Abstract
Systemic lupus erythematosus (SLE) is an autoimmune rheumatic disease characterized by a heterogeneous group of clinical aspects and laboratory abnormalities. Disease presentation appears highly variable. The quantification of disease activity is still changeably applied in clinical trials and clinical practice since no gold standard clinimetric measurement of lupus activity has been identified to date. In this chapter, we review the various instruments that are used to assess disease activity, as well as SLE prognosis, including survival, causes of death, and prognostic predictors.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Systemic lupus erythematosus (SLE) is an autoimmune rheumatic disease characterized by a heterogeneous group of clinical features and laboratory abnormalities. Disease presentation appears highly changeable [1], and quantification of disease activity is still variably applied in clinical trials and clinical practice since no gold standard clinimetric measurement of lupus activity has been identified to date.
Prognosis of SLE has dramatically improved since the 1950s; however, SLE patients still display a 4.6-fold higher standardized mortality rate and a poorer quality of life compared to their counterparts in the general population [2]. Persistent disease activity and drug side effects are associated with damage accrual, which in turn is predictive of more damage and poor prognosis.
In this chapter, we review the various instruments that are used to assess disease activity, as well as SLE prognosis, including survival, causes of death, and prognostic predictors.
2 Disease Activity Indices
Lupus activity can be defined as the sum of all clinical manifestations and serological abnormalities related to ongoing immune inflammatory pathways involved in SLE. From the clinical point of view, lupus activity encompasses inflammatory and noninflammatory manifestations and persistent serologic abnormalities including the presence of autoantibodies, especially anti-double-stranded (ds) DNA antibody and low C3 and/or C4.
The physician’s opinion is often considered the “gold standard” for the evaluation of disease activity; however, it is not reliable due to important limitations including the physician’s personal experience and the weight which has to be assigned to the involvement of various organs [3].
Since the 1980s, nearly 60 different indices for assessing SLE activity have been elaborated [3]. Among them, two types of disease activity measurements can be identified: global activity scores, which provide an overall measurement of disease activity, and specific organ/system activity scores which assess disease activity in a single organ or system. Table 15.1 reports the most commonly used SLE disease activity indices.
Global score indices are very useful for comparing cohorts of SLE patients, as well as subjects with different disease manifestations. In addition, they are easy to assess and can be used retrospectively. However, the final score of a global activity tool might be the result of low activity in several organs or high activity in one single organ. In addition, they may underscore the severity of the disease [3].
On the other hand, specific organ/system scales are able to identify the extreme variability of SLE manifestations and allow for the assessment of disease activity and clinical response in specific organs. Therefore, they may be used as a cutoff for defining entry criteria into clinical trials. However, they are not very sensitive, and training is required to improve the performance of assessors.
3 Global Score Activity Indices
The Physician Global Assessment (PGA) is an important and useful outcome evaluation of lupus disease activity that is based on the physicians’ overall judgment. It provides a score based on clinical evaluation ranging between 0 and 3, where 0 means absence of disease activity and 3 is the highest disease activity which the examiner can expect considering all patients with lupus, in other words the most active patient in the “universe of lupus” and not in that particular patient.
The systemic lupus erythematosus disease activity index (SLEDAI) was developed at the University of Toronto in 1992 and it is composed of 24 items [4]. The score of severe clinical manifestations of SLE, such as vasculitis and renal and neurological involvement, is higher compared to other less severe disease manifestations, e.g., joint and skin manifestations. Serology, including anti-dsDNA, C3, and C4, is also taken into consideration. Only descriptors that are present in the 10 days prior to evaluation should be scored. The total SLEDAI score can vary from 0 to 105.
SLEDAI provides an overall evaluation of disease activity and is especially useful for comparing cohorts of SLE patients. Notably, it allows comparisons of patients with different disease manifestations. It is easy to assess, straightforward, and can also be retrospectively evaluated. Nonetheless, the SLEDAI score has some drawbacks: first, the final score might be due to low activity in different organs or high activity in one single organ; second, the same score may be due to the involvement of different organs; third, it may underscore the severity of the disease; and, lastly, the decrease in score due to the improvement in a specific organ system may be masked by the worsening in another organ system.
