Abstract
This chapter reviews recent developments in the treatment of ischemic mitral regurgitation. Recent and ongoing studies have added to our understanding of this dynamic disease process. We discuss current investigations on outcomes of new and established approaches, including adjunctive surgical techniques.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Ischemic Mitral Regurgitation (IMR)
- Revascularization
- Mitral Valve Repair (MVR)
- Coronary Artery Bypass Grafting (CABG)
- Left Ventricular Reverse Remodeling
Introduction
Mitral regurgitation (MR) represents the most frequent valvular heart disease in the United States. Ischemic mitral regurgitation (IMR) is common after myocardial infarction and results in significantly increased risk for congestive heart failure and death. It is usually mild in severity and consequently may go undiagnosed [1]. While outcomes are worse with increasing IMR severity, even mild IMR portends a significantly increased risk of cardiovascular mortality. The Survival and Ventricular Enlargement (SAVE) study reported a cardiovascular mortality incidence of 29 % at 3.5 years after MI in those developing IMR, compared to 12 % in those without IMR (P < 0.001) [2]. The dynamic nature of IMR makes assessment and treatment selection challenging.
Pathophysiology of IMR
Pathologic condition in any one or more of the components of the mitral valve apparatus may lead to mitral regurgitation; however, the mitral valve is normal in structure in most cases of IMR. The definition of ischemic (functional) mitral regurgitation by the classic Carpentier triad requires the following: (1) patient with known coronary artery disease and a global or regional wall motion abnormality; (2) echocardiographic evidence for restricted leaflet motion in systole and/or annular dilatation, and (3) valve leaflet tethering but otherwise macroscopically normal leaflets [3]. Mitral regurgitation in the ischemic and dysfunctional LV further increases atrial pressure, which may lead to both pulmonary hypertension [4] and heart failure [5].
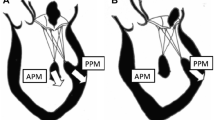
IMR is largely due to left ventricular dysfunction and dilatation. Because chordae are non-extensible, papillary-muscle displacement exerts traction on leaflets [6]. Subsequent dilatation of the mitral annulus contributes to IMR by distracting the mitral leaflets and preventing appropriate leaflet coaptation. IMR occurs due to papillary muscle rupture in a small minority of cases. More commonly, local LV remodeling in the region of papillary muscles attachment results in displacement and thus alteration of mitral valve coaptation. Commonly, myocardial infarction involving the right or circumflex coronary arteries will result in greater displacement of the posterior compared to the anterior papillary muscle [7]. This results in greater posterior leaflet tethering. After anterior MI, apical displacement of papillary muscles may result in apical tethering of both mitral valve leaflets and central mitral regurgitation [8].
Multiple lines of evidence suggest that MR in the setting of ischemic disease is associated with poor outcome [1, 5, 9, 10]. Still disputed is whether IMR intrinsically causes poor outcome or whether IMR is a surrogate for left ventricular alterations which themselves are responsible for poor outcomes. However, the association of severe ischemic mitral regurgitation with poor outcomes independent of ejection fraction, age, and presentation all suggest that regurgitation itself is a major contributor to poor outcomes.
Treatment Options
While modest improvements have been made in recent years, rates of morbidity and mortality with intervention in IMR remain high [11, 12]. Overall suboptimal outcomes reflect uncertainty with respect to surgical indications. With transcatheter interventional treatments for mitral regurgitation primarily in the experimental phase, surgery is the treatment recommended by management guidelines [13]. The most recently published ACC/AHA guidelines for CABG and Valve Disease avoid a formal decision algorithm for the treatment of IMR and at present lag behind the best available clinical evidence. 2014 guidelines advise that a concomitant mitral valve repair procedure be considered in patients with chronic moderate secondary MR undergoing other cardiac surgery (Level of Evidence: C) [14]. In this chapter, the use of coronary artery revascularization by way of CABG alone in the treatment of ischemic mitral regurgitation will be reviewed. The decision to add a concomitant mitral valve procedure will be fully discussed in the next chapter. Most broadly, the best clinical evidence available at this time support the use of revascularization with CABG alone for the treatment of moderate ischemic mitral regurgitation. We do not recommend the routine inclusion of a concomitant mitral valve procedure to CABG for the treatment of moderate ischemic mitral regurgitation.
