Abstract
The existence of nano-sized extracellular vesicles including microvesicles and exosomes has been recognized for some time, but it is only relatively recently that their relevance to cardiovascular health and disease has begun to emerge. The discovery that they can deliver not only proteins but mRNA and miRNA to recipient cells suggested that they might regulate protein levels in the heart, and therefore cardiovascular function. Indeed, several studies have now shown that stem cells release extracellular vesicles which are taken up by recipient cells in vitro and modulate their function. They may therefore mediate some of the beneficial effects of stem cell implantation that have been observed. However many fundamental technical issues relating to their isolation and characterization remain to be overcome, and many aspects of their processing in vivo remain hypothetical. This chapter will examine the evidence for a role of exosome and microvesicles in local and distant communication mechanisms in the heart, particular in ischaemic heart failure and after myocardial infarction.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The existence of extracellular vesicles (EVs), such as microvesicles and exosomes has been recognized for some time, but it is only relatively recently that their relevance to cardiovascular health and disease has emerged. In particular, the search for potential mediators of the paracrine benefit of stem cells implanted in the heart has alighted on exosomes as interesting candidate molecules which may deliver protective and regenerative signals to the heart. This chapter will provide a background understanding of exosome and microvesicle biology and practical limitations in their analysis. Data suggesting these vesicles have beneficial effects in the heart will be discussed, as will the evidence for their possible involvement in the beneficial effects of stem cell in the heart.
2 Extracellular Vesicles
All eukaryotic cells are bound by a lipid bilayer membrane. If this pinches and buds off from the cell, it will cause the formation of small, bilayer lipid membrane vesicles containing some of the membrane-localized and cytosolic proteins of the “parent” cell . Budding such as this occurs during the regulated process of apoptosis, when the cytoskeleton constricts and the plasma membrane bulges out, causing the familiar “blebbing ” appearance of dying cells. Eventually these cells fragment into “ apoptotic bodies”, which are vesicles with a typical diameter of 1–5 μm. However, living cells also release vesicles from the plasma membrane via a process of shedding or budding (Fig. 9.1). These vesicles of a diameter of between 300 nm and 1 μm are called “microvesicles”. Even smaller are “exosomes”, which can form within endosomes by invagination of the endosome membrane to form internal vesicles of 30–100 nm in diameter. These endosomes containing numerous vesicles are called “multivesicular bodies ”. When the multivesicular body fuses with the plasma membrane, the exosomes are released from the cell into the extracellular space—a process of exocytosis that is believed to be energy dependent and involve members of the Rab family of small GTPases, although the exact mechanism is not well defined and may be cell type dependent (Raposo and Stoorvogel 2013; Yellon and Davidson 2014; Kowal et al. 2014).
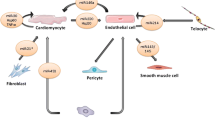
How the main types of EVs are formed. Microvesicles are released from the plasma membrane via a process of shedding or budding. Vesicles form within early endosomes by invagination of the endosome membrane, resulting in what are called multivesicular bodies. Once the multivesicular body fuses with the plasma membrane, the exosomes within are released from the cell into the extracellular space
Most, if not all, cell types appear to be capable of releasing exosomes and microvesicles (Raposo and Stoorvogel 2013), but some are more prolific than others. Platelets are some of the most prolific, as discovered by Peter Wolf in 1967, when he realised that “platelet dust ” released by activated platelets was actually microvesicles. At the time, however, they were considered to be cellular debris (Wolf 1967). In addition to releasing microvesicles, platelets also produce abundant numbers of exosomes (Heijnen et al. 1999). Exosomes were first discovered by examining the particles released by reticulocytes during their maturation into red blood cells (Johnstone et al. 1987). Again, they were initially thought to be waste products involved in “trash disposal” of proteins such as the transferrin receptor that were no longer required (Johnstone et al. 1987). Dendritic cells were also found to release exosomes, but these were shown to have a functional effect, since by presenting major histocompatibility complex antigens and generating an immune response, allogeneic donor-derived exosomes could induce immune tolerance to a heart allograft (Peche et al. 2003). In 2001, tumour cells were shown to release large numbers of exosomes which, when taken up by dendritic cells, could initiate a cytotoxic T-cell response (Wolfers et al. 2001). Exosomes have remained an intensive area of research in the cancer field, although the general opinion is now that exosomes aid the tumour cells in escaping immune surveillance by suppressing the immune response (Zhang and Grizzle 2011), and may facilitate metastasis (Hood et al. 2011).
In the cardiovascular system, exosomes have been isolated from primary adult rat cardiomyocytes (Gupta and Knowlton 2007; Wang et al. 2014) as well as the HL-1 mouse atrial cardiomyocyte cell line (Genneback et al. 2013; Waldenstrom et al. 2012). Matrix vesicles , which are secreted by vascular smooth muscle cells and are involved in initiating vascular calcification, were recently shown to be exosomes (Kapustin et al. 2015). Endothelial cells appear to release both exosomes and microvesicles (Deregibus et al. 2007). The production of exosomes is difficult to confirm in vivo. Vesicle-like structures resembling exosomes have been identified in electron micrographs of mouse heart (Barile et al. 2012), but in order to confirm their identity as exosomes and not unrelated vesicles or cross-sections of finger-like projections, for example, it would be necessary to immunolabel them with antibodies against known exosomal markers proteins . It is also very difficult to determine cell of origin in static images. Vascular smooth muscle cells secrete matrix vesicles that initiate vascular calcification . These vesicles were recently identified as exosomes containing some proteins to osteoblast-derived matrix vesicles (Kapustin et al. 2015), demonstrating an intriguing role for exosomes in vascular pathology. Finally, all body fluids contain large numbers of exosomes and microvesicles (Yellon and Davidson 2014; Thery et al. 2006). In the case of blood, the majority of EVs originate from platelets (Smalley et al. 2007), though erythrocytes, endothelium, lymphocytes or other blood cells also contribute.
3 Isolation of Extracellular Vesicles
In all studies of EVs the isolation method used is a primary consideration. This is particularly the case for exosomes, for which there is as yet no “perfect” method of purification. In fact, the International Society of Extracellular Vesicles (ISEV) released a position statement in 2014 recognizing that most isolation methods result in a mixture of different types of EVs (Lotvall et al. 2014). This is partly a consequence of the functional definition of exosomes as “vesicles that are released from multivesicular bodies”—once released from the cell there is no unique method to identify their origin. It is also experimentally challenging to use size to separate exosomes from microvesicles, which are only slightly larger, and from other vesicles, protein aggregates, and lipoproteins, which can overlap in size (Lotvall et al. 2014; Thery et al. 2006). Density gradient centrifugation is often used to separate different vesicle types based on their buoyant density (Lotvall et al. 2014; Thery et al. 2006). This can be useful for separating them from protein aggregates which do not float, but does not completely eliminate lipoproteins (HDL and LDL ) (Lotvall et al. 2014). Protein markers are commonly used to identify exosomes and microvesicles in preparations, and these are discussed under the following section. In general, cardiovascular researchers will be interested in isolating exosomes either from in vitro cultured cells , or from blood. The critical issues for each are somewhat different and are addressed separately.
