Abstract
One major challenge for the twenty-first century will be the production of sufficient food for the global human population. The negative impacts on soil–plant–microbes–environmental sustainability due to injudicious use of chemical fertilizer, pesticide, insecticide, etc. by the unaware farmers deteriorate soil and environment quality. One possible way to use efficient soil microorganisms to remediate nutrient deficiency in agricultural soils and other plant growth-promoting (PGP) activities that can be of help for plant growth and development. The Bacillus species is one the most dominant rhizospheric bacterial/rhizobacteria species like Bacillus subtilis, B. cereus, B. thuringiensis, B. pumilus, B. megaterium, etc. that can help enhance the plant growth and development by different mechanisms, which PGPR can inhibit phytopathogens is the production of hydrogen cyanide (HCN) and/or fungal cell wall degrading enzymes, e.g., chitinase and ß-1,3-glucanase. Direct plant growth promotion includes symbiotic and non-symbiotic PGPR which function through production of plant hormones such as auxins, cytokinins, gibberellins, ethylene, and abscisic acid. Mitigate the challenge by adopting eco-friendly crop production practices. Some Bacillus species function as a sink for 1-aminocyclopropane-1-carboxylate (ACC), the immediate precursor of ethylene in higher plants, by hydrolyzing it into α-ketobutyrate and ammonia and in this way promote root growth by lowering indigenous ethylene levels in the micro-rhizo environment. Bacillus species also help in solubilization of mineral phosphates, potassium, zinc, and other nutrients; rhizobacteria retain more soil organic N and other nutrients in the soil–plant system, thus reducing the need for fertilizers and enhancing release of the nutrients from indigenous or mineral sources, enhancing the economic and environmental sustainability.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Bacillus spp.
- Mineral solubilization
- Rhizosphere
- Fe sequestration
- Efficient microorganisms
- Nutrient uptake
16.1 Introduction
World food insecurity is a chronic problem and is likely to worsen with climate change and rapid population growth. It is largely due to poor yields of the cereal , pulse , and millet crops caused by factors including soil–plant–environment system. The world’s population is assumed to increase from ~7 billion now to 8.3 billion in 2025. The world will need 70–100 % more food by 2050 (Godfray et al. 2010). The increasing human population is placing greater pressure on soil and water resources and threatening our ability to produce sufficient food, feed , and fiber . As a result, there is a growing consensus within our global community that the protection of natural resources and implementation of environmentally and economically sound agriculture practices is of the utmost priority (Ahmad et al. 2016; Bahadur et al. 2016a).
Nowadays world agriculture is facing new challenges in which ecological and molecular approaches are being integrated to achieve higher crop yields while minimizing negative impacts on the environment. In this direction, enhancing nutrient availability, plant growth and yield, and plant multi-stress resistances are key strategies. Root-, soil-, and plant-associated eco-friendly numerous microorganisms produce plant growth-promoting activities with specific action against coexisting microorganisms toward the soil sustainability (Raaijmakers et al. 2009; Combes-Meynet et al. 2011; Genilloud et al. 2011; Pineda et al. 2012; Meena et al. 2013; Maurya et al. 2014; Kumar et al. 2015; Verma et al. 2015b). Global agriculture has to double food production by 2050 in order to feed the world’s growing population and at the same time reduce its reliance on mineral/inorganic agricultural inputs. To achieve this goal, there is an urgent need to harness the multiple beneficial interactions that occur between soil microorganisms , plant , and the environment. Beneficiary impacts of soil microorganisms enhance the sustainability of soil–plant–environment ecosystem (Gupta 2012; Bahadur et al. 2016b; Das and Pradhan 2016; Dominguez-Nuñez et al. 2016).
16.2 Soil Microbial Diversity
The beneficial influences of soil microorganisms on plant growth and development include nitrogen fixing (Peix et al. 2001; Riggs et al. 2001; Marino et al. 2007), phosphorus solubilization (Yasmin et al. 2004; Tajini et al. 2012; Verma et al. 2013), potassium solubilization (Phua et al. 2012; Yadegari et al. 2012; Zhang et al. 2013: Meena et al. 2014; Maurya et al. 2014; Saha et al. 2016a), zinc solubilization (Mäder et al. 2010; Saravanan et al. 2007; Bahadur et al. 2016b), and indirect mechanisms such as productio n of phytohormones (Rashedul et al. 2009; Abbasi et al. 2011) such as auxins (Verma et al. 2013), siderophores (Filippi et al. 2011; Yu et al. 2011a, b), and PGPR from the rhizosphere to screen for their growth-promoting activity in plants under axenic conditions (Datta et al. 2011; Meena et al. 2015a, 2016; Singh et al. 2015; Verma et al. 2015a;).
16.2.1 Agricultural Important Soil Microorganisms
It has been reported that biological fertilization is an efficient method to supply plants with their necessary nutrients. It is economically and eco-friendly recommendable, because its results improved the agricultural and environmental sustainability . During the past century, industrialization of agriculture has provoked a significant and essential productivity increase, which has led to a greater amount of food available to the general population. Along with this abundance, the appearance of serious environmental and social problems came with the package: problems that must be faced and solved in the not too distant future . Nowadays, it is urgent to maintain that high productivity , but it is becoming urgent to alter as little as possible the environment. Clearly we must then head for a more environmentally sustainable agriculture while maintaining ecosystems and biodiversity. One potential way to decrease negative environmental impact resulting from continued use of chemical fertilizers , herbicides , and pesticides is the use of plant growth-promoting rhizobacteria (PGPR) . This term was first defined by Kloepper and Schroth (1978) to describe soil bacteria that colonize the rhizosphere of plants, growing in, on, or around plant tissues that stimulate plant growth by several mechanisms. Since that time, research activities aimed at understanding how these bacteria perform their positive (or negative ) effect have steadily increased, and many reports have been published on these microorganisms . Although interactions between soil microorganisms, plants –rhizosphere, and the environment have important consequences for ecosystem dynamics and changes in plant communities with time occur in concert with changes in soil properties, the relationships between soil microbial community and plant community dynamics are not fully understood (Van Der Putten 2003; Saha et al. 2016b). Plants are able to modify the structure of microbial communities in their rhizosphere (Berg and Smalla 2009), while soil microbes are important regulators of plant productivity , both through direct effects and through regulation of nutrient availability (Meena et al. 2014). However, the role of such interactions in plant community dynamics with time has received little attention (Bartelt-Ryser et al. 2005; Meena et al. 2015b, c).
16.2.2 The Bacillus Diversity in Agricultural Soils
Bacillus is the most abundant genus in the rhizosphere , and the PGPR activity of some of these strains has been known for many years, resulting in a broad knowledge of the mechanisms involved (Probanza et al. 2002; Mañero et al.2003). Naturally present in the immediate vicinity of plant roots, B. subtilis is able to maintain stable contact with higher plants and promote their growth (Dotaniya et al. 2016; Jaiswal et al. 2016; Jha and Subramanian 2016). In a micro-propagated plant system, bacterial inoculation at the beginning of the acclimatization phase can be observed from the perspective of the establishment of the soil microbiota rhizosphere . B. licheniformis when inoculated on tomato and pepper shows considerable colonization and can be used as a bio-fertilizer without altering normal management in greenhouses as well as field condition (Bacon et al. 2001; Sessitsch et al. 2002; Wu et al. 2005). B. megaterium is very consistent in improving different root parameters in mint . Phosphorus-solubilizing bacteria (PSB) B. megaterium var. phosphaticum (Lavakusha et al. 2014) and potassium -solubilizing bacteria (KSB) B. mucilaginosus (Meena et al. 2014; Maurya et al. 2014) when inoculated in nutrient-limited soil showed that rock materials (P and K rocks) and both bacterial strains consistently increased mineral availability, uptake, and plant growth of pepper and cucumber , suggesting its potential use as bio-fertilizer (Han et al. 2006; Supanjani et al. 2006).
Soil is the main reservoir of the potential bacterial rhizosphere community (Berg and Smalla 2009). Evidence is increasing that plants actively select specific elements of their bacterial rhizosphere micro-flora , establishing a habitat which is favorable for the soil–plant–environment system (Robin et al. 2007; Houlden et al. 2008; Rudrappa et al. 2008). The soil–matrix is a favorable niche for bacteria since both temperature and humidity are relatively sustainable (Ranjard et al. 2000; Sessitsch et al. 2001), mineral composition (Carson et al. 2009), and agricultural practices (Rooney and Clipson 2009; Saha et al. 2016b). The neutral soil reaction is the most favorable condition for higher bacterial diversity, whereas acidic soils were least diverse; it’s favorable for fungus growth and development. Bacterial population revealed by culture-dependent techniques represents only 1–10 % of the total bacterial micro-flora present in soil and is now known as the great plate count anomaly (Amann et al. 1995; Meena et al. 2015d, e).
16.2.3 Soil–Plant –Microbe System
Soil–plant–microbe interactions in the rhizosphere soils are responsible for various processes that influence plant growth and development and nutrient mobilization (Awasthi et al. 2011; Singh 2013); a wide range of beneficial microorganisms (e.g., bacteria , fungi , and actinomycetes ) associated with plant roots have the ability to promote the growth of the host plant under natural as well as agroecosystem by various mechanisms, namely, fixation of atmospheric nitrogen (Glick et al. 2007), phosphorus (Verma et al. 2012a), potassium (Zhang et al. 2013), and zinc solubilization (Bapiri et al. 2012), and production of plant growth regulators (Meena et al. 2012; Miransari 2011; Rajkumar et al. 2012; Verma et al. 2012b). Besides, the plant-associated microbes residing in the rhizosphere enhance the mobility and availability of plant nutrients to the plants through release of chelating agents, acidification , and redox changes (Glick et al. 2007; Rajkumar et al. 2012). It is also well known that these microbes can utilize the plant-derived substances (e.g., root exudates ) comprising different compounds (e.g., organic acids , sugars , vitamins , and amino acids ) as major nutrients for their growth and development (Berendsen et al. 2012; Dakora and Phillips 2002; Ryan et al. 2001). On the other side, plants stimulate or inhibit the growth of specific microorganisms through releasing secondary metabolites (e.g., pyrones , sesquiterpenes ) into the rhizosphere (Reino et al. 2008; Berendsen et al. 2012; Chakraborty et al. 2012). An example of bacterial stimulation of maize plant root shoot growth is shown in Fig. 16.1.
