Abstract
For this study, a methodology was developed for assessing impacts of wind energy generation on populations of birds and bats at regional to national scales. The approach combines existing methods in applied ecology for prioritizing species in terms of their potential risk from wind energy facilities and estimating impacts of fatalities on population status and trend caused by collisions with wind energy infrastructure. Methods include a qualitative prioritization approach, demographic models, and potential biological removal. The approach can be used to prioritize species in need of more thorough study as well as to identify species with minimal risk. However, the components of this methodology require simplifying assumptions and the data required may be unavailable or of poor quality for some species. These issues should be carefully considered before using the methodology. The approach will increase in value as more data become available and will broaden the understanding of anthropogenic sources of mortality on bird and bat populations.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
While many studies and multiple reviews exist regarding wind energy-wildlife interactions, we know little about the population-level consequences of direct and indirect effects of wind energy generation on species (Schuster et al. 2015). To date, only a few studies address the issue of broad-scale, population-level impacts from wind turbines. For example, two studies used modeling to examine how fatalities from collision with turbines will impact populations of Egyptian vultures (Neophron percnopterus) across Spain (Carrete et al. 2009) and Red Kites (Milvus milvus) across Germany (Schaub 2012). Bellebaum et al. (2013) compared an estimate of annual Red Kite fatalities from collisions with turbines to the species’ estimated potential biological removal (described below) to assess population-level risk across Germany.
This chapter describes a methodology designed to address questions about the direct and indirect impacts of wind energy generation on populations of birds and bats. It focuses on regional- to national-scale population impacts, not impacts to local populations interacting with a single facility. As such, the methodology evaluates the cumulative impacts of many turbines within a species’ range on its overall population status or trend.
The U.S. Geological Survey (USGS) began developing this population-level methodology in 2013. During a series of meetings with organizations ranging from industry to federal agencies and conservation groups, three main ideas emerged. First, stakeholders generally agreed that a rapid method for prioritizing species was needed to help identify species with different levels of potential risk from wind energy development. Second, groups consistently echoed the need for understanding the population-level effects of wind energy facilities. Third, stakeholders expressed an interest in a methodology that could be used to forecast the impacts of future wind energy facilities on species.
Over the two years that followed, the USGS developed a methodology designed to meet these three goals. This chapter summarizes the approach, discusses how these results might be used, and points out areas of future research that would improve the methodology and help refine the understanding of wind energy impacts on species. The methodology is considered to be one of potentially many ways to estimate population-level impacts of wind energy on wildlife at regional to national spatial scales. It is recognized that the approach has shortcomings and may be limited by data availability; however, it should help focus more research on population-level effects of wind, and other forms of energy development, on wildlife.
A Summary of the Methodology
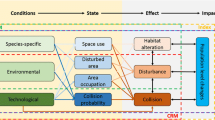
The methodology has four steps: (1) data collection, (2) a qualitative species prioritization approach, (3) demographic modeling, and (4) the potential biological removal (PBR) approach developed to manage incidental by catch of marine mammals in fisheries (Fig. 1). These steps were based on tools and techniques previously developed and commonly used in other arenas of applied ecology. After data are collected (Step 1), species are prioritized based on four risk metrics: estimates of mortality at wind turbines, impact from habitat loss, life history characteristics, and conservation status (Step 2). Those species receiving high-priority ranks are then run through two demographic components (Steps 3 and 4). The first estimates a change in the population growth rate (lambda, λ) given an estimate of the mortality rate from collisions with wind energy turbines. The second calculates the PBR and compares this, via a ratio, to the estimated number of animals killed each year by turbines.
Potential Biological Removal is defined as the maximum number of animals, not including natural mortalities that may be removed from a population while allowing that population to reach or maintain its optimum sustainable population (Wade 1998). The demographic components are then repeated using forecasts of future wind energy development to estimate expected changes under alternative future wind energy development forecasts.
Overall the methodology produces (1) a list of species ranked by their average priority score (Step 2) as well as the influence of each risk metric on the rank; and for those species with a high priority, (2) an estimate of the predicted change in probability that the population growth rate, given energy development induced mortality, will be less than 1 (Step 3); and (3) a risk ratio of PBR relative to the number of animals killed each year for both current and future levels of installed wind energy (Step 4).