With regard to the isolation of vesicles from in vitro cultured cells, most cell lines are grown in the presence of serum, which contains enormous quantities of exosomes. It is therefore essential to obtain specially filtered, certified exosome-free serum, or to prepare one’s own by overnight ultracentrifugation of the serum at 100,000 g or higher. Alternatively, it may be possible to grow some cell lines in the absence of serum during the experiment, although care must be taken that the cells remain healthy and do not undergo apoptosis, which will cause the release of apoptotic bodies that can co-purify with microvesicles or exosomes. The most efficient way of collecting exosomes from large volumes of cell medium is serial centrifugation. Sometimes, a filtration step is used first to concentrate the exosomes, however this is prone to artifactual generation of exosome-sized vesicles by fragmentation of the larger vesicles (Lotvall et al. 2014). In the process of serial centrifugation, the supernatant is collected and damaged cells and larger vesicles are removed by centrifugation at 10,000 for 30 min. The supernatant is then ultracentrifuged, typically at 100,000 g for 60 min to pellet exosomes (Hergenreider et al. 2012; Thery et al. 2006). As mentioned, the isolated exosome-rich fraction may be further purified by density gradient centrifugation on sucrose or iodixanol to enrich exosomes which have a density of between 1.13 and 1.19 g/mL (Yuana et al. 2014; Thery et al. 2006). This fraction is then diluted in buffer and re-centrifuged to remove the sucrose or iodixanol. One alternative method is to separate exosomes by size-exclusion chromatography (Boing et al. 2014). Although effective, this is less widely used due to lower throughput. A further technique is to use antibody affinity capture on beads or columns, although this requires that one is certain of the antigen marking the vesicles of interest, and the yield is much lower. Several companies now market reagents that can be used to rapidly precipitate exosomes from culture medium (or serum), the original being “Exoquick™ ”. Although yield is high, the purity is suspect (Van Deun et al. 2014), since it is difficult to wash the isolated exosome pellet washed without resorting to ultracentrifugation.
The isolation of EVs from blood is further complicated by pre-analytical considerations (Yuana et al. 2011). These are mostly designed to prevent or minimize the activation of platelets , which will otherwise release enormous numbers of microvesicles and exosomes which may influence the results. The clotting step involved in serum preparation causes massive platelet activation, which can be useful if large numbers of platelet vesicles are desired. However it is necessary to use plasma to obtain a selection of EVs representative of blood EVs (Yuana et al. 2011). Best practise is to use a relatedly large bore butterfly needle and draw blood into citrated Vacutainer® tubes, discarding the first tube. After mixing gently by inversion, the tube is centrifuged for at 1600×g for 20 min at room temperature (to minimize platelet activation) and the platelet-free plasma is carefully collected from the top. The standard protocol (Thery et al. 2006) is then to centrifuge the plasma at 10,000×g for 30 min at room temperature to pellet microvesicles. The supernatant containing exosomes is removed and ultracentrifuged at 100,000×g for 90 min at 4 °C. The supernatant is removed and the pellet resuspended in Ca2+ and Mg2+ free PBS containing citrate, and ultracentrifuged again. After a final wash, the pellet (which is not usually visible) can be vigorously resuspended for analysis of exosomes. Similar to exosomes isolated from tissue culture, density gradient purification may be performed to improve purity from protein aggregates, although contamination with high-density lipoproteins is very difficult to eliminate (Yuana et al. 2014).
Although some publications have used precipitation reagents such as Exoquick™ to isolate exosomes from blood, the standard protocol was designed for use with serum and must be adapted by the addition of Thromboplastin D in order to remove coagulative proteins and enable the use of plasma. Furthermore, the difficulty of washing the pellet containing exosomes is a major issue, and since blood contains very high concentrations of proteins, microvesicles, etc. some of these will remain in the final isolate. Despite these issues, some researchers have successfully used these methods to identify signature differences in exosome content between groups of individuals, which may be useful for diagnostic purposes. However, it must be taken into consideration that the exosomal origin of these samples remains unproven.
A promising new technique combining the convenience of precipitation and a pre-made kit with the improved separation of columns is the Exo-Spin™ kit , which appear to provide a convenient and reproducible means of eliminating >95 % of non-vesicular protein from biological fluid samples such as plasma (Welton et al. 2015).
4 Characterization of Exosomes and Microvesicles
Several approaches are necessary to unambiguously identify exosomes. Since their diameter is smaller than the wavelength of light, direct imaging of individual exosome using standard microscopy is impossible. Furthermore, they lie well below the detection threshold of flow cytometry (Nolte-’t Hoen et al. 2012). Flow cytometry is feasible for analysis of microvesicles but specialized modifications to flow cytometer equipment are necessary to optimize detection of particles at the lower range of size (Nolte-’t Hoen et al. 2012).
The most direct means of visualizing EVs is by transmission electron micrography after drying and uranyl acetate staining (Thery et al. 2006). Exosomes and other vesicles exhibit a shape that has been called “cup-shaped ” as the vesicles collapse when dried (Fig. 9.2a) (Thery et al. 2006; Raposo and Stoorvogel 2013). Contaminating objects can include lipoproteins (which appear spherical), protein aggregates, cellular organelles or viruses. Sometimes the background of the electron micrograph grid itself can resemble objects of the appropriate size range for exosomes, so it is useful to include a control grid without exosomes for comparison. Positive identification by electron micrograph is an important step in confirming the presence of exosomes and calculating the size distribution of a vesicle population.
Vesicle size can also be determined using dynamic light scattering (DLS) or nanoparticle tracking analysis (NTA) (Fig. 9.2b) (Dragovic et al. 2011). DLS can be effective but when a vesicle preparation contains populations of different sizes NTA is more appropriate as it is able to measure the size of individual particles.
Exosomes contain particular protein markers as a consequence of their release mechanism from cells, and these are often used to aid their identification (or utilized for vesicle purification by antibody affinity). Foremost amongst these proteins are the tetraspanins, particularly CD9, CD63, CD81, although it must be recognized that other vesicles can express these molecules as well (Kowal et al. 2014). It may be possible to detect these proteins by Western blot analysis, although non-reducing gels should be used because the tetraspanins are heavily glycosylated (Thery et al. 2006). If it is difficult to obtain sufficient protein for a Western blot, an effective alternative is to adhere the exosomes onto 4 μm diameter latex beads, incubate them with fluorescent antibodies against tetraspanins or other marker proteins, and measure the fluorescence of individual beads separated by flow cytometry (Thery et al. 2006). Modern flow cytometers are quite capable of analysing most microvesicles (Ayers et al. 2014), although the smallest microvesicles might be beyond the range of detection without careful optimization of the flow cytometer.
It is recommended to use several of the above approaches in order to confirm the identity and origin of the EVs.
5 Extracellular Vesicles Can Transfer Proteins and RNA
EVs have been demonstrated to possess a number of interesting capabilities including the ability to deliver proteins and RNA to cells. There is some evidence that this also occurs in vivo , despite this being much more difficult to prove. One of the first proteins demonstrated to be transferred to recipient cells in vitro was tissue factor (TF) . TF was released from platelets within microvesicles that were subsequently incorporated into the membranes of monocytes and other cells where TF exerts its biological effects (Del Conde et al. 2005; Scholz et al. 2002). An important paper demonstrated that microvesicles from tumour cells could transfer a truncated, oncogenic form of the epidermal growth factor receptor between cells, leading to activation of transforming signalling pathways (MAPK and Akt) and horizontal propagation of oncogenes and their associated transforming phenotype (Al-Nedawi et al. 2008).
Exosomes are usually found to contain primarily cytosolic and membrane-localized proteins (Mathivanan et al. 2010). Certain proteins are found almost universally in exosomes due to their being part of the packaging and/or release pathway. These include the proteins Alix, TSP101, HSP70 and tetraspanin family proteins CD9, CD63, CD81, etc., which are therefore often used as markers proteins to indicate the presence of exosomes, as described in the previous section. The degree of sorting of proteins into exosomes and microvesicles appears to be greater when the proteins contain domains that link them to the plasma membrane (Shen et al. 2011), presumably as this makes them more likely to be incorporated into EVs as the plasma membrane buds off or invaginates to produce multivesicular bodies . There is some evidence that the protein and RNA content of exosomes depends on the cellular state (de Jong et al. 2012).