Since the first publication of the SLEDAI index, several modifications have been made including the Safety of Estrogens in Lupus Erythematosus National Assessment (SELENA) – SLEDAI and the SLEDAI-2000 (SLEDAI-2K). There are also simplified English and Spanish versions of the SLEDAI without immunological tests, which make the index easier to assess: the MEX-SLEDAI [3].
SELENA-SLEDAI [5] emphasizes new or recurrent disease activity in some descriptors (seizure, cranial nerve disorders, cerebrovascular accidents, skin rash, alopecia, mucosal ulcers, proteinuria) in order to identify disease flares rather than persistent disease activity.
In 2002, Gladman et al. published an updated version of the original SLEDAI, called SLEDAI-2K [6]. The modification was made in order to identify persistent active disease manifestations and not only new onset or flares. In the SLEDAI-2K, ongoing rashes, proteinuria, alopecia, and mucosal lesions have to be scored (Table 15.2). The SLEDAI-2K is widely used in clinical practice.
On the basis of the SLEDAI score, we can subdivide disease activity into five levels: 0 = inactive disease, from 1 to 5 = mild SLE activity, from 6 to 10 = moderately active lupus, from 11 to 19 = active SLE, and ≥20 = very active lupus.
In order to better standardize the assessment of SLE activity during pregnancy, SLEDAI was modified into the SLE-Pregnancy Disease Activity Index (SLEPDAI) [7]. This includes the 24 SLEDAI descriptors, 15 of which were modified in order to differentiate signs or symptoms caused by lupus activity from those related to pregnancy. Notably, preeclampsia-eclampsia is considered an exclusion criterion for some CNS descriptors including seizure, headache and cerebral infarctions, and HELLP (hemolysis, elevated liver enzymes, and low platelets) syndrome for thrombocytopenia. SLEPDAI has not been formally validated; nevertheless, it has been used in some studies.
The European Consensus Lupus Activity Measurement (ECLAM) was developed in 1992 through the analysis of symptoms and laboratory abnormalities reported in a cohort of 704 European patients affected with SLE. It comprises 15 weighted clinical and serological descriptors. The laboratory variables are hemoglobin, white blood cells, lymphocytes, platelets, proteinuria, urinary sediment, and complement levels, which are used in routine clinical practice. ECLAM has a high inter-rater concordance and can be retrospectively calculated [8].
The Systemic Lupus Erythematosus Activity Measure (SLAM), modified later into SLAM-R [8], is composed of 31 clinical manifestations or laboratory abnormalities that are usually found in SLE. Similarly to other global scores, a numerical value is attributed to each item based on different degrees of severity, with a total sum indicative of overall disease activity.
4 Organ-Specific Activity Indices
The British Isles Lupus Assessment Group (BILAG) index was first proposed in 1988 and later revised in 2004. It includes 97 items, most of which can be assessed by physical exams; in fact, only serum creatinine, urine dipstick, and total blood cell count are scored, while no serologic tests are included [7]. Each item is evaluated qualitatively by clinical assessment or quantitatively by considering laboratory values as well. BILAG includes nine organs/systems, i.e., general, mucocutaneous, neurological, musculoskeletal, cardiovascular, respiratory, renal, hematological, and gastrointestinal.
Only features that are related to active lupus and that were present in the 4 weeks prior to evaluation should be taken into consideration. Actually, BILAG was developed on the basis of the intention-to-treat principle. Hence, each of the nine systems is scored by an alphabetic character: BILAG A stands for “Action,” BILAG B for “Beware,” BILAG C for “Containment,” BILAG D for “Discount,” and BILAG E for “no Evidence” [8]. Although it has recently been proposed, a total score obtained by attributing a numeric value to the alphabetic character has rarely been used.