The methodology was originally designed for species present in the United States during any part of their life cycle. However, the approach could be applied to species with sufficient data in any region of the world. The goal of this chapter is to present an overview of the approach so that other scientists will consider using or modifying it to study the population-level consequences of wind energy, or perhaps other anthropogenic sources of mortality on species. A detailed report of the approach, including the data sources used to parameterize the methodology and the way these are used in the United States has been published (Diffendorfer et al. 2015). This chapter provides a broad overview of the approach by describing each step and its key parameters, but does not include details on sources of data and parameterization methods. These details will vary by country, region, or taxon considered and depend on the types of data available. Data collection (Step 1) is not discussed, as the nuances of how data are generated to estimate parameters in the methodology vary by species and region. As such, our description begins with Step 2.
Step 2. Species Prioritization
Species are prioritized by combining information from four sources: conservation status and three “turbine risk” metrics (defined below). For most species, current conservation status is a consequence of existing population status and trend. At-risk species are generally less capable of handling additional negative impacts including those caused by power generation. Turbine risk is assessed as the direct and indirect risk to a species caused by wind energy facilities.
Three turbine risk metrics were designed to estimate different ways wind turbines could potentially impact a species. The metrics follow a risk framework that generally includes (1) the proportion of the population potentially impacted (exposure) and (2) a measure of the way a species might respond to the hazard. This exposure-response approach has been used in other ecological prioritization schemes (Andow and Hilbeck 2004; Parker et al. 1999; Regan et al. 2008).
Step 2 generates average risk scores, for each species by combining information from conservation status and turbine risk using a Monte Carlo simulation. The average risk scores can range from 1 to 9, with 9 being the highest risk. For each species, the approach simulates 10,000 randomly generated cutoff values for defining high, medium, or low risk scores for each metric, combines these into a single score, then estimates an average across all simulations (Beston et al. 2016; Diffendorfer et al. 2015). This approach was designed to avoid using arbitrary cut-off values for risk scores. The three turbine risk metrics are described below.
Proportion of Fatalities Due to Turbines (FT).
where n is the number of individuals killed by turbines annually, N is the total population size, and 1 − s is the adult mortality rate because s is adult survival.
The denominator, (1 − s)N, represents the predicted number of adult fatalities each year for a species and survival, s should be calculated in the absence of fatalities from wind energy. A key assumption of FT is that species with long lifespans and low adult mortality will be more likely to experience additive mortality from human-caused factors than will species with short lifespans and high adult mortality (Péron et al. 2013; Wilson et al. 2010). Thus, species with a high proportion of their fatalities caused by turbines are more likely to be at higher risk.
Fatality Risk Index (FRI)
where p is the proportion of the population exposed to turbines, m is maternity (the number of female offspring per female per year) and a is the age at first reproduction.
This metric was based on a similar approach developed by Desholm (2009) for use at individual wind farms. The ratio of maternity to age at first reproduction is a measure of a species’ life history speed and is associated with the elasticity of the population growth rate. When m:a is high, elasticity in population growth is driven primarily by changes in reproduction, but when m:a is low, survival drives change in population growth (Stahl and Oli 2006). Because wind energy impacts survival, risk should vary across species depending, in part, on their life history speed. Species with either a higher exposure to turbines, or a lower life history speed, are likely to be at more risk.
Indirect Risk Index (IRI)
where p is, again, the proportion of the population exposed and h is the number of habitats a species uses.
This metric was developed to assess risk caused by the indirect effects of habitat loss and fragmentation from wind facilities. Studies indicate that across many taxa, species with lower niche breadth (i.e., habitat specialists) are more sensitive to habitat loss and fragmentation than habitat generalists (Carrascal et al. 2013; Swihart et al. 2003, 2006; Watling and Donnelly 2007). The number of habitats, h, is an index of niche breadth. For IRI, risk is assumed to be greater when either a higher proportion of the population is exposed to turbines or the species uses fewer habitats.