Similar to microvesicles, exosomes can not only deliver signalling proteins to cells, but be taken up by other cells, incorporating the protein into the recipient membrane. For example, the Notch ligand Delta-like 4 (Dll4) is incorporated into endothelial exosomes which can transfer the Dll4 protein to other endothelial cells and incorporate it into their cell membrane, resulting in an inhibition of Notch signalling and altered angiogenesis (Sheldon et al. 2010). Of particular interest are the examples in which exosomes have been shown to deliver protective proteins to recipient cells. αB crystallin was shown to be secreted from human retinal pigment epithelium in exosomes, and taken up by adjacent photoreceptors in mouse retinal explants exposed to oxidative stress (Sreekumar et al. 2010). An important theoretical consideration is the extent to which transfer of a relatively small number of copies of a protein can be realistically expected to have a physiological effect in the recipient cell in vivo . In this respect, it is easier to imagine transfer of small quantities of mRNA or miRNA being capable of having more dramatic effects.
In a seminal paper, Valadi et al, were first to show that exosomes can transfer mRNA and miRNA between cells (Valadi et al. 2007). This study used mast cells to demonstrate the transfer of functional mRNAs that were subsequently translated in the recipient cells. In an important control experiment, exosomes were pre-treated with RNAse and trypsin, to demonstrate that mRNA was protected within the vesicles and not simply associated or co-purified. Tumour-derived microvesicles were also shown to transfer mRNA to recipient cells that could be translated (Skog et al. 2008).
The profile of miRNAs contained within exosomes appears to differ according to the cell type of origin. For example, C2C12 myoblasts and C2C12 differentiated into myotubes released exosomes with different miRNA profiles (Forterre et al. 2014). Interestingly, the miRNA profile was different from that of the donor cells, indicating that there is selective packaging of miRNA into exosomes (Forterre et al. 2014). The mechanism of selective sorting of miRNA into exosomes is only beginning to be unravelled, but is believed to involve recognition of particular sequence motifs by sumoylated heterogeneous nuclear ribonucleoprotein A2B1 (hnRNPA2B1) (Villarroya-Beltri et al. 2013). The exosomes secreted by C2C12 myotubes were functional, being able to suppress expression of Sirt1 when taken up by myoblasts, potentially modulating metabolic homeostasis and contributing to the commitment of myoblasts during differentiation (Forterre et al. 2014).
Cardiac fibroblasts secrete miRNA-enriched exosomes, including miR-21-3p—a passenger strand miRNA which normally undergoes intracellular degradation (Bang et al. 2014). However, when these exosomes were taken up by neonatal cardiomyocytes in co-culture, the cardiomyocytes increased in size indicating a hypertrophic response (Bang et al. 2014). Thus it appears that different cells in the heart may communicate to each other via exosomes. A further significant exchange of miRNAs between cells of the cardiovascular system was shown to occur between endothelial cells and smooth muscle cells . In this case, EVs were secreted by endothelial cells that had been stimulated by shear stress, which is known to be atheroprotective (Hergenreider et al. 2012). The EVs were taken up into smooth muscle cells in co-culture, delivering miR-143/145 and controlling the expression of target genes (Hergenreider et al. 2012). They also reduced atherosclerotic lesion formation in the aorta of ApoE(−/−) mice (Hergenreider et al. 2012). Since a maximum centrifugation speed of 20,500×g was used to pellet EVs, and the size range of most of the vesicles on electron micrographs ranged between 60 and 130 nm, they were referred to conservatively as “extracellular vesicles”, but presumably contained a mix of exosomes and microvesicles .
In view of the RNA content of EVs which is related to the cell type of origin, and can alter in pathological settings, they have become an attractive source of biomarkers for profiling and identification of disease markers (Cheng et al. 2014; Jansen et al. 2014), as has been reviewed elsewhere (Gaceb et al. 2014).
5.1 Extracellular Vesicles Can Transfer Proteins and RNA
EVs have been demonstrated to possess a number of interesting capabilities including the ability to deliver proteins and RNA to cells. There is some evidence that this also occurs in vivo, despite this being much more difficult to prove. One of the first proteins demonstrated to be transferred to recipient cells in vitro was tissue factor (TF). TF was released from platelets within microvesicles that were subsequently incorporated into the membranes of monocytes and other cells where TF exerts its biological effects (Del Conde et al. 2005; Scholz et al. 2002). An important paper demonstrated that microvesicles from tumour cells could transfer a truncated, oncogenic form of the epidermal growth factor receptor between cells, leading to activation of transforming signalling pathways (MAPK and Akt) and horizontal propagation of oncogenes and their associated transforming phenotype (Al-Nedawi et al. 2008).
Exosomes are usually found to contain primarily cytosolic and membrane-localized proteins (Mathivanan et al. 2010). Certain proteins are found almost universally in exosomes due to their being part of the packaging and/or release pathway. These include the proteins Alix, TSP101, HSP70 and tetraspanin family proteins CD9, CD63, CD81, etc., which are therefore often used as markers proteins to indicate the presence of exosomes, as described in the previous section. The degree of sorting of proteins into exosomes and microvesicles appears to be greater when the proteins contain domains that link them to the plasma membrane (Shen et al. 2011), presumably as this makes them more likely to be incorporated into EVs as the plasma membrane buds off or invaginates to produce multivesicular bodies. There is some evidence that the protein and RNA content of exosomes depends on the cellular state (de Jong et al. 2012).
Similar to microvesicles, exosomes can not only deliver signalling proteins to cells, but be taken up by other cells, incorporating the protein into the recipient membrane. For example, the Notch ligand Delta-like 4 (Dll4) is incorporated into endothelial exosomes which can transfer the Dll4 protein to other endothelial cells and incorporate it into their cell membrane, resulting in an inhibition of Notch signalling and altered angiogenesis (Sheldon et al. 2010). Of particular interest are the examples in which exosomes have been shown to deliver protective proteins to recipient cells. αB crystallin was shown to be secreted from human retinal pigment epithelium in exosomes, and taken up by adjacent photoreceptors in mouse retinal explants exposed to oxidative stress (Sreekumar et al. 2010). An important theoretical consideration is the extent to which transfer of a relatively small number of copies of a protein can be realistically expected to have a physiological effect in the recipient cell in vivo. In this respect, it is easier to imagine transfer of small quantities of mRNA or miRNA being capable of having more dramatic effects.
In a seminal paper, Valadi et al, were first to show that exosomes can transfer mRNA and miRNA between cells (Valadi et al. 2007). This study used mast cells to demonstrate the transfer of functional mRNAs that were subsequently translated in the recipient cells. In an important control experiment, exosomes were pre-treated with RNAse and trypsin, to demonstrate that mRNA was protected within the vesicles and not simply associated or co-purified. Tumour-derived microvesicles were also shown to transfer mRNA to recipient cells that could be translated (Skog et al. 2008).
The profile of miRNAs contained within exosomes appears to differ according to the cell type of origin. For example, C2C12 myoblasts and C2C12 differentiated into myotubes released exosomes with different miRNA profiles (Forterre et al. 2014). Interestingly, the miRNA profile was different from that of the donor cells, indicating that there is selective packaging of miRNA into exosomes (Forterre et al. 2014). The mechanism of selective sorting of miRNA into exosomes is only beginning to be unravelled, but is believed to involve recognition of particular sequence motifs by sumoylated heterogeneous nuclear ribonucleoprotein A2B1 (hnRNPA2B1) (Villarroya-Beltri et al. 2013). The exosomes secreted by C2C12 myotubes were functional, being able to suppress expression of Sirt1 when taken up by myoblasts, potentially modulating metabolic homeostasis and contributing to the commitment of myoblasts during differentiation (Forterre et al. 2014).