It should be noted that BILAG is the only transitional index where each item has to be recorded as new, same, worse, or improving rather than simply present or absent. These features are relevant when assessing the effect of a therapeutic intervention in a clinical trial.
BILAG is able to identify the extreme variability of SLE by assessing disease activity and clinical response in specific organs. It is reliable, sensitive to change, and widely used as a cutoff for entry criteria in clinical trials. Although it would appear to be the most complete SLE activity index, it is time consuming and quite complex to perform; thus, training is needed to improve the performance of the assessors. A software program (British Lupus Integrated Prospective System (BLIPS)) was created to help physicians calculate BILAG.
The Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI) was developed in 2005 to evaluate the activity and damage of lupus skin manifestations. It consists of two scores: the first summarizes the activity of skin manifestations, while the second measures disease-related skin damage. Activity is scored taking into account the following lesions: erythema, scales/hyperkeratosis, mucous membrane involvement, acute hair loss, and non-scarring alopecia. Damage is scored in terms of dyspigmentation and scarring, including scarring alopecia. The scores are calculated by a simple addition based on the extent of the lesions. The degree of involvement of each skin lesion is calculated according to the involvement of specific anatomic areas (i.e., malar area, neck, arms, etc.), scored on the basis of the worst skin lesion present within that area [9].
The Disease Activity Score (DAS)-28 is a validated and widely used clinimetric measurement in clinical trials and in routine clinical practice for patients with rheumatoid arthritis (RA). This is a standardized disease activity index that is highly correlated with the physician’s and patient’s overall assessment. By using DAS28, we can both classify patients with RA as having low, moderate, or high disease activity and define disease remission. Although it was only validated for patients with RA, DAS28 has been used for monitoring patients affected with other rheumatic conditions including SLE [10]. It assesses 28 joints (for swelling and tenderness), erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP) levels, and the patient’s overall health.
5 Evaluation of Disease Activity Over Time
Variations in disease activity over time are commonly recognized in SLE patients regardless of therapy. Unfortunately, disease activity indices measure disease activity at a single time point; thus by looking at only one measurement, we cannot have a comprehensive view of the variation of disease activity over time.
The Adjusted Mean SLEDAI-2K (AMS) was recently proposed to better summarize SLE activity over multiple visits. AMS is equivalent to the area under the curve of the SLEDAI-2K over time divided by time interval [11]. AMS is determined as follows: “(a) calculate the area under the curve between two visits: the length of time between two visits multiplied by the average of the two SLEDAI-2K values; (b) add up all the calculated areas; and (c) divide the result by the total length of the time period. AMS shares the same units as SLEDAI-2K and it is interpreted in the same way.”
By performing a longitudinal series of disease activity measurements, three different patterns of SLE activity were identified: relapsing remitting (RRD), clinical quiescent (CQD), and chronic active disease (CAD). CQD means no disease activity over time, CAD means persistent disease activity, and RRD indicates periods of disease activity interspersed with periods of inactive disease [12]. However, the frequency of disease activity patterns differs among studies as a consequence of the different definitions that are used. In our SLE cohort, we observed that every year 50 % of patients have CQD, while the remaining 50 % retained some kind of disease activity, being either CAD (in most cases) or RRD. After 7 years of follow-up, only one-third of patients could be considered in CQD, while 65 % displayed either relapsing or persistent disease activity [12].
6 Prognosis of Systemic Lupus Erythematosus
Survival of patients with SLE has improved over the past few decades [13]. Before 1955, the 5-year survival rate in SLE was less than 50 %; nowadays, the average 10-year survival rate exceeds 90 %, and the 15-year survival rate is approximately 80 % [14].
The main contributing factors toward improved survival were initially the availability of corticosteroids, dialysis, antibiotics, and antihypertensive agents. Afterward, the use of immunosuppressants and an improvement in disease classification leading to early diagnosis and treatment, and more recently the more appropriate use of conventional therapies as well as the tendency to follow patients with SLE in specialized clinics, the so-called lupus clinics, have all resulted in increased survival [4, 14–16].
