Abstract
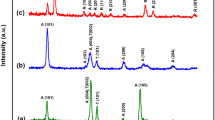
Titania nanotubes (TiO2-NTs) were prepared from anatase TiO2 nanopowder with a crystallite size of 90 nm by a hydrothermal method followed by ion exchange. The obtained TiO2-NTs were filled with NiFe2O4 quantum dots (QDs) and subjected to hydrogen at 600°C to produce TiO2-NTs filled with Ni-Fe nanoalloy. Filling TiO2-NTs with NiFe2O4 quantum dots shifted the absorption edge of the nanotubes to the visible region, narrowing the band gap of TiOx-NTs. The modified TiO2-NTs were investigated for the degradation of toxic dyes for water treatment. These TiO2NTs offered high degrees of photodegradation of the organic dyes with high visible light activity. They also were a promising corrosion inhibitor. The electrical conductivity-temperature dependences of empty anatase TiO2-NTs, anatase TiO2-NTs filled with NiFe2O4 QDs, anatase TiO2-NTs filled with Ni-Fe nanoalloy and NiFe2O4 compact disks were measured in the temperature range 25-850°C. The modified TiO2-NTs were also tested for CO2 sensing.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
I.K. Konstantinou and T. A. Albanis, “Photocatalytic Transformation of Pesticides in Aqueous Titanium Dioxide Suspensions Using Artificial and Solar Light: Intermediates and Degradation Pathways.” Appl. Catal. B-Environ., 42 (2003), 319–335.
A. Ghicov, J. M. Macak, H. Tsuchiya, J. Kunze, V. Haeublein, L. Frey, and P. Schmuki, “Ion implantation and Annealing for an Efficient N-Doping of TiO2Nanotubes.” Nano Letters, 6 (2006), 1080–1082.
X. Quan, S. Yang, X. Ruan, and H. Zhao, “Preparation of Titania Nanotubes and Their Environmental Applications as Electrode.” Environmental Science and Technology, 39 (2005), 3770–3775.
K. Meinert, C. Uerpmann, J. Matschullat, and G. Wolf, “Corrosion and Leaching of Silver Doped Ceramic Ibad Coatings on SS 316L under Simulated Physiological Conditions.” Surface and Coatings Technology, 103 (1998), 58–65.
N.O. Savage, S.A. Akbar, and P.K. Dutta, “Titanium Dioxide Based High Temperature Carbon Monoxide Selective Sensor.” Sensor. Actuat. B-Chem., 72 (2001), 239–248.
C. Wang, C. Böttcher, D.W. Bahnemann, and J.K. Dohrmann, “A Comparative Study of Nanometer Sized Fe(lii)-Doped Tio2 Photocatalysts: Synthesis, Characterization and Activity.” Journal of Materials Chemistry, 13 (2003), 2322–2329.
N. Wang, H. Lin, J. Li, X. Yang, B. Chi, and C. Lin, “Effect of Annealing Temperature on Phase Transition and Optical Property of Titanate Nanotubes Prepared by Ion Exchange Approach.” Journal of Alloys and Compounds, 424 (2006), 311–314.
M. Bahgat, A.A. Farghali, A.F. Moustafa, M.H. Khedr, and M.Y. Mohassab-Ahmed, “Electrical, Magnetic, and Corrosion Resistance Properties of TiO2 Nanotubes Filled with NiFe2O4 Quantum Dots and Ni—Fe Nanoalloy.” Appl. Nanos, (2012), 1–9.
A. Farghali, M. Khedr, and A. Moustafa, “Photocatalytic Activity and Magnetic Properties of Nanocrystallite Strontium Hexaferrite Prepared by Self-Flash Combustion.” Mater. Tech.: Advanced Pelf Mater., 23 (2008), 104–109.
J. Lee, T. Kim, J. Yu, and S. Chung, “In-Situ Alloying on Synthesis of Nanosized Ni-Fe Powder.” Nanostructured Materials, 9 (1997), 153–156.
Y. Yu, H.-H. Wu, B.-L. Zhu, S.-R. Wang, W.-P. Huang, S.-H. Wu, and S.-M. Zhang, “Preparation, Characterization and Photocatalytic Activities of F-Doped TiO2 Nanotubes.” Catalysis Letters, 121 (2008), 165–171.
N. Wang, H. Lin, J. Li, X. Yang, B. Chi, and C. Lin, “Effect of Annealing Temperature on Phase Transition and Optical Property of Titanate Nanotubes Prepared by Ion Exchange Approach.” Journal of Alloys and Compounds, 424 (2006), 311–314.
Author information
Authors and Affiliations
Editor information
Rights and permissions
Copyright information
© 2014 TMS (The Minerals, Metals & Materials Society)
About this paper
Cite this paper
Mohassab-Ahmed, M.Y., Moustafa, A.F., Farghali, A.A., Sohn, H.Y., Khedr, M.H. (2014). TiO2 Nanotubes Filled with NiFe2O4 Quantum Dots or Ni-Fe Nanoalloy: Synthesis and Applications. In: TMS 2014: 143rd Annual Meeting & Exhibition. Springer, Cham. https://doi.org/10.1007/978-3-319-48237-8_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-48237-8_5
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48593-5
Online ISBN: 978-3-319-48237-8
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)