Cardiac fibroblasts secrete miRNA-enriched exosomes, including miR-21-3p—a passenger strand miRNA which normally undergoes intracellular degradation (Bang et al. 2014). However, when these exosomes were taken up by neonatal cardiomyocytes in co-culture, the cardiomyocytes increased in size indicating a hypertrophic response (Bang et al. 2014). Thus it appears that different cells in the heart may communicate to each other via exosomes. A further significant exchange of miRNAs between cells of the cardiovascular system was shown to occur between endothelial cells and smooth muscle cells. In this case, EVs were secreted by endothelial cells that had been stimulated by shear stress, which is known to be atheroprotective (Hergenreider et al. 2012). The EVs were taken up into smooth muscle cells in co-culture, delivering miR-143/145 and controlling the expression of target genes (Hergenreider et al. 2012). They also reduced atherosclerotic lesion formation in the aorta of ApoE(−/−) mice (Hergenreider et al. 2012). Since a maximum centrifugation speed of 20,500 g was used to pellet EVs, and the size range of most of the vesicles on electron micrographs ranged between 60 and 130 nm, they were referred to conservatively as “extracellular vesicles”, but presumably contained a mix of exosomes and microvesicles.
In view of the RNA content of EVs which is related to the cell type of origin, and can alter in pathological settings, they have become an attractive source of biomarkers for profiling and identification of disease markers (Cheng et al. 2014; Jansen et al. 2014), as has been reviewed elsewhere (Gaceb et al. 2014).
6 Exosomes as a Potential Therapy for Cardiovascular Disease
Coronary artery disease is a major challenge facing health care systems around the world. In 2004, ischaemic heart disease was responsible for 7.2 million deaths, making it the leading cause of death. Myocardial infarction is a major cause of mortality and morbidity in patients with coronary artery disease. Heart failure often develops subsequently, depending on the extent of myocardial infarction. Therefore, treatment modalities that protect the heart from injury can be expected to result in major improvements not only in early mortality but on-going morbidity. Currently early reperfusion (restoration of normal coronary flow) remains the mainstay of treatment for those suffering an acute myocardial infarction. However, reperfusion also causes injury to heart muscle in what is termed “lethal reperfusion injury ” (Hausenloy and Yellon 2013). Final infarct size in patients with ST elevation myocardial infarction predicts long-term clinical outcome (Lonborg et al. 2013). Therefore, finding ways to minimize reperfusion injury is one approach that would be expected to improve the overall, long-term management of patients presenting with myocardial infarction (Braunwald and Kloner 1985; Piper et al. 1998; Hausenloy and Yellon 2013). Another theoretical approach would be to replace the contractile cardiomyocytes that were lost, by the use of stem cell therapy. As discussed elsewhere in this book, however, the results of this approach have been largely disappointing. On the other hand, some improvements in cardiac function have been observed after stem cell therapy , even in the absence of new cardiomyocytes formation . Since some benefit could also be observed experimentally after injection of conditioned medium from stem cells, the “paracrine” hypothesis developed in which it is proposed that engrafted stem cells release cytokines, growth factors and other proteins which act on cardiomyocytes and other myocardial proteins to confer improvements in function. Stem cells release a host of different factors (“the secretome”), many of which are pro-survival and proangiogenic (Makridakis et al. 2013). While many of these are released via the usual protein secretion pathways, the possibility was raised that they might be released together associated with exosomes, enabling a co-ordinated, and perhaps directed response to multiple factors.
The first study to examine whether exosomes released from stem cells are cardioprotective was performed in 2010 using an acute model of myocardial infarction . Timmers et al. had previously showed that conditioned medium from human ESC-derived mesenchymal stem cells (ESC-MSC) reduced infarct size in isolated, perfused mouse hearts subject to ischaemia and reperfusion (Timmers et al. 2007). The same group then purified exosomes from the conditioned medium by HPLC size-exclusion fractionation , and confirmed that these exosomes were sufficient to confer cardioprotection both in vitro and in vivo (Lai et al. 2010a). Long term protection with improved function after 28 days was observed after injection of at least 4 μg/kg into mice via the tail vein, just before reperfusion (Arslan et al. 2013). Interestingly, it appears that the exosomes had to be intact to induce protection, since they were no longer protective after homogenization (Arslan et al. 2013), although whether exosomes are disrupted after homogenization is not clear and was not demonstrated. Exosome administration caused an increase in the activity of cardioprotective kinases Akt and GSK3α/β after 1 h, and these remained activated until the day following treatment (Arslan et al. 2013). These kinases are known to be highly cardioprotective (Hausenloy et al. 2005). Significantly, there was also a reduction in myocardial oxidative stress, and local and systemic inflammation (Arslan et al. 2013). Exosomes isolated from MSC cells overexpressing GATA4 have also been shown to restore cardiac contractile function and reduce infarct size when injected intramyocardially at the time of infarction in a rat model (Yu et al. 2014). An increase in miR-19a was detected in the treated hearts, although it is difficult to be certain that this originated from the MSC exosomes and was not a response to treatment with exosomes (Yu et al. 2014). In turn, levels of PTEN, a predicted target of miR-19a, were reduced and Akt and ERK activation was increased (Yu et al. 2014). As an alternative cell source for large-scale production of exosomes which does not require ES cells, MSC cultures were developed from limb, kidney and liver tissues of three first trimester aborted foetuses. Despite absence of pluripotency-associated markers, these MSC were also an excellent source of cardioprotective exosomes (Lai et al. 2010b). Of interest, the protective pathways activated by exosomes appear to be applicable to other organs, since MSC exosomes (from human umbilical cord) have also been shown to protect against cisplatin-induced renal oxidative stress and apoptosis via ERK (Zhou et al. 2013), and to promote functional recovery after stroke in rats (Xin et al. 2013). Furthermore, it may be that protection is not limited to exosomes, as microvesicles derived from human adult mesenchymal stem cells also protected against ischaemia and reperfusion kidney injury (Gatti et al. 2011).
MSC are not the only type of stem cell that have been shown to release exosomes with beneficial cardiovascular effects. Intramyocardial injection of cardiac progenitor cells (CPCs) isolated from adult hearts improved cardiac function in animal models of myocardial infarction (Messina et al. 2004; Smith et al. 2007), although paracrine effects were found to contribute to the majority of the effect (Chimenti et al. 2010). Subsequently, exosomes from murine CPCs were found to reduce apoptosis when injected intramyocardially during ischaemia (Chen et al. 2013). In this study, however, exosomes were isolated by PEG precipitation (Chen et al. 2013), and therefore the more appropriate control would have been injection with PEG alone. Another study used several alternative methods of isolation of CPC-EVs: Exoquick™ precipitation, ultracentrifugation, or size-exclusion, column purification (Barile et al. 2014). Although more of tetraspanin marker proteins CD63 and CD81 were measured using Exoquick™, the three techniques were not rigorously compared, and as mentioned above, the Exoquick™ technique has some drawbacks—particularly the inability to wash the pellet to remove the Exoquick™ solution from the exosomes. Injection of these CPCs-EVs into the hearts of rats subject to permanent coronary artery ligation reduced cardiomyocyte apoptosis and scar size, increased the amount of viable tissue in the infarct area, increased blood vessel density, and prevented the loss of impairment of ventricular function between day 2 and day 7 ((Barile et al. 2014). In contrast, exosomes isolated from normal human dermal fibroblasts exhibited no benefit (Barile et al. 2014). Gray et al. used ultracentrifugation to purify exosomes from CPCs, and found that the rate of secretion of exosomes was increased after 3–12 h hypoxia (Gray et al. 2015). Intramyocardial injection of 5 μg/kg of the exosomes produced after hypoxia improved cardiac function and reduced fibrosis 21 days after ischaemia and reperfusion injury in rats (Gray et al. 2015). Interestingly, however, there was no benefit with exosomes from normoxic CPCs (Barile et al. 2014). The exosomes released after hypoxia had an altered miRNA content, and co-regulated miRNA with a beneficial profile were identified (Gray et al. 2015). Although cardiac endothelial cells and fibroblasts took up fluorescently stained exosomes in vitro, uptake was minimal in primary rat cardiomyocytes (Gray et al. 2015), suggesting either that they deliver miRNA directly to the former cells types, or that they interact with surface receptors on cardiomyocytes without delivering miRNA intracellularly. Thus, the exact mechanism of functional benefit conferred by exosomes remains unclear.