16.3 Current Nutrient Status of Agricultural Soils
A recent review of worldwide data on N use efficiency for cereal crops from researcher-managed experimental plots reported that single-year fertilizer N recovery efficiencies are ~65 % for corn , ~57 % for wheat , and ~46 % for rice . Differences in the scale of farming operations and management practices (i.e., tillage, seeding, weed and pest control, irrigation, harvesting) usually result in lower nutrient use efficiency (Kumar et al. 2016; Masood and Bano 2016; Meena et al. 2016). Nitrogen recovery in crops grown by farmers rarely exceeds ~50 % and is often much lower. A review of best available information suggests average N recovery efficiency for fields managed by farmers ranges from about 20 % to 30 % under rainfed conditions and 30 to 40 under irrigated conditions. Looked at N fertilizer recovery under different cropping systems and reported 37 % recovery for corn grown in the north central USA. They found N recovery averaged 31 % for irrigated rice grown by Asian farmers and 40 % for rice under field-specific management. In India, N recovery averaged 18 % for wheat grown under poor weather conditions, but 49 % when grown under good weather conditions (von Braun 2007; Rajkumar and Freitas 2008a, b; Khamna et al. 2010). Phosphorus (P) efficiency is also of interest because it is one of the least available and least mobile mineral nutrients. First year recovery of applied fertilizer P ranges from less than 10 % to as high as 30 % (Fig. 16.2).
However, because fertilizer P is considered immobile in the soil and reaction (fixation and/or precipitation) with other soil minerals is relatively slow, long-term recovery of P by subsequent crops can be much higher. There is little information available about potassium (K) use efficiency. However, it is generally considered to have higher use efficiency than N and P because it is immobile in most soils and is not subject to the gaseous losses that N is or the fixation reactions that affect P. First year recovery of applied K can range from 20 % to 60 % (Fig. 16.3).
16.4 PGPR Mechanism of Bacillus Species
Bacillus species have the potential to act as a PGPR, nutrient solubilization , and bioremediation , to enhance crop growth, yield, and nutrient uptake by different mechanisms that contributed through direct and indirect mechanisms in the development of sustainable soil–plant –environment systems (Schippers et al. 1995). The generally plant growth-promoting bacteria function in three different ways – synthesizing particular PGR compounds for the growth and development of plants (Zahir et al. 2004), facilitating the mineralization or solubilization of mineral from fixed form to plant available form or soil solution that can help to enhance the nutrients’ uptake from the soil (Cakmakci et al. 2006), and helping to reduce the chances of disease infection or preventing the agricultural crops from insect, pest , and diseases (Raj 2004; Saravanakumar et al. 2008; Meena et al. 2015f; Prakash and Verma et al. 2016; Priyadharsini and Muthukumar 2016).
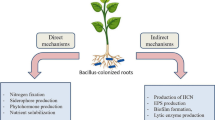
16.4.1 Direct and Indirect Mechanisms
The mechanisms of PGPB-mediated enhancement of plant growth and yield of many crops are not yet fully understood (Dey et al. 2004). However, possible explanations include (a) the ability to produce a vital enzyme , 1–aminocyclopropane–1–carboxylate (ACC) deaminase, to reduce the level of ethylene in the root of developing plants thereby increasing the root length and growth (Li et al. 2006; Meena et al. 2013; Verma et al. 2013); (b) the ability to produce hormones like auxin , i.e., indole acetic acid (IAA) (Patten and Glick 2002), abscisic acid (ABA) (Dangar and Basu 1987; Dobbelaere et al. 2003), gibberellic acid (GA) , and cytokinins (Dey et al. 2004); (c) a symbiotic nitrogen fixation (Kennedy et al. 2004); (d) antagonism against phytopathogenic bacteria by producing siderophores , ß-1,3-glucanase , chitinases , antibiotic , fluorescent pigment , and cyanide (Cattelan et al. 1999; Pal et al. 2001; Glick and Stearns 2011); (e) solubilization and mineralization of nutrients, particularly mineral phosphates and potassium (Maurya et al. 2014; Lavakusha et al. 2014; Meena et al. 2014); (f) enhanced abiotic stress (Saleem et al. 2007; Stajner et al. 1997); and (g) production of water-soluble B group vitamins such as niacin , pantothenic acid, thiamine , riboflavin, and biotin (Revillas et al. 2000; Zhuang et al. 2007; Raghavendra et al. 2016; Rawat et al. 2016; Saha et al. 2016a) (Fig. 16.4).
16.4.2 Nitrogen Fixer
The mineralization of soil organic nitrogen (N) through nitrate to gaseous N2 by soil microorganisms is a very important process in global N cycling. This cycle includes N mineralization, nitrification , denitrification , and N2 fixation . A number of bacterial species belonging to the genera Bacillus, Azospirillum , Alcaligenes , Arthrobacter , Acinetobacter , Burkholderia , Enterobacter , Erwinia , Flavobacterium , Pseudomonas , Rhizobium , and Serratia (Yu et al. 2012; Braghini Sa et al. 2012; Thepsukhon et al. 2013) are associated with the plant rhizosphere and are able to exert a beneficial effect on plant growth and development . Nowadays new techniques have identified a wide range of organisms with the plant rhizosphere with the capacity to carry out biological nitrogen fixation (BNF) – greatly expanding our appreciation of the diversity and ubiquity of N fixers – but our understanding of the rates and controls of BNF at ecosystem and global scales has not advanced at the same pace. Nevertheless, determining rates and controls of BNF is crucial to placing anthropogenic changes to the N cycle in context and to understanding, predicting, and managing many aspects of global environmental change. Here, we estimate terrestrial BNF for a preindustrial world by combining information on N fluxes with 15 N relative abundance data for terrestrial ecosystems. Our estimate is that preindustrial N fixation was 58 (range of 40–100) TgN fixed yr 21; adding conservative assumptions for geological N reduces our best estimate to 44 TgNyr 21. This approach yields substantially lower estimates than most recent calculations; it suggests that the magnitude of human alternation of the N cycle is substantially larger than has been assumed (Saha et al. 2016b; Sharma et al. 2016; Shrivastava et al. 2016).
16.4.3 Phosphorus Solubilizers
The role of phosphorus mobilizers and solubilizers is more important in soil–plant system because only ~15 % of the phosphorus fertilizer is directly available to the plant growth and development and the rest of the 85 % is lost by different processes like runoff and P fixation due to unfavorable soil conditions. However, eminent soil fertility scientists recognize that soil reactions with applied phosphate limit its direct uptake by plants in the short term; the long-term recovery can approach 90 %, because phosphorus is retained in the soil in slowly available forms (Syers 2003; Panhwar et al. 2012).
Phosphate solubilization by rhizospheric microorganisms in mineral phosphate solubilization was known as early as 1903. Since then, there have been extensive studies on the mineral phosphate solubilization by naturally abundant rhizospheric microorganisms (Fig. 16.5). Strains from bacterial genera Pseudomonas , Bacillus, Rhizobium , and Enterobacter along with Penicillium and Aspergillus fungi are the most powerful P solubilizers (Whitelaw 2000). B. megaterium , B. circulans , B. subtilis , B. polymyxa , and B. sircalmous could be referred as the most important strains (Verma et al. 2013; Meena et al. 2014; Yu et al. 2012).
The plant growth-promoting activities of Bacillus species, like potassium -solubilizing bacteria (KSB), phosphorus-solubilizing bacteria ( PSB ), iron -sequestering bacteria (siderophore -producing bacteria), cellulose -degrading activities, pectinase -producing bacteria, and phytohormone -producing bacteria (IAA , GA3, ethylene , etc.)
16.4.4 Potassium Solubilizers
K-solubilizing bacteria are able to release potassium from insoluble minerals (Sugumaran and Janarthanam 2007; Basak and Biswas 2009, 2012; Kalaiselvi and Anthoniraj 2009; Parmar and Sindhu 2013; Zarjani et al. 2013; Prajapati et al. 2013; Zhang et al. 2013; Gundala et al. 2013; Archana et al. 2012, 2013; Sindhu et al. 2012). In addition, researchers have discovered that K-solubilizing bacteria can provide beneficial effects on plant growth through suppressing pathogens and improving soil nutrients and structure. For example, certain bacteria can weather silicate minerals to release potassium, silicon , and aluminum and secrete bioactive materials to enhance plant growth (Fig. 16.5). These bacteria are widely used in biological K fertilizers and biological leaching (Lian et al. 2002; Bosecker 1997). The considerable populations of potassium-solubilizing microorganisms are present in rhizospheric soil which promotes the plant growth (Sperberg 1958).
It is generally accepted that the major mechanism of mineral K solubilization is the action of organic acids synthesized by rhizospheric microorganism. Productions of organic acids result in acidification of the microbial cell and its surroundings environment which promote the solubilization of mineral K. Silicate bacteria were found to resolve potassium , silicon , and aluminum from insoluble minerals. Silicate bacteria exert beneficial effects upon plant growth and yield. The KSB can promote K solubilization from silicate mineral and is very important to enhance the fertility status of soils. Rhizospheric microorganisms contribute directly and indirectly to the physical , chemical, and biological parameters of soil through their beneficial or detrimental activities (Meena et al. 2015g, h; Sindhu et al. 2016; Teotia et al. 2016).
16.4.5 Zinc Solubilizers
Zinc is predominantly taken up as a divalent cation, Zn2+, but in some cases of calcareous and high pH, it is believed to be taken up as a monovalent cation ZnOH+. Zinc interactions in both plants and soils are quite complex and play a major role in how and when we should apply zinc to a crop . Increasing the Zn and Fe concentration of food crop plants , resulting in better crop production and improved human health is an important global challenge. Among micronutrients , Zn deficiency is occurring in both crops and humans (White and Zasoski 1999; Welch and Graham 2004). Zinc is required in relatively small concentrations in plant tissues (5–100 mg/kg). Zn deficiency is well reported in the soils of much of the world. The deficiency of Zn in cereals especially rice is nutritionally a major problem. Cereals play in satisfying daily calorie intake in the developing world, but the Zn concentration in the grain is inherently very low, particularly when grown on Zn-deficient soils.
