Abstract
Acute promyelocytic leukemia (APL) typically presents with a life-threatening hemorrhagic diathesis. New treatment regimens for remission induction including all-trans-retinoic acid (ATRA) or arsenic trioxide (ATO) have significantly improved the mortality due to lethal bleeding, which however remains one of the major barriers to the cure of APL. Simultaneous to bleeding manifestations is often the occurrence of thrombosis, which constitutes an integral part of the thrombo-hemorrhagic syndrome (THS) accompanying the clinical presentation of the disease. The laboratory abnormalities of blood coagulation in these patients are consistent with the diagnosis of disseminated intravascular coagulation (DIC) with excess hyperfibrinolysis.
Major determinants of the coagulopathy of APL are endogenous factors expressed by the leukemic cells, including procoagulant factors, fibrinolytic proteins, and non-specific proteolytic enzymes. In addition, these cells have an increased capacity to adhere to the vascular endothelium and secrete inflammatory cytokines, which in turn stimulate the prothrombotic activities of endothelial cells and leukocytes.
This chapter will summarize our current knowledge on the epidemiology and pathogenesis of the APL-associated THS and will overview the therapeutic approaches for the management of bleeding and thrombotic complications.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Acute promyelocytic leukemia
- Thrombo-hemorrhagic syndrome (THS)
- Disseminated intravascular coagulation (DIC)
- Tissue factor
- Cancer procoagulant
- Cytokines
- Fibrinolysis
- All-trans-retinoic acid (ATRA)
- Arsenic trioxide
Introduction
Hemorrhages occur frequently in patients with acute leukemias and significantly affect morbidity and mortality [1]. Besides thrombocytopenia due to bone marrow failure, alterations of the hemostatic system contribute to the bleeding diathesis in these patients. In particular, patients with acute promyelocytic leukemia (APL) very often present with a range of laboratory abnormalities consistent with the diagnosis of disseminated intravascular coagulation (DIC) , with an excess fibrinolysis activation, and may show a variety of clinical manifestations ranging from diffuse life-threatening bleeding secondary to the consumption of coagulation factors and platelets to localized venous or arterial thrombosis [2, 3]. Bleeding and clotting manifestations may take place concomitantly as part of the same thrombo-hemorrhagic syndrome (THS) . Indeed, a profound dysregulation of the hemostatic system, due to the imbalance between procoagulant, anticoagulant, and profibrinolytic mechanisms, occurs in these patients [4]. A hemorrhagic phenotype prevails when the consumption of clotting factors and platelets and activation of fibrinolysis dominate the picture. A THS can occur to different extent in all acute myeloid leukemia subtypes [5, 6]; however, in patients with APL, hemorrhage is usually predominant and is relevant for mortality rates [7]. In recent years, the APL-associated coagulopathy has received new interest, due to the enhanced understanding of the biology of this unique myeloid differentiation disorder and to the greater sensitivity of diagnostic laboratory tests for coagulation abnormalities. In addition, most importantly, the development of new and very efficacious therapeutic drugs for APL remission induction, i.e., all-trans-retinoic acid (ATRA) and arsenic trioxide (ATO) , has attracted much attention for the beneficial effects of these therapies on the coagulation disorder. ATRA-induced differentiation of leukemic promyelocytes with remission of APL is indeed accompanied by prompt improvement of the hemorrhagic symptoms [4]. Both ATRA and ATO, as single agents, induce the molecular remission of APL and a simultaneous rapid resolution of the related coagulopathy. ATO combined to ATRA is effective in inducing APL remission in newly diagnosed patients and may provide an alternative to ATRA + chemotherapy in this disease, with less toxic effects [8].
In this chapter, we will focus on the clinical aspects, the pathogenesis, and the proposed treatments of the THS occurring in APL.
The Coagulopathy of APL
Clinical Manifestations
APL typically presents with a life-threatening hemorrhagic diathesis, the clinical and laboratory features of which are consistent with DIC [9]. The bleeding disorder is particularly severe in the microgranular variant of APL (M3v), characterized by marked hyperleukocytosis. Before the introduction of ATRA for remission induction, APL was distinguished by a high incidence of hemorrhagic death (20%), which significantly contributed to treatment failure [10].
Currently, standard treatment of APL with ATRA and chemotherapy results in more than 90% complete remission rates accompanied by a resolution of the coagulopathy, with a reduction of early hemorrhagic deaths to 2.4–6.5% [11, 12]. Despite this reduction, lethal or life-threatening hemorragic complications still occur, while the coagulopathy of APL is active. These complications are not only the most frequent cause of death early during induction therapy but can also occur before the diagnosis of APL has been made and therapy started. It should be noted that an undetermined number of patients will die before starting any differentiating agent, being acute bleeding the main cause of death in this setting. According to the data from the Swedish registry [13], 12 out of 105 patients (11.4%) had early hemorrhagic death before treatment, but this incidence is lower in the Spanish registry (3.5% out of more than 2000 cases, unpublished data). After the systematic introduction of ATRA , most early deaths have been recorded within the first 2–3 weeks [14]. A retrospective analysis showed that delays in starting ATRA led to increased early hemorrhagic death [15].
In spite of the dramatic amelioration in the rate of remission induction in patients with APL and overall improvement in survival, hemorrhage remains the most common cause of early death, accounting for 5% of the cases (37 of 736 evaluable patients) in two consecutive Programa Español de Tratamiento de las Hemopatias Malignas (PETHEMA) group studies—PETHEMA LPA96 and LPA99 [16]. Hemorrhagic deaths were almost exclusively due to intracranial (65%) and pulmonary hemorrhages (32%), with only one case of fatal gastrointestinal bleeding. It should be noted that 2 of the 24 patients with intracranial bleeding developed the hemorrhage over an extensive cerebral thrombosis. The results of a multivariate analysis of pretreatment characteristics predictive of fatal hemorrhage implicated an elevated white blood cell count (WBC > 10 × 109/L; P < 0.0001) and an abnormal creatinine level (P < 0.0004). Of note, in the LPA99 study, routine use of tranexamic acid prophylaxis (100 mg/kg/day by continuous infusion) aimed to inhibit excess fibrinolysis failed to alter the risk of hemorrhagic death (5% in both studies). Furthermore, the use of tranexamic acid was associated with a trend toward a statistically significant increase in the thrombosis rate (6% in LPA99 vs. 3% in LPA96 trial, in which tranexamic acid was not utilized; P = 0.08 in multivariate analysis). Fatal hemorrhagic events occurred from day 1 to day 23 with the majority noted in the first week (21, 57%); no lethal hemorrhages were documented beyond the fourth week of therapy.
There are no consistent data regarding the frequency of the coagulopathy accompanying APL at presentation, probably because there is no homogeneous definition for this complication. In this regard, the PETHEMA group analyzed the rate of coagulopathy in 921 patients registered in two protocols with ATRA and chemotherapy (unpublished data), using the following definition for DIC : presence of thrombocytopenia along with (1) prolonged prothrombin time and/or activated partial thromboplastin and (2) hypofibrinogenemia and/or increased levels of fibrin degradation products or D-dimer. They found that coagulopathy was present at diagnosis in 65% of patients, and additionally 12% developed this complication during induction. Specifically, hypofibrinogenemia was present in 46% of patients, and additionally 10% manifested decrease of fibrinogen levels below 170 mg/dL during induction period. The median time to resolution of DIC was 11 days from diagnosis (range, 1–53 days).