Various avenues are being explored to increase the survival and engraftment of stem cells once they have been injected into the heart. Recently, CPCs were co-delivered into ischemic mouse myocardium with a nonviral minicircle plasmid carrying HIF1, a transcription factor that mediates adaptive responses to ischemia (Ong et al. 2014). As intended, this increased the survival of CPCs after injection, and also significantly improved cardiac function 6 weeks later (Ong et al. 2014). The authors hypothesized that HIF1 was being expressed by cardiac endothelial cells, thereby modulating the host microenvironment such that CPC survival was increased. Exosomes were purified from endothelial cells overexpressing HIF1 and found to have a higher content of miR-126 and miR-210 (Ong et al. 2014). In in vitro experiments, these exosomes were taken up by CPCs, leading to activated pro-survival kinases and to a switch towards glycolysis in recipient CPCs, giving them increased tolerance when subjected to in vitro hypoxic stress (Ong et al. 2014). Antagomirs to these miRs blocked the protective effects of exosomes (Ong et al. 2014). This suggests the interesting possibility that endothelial cells can support CPS survival by exosomal transfer of miRNA.
Bone marrow CD34+ stem cells are another type of stem cell that been associated with reduced angina, improved exercise time, and reduced amputation rates when delivered to ischaemic myocardium . Exosomes isolated from the conditioned medium of CD34+ cells exerted a proangiogenic paracrine activity, both in vitro and in vivo when tested using Matrigel plug and corneal assays (Sahoo et al. 2011). In contrast, CD34-mononuclear cells did not have this effect. Mackie et al. attempted to enhance the angiogenic quality of CD34 cells by genetically modifying them to express the sonic hedgehog (Shh) protein, a developmental morphogenic protein that is also proangiogenic (see Sect. 10.6.1). CD34+ were first engineered to express Shh. When injected into the infarct border zone in mice after myocardial infarction , infarct size was reduced and border zone capillary density was increased 4 weeks later (Mackie et al. 2012). This was paralleled by improved ventricular dilation and cardiac function. In vitro studies in cells were performed to demonstrate that Shh was released from the CD34+Shh cells in exosomes, and could be transferred to recipient cells and (modestly) activate transcription. When exosomes were isolated from conditioned medium of CD34 + Shh cells using differential ultracentrifugation they were found to contain Shh (Mackie et al. 2012). When injected into mice, were also found to be protective (Mackie et al. 2012). In contrast, CD34+ cells or their exosomes showed no benefit (Mackie et al. 2012).
In all of the above studies, it is important to recall that stem cells also secrete numerous other proteins that are not associated with vesicles. These include pro-inflammatory, anti-inflammatory and pleiotropic cytokines, chemokines, angiogenic factors, growth factors, growth factor-binding proteins, extracellular matrix proteins and extracellular matrix remodelling enzymes (Makridakis et al. 2013; Skalnikova et al. 2011). Many of these, particularly the growth factors and chemokines have been shown to be cardioprotective (Hausenloy and Yellon 2009; Hausenloy et al. 2010), or, along with angiogenic factors , to improve long term recovery after infarction. Thus, there is still great interest in using stem cells as localized “factories” for production of trophic factors to maintain or improve cardiac function. On the other hand, there are some clear potential benefits to the use of exosomes as delivery vehicles (Table 9.1). Foremost amongst these, since living cells are not injected, is the absence of risk of transformation and tumourigenicity associated with the injection of stem cells. Since injected exosomes are only present transiently, and are of low immunogenicity , there should not be any need for immunosuppression. However, this does mean that there is only a transient response, and repeated administration of exosomes may be required for prolonged benefit. As there have not yet been any clinical trials administering exosomes to humans, however, most of these considerations are hypothetical at present.
The extent to which EVs from different cell types have different functions and activities is still under investigation. In some of the studies above using EVs isolated from stem cells, EVs from other cell types such as fibroblasts were used as negative controls and found not to have beneficial cardiovascular effects in the models used. Exosomes from CD34+ cells were only protective if isolated from cells overexpressing Shh (Mackie et al. 2012). This data must be reconciled with the fact that there are huge numbers of EVs present in the blood of all individuals, which could potentially deliver miRNA or receptor-ligand mediated signals to the heart. This possibility was addressed by isolating plasma exosomes from healthy individuals using differential ultracentrifugation and testing whether they were cardioprotective in rat models of ischaemia and reperfusion in vitro and in vivo (Vicencio et al. 2015). Indeed, exosomes from plasma were strongly cardioprotective, reducing infarct size after injection of all of the exosomes isolated from a donor rat into a recipient rat (Vicencio et al. 2015). Plasma exosomes were similarly protective in an isolated perfused rat heart model (Vicencio et al. 2015). Remote ischaemic preconditioning (RIC) is a technique in which several short episodes of non-lethal ischaemia and reperfusion are applied to the limb by occluding the blood flow using a tourniquet or other means (Hausenloy and Yellon 2008). In animal models this has been shown to induce protection against lethal myocardial ischaemia and reperfusion injury, via a mechanism that involves the transmission of a humoral blood factor to the heart to activate protective kinase signalling pathways (Hausenloy and Yellon 2008). As yet, the identity of the humoral factor is unclear although evidence for several candidates has been presented, including SDF-1α and Il-10 (Davidson et al. 2013; Cai et al. 2012). As vehicles able to deliver multiple signals between cells, EVs were proposed as possible candidates for carriers of the cardioprotective factor released by RIC (Yellon and Davidson 2014). A study by Giricz et al. suggested that this may be the case, since RIC was not effective when EVs were removed from medium containing the factor (Giricz et al. 2014). More recently, the numbers of EVs in the blood was shown to be significantly increased after RIC in both rats and healthy human volunteers (Vicencio et al. 2015). Experiments performed using primary adult rat cardiomyocytes demonstrated a dose dependency. The EVs released after RIC were found to be marginally, but not significantly more protective in this model (Vicencio et al. 2015), suggesting that the increase in EV number was not sufficient to explain the mechanism of RIC. However, the fact that plasma EVs in themselves were cardioprotective is important and may suggest that they signal continuously to the heart, modulating protective state. This is difficult to prove without specific methods of inhibiting EV release in vivo. This study also examined the mechanism of protection using inhibitors and Western blot analysis and identified HSP70 in the exosome membrane as essential for the protection (Vicencio et al. 2015). Protection was blocked by inhibitors of TLR4, or ERK1/2, leading to the proposal of a model in which HSP70 is recognized by TLR4, which activates ERK1/2, p38MAPK and downstream phosphorylation of small heat shock protein HSP27 (Vicencio et al. 2015). TLR4 is part of the innate immune system, and strong activation by its ligands from bacteria leads to a cell damage response and can cause cell death. However, mild activation appears to be protective (Mathur et al. 2011; Zhang et al. 2013).
7 Conclusion and Perspectives
In conclusion, exosomes and microvesicles represent an exciting new frontier in cardiovascular research. Although data suggests that they possess an important role in cell-to-cell communication, this remains to be established and defined using rigorously purified vesicle populations for which techniques are yet to be established. Furthermore, crucial experiments to define their role in vivo require the ability to inhibit EV production and/or uptake in vivo—techniques which are not yet available. However, the intense interest in this area of research, and the eagerness to identify the mechanism of potential paracrine benefit by stem cells are two factors likely to contribute to the achievement of remarkable and rapid advancements in this field.