The major reason for the widespread occurrence of zinc deficiency problems in crop plant is the low solubility of Zn in soils rather than low total amount of Zn. Zinc-solubilizing bacteria (ZSB) help to solubilize the fixed form of Zn and increase uptake of Zn leading to fortification of grains with Zn (Bapiri et al. 2012). Soil microorganisms require various nutrients for their growth and metabolism. Among the nutrients, zinc is an element present in the enzyme system as cofactor and metal activator of many enzymes (Parisi and Vallee 1969). This causes transformation of about 96–99 % of applied available zinc to various unavailable forms (Fig. 16.5). The zinc thus made unavailable can be reverted back to available form by inoculating a bacterial strain capable of solubilizing it. Since zinc is a limiting factor in crop production, importance of ZSB has an immense in zinc nutrition to plants (Bapiri et al. 2012; Verma et al. 2013).
16.4.6 Fe Sequestration
Iron (Fe) deficiency is a worldwide problem that is directly correlated with poverty and food insecurity. Approximately one third of the world’s population suffers from Fe deficiency-induced anemia, 80 % of which are in developing countries (Boccio and Iyengar 2003; Miethke and Marahiel 2007). Total Fe content in soil is relatively high , but its availability to soil microorganisms is low in aerated soils because the prevalent form (Fe3+) is poorly soluble. Plants and microorganisms have developed mechanisms to increase Fe uptake (Marschner 1995; Rajkumar et al. 2010). In plants, there are two different strategies in response to Fe deficiency. Strategy I plants (dicots and non-graminaceous monocots ) release organic acid anions which chelate Fe. Iron solubility is also increased by decreasing the rhizosphere pH, and Fe uptake is enhanced by an increased reducing capacity of the roots (Fe3+ → Fe2+). Strategy II plants ( Poaceae ) release phytosiderophores that chelate Fe3+ (Von Wiren et al. 1993; Sinha and Mukherjee 2008; Sullivan et al. 2012). Under Fe deficiency stress, soil microorganisms release organic acid anions or siderophores that chelate Fe3+. After movement of the ferrated chelate to the cell surface, Fe3+ is reduced either outside or within the cell (Neilands 1984). Microorganisms produce a range of siderophores, e.g., ferrichromes by fungi and enterobactin , pyoverdine , and ferrioxamines by bacteria (Von Wiren et al. 1993; Ma et al. 2011). Rhizobacterial strain significantly influences Fe uptake by agricultural crop (Yu et al. 2011a, b; Sadeghi et al. 2012; Socha and Guerinot 2014).
Fe was supplied either as microbial siderophores (pseudobactin [PSB ] or ferrioxamine B [FOB]) or as phytosiderophores obtained as root exudates from barley (epi-3-hydroxy-mugineic acid [HMA] ) under varied population densities of rhizosphere microorganisms (axenic , uninoculated, or inoculated with different microorganism cultures). When maize was grown under axenic conditions and supplied with FeHMA Socha and Guerinot 2014), Fe uptake rates were 100–300 times higher compared to those in plants supplied with Fe siderophores (Fig. 16.5). Fe from both sources was taken up without the involvement of an extracellular reduction process. The supply of FeHMA enhanced both uptake rate and translocation rate to the shoot (>60 % of the total uptake). However, increased density of microorganisms resulted in a decrease in Fe uptake rate (up to 65 %), presumably due to microbial degradation of the FeHMA. In contrast, when FeFOB or FePSB was used as the Fe source, increased population density of microorganisms enhanced Fe uptake. The enhancement of Fe uptake resulted from the uptake of FeFOB and FePSB by microorganisms adhering to the rhizoplane or living in the free space of cortical cells . The microbial apoplastic Fe pool was not available for root to shoot transport or, thus, for utilization by the plants (Socha and Guerinot 2014). These results, in addition to the low uptake rate under axenic conditions, are in contrast to earlier hypotheses suggesting the existence of a specific uptake system for Fe siderophores in higher plants. The bacterial siderophores PSB and FOB were inefficient as Fe sources for plants even when supplied by stem injection. It was concluded that microorganisms are involved in degradation processes of microbial siderophores , as well as in competition for Fe with higher plants (Crowley et al. 1992; Socha and Guerinot 2014).
Fe sequestration of B. megaterium in iron -deficient medium detected in the exponential phase of growth seems not to be affected by the glucose availability and was not related with the onset of endospore formation (Chincholkar et al. 2007). The carbon source affected the siderophore production by B. megaterium (Socha and Guerinot 2014). Among the carbon sources tested, the growth on glycerol promoted the highest siderophore production. The increase of argentine concentration in the culture medium did not enhance the siderophore production. The agitation had a positive effect on the growing of B. megaterium and siderophore production. To our knowledge, this is the first work that describes the physiological response of B. megaterium in terms of siderophore production (Das et al. 2007; Socha and Guerinot 2014).
16.5 Impact of Bacillus Species on Yield and Nutrient Uptake
Nowadays rapidly increasing rate of human population with reducing land holding size due to urbanizations , industrialization , and modernization, by all these increasing presser how to we increasing our food grain production in compared to population with soil–plant –environment sustainability (Ilippi et al. 2011; Sullivan et al. 2012). One possible way to use of beneficially agricultural important microorganisms , with judicious application of mineral as well as chemical fertilizer for sustainable crop production. In this context many research studies reported that Bacillus species and other rhizobacterial strains inoculated to soil significantly enhanced crop growth, yield, and nutrient uptake (Yasmeen et al. 2012b; Velázquez et al. 2016; Yadav et al. 2016).
Bacillus spp. are used as PGPR with plant growth-promoting traits like phosphate, potassium , and zinc solubilization ; N2 fixation and phytohormone production (Liu et al. 2006; Lavakusha et al. 2014; Meena et al. 2014; Maurya et al. 2014) are also being used as bio-inoculants for crop production. The Bacillus species are reported to increase the yield in wheat (de Freitas et al. 2007; Cakmakci et al. 2007), maize (Pal et al. 2001), sugar beet (Cakmakci et al. 2006), and spinach (Cakmakci et al. 2007). According to Verma et al. (2012a) observed increase in growth and yield of beans by co-inoculating Bacillus strains with other rhizobacteria significantly influenced on nodule formation in pulse crops (Lavakusha et al. 2014; Liu et al. 2006; Yadav et al. 2010) and are widely used as plant health-promoting rhizobacteria by reducing diseases and producing antibiotic (Verma et al. 2013) (Table 16.1).
16.6 Implications of Efficient Soil Microorganisms in Sustainable Agriculture
The various ways in which efficient soil microorganisms have been used over the past fifth decade to modern sustainable technology, human and animal health, food processing, food safety and quality, genetic engineering , environmental protection, agricultural biotechnology , and in more effective treatment of agricultural. However, microbial technologies have been applied to various agricultural and environmental problems with considerable success in recent years; they have not been widely accepted by the scientific community as it is often hard to consistently reproduce their beneficial effects. We can enhance soil–plant –environment sustainability through the use of efficient soil microorganisms for sustainable agricultural production (Godfray et al. 2010). As discussed above, agriculture should consider maximizing the coadaptation between soil–plant–microbes in an effort to promote soil microbial diversity (Badri et al. 2008 ; Yasin et al. 2016; Zahedi 2016).
Which implications does decoupling the coadapted soil–plant–microbial relationship have on sustainable agriculture? The soil environment is likely the most complex biological community . Efficient soil organisms are extremely diverse and contribute to a wide range of ecosystem services that are essential to the sustainable function of natural and managed ecosystems. The efficient soil organism community can have direct and indirect impacts on land productivity . Direct impacts are those where specific efficient soil microorganisms affect crop yield immediately (Broeckling et al. 2008). Indirect effects include those provided by soil organisms participating in carbon and nutrient cycles, soil structure modification, and food web interactions that generate ecosystem services that ultimately affect productivity. Research opportunities and gaps related to methodological, experimental , and conceptual approaches may be helpful to enhance sustainable agricultural production system .
16.7 Future Prospect
The Bacillus species are a major integral component of soil microbial community and play an important role in the N fixation and phosphorus, potassium , zinc , and iron cycles in soil–plant rendering the plants available forms of nutrients. These bacterial strains have enormous potential for making use of fixed form of minerals and very slowly available nutrients under soil–plant systems with low availability in tropical and subtropical countries. The mechanism of mineral solubilization by Bacillus species has been studied in detail, but the K and Zn solubilization and Fe sequestration are a complex phenomenon affected by many factors, such as potential of bacterial strain used, nutritional status of soil, mineral type, amount of mineral, size of mineral particles, and environmental factors. Moreover, the sustainability of the Bacillus species after inoculation in soil as well as seed and seedling treatment is also important for mineral availability to benefit sustainable crop growth and development. Therefore, further study is needed to understand the problem of development of efficient and indigenous Bacillus species with microbial consortium for growth and yield of crops. Another big problem is the commercial propagation of soil microorganism’s consortium and their preservation and transportation at farmer’s fields for sustainable agricultural production.
16.8 Concluding Remarks
Climate change problems have raised great interest in eco-friendly sustainable agricultural management practices. The use of growth-promoting rhizobacteria is a promising solution for sustainable soil–plant –microbes, environmentally friendly agricultural production system. The studies on Bacillus species as plant growth-promoting activities in sustainable agriculture included isolating and screening antagonists targeting different diseases , evaluating their effectiveness in greenhouse as well as field, dissecting their mechanisms, and enhancing nutrient availability in agricultural soils. Research on improvement of Bacillus species through genetic engineering is also conducted in order to increase effectiveness under unfavorable conditions. Bacillus species control the damage to plants from phytopathogens and promote the plant growth by a number of different mechanisms and enhance the availability of nutrients for sustainable growth and development of agricultural production system.
References
Abbasi, M. K., Sharif, S., Kazmi, M., Sultan, T., & Aslam, M. (2011). Isolation of plant growth promoting rhizobacteria from wheat rhizosphere and their effect on improving growth, yield and nutrient uptake of plants. Plant Biosystems, 145, 159–168.
Abdullah, A. T., Hanafy, M. S., EL-Ghawwas, E. O., & Ali, Z. H. (2012). Effect of compost and some biofertilizers on growth, yield, essential oil, productivity and chemical composition of Rosmarinus officinalis, L. plants. Journal of Horticultural Science & Ornamental Plants, 4(2), 201–214.
Abou-Shanab, R. A., Ghanem, K., Ghanem, N., & Al-Kolaibe, A. (2008). The role of bacteria on heavy-metal extraction and uptake by plants growing on multi-metal-contaminated soils. World Journal of Microbiology and Biotechnology, 24, 253–262.
Ahmad, F., Ahmad, I., & Khan, M. S. (2006). Screening of free living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiological Research, 36, 1–9.
Ahmad, M., Nadeem, S. M., Naveed, M., & Zahir, Z. A. (2016). Potassium-solubilizing bacteria and their application in agriculture. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 293–313). New Delhi: Springer.
