Abstract
Optimal resuscitation of septic shock needs to be directed by a hemodynamic monitoring. Fluid responsiveness must be evaluated by dynamic parameters. Monitoring of cardiac output is also recommended. A central venous line, an arterial catheter, and echocardiography are the minimal monitoring required. In case of no response to treatment or concomitant ALI/ARDS, transpulmonary thermodilution should be used in addition to the basic monitoring.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Dynamic parameters of fluid responsiveness
- Arterial catheter
- Echocardiography
- Pulmonary artery catheter
- Transpulmonary thermodilution
To know in septic patients:
-
1.
The limitations of older hemodynamic methods
-
2.
The advantages, interests, and limitations of newer methods
-
3.
Why prefer dynamic over static parameters for the evaluation of fluid responsiveness
-
4.
Why prefer an integrated approach with bedside ultrasound and invasive techniques
1 Introduction
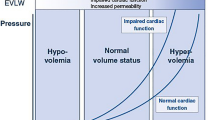
Septic shock is a complex interaction of several hemodynamic abnormalities:
-
Absolute hypovolemia
-
Relative hypovolemia due to general vasodilatation that coexists with zones of vasoconstriction
-
Myocardial dysfunction, even at an earlier stage
-
Inadequate oxygen extraction due to intracellular (probably mitochondrial) abnormalities
Septic shock is one subtype of shock states.
According to the type of shock, different abnormalities of circulatory parameters are found. Shock is commonly classified into four different subtypes with different pathophysiologies: distributive, cardiogenic, hypovolemic, and obstructive. In ◘ Table 33.1 are described the distinguishing features of the shock states. Septic shock can present one or several of these features. It is of crucial importance to diagnose these hemodynamic abnormalities to select the most appropriate treatments: fluids, vasopressors, inotropic agents, or combination of several [1]. This is always the case in septic shock. Finally, although there is no one best targeted hemodynamic goal in the resuscitations of septic shock patients, optimizing organ blood flow early before end-organ injury develops is usually associated with reduced morbidity and mortality, though no specific endpoints of resuscitations have been validated to be superior.
2 Monitoring
The goal of hemodynamic monitoring is to assess the adequacy of cardiopulmonary function, blood distribution, and oxygen delivery relative to tissue demand. In septic shock basic hemodynamic assessment (vital signs, physical exam) must be completed by invasive and noninvasive technologies.
2.1 Arterial Pressure
Monitoring blood pressure saves lives and the goal of resuscitation must always be tissue perfusion. An arterial catheter is always needed for the monitoring of septic shock patients. The mean arterial pressure should be kept around 60–65 mmHg, but a personalized blood pressure management is needed for each patient. Higher levels may be needed in older, formerly hypertensive patients. Lower levels may be enough in younger patients. A diastolic blood pressure below 40 mmHg is an indication to start immediate vasopressor therapy. Pulse pressure variation is a dynamic tool to evaluate a state of fluid responsiveness (see below).
Add (!) icon Blood pressure is the oldest form of perfusion monitoring. Values targeted during treatment must be individualized.
2.2 Prediction of Preload Responsiveness
Static measures can no longer be recommended. The followings do not reliably reflect the left ventricular filling pressure in shock states with pulmonary hypertension or compliance changes:
-
Central venous pressure
-
Pulmonary occlusion pressure
-
Ventricular end-diastolic volumes
-
Ventricular diastolic area
-
Global end-diastolic volume
-
Intrathoracic blood volume
Dynamic measures of fluid responsiveness are recommended instead. ◘ Tables 33.2, 33.3, and 33.4 present the parameters to use and the main limitations in their interpretations [2,3,4]. Ultrasound is useful to reduce the time of diagnostic uncertainty and guide resuscitation. They may be used to assess volume status, ventricular strain, and myocardial dysfunction [5,6,7,8]. Passive leg raising test can be used to predict volume responsiveness without giving a single drop of fluid. It should be titrated against changes in CO/SV rather than PP changes (◘ Tables 33.2 and 33.3).
