Abstract
Filamentous cyanobacteria are important primary producers and N2 fixers in many terrestrial environments. As reduced nitrogen is often limiting, some thalloid liverworts (Marchantiophyta), hornworts (Anthocerophyta), the water fern Azolla (Salviniales), cycads (Cycadophyta), and the angiosperm Gunnera (Gunnerales) have evolved the ability to establish stable and structurally well-defined symbioses with N2-fixing cyanobacteria. Also a wide diversity of lichen-forming fungi have cyanobacteria as photosynthetic symbionts or as N2-fixing symbionts. Cyanolichen symbioses have evolved independently in different fungal lineages, and evolution has often resulted in convergent morphologies in distantly related groups. DNA techniques have provided a wealth of new information on the diversity of symbiotic cyanobacteria and their hosts. The fact that many plants and fungi engage in many different symbioses simultaneously underlines the probable significance of diffuse evolutionary relationships between different symbiotic systems, including cyanobacterial and mycorrhizal associations. This review introduces the reader to recent research on symbiotic cyanobacteria in terrestrial ecosystems and shortly describes the astonishing range of diversity in these ecologically important associations.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction
Mutually beneficial symbiotic interactions are an inherent feature of most ecological communities. Nitrogen is essential for growth of land plants, but the availability of reduced nitrogen in the soil is often limiting. Diazotrophic bacteria are able to convert atmospheric dinitrogen (N2) to ammonia (NH3) that can be utilized by plants, and consequently, numerous land plants form an association with N2-fixing bacteria. These interactions include the morphologically and physiologically highly coevolved symbioses between legumes and rhizobia, between actinorhizal plants and Frankia, and a plethora of more casual associations between plants and prokaryotic diazotrophs (Bothe et al. 2010; Santi et al. 2013).
Filamentous cyanobacteria are important N2 fixers in aquatic ecosystems but also in many terrestrial environments (Cornelissen et al. 2007; Menge and Hedin 2009; Elbert et al. 2012; Lindo et al. 2013; Rousk et al. 2013; Arróniz-Crespo et al. 2014). However, only a restricted and highly paraphyletic assemblage of land plants establish well-defined symbioses with cyanobacteria. These include two genera of thalloid liverworts (Marchantiophyta), all hornworts (Anthocerophyta), one genus of ferns (Azolla, Salviniales), all cycads (Cycadophyta), and one isolated genus of angiosperms (Gunnera, Gunnerales). Within the symbiotic structures produced by plants, the symbiotic cyanobacteria (cyanobionts) are sustained by sugars provided by the host plant (Adams and Duggan 2008; Meeks 2009; Ekman et al. 2013). Most plant–cyanobacterium symbioses are facultative in the sense that both partners can be cultured separately, but plants in the wild invariably associate with cyanobacterial symbionts.
Nitrogen is also essential for fungal growth and not surprisingly also many fungi associate with N2-fixing prokaryotes, including diazotrophic cyanobacteria (Schneider et al. 2016). In addition, a wide diversity of lichen-forming fungi have cyanobacteria as photosynthetic symbionts (photobionts). Cyanolichen symbioses have evolved independently in many fungal lineages, and convergent evolution has often resulted in similar thallus morphologies in distantly related groups of lichen-forming fungi (Rikkinen 2002).
This review introduces the reader to recent research on terrestrial symbiotic cyanobacteria and shortly describes the range of variation in their associations with land plants and fungi. Many if not most plants and fungi engage in multiple symbioses simultaneously. This provides vast possibilities for indirect effects between different symbionts, as well as the potential for each symbiont to modify the benefits and costs the other symbionts impose on their hosts. Thus, special attention is given to possible diffuse connections between cyanobacterial and other symbioses, including the diverse mycorrhizal associations of early land plants.
Symbiotic Cyanobacteria
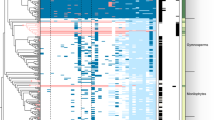
While cyanobacteria are diverse and sometimes abundant in many terrestrial ecosystems, only a small minority of them establish well-defined symbioses with eukaryote hosts. Nostoc is by far the most common genus of cyanobacteria associated with land plants and fungi (Fig. 1). Also several other nostocalean and stigonematalean genera (Howard-Azzeh et al. 2014), including Calothrix, Dichothrix, Stigonema, and the recently re-circumscribed Rhizonema (Lücking et al. 2009, 2013a; Dal-Forno et al. 2013), include symbiotic taxa. Lichen-forming ascomycetes of the predominately tropical order Lichenomycetes seem to mainly associate with Gloeocapsa, Anacystis, and other non-nostocalean cyanobacteria (Tschermak-Woess 1988; Voytsekhovich et al. 2011a, b). However, DNA studies on the biological diversity in these symbioses have only barely begun (Ortiz-Álvarez et al. 2015).
Lichen-symbiotic and free-living terrestrial cyanobacteria. A Foliose bipartite cyanolichen (Leptogium sp., Peltigerales) with cyanobionts (Nostoc) inside thin gelatinous thallus. The brown disks are apothecia of the fungal symbiont. B–D Morphological variation of Nostoc cyanobionts in three specimens of epiphytic Leptogium photographed through the thin upper cortex of hydrated thalli. Note the large heterocysts in B and C. The small translucent bodies between cyanobacterial trichomes are fungal hyphae in optical cross section. E–F Morphological variation in free-living colony-forming Nostoc. G Foliose bipartite cyanolichen (Coccocarpia sp., Peltigerales) with Rhizonema cyanobionts in the photobiont layer. Scale bars A, E–G 5 mm; B–D 5 μm
When in symbiosis, the phenotype of cyanobacteria is often modified, complicating direct comparisons between symbiotic cyanobacteria and their aposymbiotic relatives (Bergman and Hällbom 1982; Schüßler 2012; Ran et al. 2010). However, DNA methods can now be used to accurately identify symbiotic cyanobacteria both from fresh biological material (Rikkinen 2013) and dry herbarium specimens (Wright et al. 2001; Palinska et al. 2006). At the generic level, the standard DNA method used for identifying symbiotic cyanobacteria is 16S rRNA gene sequencing. However, due to many unresolved problems in cyanobacterial taxonomy, the symbiotic taxa cannot presently be identified to bacterial species (Oren and Garrity 2014; Pinevich 2015).
Symbiotic Nostoc can benefit their hosts either by providing fixed nitrogen, as in all plant symbioses and some cyanolichens, or by serving as a source of both carbon and nitrogen, as in most cyanolichens. Several studies have shown that lichen symbiontic Nostoc genotypes are closely related to plant symbiotic and free-living forms of the same genus (Paulsrud 2002; Rikkinen 2004; Papaefthimiou et al. 2008a; Yamada et al. 2012). They have also indicated that many fungal hosts are highly selective with respect to their cyanobionts. On the other hand, some lichen-forming fungi are known to associate with many different cyanobiont genotypes and frequently share them with other fungal species (Kaasalainen et al. 2012; O’Brien et al. 2013; Magain and Sérusiaux 2014; see Rikkinen 2013 for recent review). Bryophytes and cycads do not seem to be quite as selective in their cyanobiont choice and can often associate with several different Nostoc genotypes (Rikkinen and Virtanen 2008; Gehringer et al. 2010; Yamada et al. 2012).
As a whole, attempts to characterize the cyanobacterial symbiont have so far been made for a minute fraction of all known terrestrial symbioses. However, the complete genomic sequence of the symbiotically competent cyanobacterium Nostoc punctiforme (strain ATCC 29133 or PCC 73102), originally isolated from the coralloid roots of the cycad Macrozamia, has been determined (Meeks et al. 2001). Also the genome of the cyanobiont of the water fern Azolla (“Nostoc azollae” 0708) has been sequenced (Vigil-Stenman et al. 2015), and genomic sequences of several lichen-associated Nostoc genotypes are expected to become available soon (Grube et al. 2014). Metagenome sequencing of cyanolichens is also providing new insights into the genetic diversity of cyanobacterial symbioses (Kampa et al. 2013; Sigurbjörnsdóttir et al. 2015).
Cyanobacterial Adaptations to Symbiosis
Species of Nostoc form multicellular filaments in which, especially under conditions of combined nitrogen deprivation, some cells differentiate into heterocysts (Fig. 1), which have a thickened cell wall and provide a micro-aerobic environment for the functioning of the highly oxygen-sensitive enzyme nitrogenase, which is essential for the conversion of N2 to NH3 (Dodds et al. 1995; Flores and Herrero 2010). The vegetative cells and heterocysts of Nostoc are mutually interdependent; the latter import photosynthates from vegetative cells and provide nitrogen in return. Multicellularity evolved in cyanobacteria already 2.5 billion years ago (Tomitani et al. 2006; Schirrmeister et al. 2011, 2013, 2015) and has since been lost and regained several times in different cyanobacterial lineages (Flores and Herrero 2010; Claessen et al. 2014).
The vegetative cells of Nostoc can also differentiate into thick-walled resting stages (akinetes) and/or hormogonia which are short, small-celled filaments that move by gliding (Meeks et al. 2002). Hormogonia are attracted to root extracts and to certain sugars, including glucose, and can penetrate into tissues and cells of plant and fungal hosts (Meeks and Elhai 2002; Adams and Duggan 2008; Ekman et al. 2013). They differentiate in response to a range of signals, including the still uncharacterized hormogonium-inducing factor (HIF) that is released by plants under nitrogen starvation (Campbell and Meeks 1989; Nilsson et al. 2006). For more on the role and mechanisms of cyanobacterial motility in plant infection, see Adams and Duggan (2008) and Adams et al. (2013).
When inside the plant host, hormogonium production is repressed by hormogonium-repressing factors . Liaimer et al. (2015) reported that the nonribosomal peptide nostopeptolide produced by symbiotic Nostoc can function either as a hormogonium-repressing factor or as a chemoattractant, depending on its extracellular concentration. Splitt and Risser (2016) found that plant-derived sucrose or sucrose analogs can both repress hormogonia and induce the production of a polysaccharide sheath that seems to play a role in the establishment and maintenance of the symbiotic state in Nostoc. The signaling between the plant and the cyanobiont may also involve arabinogalactan proteins (Jackson et al. 2012; Adams et al. 2013).
Inside the host plant, the symbiotic cyanobacterium can undergo drastic morphological and physiological changes. The vegetative cells may be enlarged and show irregularities of shape. The rate of CO2 fixation tends to be reduced, whereas N2 fixation is stimulated and ammonium assimilation downregulated (Adams and Duggan 2008; Adams et al. 2013). In plant symbioses and some cyanolichens, the frequency of heterocysts increases, often reaching 30–40 %, which is several times higher than that typically found in free-living Nostoc (Meeks and Elhai 2002). The doubling time of symbiotic Nostoc may be slowed down to a fraction of that in free-living cyanobacteria, which ensures that the cyanobiont does not outgrow its host. For details on genetic aspects of plant–cyanobiont interactions, the reader is referred to Adams et al. (2013).
In exchange for fixed nitrogen, the plant-symbiotic cyanobacteria extract a carbon cost from their hosts. The high rate of N2 fixation cannot be maintained by the limited photosynthetic capacity of the cyanobionts themselves and must be supported by reduced carbon from the host. Symbiotic cyanobacteria are facultative heterotrophs and the sugars most frequently assimilated are fructose, glucose, and sucrose. From a carbon budget perspective, the overall costs to the host for maintaining cyanobacterial N2 fixation and respiration have not been determined, but they may be considerable. Especially in situations where soil nitrogen is easily obtained, the cost of maintaining cyanobionts may outweigh the benefits and explain why cyanobacterial symbioses are not more common among land plants (Ekman et al. 2013).
The cell surfaces of cyanobacteria are covered with complex carbohydrates which act as barriers against different types of stress and can also act as specific recognition factors (Kehr and Dittmann 2015). Recognition of compatible cyanobiont cells is believed to be performed by specific lectins produced and secreted by the plant or fungal host. Lectins have been identified from the fungal hosts of several cyanolichen species (Manoharan et al. 2012; Miao et al. 2012), and a glycosylated arginase acting as a fungal lectin has been found to bind to Nostoc from cyanolichens to function in the recruitment and adhesion of cyanobiont cells to the hyphal surface (Díaz et al. 2009, 2011; Vivas et al. 2010).
Vertical transmission of cyanobionts from one host generation to the next has evolved independently in many different lineages of cyanolichens (Rikkinen 2002), in thalloid liverworts (Rikkinen and Virtanen 2008), and in the cyanobacterium–Azolla symbiosis (Adams et al. 2013). The production of specialized symbiotic propagules helps to maintain pairwise symbiotic interactions over time and promotes the likelihood of coevolution between specific partners. The cyanobiont of Azolla spends its entire life cycle within the host plant and due to genomic erosion is now unable to grow aposymbiotically (Ran et al. 2010; Vigil-Stenman et al. 2015). Also some lichen-symbiotic Nostoc genotypes are “unculturable” and may well have lost their ability to grow outside lichen thalli (Rikkinen 2013).
Kaasalainen et al. (2012) related the high diversity of microcystin variants and corresponding genes in lichen cyanobionts to the evolutionary effects of symbiotic dispersal. When packaged into propagules of symbiotically dispersing lichens, the cyanobionts invariably experience extreme genetic bottlenecks. On the other hand, the close association with one or more fungal hosts likely promotes the selection for traits different from those typically experienced by nonsymbiotic cyanobacteria. As a whole, the recurrent bottlenecks and other populations shaping effects may have been instrumental for the evolution of the present genetic and chemical diversity in lichen cyanobionts.
Cyanobacteria produce a wide range of secondary metabolites, including many toxic compounds (Calteau et al. 2014; Dittmann et al. 2015). The cyanobionts of some cyanolichens produce hepatotoxic microcystins and nodularin in symbiosis (Oksanen et al. 2004; Kaasalainen et al. 2009, 2012, 2013), and nodularin was recently also found from the roots of cycads (Gehringer et al. 2012). These nonribosomal peptides are familiar from cyanobacterial blooms in aquatic ecosystems, where they have caused animal poisonings around the world and also pose a threat to human health. It is also possible that some lichen-symbiotic cyanobacteria can contribute to the defense against their hosts and/or parasitic fungi by producing hassalladins or other mycotoxic compounds (Vestola et al. 2014; Shishido et al. 2015).
Cyanolichens
‘Lichens are traditionally defined as ecologically obligate symbioses between fungal hosts and symbiotic green algae and/or cyanobacteria. The term “cyanolichen” is used of lichens with a cyanobacterial symbiont (cyanobiont), either as the sole photosynthetic partner (photobiont) or as a N2-fixing symbiont in addition to a primary eukaryotic photobiont. Lichen symbioses do not have independent scientific names; all partners of the symbiosis have their own names and the name of the intact “lichen species” refers to the fungal partner alone. Cyanolichen symbioses are ecologically obligate in the sense that the mycobionts of most cyanolichens cannot be cultured without the appropriate cyanobionts, and most of the cyanobionts do not appear to establish aposymbiotic populations outside lichen thalli.
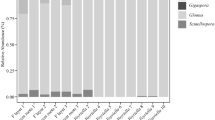
Symbioses between different fungi and cyanobacteria have evolved independently on many occasions, and in some lineages, the symbiosis seems to have been lost. Numerous cyanolichen species, possibly even the majority, have not yet been found and described (Lumbsch et al. 2011; Lücking et al. 2014; Moncada et al. 2014). A lichen-symbiotic lifestyle appears to have evolved at least ten times in the Ascomycota and five times in the Basidiomycota, with a vast majority of the ca. 18,000 currently accepted species of lichen-forming fungi belonging to the ascomycetes (Lücking et al. 2014). Only about 10 % of the currently known species of mycobionts establish lichen symbioses with cyanobacteria, with the overwhelming majority associating with green algae (Chlorophyta), especially certain groups of trebouxiophytes and Trentepohliales or rarely with other types of eukaryotic algae (Rikkinen 2002, 2015).
Among the fungi , cyanobacteria establish symbiotic associations mainly with ascomycetes (Ascomycota). Glomeromycetes (Glomeromycota) and basidiomycetes (Basidiomycota) are common partners in mycorrhizal symbioses, but associations between them and cyanobacteria are rare. Prominent exceptions include basidiomycetes of the Dictyonema clade (Dal-Forno et al. 2013; Lücking et al. 2014) and the enigmatic glomeromycete Geosiphon (Schüßler 2012), which does not well fit into the commonly used definition of a “lichen-forming fungus.” The polyphyletic chytrids (Chytridiomycota) include many parasites of cyanobacteria (Gerphagnon et al. 2013; Rohrlack et al. 2015), but no mutualistic forms have been described.
The Lichen Thallus
Within the lichen thallus, the cyanobiont provides sugar (as glucose) and/or fixed atmospheric dinitrogen (as ammonium) to the mycobiont. The fungus, in turn, provides the cyanobionts water, carbon dioxide, and stable environment that is relatively well buffered against environmental extremes and grazing invertebrates.
On the basis of their general habit, cyanolichens have traditionally been grouped into foliose, fruticose, and crustose species (Fig. 2). This division is obviously artificial and convergent forms have repeatedly evolved in different fungal lineages. The thalli of most cyanolichens are foliose, i.e., their dorsiventral thalli are flat and lobate and mainly grow horizontally. Fruticose cyanolichens produce shrubby, often upright thalli with cylindrical lobes that may be attached to the substrate only by a narrow base. Crustose cyanolichens produce relatively undifferentiated thalli that often grow very tightly attached to or even partly immersed into the substrate.
Cyanolichen growth forms. A Foliose bipartite cyanolichen (Peltigera praetextata, Peltigerales) with cyanobionts (Nostoc) in a layer below the upper cortex of the stratified thallus. B Foliose cephalodiate lichen (Peltigera leucophlebia, Peltigerales) with cyanobionts (Nostoc) in cephalodia on the upper surface of the green algal thallus. C Fruticose cephalodiate lichen (Stereocaulon sp., Lecanorales) with cyanobionts in large saccate cephalodia. D Crustose bipartite cyanolichen (Collema sp., Peltigerales). E Gelatinous bipartite cyanolichen (Leptogium saturninum, Peltigerales). Scale bars 5 mm
The thalli of most lichens are clearly stratified and have a protective upper cortex, a well-defined photobiont layer immediately below, and a loose medulla that effectively facilitates gas exchange. The lower surface may have rhizines which attach the lichen to substrate, help to maintain airspace below the thallus, and contribute to water absorption. The effective exchange of metabolites obviously requires an intimate connection between the mycobiont and photobionts. In most cyanolichens, thin-walled mycobiont hyphae penetrate the gelatinous sheaths of cyanobionts but are not in direct contact with the cyanobacterial cell wall. For more details of the anatomy of lichen thalli, the reader is referred to the recent review by Honegger (2012).
The light-absorbing fungal pigments and refractive structures in the cortex influence the quantity and quality of light that reaches the photobiont layer (Bjerke et al. 2005; Wu et al. 2014). The structural organization of the thallus also influences water relations; while thick thalli need a relatively long time to fill their internal water storage, the fully hydrated thalli can also continue photosynthesis for extended periods while drying (Gauslaa 2014; Merinero et al. 2014). In most cyanolichens, the structural characteristics of the thallus can be modified in response to the prevailing environment. The genetic regulation of lichen thallus formation and metabolite exchange is still almost unknown, but it must involve extensive molecular crosstalk between the partners (Chua et al. 2012; Kampa et al. 2013; Wang et al. 2014).
The cyanobionts of gelatinous cyanolichens or so-called jelly lichens (Oksanen et al. 2002; Otálora et al. 2013, 2014) are not organized into a distinct photobiont layer but occur more freely within the medulla (Fig. 2). The extensive gelatinous sheaths of the cyanobionts can absorb large quantities of water, and consequently, gelatinous cyanolichens typically exhibit major changes in thallus dimensions during wetting and drying and are in this respect similar to the gelatinous colonies of free-living Nostoc (Fig. 1).