Step 3. Estimating Change in a Species Population Growth Rate
For species with high-risk prioritization scores, the next step estimates the probability of change in the population growth rate. To do this, an estimate of turbine mortality rate (c, the annual chance an individual will die from a collision, or barotrauma, with a wind turbine), is combined with either an empirical or modeled estimate of population growth rate in the absence of wind energy facilities. The approach assumes density-independent population growth and that all stage/age classes and sexes are equally likely to die from wind turbines. With these assumptions, simplified population dynamics can be described as:
where N t is the population size at year t, λ is the population growth rate, c is the annual chance an individual will die from a collision with a turbine, and cλN t is the number of individuals killed each year by turbines. A λ < 1 indicates a declining population.
This can be rewritten as N t+1 = (1 − c)λNt, and N t+1/N t = (1 − c)λ. If N t+1/N t is the population growth with wind, λw, then, λw = (1 − c)λb where λb is the population growth rate in the absence of wind. Thus, the change in population growth rate caused by wind energy is simply λw = λb − cλb and λb − λw = cλb.
To estimate the predicted change in population growth rate due to fatalities from wind turbines, this approach requires an estimate of the turbine mortality rate and an estimate of population growth rate in the absence of wind energy. Turbine mortality rate, c, can be estimated as the number of animals killed by turbines/total population size, while λb could be directly observed from monitoring data or estimated as the dominant eigenvalue from a matrix population model (Diffendorfer et al. 2015). The sources of monitoring data could be, for example, the Breeding Bird Survey in the United States and southern Canada. This approach generates a point estimate of change in population growth rate and the uncertainty around the point estimate.
Because uncertainty exists around c and λ (actually, estimates of these values, c-hat, and λ-hat), this step generates a distribution—rather than a single value—of the predicted change in population growth rate due to wind energy fatalities. For each distribution of population growth rates, one with fatalities from wind turbines, and one without, the probability of λ < 1 can be estimated. The difference between the two probabilities represents the estimated change in the probability that the population growth rate is less than 1. So, change in Prob(λ < 1) = Prob(λw < 1) − Prob(λb < 1).
Step 4. Potential Biological Removal and the Risk Ratio
Potential Biological Removal was developed to manage human-caused deaths to marine mammals (Taylor et al. 2000). It is one of several ‘reference point’ approaches used to assess, and set limits on, anthropogenic sources of mortality to species (Moore et al. 2013). PBR estimates the number of individuals that could be killed before a population will fall below a size considered sustainable, typically half a population’s carrying capacity (Wade 1998). The approach has been extended to issues of harvest in birds (Johnson et al. 2012; Runge et al. 2009) and applied to at least one species in relation to wind energy (Bellebaum et al. 2013).
The PBR is calculated as:
where F is a ‘recovery factor’ set by regulatory agencies or existing law.
‘F’ adjusts the value of PBR to change the rate of recovery depending on a species’ population size or status, or to account for uncertainties in the input parameter estimates and assure the estimated value of PBR is not too high. The ‘rmax’ is the maximum annual population growth rate under ideal conditions. There are a variety of approaches to estimate r max (Slade et al. 1998; Millar and Meyer 2000; Niel and Lebreton 2005; Dillingham et al. 2016). Finally, Nmin is a lower bound on the total population size. Under the Marine Mammal Protection Act in the U.S., Nmin is set at the 20th percentile of observed abundance assuming a log-normal distribution. The 20th percentile level was based on results from a computer simulation (Wade 1998), whereas F is set by regulatory agencies at 0.1, 0.5, or 1 depending on population status.
The methodology presented here uses the PBR approach to calculate a risk ratio (RR), which is the number of annual fatalities due to wind energy divided by the PBR (Richard et al. 2011). This represents the proportion of the PBR caused by collisions with wind turbines. Risk from wind energy is low when RR is near zero. At RR equal to 1, fatalities from wind turbines are equal to the PBR. At RR greater than 1, the population is expected to decline below the level considered sustainable due to fatalities from wind energy.