Alagawadi, A. R., & Gaur, A. C. (1992). Inoculation of Azospirillum brasilense and phosphate solubilizing bacteria on yield of sorghum [Sorghum bicolor (L.) Moench] in dry land. Tropical Agriculture, 69, 347–350.
Amann, R. I., Ludwig, W., & Schleifer, K. H. (1995). Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiological Reviews, 59, 143–169.
Archana, D. S., Nandish, M. S., Savalagi, V. P., & Alagawadi, A. R. (2012). Screening of K-solubilizing bacteria (KSB) for plant growth promotional activity. Bioinfolet, 9(4), 627–630.
Archana, D. S., Nandish, M. S., Savalagi, V. P., & Alagawadi, A. R. (2013). Characterization of potassium solubilizing bacteria (KSB) from rhizosphere soil. Bioinfolet, 10, 248–257.
Awasthi, R., Tewari, R., & Nayyar, H. (2011). Synergy between plants and P-solubilizing microbes in soils: Effects on growth and physiology of crops. International Research Journal of Microbiology, 2, 484–503.
Bacon, C. W., & Hinton, D. M. (2001). Control of seedling blight in wheat by Bacillus mojavensis. Phytopathology, 91(suppl), S4.
Bacon, C. W., Yates, I. E., Hinton, D. M., & Meredith, F. (2001). Biological control of Fusarium moniliforme in maize. Environmental Health Perspectives, 109, 325–332.
Badri, D. V., Loyola-Vargas, V. M., Broeckling, C. D., De-la-Pena, C., Jasinski, M., Santelia, D., Martinoia, E., Sumner, L. W., Banta, L. M., Stermitz, F., & Vivanco, J. M. (2008). Altered profile of secondary metabolites in the root exudates of Arabidopsis ATP-binding cassette transporter mutants. Plant Physiology, 146, 762–771.
Bahadur, I., Maurya, B. R., Kumar, A., Meena, V. S., & Raghuwanshi, R. (2016a). Towards the soil sustainability and potassium-solubilizing microorganisms. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 225–266). New Delhi: Springer.
Bahadur, I., Maurya, B. R., Meena, V. S., Saha, M., Kumar, A., & Aeron, A. (2016b). Minerals release dynamics of tri-calcium phosphate (TCP) and waste muscovite (WM) by mineral solubilizing rhizobacteria (MSR) isolated from Indo-Gangetic Plain (IGP) of India. Geomicrobiology Journal. doi:10.1080/01490451.2016.1219431.
Bais, H. P., Walker, T. S., Schweizer, H. P., & Vivanco, J. M. (2002). Root specific elicitation and antimicrobial activity of rosmarinic acid in hairy root cultures of Ocimum basilicum. Plant Physiology and Biochemistry, 40, 983–995.
Bapiri, A., Asgharzadeh, A., Mujallali, H., Khavazi, K., & Pazira, E. (2012). Evaluation of zinc solubilization potential by different strains of Fluorescent Pseudomonads. Journal of Applied Sciences and Environmental Management, 16(3), 295–298.
Bartelt-Ryser, J., Joshi, J., Schmid, B., Brandl, H., & Balser, T. (2005). Soil feedbacks of plant-diversity on soil microbial communities and subsequent plant growth. Perspectives in Plant Ecology, 7, 27–49.
Basak, B. B., & Biswas, D. R. (2009). Influence of potassium solubilizing microorganism (Bacillus mucilaginosus) and waste mica on potassium uptake dynamics by sudan grass (Sorghum vulgare Pers.) grown under two Alfisols. Plant and Soil, 317, 235–255.
Basak, B. B., & Biswas, D. R. (2010). Co-inoculation of potassium solubilizing and nitrogen fixing bacteria on solubilization of waste mica and their effect on growth promotion and nutrient acquisition by a forage crop. Biology and Fertility of Soils, 46, 641–648.
Basak, B. B., & Biswas, D. R. (2012). Modification of waste mica for alternative source of potassium: Evaluation of potassium release in soil from waste mica treated with potassium solubilizing bacteria (KSB). Lambert Academic Publishing. ISBN: 978-3-659-29842-4.
Baset Mia, M. A., Shamsuddin, Z. H., Wahab, Z., & Marziah, M. (2010). Effect of plant growth promoting rhizobacterial (PGPR) inoculation on growth and nitrogen incorporation of tissue-cultured musa plantlets under nitrogen-free hydroponics condition. Australian Journal of Crop Science, 4(2), 85–90.
Berendsen, R. L., Pieterse, C. M., & Bakker, P. A. (2012). The rhizosphere microbiome and plant health. Trends in Plant Science, 17, 478–486.
Berg, G., & Smalla, K. (2009). Plant species versus soil type: Which factors influence the structure and function of the microbial communities in the rhizosphere? FEMS Microbiology Ecology, 68, 1–13.
Boccio, J. R., & Iyengar, V. (2003). Iron deficiency causes, consequences, and strategies to overcome this nutritional problem. Biological Trace Element Research, 94, 1–32.
Bosecker, K. (1997). Bioleaching: Metal solubilization by microorganisms. FEMS Microbiology Reviews, 20, 59–604.
Broeckling, C. D., Broz, A. K., Bergelson, J., Manter, D. K., & Vivanco, J. M. (2008). Root exudates regulate soil fungal community composition and diversity. Applied and Environmental Microbiology, 74, 738–744.
Brunetti, G., Farrag, K., Soler-Rovira, P., Nigro, F., & Senesi, N. (2011). Greenhouse and field studies on Cr, Cu, Pb and Zn phytoextraction by Brassica napus from contaminated soils in the Apulia region, Southern Italy. Geoderma, 160, 517–523.
Cakmakci, R., Dönmez, F., Aydın, A., & Sahin, F. (2006). Growth promotion of plants by plant growth-promoting rhizobacteria under greenhouse and two different field soil conditions. Soil Biology and Biochemistry, 38, 1482–1487.
Cakmakci, R., Dönmez, M. F., & Erdoğan, A. (2007). The effect of plant growth promoting rhizobacteria on barley seedling growth, nutrient uptake, some soil properties, and bacterial counts. Turkish Journal of Agriculture and Forestry, 31, 189–199.
Calvo, P., Ormeno-Orrillo, E., Martinez-Romero, E., & Zuniga, D. (2010). Characterization of Bacillus isolates of potato rhizosphere from Andean soils of Peru and their potential PGPR characteristics. Brazilian Journal of Microbiology, 41, 899–906.
Carson, J. K., Campbell, L., Rooney, D., Clipson, N., & Gleeson, D. B. (2009). Minerals in soil select distinct bacterial communities in their microhabitats. FEMS Microbiology Ecology, 67, 381–388.
Cattelan, A. J., Hartel, P. G., & Fuhrmann, J. J. (1999). Screening of plant growth promoting rhizobacteria to promote early soybean growth. Soil Science Society of America, 63, 1670–1680.
Chakraborty, U., Chakraborty, B. N., & Chakraborty, A. P. (2012). Induction of plant growth promotion in Camellia sinensis by Bacillus megaterium and its bioformulations. World Journal of Agricultural Sciences, 8(1), 104–112.
Chen, Y., Fan, J. B., Du, L., Xu, H., Zhang, Q. H., & He, Y. Q. (2014). The application of phosphate solubilizing endophyte Pantoea dispersa triggers the microbial community in red acidic soil. Applied Soil Ecology, 84, 235–244.
Chincholkar, S. B., Chaudhari, B. L., & Rane, M. R. (2007). Microbial siderophores: State of art. In S. B. Chincholkar & A. Varma (Eds.), Microbial siderophores (pp. 233–242). Germany: Springer Verlag.
Combes-Meynet, E., Pothier, J. F., Moënne-Loccoz, Y., & Prigent-Combaret, C. (2011). The Pseudomonas secondary metabolite 2,4-diacetylphloroglucinol is a signal inducing rhizoplane expression of Azospirillum genes involved in plant-growth promotion. Molecular Plant - Microbe Interactions, 24, 271–284.
Crowley, D. E., Romheld, V., Marschner, H., & Szanizlo, P. J. (1992). Root-microbial effect on iron uptake from siderophores and phytosiderophores. Plant and Soil, 142, 1–7.
Dakora, F. D., & Phillips, D. A. (2002). Root exudates as mediators of mineral acquisition in low-nutrient environments. Plant and Soil, 245, 35–47.
Dangar, T. K., & Basu, P. S. (1987). Studies on plant growth substances, IAA metabolism and nitrogenase activity in root nodules of Phaseolus aureus Roxb. var. mungo. Biologia Plantarum, 29, 350–354.
Das, I., & Pradhan, M. (2016). Potassium-solubilizing microorganisms and their role in enhancing soil fertility and health. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 281–291). New Delhi: Springer.
Das, A., Prasad, R., Srivastava, A., Giang, P. H., Bhatnagar, K., & Varma, A. (2007). Fungal siderophores: Structure, functions and regulation. In A. Varma & S. Chincholkar (Eds.), Soil biology (pp. 1–42). Heidelberg: Springer-Verlag.
Datta, M., Palit, R., Sengupta, C., Pandit, M. K., & Banerjee, S. (2011). Plant growth promoting rhizobacteria enhance growth and yield of chili (Capsicum annuum L.) under field conditions. Australian Journal of Crop Science, 5(5), 531–536.
de-Bashan, L. E., & Bashan, Y. (2008). Joint immobilization of plant growth-promoting bacteria and green microalgae in alginate beads as an experimental model for studying plant–bacterium interactions. Applied Environmental Microbiology, 74, 6797–6802.
de Freitas, J. R., Banerjee, M. R., & Germida, J. J. (2007). Phosphate solubilising rhizobacteria enhance the growth and yield but not phosphorus uptake of canola. Biology and Fertility of Soils, 24, 358–364.
Dey, R., Pal, K. K., Bhatt, D. M., & Chauhan, S. M. (2004). Growth promotion and yield enhancement of peanut (Arachis hypogaea L.) by application of plant growth-promoting rhizobacteria. Microbiological Research, 159, 371–394.
Dobbelaere, S., Vanderleyden, J., & Okon, Y. (2003). Plant growth-promoting effects of diazotrophs in the rhizosphere. Critical Reviews in Plant Sciences, 22, 107–149.
Dominguez-Nuñez, J. A., Benito, B., Berrocal-Lobo, M., & Albanesi, A. (2016). Mycorrhizal fungi: role in the solubilization of potassium. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 77–98). New Delhi: Springer.