Based on symbiont composition, cyanolichens have traditionally been divided into two artificial groups: bipartite cyanolichens and tripartite or cephalodiate lichens (Fig. 2). In bipartite cyanolichens , the cyanobionts typically form a photobiont layer immediately below the upper cortex. In cephalodiate lichens , the fungus produces a compound thallus with green algal and cyanobacterial symbionts (Fig. 3). In most cases, the green algal symbionts form a photobiont layer, while the cyanobionts are restricted to cephalodia, i.e., specific structures housing cyanobionts in an otherwise green algal lichen thallus (Cornejo and Scheidegger 2013). Lichen cyanobionts can deliver both sugar and fixed nitrogen to their fungal partners, and the relative importance of these two activities differs in bipartite and cephalodiate cyanolichens. In cephalodiate lichens, the green algal photobionts typically produce most photosynthate and the cephalodial cyanobionts mainly fix nitrogen (Green et al. 2008; Nash 2008). Concurrently, the cephalodial cyanobionts tend to show higher heterocyst frequencies and higher rates of N2 fixation than those of bipartite cyanolichens. As a whole, cephalodia occur in hundreds of lichen species, often hidden in the medulla or on the lower surface of the thallus, reflecting the primary role of cephalodial cyanobionts in N2 fixation.
Cephalodia and photosymbiodemes. A Cephalodia with Nostoc cyanobionts on upper surface of Peltigera aphthosa (Peltigerales). B The cephalodia are delimited by a continuous fungal cortex. C Poorly delimited cephalodia with Nostoc cyanobionts on the stems of Stereocaulon sp. (Lecanorales). D Large cephalodia with Nostoc cyanobionts on the upper surface of Placopsis sp. (Trapeliales). E Photosymbiodeme of Pseudocyphellaria sp. (Peltigerales). Green algal lobes with Nostoc cyanobionts in internal cephalodia developing from bipartite thallus of the same lichen. F Independent Dendriscocaulon cyanomorph of Sticta sp. (Peltigerales). G Photosymbiodeme of Nephroma arcticum (Peltigerales). Green algal lobes with Nostoc cyanobionts in internal cephalodia developing from bipartite thallus of the same lichen. Scale bars 5 mm
Cephalodiate cyanolichens can either have one cyanobacterial genotype in all cephalodia of a single lichen thallus (Paulsrud et al. 1998, 2001) or house different cyanobacterial genotypes in different cephalodia (Paulsrud et al. 2000; Kaasalainen et al. 2009), and some tripartite lichens have the two different photobionts in what appears to be the same photobiont layer (Henskens et al. 2012). It is possible that some mycobionts can respond to habitat variability and environmental change by selecting between different cyanobiont genotypes with different environmental optima, in a similar way as has been described from some green algal lichens (Casano et al. 2011). Some cyanolichen mycobionts can produce distinct thallus morphologies in symbiosis with compatible green algae and cyanobacteria, respectively (Fig. 3). The disparate morphs can either combine to form one chimeric thallus or grow separately. Chimeric lichens with green algae and cyanobacteria as photobionts in different parts of one thallus are called photosymbiodemes , and the two types of free-living morphs are called chloromorphs (chlorosymbiodemes) and cyanomorphs (cyanosymbiodemes), respectively. Many previously unknown photosymbiodemes have recently been described from different cyanolichen lineages (Aptroot and Schumm 2009; Magain et al. 2012; Magain and Sérusiaux 2014). They can potentially be used for studying the influence of photobiont choice on thallus morphogenesis and metabolism under identical conditions of growth and host association (Schelensog et al. 2000).
While lichens are usually perceived as pairwise interactions between one fungus and one or two photobionts, they regularly also incorporate a plethora of associated microorganisms. Lichen thalli host diverse and apparently specialized communities of non-phototrophic bacteria (Grube et al. 2009; Hodkinson and Lutzoni 2009; Hodkinson et al. 2012; Sigurbjörnsdóttir et al. 2014; Aschenbrenner et al. 2016). Also the diversity of associated microfungi is remarkable; nearly 2000 species of lichenicolous fungi have been described and thousands of additional species are believed to await description (Arnold et al. 2009; U’Ren et al. 2012; Werth et al. 2013). Many lichenicolous fungi grow only on cyanolichens (Fig. 5), either as host-specific parasites or as broad-spectrum pathogens, saprotrophs, or commensals (Lawrey and Diederich 2016). In addition, some green algal lichens regularly start their development on cyanolichen thalli (Dal-forno et al. 2013), while others associate with colonies of “free-living” cyanobacteria, presumably in order to access fixed nitrogen (Rikkinen 2002).
Ascolichens and basidiolichens. A Fertile specimen of bipartite ascolichen Fuscopannaria sp. (Peltigerales) with apothecia on the upper surface of the thallus. B Bipartite ascolichen Peltigera sp. (Peltigerales) with apothecia on upper surface of upright thallus lobes. C Bipartite ascolichen Peltigera sp. (Peltigerales) producing symbiotic propagules (soredia) in well-delimited structures (soralia) on upper surface of the thallus. D Bipartite basidiolichen Dictyonema s.lat. (Agaricales) with Rhizonema cyanobionts in corticoid basidiomata. Scale bars 5 mm
Ecology of cyanolichens. A Lichens are important winter feed for reindeer which graze heavily on terricolous green algal species. However, the ruminants avoid eating cyanolichens, possibly due to toxic compounds produced by lichen cyanobionts. B Mollusk grazing limits the distribution of some cyanolichens in boreal rain forests and other humid habitats. C Selective feeding by unidentified beetle of Nostoc cyanobionts from cephalodia of Nephroma arcticum (Peltigerales). D Unidentified lichenicolous fungus parasitizing the photobiont layer of Peltigera sp. (Peltigerales). E Secondary substances produced by many lichen-symbiotic fungi produce can play a role in defense against herbivores. This Pseudocyphellaria species produces yellow vulpinic acid and other pulvinic acid derivatives in the medulla, and the toxic compound also accumulates in the symbiotic propagules (soredia) of the lichen. Scale bars 2 mm
Ascolichens and Basidiolichens
Among the Ascomycota , cyanobacteria form symbioses especially with Lecanoromycetes (Miadlikowska et al. 2014) and Lichinomycetes (Schultz et al. 2001). An overwhelming majority of cyanolichen mycobionts belong to Lecanoromycetes (Fig. 4). Species that form bipartite symbioses with cyanobacteria are largely restricted to the Peltigerales, while cephalodiate taxa occur more sporadically among different lecanoromycete orders (Miadlikowska et al. 2014). Most fungi in the Peltigerales (e.g., Collemataceae, Nephromataceae, Pannariaceae, and Peltigeraceae) associate with symbiotic Nostoc, while others (e.g., Coccocarpiaceae) associate with other nostocalean and/or stigonematalean cyanobionts (Lücking et al. 2009, 2013b; Ekman et al. 2014; Spirbille et al. 2014; Zúñiga et al. 2015, see Rikkinen 2013 for more detailed review).
All fungi in the predominately tropical ascomycete class Lichinomycetes are lichen symbiotic, and most of them seem to establish bipartite symbioses with Gloeocapsa, Anacystis, and other non-nostocalean cyanobacteria (Tschermak-Woess 1988; Schultz et al. 2001; Voytsekhovich et al. 2011a, b). However, Ortiz-Álvarez et al. (2015) reported that the cyanobionts of two European species of Lichina (Lichinomycetes) were closely related to marine and freshwater strains of Rivularia (Nostocales).
While lichenization is generally rare among basidiomycetes, it has clearly evolved several times in unrelated groups. Seven currently accepted genera form structurally well-delimited symbioses with green algae or cyanobacteria. In addition, several other groups of basidiomycetes include lichenicolous species, some of which grow on cyanolichens (Diederich 1996; Lawrey and Diederich 2016). The major lineages of Basidiomycota that contain lichen-forming species also exhibit saprotrophic, pathogenic, mycorrhizal, and lichenicolous taxa (Lawrey et al. 2009). For example, some species of Athelia (Atheliales) associate with cyanobacteria, while others are saprophytes or grow on coccomyxoid green algae or as lichen parasites (Oberwinkler 2012).
Recent studies have revealed that especially the Dictyonema clade (Fig. 4) in the Hygrophoraceae (Agaricales) includes a large number of hitherto undescribed species (Lücking et al. 2013a, 2014). All species of this clade are lichen-forming (Dal-Forno et al. 2013, 2016). The basal clade of Dictyonema s. lat. includes fungi with appressed-filamentous thalli and a simple hyphal sheath around the cyanobacterial photobiont filaments (Lawrey et al. 2009). Dictyonema s. str. is a paraphyletic grade of species that form appressed to shelflike filamentous thalli with the cyanobiont filaments surrounded by jigsaw puzzle-shaped hyphal sheaths. Acantholichen, Corella, and Cora are nested within Dictyonema s.lat and have coiled cyanobacterial filaments forming clusters, wrapped within a dense hyphal sheath formed by jigsaw puzzle-shaped cells of the mycobiont, appearing unicellular or pseudo-colonial. Acantholichen is sister to Corella, both in turn forming a sister clade to Cora (Lawrey et al. 2009; Dal-Forno et al. 2013). The Neotropical genus Acantholichen includes microsquamulose species with dark blue to gray thalli, with spiny apical cells on both thallus surfaces, giving them a coarsely white-pruinose appearance (Jørgensen 1998).
The cyanobionts of the Dictyonema and related basidiomycetes were previously identified as Scytonema or Chroococcus, depending on whether their filaments were short coiled or not (Tschermak-Woess 1988). Lücking et al. (2009) revealed that they form a lineage among nostocalean cyanobacteria clearly distinct from Scytonema species and named them Rhizonema (Lücking et al. 2009). In Dictyonema, Cora, and Acantholichen, the cyanobionts are enveloped by fungal hyphae and penetrated by haustoria. Thus, like in parasitic chytrids and some cyanophilous ascomycetes, the basidiomycete hosts actually penetrate the cell walls of cyanobacteria in search of nutrition (Oberwinkler 1984, 2012).
Lawrey et al. (2009) pointed out that the high concentration and diversity of lichen-forming taxa in the Hygrophoraceae suggest a predisposition toward lichen symbiosis in this family, even though nutritional modes in the lineage are not markedly different from many other families in the Agaricales, including mostly saprotrophic and ectomycorrhizal fungi. However, the family included many bryophilous taxa which associate either with the plants directly or with photosynthetic microbial communities containing complex mixtures of eukaryotic algae, cyanobacteria, and moss protonemata on water-retaining peat or well-decayed wood. This may indicate a tendency to switch from a saprotrophic to a lichen-symbiotic mode of nutrition in bryophilous species of this family (Lawrey et al. 2009).
Cyanolichen Ecology
Cyanolichens are found in many types of terrestrial environments ranging from tropical rain forests and semideserts to arctic tundra (Fig. 5). Their diversity and abundance are highest in relatively humid climates. Epiphytic species do particularly well in the moist and cool conditions of higher elevations in tropical mountains and in maritime regions of higher latitudes. They are often well represented in the epiphyte communities of old-growth boreal and temperate forests where they intercept and help to retain atmospheric moisture, sequester nutrients, and provide habitat and food for many invertebrates. Many epiphytic species thrive in microhabitats that combine moderate light intensities and ample moisture with periodic drying events.
In closed canopy forests, epiphytic cyanolichens are often most abundant in the lower and mid canopy sections, where the quantity and quality of light are moderated by the overlying canopy. Cyanolichens are generally less diverse in arid climates, but can be important components of biological soil crusts in semideserts, where they help to stabilize the soil and contribute to its fertility. The N2-fixing cyanobionts of cyanolichens contribute significant amounts of nitrogen to the ecosystem (Gavazov et al. 2010; Elbert et al. 2012). Flexibility between alternative nitrogen fixation pathways in Nostoc can be significant for cyanolichens that grow on nutrient-poor substrates and get most of their mineral nutrition through aerial deposition (Darnajoux et al. 2014; Hodkinson et al. 2014). Epiphytic cyanolichens are highly susceptible to the adverse effects of air pollution and can be used as indicators of clean air. In addition, many species have been hard hit by logging and are now more or less restricted to old-growth forests and can be used as indicators of long habitat continuity. For more information on the habitat ecology of cyanolichens, see Rikkinen (2015).
Many cyanolichen species facilitate the reproduction and simultaneous dispersal of their symbiotic consortium by producing symbiotic propagules. However, the mycobionts of most cyanolichen species only produce fungal spores and must thus reestablish their symbiotic association at each reproductive cycle. A compatible partner can be obtained either from a population of free-living cyanobionts or from the thallus or symbiotic diaspore of another cyanolichen. As appropriate cyanobionts are not likely to be ubiquitously distributed, the local availability of cyanobionts can explain many patterns of cyanolichen species occurrence, and shared symbiont specificity may lead to facilitative interactions between different fungal hosts. For example, some spore-dispersed mycobionts appear to be facilitated by the prior establishment of other cyanolichen species that produce symbiotic propagules (Rikkinen et al. 2002; Rikkinen 2003, 2013; Fedrowitz et al. 2011, 2012; Belinchón et al. 2014; Dal Grande et al. 2014) or by bryophytes that house appropriate cyanobacteria (Cornejo and Scheidegger 2016).
Geosiphon pyriformis–Nostoc Symbiosis
Geosiphon pyriforme (Geosiphonaceae, Archaeosporales) is the only species of Glomeromycota that is known to form a well-defined symbiosis with cyanobacteria (Schüßler 2012). While this unique association has only been found from a few sites in central Europe, there is full reason to believe that the symbiosis type is ancient and that other similar associations still exist and await discovery. The Geosiphon pyriformis–Nostoc symbiosis seems to represent a “living fossil” of multibiont associations that once connected the cyanobacterial and mycorrhizal symbioses of early bryophytes and contributed to the evolution of early terrestrial ecosystems.
The Glomeromycota is a monophyletic group of fungi that was previously included as an order in the former Zygomycetes. Details in the taxonomic history of the lineage were recently reviewed by Redecker and Schüßler (2014). Glomeromycotan fungi establish arbuscular mycorrhizal (AM) symbioses with land plants including most vascular plants and many hornworts and liverworts. The symbiotic fungi colonize plant roots but also efficiently penetrate the soil and improve plant water and mineral nutrient uptake in return for plant-assimilated carbon (van der Heijden et al. 2015).
Geosiphon pyriformis is unique among the presently known glomeromycetan fungi not only in associating with symbiotic cyanobacteria but also because it has not yet been shown to form mycorrhizae with bryophytes or vascular plants. The substrate ecology and establishment of the Geosiphon pyriformis–Nostoc symbiosis were described by Mollenhauer and Mollenhauer (1988) who managed to maintain the symbiontic consortium in the laboratory. The fungus grows among free-living cyanobacteria and bryophytes in the topsoil of humid, nutrient-poor sites and regularly occurs in close association with moss protonemata. The Nostoc symbionts are housed endosymbiotically within small multinucleate and vacuolated fungal bladders on the soil surface, and the fungal hyphae can produce new vesicles when a specific primordial stage of Nostoc is present. The cyanobacteria are incorporated by the fungus near the hyphal tip, which then swells and develops into a pear-shaped aboveground bladder up to 2 mm long. Each bladder results from a single incorporation event and different bladders formed by the mycelium can thus house different Nostoc genotypes. Within the terminal bladders, the cyanobionts proliferate and provide photosynthate and fixed nitrogen to the host. At times the fungus forms large resting spores similar to those of related glomeromycotan fungi.
Geosiphon bladders are essentially large, multikaryotic cells with a photosynthetically active region in the exposed apex and a more restricted storage region at the base, partly embedded in the soil and attached to the underground mycelium. All cyanobionts of each bladder are in one membrane-bound cellular compartment (symbiosome). There is a rudimentary fungal cell wall between the symbiosome membrane (fungal plasma membrane) and the enclosed Nostoc cells, basically similar to the thin cell walls of arbuscules of the AM fungi within colonized plant cells. For more details on the anatomy and ultrastructure, see Schüßler (2012), and for genetic aspects, see Schüßler et al. (2006, 2007) and Ellerbeck et al. (2013).
The endosymbiotic cyanobacterium isolated by Mollenhauer was originally identified as Nostoc punctiforme. There is a level of specificity in the Geosiphon pyriformis–Nostoc association as some genotypes of Nostoc are incorporated by the fungus, while in others the development of the cyanobacterium ceases at an early stage or it is not incorporated at all (Schüßler 2012). The cyanobiont can be readily isolated and cultivated without the fungal partner. The vegetative cells of the cyanobiont within symbiotic bladders are larger than the cells of typical free-living Nostoc, apparently because of the high osmotic pressure inside the bladder, but their ultrastructure is not markedly modified. Also the heterocyst frequency of cyanobionts within the bladder is similar to that of free-living Nostoc, indicating that the cyanobionts play a principal role in photosynthesis and not in N2 fixation (Kluge et al. 1991, 1992). The cyanobionts within Geosiphon bladders get water, carbon dioxide, and all inorganic nutrients except nitrogen from the fungal host. The fungus may also offer protection against heavy metals and other abiotic stress factors (Scheloske et al. 2001; Wojtczak and Janik 2016).
While Geosiphon pyriforme is the only AM fungus known to have endosymbiotic cyanobacteria, many other AM fungi host biotrophic endobacteria in their cytoplasm (Naumann et al. 2010; Torres-Cortésa et al. 2015) and also symbiotic bladders of Geosiphon house such endosymbionts. The Mollicutes (mycoplasma-related endobacteria, MRE) are related to Mycoplasma and Phytoplasma species which are biotrophic parasites in animals and plants. The coccoid bacteria appear to possess a Gram-positive cell wall and are not surrounded by host membrane (Torres-Cortésa et al. 2015).
MRE are common in the mycelia and spores of AMF and are thus also indirectly associated with plants. Recently they have also been identified from species of Endogone (Mucoromycotina), a fungal genus that also includes plant mycorrhizal species, implying that the symbiosis between MRE and fungi would predate the divergence between glomeromycetes and mucoromycetes. They are presumed to play some role in the AM symbiosis, but the nature of their role is unknown and it thus remains unclear whether they actually are mutualistic associates or parasites of their fungal hosts (Desirò et al. 2013b, 2015; Naito et al. 2015; Toomer et al. 2015).
Bryophyte–Cyanobacterium Symbioses
Bryophytes (liverworts, mosses, and hornworts) are generally recognized as the oldest living land plants (Shaw et al. 2011; Ligrone et al. 2012). While the exact relationships between the three lineages continue to remain ambiguous (Wickett et al. 2014), the recently most widely accepted view has placed liverworts as sister to all other embryophytes (Qiu et al. 2006). Unfortunately the fossil record is extremely fragmentary for all bryophytes and is of limited value for resolving the earliest divergences and radiations (Taylor et al. 2009). Spore fossils predate plant megafossils by 40 million years and give some clues to the pioneering events in land colonization some 470 Mya (Edwards et al. 2014; Brown et al. 2015).
Among the extant bryophytes, only the hornworts and some liverworts establish structurally well-defined symbioses with cyanobacteria. The cyanobionts are usually Nostoc, although also other cyanobacterial genera have been mentioned as symbionts of some bryophyte species (West and Adams 1997; Costa et al. 2001; Rikkinen and Virtanen 2008).
Liverwort Symbioses
Liverworts include ca. 5000 extant species in three classes. Among these, the Haplomitriopsida, including Treubiales with two genera and the monotypic Haplomitriales, is sister to all other taxa. The Blasiales with two monotypic genera is sister to the remaining complex thalloid liverworts, the Marchantiopsida, with ca. 340 extant species (Wahrmund et al. 2008). All the remaining liverworts belong to the diverse Jungermanniopsida, which includes both leafy and simple thalloid forms. Villarreal et al. (2016) estimated that the complex thalloid liverworts diverged 295 Mya (250–365 Mya) and the Marchantiidae (excluding Blasiales) 262 Mya (226–327 Mya).