References
Al-Nedawi K, Meehan B, Micallef J, Lhotak V, May L, Guha A, Rak J (2008) Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat Cell Biol 10(5):619–624. doi:10.1038/ncb1725
Arslan F, Lai RC, Smeets MB, Akeroyd L, Choo A, Aguor EN, Timmers L, van Rijen HV, Doevendans PA, Pasterkamp G, Lim SK, de Kleijn DP (2013) Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/Akt pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res 10(3):301–312. doi:10.1016/j.scr.2013.01.002
Ayers L, Harrison P, Kohler M, Ferry B (2014) Procoagulant and platelet-derived microvesicle absolute counts determined by flow cytometry correlates with a measurement of their functional capacity. J Extracell Vesicles 3. doi:10.3402/jev.v3.25348
Bang C, Batkai S, Dangwal S, Gupta SK, Foinquinos A, Holzmann A, Just A, Remke J, Zimmer K, Zeug A, Ponimaskin E, Schmiedl A, Yin X, Mayr M, Halder R, Fischer A, Engelhardt S, Wei Y, Schober A, Fiedler J, Thum T (2014) Cardiac fibroblast-derived microRNA passenger strand-enriched exosomes mediate cardiomyocyte hypertrophy. J Clin Invest 124(5):2136–2146. doi:10.1172/JCI70577
Barile L, Gherghiceanu M, Popescu LM, Moccetti T, Vassalli G (2012) Ultrastructural evidence of exosome secretion by progenitor cells in adult mouse myocardium and adult human cardiospheres. J Biomed Biotechnol 2012:354605. doi:10.1155/2012/354605
Barile L, Lionetti V, Cervio E, Matteucci M, Gherghiceanu M, Popescu LM, Torre T, Siclari F, Moccetti T, Vassalli G (2014) Extracellular vesicles from human cardiac progenitor cells inhibit cardiomyocyte apoptosis and improve cardiac function after myocardial infarction. Cardiovasc Res 103(4):530–541. doi:10.1093/cvr/cvu167
Boing AN, van der Pol E, Grootemaat AE, Coumans FA, Sturk A, Nieuwland R (2014) Single-step isolation of extracellular vesicles by size-exclusion chromatography. J Extracell Vesicles 3. doi:10.3402/jev.v3.23430
Braunwald E, Kloner RA (1985) Myocardial reperfusion: a double-edged sword? J Clin Invest 76(5):1713–1719
Cai ZP, Parajuli N, Zheng X, Becker L (2012) Remote ischemic preconditioning confers late protection against myocardial ischemia-reperfusion injury in mice by upregulating interleukin-10. Basic Res Cardiol 107(4):277. doi:10.1007/s00395-012-0277-1
Chen L, Wang Y, Pan Y, Zhang L, Shen C, Qin G, Ashraf M, Weintraub N, Ma G, Tang Y (2013) Cardiac progenitor-derived exosomes protect ischemic myocardium from acute ischemia/reperfusion injury. Biochem Biophys Res Commun. doi:10.1016/j.bbrc.2013.01.015
Cheng L, Sharples RA, Scicluna BJ, Hill AF (2014) Exosomes provide a protective and enriched source of miRNA for biomarker profiling compared to intracellular and cell-free blood. J Extracell Vesicles 3. doi:10.3402/jev.v3.23743
Chimenti I, Smith RR, Li TS, Gerstenblith G, Messina E, Giacomello A, Marban E (2010) Relative roles of direct regeneration versus paracrine effects of human cardiosphere-derived cells transplanted into infarcted mice. Circ Res 106(5):971–980. doi:10.1161/CIRCRESAHA.109.210682
Davidson SM, Selvaraj P, He D, Boi-Doku C, Yellon RL, Vicencio JM, Yellon DM (2013) Remote ischaemic preconditioning involves signalling through the SDF-1alpha/CXCR4 signalling axis. Basic Res Cardiol 108(5):377. doi:10.1007/s00395-013-0377-6
de Jong OG, Verhaar MC, Chen Y, Vader P, Gremmels H, Posthuma G, Schiffelers RM, Gucek M, van Balkom BWM (2012) Cellular stress conditions are reflected in the protein and RNA content of endothelial cell-derived exosomes. J Extracell Vesicles 1. doi:10.3402/jev.v1i0.18396
Del Conde I, Shrimpton CN, Thiagarajan P, Lopez JA (2005) Tissue-factor-bearing microvesicles arise from lipid rafts and fuse with activated platelets to initiate coagulation. Blood 106(5):1604–1611. doi:10.1182/blood-2004-03-1095
Deregibus MC, Cantaluppi V, Calogero R, Lo Iacono M, Tetta C, Biancone L, Bruno S, Bussolati B, Camussi G (2007) Endothelial progenitor cell derived microvesicles activate an angiogenic program in endothelial cells by a horizontal transfer of mRNA. Blood 110(7):2440–2448. doi:10.1182/blood-2007-03-078709
Dragovic RA, Gardiner C, Brooks AS, Tannetta DS, Ferguson DJ, Hole P, Carr B, Redman CW, Harris AL, Dobson PJ, Harrison P, Sargent IL (2011) Sizing and phenotyping of cellular vesicles using nanoparticle tracking analysis. Nanomedicine 7(6):780–788. doi:10.1016/j.nano.2011.04.003
Forterre A, Jalabert A, Chikh K, Pesenti S, Euthine V, Granjon A, Errazuriz E, Lefai E, Vidal H, Rome S (2014) Myotube-derived exosomal miRNAs downregulate Sirtuin1 in myoblasts during muscle cell differentiation. Cell Cycle 13(1):78–89. doi:10.4161/cc.26808
Gaceb A, Martinez MC, Andriantsitohaina R (2014) Extracellular vesicles: new players in cardiovascular diseases. Int J Biochem Cell Biol 50:24–28. doi:10.1016/j.biocel.2014.01.018
Gatti S, Bruno S, Deregibus MC, Sordi A, Cantaluppi V, Tetta C, Camussi G (2011) Microvesicles derived from human adult mesenchymal stem cells protect against ischaemia-reperfusion-induced acute and chronic kidney injury. Nephrol Dial Transplant 26(5):1474–1483. doi:10.1093/ndt/gfr015
Genneback N, Hellman U, Malm L, Larsson G, Ronquist G, Waldenstrom A, Morner S (2013) Growth factor stimulation of cardiomyocytes induces changes in the transcriptional contents of secreted exosomes. J Extracell Vesicles 2. doi:10.3402/jev.v2i0.20167
Giricz Z, Varga ZV, Baranyai T, Sipos P, Paloczi K, Kittel A, Buzas E, Ferdinandy P (2014) Cardioprotection by remote ischemic preconditioning of the rat heart is mediated by extracellular vesicles. J Mol Cell Cardiol 68:75–78. doi:10.1016/j.yjmcc.2014.01.004
Gray WD, French KM, Ghosh-Choudhary S, Maxwell JT, Brown ME, Platt MO, Searles CD, Davis ME (2015) Identification of therapeutic covariant microRNA clusters in hypoxia-treated cardiac progenitor cell exosomes using systems biology. Circ Res 116(2):255–263. doi:10.1161/CIRCRESAHA.116.304360
Gupta S, Knowlton AA (2007) HSP60 trafficking in adult cardiac myocytes: role of the exosomal pathway. Am J Physiol Heart Circ Physiol 292(6):H3052–H3056. doi:10.1152/ajpheart.01355.2006
Hausenloy DJ, Yellon DM (2008) Remote ischaemic preconditioning: underlying mechanisms and clinical application. Cardiovasc Res 79(3):377–386. doi:10.1093/cvr/cvn114
Hausenloy DJ, Yellon DM (2009) Cardioprotective growth factors. Cardiovasc Res 83(2):179–194. doi:10.1093/cvr/cvp062