Coronary Artery Revascularization
Reported hospital mortality in patients with IMR undergoing CABG varies widely from 1.0 to 12.5 %, due to differences in baseline comorbidities, LV size and LV function [15–18]. While percutaneous coronary intervention (PCI) continues to be offered to some patients at high risk for conventional open CABG, this modality often fails to address the persistently occluded coronary arteries common in IMR patients. Ellis and colleagues reported a 28 % rate of complete revascularization with PCI in this population [19]. Ellis and colleagues suggest that maximally complete revascularization by CABG may offer better outcomes since most IMR patients suffer from severe multivessel coronary artery disease.
The response of IMR to coronary revascularization alone is known to be variable. Among patients with moderate IMR undergoing CABG alone, Lam and colleagues report a 22 % progression to severe IMR at 6 weeks [17]. By contrast, Tolis and colleagues report a similar population of patients undergoing CABG alone for mild-moderate IMR and show improvement at 3 year follow-up of MR grade from 1.7 to 0.5 [18]. This variation in outcome is hypothesized to relate to the completeness of revascularization, a standpoint supported by recent observational studies [20]. LV contractile reserve may also play a role, in that successful CABG will restore viable LV segments in the region of papillary muscle attachment that may relieve tethering of the mitral valve. Recent work examining cardiac remodeling has revealed no change in MR grade among patients without improvement in LV function or LV size following isolated CABG for IMR, supporting the thesis that maximal restoration of perfusion to viable myocardium is prerequisite for successful remodeling [16, 21, 22].
Five year survival in patients with moderate IMR undergoing CABG alone varies from 50 % [18] to 87 % [16] in recent observational studies. Survival after CABG in the IMR population is generally decreased compared to those patients undergoing isolated CABG without MR [17, 23, 24]. In their most recent work, Grossi and colleagues report that even mild IMR is an independent risk factor for worsening survival after CABG [24]. Examination of over 3000 patients treated at Duke University revealed a graded effect with increasing MR associated with increasing mortality after CABG [23]. These data again highlight that patients with increasing degrees of MR often present as more comorbid than patients without MR. Patients with MR had worse NYHA functional class and lower ejection fraction. These patients were more likely to suffer from renal insufficiency, and were more likely to have had an intra-aortic balloon pump (IABP) in place preoperatively. These factors have made appropriate risk stratification and propensity analysis challenging in determining the best course of therapy. Fortunately, recent prospective work has sought to clarify treatment strategies for this patient population.
Revascularization with CABG Alone for Ischemic MR
In patients with moderate IMR, there has been considerable controversy with respect to the appropriate course of therapy. In particular, the decision to perform a valve procedure in addition to revascularization with CABG has been the subject of much debate. The benefit of concomitant mitral valve intervention has hinged on an assessment of the extent to which revascularization alone can adequately improve valvular function. Proponents of CABG alone note that revascularization can promote left ventricular remodeling, leading to a decreased LV chamber size, restored functional integrity of the subchordal mitral valve apparatus, and thereby decreasing mitral regurgitation. Importantly, the kinetics and rate of remodeling remain to be fully elucidated. This is of particular importance in the evaluation of short-term outcomes following CABG, as near-term evaluation may not fully capture the benefit of coronary artery bypass surgery. Those who favor combined CABG and mitral intervention note that isolated CABG in the presence of scar and non-viable myocardium may not result in remodeling after revascularization, therefore necessitating mitral intervention as regurgitation will not improve despite revascularization. Recently, both updated observational studies and prospective clinical trial data have clarified the risks and benefits of each strategy in this patient population.
Castleberry et al. retrospectively reviewed patients at a single institution carrying a diagnosis of coronary artery disease and moderate or severe mitral regurgitation from 1990 to 2009 [25]. A total of 4989 patients were stratified by medical therapy alone, PCI, CABG, or CABG with concomitant mitral valve repair or replacement. After a median follow up period of 5.37 years, lower mortality was observed in patients treated with revascularization (by PCI, CABG, or CABG plus mitral valve repair or replacement) in comparison to medical therapy. Patients treated with CABG alone demonstrated the lowest risk of death.
