Abstract
Color is a visible pollutant and its presence has always been undesirable in water used for either industrial or domestic needs.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Color is a visible pollutant and its presence has always been undesirable in water used for either industrial or domestic needs. The exact period of the commencement of the art of dyeing in the world could not be ascertained correctly. However, archaeological evidence shows that dyeing was a wide-spread industrial enterprise in Egypt, India and Mesopotamia round third millennium B.C. (Ouzman 1998). Ever since the beginning of human civilization, people have been using colorants for painting and dyeing of their surroundings, skins and clothes. Until the mid 19th century, all colorants applied were from natural origin. These dyes were from plant sources, like roots, berries, bark, leaves, wood, fungi, and lichens. They fade early, because they were from natural origin and degradable. William Henry Perkin discovered the first synthetic dye stuff “Mauve” (aniline, a basic dye) while searching for a cure for malaria and thus, a new industry has opening. It was a brilliant fuchsia type color, but faded easily. Synthetic dyes were named after the chemical structure of the chromophoric group (azo dyes, anthraquinone dyes , xanthene dyes, triphenylmethane dyes, etc.) (Zollinger 2003). Amongst complex industrial wastewater with various types of coloring agents, dye wastes are predominant (Anjaneyulu et al. 2005).
Color is contributed by phenolic compounds , such as tannins, lignins (2–3 %) and organic colorants (3–4 %) (Clarke and Steinle 1995). However, synthetic dyes including dye intermediates i.e. sulfur, mordant, reactive, cationic, disperse, azo, acid, and vat dyes contribute maximum to waste waters (Raghavacharya 1997). Dyes are difficult to be decolorized due to their complex structure, synthetic origin and recalcitrant nature. Therefore, it is obligatory to remove them from industrial effluents before being disposed into water bodies. The color of water polluted with organic colorants gets reduced with the cleavage of the –C=C–, –N=N– bonds as well as heterocyclic and aromatic rings. The absorption of light by the associated molecules shifts from the visible to the ultraviolet or infrared region of the electromagnetic spectrum (Strickland and Perkins 1995). There are about 12 classes of chromogenic groups, among which, the most common are the azo dyes, which make up to 60–70 % of all textile and tannery dyestuffs produced (Carliell et al. 1996), followed by the anthraquinone type. Discharge of colorless effluents loaded with toxic and hazardous pollutants to water bodies is not usually objected. On the other hand, the discharge of colored effluents, though less toxic, is often objected by the public on the assumption that color is an indicator of pollution. It is, therefore, not surprising to note that color in wastewater has now been considered as a pollutant that needs to be treated before discharge. Wastewater treatment using biological process for decolorization of industrial effluents is still ambiguous and divergent.
2 Microbial Treatment of Waste Water Containing Dyes
Rapid industrialization releases an array of environmental pollutants which include several xenobiotics (Khan et al. 2013). These compounds are highly toxic and most of them are carcinogenic in nature. Therefore, they have been listed as highly toxic pollutants by the United States Environment Protection Agency (http://www.epa.gov). The synthetic dyes constitute the largest class and are widely used in the textile, cosmetic, printing, drug, and food processing industries. These dyes are toxic, mutagenic and carcinogenic in nature and have negative impact on the living organisms (Dos Santos et al. 2007). In the dyeing process, most of dyes remain unbound and get released into the environment with wastewater. This is an alarming situation. A dye house effluent typically contains 0.6–0.8 g dye l−1 (Gahr et al. 1994). Microbial degradation of textile dyes has become a key research area in environmental sciences. The microbes adapt to the toxic wastes and develop new resistant mechanism, they can biodegrade various toxic chemicals into less harmful forms. Many bacterial, fungal and algal species have developed the ability to absorb and/or degrade azo dyes (Pandey et al. 2007).
2.1 Bacterial Biodegradation
Microbial decolorization and degradation is an eco-friendly and cost-competitive alternative to the chemical decomposition process (McMullan et al. 2001). Most of studies on dye biodegradation have focused on bacteria and fungi, in which bacteria are found to be more efficient and effective (Chang et al. 2001; Khehra et al. 2005). In the biodegradation, the original dye structure is cleaved into fragments by the microbial cells. Sometimes, bacterial degradation achieves complete mineralization, i.e. conversion of dyes into CO2, biomass and inorganics (Kolekar et al. 2012). Thus, textile industries have to develop an effective biological effluent treatment method as an alternative to the conventional physico-chemical effluent treatment methods.