Hausenloy DJ, Yellon DM (2013) Myocardial ischemia-reperfusion injury: a neglected therapeutic target. J Clin Invest 123(1):92–100. doi:10.1172/JCI62874
Hausenloy DJ, Tsang A, Yellon DM (2005) The reperfusion injury salvage kinase pathway: a common target for both ischemic preconditioning and postconditioning. Trends Cardiovasc Med 15(2):69–75
Hausenloy DJ, Baxter G, Bell R, Botker HE, Davidson SM, Downey J, Heusch G, Kitakaze M, Lecour S, Mentzer R, Mocanu MM, Ovize M, Schulz R, Shannon R, Walker M, Walkinshaw G, Yellon DM (2010) Translating novel strategies for cardioprotection: the Hatter workshop recommendations. Basic Res Cardiol 105(6):677–686. doi:10.1007/s00395-010-0121-4
Heijnen HF, Schiel AE, Fijnheer R, Geuze HJ, Sixma JJ (1999) Activated platelets release two types of membrane vesicles: microvesicles by surface shedding and exosomes derived from exocytosis of multivesicular bodies and alpha-granules. Blood 94(11):3791–3799
Hergenreider E, Heydt S, Treguer K, Boettger T, Horrevoets AJ, Zeiher AM, Scheffer MP, Frangakis AS, Yin X, Mayr M, Braun T, Urbich C, Boon RA, Dimmeler S (2012) Atheroprotective communication between endothelial cells and smooth muscle cells through miRNAs. Nat Cell Biol 14(3):249–256. doi:10.1038/ncb2441
Hood JL, San RS, Wickline SA (2011) Exosomes released by melanoma cells prepare sentinel lymph nodes for tumor metastasis. Cancer Res 71(11):3792–3801. doi:10.1158/0008-5472.CAN-10-4455
Jansen F, Yang X, Proebsting S, Hoelscher M, Przybilla D, Baumann K, Schmitz T, Dolf A, Endl E, Franklin BS, Sinning JM, Vasa-Nicotera M, Nickenig G, Werner N (2014) MicroRNA expression in circulating microvesicles predicts cardiovascular events in patients with coronary artery disease. J Am Heart Assoc 3(6), e001249. doi:10.1161/JAHA.114.001249
Johnstone RM, Adam M, Hammond JR, Orr L, Turbide C (1987) Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem 262(19):9412–9420
Kapustin AN, Chatrou ML, Drozdov I, Zheng Y, Davidson SM, Soong DY, Furmanik M, Sanchis P, de Rosales RT, Alvarez-Hernandez D, Shroff R, Yin X, Muller KH, Skepper JN, Mayr M, Reutelingsperger C, Chester A, Bertazzo S, Schurgers LJ, Shanahan CM (2015) Vascular smooth muscle cell calcification is mediated by regulated exosome secretion. Circ Res. doi:10.1161/CIRCRESAHA.116.305012
Kowal J, Tkach M, Thery C (2014) Biogenesis and secretion of exosomes. Curr Opin Cell Biol 29:116–125. doi:10.1016/j.ceb.2014.05.004
Lai RC, Arslan F, Lee MM, Sze NS, Choo A, Chen TS, Salto-Tellez M, Timmers L, Lee CN, El Oakley RM, Pasterkamp G, de Kleijn DP, Lim SK (2010a) Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res 4(3):214–222. doi:10.1016/j.scr.2009.12.003
Lai RC, Arslan F, Tan SS, Tan B, Choo A, Lee MM, Chen TS, Teh BJ, Eng JK, Sidik H, Tanavde V, Hwang WS, Lee CN, El Oakley RM, Pasterkamp G, de Kleijn DP, Tan KH, Lim SK (2010b) Derivation and characterization of human fetal MSCs: an alternative cell source for large-scale production of cardioprotective microparticles. J Mol Cell Cardiol 48(6):1215–1224. doi:10.1016/j.yjmcc.2009.12.021
Lonborg J, Vejlstrup N, Kelbaek H, Holmvang L, Jorgensen E, Helqvist S, Saunamaki K, Ahtarovski KA, Botker HE, Kim WY, Clemmensen P, Engstrom T (2013) Final infarct size measured by cardiovascular magnetic resonance in patients with ST elevation myocardial infarction predicts long-term clinical outcome: an observational study. Eur Heart J Cardiovasc Imaging 14(4):387–395. doi:10.1093/ehjci/jes271
Lotvall J, Hill AF, Hochberg F, Buzas EI, Di Vizio D, Gardiner C, Gho YS, Kurochkin IV, Mathivanan S, Quesenberry P, Sahoo S, Tahara H, Wauben MH, Witwer KW, Thery C (2014) Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles 3:26913. doi:10.3402/jev.v3.26913
Mackie AR, Klyachko E, Thorne T, Schultz KM, Millay M, Ito A, Kamide CE, Liu T, Gupta R, Sahoo S, Misener S, Kishore R, Losordo DW (2012) Sonic hedgehog-modified human CD34+ cells preserve cardiac function after acute myocardial infarction. Circ Res 111(3):312–321. doi:10.1161/CIRCRESAHA.112.266015
Makridakis M, Roubelakis MG, Vlahou A (2013) Stem cells: insights into the secretome. Biochim Biophys Acta 1834(11):2380–2384. doi:10.1016/j.bbapap.2013.01.032
Mathivanan S, Ji H, Simpson RJ (2010) Exosomes: extracellular organelles important in intercellular communication. J Proteomics 73(10):1907–1920. doi:10.1016/j.jprot.2010.06.006
Mathur S, Walley KR, Wang Y, Indrambarya T, Boyd JH (2011) Extracellular heat shock protein 70 induces cardiomyocyte inflammation and contractile dysfunction via TLR2. Circ J 75(10):2445–2452
Messina E, De Angelis L, Frati G, Morrone S, Chimenti S, Fiordaliso F, Salio M, Battaglia M, Latronico MV, Coletta M, Vivarelli E, Frati L, Cossu G, Giacomello A (2004) Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ Res 95(9):911–921. doi:10.1161/01.RES.0000147315.71699.51
Nolte-’t Hoen EN, van der Vlist EJ, Aalberts M, Mertens HC, Bosch BJ, Bartelink W, Mastrobattista E, van Gaal EV, Stoorvogel W, Arkesteijn GJ, Wauben MH (2012) Quantitative and qualitative flow cytometric analysis of nanosized cell-derived membrane vesicles. Nanomedicine 8(5):712–720. doi:10.1016/j.nano.2011.09.006
Ong SG, Lee WH, Huang M, Dey D, Kodo K, Sanchez-Freire V, Gold JD, Wu JC (2014) Cross talk of combined gene and cell therapy in ischemic heart disease: role of exosomal microRNA transfer. Circulation 130(11 Suppl 1):S60–S69. doi:10.1161/CIRCULATIONAHA.113.007917
Peche H, Heslan M, Usal C, Amigorena S, Cuturi MC (2003) Presentation of donor major histocompatibility complex antigens by bone marrow dendritic cell-derived exosomes modulates allograft rejection. Transplantation 76(10):1503–1510. doi:10.1097/01.TP.0000092494.75313.38
Piper HM, Garcia-Dorado D, Ovize M (1998) A fresh look at reperfusion injury. Cardiovasc Res 38(2):291–300
Raposo G, Stoorvogel W (2013) Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 200(4):373–383. doi:10.1083/jcb.201211138
Sahoo S, Klychko E, Thorne T, Misener S, Schultz KM, Millay M, Ito A, Liu T, Kamide C, Agrawal H, Perlman H, Qin G, Kishore R, Losordo DW (2011) Exosomes from human CD34(+) stem cells mediate their proangiogenic paracrine activity. Circ Res 109(7):724–728. doi:10.1161/CIRCRESAHA.111.253286
Scholz T, Temmler U, Krause S, Heptinstall S, Losche W (2002) Transfer of tissue factor from platelets to monocytes: role of platelet-derived microvesicles and CD62P. Thromb Haemost 88(6):1033–1038. doi:10.1267/th02121033