Causes of death can be subdivided into those related to SLE itself and those related to disease complications. Causes related to SLE itself include active disease, i.e., active nephritis, CNS involvement, visceral vasculitis, and end-organ failure. Causes related to complications include infections, neoplasm, and atherosclerosis.
In 1976, Urowitz first reported a bimodal mortality pattern: deaths in the first few years of illness are typically caused by severe disease or infections due to immunosuppressive therapy, whereas late deaths are generally related to myocardial infarction or strokes, both attributed to accelerated atherosclerosis [17, 18]. Nowadays, deaths due to SLE have been further reduced, thanks to a more appropriate use of traditional treatments and to the introduction of new drugs such as mycophenolate mofetil, belimumab, and rituximab. However, intervening infections or cardiovascular events remain the major causes of deaths in the long term, followed by an increased incidence of malignancies [19, 20].
6.1 Prognostic Factors in SLE
It has been hypothesized that several specific factors may influence the prognosis of patients with SLE. They can be subdivided into two groups: unrelated or related to SLE.
6.2 Factors Unrelated to SLE
Prognostic factors unrelated to SLE include race, gender, age at SLE onset, socioeconomic status, and environment. In general, whites tend to have a better outcome than nonwhites, with mortality rates being higher in nonwhites. Low socioeconomic status and poor compliance seem to negatively influence the survival and morbidity of SLE patients with lupus. The effect of gender, age at disease onset, or environment on SLE prognosis remains elusive [16].
6.3 Factors Related to SLE
SLE-related factors include year of diagnosis, time from disease onset to diagnosis, disease activity and remission, SLE manifestations, disease treatment, and damage accrual.
6.3.1 Age at Diagnosis
Year of diagnosis is a critical aspect in the prognosis of SLE patients since the treatment has changed over the decades. For example, patients diagnosed with SLE in the 1950s, 1960s, and 1970s were treated more aggressively and for a longer period of time with corticosteroids and cyclophosphamide than patients diagnosed more recently.
6.3.2 Lag Time Between Symptoms and Diagnosis
In our cohort of 487 SLE patients recruited between 1970 and 2008, the mean lag time between onset and diagnosis was 59 months in patients diagnosed before 1980, a figure that is similar to what was reported by Wallace in 1981 [21], 28 months in those diagnosed between 1980 and 1989, 15 months in those diagnosed between 1990 and 1999, and 9 months after 2000. It is noteworthy that the difference was significant between the first and the second group, probably as a consequence of the introduction of antinuclear antibody (ANA) testing, and between the second and the third group, probably due to better knowledge of autoimmunity and autoimmune diseases. By contrast, the difference was not significant between the third and fourth group, supporting the concept that from 1990 until now nothing relevant has been introduced which could have improved the SLE diagnostic process. Disease progression in the time that elapses between disease onset and diagnosis is one of the major contributors to the improvement of survival and quality of life in SLE patients since early diagnosis and treatment increase SLE remission rate and improve patient prognosis [1, 14, 22].
6.3.3 Overall Disease Activity
Overall disease activity at first visit, at time of renal biopsy, or over time (AMS) proved to be an important predictor of mortality in a number of cohorts [23–26]. Moreover, Cook et al. [27] found an increase in the relative risk (RR) of mortality depending on the progressive increase in SLEDAI score: RR 1.28 for SLEDAI 1-5, RR 2.34 for SLEDAI 6-10, RR 4.74 for SLEDAI 11-19, and RR 14.11 for SLEDAI >20 (compared with SLEDAI = 0).