Concomitant to the bleeding diathesis, thrombotic events occur in APL patients with an incidence rate ranging from 2 to 15% [17,18,19]. Interestingly, ATRA increases the thrombotic risk in those patients who manifest accelerated differentiation, otherwise known as the “differentiation syndrome” [1, 20]. More recently, Mitrovic et al. described a thrombotic event rate of 20.6% (6.3% arterial and 14.3% venous) in 63 patients treated with AIDA regimens, and these events occurred mostly during induction [21]. Of note, the reported incidence of thrombo-ischemic events seems different depending on the prospective or retrospective design of the studies (Fig. 5.1). As the incidence ranges between 5 and 6% in the retrospective series [17, 22], it becomes higher in the prospective series, ranging from 13 to 20% [23]. As an example, the PETHEMA database was retrospectively analyzed for thrombotic events in 759 consecutive APL patients (LPA96 and LPA99 trials) [16, 22]. An incidence rate of thrombosis of 5.1% (39/759) was observed, and four cases were associated with the use of tranexamic acid: two patients with deep vein thrombosis, one case of hemorrhagic skin necrosis, and one of renal necrosis. In multivariate analysis, hypofibrinogenemia at presentation (<170 mg/dL) and the M3 variant subtype remained from the univariate analysis as independent prognostic factors. Thrombosis was observed to relate with a higher induction mortality (including deaths prior to the initiation of chemotherapy), 28% vs. 11%, P < 0.01 [22]. In contrast, the prospective study by the PETHEMA group performed on 921 patients (LPA2005 and LPA2012 trials) showed an incidence of 4.1% at presentation, while 9.3% developed during induction therapy (overall 13.4% incidence). The type of thrombosis was catheter-related (46%), deep venous (17%), cerebral stroke (12%), pulmonary embolism (12%), acute myocardial infarction (9%), and others (4%). Regarding the risk factors for thrombosis, the study by Breccia and colleagues showed that high WBC, BCR3 isoform, FLT3-ITD, CD2, and CD15 surface antigens were related with the development of this complication [17]. However, a large prospective study by Rodriguez-Veiga et al. showed the following risk factors for the development of non-catheter-related thrombosis at diagnosis or during induction: hypoalbuminemia , absence of hemorrhage at diagnosis, higher platelet counts, male sex, and worse performance status (ECOG scale) (unpublished data).
Hemostatic Abnormalities
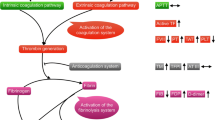
Normal hemostatic mechanisms consist of three processes strictly connected: (1) primary hemostasis, in which platelet adhere to the vessel wall lesion and, upon activation and aggregation, generate a platelet plug; (2) the coagulation cascade activation, leading to fibrin formation and platelet plug establishment, which is finely regulated by natural inhibitors (i.e., antithrombin, protein C, protein S) that prevent excessive coagulation; and (3) fibrinolysis activation, triggered by fibrin itself, which determines the degradation and dissolution of cross-linked fibrin and final restoration of vessel wall integrity. In patients with APL, laboratory coagulation abnormalities show a profound dysregulation in all of these mechanisms at the onset of the disease. Besides thrombocytopenia, routine coagulation laboratory test alterations occur, including hypofibrinogenemia , increased circulating levels of fibrinogen-fibrin degradation products (FDPs), and prolonged prothrombin and thrombin times. These abnormalities can be accentuated by the initiation of cytotoxic chemotherapy, resulting in severe hemorrhagic complications [25]. As summarized in Table 5.1, the observed alterations of routine clotting tests are not specific of any hemostatic pathway.
The results of more sensitive laboratory assays confirm the activation of all of these systems in APL. In fact, plasma levels of well-known markers of clotting activation, i.e., the prothrombin fragment F1 + 2 (F1 + 2), thrombin-antithrombin (TAT) complex, and fibrinopeptide A (FPA), are elevated in the majority of APL patients [9, 26, 27]. Additionally, plasma markers indicating ongoing hyperfibrinolysis , including high levels of FDPs and urokinase plasminogen activator (u-PA) together with low levels of plasminogen and 2-antiplasmin, are present [26, 29,29,30]. Finally, elevated plasma levels of leukocyte elastase and fibrinogen split products of elastase are detected, which demonstrate hyperactivity of non-specific proteases.
Activation of any of the three cascades (i.e., coagulation, fibrinolysis, or non-specific proteolysis) can potentially trigger the bleeding complications of APL. However, the detection of elevated levels of D-dimer, the lysis product of stabilized cross-linked fibrin, provides strong evidence for ongoing clotting activation and thrombin generation in vivo, with hyperfibrinolysis occurring secondarily to the generation of thrombin [26, 32,32,33,34]. Less evident is the occurrence of primary hyperfibrinolysis as the major event leading to the bleeding diathesis in APL [36,36,37,38]. Based on the laboratory tests currently available, it is difficult to prove the existence of primary systemic hyperfibrinolysis in APL. In fact, while reactive, or secondary, hyperfibrinolysis in response to clotting activation can be documented by the D-dimer level increment, there are no specific tests that define primary hyperfibrino(geno)lysis in vivo. The findings of profound reductions of 2-antiplasmin and plasminogen levels, sensitive to the therapeutic use of antifibrinolytic agents [35, 36], do not allow the distinction between primary and secondary hyperfibrinolysis. Menell and colleagues found that annexin II, a protein with high affinity for plasminogen (PA) and tissue-type plasminogen activator (tPA), is highly expressed by APL cells as compared to non-APL leukemic cells [37]. The expression of annexin II on circulating APL cell surface might be responsible for primary hyperfibrinolysis in vivo; however, in the same study, the assessment of systemic activation of fibrinolysis in patient plasma still relied on non-specific primary hyperfibrinolysis markers and on D-dimer levels, which rather stands for secondary hyperfibrinolysis .
In conclusion, the activation of coagulation leads to the development of a consumption coagulopathy with excess reactive activation of fibrinolysis, the mechanism of clot lysis, and dissolution. Clinical manifestations are diffuse hemorrhages, organ failure due to microthrombi, and sometimes thrombosis of large vessels. Primary hyperfibrinolysis may occur in specific districts (i.e., cerebral vessels) where annexin II is highly expressed and aggravates bleeding.
One laboratory finding that may distinguish the coagulopathy of APL from typical DIC complicating other clinical conditions (e.g., sepsis) is the maintenance of relatively normal levels of the coagulation inhibitors antithrombin (AT) . This has raised some arguments against DIC, favoring the hypothesis of primary hyperfibrinolysis as the determinant of severe bleeding in acute leukemia [39]. Of interest, however, is that reduced levels of AT in patients with APL tend to occur in patients with hepatic dysfunction, while these levels are usually normal in those with normal liver function [40]. Therefore, normal levels of AT in APL patients do not exclude DIC but may emphasize other features of this coagulopathy.