Three prospective clinical trials now corroborate many of the findings first identified in the retrospective databases (Table 6.1). The first to publish (in 2009), Fattouch et al. reported a single center Italian study of 102 patients of which 48 underwent CABG with concomitant MVR, while 54 underwent CABG alone [26]. Primary endpoints were NYHA functional class to assess clinical status, and assessment of extent of reversal of left ventricular remodeling by way of echocardiographic assessment (TTE) of left ventricular end-systolic diameter (LVESD), left ventricular end-diastolic diameter (LVEDD) and ejection fraction. At 1 year, addition of MVR to CABG resulted in lower NYHA functional class (15.5 % of patients undergoing CABG with MVR vs. 43.7 % of patients undergoing CABG alone experiencing NYHA class II or greater symptoms). CABG-alone patients experienced higher rates of post-operative MR and had no significant change in LVESD or LVEDD at the conclusion of the study, while CABG with MVR patients exhibited reversal of left-ventricular remodeling as evidenced by a decrease in both LVESD and LVEDD. Subjects had no difference in ejection fraction nor survival, though the study was underpowered for the detection of survival differences.
The 2012 Randomized Ischemic Mitral Evaluation (RIME) trial randomized a total of 73 patients in the United Kingdom and Poland with moderate MR by echocardiography to isolated CABG vs CABG with concomitant MVR [27]. The study was concluded after 73 patients of a planned 100 after the primary endpoint – peak oxygen consumption – was reached after 1 year. Peak oxygen consumption has previously been recognized as a clinically-relevant measure of functional capacity. Patients undergoing CABG + MVR had a 22 % increase in peak oxygen consumption at 1-year compared to a 5 % increase in patients undergoing isolated CABG. Patients undergoing CABG + MVR had a median NYHA functional class of I, compared to median class of II in the isolated CABG group at the conclusion of the follow up period. The RIME study did not identify any difference in overall survival between the treatment arms, though again a small sample size and short duration of follow up limit the scope of long-term conclusions. Overall, the RIME study supported the addition of a mitral intervention in addition to revascularization with CABG.
Most recently, Smith et al. have reported the results of the largest trial to date designed to ascertain if the potential benefits of a combined CABG and mitral procedure outweigh the increased risks of the added intervention [28]. The Cardiothoracic Surgical Trials Network randomly assigned 301 patients across multiple centers with moderate ischemic mitral regurgitation as determined by transthoracic echocardiography to CABG alone or CABG plus mitral-valve repair (combined procedure). The primary end point was the left ventricular end-systolic volume index (LVESVI). At 1 year, significant reductions in the LVESVI were observed in both arms of the trial, but the addition of a mitral-valve repair to CABG did not result in a higher degree of left ventricular reverse remodeling. 69 % of patients in the CABG-alone group had no mitral regurgitation or mild regurgitation at 1 year, as compared with 89 % of patients in the combined procedure group. These findings suggest that revascularization alleviates reversible ischemia in both groups. Clinical outcomes at 1 year, including functional status, quality of life, mortality, need for mitral-valve reoperation, and major adverse cardiac or cerebrovascular events did not differ significantly between groups. The combined-procedure group did experience a higher rate of serious neurologic events and had a higher rate of supraventricular arrhythmias, likely related to the atriotomy mandated by the mitral valve procedure. Given that the addition of a mitral-valve repair to CABG did not result in a higher degree of left ventricular remodeling, but did lead to an increased number of untoward events, the trial did not show a clinically meaningful advantage of adding mitral repair to CABG.
The results of these three studies are in conflict. Taken together in recent meta-analysis, the addition of MVR to CABG in patients with moderate ischemic mitral regurgitation does reduce residual MR grade in short-term outcomes, but does so with a simultaneous increase in morbidity and does not offer improvement in mortality or other clinically-meaningful metrics. The opposing outcomes reached by these trials may reflect differences in the end points assessed, the methods of classifying mitral regurgitation, and baseline characteristics such as rates of prior myocardial infarction and duration of mitral regurgitation from initial diagnosis to trial enrollment. While there may well exist a patient population with moderate ischemic mitral regurgitation that will optimally benefit from CABG with concomitant mitral valve procedure in terms of survival, functional status, or symptoms, this patient population has not yet been conclusively identified in the literature. At present, a concomitant mitral valve procedure should not be routinely added to CABG for the treatment of moderate ischemic mitral regurgitation. Longer-term analyses of these trials will be of great value and will yield further insight into the long-term impact of revascularization in IMR. In particular, further assessment of the degree to which CABG can yield both long-term reversal of ventricular remodeling and improvement in clinical outcomes will be of extreme importance. Taken together, the best available evidence to date supports isolated CABG in the short term for the treatment of moderate ischemic mitral regurgitation. These data should be considered in the context of the individual patient who is evaluated for the treatment of ischemic mitral regurgitation in order to best identify an appropriately risk-stratified and individualized treatment plan.