Blasia pusilla and Cavicularia densa (Blasiales) are the only extant liverworts that form morphologically well-defined symbioses with cyanobacteria. The Nostoc cyanobionts of the thalloid liverworts occupy spherical auricles that develop on the underside of the thallus. The auricle starts its development from a three-celled mucilage hair, and Nostoc hormogonia enter the auricle when it is a small, dome-shaped structure. Concomitant growth of cyanobiont and auricle expansion eventually results in a larger, more or less globular structure that is easily visible even with the naked eye.
Auricles of Blasia pusilla have two slime papillae, with one (the inner slime papilla) partly filling the young auricle cavity and the other (the outer slime papilla) arising from the thallus adjacent to the auricle (Fig. 6). The cyanobacteria enter the auricles as hormogonia, lose their motility, and differentiate heterocysts. As a cyanobiont colony grows, filamentous protrusions derived from the inner slime papilla grow into the cyanobiont colony and increase the surface area of contact between the cyanobiont and the bryophyte, thus enhancing nutrient exchange between the host and symbiont (Duckett et al. 1977; Renzaglia 1982; Renzaglia et al. 2000; Kimura and Nakano 1990).
Cyanobacterial symbiosis of the liverwort Blasia pusilla (Blasiales). A Young gametophyte with stellate gemmae on upper surfaces of thallus lobes. Some cyanobacterial auricles on the lower surface of the liverwort can be seen as dark spots through the thin marginal sections of thallus lobes. B Blasia pusilla is a pioneer colonizer of clayey soils. C The stellate gemmae are essentially miniature thalli and tend to be infected by symbiotic Nostoc while being still attached to the parent gametophyte. D Mature auricle with symbiotic Nostoc on the lower surface of the gametophyte. E–H Series of optical cross sections through a young uninfected auricle in the meristematic region of stellate gemma; n = Nostoc hormogonia and trichomes outside the auricle, a auricle, o outer slime papilla, x inner slime papilla. I Nostoc trichome with terminal heterocyst in contact with outer slime papilla. J Optical cross section through young uninfected auricle showing the outer slime papilla (o) and inner slime papilla (x). The Nostoc hormogonia infect the auricle through the minute opening seen just left of the inner slime papilla. K Optical cross section (from above) of Fig. 6 (continued) a recently infected young auricle with Nostoc propagating between the inner slime papilla (x) and the auricle wall (cells with chloroplasts). L Branched transfer cells (t) and detached Nostoc cells revealed by squashing a developing auricle. M Young basidioma of Blasiphalia pseudogrisella (Hymenochaetales) attached to rhizoids of Blasia pusilla. Several bottle-shaped receptacles (r) releasing ovoid, non-symbiotic gemmae have formed on the upper surface of the liverwort. Scale bars A–D, M 2 mm; E–J 10 μm; E–J 20 μm
Blasia and Cavicularia reproduce asexually by producing gemmae which facilitate the simultaneous dispersal of the host and Nostoc cyanobionts. Blasia produces ovoid gemmae in bottle-shaped receptacles and stellate gemmae on the upper surface of the tips of thallus lobes (Fig. 6). Cavicularia produces both types of gemmae in crescent-shaped receptacles forming close to the tips of mature thallus lobes (Fig. 7). The ovoid gemmae of both liverwort species are nonsymbiotic and only associate with cyanobacteria when they have dispersed and develop into thallus primordia. However, the stellate gemmae essentially represent miniature thalli, equipped with two symbiotic auricles, which are regularly infected by symbiotic Nostoc, while the propagules are attached to the parent gametophyte (Rikkinen and Virtanen 2008).
Cyanobacterial symbiosis of the liverwort Cavicularia densa (Blasiales). A Tip of thallus lobe with cyanobacterial auricles on the lower surface visible as dark spots on the upper surface. B Mature receptacle (re) producing ovoid and stellate gemmae closely associated with Nostoc colonies of mature auricles. C Close-up of two mature auricles as seen through the upper surface of the thallus. Scale bars 2 mm
During gametophyte growth, the symbiosis is continuously reestablished as young auricles are infected by Nostoc hormogonia. Concurrently, several different Nostoc genotypes are often present in single thalli. However, some Nostoc genotypes are dominant and widespread and typically shared by most bryophytes within a given locality (West and Adams 1997; Costa et al. 2001; Rikkinen and Virtanen 2008). The primary cyanobionts of Blasia and Cavicularia are closely related and the same Nostoc genotypes have also been found from terricolous cyanolichens (Rikkinen 2004, 2009, 2013; Rikkinen and Virtanen 2008; Papaefthimiou et al. 2008b).
The stellate gemmae of Blasia and Cavicularia can promote the persistence of specific cyanobacterial associations over the critical dispersal phase. During subsequent gametophyte development, new auricles are continuously formed. At this stage, some of the infecting cyanobacteria can represent suboptimal cyanobacteria from the soil (West and Adams 1997). This is not a serious problem, as only a few auricles infected by appropriate Nostoc genotypes are needed to support thallus growth. Hence, the cyanobacterial diversity of bryophyte thalli may more reflect difficulties in avoiding accidental infections, than the lack of symbiont preference (Rikkinen and Virtanen 2008).
Some leafy liverworts (Jungermanniopsida) including Frullania and Porella species regularly have cyanobacteria in the watersacs of their leaves (Dalton and Chatfield 1987). Cornejo and Scheidegger (2016) reported that Frullania asagrayana and the rare and endangered epiphytic cyanolichen Erioderma pedicellatum share identical Rhizonema cyanobionts. The liverwort is the principal substrate of Erioderma during its juvenile stage, and Rhizonema within the watersacs of liverwort leaves seems to facilitate the establishment and growth of the rare cyanolichen. Since Rhizonema has not been found “free-living,” the availability and distribution of cyanobionts likely represent a critical limit to the establishment and recruitment of ascospore-dispersed cyanolichen species (Cornejo and Scheidegger 2016). The watersacs on some liverworts are also involved in other ecological interactions, including zoophagy of protozoa (Barthlott et al. 2000).
The basidiomycete Blasiphalia pseudogrisella (Hymenochaetales) forms appressoria on the rhizoids of Blasia (Fig. 6). The germinating spores of the fungus have also been observed to infect slime papillae in the meristematic region of liverwort gemmae. The fungus belongs to the Rickenella clade with other species that associate with bryophytes or green algae or are predacious on nematodes. Blasiphalia seems to only associate with Blasia and may be dispersed via infected gemmae of the liverwort host (Redhead 1981; Larsson et al. 2006).
In Blasia and Cavicularia, the slime papillae of symbiotic auricles play a crucial role in symbiosis by attracting hormogonia and developing into transfer cells that mediate metabolite exchange between the cyanobiont and the plant host. Also the slime papillae of other liverworts may be involved in symbiotic interactions with soil microbes (Fig. 8). Such associations have not been described in any detail, but casual observations reveal that the mucilage produced by slime papillae commonly supports a diversity of bacteria and filamentous fungi and some of them also infect the papillae. It seems likely that some of the associated bacteria are diazotrophic or in other ways beneficial to the plant.
Interactions between slime papillae of leafy liverwort (Odontoschisma elongatum, Cephaloziaceae) with bacteria and fungi. A Young uninfected slime papillae at margins and surface of reduced underleaves near the growing tip of the plant; the fragile slime papillae are rarely preserved in dry herbarium material. B Bacteria (b) and fungal hyphae attached to the surface of young slime papilla; eventually the fungus infects the papilla with haustoria (h). C At later stages the slime papillae become heavily infected by fungal hyphae and are eventually shed away. Scale bars 10 μm
In this context, one must emphasize that the production of slime papillae is an apomorphic feature that characterizes all extant liverworts. Slime papillae are ephemeral structures and typically produced in specific localities in growing liverworts where there is an obvious need for fixed nitrogen and/or other growth promoters, such as at the tips of growing thalli or leaves, in gemmiferous regions, around gametangia, etc. They are also typically produced in locations where moisture conditions are favorable for prolonged microbial activity, like in between rhizoids on the lower surfaces of liverwort thalli, around water reservoirs of lobed leaves and watersacs, etc. Without roots and a vascular system, liverworts cannot effectively transport nutrients like vascular plants. Fixing nitrogen where and when it is needed and then shedding the slime papillae may be a very widespread strategy among this ancient lineage of minute land plants.
Among thalloid liverworts, associations with glomeromycete fungi are widespread in both the complex (Marchantiopsida) and simple (Jungermanniopsida, Metzgeriidae) lineages. However, these fungi appear to be absent from Blasia and Cavicularia as well as several derived lineages. The intracellular endophytes typically infect thalloid liverworts through the rhizoids and colonize particular taxon-specific regions of the thalli where they form trunk hyphae with numerous short-lived, regularly dichotomizing arbuscular side branches (Pressel et al. 2010). The swollen rhizoids in many families of leafy liverworts (e.g., Cephaloziaceae, Calypogeiaceae) can be packed with hyphae of the ascomycete Rhizoscyphus ericae (Leotiomycetes), which also forms ericoid mycorrhizas with vascular plants. Pressel et al. (2008) pointed out that the origins of the ascomycete associations in liverworts may date back to more than 250 Mya. Thus, they long predate the ericoid mycorrhizas which arose only 106–114 Mya.
Hornwort Symbioses
Hornworts (Anthocerophyta) include about 220 currently accepted species in 12 genera (Villarreal et al. 2015). Several recent phylogenies have placed hornworts as the closest extant relatives of the tracheophytes (Groth-Malonek et al. 2005; Qiu et al. 2006; Chang and Graham 2011), but lately this has again been challenged (Cox et al. 2014; Wickett et al. 2014). The monospecific Leiosporoceros is sister to all other hornworts and is currently placed in a separate class Leiosporocerotopsida (Villarreal et al. 2015). Among many other unique features, many hornworts have a chloroplast with a central pyrenoid and a carbon concentration mechanism unknown from other land plants (Li et al. 2009; Xue et al. 2010; Villarreal et al. 2013). They are widely distributed in moist temperate and tropical habitats, mainly as pioneer colonizers of nutrient-poor substrates (Fig. 9). The tropics have the highest diversity of species per area, particularly tropical Asia and the Neotropics (Villarreal et al. 2010; Villarreal and Renner 2014).
Cyanobacterial symbiosis of hornworts (Anthocerophyta). A Anthoceros agrestis (Anthocerotales) is a pioneer colonizer of clayey soils. The horn-shaped sporophytes grow from archegonia embedded in the upper surface of the gametophyte. B Mature hornwort sporophytes split into two halves lengthwise, releasing the spores. C Nostoc colonies housed in mucilage clefts can be seen as dark spots on the lower surface of thallus. Scale bars 2 mm
Cyanobacterial symbiosis with Nostoc is an universal feature in hornwort gametophytes. The association is established via apically derived clefts on the lower surface of the thallus. The cavities are connected to the thallus surface via stomata-like pores. When the Nostoc cyanobiont enters a young cleft, the middle lamella between hornwort cells separates to form a schizogenous space. As the cavity forms, it is filled with mucilage produced by surrounding plant cells (Rodgers and Stewart 1977; Renzaglia 1978; Adams 2002). Most hornworts produce mucilage clefts continuously during thallus growth and each of them becomes individually infected by Nostoc hormogonia. The presence of the cyanobiont stimulates enlargement of the symbiotic clefts and noninfected cavities do not develop further (Rodgers and Stewarts 1977). The mature cyanobiont colonies are usually small and more or less globular. However, in mature Leiosporoceros thalli, the Nostoc cyanobionts form long and branching strands that run parallel to the main axis of the thallus (Villarreal and Renzaglia 2006). Also young thalli of Leiosporoceros have mucilage clefts at the thallus apex and Nostoc invasion seems to take place through them. Later no more clefts are produced and the established cyanobacterial strands start to elongate in synchrony with thallus growth and branching (Villarreal and Renzaglia 2006).
As most hornworts continuously produce new ventral mucilage clefts near the thallus apex throughout their life span, it is likely that multiple cyanobacterial genotypes will invade different clefts of a single hornwort thallus. The presence of multiple cyanobacterial genotypes within a single hornwort thallus have indeed been confirmed using molecular markers (West and Adams 1997; Costa et al. 2001). Even young Leiosporoceros thalli typically have many lobes, each with several mucilage clefts, and this can lead to cyanobiont variability within single mature thalli (Villarreal and Renzaglia 2006). The development of integrated cyanobiont network in Leiosporoceros eliminates the need for multiple cyanobacterial invasions during thallus growth. The canals also allow extensive surface contact for exchange between the partners along the length of the strand. A single invasion of the hornwort thallus can be adaptive as it eliminates the need to constantly attract new, appropriate symbionts. On the other hand, multiple invasions could more effectively ensure successful colonization and allow for more flexibility in cyanobiont choice (Villarreal and Renzaglia 2006).
Hornworts establish mycorrhizal symbioses with both Glomeromycota and Mucoromycotina fungi, often simultaneously (Ligrone 1988; Schüßler 2000; Desirò et al. 2013a; Bidartondo et al. 2011). Interestingly, the principal mode of fungal entry in the hornworts is via mucilage clefts and not through rhizoids as in most liverworts. Desirò et al. (2013a) noted that the fungal hyphae occur in close association with symbiotic Nostoc within the mucilage, possibly indicating a relationship resembling that of Geosiphon. They also found that the more abundant the cyanobacteria, the less likely the hornworts were to harbor mycorrhizal fungi. For example, the thalli of Leiosporoceros were found to be fungus-free (Desirò et al. 2013a).
Moss Symbioses
The mosses (Bryophyta) are the largest group of bryophytes consisting of some 10,000 species in three or four distinct clades usually recognized as classes. Although the order of diversification is still controversial, it is clear that the peat mosses (Sphagnopsida) with four genera and the monogeneric Takakiopsida are sister to all other mosses, followed by the Andreaeopsida with two genera and then the remainder of more advanced orders (Cox et al. 2010).
Many mosses regularly house epiphytic cyanobacterial colonies on their surfaces, especially in sheltered spots between leaves and rhizoids (Fig. 10). While most of these associations are undoubtedly facultative, they definitely play an important role in N2 fixation, especially in boreal and arctic and in some temperate ecosystems (Solheim et al. 1996; Zackrisson et al. 2004; Gavazov et al. 2010; Lindo and Whiteley 2011; Turetsky et al. 2012; Rousk et al. 2013; Arróniz-Crespo et al. 2014). Feather mosses in boreal forests commonly house heterocystous cyanobacteria (e.g., Nostoc, Calothrix, Stigonema) in addition to other N2-fixing bacteria (Houle et al. 2006; Cornelissen et al. 2007; Ininbergs et al. 2011). In peat bogs, cyanobacteria are often present in the dead and water-filled hyaline cells in the leaves and stems of Sphagnum. With an elevated pH, the hyaline cells may represent a favorable microhabitat for cyanobacteria in acidic peatlands (Berg et al. 2013; Kostka et al. 2016). Lindo et al. (2013) concluded that bryophyte–cyanobacterium associations carry out several important functions in nitrogen-limited boreal and arctic ecosystems through their production of recalcitrant litter, thermal protection of soils, and role as the primary source of nitrogen through N2 fixation. The composition of nitrogen-fixing moss-associated cyanobacterial communities differs between moss species, sites, and seasons, but the exact patterns remain largely unexplored (Ininbergs et al. 2011; Bay et al. 2013; Leppänen et al. 2013; Warshan et al. 2016).
Cyanobacterial symbioses of mosses (Bryophyta ). A Sphagnum riparium (Sphagnales) and other peat mosses can house cyanobacteria in the water-filled hyaline cells of their leaves and stems. B Moss-associated cyanobacteria play an important role in biological N2 fixation in arctic and boreal regions but also in montane rain forests. C Hylocomium splendens (Hypnales) and other feather mosses commonly have heterocystous cyanobacteria and other N2-fixing prokaryotes in sheltered spots between their leaves. Scale bars 2 mm
While many specialized fungi grow on mosses (Döbbeler 1997; Davey and Currah 2006; Kauserud et al. 2008) and many of them appear to be species-specific (Higgins et al. 2007; Stenroos et al. 2010), mutualistic symbioses between mosses and fungi have not been found (Pressel et al. 2010). Thus, all fungal associates of mosses seem to be either parasites or saprotrophs, and mosses represent the only division of land plants that does not seem to establish mycorrhizal symbioses with Glomeromycota. This is quite peculiar considering the early divergence and wide extant diversity of mosses (Liu et al. 2011; Taylor et al. 2015a).
Azolla Symbiosis
The genus Azolla (Salviniales, Pteridophyta) contains seven extant species of small aquatic ferns (Evrard and Van Hove 2004; Metzgar et al. 2007; Pereira et al. 2011). Several species are widely distributed in tropical and warm temperate regions (Fig. 11). The genus has an extensive fossil history starting from the Cretaceous based principally on isolated megaspores (Hall 1974; Collison 1980; Taylor et al. 2009).
Cyanobacterial symbiosis of the water fern Azolla (Salviniales). A Azolla filiculoides is native to warm temperate and tropical parts of the Americas and Australasia and has become invasive in several regions including western Europe and Africa. Scale bar 5 mm. B Azolla rubra is widespread throughout the Pacific and in southern Asia. In New Zealand, it is common in shallow water bodies such as ponds, lake margins, and slow-flowing streams and also grows in swamps on muddy ground. C The introduced Azolla filiculoides (Azolla section Azolla) and the larger native Azolla nilotica (Azolla section Rhizosperma) growing together on the muddy shore of Lake Naivasha, Kenya
Azolla has branching rhizomes with adventitious roots and alternate bilobed leaves (Fig. 11). The transparent ventral lobe of the lead helps the plant to float, whereas the dorsal lobe is photosynthetic and contains a cavity with symbiotic cyanobacteria. In mature Azolla plants, the cyanobionts are located in the periphery of the leaf cavity in mucilage between internal and external envelopes (Adams et al. 2013). The adaxial epidermis of the leaf cavity contains a pore with an opening that is larger in younger leaves. The growth of the cyanobionts is coordinated with the growth of the host plant. The apical meristem of each branch contains a colony of undifferentiated cyanobionts. Cyanobacteria from the colony are introduced into the leaf primordium before the development of the leaf cavity is complete. During aging, the cyanobionts show decreases in cell division, increases in the size of vegetative cells, and an increase in heterocyst frequency up to 20–30 % in mature leaves (Adams et al. 2013). Simple hair cells are involved in the transport of sugars (sucrose) from the photosynthetic mesophyll cells of Azolla to the leaf cavity. Also, primary branched hair cells with transfer cell morphology may be involved in nutrient transfer. Ekman et al. (2008) studied the proteomics of the Azolla cyanobiont and found that processes related to energy production, nitrogen and carbon metabolism, and stress-related functions were upregulated compared with free-living cyanobacteria, whereas photosynthesis and metabolic turnover rates were downregulated. In addition to the primary cyanobiont, the leaf cavities of Azolla regularly house diverse communities of other bacteria which may also play a role in the symbiosis (Nierzwicki-Bauer and Aulfinger 1991; Lechno-Yossef and Nierzwicki-Bauer 2002; Zheng et al. 2008).
The Azolla cyanobionts retain their association with the fern throughout its life cycle. The vertical transfer of the cyanobiont to new generations is facilitated by the female reproductive structure (megasporocarp) of the heterosporous fern. During colonization of the megasporocarp, cyanobiont hormogonia enter through pores at the top of the indusium and then differentiate into akinetes in a synchronized manner. The details of this process are described in Zheng et al. (2009). Papaefthimiou et al. (2008b) and Sood et al. (2008) studied cyanobacterial diversity in Azolla and found different cyanobacterial genotypes from different species and also diversity within a single Azolla species. The cyanobacteria differed most markedly between members of the two sections of the genus Azolla.
