Abstract
Polypharmacy occurs commonly among older patients with human immunodeficiency virus (HIV).
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Human Immunodeficiency Virus
- Integrase Inhibitor
- Human Immunodeficiency Virus Viral Load
- Excessive Polypharmacy
- Human Immunodeficiency Virus Positive Individual
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
FormalPara Key Points-
Polypharmacy occurs commonly among older patients with human immunodeficiency virus (HIV).
-
It is an independent and under-appreciated contributor to aging and drug-related complications.
-
It requires regular and careful medication review.
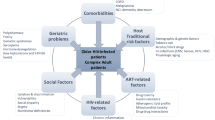
Polypharmacy, commonly defined in the general population as taking five or more different medications on a daily basis [1], is an important but still poorly understood clinical problem in patients with HIV, particularly among those older than 50 years of age [2, 3]. This is not a new phenomenon in HIV disease, as patients have always needed to take multiple medications in order to remain stable. In the mid-1990s the first generation of highly active antiretroviral therapy (HAART) regimens included three different antiretrovirals (ARVs), each consisting of several tablets. All of these pills had to be taken three to four times daily, often according to strict dietary restrictions. These ARVs were taken in addition to the drugs patients often took either for primary or secondary prophylaxis of various opportunistic infections, as well as for symptomatic management of drug-related toxicities, or for the acute management of intercurrent complications.
It was demonstrated early in the HIV epidemic that polypharmacy increased the risk of poor adherence to HAART [4]. With the early HAART regimens, particularly the protease inhibitor (PI) based treatments, taking more than 95 % of the pills as prescribed was required in order to achieve undetectable HIV RNA levels. Suboptimal adherence to HAART predicts the development of drug resistance [5] as well as clinical progression and mortality [6]. In early clinical studies of antiretroviral therapy (ART) naïve patients, who were monitored and followed carefully, more than 80 % remained on their first HAART regimens at one year follow-up. In the ‘real-world’ experience of busy clinic-based care, often significantly less than two-thirds of patients were still taking their drugs as prescribed at 1 year of follow-up [7].
HAART has been greatly simplified over the past 10–15 years, and patients now are often able to take daily regimens. As well, several single-tablet regimens (STR) are available, each containing three distinct ARVs [8]. Currently available ARVs are better tolerated, have significantly less toxicity, and require fewer dietary restrictions, although some remain [9]. These drug regimens are also more forgiving in terms of strict adherence and 80–95 % adherence rates are increasingly associated with undetectable HIV viral loads. These changes in drug design and formulation allow for improved compliance and result in greater durability of response. Nevertheless, patients with HIV still have a greater prevalence of polypharmacy than age-matched controls [10, 11]. Furthermore, the prevalence of what has been termed excessive polypharmacy, the use of 10 or more medications, appears to be a particularly worrisome problem among older patients with HIV [10, 12].
The need to take multiple drugs in currently treated patients with HIV occurs for several reasons, including the increased prevalence of non-AIDS defining comorbidities, which often require medical therapy either for prevention or active therapy [13]. Treatments typically include drugs for cardiovascular disease, hypertension, gastrointestinal disorders, psychoactive disorders, non-HIV-related infections, non-AIDS-defining cancers, dyslipidemia, and type II diabetes. An important limitation in understanding the scope of the problem is that the manner in which the use of multiple medications is reported is inconsistent and therefore potentially confusing. In a study by Edelman et al. [3] the median number of drugs taken by all patients with HIV was between five and nine. Another study reported that between 15 and 75 % of treated patients with HIV in their 60s meet criteria for polypharmacy and that 14 % of patients older than 65 years of age were taking four or more non-ARV drugs, most of which were vitamins, supplements, or drugs for cardiovascular or neurologic disorders [14]. A further study focusing on older patients with HIV found that in a cohort, with a median age of 64 years, the median number of drugs patients were taking was 13, of which only four were ARVs [10]. In the general population older patients taking this many drugs have an increased risk of falls, impaired cognition, frailty, hospitalization, and mortality [1].