2.1.1 Decolorization Under Aerobic Conditions
Several bacterial strains, that can aerobically decolorize azo dyes, have been isolated during the past few years. Many of these strains require organic carbon sources, as they cannot utilize dye as the growth substrate (Stolz 2001). Pseudomonas aeruginosa decolorized a commercial tannery and textile dye, Navitan Fast blue S5R, in the presence of glucose under aerobic conditions along with other azo dyes (Nachiyar and Rajkumar 2003). In the degradation of dyes, these bacteria cleave –N=N– bonds reductively and utilize amines as the source of carbon and energy for their growth. Such organisms are very specific to their substrate. Other bacterial strains with this trait are Xenophilus azovorans KF46 (previously known as Pseudomonas sp. KF46) and Pigmentiphaga kullae K24 (previously known as Pseudomonas sp. K24), which were grown aerobically on carboxy-orange I and carboxy-orange II, respectively (Zimmermann et al. 1982). These organisms, however, could not grow on structurally analogous sulfonated dyes, acid orange 20 and acid orange 7. Long adaptation of 4-aminobenzene sulfonate degrading Hydrogenophaga intermedia for growth on 4-carboxy-4′-sulfoazobenzene as the sole organic carbon source led to the isolation of other strain which reduced the dye and utilized the two amine metabolites formed (Blumel et al. 1998). Sphingomonas sp. strain 1CX, an obligate aerobic bacterium, was able to grow on azo dye, acid orange 7, as sole source of carbon, energy and nitrogen (Coughlin et al. 1999). This strain degraded only one of the component called amines (1-amino 2-naphthol) formed during decolorization of acid orange 7 and 4-aminobenzene sulfonate degradation.
2.1.2 Decolorization Under Anaerobic Conditions
Methanogenesis of complex organic compounds requires the co-ordinated participation of many different groups of bacteria, including acidogenic, acetogenic and methanogenic bacteria (Wuhrmann et al. 1980). Dye decolorization under these conditions requires an organic carbon/energy source. Simple substrates, like glucose, starch, acetate, ethanol and more complex ones, such as whey and tapioca, have been used for dye decolorization under methanogenic conditions (Yoo et al. 2001; van der Zee and Villaverde 2005). Extensive studies have been carried out to determine the role of the diverse groups of bacteria associated with the decolorization of azo dyes under methanogenesis (Carliell et al. 1996). However, a few investigators have shown that acidogenic as well as methanogenic bacteria also contribute to dye decolorization. Reduction under anaerobic conditions appears to be non-specific, as a group of azo compounds are decolorized. In such situation, the rate of decolorization is dependent on the added organic carbon source , as well as the dye structure (Stolz 2001). Furthermore, there is no correlation between decolorization rate and molecular weight, indicating that decolorization is not a specific process and cell permeability is not important for decolorization. Thus, anaerobic azo dye decolorization is the process where dye might act as an acceptor of electrons supplied by carriers of the electron transport chain. Alternatively, decolorization might be also attributed to non-specific extracellular reactions occurring between reduced compounds generated by the anaerobic biomass (van der Zee et al. 2001).
2.1.3 Decolorization Under Anoxic Conditions
Anoxic decolorization of various azo dyes by mixed aerobic and facultative anaerobic microbial consortia has been reported by various workers (Nigam et al. 1996; Khehra et al. 2005; Moosvi et al. 2005). Although many of these cultures were able to grow aerobically, decolorization was achieved only under anoxic conditions. Pure bacterial strains, such as Pseudomonas luteola, Aeromonas hydrophila , Bacillus subtilis, Proteus mirabilis and Pseudomonas sp. decolorized azo dyes under anoxic conditions (Chang et al. 2001; Yu et al. 2001; Chen et al. 2003). Azo dye decolorization by mixed as well as pure cultures generally required complex organic sources, such as yeast extract, peptone, or a combination of complex organic sources (Chen et al. 2003; Khehra et al. 2005). Glucose is the preferred substrate in anaerobic dye decolorization under methanogenic conditions, but its suitability for anoxic dye decolorization by facultative anaerobes and fermenting bacteria seems to vary depending on the bacterial culture. Decolorization of Mordant Yellow 3 by Sphingomonas xenophaga strain BN6 was greatly enhanced by glucose, whereas a significant decrease in azo dye decolorization in its presence was reported for Pseudomonas luteola, Aeromonas sp. and a few other mixed cultures (Haug et al. 1991; Chang et al. 2001). The negative effect of glucose on anoxic decolorization has been attributed either to a decrease in pH due to acid formation, or to catabolic repression (Chen et al. 2003).