Dotaniya, M. L., Meena, V. D., Basak, B. B., & Meena, R. S. (2016). Potassium uptake by crops as well as microorganisms. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 267–280). New Delhi: Springer.
Earanna, N. (2001). VA mycorrhizal association in Medicinal plants of South Eastern Dry Zone of Karnataka and Response of Phyllanthus amarus and Withania somnifera to inoculation with VAM fungi and plant growth promoting Rhizomicro-organisms. Ph.D. thesis submitted to the University of Agricultural Sciences, GKVK, Bangalore.
Ekin, Z. (2010). Performance of phosphate solubilizing bacteria for improving growth and yield of sunflower (Helianthus annuus L.) in the presence of phosphorus fertilizer. African Journal of Biotechnology, 9, 3794–3800.
Esitken, A., Yildiz, H. E., Ercisli, S., Donmez, M. F., Turan, M., & Gunes, A. (2010). Effects of plant growth promoting bacteria (PGPB) on yield, growth and nutrient contents of organically grown strawberry. Scientia Horticulturae, 124, 62–66.
Erturk, Y., Ercisli, S., Haznedar, A., & Cakmakci, R. (2010). Effects of plant growth promoting rhizobacteria (PGPR) on rooting and root growth of kiwifruit (Actinidia deliciosa) stem cuttings. Biological Resaerch, 42, 91–98.
Filippi, M. C. C., da Silva, G. B., Silva-Lobo, V. L., Côrtes, M. V. C. B., Moraes, A. J. G., & Prabhu, A. S. (2011). Leaf blast (Magnaporthe oryzae) suppression and growth promotion by rhizobacteria on aerobic rice in Brazil. Biological Control, 58, 160–166.
Genilloud, O., González, I., Salazar, O., Martín, J., Tormo, J. R., & Vicente, F. (2011). Current approaches to exploit actinomycetes as a source of novel natural products. Journal of Industrial Microbiology & Biotechnology, 38, 375–389.
Glick, B. R., & Stearns, J. C. (2011). Making phytoremediation work better: Maximizing a plant’s growth potential in the midst of adversity. International Journal of Phytoremediation, 13(1), 4–16.
Glick, B. R., Cheng, Z., Czarny, J., & Duan, J. (2007). Promotion of plant growth by ACC deaminase-producing soil bacteria. European Journal of Plant Pathology, 119(3), 329–339.
Godfray, H. C. J., Beddington, J. R., Crute, I. R., Haddad, L., Lawrence, D., Muir, J. F., et al. (2010). Food security: The challenge of feeding 9 billion people. Science, 327, 812–818.
Gundala, P. B., Chinthala, P., & Sreenivasulu, B. (2013). A new facultative alkaliphilic, potassium solubilizing, Bacillus Sp. SVUNM9 isolated from mica cores of Nellore District, Andhra Pradesh, India. Research & Reviews: Journal of Microbiology and Biotechnology, 2(1), 1–7.
Gupta, V. (2012). Beneficial microorganisms for sustainable agriculture. Microbiology Australia, 3, 113–115.
Gutiérrez Mañero, F. J., Probanza, A., Ramos, B., Colón Flores, J. J., & Lucas García, J. A. (2003). Ecology, genetic diversity and screening strategies of plant growth promoting rhizobacteria (PGPR). Journal of Plant Nutrition, 26, 1101–1115.
Han, H. S., Supanjani, & Lee, K. D. (2006). Effect of co-inoculation with phosphate and potassium solubilizing bacteria on mineral uptake and growth of pepper and cucumber. Plant, Soil and Environment, 52, 130–136.
Hassan, E. A., Hassan, E. A., & Hamad, E. H. (2010). Microbial solubilization of phosphate-potassium rocks and their effect on khella (Ammi visnaga) growth. Annals of Agricultural Sciences, 55(1), 37–53.
Hemashenpagam, N., & Selvaraj, T. (2011). Effect of arbuscular mycorrhizal (AM) fungus and plant growth promoting rhizomicroorganisms (PGPR’s) on medicinal plant Solanum viarum seedlings. Journal of Environmental Biology, 32, 579–583.
Houlden, A., Timms-Wilson, T. M., Day, M. J., & Bailey, M. J. (2008). Influence of plant developmental stage on microbial community structure and activity in the rhizosphere of three field crops. FEMS Microbiology Ecology, 65, 193–201.
Ilippi, M. C. C., da Silva, G. B., Silva-Lobo, V. L., Cortes, M., Moraes, A. J. G., & Prabhu, A. S. (2011). Leaf blast (Magnaporthe oryzae) suppression and growth promotion by rhizobacteria on aerobic rice in Brazil. Biological Control, 58, 160–166.
Jaiswal, D. K., Verma, J. P., Prakash, S., Meena, V. S., & Meena, R. S. (2016). Potassium as an important plant nutrient in sustainable agriculture: A state of the art. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 21–29). New Delhi: Springer.
Jha, Y., & Subramanian, R. B. (2016). Regulation of plant physiology and antioxidant enzymes for alleviating salinity stress by potassium-mobilizing bacteria. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 149–162). New Delhi: Springer.
Kalaiselvi, P., & Anthoniraj, S. (2009). In vitro solubilization of silica and potassium from silicate minerals by silicate solubilizing bacteria. Journal of Ecobiology, 24(2), 159–168.
Karakurt, H., Kotan, R., Dadasoglu, F., Aslantaş, R., & Şahin, F. (2011). Effects of plant growth promoting rhizobacteria on fruit set, pomological and chemical characteristics, color values, and vegetative growth of sour cherry (Prunus cerasus cv. Kutahya). Turkish Journal of Biology, 35, 283–291.
Kaymak, H. C., Yarali, F., Guvenc, I., & Figen, D. M. (2008). The effect of inoculation with plant growth rhizobacteria (PGPR) on root formation of mint (Mentha piperita L.) cuttings. African Journal of Biotechnology, 7(24), 4479–4483.
Kaymak, H. C., Guvenc, I., Yarali, F., & Donmez, M. F. (2009). The effects of biopriming with PGPR on germination of radish (Raphanus sativus L.) seeds under saline conditions. Turkish Journal of Agriculture and Forestry, 33(2), 173–179.
Kennedy, I. R., Chouhury, A. T. M. A., & Kecskes, M. L. (2004). Non-symbiotic bacterial diazotrophs in crop farming systems; can their potential for plant growth promotion be latter exploited? Soil Biology and Biochemistry, 36, 1229–1244.
Khamna, S., Yokota, A., Peberdy, J. F., & Lumyong, S. (2010). Indole-3-acetic acid production by Streptomyces sp isolated from some thai medicinal plant rhizosphere soils. EurAsian Journal of BioSciences, 4, 23–32.
Khan, M. S., Zaidi, A., & Wani, P. A. (2007). Role of phosphate-solubilizing microorganisms in sustainable agriculture – a review. Agronomy for Sustainable Development, 27, 29–43.
Kloepper, J. W., & Schroth, M. N. (1978). Plant growth promoting rhizobacteria on radishes (Angers, Edt), Proceeding of the Fourth International Conference on Plant Pathogenic Bacteria Gibbert-Clarey, Tours, pp. 24, 879–882.
Kumar, A., Kumar, A., Devi, S., Patil, S., Payal, C., & Negi, S. (2012a). Isolation, screening and characterization of bacteria from rhizospheric soils for different plant growth promotion (PGP) activities an in-vitro study. Recent Research in Science and Technology, 4, 1–5.
Kumar, P., Dubey, R. C., & Maheshwari, D. K. (2012b). Bacillus strains isolated from rhizosphere showed plant growth promoting and antagonistic activity against phytopathogens. Microbiological Research, 67, 493–499.
Kumar, A., Bahadur, I., Maurya, B. R., Raghuwanshi, R., Meena, V. S., Singh, D. K., & Dixit, J. (2015). Does a plant growth-promoting rhizobacteria enhance agricultural sustainability? Journal of Pure and Applied Microbiology, 9(1), 715–724.
Kumar, A., Patel, J. S., Bahadur, I., & Meena, V. S. (2016). The molecular mechanisms of KSMs for enhancement of crop production under organic farming. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 61–75). New Delhi: Springer.
Kundu, B. S., & Gaur, A. C. (1980). Effect of phosphobacteria on the yield and phosphate uptake of potato crop. Current Science, 49, 159–160.
Lavakusha, Yadav, J., Verma, J. P., Jaiswal, D. K., & Kumar, A. (2014). Evaluation of PGPR and different concentration of phosphorus level on plant growth, yield and nutrient content of rice (Oryza sativa). Ecological Engineering, 62, 123–128.
Li, F. C., Li, S., Yang, Y. Z., & Cheng, L. J. (2006). Advances in the study of weathering products of primary silicate minerals, exemplified by mica and feldspar. Acta Petrologica et Mineralogica, 25, 440–448.
Lian, B., Fu, P. Q., Mo, D. M., & Liu, C. Q. (2002). A comprehensive review of the mechanism of potassium release by silicate bacteria. Acta Mineral Sinica, 22, 179–182.
Liu, W., Xu, X., Wu, S., Yang, Q., Luo, Y., & Christie, P. (2006). Decomposition of silicate minerals by Bacillus mucilaginosus in liquid culture. Environmental Geochemistry and Health, 28, 133–140.
Lopez, B. R., Tinoco-Ojanguren, C., Bacilioa, M., Mendozac, A., & Bashana, Y. (2012). Endophytic bacteria of the rock-dwelling cactus Mammillaria fraileana affect plant growth and mobilization of elements from rocks. Environmental and Experimental Botany, 81, 26–36.
Ma, Y., Prasad, M. N. V., Rajkumar, M., & Freitas, H. (2011). Plant growth promoting rhizobacteria and endophytes accelerate phytoremediation of metalliferous soils. Biotechnology Advances, 29, 248–258.
Mäder, P., Kiser, F., Adholeya, A., Singh, R., Uppal, H. S., Sharma, A. K., Srivastava, R., Sahai, V., Aragno, M., Wiemkein, A., Johri, B. N., & Fried, P. M. (2010). Inocula-tion of root microorganisms for sustainable wheat–rice and wheat–black gram rotations in India. Soil Biology and Biochemistry, 43, 609–619.
Marino, D., Frendo, P., González, E. M., Puppo, A., & Arrese-Igor, C. (2007). NADPH recycling systems in oxidative stressed nodules: A key role for the NADP+-dependent isocitrate dehydrogenase. Planta, 225, 413–421.
