7):674–80.
Zuhr O, Bäumer D, Hürzeler M. The addition of soft tissue replacement grafts in plastic periodontal and implant surgery: critical elements in design and execution. J Clin Periodontol. 2014;41(Suppl 15):S123–42.
Monnet-Corti V, Santini A, Glise JM, Fouque-Deruelle C, Dillier FL, Liébart MF, Borghetti A. Connective tissue graft for gingival recession treatment: assessment of the maximum graft dimensions at the palatal vault as a donor site. J Periodontol. 2006;77(5):899–902.
Benninger B, Andrews K, Carter W. Clinical measurements of hard palate and implications for subepithelial connective tissue grafts with suggestions for palatal nomenclature. J Oral Maxillofac Surg. 2012;70(1):149–53.
Zucchelli G, Mele M, Stefanini M, Mazzotti C, Marzadori M, Montebugnoli L, de Sanctis M. Patient morbidity and root coverage outcome after subepithelial connective tissue and de-epithelialized grafts: a comparative randomized-controlled clinical trial. J Clin Periodontol. 2010;37(8):728–38.
Gapski R, Satheesh K, Cobb CM. Histomorphometric analysis of bone density in the maxillary tuberosity of cadavers: a pilot study. J Periodontol. 2006;77(6):1085–90.
Müller HP, Schaller N, Eger T. Ultrasonic determination of thickness of masticatory mucosa: a methodologic study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88(2):248–53.
Harris RJ. A comparison of two techniques for obtaining a connective tissue graft from the palate. Int J Periodontics Restorative Dent. 1997;17(3):260–71.
Lorenzana ER, Allen EP. The single-incision palatal harvest technique: a strategy for esthetics and patient comfort. Int J Periodontics Restorative Dent. 2000;20(3):297–305.
Robinson RE. The distal wedge operation. Periodontics. 1966;4(5):256–64.
Gottlow J, Nyman S, Karring T, Lindhe J. Treatment of localized gingival recessions with coronally displaced flaps and citric acid. An experimental study in the dog. J Clin Periodontol. 1986;13(1):57–63.
Wennström JL. Mucogingival therapy. Ann Periodontol. 1996;1(1):671–701. Review
Wilderman N, Wentz M. Repair of a dentogingival defect with a pedicle flap. J Periodontol. 1965;36:218–36.
Nobuto T, et al. Microvascularization of the free gingival autograft. J Periodontal Res. 1988;59:639–46.
Oliver R, et al. Microscopic evaluation of healing and revascularisation of free gingival grafts. J Periodontal Res. 1968;3:84–95.
Zucchelli G, Cesari C, Amore C, Montebugnoli L, De Sanctis M. Laterally moved, coronally advanced flap: a modified surgical approach for isolated recession-type defects. J Periodontol. 2004;75(12):1734–41. Erratum in: J Periodontol. 2005 Aug;76(8):1425
Cohen DW, Ross S. The double papillae repositioned flap in periodontal therapy. J Periodontol. 1968;39(2):65–70.
Tonetti MS, Jepsen S, Working Group 2 of the European Workshop on Periodontology. Clinical efficacy of periodontal plastic surgery procedures: consensus report of Group 2 of the 10th European Workshop on Periodontology. J Clin Periodontol. 2014;41(Suppl 15):S36–43.
Matter J, Cimasoni G. Creeping attachment after free gingival grafts. J Periodontol. 1976;47(10):574–9.
Matter J. Creeping attachment of free gingival grafts. A five-year follow-up study. J Periodontol. 1980;51(12):681–5.
Brustein DD. Cosmetics periodontics- coronally repositioned pedicle graft. Dent Surv. 1970;46:22–5.
Allen EP, Miller PD. Coronal positioning of existing gingival. Short term result in the treatment of shallow marginal tissue recession. J Periodontol. 1989;60:316–9.
de Sanctis M, Zucchelli G. Coronally advanced flap: a modified surgical approach for isolated recession-type defects: three-year results. J Clin Periodontol. 2007;34(3):262–8.
Cairo F, Nieri M, Pagliaro U. Efficacy of periodontal plastic surgery procedures in the treatment of localized facial gingival recessions. A systematic review. J Clin Periodontol. 2014;41(Suppl 15):S44–62.
Maynard JG Jr. Coronal positioning of a previously placed autogenous gingival graft. J Periodontol. 1977;48(3):151–5.
Miller PD Jr. A classification of marginal tissue recession. Int J Periodontics Restorative Dent. 1985;5(2):8–13.
Tarnow DP. Semilunar coronally positioned flap. J Clin Periodontol. 1986;13:182–5.
Raetzke PB. Covering localized areas of root exposure employing the “envelope” technique. J Periodontol. 1985;56(7):397–402.
Zucchelli G, De Sanctis M. Treatment of multiple recession-type defects in patients with esthetic demands. J Periodontol. 2000;71(9):1506–14.
Allen AL. Use of the supraperiosteal envelope in soft tissue grafting for root coverage. I. Rationale and technique. Int J Periodontics Restorative Dent. 1994;14(3):216–27.
Zabalegui I, Sicilia A, Cambra J, Gil J, Sanz M. Treatment of multiple adjacent gingival recessions with the tunnel subepithelial connective tissue graft: a clinical report. Int J Periodontics Restorative Dent. 1999;19(2):199–206.
Aroca S, Keglevich T, Nikolidakis D, Gera I, Nagy K, Azzi R, Etienne D. Treatment of class III multiple gingival recessions: a randomized-clinical trial. J Clin Periodontol. 2010;37(1):88–97.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Windisch, P., Molnár, B. (2018). Recession Coverage Using Autogenous Grafts. In: Kasaj, A. (eds) Gingival Recession Management. Springer, Cham. https://doi.org/10.1007/978-3-319-70719-8_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-70719-8_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-70717-4
Online ISBN: 978-3-319-70719-8
eBook Packages: MedicineMedicine (R0)