Ran et al. (2010) provided evidence that there has been selective streamlining of the primary cyanobiont genome which has resulted in an organism devoted to nitrogen fixation and devoid of autonomous growth. This has led to marked loss of function within gene categories for basic metabolic processes such as glycolysis, replication, and nutrient uptake. Phylogenetic analysis grouped the Azolla cyanobiont (Nostoc azollae 0708) with Raphidiopsis and Cylindrospermopsis, which have the smallest known genomes among multicellular cyanobacteria (Stucken et al. 2010). Other molecular characteristics indicate a close phylogenetic relationship to Nostoc and Anabaena strains (Baker et al. 2003; Papaefthimiou et al. 2008b; Ran et al. 2010). For more information on the identity of the Azolla-associated cyanobacteria, the reader is referred to recent reviews by Adams et al. (2013) and Pereira and Vasconcelos (2014).
The Azolla symbiosis is of considerable economic importance (Brouwer et al. 2014). The symbiotic plants are used as a nitrogen-rich biofertilizer especially in rice paddies and/or fertilization of fields. Owing to its high protein content, the plants can also be used as a fodder for pigs, ducks, and other domestic animals. Finally, due to its rapid growth and floating lifestyle, Azolla can also be used to remove nitrates, phosphorous, and heavy metals from polluted water. On the other hand, the mat-forming and easily spreading plants have also become noxious weeds in many tropical and temperate regions (Fig. 11).
Cycad Symbioses
Cycads (Cycadales) are an ancient lineage of evergreen, palmlike gymnosperms that can be traced back to the Paleozoic (Fig. 12). The oldest cycad fossils are from the Pennsylvanian and the general morphology of the group has remained relatively unchanged since then (Taylor et al. 2009). Cycads and Ginkgo diverged in the late Carboniferous or early Permian, and the most recent common ancestor of the living cycads lived in the late Permian. They diversified and became more abundant in the Triassic when primitive seed-plant floras were replaced by conifers and cycads after the Permian–Triassic mass extinction. Cycads reached a diversity peak during the Jurassic and remained relatively stable in terms of diversity during the Cretaceous when the extant lineages were established. Although all extant cycad lineages are ancient, the modern species do not seem to be much older than 12 million years (Nagalingum et al. 2011; Salas-Leiva et al. 2013; Xi et al. 2013; Condamine et al. 2015; Silvestro et al. 2015).
Cycads are insect pollinated and their early diversification may have been linked to the cycad insect pollinator of the order Thysanoptera originating and diversifying in the late Permian. While some cycad–insect interactions may be ancient, this does not seem to be the case for all associations between cycads and beetles (Schneider et al. 2002; Terry et al. 2007; Downie et al. 2008; Peñalver et al. 2012). Dinosaurs have been proposed as key dispersers of cycad seeds during the Mesozoic, and temporal variation in cycad diversity and abundance has been linked to faunal changes. However, when assessing the fossil evidence, Butler et al. (2009) could not find unequivocal support for coevolutionary interactions between cycads and herbivorous dinosaurs.
Extant cycads include ca. 300 species that are classified in 12 genera in three families, widely but patchily distributed in tropical and subtropical regions of the Americas, Africa, Southeast Asia, and Australia (Wang and Ran 2014). Most cycads have a stout trunk with a large crown of tough leaves and can vary in height from 20 cm to almost 20 m at maturity and a thick taproot that can extend many meters into the soil. They also produce lateral roots, some of which develop into specialized coralloid roots that house the cyanobacterial symbionts (Fig. 12). The coralloid roots grow sideways or upward toward the soil surface. The cycad–cyanobacterium symbiosis is still the only known example of a naturally occurring plant root–cyanobacterium symbiosis in plants, and the ability of many cycads to thrive in nutrient-poor soils is generally attributed to the cyanobacterial symbiont (Lindblad 1990, 2009; Costa et al. 1999; Adams et al. 2013).
Symbiotic cyanobacteria are not present in precoralloid roots of cycad seedlings, but their presence is required for further development into coralloid roots (Lindblad 2009; Adams et al. 2013). Precoralloid roots likely release chemicals that induce hormogonium formation in and act as chemoattractants for symbiotic cyanobacteria (Ow et al. 1999). The invasion of plant tissue is thought to occur through apical lenticels and/or injured epidermal cells, and also other soil microbes may be involved in the process (Nathanielsz and Staff 1975; Lobakova et al. 2003; Lindblad 2009). Once inside the root, the cyanobacteria migrate inward through the outer cortex and eventually establish a well-defined zone between the inner and outer cortices of the coralloid root. Specialized plant cells within the cyanobacterial zone facilitate the transfer of nutrients between the symbiotic partners (Nathanielsz and Staff 1975; Ahern and Staff 1994; Lindblad 2009; Adams et al. 2013).
As the coralloid roots of most cycad species are deep beneath the soil surface, the cyanobionts live in complete darkness. However, they retain a full photosynthetic apparatus, associated pigments, and carbon-fixing potential (Lindblad et al. 1985). Nitrogenase activity is several folds higher in cycad-symbiotic Nostoc than in free-living forms and increases with increasing heterocyst frequency, until reaching a maximum at heterocyst frequencies of around 25–35 % (Lindblad et al. 1991). There is a developmental gradient from comparatively low heterocyst frequency in the growing tips of the coralloid roots to very high (up to 46 %) in the older parts of the roots. Many heterocysts in the older parts are inactive and this explains the decrease in nitrogenase activity of aging coralloid roots (see Lindblad 2009 and Adams et al. 2013 for further details).
The cyanobionts of cycads are usually Nostoc, but also Calothrix has sometimes been found. Cycads can house multiple Nostoc genotypes in single plants as well as in single roots, and there seems to be little specificity between cycad species and their cyanobionts (Lindblad et al. 1989; Grobbelaar et al. 1987; Costa et al. 1999, 2004; Zheng et al. 2002; Gehringer et al. 2010; Thajuddi et al. 2010; Yamada et al. 2012). Most symbiotic Nostoc genotypes reported from cycads are not identical to those typically found in thalloid bryophytes and/or lichen-forming fungi and are more closely related to some free-living Nostoc genotypes (Rikkinen 2004; Rikkinen and Virtanen 2008; Gehringer et al. 2010). However, the cyanobionts within cycad roots may differ markedly from those found in the surrounding soil (Cuddy et al. 2012).
Associations Between Angiosperms and Cyanobacteria
As pointed out by Osborne and Bergman (2009), structurally well-defined symbioses between angiosperms and nitrogen-fixing bacteria are relatively uncommon in nature. The Gunnera–Nostoc symbiosis remains the only known symbiosis between angiosperms and cyanobacteria. It differs markedly from other plant–cyanobacterium associations in being a true endosymbiosis with Nostoc cyanobionts housed within the cells of the host plant.
Gunnera Symbiosis
All species of Gunnera establish endosymbiotic associations with Nostoc (Bergman 2002; Osborne and Sprent 2002; Khamar et al. 2010; Fernández-Martínez et al. 2013). The genus includes ca. 60 species of small, medium, or sometimes huge herbs with spiral leaves that have wide blades with prominent palmate venation and toothed margins (Fig. 13). They also have stipule-like “scale leaves” and relatively large and branched inflorescences bearing numerous small and inconspicuous, almost exclusively wind-pollinated flowers. Most species are perennial and some can reach a considerable age; the only annual species is sister to the rest of the genus (Wanntorp et al. 2001, 2003; Wanntorp and Ronse De Craene 2005; Rutishauser et al. 2004).
The order Gunnerales occupies an isolated position among the core eudicots and the divergence from the stem lineage occurred already ca. 115 Mya (Soltis et al. 2003; Magallón and Castillo 2009; Vekemans et al. 2014). In addition to the genus Gunnera (Gunneraceae), the lineage only includes the genus Myrothamnus (Myrothamnaceae) with two species of small xerophytic shrubs in southern Africa and Madagascar (Apg 2016). The natural range of extant Gunnera species includes South America, Africa, and the Australasian region, with some species reaching Hawaii and southern Mexico in the north (Wanntorp and Wanntorp 2003). This range indicates that the plants were widely distributed in Gondwana before its final breakup (Wanntorp and Wanntorp 2003).
Gunnera also has a long fossil history based on its characteristic pollen going back to the Early Cretaceous. Fossil pollen has been identified from all continents of the Southern Hemisphere but also from India and North America, as well as sediments in the Indian and South Atlantic Oceans (Wanntorp et al. 2004a, b). Several species are cultivated as garden ornamentals, and especially Gunnera tinctoria, originally a native of Chile, has naturalized and become invasive in several regions with humid temperate climates, including Ireland, the UK, the Azores, the USA, and New Zealand (Osborne and Sprent 2002; Gioria and Osborne 2009, 2013; Fennell et al. 2013, 2014).
The Nostoc cyanobionts of Gunnera are maintained in the stem cortex of the rhizome of larger species and the stolon of smaller species (Bergman 2002; Osborne and Sprent 2002; Khamar et al. 2010). Special glands are formed on the stem immediately below the leaves. The gland is formed by up to nine papillae surrounding one central papilla. The papillae secrete thick mucilage which induces hormogonium differentiation in Nostoc. Between the papillae, and leading into the stem tissue, are deep channels through which the mucilage is released. The mucilage attracts hormogonia between the papillae and further into the interior of the gland where appropriate Nostoc genotypes are taken into plant cells (Söderbäck and Bergman 1993; Johansson and Bergman 1994; Rasmussen et al. 1994, 1996; Uheda and Silvester 2001).
Within Gunnera cells, the Nostoc hormogonia develop into filaments with heterocysts . The cyanobiont proliferates and the infected plant cells divide repeatedly to form internal colonies within the stem cortex. The glands are the only known entry point for Nostoc hormogonia into the cortical tissue of Gunnera. Along the rhizomes, the cyanobionts occur as well-defined colonies and show different stages of development, indicating that they were each formed through successive and separate infection processes involving different glands. Within the plant cells, the cyanobionts are surrounded by the host cell plasmalemma and the membrane acts as the interface through which the exchange of metabolites takes place. Although only young glands can incorporate Nostoc hormogonia, new glands continue to develop at the base of each new leaf when the stem grows. Gland development in Gunnera also takes place in the absence of symbiotic cyanobacteria, but nitrogen limitation seems to be a prerequisite for their development (Bergman 2002; Wang et al. 2004; Chiu et al. 2005; Osborne and Bergman 2009; Khamar et al. 2010; Adams et al. 2013).
Molecular studies have confirmed that the cyanobionts of Gunnera belong to Nostoc and are closely related to some symbiotic and free-living genotypes of the genus (Rasmussen and Svenning 2001; Svenning et al. 2005; Papaefthimiou et al. 2008a). There is considerable phenotypic and genotypic variation among the cyanobionts (Bergman et al. 1992). Several different Nostoc genotypes may sometimes be present in a single Gunnera plant (Nilsson et al. 2000), but in most cases, each plant has housed only one Nostoc genotype (Guevara et al. 2002). Fernández-Martínez et al. (2013) found no genetic variability among Nostoc cyanobionts within single Gunnera plants, while the cyanobionts of neighboring plants could be markedly different. The Nostoc genotypes were closely related to those found in terricolous cyanolichens and bryophytes. Nostoc isolates from cycads and bryophytes also readily invade Gunnera cells and vice versa (Adams et al. 2013).
The evolutionary origin of the Gunnera glands is a mystery. The bright red glands form at right angles to successive petioles and are clearly visible at the developing cotyledon (for images, see Osborne and Bergman 2009 and Adams et al. 2013). The gland originates internally from the lateral meristem and eventually ruptures the epidermis, exposing the surface of epidermal cells. Osborne and Bergman (2009) pointed out that at later stages of development, the glands are often closely associated with adventitious root formation. The glands in young seedlings are initiated prior to shoot emergence and are evident as a dome-shaped structure with red (anthocyanin) pigmentation. A similar pigmentation is also present in the tips of developing adventitious roots, suggesting that the glands might evolve as highly modified adventitious roots. In any case, the specialized morphology and sugar accumulation in the glands of Gunnera seedlings are crucial for early stages in the establishment of the Nostoc endosymbiosis.
Khamar et al. (2010) investigated changes in the carbohydrate metabolism during Gunnera gland development and discovered that plant cells within the mature glands accumulated high levels of soluble sugars (including high concentration of glucose and fructose and lower concentration of sucrose) prior to the arrival of cyanobacteria and that the various sugars affected the Nostoc cyanobionts’ ability to form motile hormogonia. They concluded that in a nitrogen-limiting environment, Gunnera glands secrete mucilage with minute of soluble sugars, which attract Nostoc to the gland surface but does not interfere with hormogonium formation. The hormogonia penetrate into the gland and enter cells at the base of the gland presumably because they are attracted by the high levels of glucose and fructose inside the cells, while within the plant cells, further hormogonia are not formed because of the high levels of soluble sugars (Khamar et al. 2010).
The accumulation of soluble sugars in the cells of mature but still noninfected Gunnera gland indicates that the nonsugar-secreting structure could have originally evolved from a sugar-secreting gland functionally similar but not necessarily homologous to extrafloral (EF) nectaries of other eudicots (Marazzi et al. 2013). Also the conspicuous red pigmentation of the glands in Gunnera seedlings might play a role in attracting flying insects capable of vectoring symbiotically compatible Nostoc genotypes from the decomposing old rhizomes of well-established Gunnera colonies to the germinating seedlings. While the mucilage secreted by the glands largely consists of nondigestible carbohydrates (Rasmussen et al. 1994), with barely detectible levels of soluble sugars (Khamar et al. 2010), the gland tissue itself is rich in soluble sugars that may well attract insects. The possible role of insect vectors in the dispersal ecology of the Nostoc–Gunnera symbiosis should be elucidated in field studies within the natural range of Gunnera species.
It is possible that the original function of Gunnera glands could have been comparable to those of EF nectaries that are produced on the leaves, stems, and virtually any other parts of thousands of plant species. These structures have been described for nearly 4000 vascular plant species representing nearly 750 genera in over 100 families (Weber and Keeler 2013). Their primary function is to attract aggressive and often mutualistic insects, particularly ants which, while foraging for nectar, protect the host plant from herbivorous animals (Heil et al. 2004; Villamil et al. 2013; Weber and Agrawal 2014). Besides ants, the EF nectaries of different plants attract a wide variety of other arthropods including many Diptera. Some of them also provide visual cues for foraging arthropods and also the emission of odors can attract mutualistic insects (Koptur 1992).
Weber and Keeler (2013) estimated that extrafloral nectaries have evolved independently at least 457 times in vascular plants. Even among the ferns, extrasoral nectaries have arisen independently several times, and they first evolved long before floral nectar and long before ants appeared in the fossil record (Marazzi et al. 2012; Koptur et al. 2013). Therefore, their original function could not have involved ants or other social hymenopterans. Whatever the primary role, the sugar secreted has impacted the diversification of several groups on arthropods and fungi. For example, several lineages of sooty molds have specialized to use plant exudates and insect honey dew for nutrition (Rikkinen et al. 2003; Schmidt et al. 2014).
According to the EF nectary hypothesis, the secretions of ancestral Gunnera glands would have been involved in an indirect anti-herbivore defense by attracting and hence increasing the presence of putative plant defenders on the plant stem. Thus, the mutualism between the ancestral Gunnera species and the putative (facultative) insect associates would represent the original state, whereas the association between the plant and Nostoc would have derived later and then evolved into an ecologically obligate cyanobacterial symbiosis.
Interestingly, Campbell et al. (2013) reported that some epiphytic cyanolichens on conifers may utilize glucose excreted by the EF nectaries of neighboring angiosperms. They proposed that the exogenous source of carbon could help to sustain the lichen mycobionts during drought-induced periods of inactivity of their cyanobacterial symbionts.
Other Angiosperms
Many angiosperms regularly house epiphytic cyanobacteria on their stems, leaves, or root surfaces. While most of these associations are probably accidental and of no particular significance to the plants, in some cases, N2 fixation of associated cyanobacteria might be significant. For example, cyanobacteria including Nostoc and Calothrix are commonly seen on the exposed roots of tropical epiphytic orchids (Fig. 14), and sometimes cyanobacteria and associated fungi can form sheaths around aerial roots (Tsavkelova et al. 2003). As the cyanobacteria are capable of N2 fixation, they may benefit the plant and/or the endomycorrhizal fungi that live within the specialized velamen of the aerial roots (Teixeira da Silva et al. 2015).
Cyanobacterial associations of tracheophytes . A Heterocystous cyanobacteria and other N2-fixing prokaryotes often grow on and within the aerial roots of tropical orchids, with presumed benefits to the plants or their mycorrhizal fungi. B Nostoc hormogonia are attracted to nitrogen-deprived vascular plants. Here lichen-symbiotic Nostoc isolated from a Peltigera species and cultured on nitrogen-deficient medium (Z8) form an artificial association with a somatic embryo plant of Scots pine (Pinus sylvestris)
Establishment of artificial associations between nitrogen-fixing microorganisms and agricultural crop plants could potentially reduce the demand for chemically produced nitrogen fertilizers (Gusev et al. 2002; Prasanna et al. 2009). Nostoc hormogonia are known to be attracted to root extracts of many nitrogen-deprived vascular plants (Fig. 14), including rice and wheat (Gantar et al. 1995; Nilsson et al. 2002, 2005). Floating cyanobacterial communities on the soil–water surface are widely used in agriculture as nitrogen-supplementing biofertilizers in rice paddy fields, especially in Southeast Asia, and nitrogen accretions of 25–30 kg N/ha have been attributed to their N2 fixation (Prasanna et al. 2009).
Early Evolution of Terrestrial Symbiotic Systems
Through endosymbiosis , certain cyanobacteria evolved into the plastids of photoautotrophic eukaryotes some 900 million years ago (Deusch et al. 2008; Dagan et al. 2013; Shih and Matzke 2013; Ochoa de Alda et al. 2014; Ruhfel et al. 2014; Ku et al. 2015). Already much earlier cyanobacteria evolved oxygenic photosynthesis and changed the Earth’s atmosphere from anoxic to oxic. As nitrogenase is highly sensitive to oxygen, most N2-fixing prokaryotes became confined to anoxic environmental niches. Cyanobacteria themselves evolved several strategies to protect nitrogenase from oxygen, including a temporal separation of oxygenic photosynthesis and N2 fixation in unicellular forms and the differentiation of a specialized cell, the heterocyst, in some filamentous forms. The sensitivity of nitrogenase to oxygen may also have enhanced the early evolution of cyanobacterial symbioses, as many symbiotic structures represent micro-aerobic environments that help to protect cyanobacterial cells from atmospheric oxygen (Rikkinen 2002).
It is possible that the evolution of some cyanobacterial symbioses correlated temporally with geological periods of increased oxygen in the atmosphere. High atmospheric oxygen levels (over 30 %) were experienced during the Carboniferous, and this may have seriously interfered with nitrogen fixation and given adaptive value to any structures and mechanisms that helped to isolate N2-fixing cells from the atmosphere. Hence the early evolution of some symbiotic structures, like the coralloid roots of cycads, could parallel the development of arthropod and amphibian gigantism, which was directly facilitated by the hyperoxic atmosphere (Payne et al. 2009, 2011, 2012). Unfortunately, no fossilized roots of early cycads have yet been identified (Taylor et al. 2009).
The mycelia of mycorrhizal fungi associate with soil bacteria, many of which are capable of N2 fixation (Scheublin et al. 2010; Valverde et al. 2016). Such associations probably experienced a triumph in the late Permian, when soil bacteria, following the atmospheric transition to hypoxia, became more efficient in nitrogen fixation. While giant amphibians and arthropods disappeared, the cyanobacterial symbioses did not and the reduction in atmospheric O2 levels did not directly threaten them. However, the competitive edge and ecological significance of cyanobacterial N2 fixation may have started to dwindle when diffuse rhizosphere associations with soil bacteria became a more viable option for nitrogen-starved plants (Rikkinen 2002). Rhizosphere associations between fungi and bacteria must have promoted the evolution of mycorrhizal symbioses and the subsequent diversification of seed plants. The radiation of modern ferns, bryophytes, fungal endophytes, and lichens was in turn facilitated by the diversification of seed plants (Schneider et al. 2004; Schuettpelz and Pryer 2009; Feldberg et al. 2014; Li et al. 2014; Lóriga et al. 2014; Sundue et al. 2016).