Sheldon H, Heikamp E, Turley H, Dragovic R, Thomas P, Oon CE, Leek R, Edelmann M, Kessler B, Sainson RC, Sargent I, Li JL, Harris AL (2010) New mechanism for Notch signaling to endothelium at a distance by Delta-like 4 incorporation into exosomes. Blood 116(13):2385–2394. doi:10.1182/blood-2009-08-239228
Shen B, Wu N, Yang JM, Gould SJ (2011) Protein targeting to exosomes/microvesicles by plasma membrane anchors. J Biol Chem 286(16):14383–14395. doi:10.1074/jbc.M110.208660
Skalnikova H, Motlik J, Gadher SJ, Kovarova H (2011) Mapping of the secretome of primary isolates of mammalian cells, stem cells and derived cell lines. Proteomics 11(4):691–708. doi:10.1002/pmic.201000402
Skog J, Wurdinger T, van Rijn S, Meijer DH, Gainche L, Sena-Esteves M, Curry WT Jr, Carter BS, Krichevsky AM, Breakefield XO (2008) Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol 10(12):1470–1476. doi:10.1038/ncb1800
Smalley DM, Root KE, Cho H, Ross MM, Ley K (2007) Proteomic discovery of 21 proteins expressed in human plasma-derived but not platelet-derived microparticles. Thromb Haemost 97(1):67–80
Smith RR, Barile L, Cho HC, Leppo MK, Hare JM, Messina E, Giacomello A, Abraham MR, Marban E (2007) Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens. Circulation 115(7):896–908. doi:10.1161/CIRCULATIONAHA.106.655209
Sreekumar PG, Kannan R, Kitamura M, Spee C, Barron E, Ryan SJ, Hinton DR (2010) AlphaB crystallin is apically secreted within exosomes by polarized human retinal pigment epithelium and provides neuroprotection to adjacent cells. PLoS One 5(10), e12578. doi:10.1371/journal.pone.0012578
Thery C, Amigorena S, Raposo G, Clayton A (2006) Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol Chapter 3:Unit 3.22. doi:10.1002/0471143030.cb0322s30
Timmers L, Lim SK, Arslan F, Armstrong JS, Hoefer IE, Doevendans PA, Piek JJ, El Oakley RM, Choo A, Lee CN, Pasterkamp G, de Kleijn DP (2007) Reduction of myocardial infarct size by human mesenchymal stem cell conditioned medium. Stem Cell Res 1(2):129–137. doi:10.1016/j.scr.2008.02.002
Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9(6):654–659. doi:10.1038/ncb1596
Van Deun J, Mestdagh P, Sormunen R, Cocquyt V, Vermaelen K, Vandesompele J, Bracke M, De Wever O, Hendrix A (2014) The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling. J Extracell Vesicles 3. doi:10.3402/jev.v3.24858
Vicencio JM, Yellon DM, Sivaraman V, Das D, Boi-Doku C, Arjun S, Zheng Y, Riquelme JA, Kearney J, Sharma V, Multhoff G, Hall AR, Davidson SM (2015) Plasma exosomes protect the myocardium from ischemia-reperfusion injury. J Am Coll Cardiol 65(15):1525–1536
Villarroya-Beltri C, Gutierrez-Vazquez C, Sanchez-Cabo F, Perez-Hernandez D, Vazquez J, Martin-Cofreces N, Martinez-Herrera DJ, Pascual-Montano A, Mittelbrunn M, Sanchez-Madrid F (2013) Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat Commun 4:2980. doi:10.1038/ncomms3980
Waldenstrom A, Genneback N, Hellman U, Ronquist G (2012) Cardiomyocyte microvesicles contain DNA/RNA and convey biological messages to target cells. PLoS One 7(4), e34653. doi:10.1371/journal.pone.0034653
Wang X, Huang W, Liu G, Cai W, Millard RW, Wang Y, Chang J, Peng T, Fan GC (2014) Cardiomyocytes mediate anti-angiogenesis in type 2 diabetic rats through the exosomal transfer of miR-320 into endothelial cells. J Mol Cell Cardiol 74:139–150. doi:10.1016/j.yjmcc.2014.05.001
Welton JL, Webber JP, Botos LA, Jones M, Clayton A (2015) Ready-made chromatography columns for extracellular vesicle isolation from plasma. J Extracell Vesicles 4:27269. doi:10.3402/jev.v4.27269
Wolf P (1967) The nature and significance of platelet products in human plasma. Br J Haematol 13(3):269–288
Wolfers J, Lozier A, Raposo G, Regnault A, Thery C, Masurier C, Flament C, Pouzieux S, Faure F, Tursz T, Angevin E, Amigorena S, Zitvogel L (2001) Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat Med 7(3):297–303. doi:10.1038/85438
Xin H, Li Y, Cui Y, Yang JJ, Zhang ZG, Chopp M (2013) Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab 33(11):1711–1715. doi:10.1038/jcbfm.2013.152
Yellon DM, Davidson SM (2014) Exosomes: nanoparticles involved in cardioprotection? Circ Res 114(2):325–332. doi:10.1161/CIRCRESAHA.113.300636
Yu B, Kim HW, Gong M, Wang J, Millard RW, Wang Y, Ashraf M, Xu M (2014) Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int J Cardiol 182C:349–360. doi:10.1016/j.ijcard.2014.12.043
Yuana Y, Bertina RM, Osanto S (2011) Pre-analytical and analytical issues in the analysis of blood microparticles. Thromb Haemost 105(3):396–408. doi:10.1160/TH10-09-0595
Yuana Y, Levels J, Grootemaat A, Sturk A, Nieuwland R (2014) Co-isolation of extracellular vesicles and high-density lipoproteins using density gradient ultracentrifugation. J Extracell Vesicles 3. doi:10.3402/jev.v3.23262
Zhang HG, Grizzle WE (2011) Exosomes and cancer: a newly described pathway of immune suppression. Clin Cancer Res 17(5):959–964. doi:10.1158/1078-0432.CCR-10-1489
Zhang Y, Zhang X, Shan P, Hunt CR, Pandita TK, Lee PJ (2013) A protective Hsp70-TLR4 pathway in lethal oxidant lung injury. J Immunol 191(3):1393–1403. doi:10.4049/jimmunol.1300052
Zhou Y, Xu H, Xu W, Wang B, Wu H, Tao Y, Zhang B, Wang M, Mao F, Yan Y, Gao S, Gu H, Zhu W, Qian H (2013) Exosomes released by human umbilical cord mesenchymal stem cells protect against cisplatin-induced renal oxidative stress and apoptosis in vivo and in vitro. Stem Cell Res Ther 4(2):34. doi:10.1186/scrt194
Acknowledgements
This work was funded by a grant from the Medical Research Council [MR/K002066/1] and the British Heart Foundation [RG/08/015/26411]. I am grateful for the ongoing support of Prof Derek Yellon, and co-workers at the Hatter Cardiovascular Institute.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Davidson, S. (2016). Microvesicles and Exosomes in Local and Distant Communication with the Heart. In: Madonna, R. (eds) Stem Cells and Cardiac Regeneration. Stem Cell Biology and Regenerative Medicine. Springer, Cham. https://doi.org/10.1007/978-3-319-25427-2_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-25427-2_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-25425-8
Online ISBN: 978-3-319-25427-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)