Marschner, H. (1995). Mineral nutrition of higher plants. London: Academic.
Masood, S., & Bano, A. (2016). Mechanism of potassium solubilization in the agricultural soils by the help of soil microorganisms. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 137–147). New Delhi: Springer.
Maurya, B. R., Meena, V. S., & Meena, O. P. (2014). Influence of inceptisol and alfisol’s potassium solubilizing bacteria (KSB) isolates on release of K from waste mica. Vegetos, 27(1), 181–187.
Medina, A., Probanza, A., Gutierrez Mañero, F. J., & Azcón, R. (2003). Interactions of arbuscular-mycorrhizal fungi and Bacillus strains and their effects on plant growth, microbial rhizosphere activity (thymidine and leucine incorporation) and fungal biomass (ergosterol and chitin). Applied Soil Ecology, 22, 15–28.
Meena, O. P., Maurya, B. R., & Meena, V. S. (2013). Influence of K- solubilizing bacteria on release of potassium from waste mica. Agronomy for Sustainable Development, 1(1), 53–56.
Meena, V. S., Maurya, B. R., & Verma, J. P. (2014). Does a rhizospheric microorganism enhance K+ availability in agricultural soils? Microbiological Research, 169, 337–347.
Meena, R. K., Singh, R. K., Singh, N. P., Meena, S. K., & Meena, V. S. (2015a). Isolation of low temperature surviving plant growth-promoting rhizobacteria (PGPR) from pea (Pisum sativum L.) and documentation of their plant growth promoting traits. Biocatalysis and Agricultural Biotechnology. doi:10.1016/j.bcab.2015.08.006.
Meena, R. S., Meena, V. S., Meena, S. K., & Verma, J. P. (2015b). The needs of healthy soils for a healthy world. Journal of Cleaner Production, 102, 560–561.
Meena, R. S., Meena, V. S., Meena, S. K., & Verma, J. P. (2015c). Towards the plant stress mitigate the agricultural productivity: A book review. Journal of Cleaner Production, 102, 552–553.
Meena, R. S., Yadav, R. S., Meena, H., Kumar, S., Meena, Y. K., & Singh, A. (2015d). Towards the current need to enhance legume productivity and soil sustainability worldwide: A book review. Journal of Cleaner Production, 104, 513–515.
Meena, V. S., Maurya, B. R., Verma, J. P., Aeron, A., Kumar, A., Kim, K., & Bajpai, V. K. (2015e). Potassium solubilizing rhizobacteria (KSR): Isolation, identification, and K-release dynamics from waste mica. Ecological Engineering, 81, 340–347.
Meena, V. S., Meena, S. K., Verma, J. P., Meena, R. S., & Ghosh, B. N. (2015f). The needs of nutrient use efficiency for sustainable agriculture. Journal of Cleaner Production, 102, 562–563.
Meena, V. S., Meena, S. K., Verma, J. P., Meena, R. S., & Jat, L. K. (2015g). Current trend to mitigate climate change and sustainable agriculture: A book review. Journal of Cleaner Production, 102, 548–549.
Meena, V. S., Verma, J. P., & Meena, S. K. (2015h). Towards the current scenario of nutrient use efficiency in crop species. Journal of Cleaner Production, 102, 556–557.
Meena, R. S., Bohra, J. S., Singh, S. P., Meena, V. S., Verma, J. P., Verma, S. K., & Sihag, S. K. (2016). Towards the prime response of manure to enhance nutrient use efficiency and soil sustainability a current need: A book review. Journal of Cleaner Production, 112(1), 1298–1260.
Meena, V. S., Bahadur, I., Maurya, B. R., Kumar, A., Meena, R. K., Meena, S. K., et al. (2016). Potassium-solubilizing microorganism in evergreen agriculture: An overview. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 1–20). New Delhi: Springer.
Miethke, M., & Marahiel, M. A. (2007). Siderophore-based iron acquisition and pathogen control. Microbiology and Molecular Biology Reviews, 71, 413–451.
Miransari, M. (2011). Hyperaccumulators, arbuscular mycorrhizal fungi and stress of heavy metals. Biotechnology Advances, 29, 645–653.
Nadeem, S. M., Shaharoona, B., Arshad, M., & Crowley, D. E. (2012). Population density and functional diversity of plant growth promoting rhizobacteria associated with avocado trees in saline soils. Applied Soil Ecology, 62, 147–154.
Neilands, J. B. (1984). Siderophores of bacteria and fungi. Microbiological Sciences, 1, 9–14.
Ngoma, L., Mogatlanyane, K., & Babalola, O. O. (2014). Screening of endophytic bacteria towards the development of cottage industry: An in vitro study. Journal of Human Ecology, 47(1), 45–63.
Nihorimbere, V., Ongena, M., Cawoy, H., Brostaux, Y., Kakana, P., Jourdan, E., & Thonart, P. (2010). Beneficial effects of Bacillus subtilis on field-grown tomato in Burundi: Reduction of local Fusarium disease and growth promotion. African Journal of Microbiology Research, 4(11), 1135–1142.
Nihorimbere, V., Ongena, M., Smargiassi, M., & Thonart, P. (2011). Beneficial effect of the rhizosphere microbial community for plant growth and health. Biotechnology, Agronomy, Society and Environment, 15(2), 327–337.
Orhan, E., Esitken, A., Ercisli, S., Turan, M., & Sahin, F. (2006). Effects of plant growth promoting rhizobacteria (PGPR) on yield, growth and nutrient contents in organically growing raspberry. Scientia Horticulturae, 111(1), 38–43.
Pal, K. K., Tilak, K. V. B. R., Saxena, A. K., Dey, R., & Singh, C. S. (2001). Suppression of maize root diseases caused by Macrophomina phaseolina, Fusarium moniliforme and Fusarium graminearum by plant growth promoting rhizobacteria. Microbiological Research, 156, 209–223.
Panhwar, Q. A., Othman, R., Rahman, Z. A., Meon, S., & Ismail, M. R. (2012). Isolation and characterization of phosphate-solubilizing bacteria from aerobic rice. African Journal of Biotechnology, 11(11), 2711–2719.
Parisi, B., & Vallee, B. L. (1969). Metal enzyme complexes activated by zinc. The Journal of Biological Chemistry, 179, 803–807.
Parmar, P., & Sindhu, S. S. (2013). Potassium solubilization by rhizosphere bacteria: Influence of nutritional and environmental conditions. Journal of Microbiology Research, 3(1), 25–31.
Patten, C. L., & Glick, B. R. (2002). Role of Pseudomonas putida indole-acetic acid in development of the host plant root system. Applied and Environmental Microbiology, 68, 3795–3801.
Peix, A., Mateos, P. F., Rodríguez-Barrueco, C., Martínez-Molina, E., & Velázquez, E. (2001). Growth promotion of common bean (Phaseolus vulgaris L.) by a strain of Burkholderia cepacia under growth chamber conditions. Soil Biology and Biochemistry, 33, 1927–1935.
Pereira, P., Ibanez, F., Rosenblueth, M., Etcheverry, M., Martinez-Romero, E. (2011). Analysis of the bacterial diversity associated with the roots of maize (Zea mays L.), through culture dependent and culture-independent methods. International Scholarly Research Network Ecology pp. 1–10.
Phua, C. K. H., Abdul Wahid, A. N., & Abdul Rahim, K. (2012). Development of multifunctional biofertilizer formulation from indigenous microorganisms and evaluation of their N2-fixing capabilities on Chinese cabbage using 15N tracer technique. Pakistan Journal of Agricultural Sciences, 35(3), 673–679.
Pineda, A., Zheng, S. J., van Loon, J. J. A., & Dicke, M. (2012). Rhizobacteria modify plant–aphid interactions: A case of induced systemic susceptibility. Plant Biology, 14(1), 83–90.
Prajapati, K., Sharma, M. C., & Modi, H. A. (2013). Growth promoting effect of potassium solubilizing microorganisms on Abelmoschus esculentus. International Journal of Agriculture Sciences, 3(1), 181–188.
Prakash, S., & Verma, J. P. (2016). Global perspective of potash for fertilizer production. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 327–331). New Delhi: Springer.
Priyadharsini, P., & Muthukumar, T. (2016). Interactions between arbuscular mycorrhizal fungi and potassium-solubilizing microorganisms on agricultural productivity. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 111–125). New Delhi: Springer.
Probanza, A., García, J. A. L., Palomino, M. R., Ramos, B., & Mañero, F. J. G. (2002). Pinus pinea L. seedling growth and bacterial rhizosphere structure after inoculation with PGPR Bacillus (B. licheniformis CECT 5106 and B. pumilus CECT 5105). Applied Soil Ecology, 20(2), 75–84.
Puente, M. E., Li, C. Y., & Bashan, Y. (2009a). Rock-degrading endophytic bacteria in cacti. Environmental and Experimental Botany, 66, 389–401.
Puente, M. E., Li, C. Y., & Bashan, Y. (2009b). Endophytic bacteria in cacti seeds can improve the development of cactus seedlings. Environmental and Experimental Botany, 66, 402–408.
Raaijmakers, J. M., Paulitz, T. C., Steinberg, C., Alabouvette, C., & Moënne-Loccoz, Y. (2009). The rhizosphere: A playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant and Soil, 321, 341–361.
Raghavendra, M. P., Nayaka, S. C., & Nuthan, B. R. (2016). Role of rhizosphere microflora in potassium solubilization. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 43–59). New Delhi: Springer.
Raj, S. A. (2004). Solubilization of silicate and concurrent release of phosphorus and potassium in rice ecosystem. Book chapter-conference paper Biofertilizers technology, Coimbatore, pp. 372–378.
Rajasekar, S., & Elango, R. (2011). Effect of microbial consortium on plant growth and improvement of alkaloid content in Withania somnifera (Ashwagandha). Current Botany, 2(8), 27–30.
Rajkumar, M., & Freitas, H. (2008a). Effects of inoculation of plant-growth promoting bacteria on Ni uptake by Indian mustard. Bioresource Technology, 99, 3491–3498.
Rajkumar, M., & Freitas, H. (2008b). Influence of metal resistant-plant growth-promoting bacteria on the growth of Ricinus communis in soil contaminated with heavy metals. Chemosphere, 71, 834–842.