6.3.4 Disease Manifestations
In Cook’s cohort [27], some SLEDAI descriptors were found to be risk factors for death, including organic brain syndrome, retinal changes, cranial nerve disorders, proteinuria, pleurisy, fever, thrombocytopenia, and leukopenia. Moreover, Doria et al. [14] pointed out that the survival curves of patients affected with mild SLE (i.e., skin and musculoskeletal involvement) are similar to those of patients with severe lupus (i.e., renal involvement and CNS lupus) but only for up to 15 years after diagnosis; thereafter, the survival curves of patients with severe SLE rapidly worsen. By contrast, patients with mild disease have a survival rate similar to their counterparts in the general population [14].
6.3.5 Disease Remission
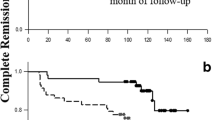
As shown by a large multicenter inception SLE cohort, disease remission and particularly early remission are predictive of better outcome in SLE patients [28]. As an example, renal survival is higher in patients who achieve either complete or partial disease remission as compared to severely affected patients, and overall survival at 20 years is significantly increased only in SLE patients who have complete disease remission [25, 29].
Indeed, remission within 1 year from disease onset is associated with a significant reduction in disease flares, organ damage, and overall cumulative dosage of corticosteroids [28], meaning a greater chance for lasting disease quiescence.
Unfortunately, remission in SLE is not clearly defined by any of the available disease activity scores. According to SLEDAI, remission may be defined as SLEDAI = 0, whereas SLEDAI ≥3 is considered persistent disease activity. Many authors have raised the question regarding what significance should be given to serological abnormalities such as anti-dsDNA antibodies or complement levels in patients without clinical manifestations of lupus, the so-called serologically active clinical quiescent disease. Based on clinical practice, three levels of remission in SLE can be identified: (a) complete remission may be defined as no clinical or serological signs of disease activity in patients who are treatment-free; (b) clinical remission off corticosteroids can be defined as the absence of signs and symptoms of urinary and hematological abnormalities in patients with or without serological abnormalities who are corticosteroid-free; and (c) clinical remission on corticosteroids can be defined as the absence of signs and symptoms of urinary and hematological abnormalities in patients with or without serological abnormalities who are taking low-dose corticosteroids (≤5 mg/day prednisone or equivalent) [30].
6.3.6 Disease Treatment
Improved therapeutic approaches for SLE have led to better disease control, thereby reducing mortality related to active disease. However, they can be responsible for drug-related adverse events which may affect long-term prognosis. The drugs which mostly seem to affect long-term prognosis are corticosteroids and immunosuppressants which are able to decrease disease activity, but their prolonged use has been associated with increased cholesterol levels and a high frequency of cardiovascular events and infections [16]. On the other hand, hydroxychloroquine seems to be associated with lower cholesterol levels, suggesting a protective role of this drug against coronary artery disease [17].
6.3.7 Damage Accrual
Damage is defined as irreversible tissue injury occurring after SLE diagnosis and lasting at least 6 months. Damage is evaluated by the SLICC (Systemic Lupus International Collaborating Clinics) damage index (SDI), which encompasses 12 organ systems [31]. While disease activity tends to decline over time in patients with SLE, damage tends to increase. Since lupus patients nowadays live longer than they did a few decades ago, they tend to accumulate more damage, which is secondary to both active disease and long-standing treatment, especially corticosteroid chronic exposure [32, 33]. Notably, SLE patients with active disease (CAD or RRD) accumulate more organ damage compared to patients in remission. It has been shown that long-standing active SLE may drive an 8 % increased risk of organ damage [30]. High AMS scores were also found to be associated with damage and cardiovascular disease in SLE [34].
Damage is one of the main determinants of poor long-term prognosis in SLE patients. Indeed, damage accrual can lead to more damage, which, in turn, leads to disability, productivity loss, and, in the most severe cases, death [30].
A prospective study showed that 25 % of lupus patients with a SLICC-DI >0 at baseline had died at 10 years vs. only 7.3 % of patients with no damage at study entry; notably, renal damage and cardiovascular disease were associated with a higher risk of death during the follow-up [35]. In another prospective follow-up study, an increase in SLICC-DI ≥2 up to the third year of disease was found to increase the RR of mortality to 7.7 [36]; renal failure and cardiovascular disease were again the major predictors of death.