Final Output, Use of the Methodology, and Areas for Future Research
Ultimately, the methodology produces (1) a list of species ranked according to their average risk score calculated in the prioritization step. In addition, for those species further evaluated in Steps 3 and 4, the methodology produces (2) an estimate of the predicted change in probability the population will decline in the face of mortality from wind energy (i.e. the population growth rate will be less than 1), and (3) an estimate of the risk ratio based on PBR. It is also possible to use Steps 2 and 3 to forecast the effects of future growth in wind energy generation on species (Diffendorfer et al. 2015). To do so, the values of variables c (used in Step 3, the demographic model), and the number of animals killed annually (used in Step 4, PBR) must be updated based on scenarios of future wind facility development. Table 1 shows an example of output for a case study of six species.
To model future growth in wind energy, the average and 95% upper limit of 11 projections of installed wind energy capacity were used as the medium and high growth scenarios (Diffendorfer et al. 2016). Installed capacity in gigawatts (GW) is current (2014) scenario, 62.3 GW; medium scenario for 2025, 94 GW; high scenario for 2025, 121 GW. Risk-ratio values are presented as an average, and values in parentheses represent the 95% confidence interval. NA is not applicable; PBR is potential biological removal; and λ is the population growth rate.
Three of the species (bird 4, 5 and 6) had relatively low direct risk during the prioritization process and were not further analyzed. Of the remaining three species, bird 1 showed low risk, with predicted changes in λ < 1 smaller than 0.5% and a risk ratio near zero. Bird 2, however, had a much larger predicted change in population growth and the confidence interval around the risk ratio included 1 in all three scenarios, with the mean risk ratio greater than 1 in the high scenario. Thus, for Bird 2, PBR may be currently exceeded by mortality caused by collisions and is likely to be exceeded if future levels of wind energy development follow our scenarios. Bird 3’s results were intermediate in comparison to the bird 1 and bird 2. Small changes in population growth for bird 3 were predicted while mean risk ratios were <1 for all scenarios, and the 95% Confidence intervals overlapped 1 for medium and high scenarios.
What sorts of decision making can the methodology support? We first distinguish between absolute and relative risk. Absolute risk is the actual risk to population status and trend from the impacts of wind energy generation on a species. Relative risk is a comparison of risk level between species. The prioritization component of the methodology is a first attempt at estimating relative risk. Species with higher average risk scores are expected to be at more relative risk than species with lower average risk scores. Furthermore, the approach produces additional information about the possible cause of the risk by identifying which turbine risk metric has the most influence on the risk ranking. For example, a species might rank highly because the Indirect Risk Index is high but the remaining metrics that include collision fatalities are low. In this case, the species may be a habitat specialist whose range overlaps turbines, but is rarely recorded as colliding with turbine blades. If studies show this species is not displaced from areas with turbines, or that breeding success is not affected by facility infrastructure, then impacts to the species may be limited to direct habitat loss caused by the turbine. On the other hand, if FT (proportion of fatalities from turbines) is high and heavily influences a species risk rank, then additional research on population trends and demography may be required.
The approach may also perform well at identifying species at low risk. Species with low average risk scores, very small or negligible estimated changes in population growth, and small risk ratios are less likely to be impacted by fatalities from turbines. In these cases, observed mortality will likely be small relative to population size and productivity.
More species must be included and additional analyses performed to understand how finely the prioritization approach can separate species. For example, given uncertainties in the input data, the methodology may only be able to distinguish five distinct levels of risk, despite ranking hundreds of bird species. Higher uncertainty associated with the input data will likely result in higher uncertainty around the average risk scores, more overlap between species in the distributions of average risk score, and thus lower resolution of relative risk across species. Ultimately, additional research, and simulation of the prioritization approach, is required to address this issue.
Both the change in population growth rate and the risk ratio will produce accurate estimates of absolute risk for a species, but only if the input data are accurate and if the assumptions of the approaches are met. It is suspected that the data quality for at least some parameters might be low for many bird and bat species. This data limitation, coupled with the generalized structure of the population growth and risk ratio approaches, likely means Steps 3 and 4 will produce only approximate estimates of absolute risk. In these cases, it would be prudent to consider the results to be working hypotheses about the impacts of wind energy facilities on a species that require additional research. Such research could include more realistic models that better match a species’ life history and behavior in relation to wind turbines, and field studies to better estimate model parameters. Thus, in cases with high uncertainty, the approach may be best used to identify species potentially at risk and to highlight them for additional work.