Thalloid liverworts, hornworts, and cycads together represent a rather conspicuous proportion of all extant plant lineages that can be reliably traced back to pre-Permian times. Also Azolla and Gunnera can be traced back to the Cretaceous (Taylor et al. 2009). Also some lineages of extant cyanobacterial lichens are probably ancient. The oldest fossil of a stratified cyanolichen is from the Lower Devonian (Honegger et al. 2012). While very few Paleozoic or Mesozoic lichen fossils have so far been found, many amber fossils demonstrate that many extant genera of lichen-forming fungi had already evolved by the Tertiary (Rikkinen and Poinar 2002, 2008; Kaasalainen et al. 2015; Lumbsch and Rikkinen 2016). The initial diversification of the Pezizomycotina (Ascomycota) occurred already in the Ordovician, followed by repeated splits of lineages throughout the Phanerozoic (Beimforde et al. 2014).
The earliest fungal symbioses may have in many respects resembled some extant crustose cyanolichens, especially salt-tolerant forms that live on rocky ocean shores. Littoral habitats along ancient shorelines brought cyanobacteria, green algae, and fungi into close contact under conditions where there were both a need and opportunity for establishing new symbiotic interactions (Rikkinen 2002; Edwards and Kenrick 2015). The earliest lichen-like symbioses probably evolved long before the initial evolution of mycorrhizal symbioses, the subsequent rise of vascular plants, and the later diversification of parasitic and saprophytic fungi. Accordingly, some modern cyanolichens may preserve biological features that closely mirror those that evolved in similar cyanobacterial symbioses at early stages of terrestrial evolution.
The fact that most vascular plants, liverworts, and hornworts include species that form symbioses with AM fungi indicates that also these symbioses evolved during very early stages in the evolution of land plants. The AM fungi have a definite fossil record extending to the early Devonian ca. 400 Mya, which is before early vascular plants had acquired roots (Kenrick and Strullu-Derrien 2014; Strullu-Derrien et al. 2014), and the oldest fossil spores of possible AM fungi date back to the Ordovician ca. 460 Mya (Taylor et al. 2015b). Molecular data indicate that glomeromycetes have always been symbiotic and suggest that the first mycorrhizae may have involved mucoromycetes, the sister lineage glomeromycetes (Bidartondo et al. 2011; Strullu-Derrien et al. 2014). The mycobiont of an early lichen-like symbiosis from the Early Devonian with nonseptate hyphae may have belonged to one of these groups (Taylor et al. 1997, 2015b), and already the common ancestor of Glomeromycota and Mucoromycota was probably capable of establishing symbioses with cyanobacteria and/or early photosynthetic eukaryotes (Delaux et al. 2013, 2015; Field et al. 2015a, b; Tang et al. 2016).
The Geosiphon pyriformis –Nostoc endosymbiosis may represent one living example of early symbioses between glomeromycetes, cyanobacteria, and bryophytes (Schüßler and Walker 2011; Schüßler 2012). Such symbioses could be seen as “evolutionary precursors” to more modern types of AM symbioses (Schüßler and Walker 2011; Selosse et al. 2015; Wojtczak and Janik 2016). It is noteworthy that a symbiotic cyanobacterium has been identified from the cortex of Aglaophyton major, one of the earliest early land plants in the Devonian (Taylor and Krings 2005). Somewhat resembling the situation seen in modern hornworts, the filamentous cyanobacterium seems to have entered its host via stomatal pores, colonized the substomatal chambers, and then spread throughout the outer cortical tissue, where it has been preserved in fossil specimens.
Concluding Remarks
Symbiotic interactions between cyanobacteria, plants, and fungi have evolved repeatedly during the course of evolution. A trend toward versatile and diffuse associations with non-photosynthetic bacteria may have reduced the relative importance of cyanobacterial symbioses during fungal and vascular plant evolution. However, cyanolichens are still an important component of biodiversity in many terrestrial ecosystems and have provided unique opportunities for the diversification of both symbiotic cyanobacteria and fungi. During their evolution, many lichen-forming fungi may have established associations with plant-symbiotic Nostoc genotypes and/or vice versa, leading to repeated cyanobiont switches between terricolous lichens, thalloid bryophytes, cycads, and/or Gunnera. Likely these cyanobacterial symbioses have also had many diffuse effects on mycorrhizal and plant–insect mutualisms. Clearly we are only beginning to unravel the complex network of biological interactions and evolutionary processes in which symbiotic cyanobacteria have evolved. DNA techniques offer accurate methods for studying the genetic diversity of symbiotic cyanobacteria in situ and for making comparisons between lichen cyanobionts and their nonsymbiotic relatives. In the future, we can expect many interesting new findings from previously ignored cyanobiont lineages and unexplored ecological settings. They can add significantly to what we already know about symbiotic cyanobacteria and their diverse hosts.
References
Adams DG (2002) Cyanobacteria in symbiosis with hornworts and liverworts. In: Rai AN, Bergman B, Rasmussen U (eds) Cyanobacteria in symbiosis. Kluwer, Dordrecht, pp 117–135
Adams DG, Duggan PS (2008) Cyanobacteria-bryophyte symbioses. J Exp Bot 59:1047–1058. doi:10.1093/jxb/ern005
Adams D, Bergman B, Nierzwicki-Bauer S et al (2013) Cyanobacterial-plant symbioses. In: Rosenberg E, DeLong E, Lory S et al (eds) The prokaryotes. Springer, Berlin, pp 359–400
Ahern CP, Staff IA (1994) Symbiosis in cycads: the origin and development of coralloid roots in Macrozamia communis (Cycadaceae). Am J Bot 81:1559–1570
Apg IV (2016) An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot J Linn Soc 181:1–80
Aptroot A, Schumm F (2009) Chimeras occur on the pantropical lichinomycete Phyllopeltula corticola. Lichenologist 42:307–310. doi:10.1017/S0024282909990351
Arnold AE, Miadlikowska J, Higgins KL et al (2009) A phylogenetic estimation of trophic transition networks for ascomycetous fungi: are lichens cradles of symbiotrophic fungal diversification? Syst Biol 58:283–297. doi:10.1093/sysbio/syp001
Arróniz-Crespo M, Pérez-Ortega S, De Los RA et al (2014) Bryophyte-cyanobacteria associations during primary succession in recently deglaciated areas of Tierra del Fuego (Chile). PLoS One 9(5):e96081. doi:10.1371/journal.pone.0096081
Aschenbrenner IA, Cernava T, Berg G et al (2016) Understanding microbial multi-species symbioses. Front Microbiol 7:180. doi:10.3389/fmicb.2016.00180
Baker JA, Entsch B, McKay DB (2003) The cyanobiont in an Azolla fern is neither Anabaena nor Nostoc. FEMS Microbiol Lett 229:43–47. doi:10.1016/S0378-1097(03)00784-5
Barthlott W, Fischer E, Frahm J-P (2000) First experimental evidence for zoophagy in the hepatic Colura. Plant Biol 2:93–97. doi:10.1055/s-2000-9150
Bay G, Nahar N, Oubre M, Whitehouse MJ et al (2013) Boreal feather mosses secrete chemical signals to gain nitrogen. New Phytol 200:54–60. doi:10.1111/nph.12403
Beimforde C, Feldberg K, Nylinder S et al (2014) Estimating the Phanerozoic history of the Ascomycota lineages: combining fossil and molecular data. Mol Phylogenet Evol 78:386–398. doi:10.1016/j.ympev.2014.04.024
Belinchón R, Yahr R, Ellis CJ (2014) Interactions among species with contrasting dispersal modes explain distributions for epiphytic lichens. Ecography 37:001–007. doi:10.1111/ecog.01258
Berg A, Danielsson A, Svensson BH (2013) Transfer of fixed-N from N2-fixing cyanobacteria associated with the moss Sphagnum riparium results in enhanced growth of the moss. Plant Soil 362:271–278. doi:10.1007/s11104-012-1278-4
Bergman B (2002) Nostoc-Gunnera symbiosis. In: Rai AN, Bergman B, Rasmussen U (eds) Cyanobacteria in symbiosis. Kluwer, Dordrecht, pp 207–232
Bergman B, Hällbom L (1982) Nostoc of Peltigera canina when lichenized and isolated. Can J Bot 60:2092–2098
Bergman B, Johansson C, Söderbäck E (1992) The Nostoc-Gunnera symbiosis. New Phytol 122:379–400
Bidartondo MI, Read DJ, Trappe JM et al (2011) The dawn of symbiosis between plants and fungi. Biol Lett 7:574–577. doi:10.1098/rsbl
Bjerke JW, Gwynn-Jones D, Callaghan TV (2005) Effects of enhanced UV-B radiation in the field on the concentration of phenolics and chlorophyll fluorescence in two boreal and arctic-alpine lichens. Environ Exp Bot 53:139–149. doi:10.1016/j.envexpbot.2004.03.009
Bothe H, Schmitz O, Yates MG et al (2010) Nitrogen fixation and hydrogen metabolism in cyanobacteria. Microbiol Mol Biol Rev 74:529–551. doi:10.1128/MMBR.00033-10
Brouwer P, Bräutigam A, Külahoglu C et al (2014) Azolla domestication towards a biobased economy? New Phytol 202:1069–1082. doi:10.1111/nph.12708
Brown RC, Lemmon BE, Shimamura M et al (2015) Spores of relictual bryophytes: diverse adaptations to life on land. Rev Palaeobot Palynol 216:1–17. doi:10.1016/j.revpalbo.2015.01.004
Butler RJ, Barrett PM, Kenrick P et al (2009) Testing co-evolutionary hypotheses over geological timescales: interactions between Mesozoic non-avian dinosaurs and cycads. Biol Rev Camb Philos Soc 84:73–89. doi:10.1111/j.1469-185X.2008.00065.x
Calteau A, Fewer DP, Latifi A et al (2014) Phylum-wide comparative genomics unravel the diversity of secondary metabolism in Cyanobacteria. BMC Genomics 15:977. doi:10.1186/1471-2164-15-977
Campbell EL, Meeks JC (1989) Characteristics of hormogonia formation by symbiotic Nostoc spp. in response to the presence of Anthoceros punctatus or its extracellular products. Appl Environ Microbiol 55:125–131
Campbell J, Bengtson P, Fredeen AL et al (2013) Does exogenous carbon extend the realized niche of canopy lichens? Evidence from sub-boreal forests in British Columbia. Ecology 94:1186–1195. doi:10.2307/23435960
Casano LM, del Campo EM, García-Breijo FJ et al (2011) Two Trebouxia algae with different physiological performances are ever-present in lichen thalli of Ramalina farinacea. Coexistence versus competition? Environ Microbiol 13:806–818. doi:10.1111/j.1462-2920.2010.02386.x
Chang Y, Graham SW (2011) Inferring the higher-order phylogeny of mosses (Bryophyta) and relatives using a large, multigene plastid data set. Am J Bot 98:839–849. doi:10.3732/ajb.0900384
Chiu WL, Peters GA, Levieille G et al (2005) Nitrogen deprivation stimulates symbiotic gland development in Gunnera manicata. Plant Physiol 139:224–230. doi:10.1104/pp.105.064931
Chua JPS, Wallace EJS, Yardley JA et al (2012) Gene expression indicates a zone of heterocyst differentiation within the thallus of the cyanolichen Pseudocyphellaria crocata. New Phytol 196:862–872. doi:10.1111/j.1469-8137.2012.04272.x
Claessen D, Rozen DE, Kuipers OP et al (2014) Bacterial solutions to multicellularity: a tale of biofilms, filaments and fruiting bodies. Nat Rev Microbiol 12:115–124. doi:10.1038/nrmicro3178
Collison ME (1980) A new multiple-floated Azolla from the Eocene of Britain with a brief review of the genus. Paleontology 23:213–229
Condamine FL, Nagalingum NS, Marshall CR, Morlon H (2015) Origin and diversification of living cycads: a cautionary tale on the impact of the branching process prior in Bayesian molecular dating. BMC Evol Biol 15:65. doi:10.1186/s12862-015-0347-8
Cornejo C, Scheidegger C (2013) Morphological aspects associated with repair and regeneration in Lobaria pulmonaria and L. amplissima (Peltigerales, Ascomycota). Lichenologist 45:285–289. doi:10.1017/S0024282912000813
Cornejo C, Scheidegger C (2016) Cyanobacterial gardens: the liverwort Frullania asagrayana acts as a reservoir of lichen photobionts. Environ Microbiol Rep. doi:10.1111/1758-2229.12386 [Epub ahead of print]
Cornelissen JHC, Lang MH, Soudzilovskaia NA et al (2007) Comparative cryptogam ecology: a review of bryophyte and lichen traits that drive biogeochemistry. Ann Bot 99:987–1001. doi:10.1093/aob/mcm030
Costa JL, Paulsrud P, Lindblad P (1999) Cyanobiont diversity within coralloid roots of selected cycad species. FEMS Microbiol Ecol 28:85–91. doi:10.1016/S0168-6496(98)00095-6
Costa J-L, Paulsrud P, Rikkinen J et al (2001) Genetic dversity of Nostoc symbionts endophytically associated with two bryophyte species. Appl Environ Microbiol 67:4393–4396. doi:10.1128/AEM.67.9.4393-4396.2001
Costa JL, Romero EM, Lindblad P (2004) Sequence based data supports a single Nostoc strain in individual coralloid roots of cycads. FEMS Microbiol Ecol 49:481–487. doi:10.1016/j.femsec.2004.05.001
Cox CJ, Goffinet B, Wickett NJ et al (2010) Moss diversity: a molecular phylogenetic analysis of genera. Phytotaxa 9:175–195
Cox CJ, Li B, Foster PG et al (2014) Conflicting phylogenies for early land plants are caused by composition biases among synonymous substitutions. Syst Biol 63:272–279. doi:10.3732/ajb.0900384
Cuddy WS, Neilan BA, Gehringer MM (2012) Comparative analysis of cyanobacteria in the rhizosphere and as endosymbionts of cycads in drought-affected soils. FEMS Microbiol Ecol 80:204–215. doi:10.1111/j.1574-6941.2011.01288.x
Dagan T, Roettger M, Stucken K et al (2013) Genomes of Stigonematalean cyanobacteria (subsection V) and the evolution of oxygenic photosynthesis from prokaryotes to plastids. Genome Biol Evol 5:31–44. doi:10.1093/gbe/evs117
Dal Grande F, Beck A, Cornejo C et al (2014) Molecular phylogeny and symbiotic selectivity of the green algal genus Dictyochloropsis s.l. (Trebouxiophyceae): a polyphyletic and widespread group forming photobiont-mediated guilds in the lichen family Lobariaceae. New Phytol 202:447–455. doi:10.1111/nph.12678
Dal-Forno M, Lawrey JD, Sikaroodi M et al (2013) Starting from scratch, evolution of the lichen thallus in the basidiolichens Dictyonema (Agaricales, Hygrophoraceae). Fungal Biol 117:584–598. doi:10.1016/j.funbio.2013.05.006
Dal-Forno M, Lücking R, Bungartz F et al (2016) From one to six: unrecognized species diversity in the genus Acantholichen (lichenized Basidiomycota: Hygrophoraceae). Mycologia 108:38–55. doi:10.3852/15-060
Dalton DA, Chatfield JM (1987) A new nitrogen-fixing cyanophyte–hepatic association: Nostoc and Porella. Am J Bot 72:781–784
Darnajoux R, Constantin J, Miadlikowksa J et al (2014) Is vanadium a biometal for boreal cyanolichens? New Phytol 202:765–771. doi:10.1111/nph.12777
Davey ML, Currah RS (2006) Interactions between mosses (Bryophyta) and fungi. Botany 84:1509–1519. doi:10.1139/B10-004
Delaux PM, Séjalon-Delmas N, Bécard G et al (2013) Evolution of the plant-microbe symbiotic ‘toolkit’. Trends Plant Sci 18:298–304. doi:10.1016/j.tplants.2013.01.008
Delaux PM, Radhakrishnan J et al (2015) Algal ancestor of land plants was preadapted for symbiosis. Proc Natl Acad Sci U S A 112:13390–13395. doi:10.1073/pnas.1515426112
Desirò A, Duckett JG, Pressel S et al (2013a) Fungal symbioses in hornworts: a chequered history. Proc R Soc B 280:20130207. doi:10.1098/rspb.2013.0207
Desirò A, Naumann M, Epis S et al (2013b) Mollicutes-related endobacteria thrive inside liverwort-associated arbuscular mycorrhizal fungi. Environ Microbiol 15:822–836. doi:10.1111/j.1462-2920.2012.02833.x
Desirò A, Faccio A, Kaech A et al (2015) Endogone, one of the oldest plant-associated fungi, host unique Mollicutes-related endobacteria. New Phytol 205:1464–1472. doi:10.1111/nph.13136
Deusch O, Landan G, Roettger M et al (2008) Genes of cyanobacterial origin in plant nuclear genomes point to a heterocyst-forming plastid ancestor. Mol Biol Evol 25:748–761. doi:10.1093/molbev/msn022
Díaz EM, Sacristan M, Legaz ME et al (2009) Isolation and characterization of a cyanobacterium-binding protein and its cell wall receptor in the lichen Peltigera canina. Plant Signal Behav 4:598–603. doi:10.4161/psb.4.7.9164
Díaz EM, Vicente-Manzanares M, Sacristan M et al (2011) Fungal lectin of Peltigera canina induces chemotropism of compatible Nostoc cells by constriction-relaxation pulses of cyanobiont cytoskeleton. Plant Signal Behav 6:1525–1536. doi:10.4161/psb.6.10.16687
Diederich P (1996) The lichenicolous heterobasidiomycetes. Bibl Lichenol 61:1–198
Dittmann E, Gugger M, Sivonen K et al (2015) Natural product biosynthetic diversity and comparative genomics of the cyanobacteria. Trends Microbiol 23(10):642–652. doi:10.1016/j.tim.2015.07.008
Döbbeler P (1997) Biodiversity of bryophilous ascomycetes. Biodivers Conserv 6:721–738. doi:10.1023/A:1018370304090
Dodds WK, Gudder DA, Mollenhauer D (1995) The ecology of Nostoc. J Phycol 31:2–18
Downie DA, Donaldson JS, Oberprieler RG (2008) Molecular systematics and evolution in an African cycad-weevil interaction: Amorphocerini (Coleoptera: Curculionidae: Molytinae) weevils on Encephalartos. Mol Phylogenet Evol 47:102–116. doi:10.1016/j.ympev.2008.01.023
Duckett JG, Prasad AKSK, Davies DA et al (1977) A cytological analysis of the Nostoc–bryophyte relationship. New Phytol 79:349–362. doi:10.1111/j.1469-8137.1977.tb02215.x
Edwards D, Kenrick P (2015) The early evolution of land plants, from fossils to genomics: a commentary on Lang (1937) ‘On the plant-remains from the Downtonian of England and Wales’. Philos Trans R Soc Lond B Biol Sci 370. doi:10.1098/rstb.2014.0343