References
Lamas GA, Mitchell G, Flaker GC, et al. Clinical significance of mitral regurgitation after acute myocardial infarction. Circulation. 1997;96:827–33.
Aronson D, Goldsher N, Zukermann R, et al. Ischemic mitral regurgitation and risk of heart failure after myocardial infarction. Arch Intern Med. 2006;166:2362–8.
Carpentier A. Cardiac valve surgery—the “French correction”. J Thorac Cardiovasc Surg. 1983;86:323–37.
LA Pierard LP. The role of ischemic mitral regurgitation in the pathogenesis of acute pulmonary edema. N Engl J Med. 2004;351:1627–34.
Grigioni F, Detaint D, Avierinos J, Scott C, Tajik J, Enriquez-Sarano M. Contribution of ischemic mitral regurgitation to congestive heart failure after myocardial infarction. J Am Coll Cardiol. 2005;45(2):260–7.
Hung J, Guerrero J, Handschumacher MD, Supple G, et al. Reverse ventricular remodeling reduces ischemic mitral regurgitation: echo-guided device application in the beating heart. Circulation. 2002;106:2594–600.
Kaji S, Nasu M, Yamamuro A, et al. Annular geometry in patients with chronic ischemic mitral regurgitation. Three-dimensional magnetic resonance imaging study. Circulation. 2005;112(Suppl I):I-409–14.
Yu H-Y, Su M-Y, Liao T-Y, et al. Functional mitral regurgitation in chronic ischemic coronary artery disease: analysis of geometric alterations of mitral apparatus with magnetic resonance imaging. J Thorac Cardiovasc Surg. 2004;128:543–51.
Bursi F, Enriquez-Sarano M, Nkomo VT, Jacobsen SJ, Weston SA, Meverden RA, Roger VL. Heart failure and death after myocardial infarction in the community: the emerging role of mitral regurgitation. Circulation. 2005;111:295–301.
Lancellotti P, Troisfontaines P, Toussaint AC, Pierard LA. Prognostic importance of exercise-induced changes in mitral regurgitation in patients with chronic ischemic left ventricular dysfunction. Circulation. 2003;108:1713–17.
Grossi EA, Bizekis C, LaPietra A, et al. Late results of isolated mitral annuloplasty for “functional” ischemic mitral insufficiency. J Card Surg. 2001;16:328–32.
Johnston DR, Gillinov A, Blackstone EH, Griffin B, Stewart W, Sabik 3rd JF, Mihaljevic T, Svensson LG, Houghtaling PL, Lytle BW. Surgical repair of posterior mitral valve prolapse: implications for guidelines and percutaneous repair. Ann Thorac Surg. 2010;89(5):1385–94.
Bonow RO, Carabello B, Chatterjee K, et al. ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease) developed in collaboration with the Society of Cardiovascular Anesthesiologists endorsed by the Society for Cardiovascular Angiography and Interventions and the Society of Thoracic Surgeons. J Am Coll Cardiol. 2006;48:1–148.
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin 3rd JP, Guyton RA, O’Gara PT, Ruiz CE, Skubas NJ, Sorajja P, Sundt 3rd TM, Thomas JD. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Thorac Cardiovasc Surg. 2014;148(1):e1–e132.
Aklog L, Filsoufi F, Flores KQ, Chen RH, Cohn LH, Nathan NS, Byrne JG, Adams DH. Does coronary artery bypass grafting alone correct moderate ischemic mitral regurgitation? Circulation. 2001;104(90001):I-68–75. doi:10.1161/hc37t1.094706.