One approach to validate the population growth rate and risk ratio components is to compare them to alternative approaches for estimating population-level impacts. For example, relatively complex models have been produced for Red Kites (Schaub 2012) and Egyptian vultures (Carrete et al. 2009), linking population growth to fatalities from wind turbines. These species could be evaluated using the methodology described here, and the results from the more simplistic models could be compared to the more complex models previously used.
The assessment methodology is considered a work in progress. The approach has been purposely based on existing methods successfully used in other applications. However, for many species, the methodology is limited in use by missing or poor-quality information on species-specific turbine collision mortality, population size, and demographic rates. We also know little about the distribution, local movements, or long-distance migratory patterns of many species killed by wind turbines, especially bats. This hampers our ability to estimate the proportion of the population exposed to wind turbines.
We currently recommend using this approach to identify species at high and low risk of experiencing population-level impacts of wind energy, and then performing more detailed investigation of the species identified as high risk. However, this recommendation should be investigated with simulations that explore how robust the outputs of the methodology are to parameter uncertainty. In some cases, such as the steps to calculate population growth rate and PBR, much of this uncertainty can be transparently carried forward and expressed as uncertainty in the modelled outputs. Development, use, and refinement of methods such as these will be crucial in efforts to understand and mitigate the risks posed to vertebrate populations by the development of wind and other energy sources.
References
Andow DA, Hilbeck A (2004) Science-based risk assessment for nontarget effects of transgenic crops. Bioscience 54:637–649
Bellebaum J, Korner-Nievergelt F, Dürr T, Mammen U (2013) Wind turbine fatalities approach a level of concern in a raptor population. J Nat Conserv 21:394–400
Beston JA, Diffendorfer JE, Loss S, Johnson DH (2016) Prioritizing avian species for their risk of population-level consequences from wind energy development. PLoS ONE 11:Article e0150813
Carrascal LM, Galván I, Sánchez-Oliver JS, Benayas JMR (2013) Regional distribution patterns predict bird occurrence in Mediterranean cropland afforestations. Ecol Res 29:203–211
Carrete M, Sánchez-Zapata JA, Benítez JR, Lobón M, Donázar JA (2009) Large scale risk-assessment of wind-farms on population viability of a globally endangered long-lived raptor. Biol Conserv 142:2954–2961
Desholm M (2009) Avian sensitivity to mortality; prioritizing migratory bird species for assessment at proposed wind farms. J Environ Manage 90:2672–2679
Diffendorfer JE, Beston JA, Merrill MD, Stanton JC, Corum MD, Loss SR, Thogmartin WE, Johnson DH, Erickson RA, Heist KW (2015) Preliminary methodology to assess the national and regional impact of U.S. wind energy development on birds and bats: U.S. Geological Survey Scientific Investigations Report 2015–5066, p 40. http://dx.doi.org/10.3133/sir20155066
Dillingham DW, Moore JE, Fletcher D, Cortés E, Curtis KA, James KC, and Lewison, RL (2016) Improved estimation of intrinsic growth rmax for long-lived species: integrating matrix models and allometry. Ecol Appl 26:322–333. http://dx.doi.org/10.1890/14-1990
Johnson FA, Walters MAH, Boomer GS (2012) Allowable levels of take for the trade in Nearctic songbirds. Ecol Appl 22:1114–1130
Millar RB, Meyer R (2000) Non-linear state space modelling of fisheries biomass dynamics by using Metropolis-Hastings within-Gibbs sampling. J Roy Stat Soc: Ser C (Appl Stat) 49:327–342
Moore JE, Curtis KA, Lewison RL, Dillingham PW, Cope JM, Fordham SV, Heppell SS, Pardo SA, Simpfendorfer CA, Tuck GN, Zhou S (2013) Evaluating sustainability of fisheries by catch mortality for marine megafauna; A review of conservation reference points for data-limited populations. Environ Conserv 40:329–344