2.1.4 Anaerobic Azo Dye Reduction
Anaerobic azo dye reduction is the reductive cleavage of azo linkages resulting in the formation of aromatic amines . As aromatic amines are generally colorless, azo dye reduction is also referred to as azo dye decolorization. The first study on azo dye reduction was published as early as 1937, when the decolorization of food azo dyes by lactic acid bacteria isolated from the human gut was reported (Brohm and Frohwein 1937). Hence, as the formation of toxic aromatic amines in humans was a matter of concern, research on bacterial azo dye reduction has focused on the activity of facultative anaerobic bacteria from mammalian intestines (Walker 1970; Chung et al. 1992). Later, when the removal of dyes from wastewater became a problem, bacteria from other origins were also used to investigate anaerobic azo dye reduction by pure cultures (Wuhrmann et al. 1980), mixed cultures (Haug et al. 1991), anaerobic sediments, digester sludge (Brown and Laboureur 1983), anaerobic granular sludge and activated sludge under anaerobic conditions. A large number of azo dyes that can be reduced by different bacteria indicate that azo dye reduction is a non-specific reaction and that the capability of reducing azo dye can be considered as a universal property of anaerobic bacteria.
2.1.5 Aerobic Oxidation of Aromatic Amines
Various substituted amino-benzene, amino-naphthalene and amino-benzidine compounds have been found aerobically biodegradable (Ekici et al. 2001). The conversion of these compounds generally requires enrichment of specialized aerobes. In some cases, biodegradation was only achieved in nitrogen-free medium (Konopka 1993). Especially sulfonated aromatic amines are very difficult to be degraded. Their low biodegradability is due to the hydrophilic nature of the sulfonate group which obstructs membrane transport. The biodegradation of sulfonated aromatic amines has been demonstrated for relatively simple sulfonated aminobenzene and aminonaphthalene compounds. Another transformation, that aromatic amines may undergo, is auto oxidation when exposed to oxygen. Especially aromatic amines with ortho-substituted hydroxyl groups are susceptible to auto-oxidation (Kudlich et al. 1999). Many aromatic amines, e.g. substituted anilines , amino-benzidines and naphthylamines, have been found to oxidize initially oligomers and eventually dark-colored polymers with low solubility which are easily removed from the water phase (Field et al. 1995).
2.1.6 Combined Anaerobic-Aerobic Treatment
The prerequisite for oxidative degradation of azo dye is reductive fission of the azo linkage under anaerobic condition, followed by aerobic degradation for complete removal of azo dyes (Field et al. 1995). Two different approaches can be discerned: sequential treatment in separate reactors and integrated treatment in a single reactor. The integrated approach is based on temporal separation of the anaerobic and the aerobic phase, as in sequencing batch reactors or on the principle that diffusion of oxygen in microbial biofilms is usually limited to 10–100 μm, so that anaerobic and aerobic conditions co-exist in a single environment (Lens et al. 1995). The removal of color in the anaerobic stage was generally high, mostly higher than 70 % and in several cases, even achieved almost 100 %. Color removal efficiencies differed widely in dyes, when the removal of different azo dyes was tested under similar conditions.
The reaction time is an important factor in the anaerobic removal of azo dyes. A decrease in the hydraulic retention time of the anaerobic stage was found to result in lower color removal efficiency and anaerobic azo dye reduction is also slow reaction (An et al. 1996). The biomass concentration also plays an important role in the anaerobic removal of azo dyes. Decrease in biomass and retention time of a sequencing batch reactor results in lower color removal efficiency (Lourenço et al. 2000). The percent recovery of aromatic amines ranged between <1 % to almost 100 %. A wide range in percent recovery may partly be explained by the difficulties encountered in analyzing these often chemically unstable compounds. Partial or complete removal of many aromatic amines can be suspected from the decrease or disappearance of the sometimes unidentified peaks in HPLC chromatograms (O’Neill et al. 1999; Lourenço et al. 2000) as well as from the decrease in UV absorbance. Moreover, a large decrease in toxicity to aerobic bacterial activity was measured between the effluents of anaerobic and aerobic stage (O’Neill et al. 1999). In summary, combined anaerobic-aerobic biological treatment holds to be a promising method to remove azo dyes from wastewater. However, there are two possible bottlenecks: (i) anaerobic azo dye reduction is a time-consuming process as reflected by the requirement of long reaction time and (ii) the fate of aromatic amines during aerobic treatment is not conclusively elucidated.