Edwards D, Morris JL, Richardson JB et al (2014) Cryptospores and cryptophytes reveal hidden diversity in early land floras. New Phytol 202:50–78. doi:10.1111/nph.12645
Ekman M, Tollbäck P, Bergman B (2008) Proteomic analysis of the cyanobacterium of the Azolla symbiosis: identity, adaptation, and NifH modification. J Exp Bot 59:1023–1034. doi:10.1093/jxb/erm282
Ekman M, Picossi S, Campbell EL et al (2013) A Nostoc punctiforme sugar transporter necessary to establish a cyanobacterium-plant symbiosis. Plant Physiol 161:1984–1992. doi:10.1104/pp.112.213116
Ekman S, Wedin M, Lindblom L et al (2014) Extended phylogeny and a revised generic classification of the Pannariaceae (Peltigerales, Ascomycota). Lichenologist 46:627–656. doi:10.1017/S002428291400019X
Elbert W, Weber B, Burrows S et al (2012) Contribution of cryptogamic covers to the global cycles of carbon and nitrogen. Nat Geosci 5:459–462. doi:10.1038/ngeo1486
Ellerbeck M, Schüßler A, Brucker D et al (2013) Characterization of three ammonium transporters of the glomeromycotan fungus Geosiphon pyriformis. Eukaryot Cell 12:1554–1562. doi:10.1128/EC.00139-13
Evrard C, Van Hove C (2004) Taxonomy of the American Azolla species (Azollaceae): a critical review. Syst Geogr Plant 74:301–318
Fedrowitz K, Kaasalainen U, Rikkinen J (2011) Genotype variability of Nostoc symbionts in three epiphytic Nephroma species in a boreal forest landscape. Bryologist 114:220–230. doi:10.1639/0007-2745-114.1.220
Fedrowitz K, Kaasalainen U, Rikkinen J (2012) Geographic mosaic of symbiont selectivity in a genus of epiphytic cyanolichens. Ecol Evol 2:2291–2303. doi:10.1002/ece3.343
Feldberg K, Schneider H2, Stadler T et al (2014) Epiphytic leafy liverworts diversified in angiosperm-dominated forests. Sci Rep 4:5974. doi:10.1038/srep05974
Fennell M, Murphy JE, Gallagher T et al (2013) Simulating the effects of climate change on the distribution of an invasive plant, using a high resolution, local scale, mechanistic approach: challenges and insights. Glob Chang Biol 19:1262–1274. doi:10.1111/gcb.12102
Fennell M, Gallagher T, Vintro LL et al (2014) Using soil seed banks to assess temporal patterns of genetic variation in invasive plant populations. Ecol Evol 4:1648–1658. doi:10.1002/ece3.1043
Fernández-Martínez MA, de Los RA, Sancho LG et al (2013) Diversity of endosymbiotic Nostoc in Gunnera magellanica from Tierra del Fuego, Chile. Microb Ecol 66:335–350. doi:10.1007/s00248-013-0223-2
Field KJ, Rimington WR, Bidartondo MI (2015a) First evidence of mutualism between ancient plant lineages (Haplomitriopsida liverworts) and Mucoromycotina fungi and its response to simulated Palaeozoic changes in atmospheric CO2. New Phytol 205:743–756. doi:10.1111/nph.13024
Field KJ, Rimington WR, Bidartondo MI (2015b) Functional analysis of liverworts in dual symbiosis with Glomeromycota and Mucoromycotina fungi under a simulated Palaeozoic CO2 decline. ISME J. doi:10.1038/ismej.2015.204
Flores E, Herrero A (2010) Compartmentalized function through cell differentiation in filamentous cyanobacteria. Nat Rev Microbiol 8:39–50. doi:10.1038/nrmicro2242
Gantar M, Rowell P, Kerby NW, Sutherland IW (1995) Role of extracellular polysaccharides in the colonization of wheat (Triticum vulgare L.) roots by N2-fixing cyanobacteria. Biol Fert Soils 19:41–48. doi:10.1007/BF00336345
Gauslaa Y (2014) Rain, dew, and humid air as drivers of morphology, function and spatial distribution in epiphytic lichens. Lichenologist 46:1–16. doi:10.1017/S0024282913000753
Gavazov KS, Soudzilovskaia NA, van Logtestijn RSP et al (2010) Isotopic analysis of cyanobacterial nitrogen fixation associated with subarctic lichen and bryophyte species. Plant Soil 333:507–517. doi:10.1007/s11104-010-0374-6
Gehringer MM, Pengelly JJ, Cuddy WS et al (2010) Host selection of symbiotic cyanobacteria in 31 species of the Australian cycad genus: Macrozamia (Zamiaceae). Mol Plant-Microbe Interact 23:811–822. doi:10.1094/MPMI-23-6-0811
Gehringer MM, Adler L, Roberts AA et al (2012) Nodularin, a cyanobacterial toxin, is synthesized in planta by symbiotic Nostoc sp. ISME J 6:1834–1847. doi:10.1038/ismej.2012.25
Gentili F, Nilsson M-C, Zackrisson O et al (2005) Physiological and molecular diversity of feather moss associative N2-fixing cyanobacteria. J Exp Bot 56:3121–3127. doi:10.1093/jxb/eri309
Gerphagnon M, Latour D, Colombet J, Sime-Ngando T (2013) Fungal parasitism: life cycle, dynamics and impact on cyanobacterial blooms. PLoS One 8(4), e60894. doi:10.1371/journal.pone.0060894
Gioria M, Osborne BA (2009) The impact of Gunnera tinctoria (Molina) Mirbel invasions on soil seed bank communities. J Plant Ecol 2:153–167. doi:10.1093/jpe/rtp013
Gioria M, Osborne BA (2013) Biological flora of the British Isles: Gunnera tinctoria. J Ecol 101:243–264. doi:10.1111/1365-2745.12022
Green TGA, Nash TH III, Lange OL (2008) Physiological ecology of carbon dioxide exchange. In: Nash TH III (ed) Lichen biology, 2nd edn. Cambridge University Press, Cambridge, pp 152–181
Grobbelaar N, Scott WE, Hattingh W, Marshall J (1987) The identification of the coralloid endophytes of the southern African cycads and the ability of the isolates to fix dinitrogen. S Afr J Bot 53:111–118
Groth-Malonek M, Pruchner D, Grewe F et al (2005) Ancestors of trans-splicing mitochondrial introns support serial sister group relationships of hornworts and mosses with vascular plants. Mol Biol Evol 22:117–125. doi:10.1093/molbev/msh259
Grube M, Cardinale M, de Castro JV et al (2009) Species-specific structural and functional diversity of bacterial communities in lichen symbioses. ISME J 3:1105–1115. doi:10.1038/ismej.2009.63
Grube M, Berg G, Andrésson ÓS et al (2014) Lichen genomics: prospects and progress. In: Martin F (ed) The ecological genetics of fungi. Wiley, pp 191–212. doi:10.1002/9781118735893.ch9
Guevara R, Armesto JJ, Caru M (2002) Genetic diversity of Nostoc microsymbionts from Gunnera tinctoria revealed by PCR-STRR fingerprinting. Microb Ecol 44:127–136. doi:10.1007/s00248-002-1019-y
Gusev MV, Baulina OI, Gorelova OA, Lobakova ES, Korzhenevskaya TG (2002) Artificial cyanobacterial–plant symbioses In: Rai AN, Bergman B, Rasmussen U (eds) Cyanobacteria in Symbiosis. Kluwer, Dordrecht, pp 253–312. doi:10.1007/0-306-48005-0_14
Hall JW (1974) Cretaceous salviniaceae. Ann Miss Bot Gard 61:354–367
Heil M, Greiner S, Meimberg H (2004) Evolutionary change from induced to constitutive expression of an indirect plant resistance. Nature 430:205–208. doi:10.1038/nature02703
Henskens FL, Green TGA, Wilkins A (2012) Cyanolichens can have both cyanobacteria and green algae in a common layer as major contributors to photosynthesis. Ann Bot 110:555–563. doi:10.1093/aob/mcs108
Higgins KL, Arnold AE, Miadlikowska J et al (2007) Phylogenetic relationships, host affinity, and geographic structure of boreal and arctic endophytes from three major plant lineages. Mol Phylogenet Evol 42:543–555. doi:10.1016/j.ympev.2006.07.012
Hodkinson BP, Lutzoni F (2009) A microbiotic survey of lichen-associated bacteria reveals a new lineage from the Rhizobiales. Symbiosis 49:163–180. doi:10.1007/s13199-009-0049-3
Hodkinson BP, Gottel NR, Schadt CW et al (2012) Photoautotrophic symbiont and geography are major factors affecting highly structured and diverse bacterial communities in the lichen microbiome. Environ Microbiol 14:147–161. doi:10.1111/j.1462-2920.2011.02560.x
Hodkinson BP, Allen JL, Forrest L et al (2014) Lichen-symbiotic cyanobacteria associated with Peltigera have an alternative vanadium-dependent nitrogen fixation system. Eur J Phycol 49:11–19. doi:10.1080/09670262.2013.873143
Honegger R (2012) The symbiotic phenotype of lichen-forming ascomycetes and their endo- and epibionts. In: Hock B (ed) The Mycota IX—Fungal associations, 2nd edn. Springer, Berlin. doi:10.1007/978-3-642-30826-0_15
Honegger R, Edwards D, Axe L (2012) The earliest records of internally stratified cyanobacterial and algal lichens from the lower Devonian of the Welsh Borderland. New Phytol 197:264–275. doi:10.1111/nph.12009
Houle D, Gauthier SB, Paguet S et al (2006) Identification of two genera of N2-fixing cyanobacteria growing on three feather moss species in Boreal forests of Quebec, Canada. Can J Bot 84:1025–1029. doi:10.1139/b06-059
Howard-Azzeh M, Shamseer L, Schellhorn HE et al (2014) Phylogenetic analysis and molecular signatures defining a monophyletic clade of heterocystous cyanobacteria and identifying its closest relatives. Photosynth Res 122:171–185. doi:10.1007/s11120-014-0020-x
Ininbergs K, Bay G, Rasmussen U et al (2011) Composition and diversity of nifH genes of nitrogen-fixing cyanobacteria associated with boreal forest feather mosses. New Phytol 192:507–517. doi:10.1111/j.1469-8137.2011.03809.x
Jackson O, Taylor O, Adams DG et al (2012) Arabinogalactan proteins occur in the free-living cyanobacterium genus Nostoc and in plant-Nostoc symbioses. Mol Plant Microbe Interact 25:1338–1349. doi:10.1094/MPMI-04-12-0095-R
Johansson C, Bergman B (1994) Reconstitution of the Gunnera manicata linde symbiosis: cyanobacterial specificity. New Phytol 126:643–652. doi:10.1111/j.1469-8137.1994.tb02960.x
Jørgensen PM (1998) Acantholichen pannarioides, a new basidiolichen from South America. Bryologist 101:444–447. doi:10.2307/3244185
Kaasalainen U, Jokela J, Fewer DP et al (2009) Microcystin production in the tripartite cyanolichen Peltigera leucophlebia. Mol Plant Microb Interact 22:695–702. doi:10.1094/MPMI-22-6-0695
Kaasalainen U, Fewer DP, Jokela J et al (2012) Cyanobacteria produce a high variety of hepatotoxic peptides in lichen symbiosis. Proc Natl Acad Sci U S A 109:5886–5891. doi:10.1073/pnas.1200279109
Kaasalainen U, Fewer DP, Jokela J et al (2013) Lichen species identity and diversity of cyanobacterial toxins in symbiosis. New Phytol 198:647–651. doi:10.1111/nph.12215
Kaasalainen U, Heinrichs J, Krings M et al (2015) Alectorioid morphologies in paleogene lichens: new evidence and re-evaluation of the fossil Alectoria succini Mägdefrau. PLoS One 10(6), e0129526. doi:10.1371/journal.pone.0129526
Kampa A, Gagunashvili AN, Gulder TA et al (2013) Metagenomic natural product discovery in lichen provides evidence for a family of biosynthetic pathways in diverse symbioses. Proc Natl Acad Sci U S A 110:E3129–E3137. doi:10.1073/pnas.1305867110
Kauserud H, Mathiesen C, Ohlson M (2008) High diversity of fungi associated with living parts of boreal forest bryophytes. Botany 86:1326–1333. doi:10.1139/B08-102
Kehr JC, Dittmann E (2015) Biosynthesis and function of extracellular glycans in cyanobacteria. Life 5:164–180. doi:10.3390/life5010164
Kenrick P, Strullu-Derrien C (2014) The origin and early evolution of roots. Plant Physiol 166:570–580. doi:10.1104/pp.114.244517
Khamar HJ, Breathwaite EK, Prasse CE et al (2010) Multiple roles of soluble sugars in the establishment of Gunnera-Nostoc endosymbiosis. Plant Physiol 154:1381–1389. doi:10.1104/pp.110.162529
Kimura J, Nakano T (1990) Reconstitution of a Blasia-Nostoc symbiotic association under axenic conditions. Nova Hedwig 50:191–200
Kluge M, Mollenhauer D, Mollenhauer R (1991) Photosynthetic carbon assimilation in Geosiphon pyriforme (Kützing) F.v. Wettstein, an endosymbiotic association of fungus and cyanobacterium. Planta 185:311–315. doi:10.1007/BF00201049
Kluge M, Mollenhauer D, Mollenhauer R et al (1992) Geosiphon pyriforme, an endosymbiotic consortium of a fungus and a cyanobacterium (Nostoc), fixes nitrogen. Bot Acta 105:343–344. doi:10.1111/j.1438-8677.1992.tb00309.x
Koptur S (1992) Extrafloral nectary-mediated interactions between insects and plants. In: Bernays E (ed) Insect–plant interactions, vol IV. CRC Press, Boca Raton, pp 81–129
Koptur S, Palacios-Rios M, Díaz_Castelazo et al (2013) Nectar secretion on fern fronds associated with lower levels of herbivore damage: field experiments with a widespread epiphyte of Mexican cloud forest remnants. Ann Bot 111:1277–1283. doi:10.1093/aob/mct063
Kostka JE, Weston DJ, Glass JB (2016) The Sphagnum microbiome: new insights from an ancient plant lineage. New Phytol. doi:10.1111/nph.13993. [Epub ahead of print]
Ku C, Nelson-Sathi S, Roettger M et al (2015) Endosymbiotic origin and differential loss of eukaryotic genes. Nature 524:427–432. doi:10.1038/nature14963
Larsson KH, Parmasto E, Fischer M et al (2006) Hymenochaetales: a molecular phylogeny for the hymenochaetoid clade. Mycologia 98:926–936. doi:10.3852/mycologia.98.6.926
Lawrey JD, Diederich P (2016) Lichenicolous fungi–worldwide checklist, including isolated cultures and sequences available. http://www.lichenicolous.net [5/27/2016]. Accessed 5 June 2016
Lawrey JD, Lücking R, Sipman HJM et al (2009) High concentration of basidiolichens in a single family of agaricoid mushrooms (Basidiomycota: Agaricales: Hygrophoraceae). Mycol Res 113:1154–1171. doi:10.1016/j.mycres.2009.07.016
Lechno-Yossef S, Nierzwicki-Bauer SA (2002) Azolla-Anabaena symbiosis. In: Rai AN, Bergman B, Rasmussen U (eds) Cyanobacteria in symbiosis. Kluwer, Dordrecht, pp 153–178
Leppänen SM, Salemaa M, Smolander A et al (2013) Nitrogen fixation and methanotrophy in forest mosses along a N deposition gradient. Environ Exp Bot 90:62–69. doi:10.1016/j.envexpbot.2012.12.006
Li L, Wang B, Liu Y, Qiu YL (2009) The complete mitochondrial genome sequence of the hornwort Megaceros aenigmaticus shows a mixed mode of conservative yet dynamic evolution in early land plant mitochondrial genomes. J Mol Evol 68:665–678. doi:10.1007/s00239-009-9240-7
Li FW, Villarreal JC, Kelly S et al (2014) Horizontal transfer of an adaptive chimeric photoreceptor from bryophytes to ferns. Proc Natl Acad Sci U S A 111:6672–6677. doi:10.1073/pnas.1319929111
Liaimer A, Helfrich EJ, Hinrichs K et al (2015) Nostopeptolide plays a governing role during cellular differentiation of the symbiotic cyanobacterium Nostoc punctiforme. Proc Natl Acad Sci U S A 112:1862–1867. doi:10.1073/pnas.1419543112
Ligrone R (1988) Ultrastructure of a fungal endophyte in Phaeoceros laevis (L.) Prosc. (Anthocerophyta). Bot Gaz 149:92–100. doi:10.1086/337695
Ligrone R, Duckett JG, Renzaglia KS (2012) Major transitions in the evolution of early land plants: a bryological perspective. Ann Bot 109:851–871. doi:10.1093/aob/mcs017
Lindblad P (1990) Nitrogen and carbon metabolism in coralloid roots of cycads. Mem NY Bot Gard 57:104–113
Lindblad P (2009) Cyanobacteria in symbiosis with cycads. In: Pawlowski K (ed) Prokaryotic symbionts in plants. Springer, Berlin, pp 225–233. doi:10.1007/7171_118
Lindblad P, Bergman B, Hofsten AV et al (1985) The cyanobacterium-Zamia symbiosis: an ultrastructural study. New Phytol 101:707–716. doi:10.1111/j.1469-8137.1985.tb02876.x
Lindblad P, Haselkorn R, Bergman B et al (1989) Comparison of DNA restriction fragment length polymorphisms of Nostoc strains in and from cycads. Arch Microbiol 152:20–24. doi:10.1007/BF00447006
Lindblad P, Atkins CA, Pate JS (1991) N-2-fixation by freshly isolated Nostoc from coralloid roots of the cycad Macrozamia riedlei (Fisch. ex Gaud.) Gardn. Plant Physiol 95:753–759
Lindo Z, Whiteley JA (2011) Old trees contribute bio-available nitrogen through canopy bryophytes. Plant Soil 342:141–148. doi:10.1007/s11104-010-0678-6
Lindo Z, Nilsson MC, Gundale MJ (2013) Bryophyte-cyanobacteria associations as regulators of the northern latitude carbon balance in response to global change. Glob Chang Biol 19:2022–2035. doi:10.1111/gcb.12175
Liu Y, Xue JY, Wang B, Li L, Qiu YL (2011) The mitochondrial genomes of the early land plants Treubia lacunosa and Anomodon rugelii: dynamic and conservative evolution. PLoS One 6(10), e25836. doi:10.1371/journal.pone.0025836
Lobakova ES, Orazova MK, Dobrovol’skaya TG (2003) Microbial complexes occurring on the apogeotropic roots and in the rhizosphere of cycad plants. Microbiology 72:628–633
Lóriga J, Schmidt AR, Moran RC, Feldberg K, Schneider H, Heinrichs J (2014) The first fossil of a bolbitidoid fern belongs to the early-divergent lineages of Elaphoglossum (Dryopteridaceae). Am J Bot 101:1466–1475. doi:10.3732/ajb.1400262
Lücking R, Lawrey JD, Sikaroodi M et al (2009) Do lichens domesticate photobionts like farmers domesticate crops? Evidence from a previously unrecognized lineage of filamentous cyanobacteria. Am J Bot 96:1409–1418. doi:10.3732/ajb.0800258
Lücking R, Barrie FR, Genney D (2013a) Dictyonema coppinsii, a new name for the European species known as Dictyonema interruptum (Basidiomycota: Agaricales: Hygrophoraceae), with a validation of its photobiont Rhizonema (Cyanoprokaryota: Nostocales: Rhizonemataceae). Lichenologist 46:261–267. doi:10.1017/S0024282913000352
Lücking R, Dal-Forno M, Lawrey JD et al (2013b) Ten new species of lichenized Basidiomycota in the genera Dictyonema and Cora (Agaricales: Hygrophoraceae), with a key to all accepted genera and species in the Dictyonema clade. Phytotaxa 139:1–38. doi:10.11646/phytotaxa.139.1.1
Lücking R, Dal-Forno M, Sikaroodi M (2014) A single macrolichen constitutes hundreds of unrecognized species. Proc Natl Acad Sci U S A 111:11091–11096. doi:10.1073/pnas.1403517111
Lumbsch T, Rikkinen J. (2016) Evolution of lichens. In: Dighton J, White Jr JF, Oudemans P (eds) The fungal community: its organization and role in the ecosystem, 4th edn. CRC Press (in press)