Pathogenesis of the Coagulopathy of APL
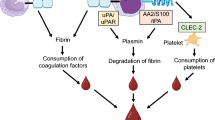
Many exogenous factors, including cytotoxic chemotherapy and concomitant infections, can impair the normal delicate balance between procoagulant and anticoagulant forces in the hemostatic system of patients with APL. However, the major determinants of coagulopathy in these patients are the intrinsic hemostatic properties of leukemic cells and their interactions with the host coagulation system, as well as the host vascular cells. These properties include (1) expression of procoagulant factors and the release of procoagulant microparticles, (2) expression of fibrinolytic proteins and proteolytic enzymes, and (3) secretion of inflammatory cytokines [i.e., interleukin-1b (IL-1β) and tumor necrosis factor (TNFα)] and expression of adhesion molecules to bind to vascular endothelium and other blood cells (Fig. 5.2).
Pathogenesis of coagulopathy in APL. APL cells interact with the host hemostatic system by (1) the expression of procoagulant, fibrinolytic, and proteolytic activities; (2) the release of inflammatory cytokines and angiogenic factors and the expression of cell membrane adhesion receptors, which induce the procoagulant and proadhesive properties of normal vascular cells and mediate APL cell-vascular cell direct interaction. All these events reflect in the peripheral blood with alterations in the levels of circulating biomarkers of hypercoagulation, hyperfibrinolysis, proteolysis, and inflammation. TAT thrombin-antithrombin complex, F1 + 2 prothrombin fragment 1 + 2, FPA fibrinopeptide A, FPB fibrinopeptide B, FDP fibrinogen-fibrin degradation product, tPA tissue-type plasminogen activator, PAI-1 plasminogen activator inhibitor 1
Increasing evidence from molecular studies of experimental models of human cancers shows that oncogene and repressor gene-mediated neoplastic transformation activate the clotting system as an integral feature of neoplastic transformation [41]. Triggering signaling pathways by one or more of these genes results in activation of the coagulation cascade and platelet function and/or suppression of fibrinolysis, which altogether can produce thrombosis and/or DIC in these models [43,43,44]. Along this line, oncogenic events may drive the expression of cellular procoagulant activities in APL cells, in which the typical PML/RARα genetic lesion is associated with the overexpression of cellular procoagulant activities and the appearance of the coagulopathy, while cellular differentiation leads to the loss of cellular procoagulant potential [1].
Procoagulant Activity
The principal cancer cell-associated clotting activating mechanisms include the expression of procoagulant factors: tissue factor (TF) and cancer procoagulant (CP). These proteins are highly expressed by APL cells.
TF is a transmembrane glycoprotein that is the primary initiator of normal blood coagulation. TF forms a complex with factor VIIa (TF/FVIIa) to trigger blood coagulation by proteolytically activating FIX and FX. Normal cells, including endothelial cells (EC) and monocyte-macrophages, do not express TF, unless they are adequately stimulated [45]. Differently, a constitutive TF expression characterizes numerous malignant tumor tissues, including APL. TF was identified in the NB4 cell line, the first human APL cell line containing the typical t(15;17) chromosomal balanced translocation. Thereafter TF has been characterized in APL cells by several laboratories [26, 27, 47,47,48,49]. The underlying molecular mechanisms are unclear; however in vitro the TF promoter is active in the PML-RARα-positive NB4 APL cell line, and it is possible that this relates to the fusion protein. Furthermore, bone marrow cells from mice transgenic for the fusion genes PLZF-RARα or NPM-RARα do express the TF gene, whereas cells derived from mice without those fusion genes do not express the TF gene [50]. A strong link is found between the regulation of TF gene expression in APL cells and the malignant transforming events. Additionally, increased levels of TF-bearing microparticles (TF-MPs) have been described in APL patients [51]. As in solid tumors, the TF-MPs found in APL exhibit procoagulant activity as shown by thrombin generation measurements [52].
CP is a cysteine protease that directly activates factor X in the absence of activated factor VII. CP is synthesized by malignant cells, and its activity has been found in extracts of different tumors [54,54,55]. CP is expressed in patient leukemic blasts of various phenotypes and is found at the highest levels in APL blasts. Accordingly, it is highly expressed in the NB4 cell line APL [56]. In APL cells, at the onset of disease, the levels of CP are found elevated compared to cells obtained from patients at the time of complete remission, confirming the association of a procoagulant protein expression with the malignant phenotype [57].
Fibrinolytic and Proteolytic Properties
Leukemic cells can express on their surface all constituents of the fibrinolytic system , which are relevant for maintenance of proper hemostasis. There are specific receptors that support the assembly of all fibrinolysis proteins on leukemic cells thus facilitating the activation of the fibrinolytic cascade [58].
APL cells are capable to interact with the host fibrinolytic system, owing to the expression of plasminogen activators (u-PA and tPA); their inhibitors, i.e., plasminogen activator inhibitor 1 and 2 (PAI-1 and PAI-2); and receptors such as u-PAR and annexin II (a co-receptor for PA and tPA) [60,60,61]. Annexin II is overexpressed on t(15;17)-positive APL cells [37, 62] and can cause a marked increase in tPA-dependent plasmin generation compared to non-APL cells in vitro [62]. However, the study of 26 patients failed to show any correlation between increased cellular annexin II expression and increased systemic fibrinolytic activity measured by FDP, plasminogen, and fibrinogen levels [62]. Increased levels of MPs expressing annexin II and tPA have been found in the circulation of patients with APL [51]. Abnormalities of the fibrinolytic system have been proposed to play a major role in the pathogenesis of the APL coagulopathy and to be responsible for the commonly observed hemorrhagic complications. Likely, the increased expression of annexin II on cerebral endothelial cells [63] may contribute to the high incidence of intracranial hemorrhage in APL [64]. Relevant in this context are also changes in other fibrinolytic proteins in APL, including increased expression of u-PA, tPA, and the urokinase-type plasminogen activator receptor (u-PAR) [65].
In addition, an increased proteolysis by non-specific proteases, such as elastase, can occur in APL. Increased plasma levels of elastase, detected as elastase-inhibitor complex, are found in these patients [66, 67]. These enzymes can interfere with coagulation by degrading clotting factors and cleaving inhibitors of fibrinolysis [67, 68]. Elastase can degrade fibrinogen, producing a pattern of FDPs different from those produced by plasmin cleavage [69, 70]. Varieties of proteases, which can be elaborated by APL cells, have been implicated in the pathogenesis of the bleeding syndrome. In an in vitro study, freshly isolated APL blasts expressed lower fibrinolytic and proteolytic activities compared to mature neutrophils. Plasma elastase levels are elevated at the onset of APL, most likely as the result of cell degranulation and lysis [31].
Cytokine Release and Adhesive Properties
Several cytokines are secreted by APL cells, including interleukin (IL)-1β and tumor necrosis factor alpha (TNFα) [71]. Both TNFα and IL-1β induce a procoagulant endothelium by upregulating the expression of TF and downregulating the expression of the anticoagulant thrombomodulin (TM) on endothelial cells (EC) [72]. In addition, these cytokines increase the production of endothelial PAI-1, the inhibitor of fibrinolysis. Upregulation of TF and PAI-1 and downregulation of TM lead the shift of the vessel wall to a prothrombotic phenotype. An increased secretion of IL-1β has been observed in leukemic promyelocytes from patients with DIC compared to patients without DIC [73].