2.2 Fungal Biodegradation
Lignin-degrading fungi , white-rot fungi , can degrade a wide range of aromatics. In earlier reports, the dyes were degraded efficiently by wood-rotting fungi (e.g. Phanerochaete chrysosporium, Trametes sp.). Eventually, they were also found to be responsible for the degradation of lignin (David et al. 1994). In the fungal degradation, there are a few reports of adsorption of dyes on the fungal cells which forms sludge which requires further treatment. In this scenario, the fungal treatment of dye containing effluents is usually time consuming and difficult also (Erden el al. 2011). This property is mainly due to the relatively non-specific activity of fungal lignolytic enzymes, such as lignin peroxidase, manganese peroxidase and laccase. The reactions catalyzed by these extracellular enzymes are oxidation reactions, e.g. lignin peroxidase catalyses the oxidation of non-phenolic aromatics, whereas manganese peroxidase and laccase catalyze the oxidation of phenolic compounds (McMullan et al. 2001).
The degradation of dyes by white-rot fungi was first reported as early as in 1983 (Glenn and Gold 1983). Since then, it has been the subject of many research projects. Virtually all dyes from chemically distinct groups are prone to fungal oxidation, but there has been a wide difference among fungal species with respect to their catalyzing power and dye selectivity. Fungal degradation of aromatic structures is a secondary metabolic event which starts when nutrients (C, N and S) become limiting factors (Kirk and Farrell 1987). Therefore, the enzymes are optimally expressed under the starving conditions. However, supplementation of energy substrates and nutrients are necessary for the propagation of cultures. Other important factors for the cultivation of white-rot fungi and expression of lignolytic activity are the availability of enzyme co-factors and the pH of the environment. Although stable operation of continuous fungal bioreactors for the treatment of synthetic dye solutions has been achieved (Mielgo et al. 2001), but application of white-rot fungi for the removal of dyes from textile wastewater faces many problems. As the wastewater is not natural, the enzyme production in white-rot fungi may be unreliable and the biomass growth and retention in bioreactors may be a matter of serious concern (Stolz 2001). Treatment of large volumes of wastewater may be very difficult, as extraction and concentration of dyes are necessary factors prior for the fungal treatment (Nigam et al. 2000). Furthermore, the low optimum pH for lignin peroxidase (4.5–5.0) requires extensive acidification of the usually highly alkaline textile wastewater which causes inhibition of other useful microorganisms, like bacteria. Moreover, other wastewater constituents, especially aromatics, may also interfere with fungal dye degradation (Stolz 2001).
2.3 Algal Biodegradation
Degradation of azo dyes by algae has been reported only in few studies. The degradation pathway is thought to involve reductive cleavage of the azo linkage, followed by further degradation (mineralization) of aromatic amines formed. Hence, algae have been demonstrated to degrade several aromatic amines, even sulfonated azo dyes . In open wastewater treatment systems, especially in (shallow) stabilization ponds, algae may, therefore, contribute to the removal of azo dyes and aromatic amines from the wastewater. However, algal biodegradation of dyes has some limitations: one of very common problem is the adsorption. It was reported that Chlorella vulgaris could absorb Supranol Red 3BW (35.62 mg dye g−1 biomass), Lanaset Red 2GA (44.98 mg dye g−1 biomass), Levafix Navy Blue EBNA (43.17 mg dye g−1 biomass), but the degradation from algal biomass is not competitive if we compare it with the bacterial degradation (Lim et al. 2010). Oscillatoria curviceps can alone degrade Acid Black (100 ppm) up to 84 % in 8 days (Priya et al. 2011). The efficiency of adsorption is highly influenced by the structure of the dye, species of algae and physical parameters, like pH, temperature, etc. (Solís et al. 2012).
2.4 Bioreactor for Effective Decolorization of Textile Dyes
Many bioreactors for the degradation of textile dyes have been developed by many researchers and a fair amount of research was conducted to assess their viability for biodegradation study. Different types of bioreactors were developed, such as upflow anaerobic sludge blanket reactor (UASB), activated sludge reactors, etc. The UASB process was developed in the late 1970s (Lettinga et al. 1980). Initially the reactor was designed to treat concentrated industrial wastewater and its application was later extended to sewage treatment also. In the UASB reactor, wastewater flows upward through an anaerobic sludge bed, consisting of semi-immobilized microbial communities. The critical elements of the UASB reactor design are the influent distribution system, the gas-solid separator, and the effluent withdrawal system. The UASB process, the most popular among anaerobic treatment processes, relies on the establishment of a dense sludge bed or digestion zone at the bottom of the reactor, where the anaerobic degradation of the wastewater organics occurs and biogas is produced (Chinwetkitvanich et al. 2000).