7 Quality of Life and Loss of Productivity
Health-related quality of life (HRQL) in SLE patients is usually evaluated by the SF-36 (Medical Outcomes Study Short Form) which consists of 36 items among 8 domains measuring diverse components of psychophysical health, i.e., physical function, role limitations due to emotional or physical problems, social function, mental health, general health perception, vitality, and pain [37]. HRQL has been shown to be lower in SLE patients as compared to their counterparts in the general population. Organ damage was associated with an overall decrease in physical, social, and mental performance, and poor coping strategies [38, 39] and new organ damage were a predictor of a further decline in HRQL [38].
Everyday activities and the ability to work are also affected in SLE patients [22, 40, 41]. It has been shown that two-third of SLE patients have a decreased ability to perform routine activities, both at home and in the workplace, thus worsening their psychological and financial well-being [42]. One-third of patients with SLE had an occupational disability with a decrease in the number of working hours per week and 50 % of patients left their jobs within 15 years of SLE diagnosis [43]. A number of studies showed SLE-related damage to be associated with work disability and loss of productivity [44–46], which was particularly significant in case of neuropsychiatric SLE or deforming arthritis [46]. Longer disease duration, fatigue, comorbidities, and poor mental status also negatively influence the patients’ productivity [46].
8 Conclusions
Despite their limitations, disease activity and damage indices have helped physicians to better standardize the assessment and management of SLE, which has certainly contributed to the improvement in survival of SLE patients in the last decades. Unfortunately, disease mortality is still high and is mostly due to disease complications, especially in patients with active and severe disease and damage accrual. Moreover, the HRQL of SLE patients is still poorer than that of their healthy counterparts. On the other hand, some indicators of a favorable outcome have emerged in the last few years. Early diagnosis and treatment, close control of disease activity, follow-up of patients in specialized lupus clinics, and prolonged remission all seem to be associated with better outcome.
References
Doria A, Zen M, Canova M, Bettio S, Bassi N, Nalotto L, Rampudda M, Ghirardello A, Iaccarino L (2010) SLE diagnosis and treatment: when early is early. Autoimmun Rev 10:55–60
Lopez R, Davidson JE, Beeby MD et al (2012) Lupus disease activity and the risk of subsequent organ damage and mortality in a large lupus cohort. Rheumatology 51:491–498
Griffiths B, Mosca M, Gordon C (2005) Assessment of patients with systemic lupus erythematosus and the use of lupus disease activity indices. Best Pract Res Clin Rheumatol 19:685–708
Urowitz MB, Gladman DD (1998) Measures of disease activity and damage in SLE. Clin Rheumatol 12:405–413
Buyon JP, Petri MA, Kim MY et al (2005) The effect of combined estrogen and progesterone hormone replacement therapy on disease activity in systemic lupus erythematosus: a randomized trial. Ann Intern Med 142:953–962
Gladman DD, Ibañez D, Urowitz MB (2002) Systemic lupus erythematosus disease activity index 2000. J Rheumatol 29:288–291
Buyon GP, Kalunian CK, Ramsey-Goldman R et al (1999) Assessing disease activity in SLE patients during pregnancy. Lupus 8:677–684