Add (!) icon Dynamic parameters must be preferred over static parameters. Passive leg raising test evaluates the potential efficacy of fluid challenge without using a single drop of fluid.
2.3 Cardiac Output Monitoring
2.3.1 Noninvasive
Indirect measures of CO can be assessed using transesophageal Doppler or thoracic cutaneous bioimpedance or bioreactance (NICOM) for the latter further validation is required in shock state.
2.3.2 Invasive and Minimally Invasive Systems [9,10,11]
The pulmonary artery catheter has still a place and is recommended in the context of refractory shock, severe pulmonary artery hypertension or right ventricular dysfunction [11, 12]. These situations are not rare in septic shock. Other systems are capable to calculate stroke volume (SV) and CO using the pulse contour waveform analysis. They provide real-time, beat-by-beat arterial blood pressure and SV monitoring. The devices provide also parameters for the assessment of fluid responsiveness.
Pulse contour analysis-derived CO: Calibrated devices (PICCO, LIDCO, and volume view) are recommended. Non-calibrated devices (FlowTrac/Vigileo, ProAQT, LiDCO Rapid, MostCare) should not be used in situations where changes in ventricular strain are expected. Calibrated system can use transpulmonary thermodilution (PICCO, volume view) or lithium dilution. Calibration is needed on a regular basis and every time there are significant hemodynamic changes (starting a vasopressor, etc.).
Add (!) icon Transpulmonary thermodilution is the method of choice and should be preferred over non-calibrated methods.
2.4 CO and Mixed (Central) O2 Saturation Monitoring
CO is the vehicle for oxygen and nutriments to the cells. Regardless of the nominal value, CO is either adequate or inadequate to meet the metabolic demand. SVO2 or ScVO2 is a reasonable indicator of the adequacy of the CO, when it is low or normal. Based on the results of three multicenter studies, a resuscitation protocol targeting a specific value of ScVO2 does not offer survival advantage; the Surviving Sepsis Campaign Guidelines no longer recommend the use of ScVO2 in the management of septic shock patients [13,14,15]. We believe this is unfortunate because ScVO2 provides valuable information for the interpretation of the adequacy of CO. Anyway, once a central venous catheter is in place, why not draw blood samples for ScVO2 measurements.
Add (!) icon CO and ScVO2 evaluated together are helpful for the management of septic shock.
2.5 Microcirculation Monitoring
Sublingual microcirculation monitoring opens a window on the assessment of microcirculation. Persistent abnormalities predict the development of organ dysfunction and subsequent mortality; unfortunately, at the present time, there are no specific strategies to alter the microcirculation [16].
2.6 Recommendations for Practice
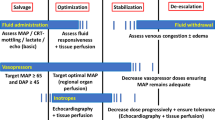
In ◘ Fig. 33.1 is proposed a two-step protocol for the optimal choice of monitoring septic shock patients.
Minimal or advanced monitoring for the diagnosis and treatment of septic shock. ScvO2 central venous blood oxygen saturation, SP systolic pressure, DP diastolic pressure, MAP mean arterial pressure, PP pulse pressure, ALI acute lung Injury, ARDS acute respiratory distress syndrome, CO cardiac output, ΔPP delta pulse pressure, SVV stroke volume variation, EVLW extravascular lung water, SVR systemic vascular resistance
Take-Home Messages
-
Blood pressure is the oldest form of perfusion monitoring. Values targeted during treatment must be individualized.
-
Dynamic parameters must be preferred over static parameters. Passive leg raising test evaluates the potential efficacy of fluid challenge without using a single drop of fluid.
-
Transpulmonary thermodilution is the method of choice and should be preferred over non-calibrated methods.
-
CO and ScVO2 evaluated together are helpful for the management of septic shock.