The expression on the surface of leukemic cells of adhesion molecules and/or their counter-receptors permits the direct interaction of these cells with the host endothelial cells (EC), platelets, and leukocytes. The attachment of malignant cells to vascular EC favors the localization of clotting activation to the vessel wall with on-site release of cytokines and the activation of the endothelium. This, in turn, increases the expression of endothelial counter-receptors, i.e., ICAM-1 or VCAM-1, which bind to the leukemic cell membrane adhesion integrins, such as LFA-1 and Mac-1. Attachment of leukemic cells to the vessel wall via these adhesion mechanisms, and subsequent trans-endothelial migration, represents one potential mechanism to explain the higher incidence of vascular complications in association with high WBC count. Experimental evidence supports the concept that adhesive mechanisms of APL cells promote the localization of clotting activation to the vessel wall, WBC and platelet aggregation, thrombin generation (and hyperfibrinolysis ), and further endothelium activation. Accordingly, leukemic cells can activate platelets by cell-cell interaction, generation of thrombin, and secretion of platelet-activating molecules. Recent data show that podoplanin , a type I transmembrane sialomucin-like glycoprotein expressed by several tumor cells and capable to induce platelet aggregation [74], is aberrantly expressed by APL promyelocytes. This might contribute to abnormal platelet aggregation and possibly to APL-related bleeding [75]. Due to the leukemia-related severe thrombocytopenia, the relevance of this mechanism in APL coagulopathy remains to be clarified.
The Effects of ATRA and ATO on the Coagulopathy of APL
Differentiating therapy with ATRA and ATO for remission induction therapy exerts overall beneficial effects on the APL-associated coagulopathy. Several studies document the decrease or normalization of clotting variables, such as F1 + 2, TAT, FPA, and D-dimer, during therapy with ATRA. Additionally, ATRA inhibits fibrinolysis by inducing the synthesis of PAI-1 and by reducing the synthesis of annexin II (with consequent reduction of receptor-bound PA) [26, 32,32,33]. Furthermore, the proteolysis of von Willebrand factor (vWF) is reduced by ATRA treatment [76]. These beneficial effects of ATRA on markers of coagulation, fibrinolysis, and proteolysis activation are associated with improvement in clinical signs of bleeding in the same patients. The benefits persist when ATRA is given in combination with chemotherapy. Induction therapy with ATRA has a long-term benefit on both disease-free and overall survival in APL, while serious bleeding at the time of presentation remains a negative prognostic finding [77]. Rapid reversal of the coagulopathy with ATRA may improve survival in some of those poor prognosis patients.
In vitro studies show that ATRA interferes with APL procoagulant and fibrinolytic mechanisms. ATRA-induced APL cell differentiation in vitro causes loss of expression of procoagulant proteins like CP [78] and TF [79, 80]. This occurs also in vivo, in the bone marrow cells of APL patients given ATRA for remission induction therapy [26]. Reduction of leukemic cell procoagulant activity by ATRA appears to be one important mechanism involved in the resolution of the coagulopathy. An in vitro study demonstrated that, after ATRA treatment, CP activity is downregulated only in those NB4 cells that are sensitive to ATRA-induced cytodifferentiation, and not in ATRA-resistant cells that do not differentiate. However, TF activity was significantly reduced in all cell lines in response to ATRA , regardless of sensitivity to ATRA-induced differentiation [81]. TF expression can be downregulated by ATRA in both APL cells and in other types of leukemic cells [82] and also in normally differentiated cells [84,84,85,86]. Nuclear run-on experiments in human monocytes and monocytic leukemia cells support the concept that ATRA inhibits induction of TF expression at the level of transcription [85] but independently of the common transcription factors AP-1 or NF-kB [85]. The destabilization of TF mRNA induced by ATRA in NB4 cells is partially dependent upon protein synthesis [86], and ATRA induces synthesis of a protein in NB4 cells that selectively degrades PML/RARα fusion protein [87]. Therefore, one or more proteins induced by ATRA in leukemic cells may also destabilize TF mRNA [88]. Additionally these data provide strong support for the hypothesis that downregulation of TF gene expression is a direct result of the mechanism of the ATRA effect on oncogene expression. Recently, Fang et al. demonstrated that ATRA treatment of NB4 APL cells leads to reduced microparticle delivery of TF to endothelial cells, underlying the importance of immediate treatment with ATRA [89]. Similarly, circulating markers of clotting activation are downregulated after induction therapy with ATRA [26, 27].
Concerning fibrinolysis, ATRA inhibits the expression of annexin II by APL blasts [63]. Furthermore, retinoids induce a rapid increase of u-PA activity on APL cell surface, which is however promptly downregulated by an increased production of PA inhibitors, including PAI-1 and PAI-2 [90]. Overall, these mechanisms can contribute to a reduction of fibrinolytic activity in APL cells in response to ATRA. These results agree with the findings of normal plasma fibrinolytic activity, measured by the assay of “euglobulin lysis area ,” in APL patients receiving ATRA [26]. In conclusion, hyperfibrinolysis may reflect activation of the fibrinolytic system on the surface of the leukemic cells, where specific receptors favor the assembly of all the fibrinolytic components. ATRA acts initially to enhance this fibrinolytic activity by increasing the synthesis of u-PA. Thereafter, however, ATRA-induced synthesis of PA inhibitors and inhibition of annexin II synthesis may be favored, contributing to the downregulation of receptor-bound plasminogen activators. On balance, therefore, no change in plasma total fibrinolytic activity occurs in most patients in response to ATRA.
No relation has been observed between plasma elastase concentration and the levels of D-dimer or other hemostatic variables during treatment with ATRA. These data, together with the data of De Stefano et al. [80], cast doubt on the earlier hypothesis that elastase makes an important contribution to the bleeding disorder of patients with APL [66].
Further, ATRA upregulates the ability of leukemic cells to produce cytokines [72]. This effect should shift the balance at the endothelium to the prothrombotic side of the equation. However, ATRA also appears to protect the endothelium in vitro against the prothrombotic assault of inflammatory cytokines, because ATRA prevents both the downregulation of TM and the upregulation of TF induced by TNFα [83] and by IL-1β in the endothelial cells [84]. Therefore, although ATRA increases cytokine synthesis by APL cells, it appears to protect the endothelium against the prothrombotic stimulus of these mediators through a complex set of interactions.
Finally, ATRA increases the adhesion capacity of APL cells to the endothelium in vitro [91], although pretreatment of ECs with ATRA reverses this effect and actually results in impaired adhesion of APL cells to ECs, due to the downregulation of EC counter-receptors by ATRA. Perhaps ATRA is unable to exert this same protective effect on the specialized endothelium of the lung, thus explaining the unusual features of the differentiation syndrome, occurring in APL patients with elevated WBC count under ATRA treatment. Both early mortality and the differentiation syndrome (characterized by unexplained fever, weight gain, respiratory distress, interstitial pulmonary infiltrates, pleural and pericardial effusions, episodic hypotension, and acute renal failure) are correlated with high WBC count, the expression of adhesion molecules, and/or the release of cytokines [93,93,94,95,96]. It seems likely that a further understanding of the pathogenesis of the differentiation syndrome and its prevention, as well as a better strategy for the treatment of the consumptive coagulopathy of APL, will evolve from an improved appreciation of the biological properties of the fusion proteins of PML-RARα [97].