Conventional activated sludge treatment of industrial wastes is an effective and highly economical system for the degradation of dyes and other organic pollutants from the wastewater. A lot of work has been done on the assessment of activated sludge system for the degradation of industrial effluents. The activated sludge treatment is the process in which the organic matter from wastewater is utilized as an energy source for the survival and multiplication of microorganisms in an aerobic environment. The microbes convert carbon into cell mass and oxidize the dyes finally to carbon dioxide and water. In addition, a limited number of microorganisms may exist in the activated sludge which get energy by oxidizing ammonia to nitrate, a process known as nitrification (Adav et al. 2009).
Aerobic sludge granulation is a biotechnological process used in biological wastewater treatment and treating chemical industrial influent (Adav et al. 2008). Now-a-days microbial granules play a major role in biological wastewater treatment due to several advantages over the conventional sludge flocks which include a denser and stronger aggregate structure, better settleability and ensured solid-effluent separation, higher biomass concentration and greater ability to withstand high organic loading, etc. (Lin et al. 2005; Adav et al. 2009).
The continuous aeration and feeding lead to an increase in bacterial cell mass and granulation was initiated at around 20 days and reached to mature granules in 60 days of reactor operation (Fig. 1a). High hydraulic selection pressure will retain denser, heavier, compact and smooth granules with improved metabolic activity (Chen et al. 2007). The SEM images of granules during the granulation process in the reactor depict average size of granules in the range of 5–8 mm (Fig. 1b).
a Systematic diagram of aerobic bioreactor for formation of bacterial granules. b SEM images of aerobic granules during the stages of granulation. (i) Cultures before granulation (2 days) and different stages of granulation (ii) 20 days, (iii) 40 days and (iv) 60 days (Kolekar 2010)
Biodegradation can be coupled with other degradation methods like, physical or chemical treatments. A few investigations have shown that azo dyes can be completely decolorized and some intermediates, such as aromatic amines with side groups (–SO3, –OH, –COOH, –Cl) containing metabolites were quantitatively detected (O’Neill et al. 1999). Actinomycetes were also tested for decolorization and degradation of textile dyes, as they produce peroxidases and also catalyses hydroxylation , oxidation, and dealkylation during various xenobiotic biotransformation (Goszczynski et al. 1994). McMullan et al. (2001) have reviewed the underlying mechanisms by which diverse categories of microorganisms from bacterial and fungal domains degrade dyestuffs. In nature, the biodegradation of pollutants (including textile dyes) was efficiently performed by a group of microorganisms in synergism. Hence, microbial consortium could be highly efficient than individual single microbe. Aerobic granules consist of dense microbial consortia packed with different microbial species and typically contain millions of organisms per gram of biomass. Thus, treating textile wastewater with environment friendly aerobic sludge granules could enhance decolorization and degradation of dyestuff, but it has been rarely documented. Recently, we have developed textile dyes degrading aerobic sludge granules in a batch reactor and evaluated their potential for textile dye removal and degradation. It was found that the dye degradation efficiency was very high as compared to individual cultures (Kolekar et al. 2012).
3 Factors Effecting Degradation Process
3.1 Microbial Growth and Energy Requirement for Degradation
The biodegradation efficiency depends on having the right microbes in the right place with the right environmental factors for degradation. The microbes (bacteria or fungi) have the physiological and metabolic potential to biodegrade the synthetic dyes (Boopathy 2000). The degradation process depends on biomass concentration (population diversity, enzyme activities), substrate (physico-chemical characteristics, molecular structure, and concentration) and a range of environmental factors like, dissolved oxygen, nitrate concentration, metals, salts, pH, temperature, moisture content, availability of electron acceptors and carbon and energy sources (Uddin et al. 2007; Phugare et al. 2010). Microorganisms are affected by changes in these parameters and consequently, their biodegradation capabilities are also affected. In the biodegradation process, the contaminant serves as an effective energy source. The contaminant may take part as primary, secondary or as a co-metabolite in the bacterial metabolism (Kolekar et al. 2008). The structure of contaminant has a high impact on degradation process, followed by its purity and solubility (Khan et al. 2013).