Conclusion
The ability to monitor the different hemodynamic abnormalities of septic shock, as well as the efficacy of treatments, is of crucial importance to improve outcome. The pathophysiology of septic shock is always complex, but the correct use of monitoring techniques, always in combination, can help prescribe the optimal treatments. There is no one best monitoring modality for septic shock patients. All are subject to measurement errors, and results may be affected by patient pathophysiology or interaction with mechanical ventilation. For the less severe forms of septic shock, minimal monitoring with a central venous line, an arterial catheter, and echocardiography is recommended. For more severe forms, or in case of associated ALI/ARDS, the use of transpulmonary thermodilution, or pulmonary artery catheter, is needed to select and monitor the appropriate treatments.
References
Cecconi M, De Backer D, Antonelli M, et al. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014;40:1795–815.
Marik PE, Cavallazzi R, Vasu T, et al. Dynamic changes in arterial waveform derived variables and fluid responsiveness in mechanically ventilated patients: a systematic review of the literature. Crit Care Med. 2009;37:2642–7.
Cavallaro F, Sandroni C, Marano C, et al. Diagnostic accuracy of passive leg raising for prediction of fluid responsiveness in adults: systematic review and meta-analysis of clinical studies. Intensive Care Med. 2010;36:1475–83.
Marik PE, Baram M, Vahid B. Does central venous pressure predict fluid responsiveness? A systematic review of the literature and the tale of seven mares. Chest. 2008;134:172.
Levitov A, Frankel HL, Blaivas M, et al. Guidelines for the appropriate use of bedside general and cardiac ultrasonography in the evaluation of critically ill patients-part II: cardiac ultrasonography. Crit Care Med. 2016;44:1206–27.
Barbier C, Loubières Y, Schmit C, et al. Respiratory changes in inferior vena cava diameter are helpful in predicting fluid responsiveness in ventilated septic patients. Intensive Care Med. 2004;30:1740–6.
Spencer KT, Kimura BJ, Korcarz CE, et al. Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26:567–81.
Monnet X, Marik P, Teboul JL. Passive leg raising for predicting fluid responsiveness: a systematic review and meta-analysis. Intensive Care Med. 2016;42:1935–47.
Monnet X, Anguel N, Naudin B, et al. Arterial pressure-based cardiac output in septic patients: different accuracy of pulse contour and uncalibrated pressure waveform devices. Crit Care. 2010;14:R109.
Monnet X, Teboul JL. Transpulmonary thermodilution: advantages and limits. Crit Care. 2017;21:147.
Connors AF Jr, Speroff T, Dawson NV, et al. Support Investigators. The effectiveness of right heart catheterization in the initial care of critically ill patients. JAMA. 1996;276:889–97.
Pinsky MR, Vincent JL. Let us use the pulmonary artery catheter correctly and only when we need it. Crit Care Med. 2005;33:1119–22.
Rhodes A, Evans LE, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45:486–552.
The PRISM investigators. Early, goal-directed therapy for septic shock – a patient-level meta-analysis. N Engl J Med. 2017;376:2223–34.
Hernandez G, Teboul JL. Fourth surviving sepsis campaign’s hemodynamic recommendations: a step forward or a return to chaos? Crit Care. 2017;21:133.
Kiyatkin ME, Bakker J. Lactate and microcirculation as suitable targets for hemodynamic optimization resuscitation of circulatory shock. Curr Opin Crit Care. 2017;23:348–54.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 European Society of Intensive Care Medicine
About this chapter
Cite this chapter
Martin, C., Duclos, G., Leone, M. (2019). Cardiopulmonary Monitoring of Septic Shock. In: Pinsky, M.R., Teboul, JL., Vincent, JL. (eds) Hemodynamic Monitoring. Lessons from the ICU. Springer, Cham. https://doi.org/10.1007/978-3-319-69269-2_33
Download citation
DOI: https://doi.org/10.1007/978-3-319-69269-2_33
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-69268-5
Online ISBN: 978-3-319-69269-2
eBook Packages: MedicineMedicine (R0)