Lumbsch HT, Ahti T, Altermann S et al (2011) One hundred new species of lichenized fungi: a signature of undiscovered global diversity. Phytotaxa 18:1–127
Magain N, Sérusiaux E (2014) Do photobiont switch and cephalodia emancipation act as evolutionary drivers in the lichen symbiosis? A case study in the Pannariaceae (Peltigerales). PLoS One 9(2):e89876. doi:10.1371/journal.pone.0089876
Magain N, Goffinet B, Sérusiaux E (2012) Further photomorphs in the lichen family Lobariaceae from Reunion (Mascarene archipelago) with notes on the phylogeny of Dendriscocaulon cyanomorphs. Bryologist 115:243–254. doi:10.1639/0007-2745-115.2.243
Magallón SS, Castillo AA (2009) Angiosperm diversification through time. Am J Bot 96:349–365
Manoharan SS, Miao VPW, Andrésson ÓS (2012) LEC-2, a highly variable lectin in the lichen Peltigera membranacea. Symbiosis 58:91–98. doi:10.1007/s13199-012-0206-y
Marazzi B, Ané C, Simon MF et al (2012) Locating evolutionary precursors on a phylogenetic tree. Evolution 66:3918–3930. doi:10.1111/j.1558-5646.2012.01720.x
Marazzi B, Bronstein JL, Koptur S (2013) The diversity, ecology and evolution of extrafloral nectaries: current perspectives and future challenges. Ann Bot 111:1243–1250. doi:10.1093/aob/mct109
Meeks JC (2009) Physiological adaptations in nitrogen-fixing Nostoc-plant symbiotic associations. In: Pawlowski K (ed) Prokaryotic symbionts in plants. Springer, Berlin, pp 181–205. doi:10.1007/7171_2007_101
Meeks JC, Elhai J (2002) Regulation of cellular differentiation in filamentous cyanobacteria in free-living and plant-associated symbiotic growth states. Microbiol Mol Biol Rev 66:94–121. doi:10.1128/MMBR.66.1.94-121.2002
Meeks JC, Elhai J, Thiel T et al (2001) An overview of the genome of Nostoc punctiforme, a multicellular, symbiotic cyanobacterium. Photosynth Res 70:85–106. doi:10.1023/A:1013840025518
Meeks JC, Campbell EL, Summers ML et al (2002) Cellular differentiation in the cyanobacterium Nostoc punctiforme. Arch Microbiol 178:395–403. doi:10.1007/s00203-002-0476-5
Menge DN, Hedin LO (2009) Nitrogen fixation in different biogeochemical niches along a 120 000-year chronosequence in New Zealand. Ecology 90:2190–2201
Merinero S, Hilmo O, Gauslaa Y (2014) Size is a main driver for hydration traits in cyano- and cephalolichens of boreal rainforest canopies. Fungal Ecol 7:59–66. doi:10.1016/j.funeco.2013.12.001
Metzgar J, Schneider H, Pryer K (2007) Phylogeny and divergence time estimates for the fern genus Azolla (Salviniaceae). Int J Plant Sci 168:1045–1053. doi:10.1086/519007
Miadlikowska J, Kauff F, Högnabba F et al (2014) A multigene phylogenetic synthesis for the class Lecanoromycetes (Ascomycota): 1307 fungi representing 1139 infrageneric taxa, 312 genera and 66 families. Mol Phylogenet Evol 79:132–168. doi:10.1016/j.ympev.2014.04.003
Miao VPW, Manoharan SS, Snæbjarnarson V et al (2012) Expression of lec-1, a mycobiont gene encoding a galectin-like protein in the lichen Peltigera membranacea. Symbiosis 57:23–31. doi:10.1007/s13199-012-0175-1
Mollenhauer D, Mollenhauer R (1988) Geosiphon cultures ahead. Endocyt Cell Res 5:69–73
Moncada B, Ready B, Lücking R (2014) A phylogenetic revision of Hawaiian Pseudocyphellaria sensu lato (lichenized Ascomycota: Lobariaceae) reveals eight new species and a high degree of inferred endemism. Bryologist 117:119–160. doi:10.1639/0007-2745-117.2.119
Nagalingum NS, Marshall CR, Quental TB et al (2011) Recent synchronous radiation of a living fossil. Science 334:796–799. doi:10.1126/science.1209926
Naito M, Morton JB, Pawlowska TE (2015) Minimal genomes of mycoplasma-related endobacteria are plastic and contain host-derived genes for sustained life within Glomeromycota. Proc Natl Acad Sci U S A 112:7791–7796. doi:10.1073/pnas.1501676112
Nash TH III (2008) Nitrogen, its metabolism and potential contribution to ecosystems. In: Nash TH III (ed) Lichen biology, 2nd edn. Cambridge University Press, Cambridge, pp 216–233
Nathanielsz CP, Staff IA (1975) A mode of entry of blue-green algae into the apogeotrophic roots of Macrozamia communis. Am J Bot 62:232–235
Naumann M, Schüßler A, Bonfante P et al (2010) The obligate endobacteria of arbuscular mycorrhizal fungi are ancient heritable components related to the Mollicutes. ISME J 4:862–871. doi:10.1038/ismej.2010.21
Nierzwicki-Bauer SA, Aulfinger H (1991) Occurrence and ultrastructural characterization of bacteria in association with and isolated from Azolla caroliniana. Appl Environ Microbiol 57:3629–3636
Nilsson M, Bergman B, Rasmussen U (2000) Cyanobacterial diversity in geographically related and distant host plants of the genus Gunnera. Arch Microbiol 173:97–102. doi:10.1007/s002039900113
Nilsson M, Bhattacharya J, Rai AN, Bergman B (2002) Colonization of roots of rice (Oryza sativa) by symbiotic Nostoc strains. New Phytol 156:517–525. doi:10.1046/j.1469-8137.2002.00534.x
Nilsson M, Rasmussen U, Bergman B (2005) Competition among symbiotic cyanobacterial Nostoc strains forming artificial associations with rice (Oryza sativa). FEMS Microbiol Lett 245:139–144. doi:10.1016/j.femsle.2005.03.010
Nilsson M, Rasmussen U, Bergman B (2006) Cyanobacterial chemotaxis to extracts of host and nonhost plants. FEMS Microbiol Ecol 55:382–390. doi:10.1111/j.1574-6941.2005.00043.x
O’Brien HE, Miadlikowska J, Lutzoni F (2013) Assessing population structure and host specialization in lichenized cyanobacteria. New Phytol 198:557–566. doi:10.1111/nph.12165
Oberwinkler F (1984) Fungus-alga interactions in basidiolichens. Nova Hedw 79:739–774
Oberwinkler F (2012) Basidiolichens. In: Hock B (ed) The Mycota vol IX, fungal associations, 2nd edn. Springer, Berlin, pp 341–362
Ochoa de Alda JA, Esteban R, Diago ML et al (2014) The plastid ancestor originated among one of the major cyanobacterial lineages. Nat Commun 5:4937. doi:10.1038/ncomms5937
Oksanen I, Lohtander K, Paulsrud P et al (2002) A molecular approach to cyanobacterial diversity in a rock-pool community involving gelatinous lichens and free-living Nostoc colonies. Ann Bot Fennici 39:93–99
Oksanen I, Jokela J, Fewer DP et al (2004) Discovery of rare and highly toxic microcystins from lichen associated cyanobacterium Nostoc sp. strain IO-102-I. Appl Environ Microbiol 70:5756–5763. doi:10.1128/AEM.70.10.5756-5763.2004
Oren A, Garrity GM (2014) Proposal to change general consideration 5 and principle 2 of the international code of nomenclature of prokaryotes. Int J Syst Evol Microbiol 64:309–310. doi:10.1099/ijs.0.059568-0
Ortiz-Álvarez R, de Los RA, Fernández-Mendoza F et al (2015) Ecological specialization of two photobiont-specific maritime cyanolichen species of the genus Lichina. PLoS One 10(7), e0132718. doi:10.1371/journal.pone.0132718
Osborne B, Bergman B (2009) Why does Gunnera do it and other angiosperms don’t? An evolutionary perspective on the Gunnera-Nostoc symbiosis. In: Pawlowski K (ed) Prokaryotic symbionts in plants. Springer, Berlin, pp 207–224. doi:10.1007/7171_2007_116
Osborne BA, Sprent JI (2002) Ecology of Nostoc-Gunnera symbiosis. In: Rai AN, Bergman B, Rasmussen U (eds) Cyanobacteria in symbiosis. Kluwer, Dordrecht, pp 233–251. doi:10.1007/0-306-48005-0_13
Otálora MA, Aragoń G, Martinez I, Wedin M (2013) Cardinal characters on a slippery slope—a reevaluation of phylogeny, character evolution, and evolutionary rates in the jelly lichens (Collemataceae s. str). Mol Phylogenet Evol 68:185–198. doi:10.1016/j.ympev.2013.04.004
Otálora MA, Jørgensen PM, Wedin M (2014) A revised generic classification of the jelly lichens, collemataceae. Fungal Div 64:275–293. doi:10.1007/s13225-013-0266-1
Ow MC, Gantar M, Elhai J (1999) Reconstitution of a cycad-cyanobacterial association. Symbiosis 27:125–134
Palinska KA, Thomasius CF, Marquardt J et al (2006) Phylogenetic evaluation of cyanobacteria preserved as historic herbarium exsiccata. Int J Syst Evol Microbiol 56:2253–2263. doi:10.1099/ijs.0.64417-0
Papaefthimiou D, Hrouzek P, Mugnai MA et al (2008a) Differential patterns of evolution and distribution of the symbiotic behaviour in nostocacean cyanobacteria. Int J Syst Evol Microbiol 58:553–564. doi:10.1099/ijs.0.65312-0
Papaefthimiou D, Van Hove C, Lejeune A et al (2008b) Diversity and host specificity of Azolla cyanobionts. J Phycol 44:60–70. doi:10.1111/j.1529-8817.2007.00448.x
Paulsrud P (2002) The Nostoc symbiont of lichens: diversity, specificity and cellular modifications. PhD Thesis, Uppsala University, Sweden
Paulsrud P, Rikkinen J, Lindblad P (1998) Cyanobiont specificity in some Nostoc-containing lichens and in a Peltigera aphthosa photosymbiodeme. New Phytol 139:517–524. doi:10.1046/j.1469-8137.1998.00220.x
Paulsrud P, Rikkinen J, Lindblad P (2000) Spatial patterns of photobiont diversity in some Nostoc-containing lichens. New Phytol 146:291–299. doi:10.1046/j.1469-8137.2000.00647.x
Paulsrud P, Rikkinen J, Lindblad P (2001) Field investigations on cyanobacterial specificity in Peltigera aphthosa. New Phytol 152:117–123. doi:10.1046/j.0028-646x.2001.00234.x
Payne JL, Boyer AG, Brown JH et al (2009) Two-phase increase in the maximum size of life over 3.5 billion years reflects biological innovation and environmental opportunity. Proc Natl Acad Sci U S A 106:24–27. doi:10.1073/pnas.0806314106
Payne JL, McClain CR, Boyer AG et al (2011) The evolutionary consequences of oxygenic photosynthesis: a body size perspective. Photosynth Res 107:37–57. doi:10.1007/s11120-010-9593-1
Payne JL, Groves JR, Jost AB et al (2012) Late paleozoic fusulinoidean gigantism driven by atmospheric hyperoxia. Evolution 66:2929–2939. doi:10.1111/j.1558-5646.2012.01626.x
Peñalver E, Labandeira CC, Barrón E et al (2012) Thrips pollination of mesozoic gymnosperms. Proc Natl Acad Sci U S A 109:8623–8628. doi:10.1073/pnas.1120499109
Pereira AL, Vasconcelos V (2014) Classification and phylogeny of the cyanobiont Anabaena azollae strasburger: an answered question? Int J Syst Evol Microbiol 64:1830–1840. doi:10.1099/ijs.0.059238-0
Pereira AL, Martins M, Oliveira MM et al (2011) Morphological and genetic diversity of the family Azollaceae inferred from vegetative characters and RAPD markers. Plant Syst Evol 297:213–226. doi:10.1007/s00606-011-0509-0
Pinevich AV (2015) Proposal to consistently apply the International Code of Nomenclature of Prokaryotes (ICNP) to names of the oxygenic photosynthetic bacteria (cyanobacteria), including those validly published under the International Code of Botanical Nomenclature (ICBN)/International Code of Nomenclature for algae, fungi and plants (ICN), and proposal to change Principle 2 of the ICNP. Int J Syst Evol Microbiol 65:1070–1074. doi:10.1099/ijs.0.000034
Prasanna R, Jaiswal P, Nayak S et al (2009) Cyanobacterial diversity in the rhizosphere of rice and its ecological significance. Indian J Microbiol 49:89–97. doi:10.1007/s12088-009-0009-x
Pressel S, Ligrone R, Duckett JG (2008) The ascomycete Rhizoscyphus ericae elicits a range of host responses in the rhizoids of leafy liverworts; an experimental and cytological analysis. Fieldiana 47:59–72. doi:10.3158/0015-0746-47.1.59
Pressel S, Bidartondo MI, Ligrone R et al (2010) Fungal symbioses in bryophytes: new insights in the twenty first century. Phytotaxa 9:238–253
Qiu Y, Li L, Wang B et al (2006) The deepest divergences in land plants inferred from phylogenomic evidence. Proc Natl Acad Sci U S A 103:15511–15516. doi:10.1073/pnas.0603335103
Ran L, Larsson J, Vigil-Stenman T et al (2010) Genome erosion in nitrogen-fixing vertically transmitted endosymbiotic multicellular cyanobacterium. PLoS One 5, e11486. doi:10.1371/journal.pone.0011486
Rasmussen U, Svenning MM (2001) Characterization by genotypic methods of symbiotic Nostoc strains isolated from five species of Gunnera. Arch Microbiol 176:204–210. doi:10.1007/s002030100313
Rasmussen U, Johansson C, Bergman B (1994) Early communication in the Gunnera-Nostoc symbiosis: plant-induced cell differentiation and protein synthesis in the cyanobacterium. MPMI 7:696–702
Rasmussen U, Johansson C, Renglin A et al (1996) A molecular characterization of the Gunnera-Nostoc symbiosis: comparison with Rhizobium- and Agrobacterium-plant interactions. New Phytol 133:391–398. doi:10.1111/j.1469-8137.1996.tb01906.x
Redecker D, Schüßler (2014) Glomeromycota. In: McLaughlin DJ, Spatafora JW (eds) The Mycota VII part A, systematics and evolution, 2nd edn. Springer, Berlin, pp 251–269
Redhead SA (1981) Parasitism of bryophytes by agarics. Can J Bot 59:63–67
Renzaglia KS (1978) A comparative morphology and developmental anatomy of the Anthocerotophyta. J Hattori Bot Lab 44:31–90
Renzaglia KS (1982) A comparative developmental investigation of the gametophyte generation in the Metzgeriales (Hepatophyta). Bryophyt Bibl 24:1–238
Renzaglia KS, Duff RJ, Nickrent DL et al (2000) Vegetative and reproductive innovations of early land plants: implications for a unified phylogeny. Trans R Soc Lond 355:769–793. doi:10.1098/rstb.2000.0615
Rikkinen J (2002) Cyanolichens: an evolutionary overview. In: Rai AN, Bergman B, Rasmussen U (eds) Cyanobacteria in symbiosis. Kluwer, Dordrecht, pp 73–96
Rikkinen J (2003) Ecological and evolutionary role of photobiont-mediated guilds in lichens. Symbiosis 34:99–110
Rikkinen J (2004) Ordination analysis of tRNALeu (UAA) intron sequences in lichen-forming Nostoc strains and other cyanobacteria. Symb Bot Upsal 34:377–391
Rikkinen J (2009) Relations between cyanobacterial symbionts in lichens and plants. In: Pawlowski K (ed) Prokaryotic symbionts in plants. Springer, Berlin, pp 265–270. doi:10.1007/7171_2007_102
Rikkinen J (2013) Molecular studies on cyanobacterial diversity in lichen symbioses. MycoKeys 6:3–32. doi:10.3897/mycokeys.6.3869
Rikkinen J (2015) Cyanolichens. Biodivers Conserv 24:973–993. doi:10.1007/s10531-015-0906-8
Rikkinen J, Poinar G (2002) Fossilised Anzia (Lecanorales, lichen-forming Ascomycota) from European tertiary amber. Mycol Res 106:984–990. doi:10.1017\S0953756202005907
Rikkinen J, Poinar G (2008) A new species of Phyllopsora (Lecanorales, lichenforming Ascomycota) from Dominican amber, with remarks on the fossil history of lichens. J Exp Bot 59:1007–1011. doi:10.1093/jxb/ern004
Rikkinen J, Virtanen V (2008) Genetic diversity in cyanobacterial symbionts of thalloid bryophytes. J Exp Bot 59:1013–1021. doi:10.1093/jxb/ern003
Rikkinen J, Oksanen I, Lohtander K (2002) Lichen guilds share related cyanobacterial symbionts. Science 297:357. doi:10.1126/science.1072961
Rikkinen J, Dörfelt H, Schmidt AR et al (2003) Sooty moulds from European tertiary amber, with notes on the systematic position of Rosaria (‘Cyanobacteria’). Mycol Res 107:251–256. doi:10.1017/S0953756203007330
Rodgers GA, Stewart WD (1977) The cyanophyte-hepatic symbiosis. I. Morphology and physiology. New Phytol 78:441–458. doi:10.1111/j.1469-8137.1977.tb04851.x
Rohrlack T, Haande S, Molversmyr Å et al (2015) Environmental conditions determine the course and outcome of phytoplankton chytridiomycosis. PLoS One 10(12):e0145559. doi:10.1371/journal.pone.0145559
Rousk K, Jones DL, Deluca TH (2013) Moss-cyanobacteria associations as biogenic sources of nitrogen in boreal forest ecosystems. Front Microbiol 4:150. doi:10.3389/fmicb.2013.00150
Ruhfel BR, Gitzendanner MA, Soltis PS et al (2014) From algae to angiosperms-inferring the phylogeny of green plants (Viridiplantae) from 360 plastid genomes. BMC Evol Biol 14:23. doi:10.1186/1471-2148-14-23
Rutishauser R, Wanntorp L, Pfeifer E (2004) Gunnera herteri – developmental morphology of a dwarf from Uruguay and S Brazil (Gunneraceae). Plant Syst Evol 248:219–241. doi:10.1007/s00606-004-0182-7
Salas-Leiva DE, Meerow AW, Calonje M et al (2013) Phylogeny of the cycads based on multiple single-copy nuclear genes: congruence of concatenated parsimony, likelihood and species tree inference methods. Ann Bot 112:1263–1278. doi:10.1093/aob/mct192
Santi C, Bogusz D, Franche C (2013) Biological nitrogen fixation in non-legume plants. Ann Bot 111:743–767. doi:10.1093/aob/mct048
Schelensog M, Schroeter B, Green TGA (2000) Water dependent photosynthetic activity of lichens from New Zealand: differences in the green algal and the cyanobacterial thallus parts of photosymbiodemes. Bibl Lichenol 75:149–160
Scheloske S, Maetz M, Schüßler A (2001) Heavy metal uptake of Geosiphon pyriforme. Nucl Instrum Method B 181:659–663. doi:10.1016/S0168-583X(01)00534-1
Scheublin TR, Sanders IR, Keel C et al (2010) Characterisation of microbial communities colonising the hyphal surfaces of arbuscular mycorrhizal fungi. ISME J 4:752–763. doi:10.1038/ismej.2010.5
Schirrmeister BE, Antonelli A, Bagheri HC (2011) The origin of multicellularity in cyanobacteria. BMC Evol Biol 11:45. doi:10.1186/1471-2148-11-45
Schirrmeister BE, de Vos JM, Antonelli A (2013) Evolution of multicellularity coincided with increased diversification of cyanobacteria and the great oxidation event. Proc Natl Acad Sci U S A 110:1791–1796. doi:10.1073/pnas.1209927110
Schirrmeister BE, Gugger M, Donoghue PC (2015) Cyanobacteria and the great oxidation event: evidence from genes and fossils. Palaeontology 58:769–785. doi:10.1111/pala.12178
Schmidt AR, Beimforde C, Seyfullah LJ et al (2014) Amber fossils of sooty moulds. Rev Palaeobot Palynol 200:53–64. doi:10.1016/j.revpalbo.2013.07.002
Schneider D, Wink M, Sporer F et al (2002) Cycads: their evolution, toxins, herbivores and insect pollinators. Naturwissenschaften 89:281–294