ATO induces apoptosis and differentiation of APL cells [98]. Current data show that ATO, as well as ATRA, can reduce TF expression and procoagulant activity of APL blast cells in vitro and in vivo. ATO treatment induces rapid loss of membrane procoagulant activity and TF mRNA leading to beneficial effects in vivo on the coagulopathy of APL patients [99, 100]. Data from our laboratory show that ATO alone reduces both TF and CP expression in NB4 cells, although to a lesser extent than ATRA. Additionally, the ATO + ATRA combination is as effective as ATRA alone in reducing the procoagulant activity, suggesting no additive effect between the two drugs (Fig. 5.3). A dual role for the ATO molecular-targeted therapy for the control of both the disease remission and the APL-associated coagulopathy can be investigated and support the importance of ATO in the cure of APL.
Modulation of procoagulant activity of NB4 cells by ATO and ATRA. NB4 cells were incubated for 96 h with ATO (0.1 μM) +/− ATRA (1 μM), while control cells received the vehicle alone (DMSO). Total procoagulant activity and FVII-independent procoagulant activity were evaluated by the clotting assays of normal human plasma (NHP) and FVII-deficient plasma (FVII-DP). Results are expressed as mean ± SD of three separate experiments performed in duplicate. * = P < 0.01 vs. controls, by Student’s t-test on paired samples
Management of the Coagulopathy
APL is a medical emergency, and aggressive measures to support the bleeding complications should begin at first suspicion of APL. Modern recommendations indicate that three simultaneous actions must be immediately undertaken when a diagnosis of APL is suspected: (1) start ATRA or ATO therapy, (2) administer supportive care, and (3) confirm genetic diagnosis [101] (Fig. 5.4).
The main strategy in the management of APL coagulopathy is early initiation of ATRA or ATO, which results in resolution of the bleeding tendency and rapid normalization of coagulation tests and fibrinogen level. It is mandatory to start as soon as possible ATRA therapy and supportive measures aimed at counteracting the coagulopathy. In high-risk patients, a delay in ATRA administration appears to contribute to bleeding and early death rate [15].
The responses to the supportive treatments for DIC in APL are the most disparate. The most important supportive tool is the judicious use of platelet transfusion, whereas the use of anticoagulants and antifibrinolytic agents remains a hotly debated issue [103,103,104]. The advent of ATRA treatment has ushered in a new era in the management of the coagulopathy of APL.
Platelet Transfusions, Heparin, and Antifibrinolytic Agents
Platelet transfusions represent an essential part of the modern supportive care for APL patients, even if there is no study that specifically addressed the threshold for platelet transfusion. Prophylactic transfusion of platelets has resulted in a significant decrease in the incidence of fatal bleeding and, therefore, a prolongation of survival. In patients with APL, the bleeding risk and platelet transfusion requirements remain high even in the ATRA era [1, 105]. Current recommendations for patients with APL suggest that platelets should be transfused to maintain the platelet count above 30 × 109/L in patients not actively bleeding and above 50 × 109/L in those with active bleeding [102, 105].
In order to maintain the fibrinogen level above 100–150 mg/dL, transfusion of frozen plasma, fibrinogen, and/or cryoprecipitate has been recommended [106, 107]. These and other supportive measures should be rapidly instituted and maintained until disappearance of all clinical and laboratory signs of coagulopathy. Routine use of heparin, tranexamic acid, or other anticoagulant or antifibrinolytic therapies is not recommended. The role of heparin therapy in the treatment of the coagulopathy complicating APL is uncertain and has never been ruled out in a prospective randomized trial. Before the ATRA era, several studies concluded that the use of heparin reduced the rate of hemorrhagic death and improved long-term survival. However, the GIMEMA group, in a large retrospective analysis of 268 APL patients, demonstrated no benefit for the prevention of early hemorrhagic deaths or overall survival [108]. Additionally, in a series of 65 adults with APL, the complete remission rate was higher in patients transfused intensively with platelets and not given heparin suggesting that the correction of thrombocytopenia with platelet transfusion is of special importance and may obviate the need for heparin therapy [109]. Although markers of coagulation activation and fibrinolysis decrease rapidly, following the start of ATRA, there appears to be a slower resolution of the clinical manifestations, suggesting the persistence of a prothrombotic state during the initial period of treatment. These evidences suggest that prophylactic use of low-molecular-weight heparin (LMWH) or even the factor Xa inhibitor fondaparinux could be considered once the bleeding manifestations have been resolved [110].
It would seem logical to consider the use of antifibrinolytic agents such as epsilon-aminocaproic acid (EACA, Amicar) or tranexamic acid and/or protease inhibitors, such as aprotinin (Trasylol) in the management of APL patients with bleeding, because of a potential role played by fibrinolytic activators and other proteases. Although several small studies concluded that the use of antifibrinolytic agents was beneficial in the management of bleeding, large studies of patients treated with ATRA have shown no reduction in early hemorrhagic deaths associated with the routine use of these agents. In addition, a PETHEMA retrospective study performed on 759 patients showed that the use of prophylactic tranexamic acid was an independent risk factor for thrombosis (odds ratio 1.96). Thus, it has been suggested that antifibrinolytic agents should be reserved for patients with retinal or intracranial and other life-threatening bleeding [7, 110].
The role of factor VIIa and prothrombin complex concentrates to treat or prevent the hemorrhagic episodes is controversial, as they may enhance the thrombotic risk. Some anecdotal use of recombinant factor VIIa in patients with APL was reported, being effective for life-threatening hemorrhages [111, 112]. The use of prothrombin complex to correct coagulopathy, instead of fresh frozen plasma, could be recommended only in patients with fluid overload or DS. In any case, the use of recombinant factor VIIa or prothrombin complex should be restricted to clinical trials.
The treatment of thrombotic episodes occurring in APL patients remains a challenge, especially because this complication mainly occur during induction phase, in a patient that presents concomitant DIC and severe thrombocytopenia. In fact, no ad hoc studies or guidelines are available for the management of such complication. However, the clinician should have in mind that the hemorrhagic risk is predominant in APL, as it is the major cause of death. When a catheter-related thrombosis occurs, the central venous line should be removed as soon as possible, and a catheter-associated infection must be ruled out. The use of unfractionated heparin could be recommended in case of cerebral stroke, in view of the high risk of hemorrhagic transformation and the possibility of rapid reversion using protamine sulfate. If a standard low-molecular-weight heparin is used, the dose could be adapted to the platelet counts (e.g., 70–80% if <70 × 109/L, 50% if <50 × 109/L, stop if <30 × 109/L).