Rajkumar, M., Nagendran, R., Lee, K. J., Lee, W. H., & Kim, S. Z. (2006). Influence of plant growth promoting bacteria and Cr6+ on the growth of Indian mustard. Chemosphere, 62, 741–748.
Rajkumar, M., Ma, Y., & Freitas, H. (2008). Characterization of metal-resistant plant-growth promoting Bacillus weihenstephanensis isolated from serpentine soil in Portugal. Journal of Basic Microbiology, 48, 1–9.
Rajkumar, M., Ae, N., & Freitas, H. (2009). Endophytic bacteria and their potential to enhance heavy metal phytoextraction. Chemosphere, 77, 153–160.
Rajkumar, M., Ae, N., Prasad, M. N. V., & Freitas, H. (2010). Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends in Biotechnology, 28, 142–149.
Rajkumar, M., Sandhya, S., Prasad, M. N. V., & Freitas, H. (2012). Perspectives of plant associated microbes in heavy metal phytoremediation. Biotechnology Advances, 30, 1562–1574.
Ranjard, L., Poly, F., Combrisson, J., Richaume, A., Gourbière, F., Thioulouse, J., & Nazaret, S. (2000). Heterogeneous cell density and genetic structure of bacterial pools associated with various soil microenvironments as determined by enumeration and DNA fingerprinting approach (RISA). Microbial Ecology, 39, 263–272.
Rashedul, I. M., Madhaiyan, M., Deka Boruah, H. P., Yim, W., Lee, G., Saravanan, V. S., et al. (2009). Characterization of plant growth-promoting traits of free-living diazotrophic bacteria and their inoculation effects on growth and nitrogen uptake of crop plants. Microbial Biotechnology, 19, 1213–1222.
Rawat, J., Sanwal, P., & Saxena, J. (2016). Potassium and its role in sustainable agriculture. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 235–253). New Delhi: Springer.
Reino, J. L., Guerrero, R. F., Hernandez-Galan, R., & Collado, I. G. (2008). Secondary metabolites from species of the biocontrol agent Trichoderma. Phytochemistry Reviews, 7, 89–123.
Revillas, J. J., Rodelas, B., Pozo, C., Martinez-Toledo, M. V., & Gonzalez, L. J. (2000). Production of B-group vitamins by two Azotobacter strains with phenolic compounds as sole carbon source under diazotrophic and adiazotrophic conditions. Journal of Applied Microbiology, 89, 486–493.
Riggs, P. D., Mikulich, S. K., & Hall, S. (2001). Effects of Pemoline on ADHD, antisocial behaviors, and substance use in adolescents with conduct disorder and substance use disorder. In College on problems of drug dependence: 63rd annual scientific meeting. Rockville: National Institute on Drug Abuse.
Robin, A., Mazurier, S., Mougel, C., Vansuyt, G., Corberand, T., Meyer, J. M., & Lemanceau, P. (2007). Diversity of root-associated fluorescent Pseudomonads as affected by ferritin overexpression in tobacco. Environmental Microbiology, 9, 1724–1737.
Rooney, D. C., & Clipson, N. J. W. (2009). Phosphate addition and plant species alters microbial community structure in acidic upland grassland soil. Microbial Ecology, 57, 4–13.
Rudrappa, T. T., Czymmek, K. J. K., Pare, P. W. P., & Bais, H. P. H. (2008). Root-secreted malic acid recruits beneficial soil bacteria. Plant Physiology, 148(3), 1547–1556.
Ryan, P. R., Delhaize, E., & Jones, D. L. (2001). Function and mechanism of organic anion exudation from plant roots. Annual Review of Plant Physiology and Plant Molecular Biology, 52, 527–560.
Sa, A. L. B., Disa, A. C. F., Teixerira, M. D. A., & Vieira, R. F. (2012). Contribution of N2 fixation for the World Agriculture. In Bacteria in agrobiology: Plant probiotics, (pp. 315–324).
Sadeghi, A., Karimi, E., Dahaji, P. A., Javid, M. G., Dalvand, Y., & Askari, H. (2012). Plant growth promoting activity of an auxin and siderophore producing isolate of Streptomyces under saline soil conditions. World Journal of Microbiology and Biotechnology, 28(4), 1503–1509.
Saha, M., Maurya, B. R., Bahadur, I., Kumar, A., & Meena, V. S. (2016a). Can potassium-solubilising bacteria mitigate the potassium problems in India? In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 127–136). New Delhi: Springer.
Saha, M., Maurya, B. R., Meena, V. S., Bahadur, I., & Kumar, A. (2016b). Identification and characterization of potassium solubilizing bacteria (KSB) from Indo-Gangetic Plains of India. Biocatalysis and Agricultural Biotechnology, 7, 202–209.
Saleem, M., Arshad, M., Hussain, S., & Bhatti, A. S. (2007). Perspective of plant growth promoting rhizobacteria (PGPR) containing ACC deaminase in stress agriculture. Journal of Industrial Microbiology & Biotechnology, 34, 635–648.
Saravanakumar, D., Lavanya, N., Muthumeena, B., Raguchander, T., Suresh, S., & Samiyappan, R. (2008). Pseudomonas fluorescens enhances resistance and natural enemy population in rice plants against leaf folder pest. Journal of Applied Entomology, 132(6), 469–479.
Saravanan, V. S., Madhaiyan, M., & Thangaraju, M. (2007). Solubilization of zinc compounds by the diazotrophic, plant growth promoting bacterium Gluconacetobacter diazotrophicus. Chemosphere, 66, 1794–1798.
Schippers, B., Scheffer, R. J., Lugtenberg, J. J., & Weisbek, P. J. (1995). Biocoating of seed with plant growth promoting rhizobacteria to improve plant establishment. Outlook on Agriculture, 24, 179–185.
Selvaraj, T., Rajeshkumar, S., Nisha, M. C., Wondimu, L., & Tesso, M. (2008). Effect of Glomus mosseae and plant growth promoting rhizomicroorganisms (PGPR’s) on growth, nutrients and content of secondary metabolites in Begonia malabarica Lam. Maejo International Journal of Science and Technology, 2(3), 516–525.
Sessitsch, A., Weilharter, A., Gerzabek, M. H., Kirchmann, H., & Kandeler, E. (2001). Microbial population structures in soil particle size fractions of a long-term fertilizer field experiment. Applied and Environmental Microbiology, 67, 4215–4224.
Sessitsch, A., Reitner, B., Pfeifer, U., & Wilhelm, E. (2002). Cultivation independent population analysis of bacterial endophyte in three potatoes varieties based on eubacterial and actinomycetes specific PCR of 16S rRNA genes. Microbiology Ecology, 39, 23–32.
Sharma, A., Shankhdhar, D., & Shankhdhar, S. C. (2016). Potassium-solubilizing microorganisms: mechanism and their role in potassium solubilization and uptake. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 203–219). New Delhi: Springer.
Shrivastava, M., Srivastava, P. C., & D’Souza, S. F. (2016). KSM soil diversity and mineral solubilization, in relation to crop production and molecular mechanism. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 221–234). New Delhi: Springer.
Sheng, X. F., & He, L. Y. (2006). Solubilization of potassium bearing minerals by a wild type strain of Bacillus edaphicus and its mutants and increased potassium uptake by wheat. Canadian Journal of Microbiology, 52(1), 66–72.
Sindhu, S. S., Parmar, P., & Phour, M. (2012). Nutrient cycling: Potassium solubilization by microorganisms and improvement of crop growth. In N. Parmar & A. Singh (Eds.), Geomicrobiology and biogeochemistry: Soil biology. Germany: Springer-Wien.
Sindhu, S. S., Parmar, P., Phour, M., & Sehrawat, A. (2016). Potassium-solubilizing microorganisms (KSMs) and its effect on plant growth improvement. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 171–185). New Delhi: Springer.
Singh, J. S. (2013). Plant growth promoting rhizobacteria. Resonance, 18, 275–281.
Singh, N. P., Singh, R. K., Shahi, J. P., Jaiswal, H. K., & Singh, T. (2013). Application of bacterial endophytes as bioinoculant enhances germination, seedling growth and yield of maize (Zea mays L.). Range Management and Agroforestry, 34(2), 171–174.
Singh, N. P., Singh, R. K., Meena, V. S., & Meena, R. K. (2015). Can we use maize (Zea mays) rhizobacteria as plant growth promoter? Vegetos, 28(1), 86–99.
Sinha, S., & Mukherjee, S. K. (2008). Cadmium-induced siderophore production by a high Cd-resistant bacterial strain relieved Cd toxicity in plants through root colonization. Current Microbiology, 56, 55–60.
Socha, A. L., & Guerinot, M. L. (2014). Mn-euvering manganese: The role of transporter gene family members in manganese uptake and mobilization in plants. Frontiers in Plant Science, 5, 106.
Sperberg, J. I. (1958). The incidence of apatite-solubilizing organisms in the rhizosphere and soil. Australian Journal of Agricultural And Resource Economics, 9(6), 778–781.
Stajner, D., Kevreaan, S., Gasaić, O., Mimica-Dudić, N., & Zongli, H. (1997). Nitrogen and Azotobacter chroococcum enhance oxidative stress tolerance in sugar beet. Biologia Plantarum, 39, 441–445.
Sugumaran, P., & Janarthanam, B. (2007). Solubilization of potassium containing minerals by bacteria and their effect on plant growth. World Journal of Agricultural Sciences, 3(3), 350–355.
Sullivan, T. S., Ramkissoon, S., Garrison, V. H., Ramsubhag, A., & Thies, J. E. (2012). Siderophore production of African dust microorganisms over Trinidad and Tobago. Aerobiologia, 28, 391–401.
Sumathi, C. S., Ramesh, N., Balasubramanian, V., & Rajesh, K. V. (2011). Microbial bioinoculant potential on turmeric (Curcuma Longa L.) growth improvement under tropical nursery conditions. Asian Journal of Experimental Biological Sciences, 2(4), 612–623.
Sumithra, P., & Selvaraj, T. (2011). Influence of Glomus walkeri and plant growth promoting rhizomicroorganisms on growth, nutrition and content of secondary metabolites in Sphaeranthus amaranthoides (L.) Burm. Journal of Agricultural Technology, 7(6), 1685–1692.
Supanjani Han, H. S., Jung, S. J., & Lee, K. D. (2006). Rock phosphate potassium and rock solubilizing bacteria as alternative sustainable fertilizers. Agronomy for Sustainable Development, 26, 233–240.