Non-medically prescribed drug use is very common and often under-reported or misrepresented in the general population, and is a particular problem in patients with HIV. In addition to over-the-counter (OTC) drugs, patients often take recreational drugs and alternative care-related drugs. A large discrepancy exists between what the patient is actually taking and what their provider believes they are taking [15]. Care centers following large numbers of injection drug users will experience an increased frequency of the use of pain medications and methadone. The anticipated increasing availability of potentially curative short-term regimens for hepatitis C co-infection will further add to the increased use of drugs [16]. All of these factors combine to increase the pill burden, overall cost, and the complications related to polypharmacy.
The consequences of polypharmacy in the general elderly population are well-known, and typically include poor adherence to necessary drugs, increased incidence of adverse drug events (ADEs), drug–drug interactions (DDI), and use of inappropriate medications. Fifteen percent of treated patients with HIV may take drugs with potential anticholinergic toxicity [10]. Patients with HIV may be particularly susceptible to the above noted complications because of increased rates of renal and hepatic dysfunction, ongoing systemic immune activation, and associated chronic inflammation as well as sub-clinical impaired organ function. It is therefore not surprising that several of these common age-related and polypharmacy-associated complications also occur in younger treated patients with HIV. These include frailty [17] and impaired cognition [18]. Medications have been shown to specifically contribute to increased risk of poor mobility and falls in HIV positive individuals [19, 20]. Although polypharmacy predisposes to frailty in older seronegative people [21], it is presently unknown whether polypharmacy independently contributes to the development of frailty and impaired cognition in patients with HIV. Although several studies have shown that overall adherence to medications is higher in older patients with HIV compared to younger patients, risk factors for poor adherence are also increased in older patients [22]. This is a particular problem in HIV patients with cognitive impairment, where it has been shown that adherence is decreased primarily in older patients who perform poorly on tests of executive function [22]. Although the incidence of HIV-associated dementia has significantly decreased with HAART, rates of mild cognitive impairment and asymptomatic cognitive decline are worryingly high and may be greater compared to appropriately selected control subjects [23]. Other risk factors for poor adherence and drug toxicity in older patients with HIV, as in the general population, include social isolation, substance abuse, and limited financial resources, which are increased in certain subgroups with HIV.
The large number of drugs taken by older treated patients with HIV increases the risk of important pharmacokinetic interactions with ARVs, leading to important DDIs. Several examples highlight these negative interactions. Cardiovascular disease is the most common non-AIDS complication in treated patients, often requiring drug therapy. Interactions requiring dose adjustment and careful monitoring are required for patients with HIV taking antiarrthymics, several classes of antihypertensives, and warfarin, depending on the specific HAART components used [2]. Many treated patients with HIV develop dyslipidemia, and meet current criteria for use of statins. There is a recognized risk of statin-induced myopathy and rhabdomyolysis in patients on certain PI drugs that are potent inhibitors of CYP3-A4 and can thereby increase statin levels normally metabolized via cytochrome P450. Simvastatin and lovastatin are thus contraindicated in patients taking PIs, and only low doses of pravastatin, rosuvastatin, and atorvastatin are recommended, although higher doses are often prescribed to HIV-negative patients because of their improved efficacy. Atorvastatin may be used safely in usual doses along with the integrase inhibitor raltegravir [24]. It is unknown what interactions occur with higher doses of this statin or whether any interactions occur with the newest integrase inhibitor, dolutegravir. Proton pump inhibitors (PPIs), some of which are available OTC, are also frequently used by patients with HIV and may lower serum levels of the important PI, atazanavir, potentially leading to therapeutic failure and drug resistance [16]. Both PPIs and H2 blockers decrease absorption of rilpivirine, a recently introduced non-nucleoside reverse-transcriptase inhibitor (NNRTI) that is co-formulated in one of the currently available STR drugs. The risk of developing adrenal insufficiency and Cushing’s syndrome with the concurrent use of inhaled or intranasal fluticasone and any ritonavir-boosted PIs is well described and must be avoided. Sleep disturbances and pain syndromes occur commonly in the elderly, and have also been described in patients with HIV [25, 26]. Patients are frequently treated with drugs associated with increased risk of DDI. Recently, the co-administration of opioids and benzodiazepines has been shown to increase mortality in treated patients with HIV [27].