Conclusions
The pathogenesis of the coagulopathy in patients with APL is complex and multifactorial. A prominent role is played by leukemic cell-specific properties interfering with the patient hemostatic system. In APL, bleeding manifestations prevail, although localized thrombosis of large vessels can coexist. ATRA and ATO treatments for remission induction have improved hemorrhagic accident rates and overall mortality in APL patients. However, early hemorrhagic death still remains one of the major causes of induction treatment failures. Low fibrinogen levels, prolongation of the PT and TT, and abnormal plasma levels of markers of hypercoagulation, hyperfibrinolysis, and non-specific proteolysis characterize the coagulopathy of APL. The nearly ubiquitous presence of elevated levels of fibrin D-dimer clearly demonstrates the occurrence of secondary or reactive hyperfibrinolysis in response to activation of blood coagulation and thrombin generation. Primary mechanisms of hyperfibrinolysis may take place in some specific districts, particularly in the cerebral tissues.
Reducing the bleeding-related mortality is an important task and remains a major challenge in the cure of APL. The immediate start of ATRA and the use of prophylactic platelet transfusions are highly recommended. In contrast, the routine use of anticoagulants and/or antifibrinolytic agents in the control or prevention of DIC cannot be recommended at this time.
References
Falanga A, Rickles FR. Pathogenesis and management of the bleeding diathesis in acute promyelocytic leukaemia. Best Pract Res Clin Haematol. 2003;16(3):463–82.
Falanga A. Predicting APL lethal bleeding in the ATRA era. Blood. 2017;129(13):1739–40.
Levi M. Cancer-related coagulopathies. Thromb Res. 2014;133(Suppl 2):S70–5.
Falanga A, Russo L, Tartari CJ. Pathogenesis and treatment of thrombohemorrhagic diathesis in acute promyelocytic leukemia. Mediterr J Hematol Infect Dis. 2011;3(1):e2011068.
Falanga A, Marchetti M. Venous thromboembolism in the hematologic malignancies. J Clin Oncol. 2009;27(29):4848–57.
Libourel EJ, et al. Disseminated intravascular coagulation at diagnosis is a strong predictor for both arterial and venous thrombosis in newly diagnosed acute myeloid leukemia. Blood. 2016.
Tallman MS, Abutalib SA, Altman JK. The double hazard of thrombophilia and bleeding in acute promyelocytic leukemia. Semin Thromb Hemost. 2007;33(4):330–8.
Lo-Coco F, et al. Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. N Engl J Med. 2013;369(2):111–21.
Tallman MS, Kwaan HC. Reassessing the hemostatic disorder associated with acute promyelocytic leukemia. Blood. 1992;79(3):543–53.
Fenaux P. Management of acute promyelocytic leukemia. Eur J Haematol. 1993;50(2):65–73.
Barbui T, Finazzi G, Falanga A. The impact of all-trans-retinoic acid on the coagulopathy of acute promyelocytic leukemia. Blood. 1998;91(9):3093–102.
Castaigne S, et al. All-trans retinoic acid as a differentiation therapy for acute promyelocytic leukemia. I. Clinical results. Blood. 1990;76(9):1704–9.
Lehmann S, et al. Continuing high early death rate in acute promyelocytic leukemia: a population-based report from the Swedish Adult Acute Leukemia Registry. Leukemia. 2011;25(7):1128–34.
Breccia M, Lo Coco F. Thrombo-hemorrhagic deaths in acute promyelocytic leukemia. Thromb Res. 2014;133(Suppl 2):S112–6.
Altman JK, et al. Administration of ATRA to newly diagnosed patients with acute promyelocytic leukemia is delayed contributing to early hemorrhagic death. Leuk Res. 2013;37(9):1004–9.
de la Serna J, et al. Causes and prognostic factors of remission induction failure in patients with acute promyelocytic leukemia treated with all-trans retinoic acid and idarubicin. Blood. 2008;111(7):3395–402.
Breccia M, et al. Occurrence of thrombotic events in acute promyelocytic leukemia correlates with consistent immunophenotypic and molecular features. Leukemia. 2007;21(1):79–83.
Chang H, et al. Acute promyelocytic leukemia-associated thrombosis. Acta Haematol. 2013;130(1):1–6.
Rashidi A, et al. Thrombosis in acute promyelocytic leukemia. Thromb Res. 2013;131(4):281–9.
Montesinos P, et al. Differentiation syndrome in patients with acute promyelocytic leukemia treated with all-trans retinoic acid and anthracycline chemotherapy: characteristics, outcome, and prognostic factors. Blood. 2009;113(4):775–83.
Mitrovic M, et al. Thrombotic events in acute promyelocytic leukemia. Thromb Res. 2015;135(4):588–93.
Montesinos P, de la Serna J, Vellenga E, Rayon C, Bergua J, Parody R, Esteve J, Gonzalez M, Brunet S, Sanz M. Incidence and Risk Factors for Thrombosis in Patients with Acute Promyelocytic Leukemia. Experience of the PETHEMA LPA96 and LPA99 Protocols. Blood. 2006;108:1503.
De Stefano V, et al. The risk of thrombosis in patients with acute leukemia: occurrence of thrombosis at diagnosis and during treatment. J Thromb Haemost. 2005;3(9):1985–92.
Falanga A. Mechanisms of hypercoagulation in malignancy and during chemotherapy. Haemostasis. 1998;28(Suppl 3):50–60.
Falanga A, et al. Loss of blast cell procoagulant activity and improvement of hemostatic variables in patients with acute promyelocytic leukemia administered all-trans-retinoic acid. Blood. 1995;86(3):1072–81.
Tallman MS, et al. Effects of all-trans retinoic acid or chemotherapy on the molecular regulation of systemic blood coagulation and fibrinolysis in patients with acute promyelocytic leukemia. J Thromb Haemost. 2004;2(8):1341–50.
Booth NA, Bennett B. Plasmin-alpha 2-antiplasmin complexes in bleeding disorders characterized by primary or secondary fibrinolysis. Br J Haematol. 1984;56(4):545–56.
Reddy VB, et al. Global and molecular hemostatic markers in acute myeloid leukemia. Am J Clin Pathol. 1990;94(4):397–403.
Speiser W, et al. Hemostatic and fibrinolytic parameters in patients with acute myeloid leukemia: activation of blood coagulation, fibrinolysis and unspecific proteolysis. Blut. 1990;61(5):298–302.
Dombret H, et al. Coagulation disorders associated with acute promyelocytic leukemia: corrective effect of all-trans retinoic acid treatment. Leukemia. 1993;7(1):2–9.
Dombret H, et al. In vivo thrombin and plasmin activities in patients with acute promyelocytic leukemia (APL): effect of all-trans retinoic acid (ATRA) therapy. Leukemia. 1995;9(1):19–24.
Kawai Y, et al. Rapid improvement of coagulopathy by all-trans retinoic acid in acute promyelocytic leukemia. Am J Hematol. 1994;46(3):184–8.
Watanabe R, et al. Long-term follow-up of hemostatic molecular markers during remission induction therapy with all-trans retinoic acid for acute promyelocytic leukemia. Keio Hematology-Oncology Cooperative Study Group (KHOCS). Thromb Haemost. 1997;77(4):641–5.
Avvisati G, et al. Acquired alpha-2-antiplasmin deficiency in acute promyelocytic leukaemia. Br J Haematol. 1988;70(1):43–8.