Syers, J. K. (2003). Potassium in soils: Current concepts. In A. E. Johnston (Ed.), Proceedings of the IPI Golden Jubilee Congress 1952–2002 held at Basel Switzerland 8–10 Oct 2002. Feed the soil to feed the people. The role of potash in sustainable agriculture (pp. 301–310). Basel: International Potash Institute.
Tajini, F., Trabelsi, M., & Drevon, J. J. (2012). Combined inoculation with Glomus intraradices and Rhizobium tropici CIAT899 increases phosphorus use efficiency for symbiotic nitrogen fixation in common bean (Phaseolus vulgaris L.). Saudi Journal of Biological Sciences, 19, 157–163.
Teotia, P., Kumar, V., Kumar, M., Shrivastava, N., & Varma, A. (2016). Rhizosphere microbes: potassium solubilization and crop productivity-present and future aspects. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 315–325). New Delhi: Springer.
Thepsukhon, A., Choonluchanon, S., Tajima, S., Nomura, M., & Ruamrungsri, S. (2013). Identification of endophytic bacteria associated with N2 fixation and indole acetic acid synthesis as growth promoters in curcuma alismatifolia gagnep. Journal of Plant Nutrition, 36, 1424–1438.
Upadhyay, S. K., Singh, D. P., & Saikia, R. (2009). Genetic diversity of plant growth promoting rhizobacteria isolated from rhizospheric soil of wheat under saline condition. Current Microbiology, 59, 489–496.
Upadhyay, S. K., Singh, J. S., & Singh, D. P. (2011). Exopolysaccharide-producing plant growth promoting rhizobacteria under salinity condition. Pedosphere, 2, 214–222.
Upadhyay, S. K., Singh, J. S., Saxena, A. K., & Singh, D. P. (2012). Impact of PGPR inoculation on growth and antioxidants status of wheat plant under saline condition. Plant Biology, 4, 605–611.
Van Der Putten, W. H. (2003). Plant defense belowground and spatiotemporal processes in natural vegetation. Ecology, 84, 2269–2280.
Velázquez, E., Silva, L. R., Ramírez-Bahena, M. H., & Peix, A. (2016). Diversity of potassium-solubilizing microorganisms and their interactions with plants. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 99–110). New Delhi: Springer.
Verma, J. P., Yadav, J., & Tiwari, K. N. (2012a). Enhancement of nodulation and yield of chickpea by co-inoculation of indigenous Mesorhizobium spp. and plant growth-promoting rhizobacteria in eastern Uttar Pradesh. Communications in Soil Science and Plant Analysis, 43, 605–621.
Verma, J. P., Yadav, J., Tiwari, K. N., & Kumar, A. (2012b). Effect of indigenous Mesorhizobium spp. and plant growth promoting rhizobacteria on yields and nutrients uptake of chickpea (Cicer arietinum L.) under sustainable agriculture. Ecological Engineering, 51, 282–286.
Verma, J. P., Yadav, J., Tiwari, K. N., & Kumar, A. (2013). Effect of indigenous Mesorhizobium spp. and plant growth promoting rhizobacteria on yields and nutrients uptake of chickpea (Cicer arietinum L.) under sustainable agriculture. Ecological Engineering, 51, 282–286.
Verma, J. P., Jaiswal, D. K., Meena, V. S., Kumar, A., & Meena, R. S. (2015a). Issues and challenges about sustainable agriculture production for management of natural resources to sustain soil fertility and health. Journal of Cleaner Production, 107, 793–794.
Verma, J. P., Jaiswal, D. K., Meena, V. S., & Meena, R. S. (2015b). Current need of organic farming for enhancing sustainable agriculture. Journal of Cleaner Production, 102, 545–547.
von Braun, J. (2007). The world food situation: New driving forces and required actions. Food policy report. Washington, DC: International Food Policy Research Institute.
Von Wiren, N., Roemheld, V., Morel, J. L., Guckert, A., & Marschner, H. (1993). Influence of microorganisms on iron acquisition in maize. Soil Biology and Biochemistry, 25, 371–376.
Wahyudi, A. T., Astuti, R. P., Widyawati, A., Meryandini, A. A., & Nawangsih, A. A. (2011). Characterization of Bacillus sp. strains isolated from rhizosphere of soybean plants for their use as potential plant growth for promoting rhizobacteria. Journal of Microbiology and Antimicrobials, 3(2), 34–40.
Wei, L., Kloepper, J. W., & Tuzun, S. (1996). Induced systemic resistance to cucumber diseases and increased plant growth by plant growth-promoting rhizobacteria under field conditions. Phytopathology, 86, 221–224.
Welch, R. M., & Graham, R. D. (2004). Breeding for micronutrients in staple food crops from a human nutrition perspective. Journal of Experimental Botany, 55(396), 353–364.
White, J. G., & Zasoski, R. J. (1999). Mapping soil micronutrients. Field Crops Research, 60, 11–26.
Whitelaw, M. A. (2000). Growth promotion of plants inoculated with phosphate solubilizing fungi. Advances in Agronomy, 69, 99–151.
Wu, S. C., Cao, Z. H., Li, Z. G., Cheung, K. C., & Wong, M. H. (2005). Effects of biofertilizer containing N-fixer, P and K solubilizers and AM fungi on maize growth: A greenhouse trial. Geoderma, 125, 155–166.
Wu, X. Q., Hou, L. L., Sheng, J. M., Ren, J. H., Zheng, L., Chen, D., & Ye, J. R. (2011). Effects of ectomycorrhizal fungus Boletus edulis and mycorrhiza helper Bacillus cereus on the growth and nutrient uptake by Pinus thunbergii. Biology and Fertility of Soils, 48, 385–391.
Yadav, J., Verma, J. P., & Tiwari, K. N. (2010). Effect of plant growth promoting Rhizobacteria on seed germination and plant growth Chickpea (Cicer arietinum L.) under in vitro conditions. Biological Forum-An International Journal, 2(2), 15–18.
Yadav, B. K., & Sidhu, A. S. (2016). Dynamics of potassium and their bioavailability for plant nutrition. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 187–201). New Delhi: Springer.
Yadegari, M., Farahani, G. H. N., & Mosadeghzad, Z. (2012). Biofertilizers effects on quantitative and qualitative yield of Thyme (Thymus vulgaris). African Journal of Agricultural Research, 7(34), 4716–4723.
Yasin, M., Munir, I., & Faisal, M. (2016). Can Bacillus spp. enhance K+ uptake in crop species. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 163–170). New Delhi: Springer.
Yasmeen, A., Basra, S. M. A., Wahid, A., Nouman, W., & Rehman, H. (2012). Exploring the potential of moringa (Moringa oleifera) leaf extract (MLE) as seed priming agent in improving wheat performance. Turkish Journal of Botany, 37, 512–520.
Yasmin, S., Bakar, M. A. R., Malik, K. A., & Hafeez, F. Y. (2004). Isolation, characterization and beneficial effects of rice associated plant growth promoting bacteria from Zanzibar soils. Journal of Basic Microbiology, 3, 241–252.
Yu, X., Liu, X., Zhu, T. H., Liu, G. H., & Mao, C. (2011a). Isolation and characterization of phosphate-solubilizing bacteria from walnut and their effect on growth and phosphorus mobilization. Biology and Fertility of Soils, 47, 437–446.
Yu, X. M., Ai, C. X., Xin, L., & Zhou, G. F. (2011b). The siderophore-producing bacterium, Bacillus subtilis CAS15, has a biocontrol effect on Fusarium wilt and promotes the growth of pepper. European Journal of Soil Biology, 47, 138–145.
Yu, X., Liu, X., Zhu, T. H., Liu, G. H., & Mao, C. (2012). Co-inoculation with phosphate-solubilizing and nitrogen-fixing bacteria on solubilization of rock phosphate and their effect on growth promotion and nutrient uptake by walnut. European Journal of Soil Biology, 50, 112–117.
Zahedi, H. (2016). Growth-promoting effect of potassium-solubilizing microorganisms on some crop species. In V. S. Meena, B. R. Maurya, J. P. Verma, & R. S. Meena (Eds.), Potassium solubilizing microorganisms for sustainable agriculture (pp. 31–42). New Delhi: Springer.
Zahir, A. Z., Arshad, M., & Frankenberger, W. T., Jr. (2004). Plant growth promoting rhizobacteria: Application and perspectives in agriculture. Advances in Agronomy, 81, 97–168.
Zaidi, S., Usmani, S., Singh, B. R., & Musarrat, J. (2006). Significance of Bacillus subtilis strain SJ-101 as a bioinoculant for concurrent plant growth promotion and nickel accumulation in Brassica juncea. Chemosphere, 64, 991–997.
Zarjani, J. K., Aliasgharzad, N., Oustan, S., Emadi, M., & Ahmadi, A. (2013). Isolation and characterization of potassium solubilizing bacteria in some Iranian soils. Archives of Agronomy and Soil Science, 59, 1713–1723.
Zhang, H., Kim, M. S., Sun, Y., Dowd, S. E., Shi, H., & Paré, P. W. (2008). Soil bacteria confer plant salt tolerance by tissue-specific regulation of the sodium transporter HKT1. Molecular Plant - Microbe Interactions, 21, 737–744.
Zhang, A., Zhao, G., Gao, T., Wang, W., Li, J., Zhang, S., & Zhu, B. (2013). Solubilization of insoluble potassium and phosphate by Paenibacillus kribensis CX-7: A soil microorganism with biological control potential. African Journal of Microbiology Research, 7(1), 41–47.
Zhao, Q. Y., Shen, Q. R., Ran, W., Xiao, T. J., Xu, D. B., & Xu, Y. C. (2011). Inoculation of soil by Bacillus subtilis Y-IVI improves plant growth and colonization of the rhizosphere and interior tissues of muskmelon (Cucumis melo L.). Biology and Fertility of Soils, 47, 507–514.
Zhuang, X., Chen, J., Shim, H., & Bai, Z. (2007). New advances in plant growth-promoting rhizobacteria for bioremediation. Environment International, 33, 406–413.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing AG
About this chapter
Cite this chapter
Meena, V.S. et al. (2016). Can Bacillus Species Enhance Nutrient Availability in Agricultural Soils?. In: Islam, M., Rahman, M., Pandey, P., Jha, C., Aeron, A. (eds) Bacilli and Agrobiotechnology. Springer, Cham. https://doi.org/10.1007/978-3-319-44409-3_16
Download citation
DOI: https://doi.org/10.1007/978-3-319-44409-3_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-44408-6
Online ISBN: 978-3-319-44409-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)