Romero-Diaz J, Isenberg D, Ramsey-Goldman R (2011) Measures of adult systemic lupus erythematosus: updated version of British Isles Lupus Assessment Group (BILAG 2004), European Consensus Lupus Activity Measurements (ECLAM), Systemic Lupus Activity Measure, Revised (SLAM-R), Systemic Lupus Activity Questionnaire for Population Studies (SLAQ), Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2 K), and Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index (SDI). Arthritis Care Res (Hoboken) 63(Suppl 11):S37–S46
Albrecht JL, Taylor L, Berlin JA et al (2005) The CLASI (Cutaneous Lupus Erythematosus Disease Area and Severity Index): an outcome instrument for cutaneous lupus erythematosus. Invest Dermatol 125:889–894
Ceccarelli F, Perricone C, Massaro L et al (2014) The role of disease activity score 28 in the evaluation of articular involvement in systemic lupus erythematosus. ScientificWorldJournal 2014:236842
Ibañez D, Urowitz MB, Gladman DD (2003) Summarizing disease features over time: I. Adjusted mean SLEDAI derivation and application to an index of disease activity in lupus. J Rheumatol 30:1977–1982
Zen M, Bassi N, Nalotto L et al (2012) Disease activity patterns in a monocentric cohort of SLE patients: a seven-year follow-up study. Clin Exp Rheumatol 30:856–3
Ruiz-Irastorza G, Khamashta MA, Castellino G, Hughes GR (2001) Systemic lupus erythematosus. Lancet 357:1027–1032
Doria A, Iaccarino L, Ghirardello A et al (2006) Long-term prognosis and causes of death in systemic lupus erythematosus. Am J Med 119:700–706
Doria A, Canova M, Tonon M et al (2008) Infections as triggers and complications of systemic lupus erythematosus. Autoimmun Rev 8:24–28
Wallace DJ, Hahn BH (2007) Dubois’ lupus erythematosus, 7th edn. Lippincott Williams & Wilkins, Philadelphia, Chapter 69
Urowitz MB, Bookman AA, Koehler BE, Gordon DA, Smythe HA, Ogryzlo MA (1976) The bimodal mortality pattern of systemic lupus erythematosus. Am J Med 60:221–225
Doria A, Shoenfeld Y, Wu R et al (2003) Risk factors for subclinical atherosclerosis in a prospective cohort of patients with systemic lupus erythematosus. Ann Rheum Dis 62:1071–1077
Iaccarino L, Bettio S, Zen M, Nalotto L, Gatto M, Ramonda R et al (2013) Premature coronary heart disease in SLE: can we prevent progression? Lupus 22:1232–1242
Fei Y, Shi X, Gan F, Li X, Zhang W, Li M et al (2014) Death causes and pathogens analysis of systemic lupus erythematosus during the past 26 years. Clin Rheumatol 33:57–63
Pistiner M, Wallace DJ, Nessim S, Metzger AL, Klinenberg JR (1991) Lupus erythematosus in the 1980s: a survey of 570 patients. Semin Arthritis Rheum 21:55–64
Doria A, Rinaldi S, Ermani M, Salaffi F, Iaccarino L, Ghirardello A et al (2005) Health related quality of life in Italian patients with systemic lupus erythematosus. II. Role of clinical, immunological and psychological determinants. Rheumatology 43:1580–1586
Abu-Shakra M, Urowitz MB, Gladman DD et al (1995) Mortality studies in systemic lupus erythematosus. Results from a single center. II Predictors variables for mortality. J Rheumatol 2:1265–1270
Golet JR, Mackenzie T, Levinton C et al (1993) The long term prognosis of lupus nephritis; the impact of disease activity. J Rheumatol 20:59–65
Moroni G, Quaglini S, Gallelli B et al (2007) The long-term outcome of 93 patients with proliferative lupus nephritis. Nephrol Dial Transplant 22:2531–2539
Ibanez D, Urowitz MB, Gladman DD et al (2003) Summarizing disease features over time: I. adjusted mean SLEDAI derivation and application to an index of disease activity in lupus. J Rheumatol 30:1977–1982
CooK RJ, Gladman DD, Pericak D et al (2000) Prediction of short term mortality in SLE with time dependent measures of disease activity. J Rheumatol 27:1892–1895
Nossent J, Kiss E, Rozman B et al (2010) Disease activity and damage accrual during the early disease course in a multinational inception cohort of patients with systemic lupus erythematosus. Lupus 19:949–956