Schwartz BS, et al. Epsilon-aminocaproic acid in the treatment of patients with acute promyelocytic leukemia and acquired alpha-2-plasmin inhibitor deficiency. Ann Intern Med. 1986;105(6):873–7.
Menell JS, et al. Annexin II and bleeding in acute promyelocytic leukemia. N Engl J Med. 1999;340(13):994–1004.
Kwaan HC. The unique hemostatic dysfunction in acute promyelocytic leukemia. Semin Thromb Hemost. 2014;40(3):332–6.
Avvisati G. Coagulopathy in APL: a step forward? Blood. 2012;120(1):4–6.
Rodeghiero F, et al. Liver dysfunction rather than intravascular coagulation as the main cause of low protein C and antithrombin III in acute leukemia. Blood. 1984;63(4):965–9.
Falanga A, Barbui T, Rickles FR. Hypercoagulability and tissue factor gene upregulation in hematologic malignancies. Semin Thromb Hemost. 2008;34(2):204–10.
Boccaccio C, et al. The MET oncogene drives a genetic programme linking cancer to haemostasis. Nature. 2005;434(7031):396–400.
Rong Y, et al. PTEN and hypoxia regulate tissue factor expression and plasma coagulation by glioblastoma. Cancer Res. 2005;65(4):1406–13.
Yu JL, et al. Oncogenic events regulate tissue factor expression in colorectal cancer cells: implications for tumor progression and angiogenesis. Blood. 2005;105(4):1734–41.
Furie B, Furie BC. Mechanisms of thrombus formation. N Engl J Med. 2008;359(9):938–49.
Andoh K, et al. Tissue factor activity in leukemia cells. Special reference to disseminated intravascular coagulation. Cancer. 1987;59(4):748–54.
Gouault Heilmann M, et al. The procoagulant factor of leukaemic promyelocytes: demonstration of immunologic cross reactivity with human brain tissue factor. Br J Haematol. 1975;30(2):151–8.
Bauer KA, et al. Tissue factor gene expression in acute myeloblastic leukemia. Thromb Res. 1989;56(3):425–30.
Hair GA, et al. Tissue factor expression in human leukemic cells. Leuk Res. 1996;20(1):1–11.
Cheng GX, et al. Distinct leukemia phenotypes in transgenic mice and different corepressor interactions generated by promyelocytic leukemia variant fusion genes PLZF-RARalpha and NPM-RARalpha. Proc Natl Acad Sci U S A. 1999;96(11):6318–23.
Kwaan HC, Rego EM. Role of microparticles in the hemostatic dysfunction in acute promyelocytic leukemia. Semin Thromb Hemost. 2010;36(8):917–24.
Ma G, et al. Increased promyelocytic-derived microparticles: a novel potential factor for coagulopathy in acute promyelocytic leukemia. Ann Hematol. 2013;92(5):645–52.
Falanga A, Gordon SG. Isolation and characterization of cancer procoagulant: a cysteine proteinase from malignant tissue. Biochemistry. 1985;24(20):5558–67.
Donati MB, et al. Cancer procoagulant in human tumor cells: evidence from melanoma patients. Cancer Res. 1986;46(12 Pt 1):6471–4.
Mielicki W, et al. Procoagulant activity of human stomach and colon cancers. Oncology. 1990;47(4):299–302.
Falanga A, et al. A new procoagulant in acute leukemia. Blood. 1988;71(4):870–5.
Donati MB, et al. Cancer procoagulant in acute non lymphoid leukemia: relationship of enzyme detection to disease activity. Thromb Haemost. 1990;64(1):11–6.
Hajjar KA. Cellular receptors in the regulation of plasmin generation. Thromb Haemost. 1995;74(1):294–301.
Bennett B, et al. The bleeding disorder in acute promyelocytic leukaemia: fibrinolysis due to u-PA rather than defibrination. Br J Haematol. 1989;71(4):511–7.
Francis RB Jr, Seyfert U. Tissue plasminogen activator antigen and activity in disseminated intravascular coagulation: clinicopathologic correlations. J Lab Clin Med. 1987;110(5):541–7.
Stephens R, et al. Production of an active urokinase by leukemia cells: a novel distinction from cell lines of solid tumors. Leuk Res. 1988;12(5):419–22.
Liu Y, et al. The expression of annexin II and its role in the fibrinolytic activity in acute promyelocytic leukemia. Leuk Res. 2011;35(7):879–84.
Kwaan HC, Wang J, Weiss I. Expression of receptors for plasminogen activators on endothelial cell surface depends on their origin. J Thromb Haemost. 2004;2(2):306–12.
Stein E, et al. The coagulopathy of acute promyelocytic leukaemia revisited. Best Pract Res Clin Haematol. 2009;22(1):153–63.
Graf M, et al. High expression of urokinase plasminogen activator receptor (UPA-R) in acute myeloid leukemia (AML) is associated with worse prognosis. Am J Hematol. 2005;79(1):26–35.
Egbring R, et al. Demonstration of granulocytic proteases in plasma of patients with acute leukemia and septicemia with coagulation defects. Blood. 1977;49(2):219–31.
Nevo S, et al. Acute bleeding after bone marrow transplantation (BMT)- incidence and effect on survival. A quantitative analysis in 1,402 patients. Blood. 1998;91(4):1469–77.
Brower MS, Harpel PC. Proteolytic cleavage and inactivation of alpha 2-plasmin inhibitor and C1 inactivator by human polymorphonuclear leukocyte elastase. J Biol Chem. 1982;257(16):9849–54.
Sterrenberg L, et al. Anticoagulant properties of purified X-like fragments of human fibrinogen produced by degradation with leukocyte elastase. Thromb Haemost. 1984;51(3):398–402.
Sterrenberg L, Nieuwenhuizen W, Hermans J. Purification and partial characterization of a D-like fragment from human fibrinogen, produced by human leukocyte elastase. Biochim Biophys Acta. 1983;755(2):300–6.
Griffin JD, et al. Secretion of interleukin-1 by acute myeloblastic leukemia cells in vitro induces endothelial cells to secrete colony stimulating factors. Blood. 1987;70(4):1218–21.
Khan MM, et al. Upregulation of tissue factor in monocytes by cleaved high molecular weight kininogen is dependent on TNF-alpha and IL-1beta. Am J Physiol Heart Circ Physiol. 2010;298(2):H652–8.
Cozzolino F, et al. Potential role of interleukin-1 as the trigger for diffuse intravascular coagulation in acute nonlymphoblastic leukemia. Am J Med. 1988;84(2):240–50.
Raica M, Cimpean AM, Ribatti D. The role of podoplanin in tumor progression and metastasis. Anticancer Res. 2008;28(5B):2997–3006.
Lavallée VP, Marquis M, Bordeleau ME, Chagraoui J, MacRae T, Boivin I, Boucher G, Gendron P, Lemieux S, Bonnefoy A, Rivard GE, Hébert J, Sauvageau G. Transcriptional landscape of APL identifies aberrant podoplanin expression as a defining feature and missing link for the bleeding disorder of this disease. Blood. 2016;128:1075.
Federici AB, et al. Proteolysis of von Willebrand factor is decreased in acute promyelocytic leukaemia by treatment with all-trans-retinoic acid. Br J Haematol. 1996;92(3):733–9.