One-third of treated patients with chronically suppressed HIV-viral load (HIV-VL) do not achieve immune recovery to more than 500 CD4+ cells. This occurs more frequently in older patients with HIV who remain at increased risk of both AIDS as well as non-AIDS-related complications compared to patients with more than 500 CD4+ cells. Some older patients with very low plateau CD4 counts may require primary prophylaxis of common opportunistic infections, such as Pneumocystis pneumonia and central nervous system toxoplasmosis. Low dose sulfamethoxazole is effective as the first-line drug for primary prophylaxis of these infections, which adds to the pill burden and risk of DDI, and thus requires careful monitoring (eg, in patients taking warfarin).
In summary, polypharmacy is very common in patients with HIV and is an essential component of comprehensive patient management. Polypharmacy independently contributes to morbidity and mortality and demands increased vigilance on the part of providers involved in the complex care of aging patients with HIV. It is highly recommended that all patients review their medications at every regular visit and have an annual medical reconciliation. This is best carried out by knowledgeable pharmacists, who are essential members of the HIV treating team.
References
Gnjidic D, Hilmer SN, Blyth FM, et al. Polypharmacy cutoff and outcomes: five or more medicines were used to identify community-dwelling older men at risk of different adverse outcomes. J Clin Epidemiol. 2012;65:989–95.
Gleason LJ, Luque AE, Shah K. Polypharmacy in the HIV-infected older adult population. Clin Interv Aging. 2013;8:749–63.
Edelman EJ, Gordon KS, Glover J, McNicholl IR, Fiellin DA, Justice AC. The next therapeutic challenge in HIV: polypharmacy. Drugs Aging. 2013;30:613–28.
Stone VE, Jordan J, Tolson J, Miller R, Pilon T. Perspectives on adherence and simplicity for HIV-infected patients on antiretroviral therapy: self-report of the relative importance of multiple attributes of highly active antiretroviral therapy (HAART) regimens in predicting adherence. J Acquir Immune Defic Syndr. 2004;36:808–16.
Mannheimer S, Friedland G, Matts J, Child C, Chesney M. The consistency of adherence to antiretroviral therapy predicts biologic outcomes for human immunodeficiency virus-infected persons in clinical trials. Clin Infect Dis. 2002;34:1115–21.
Wood E, Hogg RS, Yip B, Harrigan PR, O'Shaughnessy MV, Montaner JS. Effect of medication adherence on survival of HIV-infected adults who start highly active antiretroviral therapy when the CD4+ cell count is 0.200 to 0.350 x 10(9) cells/L. Ann Intern Med. 2003;139:810–6.
Bangsberg DR, Perry S, Charlebois ED, et al. Non-adherence to highly active antiretroviral therapy predicts progression to AIDS. AIDS. 2001;15:1181–3.
Geretti AM, Tsakiroglou M. HIV: new drugs, new guidelines. Curr Opin Infect Dis. 2014;27:545–53.
Gunthard HF, Aberg JA, Eron JJ, et al. Antiretroviral treatment of adult HIV infection: 2014 recommendations of the International Antiviral Society-USA Panel. JAMA. 2014;312:410–25.
Greene M, Steinman MA, McNicholl IR, Valcour V. Polypharmacy, drug-drug interactions, and potentially inappropriate medications in older adults with human immunodeficiency virus infection. J Am Geriatr Soc. 2014;62:447–53.