Chen YE, Korbet SM, Katz RS et al (2008) Value of a complete or partial remission in severe lupus nephritis. Clin J Am Soc Nephrol 3:46–53
Doria A, Gatto M, Zen M, Iaccarino L, Punzi L (2014) Optimizing outcome in SLE: treating-to-target and definition of treatment goals. Autoimmun Rev 13:770–777
Gladman DD, Urowitz MB, Goldsmith CH, Fortin P, Ginzler E, Gordon C et al (1997) The reliability of the Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index in patients with systemic lupus erythematosus. Arthritis Rheum 40:809–813
Thamer M, Hernán MA, Zhang Y et al (2009) Prednisone, lupus activity, and permanent organ damage. J Rheumatol 36:560–564
Stoll T, Sutcliffe N, Mach J et al (2004) Analysis of the relationship between disease activity and damage in patients with systemic lupus erythematosus – a 5-yr prospective study. Rheumatology 43:1039–4
Ibañez D, Gladman DD, Urowitz MB (2005) Adjusted mean Systemic Lupus Erythematosus Disease Activity Index-2K is a predictor of outcome in SLE. J Rheumatol 32:824–827
Rahman P, Gladman DD, Urowitz MB et al (2001) Early damage as measured by the SLICC/ACR damage index is a predictor of mortality in systemic lupus erythematosus. Lupus 10:93–96
Manger K, Manger B, Repp R et al (2002) Definition of risk factors for death, end stage renal disease, and thromboembolic events in a monocentric cohort of 338 patients with systemic lupus erythematosus. Ann Rheum Dis 61:1065–1070
Ware JE Jr, Sherbourne CD (1992) The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 30:473–483
Mok CC, Ho LY, Cheung MY, Yu KL, To CH (2009) Effect of disease activity and damage on quality of life in patients with systemic lupus erythematosus: a 2-year prospective study. Scand J Rheumatol 38:121–127
Rinaldi S, Ghisi M, Iaccarino L et al (2006) Influence of coping skills on health-related quality of life in patients with systemic lupus erythematosus. Arthritis Rheum 55:427–433
Rinaldi S, Doria A, Salaffi F, Ermani M, Iaccarino L, Ghirardello A et al (2004) Health related quality of life in Italian patients with systemic lupus erythematosus. I. Relationship between physical and metal dimension and impact of age. Rheumatology 43:1574–1579
Khanna S, Pal H, Pandey RM, Handa R (2004) The relationship between disease activity and quality of life in systemic lupus erythematosus. Rheumatology 43:1536–1540
Bertsias G, Ioannidis JP, Boletis J et al (2008) EULAR recommendations for the management of systemic lupus erythematosus. Report of a Task Force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics. Ann Rheum Dis 67:195–205
Yelin E, Trupin L, Katz P, Criswell L, Yazdany J, Gillis J et al (2007) Work dynamics among persons with systemic lupus erythematosus. Arthritis Rheum 57:56–63
Bertoli AM, Fernández M, Alarcón GS, Vilá LM, Reveille JD (2007) Systemic lupus erythematosus in a multiethnic US cohort LUMINA (XLI): factors predictive of self reported work disability. Ann Rheum Dis 66:12–17
Mok CC, Cheung MY, Ho LY, Yu KL, To CH (2008) Risk and predictors of work disability in Chinese patients with systemic lupus erythematosus. Lupus 17:1103–1107
Al Dhanhani AM, Gignac MA, Su J, Fortin PR (2009) Work disability in systemic lupus erythematosus. Arthritis Rheum 61:378–385
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Iaccarino, L., Larosa, M., Doria, A. (2016). Disease Activity Indices and Prognosis of Systemic Lupus Erythematosus. In: Roccatello, D., Emmi, L. (eds) Connective Tissue Disease. Rare Diseases of the Immune System. Springer, Cham. https://doi.org/10.1007/978-3-319-24535-5_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-24535-5_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-24533-1
Online ISBN: 978-3-319-24535-5
eBook Packages: MedicineMedicine (R0)