3.2 Oxygen Availability
The decolorization of various textile dyes by mixed aerobic and facultative anaerobic microbial consortia has been also reported earlier (Nigam et al. 1996; Moosvi et al. 2005). Although many of these cultures could grow aerobically, decolorization was achieved only under anaerobic or static anoxic conditions (Kolekar and Kodam 2012). Pure bacterial strains, such as Pseudomonas luteola, Aeromonas hydrophila , Bacillus subtilis, Pseudomonas sp., Proteus mirabilis , Bacillus fusiformis and Alishewanella sp. decolorized dyes under both anoxic and static anoxic conditions (Chang et al. 2001; Kolekar et al. 2008; Kolekar and Kodam 2012). It was observed that under static anoxic conditions, the dye decolorization of different dyes was more than 75 % as compared to agitation , which caused less than 30 % decolorization (Kolekar et al. 2008; Chaudhari et al. 2013). This indicates that azo dye degradation primarily starts in anoxic conditions. The facultative anaerobes were able to grow under both aerobic and anaerobic conditions and capable of decolorizing dyes under anaerobic condition (Nigam et al. 1996). In the first step of biodegradation, a reductive cleavage of the azo bond gives rise to the production of colorless metabolites, mainly aromatic amines which are further degraded in aerobic conditions. The first step usually occurs in anaerobic/static anoxic conditions; however, it was observed by several researchers that aerobic microbes were able to cleave the azo group by synthesizing oxygen insensitive azoreductases in the presence of molecular oxygen (Nachiyar and Rajkumar 2005). Bacterial strains, that decolorize textile dyes aerobically, were reported for few years. Many of these cultures require external carbon sources, because they cannot utilize dye as terminal electron acceptor (Stolz 2001). Textile dye was decolorized by P. aeruginosa in the presence of glucose under aerobic conditions (Nachiyar and Rajkumar 2003). Azo dye decolorization by mixed, as well as pure cultures generally requires complex organic sources, such as yeast extract, peptone, or a combination of complex organic source and carbohydrate (Chen et al. 2003; Khehra et al. 2005).
3.3 Optimization of Biodegradation Conditions
Microbial degradation depends on various physico-chemical operational conditions like, pH, temperature, salinity, heavy metals, radioactive molecules , chlorinated compounds and other contaminants (Boopathy 2000). In this scenario, wastewater contains many organic and inorganic pollutants. As textile wastewaters result from different classes of dyes, they vary in their composition. Every microorganism has some physico–chemical properties to work for biodegradation process, even if the microbes are in consortia these physico–chemical properties influence the biodegradation process (Mohana et al. 2008).
One of the factors, like pH has a major effect on the efficiency of textile dye degradation and the optimal pH for color removal in bacteria is often between pH 6.0 and 10.0 (Yoo et al. 2001; Kolekar et al. 2008; Kolekar and Kodam 2012). Most of fungi and yeast showed better decolorization at acidic or neutral pH (Mielgo et al. 2001). Many reports showed that the rate of dye degradation is higher at the optimal pH and gradually decreases at either side of optimum pH (Yoo et al. 2001; Saratale et al. 2009; Chaudhari et al. 2013). The effects of pH may be related to the transport of dye molecule across the cell membrane, which is considered as the rate limiting step for the decolorization (Kodam et al. 2005). The decolorization of Brilliant blue G by consortium of Galactomyces geotrichum and Bacillus sp. was found to have broad pH range from 5.0 to 9.0 (Jadhav et al. 2008).
During the biodegradation process, temperature also affects the biodegradation efficiency. The maximum rate of dye degradation by most of microbes depends on the optimum growth and temperature. It was observed that degradation potential was proportional to an increase of temperature, but within the optimum temperature range (Nachiyar and Rajkumar 2003; Kolekar et al. 2008). However, the rate of decolorization was reduced beyond optimum temperature. The reduction in decolorization can be attributed to the loss of cell viability or the denaturation of azoreductase enzyme which is a key enzyme for dye degradation (Saratale et al. 2009). It was also found that the dye degradation depended largely on the microbial optimum growth temperature (Stolz 2001; Khan et al. 2013). Recently, the degradation of reactive Orange M2R dye and chromate by Lysinibacillus sp. KMK-A was reported in the temperature range of 20–50 °C, but the degradation gradually decreased with increasing temperature (Chaudhari et al. 2013).
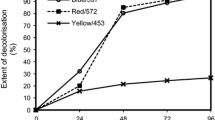
Microbial decolorization often gets affected in the presence of high amounts of salts (up to 10 g l−1 NaCl or Na2SO4) (Uddin et al. 2007). Many microbial species are able to decolorize azo dyes within a certain limit of salts. But in most of cases, they were unable to decolorize azo dyes at high salinity conditions. High salt concentration may cause plasmolysis or loss of activity of cells. It was observed that 85–95 % decolorization of acid orange 10 and disperse blue 79 by Bacillus fusiformis was found within 48 h for 0.5–3 % salt concentration, However, further increase in the salt (4 %) significantly decreased dye decolorization (Kolekar et al. 2008). There are only some bacteria which have the potential to grow, multiply in high salt concentrations and can degrade dye efficiently. Gracilibacillus sp. GTY strain, grown in the media containing 15 % (w/v) of NaCl, showed the best performance in dye decolorization (Uddin et al. 2007).