Gimeno-Gracia M, Crusells-Canales MJ, Javier Armesto-Gómez F, Rabanaque-Hernández MJ. Prevalence of concomitant medications in older HIV+ patients and comparison with general population. HIV Clin Trials. 2015;16:117–24.
Tseng A, Szadkowski L, Walmsley S, Salit I, Raboud J. Association of age with polypharmacy and risk of drug interactions with antiretroviral medications in HIV-positive patients. Ann Pharmacother. 2013;47:1429–39.
Guaraldi G, Orlando G, Zona S, et al. Premature age-related comorbidities among HIV-infected persons compared with the general population. Clin Infect Dis. 2011;53:1120–6.
Hasse B, Ledergerber B, Furrer H, et al. Morbidity and aging in HIV-infected persons: the Swiss HIV cohort study. Clin Infect Dis. 2011;53:1130–9.
Furler MD, Einarson TR, Walmsley S, Millson M, Bendayan R. Polypharmacy in HIV: impact of data source and gender on reported drug utilization. AIDS Patient Care STDS. 2004;18:568–86.
Foy M, Sperati CJ, Lucas GM, Estrella MM. Drug interactions and antiretroviral drug monitoring. Curr HIV/AIDS Rep. 2014;11:212–22.
Brothers TD, Kirkland S, Guaraldi G, et al. Frailty in people aging with human immunodeficiency virus (HIV) infection. J Infect Dis. 2014;210:1170–9.
Clifford DB, Ances BM. HIV-associated neurocognitive disorder. Lancet Infect Dis. 2013;13:976–86.
Erlandson KM, Allshouse AA, Jankowski CM, et al. Risk factors for falls in HIV-infected persons. J Acquir Immune Defic Syndr. 2012;61:484–9.
Richert L, Brault M, Mercié P, et al. Decline in locomotor functions over time in HIV-infected patients. AIDS. 2014;28:1441–9.
Herr M, Robine JM, Pinot J, Arvieu JJ, Ankri J. Polypharmacy and frailty: prevalence, relationship, and impact on mortality in a French sample of 2350 old people. Pharmacoepidemiol Drug Saf. 2015;24:637–46.
Ettenhofer ML, Hinkin CH, Castellon SA, et al. Aging, neurocognition, and medication adherence in HIV infection. Am J Geriatr Psychiatry. 2009;17:281–90.
Heaton RK, Franklin Jr DR, Deutsch R, et al. Neurocognitive change in the era of HIV combination antiretroviral therapy: the longitudinal CHARTER study. Clin Infect Dis. 2015;60:473–80.
Blonk M, van Beek M, Colbers A, Schouwenberg B, Burger D. Pharmacokinetic drug-drug interaction study between Raltegravir and Atorvastatin 20 mg in healthy volunteers. J Acquir Immune Defic Syndr. 2015;69:44–51.
Payne BA, Hateley CL, Ong EL, et al. HIV-associated fatigue in the era of highly active antiretroviral therapy: novel biological mechanisms? HIV Med. 2013;14:247–51.
Vosvick M, Gore-Felton C, Ashton E, et al. Sleep disturbances among HIV-positive adults: the role of pain, stress, and social support. J Psychosom Res. 2004;57:459–63.
Weisberg DF, Gordon KS, Barry DT, et al. Long-term prescription of opioids and/or benzodiazepines and mortality among HIV-infected and uninfected patients. J Acquir Immune Defic Syndr. 2015;69:223–33.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Falutz, J. (2016). HIV, Aging, and Polypharmacy. In: Guaraldi, G., Falutz, J., Mussi, C., Silva, A. (eds) Managing the Older Adult Patient with HIV. Adis, Cham. https://doi.org/10.1007/978-3-319-20131-3_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-20131-3_11
Published:
Publisher Name: Adis, Cham
Print ISBN: 978-3-319-20130-6
Online ISBN: 978-3-319-20131-3
eBook Packages: MedicineMedicine (R0)