Tallman MS, et al. All-trans retinoic acid in acute promyelocytic leukemia: long-term outcome and prognostic factor analysis from the North American Intergroup protocol. Blood. 2002;100(13):4298–302.
Falanga A, et al. Cancer procoagulant in the human promyelocytic cell line NB4 and its modulation by all-trans-retinoic acid. Leukemia. 1994;8(1):156–9.
Koyama T, et al. All-trans retinoic acid upregulates thrombomodulin and downregulates tissue-factor expression in acute promyelocytic leukemia cells: distinct expression of thrombomodulin and tissue factor in human leukemic cells. Blood. 1994;84(9):3001–9.
De Stefano V, et al. Effect of all-trans retinoic acid on procoagulant and fibrinolytic activities of cultured blast cells from patients with acute promyelocytic leukemia. Blood. 1995;86(9):3535–41.
Falanga A, et al. Cancer procoagulant and tissue factor are differently modulated by all-trans-retinoic acid in acute promyelocytic leukemia cells. Blood. 1998;92(1):143–51.
Saito T, et al. Anticoagulant effects of retinoic acids on leukemia cells. Blood. 1996;87(2):657–65.
Ishii H, et al. Retinoic acid counteracts both the downregulation of thrombomodulin and the induction of tissue factor in cultured human endothelial cells exposed to tumor necrosis factor. Blood. 1992;80(10):2556–62.
Falanga A, et al. All-trans-retinoic acid counteracts endothelial cell procoagulant activity induced by a human promyelocytic leukemia-derived cell line (NB4). Blood. 1996;87(2):613–7.
Oeth P, et al. Retinoic acid selectively inhibits lipopolysaccharide induction of tissue factor gene expression in human monocytes. Blood. 1998;91(8):2857–65.
Zhu J, et al. Tissue factors on acute promyelocytic leukemia and endothelial cells are differently regulated by retinoic acid, arsenic trioxide and chemotherapeutic agents. Leukemia. 1999;13(7):1062–70.
Raelson JV, et al. The PML/RAR alpha oncoprotein is a direct molecular target of retinoic acid in acute promyelocytic leukemia cells. Blood. 1996;88(8):2826–32.
Brand K, et al. Tissue factor mRNA in THP-1 monocytic cells is regulated at both transcriptional and posttranscriptional levels in response to lipopolysaccharide. Mol Cell Biol. 1991;11(9):4732–8.
Fang Y, et al. PML-RARa modulates the vascular signature of extracellular vesicles released by acute promyelocytic leukemia cells. Angiogenesis. 2016;19(1):25–38.
Tapiovaara H, et al. Induction of differentiation of promyelocytic NB4 cells by retinoic acid is associated with rapid increase in urokinase activity subsequently downregulated by production of inhibitors. Blood. 1994;83(7):1883–91.
Marchetti M, et al. All-trans-retinoic acid increases adhesion to endothelium of the human promyelocytic leukaemia cell line NB4. Br J Haematol. 1996;93(2):360–6.
Vahdat L, et al. Early mortality and the retinoic acid syndrome in acute promyelocytic leukemia: impact of leukocytosis, low-dose chemotherapy, PMN/RAR-alpha isoform, and CD13 expression in patients treated with all-trans retinoic acid. Blood. 1994;84(11):3843–9.
Di Noto R, et al. All-trans retinoic acid promotes a differential regulation of adhesion molecules on acute myeloid leukaemia blast cells. Br J Haematol. 1994;88(2):247–55.
Larson RS, Brown DC, Sklar LA. Retinoic acid induces aggregation of the acute promyelocytic leukemia cell line NB-4 by utilization of LFA-1 and ICAM-2. Blood. 1997;90(7):2747–56.
Dubois C, et al. Modulation of IL-8, IL-1 beta, and G-CSF secretion by all-trans retinoic acid in acute promyelocytic leukemia. Leukemia. 1994;8(10):1750–7.
Marchetti M, et al. All-trans retinoic acid modulates microvascular endothelial cell hemostatic properties. Haematologica. 2003;88(8):895–905.
Melnick A, Licht JD. Deconstructing a disease: RARalpha, its fusion partners, and their roles in the pathogenesis of acute promyelocytic leukemia. Blood. 1999;93(10):3167–215.
Emadi A, Gore SD. Arsenic trioxide—an old drug rediscovered. Blood Rev. 2010;24(4–5):191–9.
Zhou J, et al. Phosphatidylserine exposure and procoagulant activity in acute promyelocytic leukemia. J Thromb Haemost. 2010;8(4):773–82.
Zhang X, et al. [The impact of arsenic trioxide or all-trans retinoic acid treatment on coagulopathy in acute promyelocytic leukemia]. Zhonghua Nei Ke Za Zhi. 2001;40(12):829–33.
Watts JM, Tallman MS. Acute promyelocytic leukemia: what is the new standard of care? Blood Rev. 2014;28(5):205–12.
Squizzato A, et al. Supportive management strategies for disseminated intravascular coagulation. An international consensus. Thromb Haemost. 2016;115(5):896–904.
Sanz MA, Lo-Coco F. Modern approaches to treating acute promyelocytic leukemia. J Clin Oncol. 2011;29(5):495–503.
Sanz MA, et al. Risk-adapted treatment of acute promyelocytic leukemia with all-trans retinoic acid and anthracycline monochemotherapy: long-term outcome of the LPA 99 multicenter study by the PETHEMA Group. Blood. 2008;112(8):3130–4.
Rickles FR, et al. Bleeding and thrombosis in acute leukemia: what does the future of therapy look like? Thromb Res. 2007;120(Suppl 2):S99–106.
Sanz MA, Montesinos P. Open issues on bleeding and thrombosis in acute promyelocytic leukemia. Thromb Res. 2010;125(Suppl 2):S51–4.
Sanz MA, et al. Management of acute promyelocytic leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood. 2009;113(9):1875–91.
Rodeghiero F, et al. Early deaths and anti-hemorrhagic treatments in acute promyelocytic leukemia. A GIMEMA retrospective study in 268 consecutive patients. Blood. 1990;75(11):2112–7.
Bassan R, et al. Short-term treatment for adult hypergranular and microgranular acute promyelocytic leukemia. Leukemia. 1995;9(2):238–43.
Arbuthnot C, Wilde JT. Haemostatic problems in acute promyelocytic leukaemia. Blood Rev. 2006;20(6):289–97.
Zver S, Andoljsek D, Cernelc P. Effective treatment of life-threatening bleeding with recombinant activated factor VII in a patient with acute promyelocytic leukaemia. Eur J Haematol. 2004;72(6):455–6.
Alimoghaddam K, Ghavamzadeh A, Jahani M. Use of Novoseven for arsenic trioxide-induced bleeding in PML. Am J Hematol. 2006;81(9):720.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Falanga, A., Russo, L., Montesinos, P. (2018). APL Coagulopathy. In: Abla, O., Lo Coco, F., Sanz, M. (eds) Acute Promyelocytic Leukemia . Springer, Cham. https://doi.org/10.1007/978-3-319-64257-4_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-64257-4_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-64256-7
Online ISBN: 978-3-319-64257-4
eBook Packages: MedicineMedicine (R0)