The dye decolorization affects with contaminants present in the wastewater. Many heavy metals are known to be toxic and can reduce the microbial growth which affects the biodegradation process. There are strict regulations to limit the amount of heavy metal present in the dye effluent in different countries, as some of them are known to be carcinogenic. The emphasis is currently on the reduction of trace metals in textile processing effluent. It has been suggested that the heavy metals present in the dye bath would not affect the performance of bacteria during the degradation of the organic matters in the biochemical process (Boopathy 2000; Uddin et al. 2007).
4 Involvement of Oxidoreductive Enzymes in Degradation Process
In recent years, the enzymatic approach has attracted much interest in the biodegradation and decolorization of textile and other industrial wastewater. Enzymatic treatment is usually very effective in the degradation of pollutants. The potential of the enzymes like cytochrome P450, manganese peroxidases, lignin peroxidases, laccases, polyphenol oxidases , and azoreductases, has been studied in the decolorization and degradation of dyes. The expression of the enzymes involved in dye degradation may vary with time, but dependent on the growth phase of the microorganisms, and is also influenced by other compounds present in the effluent. There is an increasing industrial value for the alternative cost effective method for treatment of a large volume of wastewater. The application of enzyme-based systems in waste water treatment is highly valuable (Husain 2006).
4.1 Cytochrome P450
Cytochrome P450 (CYP) represents one of the largest and oldest gene super families coding for enzymes present in the genomes of all biological kingdoms. The terminology CYP is uncommon for enzymes, because it is not based on function, but describes originally the spectral properties of b-type heme containing red pigments, which display a typical absorption band at 450 nm of their reduced carbon-monoxide bound form (Omura and Sato 1962). In mammals, azo dyes are reduced to aryl amines by CYP and a flavin dependent cytosolic reductase. The reactions catalysed can be extremely diverse as hydroxylations, N–, O–, and S-dealkylations, sulfoxidations, epoxidations, deaminations, desulfurations, dehalogenations, peroxidations, and N-oxide reductions (Bernhardt 2006). Since many of the individual CYP catalyse multiple reactions, the usual method of naming enzymes is inadequate for this group of proteins, and a systematic nomenclature has been derived based on structural homology. The CYP has been exclusively studied for detoxification and degradation. The degradation of various textile dyes by Alishewanella sp. KMK6 showed an induction in CYP activities in the cells after dye decolorization indicating the role of these enzymes (Kolekar and Kodam 2012). Recently Exiguobacterium sp. MG2 cytochrome P450 participated in the degradation of malachite green (N-methylated diaminotriphenylmethane), one of the most common dyes used in the textile industry (Wang et al. 2012).
4.2 Azoreductase
Azoreductase is also one of the biotransforming enzymes involved in decolorization of azo dyes by breaking azo bond. Azo dyes can be used by microorganisms as sole source of carbon, energy and nitrogen with the initial cleavage by azoreductase enzyme. Azoreductase requires NADH/NADPH as an electron donor to catalyze the reductive cleavage of azo bond. Recently, Nachiyar and Rajkumar (2005) have reported that purified azoreductase catalyzes the reductive cleavage of the azo bond of Navitan fast blue S5R in the presence of NADH as an electron donor, yielding metanilic acid and peri acid as degradation products (Fig. 2).
Cleavage of azo bond by purified azoreductase from Pseudomonas aeruginosa (Nachiyar and Rajkumar 2005)
It has been known for a long time that metabolic processes, such as azo reduction, can activate and sometimes also detoxify azo compounds. The reduction of azo bonds in azo dyes is important in their toxicity, mutagenicity and carcinogenicity (Garner et al. 1984). It has been shown that intestinal bacteria carried out reduction of azo compounds to produce aromatic amines. Some 45 different intestinal bacteria have been found to express azoreductases in a diverse collection of bacteria, including both strictly anaerobic Clostridium species and facultative anaerobic Enterobacteriaceae. It is not yet clear that how many different types of azoreductase enzymes are found in the human gut, or which of them are relevant for metabolism of azo dyes.
During the aerobic, “semi-aerobic” (in static anoxic) or anaerobic incubation of bacteria with azo compounds, amines were often detected which originated from a reductive cleavage of the azo bond. The azoreductase from Escherichia coli CD-2 was found to be responsible for the decolorization of azo dyes. The enzyme used methyl red as a substrate and the optimal pH value and temperature were pH 6.5 and 37 °C, respectively. The enzyme was stable under different physico-chemical conditions (Cui et al. 2012). Temperature stability studies showed that the enzyme was stable below 55 °C. However, azoreductase from Enterobacter agglomerans and P. aeruginosa lost most of their activities when the temperature reached to 50 °C (Nachiyar and Rajkumar 2005).