Kang DH, et al. Mitral valve repair versus revascularization alone in the treatment of ischemic mitral regurgitation. Circulation. 2006;114(1 Suppl):I499–503.
Lam B-K, Gillinov A, Blackstone EH, et al. Importance of moderate ischemic mitral regurgitation. Ann Thorac Surg. 2005;79:462–70.
Tolis GA, Korkolis D, Kopf GS, et al. Revascularization alone (without mitral valve repair) suffices in patients with advanced ischemic cardiomyopathy and mild-moderate mitral regurgitation. Ann Thorac Surg. 2002;74:1476–81.
Ellis SG, Whitlow P, Raymond RE, Schneider JP. Impact of mitral regurgitation on long-term survival after percutaneous coronary intervention. Am J Cardiol. 2002;89:315–18.
Campwala SZ, Bansal R, Wang N, et al. Mitral regurgitation progression following isolated coronary artery bypass surgery: frequency, risk factors, and potential prevention strategies. Eur J Cardiothorac Surg. 2006;29:348.
Buja P, Tarantini G, Bianco FD, et al. Moderate-to severe ischemic mitral regurgitation and multivessel coronary artery disease: impact of different treatment on survival and rehospitalization. Int J Cardiol. 2006;111:26–33.
Jones RH, Velazquez E, Michler RE, et al. Coronary bypass surgery with or without surgical ventricular reconstruction. N Engl J Med. 2009;360(17):1705–17.
Schroder JN, Williams M, Hata JA, et al. Impact of mitral valve regurgitation evaluated by intraoperative transesophageal echocardiography on long-term outcomes after coronary artery bypass grafting. Circulation. 2005;112(9_suppl):I-293–8. doi:10.1161/circulationaha.104.523472.
Grossi EA, Crooke G, Di Giorgi PL, et al. Impact of moderate functional mitral insufficiency in patients undergoing surgical revascularization. Circulation. 2006;114:I573.
Castleberry AW, Williams JB, Daneshmand MA, Honeycutt E, Shaw LK, Samad Z, Lopes RD, Alexander JH, Mathew JP, Velazquez EJ, Milano CA, Smith PK. Surgical revascularization is associated with maximal survival in patients with ischemic mitral regurgitation: a 20-year experience. Circulation. 2014;129(24):2547–56. doi:10.1161/CIRCULATIONAHA.113.005223.
Fattouch K, Guccione F, Sampognaro R, Panzarella G, Corrado E, Navarra E, Calvaruso D, Ruvolo G. POINT: efficacy of adding mitral valve restrictive annuloplasty to coronary artery bypass grafting in patients with moderate ischemic mitral valve regurgitation: a randomized trial. J Thorac Cardiovasc Surg. 2009;138(2):278–85.
Chan KM, Punjabi PP, Flather M, Wage R, Symmonds K, Roussin I, Rahman-Haley S, Pennell DJ, Kilner PJ, Dreyfus GD, Pepper JR. Coronary artery bypass surgery with or without mitral valve annuloplasty in moderate functional ischemic mitral regurgitation: final results of the Randomized Ischemic Mitral Evaluation (RIME) trial. Circulation. 2012;126(21):2502–10.
Smith PK, Puskas JD, Ascheim DD, Voisine P, Gelijns AC, Moskowitz AJ, Hung JW, Parides MK, Ailawadi G, Perrault LP, Acker MA, Argenziano M, Thourani V, Gammie JS, Miller MA, Pagé P, Overbey JR, Bagiella E, Dagenais F, Blackstone EH, Kron IL, Goldstein DJ, Rose EA, Moquete EG, Jeffries N, Gardner TJ, O’Gara PT, Alexander JH, Michler RE. Surgical treatment of moderate ischemic mitral regurgitation. N Engl J Med. 2014;371(23):2178–88. doi:10.1056/NEJMoa1410490.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Mulvihill, M.S., Smith, P.K. (2017). Treatment of Functional Ischemic Mitral Regurgitation by Coronary Artery Bypass Grafting. In: Chan, K. (eds) Functional Mitral and Tricuspid Regurgitation. Springer, Cham. https://doi.org/10.1007/978-3-319-43510-7_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-43510-7_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-43508-4
Online ISBN: 978-3-319-43510-7
eBook Packages: MedicineMedicine (R0)