Schneider H, Schuettpelz E, Pryer KM et al (2004) Ferns diversified in the shadow of angiosperms. Nature 428:553–557. doi:10.1038/nature02361
Schneider K, Resl P, Spribille T (2016) Escape from the cryptic species trap: lichen evolution on both sides of a cyanobacterial acquisition event. doi:10.1111/mec.13636
Schuettpelz E, Pryer KM (2009) Evidence for a cenozoic radiation of ferns in an angiosperm-dominated canopy. Proc Natl Acad Sci U S A 106:11200–11205. doi:10.1073/pnas.0811136106
Schultz M, Arendholz WR, Büdel B (2001) Origin and evolution of the lichenized ascomycete order Lichinales: monophyly and systematic relationships inferred from ascus, fruiting body and SSU rDNA evolution. Plant Biol 3:116–123
Schüßler A (2000) Glomus claroideum forms an arbuscular mycorrhiza-like symbiosis with the hornwort Anthoceros punctatus. Mycorrhiza 10:15–21. doi:10.1007/s005720050282
Schüßler A (2012) The Geosiphon–Nostoc endosymbiosis and its role as a model for arbuscular mycorrhiza research. In: Hock B (ed) The Mycota IX—fungal associations, 2nd edn. Springer, Berlin. doi:10.1007/978-3-642-30826-0_5
Schüßler A, Walker C (2011) Evolution of the ‘plant-symbiotic’ fungal phylum, Glomeromycota. In: Pöggeler S, Wöstemeyer J (eds) The Mycota VII—evolution of fungi and fungal-like organisms. Springer, Berlin, pp 163–185. doi:10.1007/978-3-642-19974-5_7
Schüßler A, Martin H, Cohen D et al (2006) Characterization of a carbohydrate transporter from symbiotic glomeromycotan fungi. Nature 444:933–936. doi:10.1038/nature05364
Schüßler A, Martin H, Cohen D et al (2007) Arbuscular mycorrhiza: studies on the Geosiphon symbiosis lead to the characterization of the first glomeromycotean sugar transporter. Plant Signal Behav 2:431–434. doi:10.4161/psb.2.5.4465
Selosse MA, Strullu-Derrien C, Martin FM et al (2015) Plants, fungi and oomycetes: a 400-million year affair that shapes the biosphere. New Phytol 206:501–506. doi:10.1111/nph.13371
Shaw AJ, Szövényi P, Shaw B (2011) Bryophyte diversity and evolution: windows into the early evolution of land plants. Am J Bot 98:352–369. doi:10.3732/ajb.1000316
Shih PM, Matzke NJ (2013) Primary endosymbiosis events date to the later proterozoic with cross-calibrated phylogenetic dating of duplicated ATPase proteins. Proc Natl Acad Sci U S A 110:12355–12360. doi:10.1073/pnas.1305813110
Shishido TK, Humisto A, Jokela J et al (2015) Antifungal compounds from cyanobacteria. Mar Drugs 13:2124–2140. doi:10.3390/md13042124
Sigurbjörnsdóttir MA, Heiðmarsson S, Jónsdóttir AR et al (2014) Novel bacteria associated with arctic seashore lichens have potential roles in nutrient scavenging. Can J Microbiol 60:307–317. doi:10.1139/cjm-2013-0888
Sigurbjörnsdóttir MA, Andrésson ÓS, Vilhelmsson O (2015) Analysis of the Peltigera membranacea metagenome indicates that lichen-associated bacteria are involved in phosphate solubilization. Microbiology 161:989–996. doi:10.1099/mic.0.000069
Silvestro D, Cascales-Miñana B, Bacon CD, Antonelli A (2015) Revisiting the origin and diversification of vascular plants through a comprehensive bayesian analysis of the fossil record. New Phytol 207:425–436. doi:10.1111/nph.13247
Söderbäck E, Bergman B (1993) The Nostoc-Gunnera symbiosis: carbon fixation and translocation. Physiol Plant 89:125–132. doi:10.1111/j.1399-3054.1993.tb01795.x
Solheim B, Endal A, Vigstad H (1996) Nitrogen fixation in arctic vegetation and soils from Svalbard, Norway. Polar Biol 16:35–40. doi:10.1007/BF02388733
Soltis DE, Senters AE, Zanis MJ et al (2003) Gunnerales are sister to other core eudicots: implications for the evolution of pentamery. Am J Bot 90:461–470. doi:10.3732/ajb.90.3.461
Sood A, Prasanna R, Prasanna BM, Singh PK (2008) Genetic diversity among and within cultured cyanobionts of diverse species of Azolla. Folia Microbiol 53:35–43. doi:10.1007/s12223-008-0005-2
Spirbille T, Tønsberg T, Stebentheiner E et al (2014) Reassessing evolutionary relationships in the filamentous cyanolichen genus Spilonema (Peltigerales, Lecanoromycetes). Lichenologist 46:373–388. doi:10.1017/S0024282913000601
Splitt SD, Risser DD (2016) The non-metabolizable sucrose analog sucralose is a potent inhibitor of hormogonium differentiation in the filamentous cyanobacterium Nostoc punctiforme. Arch Microbiol 198:137–147. doi:10.1007/s00203-015-1171-7
Stenroos S, Laukka T, Huhtinen S et al (2010) Multiple origins of symbioses between ascomycetes and bryophytes suggested by a five-gene phylogeny. Cladistics 26:281–300. doi:10.1111/j.1096-0031.2009.00284.x
Strullu-Derrien C, Kenrick P, Pressel S et al (2014) Fungal associations in Horneophyton ligneri from the Rhynie Chert (c. 407 million year old) closely resemble those in extant lower land plants: novel insights into ancestral plant-fungus symbioses. New Phytol 203:964–979. doi:10.1111/nph.12805
Stucken K, John U, Cembella A et al (2010) The smallest known genomes of multicellular and toxic cyanobacteria: comparison, minimal gene sets for linked traits and the evolutionary implications. PLoS One 5, e9235. doi:10.1371/journal.pone.0009235
Sundue MA, Testo WL, Ranker TA (2016) Morphological innovation, ecological opportunity, and the radiation of a major vascular epiphyte lineage. Evolution 69:2482–2495. doi:10.1111/evo.12749
Svenning MM, Eriksson T, Rasmussen U (2005) Phylogeny of symbiotic cyanobacteria within the genus Nostoc based on 16S rDNA sequence analyses. Arch Microbiol 183:19–26. doi:10.1007/s00203-004-0740-y
Tang N, San Clemente H, Roy S (2016) A survey of the gene repertoire of Gigaspora rosea unravels conserved features among glomeromycota for obligate biotrophy. Front Microbiol 7:233. doi:10.3389/fmicb.2016.00233
Taylor TN, Krings M (2005) Fossil microorganisms and land plants: associations and interactions. Symbiosis 40:119–135
Taylor TN, Hass H, Kerp H (1997) A cyanolichen from the lower devonian rhynie chert. Am J Bot 84:992–1004
Taylor NT, Taylor EL, Krings M (2009) Paleobotany. The biology and evolution of fossil plants, 2nd edn. Academic, London
Taylor NT, Krings M, Taylor EL (2015a) Fossil fungi. Academic, London
Taylor ZN, Rice DW, Palmer JD (2015b) The complete moss mitochondrial genome in the angiosperm Amborella is a chimera derived from two moss whole-genome transfers. PLoS One 10(11), e0137532. doi:10.1371/journal.pone.0137532
Teixeira da Silva JA, Tsavkelova EA, Zeng S et al (2015) Symbiotic in vitro seed propagation of Dendrobium: fungal and bacterial partners and their influence on plant growth and development. Planta 242:1–22. doi:10.1007/s00425-015-2301-9
Terry I, Walter GH, Moore C et al (2007) Odor-mediated push-pull pollination in cycads. Science 318:70. doi:10.1126/science.1145147
Thajuddi N, Muralithara G, Sundaramoorth M et al (2010) Morphological and genetic diversity of symbiotic cyanobacteria from cycads. J Basic Microbiol 50:254–265. doi:10.1002/jobm.200900343
Tomitani A, Knoll AH, Cavanaugh et al (2006) The evolutionary diversification of cyanobacteria: molecular-phylogenetic and paleontological perspectives. Proc Natl Acad Sci USA 103:5442–5447. doi:10.1073/pnas.0600999103
Toomer KH, Chen X, Naito M et al (2015) Molecular evolution patterns reveal life history features of mycoplasma-related endobacteria associated with arbuscular mycorrhizal fungi. Mol Ecol 24:3485–3500. doi:10.1111/mec.13250
Torres-Cortésa G, Ghignoneb S, Bonfantec P et al (2015) Mosaic genome of endobacteria in arbuscular mycorrhizal fungi: transkingdom gene transfer in an ancient mycoplasma-fungus association. Proc Natl Acad Sci U S A 112:7785–7790. doi:10.1073/pnas.1501540112
Tsavkelova EA, Lobakova ES, Kolomeĭtseva GL et al (2003) Localization of associative cyanobacteria on the roots of epiphytic orchids. Microbiology 72:86–91. doi:10.1023/A:1022286225013
Tschermak-Woess E (1988) The algal partner. In: Galun M (ed) CRC handbook of lichenology, vol I. CRC Press, Boca Raton, pp 39–92
Turetsky MR, Bond-Lamberty B, Euskirchen E et al (2012) The resilience and functional role of moss in boreal and arctic ecosystems. New Phytol 196:49–67. doi:10.1111/j.1469-8137.2012.04254.x
U’Ren JM, Lutzoni F, Miadlikowska J et al (2012) Host- and geographic structure of endophytic and endolichenic fungi at a continental scale. Am J Bot 99:898–914. doi:10.3732/ajb.1100459
Uheda E, Silvester WB (2001) The role of papillae during the infection process in the Gunnera – Nostoc symbiosis. Plant Cell Physiol 42:780–783. doi:10.1093/pcp/pce097
Valverde A, De Maayer P, Oberholster T et al (2016) Specific microbial communities associate with the rhizosphere of Welwitschia mirabilis, a living fossil. PLoS One 11, e0153353. doi:10.1371/journal.pone.0153353
van der Heijden MG, Martin FM, Selosse MA et al (2015) Mycorrhizal ecology and evolution: the past, the present, and the future. New Phytol 205:1406–1423. doi:10.1111/nph.13288
Vekemans D, Proost S, Vanneste K et al (2014) Gamma paleohexaploidy in the stem lineage of core eudicots: significance for MADS-box gene and species diversification. Mol Biol Evol 29:3793–3806. doi:10.1093/molbev/mss183
Vestola J, Shishido TK, Jokela J et al (2014) Hassallidins, antifungal glycolipopeptides, are widespread among cyanobacteria and are the end-product of a nonribosomal pathway. Proc Natl Acad Sci U S A 111:E1909–E1917. doi:10.1073/pnas.1320913111
Vigil-Stenman T, Larsson J, Nylander JA et al (2015) Local hopping mobile DNA implicated in pseudogene formation and reductive evolution in an obligate cyanobacteria-plant symbiosis. BMC Genomics 16:193. doi:10.1186/s12864-015-1386-7
Villamil N, Márquez-Guzmán J, Boege K (2013) Understanding ontogenetic trajectories of indirect defence: ecological and anatomical constraints in the production of extrafloral nectaries. Ann Bot 112:701–709. doi:10.1093/aob/mct005
Villarreal JC, Renner SS (2014) A review of molecular-clock calibrations and substitution rates in liverworts, mosses, and hornworts, and a timeframe for a taxonomically cleaned-up genus Nothoceros. Mol Phylogenet Evol 78:25–35. doi:10.1016/j.ympev.2014.04.014
Villarreal AJC, Renzaglia KS (2006) Structure and development of Nostoc strands in Leiosporoceros dussii (Anthocerotophyta): a novel symbiosis in land plants. Am J Bot 93:693–705. doi:10.3732/ajb.93.5.693
Villarreal JC, Cargill DC, Hagborg A et al (2010) A synthesis of hornwort diversity: patterns, causes and future work. Phytotaxa 9:150–166. doi:10.11646/phytotaxa.9.1.8
Villarreal JC, Forrest LL, Wickett N, Goffinet B (2013) The plastid genome of the hornwort Nothoceros aenigmaticus (Dendrocerotaceae): phylogenetic signal in inverted repeat expansion, pseudogenization, and intron gain. Am J Bot 100:467–477. doi:10.3732/ajb.1200429
Villarreal JC, Cusimano N, Renner SS (2015) Biogeography and diversification rates in hornworts: the limitations of diversification modeling. Taxon 64:229–238. doi:10.12705/642.7
Villarreal AJC, Crandall-Stotler BJ, Hart ML et al (2016) Divergence times and the evolution of morphological complexity in an early land plant lineage (Marchantiopsida) with a slow molecular rate. New Phytol 209:1734–1746. doi:10.1111/nph.13716
Vivas M, Sacristan M, Legaz ME et al (2010) The cell recognition model in chlorolichens involving a fungal lectin binding to an algal ligand can be extended to cyanolichens. Plant Biol 12:615–621. doi:10.1111/j.1438-8677.2009.00250.x
Voytsekhovich A, Mikhailyuk TI, Darienko TM (2011a) Lichen photobionts. 1: biodiversity, ecophysiology and co-evolution with the mycobiont. Algologia 21:3–26
Voytsekhovich A, Mikhailyuk TI, Darienko TM (2011b) Lichen photobionts. 2: origin and correlation with mycobiont. Algologia 21:151–177
Wahrmund U, Groth-Malonek M, Knoop V (2008) Tracing plant mitochondrial DNA evolution: rearrangements of the ancient mitochondrial gene cluster trnA-trnT-nad7 in liverwort phylogeny. J Mol Evol 66:621–629. doi:10.1007/s00239-008-9114-4
Wang XQ, Ran JH (2014) Evolution and biogeography of gymnosperms. Mol Phylogenet Evol 75:24–40. doi:10.1016/j.ympev.2014.02.005
Wang CM, Ekman M, Bergman B (2004) Expression of cyanobacterial genes involved in heterocyst differentiation and dinitrogen fixation along a plant symbiosis development profile. Mol Plant Microbe Interact 17:436–443. doi:10.1094/MPMI.2004.17.4.436
Wang YY, Liu B, Zhang XY et al (2014) Genome characteristics reveal the impact of lichenization on lichen-forming fungus Endocarpon pusillum Hedwig (Verrucariales, Ascomycota). BMC Genomics 15:34. doi:10.1186/1471-2164-15-34
Wanntorp L, Ronse De Craene LP (2005) The Gunnera flower: key to eudicot diversification or response to pollination mode? Int J Plant Sci 166:945–953
Wanntorp L, Wanntorp H-E (2003) The biogeography of Gunnera L.: vicariance and dispersal. J Biogeogr 30:979–987. doi:10.1046/j.1365-2699.2003.00895.x
Wanntorp L, Wanntorp H-E, Oxelman B et al (2001) Phylogeny of Gunnera. Plant Syst Evol 226:85–107. doi:10.1007/s006060170075
Wanntorp L, Wanntorp H-E, Rutishauser R (2003) On the homology of the scales in Gunnera (Gunneraceae). Bot J Linnean Soc 142:301–308. doi:10.1046/j.1095-8339.2003.00185.x
Wanntorp L, Dettmann ME, Jarzen DM (2004a) Tracking the mesozoic distribution of Gunnera: comparison with the fossil pollen species Tricolpites reticulatus cookson. Rev Palaeobot Palynol 132:163–174. doi:10.1016/j.revpalbo.2004.06.001
Wanntorp L, Praglowski J, Grafström E (2004b) New insights into the pollen morphology of the genus Gunnera (Gunneraceae). Grana 43:15–21
Warshan D, Bay G, Nahar N et al (2016) Seasonal variation in nifH abundance and expression of cyanobacterial communities associated with boreal feather mosses. ISME J. doi:10.1038/ismej.2016.17 [Epub ahead of print]
Weber MG, Agrawal AA (2014) Defense mutualisms enhance plant diversification. Proc Natl Acad Sci U S A 111:16442–16447. doi:10.1073/pnas.1413253111
Weber MG, Keeler KH (2013) The phylogenetic distribution of extrafloral nectaries in plants. Ann Bot 111:1251–1261. doi:10.1093/aob/mcs225
Werth S, Millanes AM, Wedin M et al (2013) Lichenicolous fungi show population subdivision by host species but do not share population history with their hosts. Fungal Biol 117:71–84. doi:10.1016/j.funbio.2012
West N, Adams DG (1997) Phenotypic and genotypic comparison of symbiotic and free-living cyanobacteria from a single field site. Appl Environ Microbiol 63:4479–4484
Wickett NJ, Mirarab S, Nguyen N (2014) Phylotranscriptomic analysis of the origin and early diversification of land plants. Proc Natl Acad Sci U S A 111:E4859–E4868. doi:10.1073/pnas.1323926111
Wojtczak G, Janik P (2016) Phytoremediation with Geosiphon-like symbiosis? Environ Sci Pollut Res Int 23:5992–5994. doi:10.1007/s11356-016-6135-1
Wolfe BE, Husband BC, Klironomos JN (2005) Effects of a belowground mutualism on an aboveground mutualism. Ecol Lett 8:218–223. doi:10.1111/j.1461-0248.2004.00716.x
Wright D, Prickett T, Heim RF et al (2001) Form species Nostoc commune (Cyanobacteria). Int J Syst Evol Microbiol 51:1839–1852. doi:10.1099/00207713-51-5-1839
Wu L, Zhang G, Lan S et al (2014) Longitudinal photosynthetic gradient in crust lichens’ thalli. Microb Ecol 67:888–896. doi:10.1007/s00248-014-0366-9
Xi Z, Rest JS, Davis CC (2013) Phylogenomics and coalescent analyses resolve extant seed plant relationships. PLoS One 8(11):e80870. doi:10.1371/journal.pone.0080870
Xue JY, Liu Y, Li L, Wang B, Qiu YL (2010) The complete mitochondrial genome sequence of the hornwort Phaeoceros laevis: retention of many ancient pseudogenes and conservative evolution of mitochondrial genomes in hornworts. Curr Genet 56:53–61. doi:10.1007/s00294-009-0279-1
Yamada S, Ohkubo S, Miyashita et al (2012) Genetic diversity of symbiotic cyanobacteria in Cycas revoluta (Cycadaceae). FEMS Microbiol Ecol 81:696–706. doi:10.1111/j.1574-6941.2012.01403.x
Zackrisson O, DeLuca TH, Nilsson M-C et al (2004) Nitrogen fixation increases with successional age in boreal forests. Ecology 85:3327–3334. doi:10.1890/04-0461
Zheng WW, Song TY, Bao XD et al (2002) High cyanobacterial diversity in coralloid roots of cycads revealed by PCR fingerprinting. FEMS Microbiol Ecol 40:215–222. doi:10.1111/j.1574-6941.2002.tb00954.x
Zheng S-P, Chen B, Guan X et al (2008) Diversity analysis of endophytic bacteria within Azolla microphylla using PCR-DGGE and electron microscopy. Chin J Agric Biotechnol 16:508–514. doi:10.1017/S1479236208002441
Zheng W, Bergman B, Chen B et al (2009) Cellular responses in the cyanobacterial symbiont during its vertical transfer between plant generations in the Azolla microphylla symbiosis. New Phytol 181:53–61. doi:10.1111/j.1469-8137.2008.02644.x
Zúñiga C, Leiva D, Ramírez-Fernández L et al (2015) Phylogenetic diversity of Peltigera cyanolichens and their photobionts in southern Chile and Antarctica. Microbes Environ 30:172–179. doi:10.1264/jsme2.ME14156
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Rikkinen, J. (2017). Cyanobacteria in Terrestrial Symbiotic Systems. In: Hallenbeck, P. (eds) Modern Topics in the Phototrophic Prokaryotes. Springer, Cham. https://doi.org/10.1007/978-3-319-46261-5_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-46261-5_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-46259-2
Online ISBN: 978-3-319-46261-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)