Niel C, Lebreton JD (2005) Using demographic invariants to detect overhear vested bird populations from incomplete data. Conserv Biol 19:826–835
Parker IM, Simberloff D, Lonsdale WM, Goodell K, Wonham M, Kareiva PM, Williamson MH, Von Holle B, Moyle PB, Byers JE, Goldwasser L (1999) Impact: toward a framework for understanding the ecological effects of invaders. Biol Invasions 1:3–19
Péron G, Hines JE, Nichols JD, Kendall WL, Peters KA, Mizrahi DS (2013) Estimation of bird and bat mortality at wind-power farms with super population models. J Appl Ecol 50:902–911
Regan HM, Hierl LA, Franklin J, Deutschman DH, Schmalbach HL, Winchell CS, Johnson BS (2008) Species prioritization for monitoring and management in regional multiple species conservation plans. Divers Distrib 14:462–471
Richard Y, Edward R, Filippi A, Filippi D (2011) Assessment of the risk to seabird populations from New Zealand commercial fisheries. Final Research Report for New Zealand Ministry of Fisheries projects IPA2009/19 and IPA2009/20 (Unpublished report held by the Ministry of Fisheries, Wellington), p 66. Available via http://www.dragonfly.co.nz/publications/pdf/Richardetal_2011a_IPA2009-20.pdf. Accessed on 28 Sept 2015
Runge MC, Sauer JR, Avery ML, Blackwell BF, Koneff MD (2009) Assessing allowable take of migratory birds. J Wildlife Manage 73:556–565
Schaub M (2012) Spatial distribution of wind turbines is crucial for the survival of red kite populations. Biol Conserv 155:111–118
Schuster E, Bulling L, Koppel J (2015) Consolidating the state of knowledge: a syntopic review of wind energy’s wildlife effects. Environ Manage 56:300–331
Slade NA, Gomulkiewicz R, Alexander HM (1998) Alternatives to Robinson and Redford’s method of assessing overharvest from incomplete demographic data. Conserv Biol 12:148–155
Stahl JT, Oli MK (2006) Relative importance of avian life-history variables to population growth rate. Ecol Model 198:183–194
Swihart RK, Gehring TM, Kolozsvary MB, Nupp TE (2003) Responses of “resistant” vertebrates to habitat loss and fragmentation: the importance of niche breadth and range boundaries. Divers Distrib 9:1–18
Swihart RK, Lusk JJ, Duchamp JE, Rizkalla CE, Moore JE (2006) The roles of landscape context, niche breadth, and range boundaries in predicting species responses to habitat alteration. Divers Distrib 12:277–287
Taylor BL, Wade PR, De Master DP, Barlow J (2000) Incorporating uncertainty into management models for marine mammals. Conserv Biol 14:1243–1252
Wade PR (1998) Calculating limits to the allowable human-caused mortality of cetaceans and pinnipeds. Mar Mammal Sci 14:1–37
Watling JI, Donnelly MA (2007) Multivariate correlates of extinction proneness in a naturally fragmented landscape. Divers Distrib 13:372–378
Wilson JC, Elliott M, Cutts ND, Mander L, Mendão V, Perez-Dominguez R, Phelps A (2010) Coastal and offshore wind energy generation: is it environmentally benign? Energies 3:1383–1422
Acknowledgements
We thank V. Bennett, P. Cryan, D. Houseknect, T. Katzner, and M. Runge for reviewing early versions of the methodology. We particularly thank M. Runge for asking us to think harder about the utility of PBR. We are extremely grateful for external review panel members, T. Allison, W. Erickson, A. Hale, and F. Bennet. J. Havens developed nearly all the complex graphics and we appreciate his help. Finally, we thank the organizers of the CWW conference in Berlin and the many individuals who gave us excellent feedback on the methodology.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Diffendorfer, J.E. et al. (2017). A Method to Assess the Population-Level Consequences of Wind Energy Facilities on Bird and Bat Species. In: Köppel, J. (eds) Wind Energy and Wildlife Interactions. Springer, Cham. https://doi.org/10.1007/978-3-319-51272-3_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-51272-3_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-51270-9
Online ISBN: 978-3-319-51272-3
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)