The aerobic reductive metabolism of azo dyes requires specific enzymes (aerobic azoreductases) that catalyze these reactions in the presence of molecular oxygen. In contrast to few reports of aerobic decolorization of azo dyes, a wide range of organisms are able to reduce azo compounds under anaerobic/anoxic conditions. This has been shown for purely anaerobic (e.g. Bacteroides sp., Eubacterium sp., Clostridium sp.), facultative anaerobic (e.g. Proteus vulgaris, Streptococcus faecalis ), and aerobic (e.g. Bacillus sp., Sphingomonas sp.) bacteria, yeasts, and even tissues from higher organisms (Walker 1970). Cell extracts generally showed much higher rates of anaerobic reduction of azo dyes than of resting cells (Wuhrmann et al. 1980). Reductases for NADH-DCIP (NADH-dichlorophenol indophenols), azo and riboflavin were reported for the degradation of different dyes, but the actual role of different reductase is still unknown (Fig. 3). FMN-dependent NADH-azoreductase, found to degrade azo compounds , catalyses the reductive cleavage of an azo group by a ping pong mechanism (Ooi et al. 2009). The degradation of reactive blue 59 (RB59) by Alishewanella sp. KMK6 showed an increase in azoreductase and DCIP reductase activities by 10 fold in the cells after dye decolorization, indicating the role of these enzymes in the biodegradation (Kolekar et al. 2013).
DCIP reductase activity by using DCIP as a substrate (Bidoia et al. 2010)
4.3 Peroxidases
In recent years, research has developed processes in which peroxidases are employed to remove/transform phenolic compounds from polluted wastewater. The application of enzyme preparations shows considerable benefits over the use of microorganisms. Textile dyes can be efficiently degraded by various peroxidases as well. The enzymes, produced by the fungus, are lignin peroxidase (LiP) and manganese peroxidase (MnP). The lignin peroxidase and manganese peroxidase, produced by the white rot fungus Phanerochaete chrysosporium , have a potential to oxidize the substrates by an electron transfer process or by radicals generated during the enzyme catalytic cycle. There are various peroxidases which are commonly used for biodegradation of textile wastewater. A common mechanism, in which the heme groups are the main constituents, catalyzes the reactions in the presence of hydrogen peroxide (Jamal et al. 2012; Zucca et al. 2012). The decolorization of dyes by white-rot fungi was first time reported by Glenn and Gold (1983) and been used by others to rapidly assess the biodegradative potential of a diverse group of white-rot fungi (Gao et al. 2006). Now, white-rot fungus has become a model for degradation research (Pazarlioglu et al. 2010).
The modified lignin has been used as a dispersing agent in synthesizing many textile dyes, Lignin peroxidase (LiP), also known as ligninase, was first discovered for partial decolorization of methylated lignin in vitro (Tien and Kirk 1983). The use of lignin degrading fungus has attracted increasing scientific interest, as these organisms are able to degrade a wide range of recalcitrant organic pollutants because they do not require pre-conditioning before the degradation of pollutants and the enzyme secretion depends on nutrient limitation rather than presence of pollutant (Wesenberg et al. 2003). The extracellular enzyme system from fungus also enables them to tolerate high concentration of pollutants.
Manganese peroxidase (MnP) from the lignin-degrading fungus Phanerochaete chrysosporium can oxidize a variety of organic compounds including dyes, but only in the presence of Mn(II). MnP catalysis occurs in a series of irreversible oxidation-reduction (redox) reactions which follow a ping-pong mechanism . MnP catalyzes the oxidation of several phenols and aromatic amine dyes (Mielgo et al. 2003). There are also reports of azo dye degradation by MnP from Bjerkandera adusta and Pleurotus eryngii . The reactions with the dyes were characterized by their apparent K m values ranging from 4 to 16 μM and specific activities ranging from 3.2 to 10.9 U/mg (Heinfling et al. 1997). Recently, it has been reported that Bjerkandera sp. BOS55 (ATCC 90940) was able to decolorize high dye concentration (up to 1,500 mg l−1) within 10 min reaction time. This is not because of change in pH, but has a specific oxidation capacity as high as 10 mg dye degraded per unit of MnP consumed which caused more than 90 % decolorization (Mielgo et al. 2003).
4.4 Laccases
Laccases are copper containing enzymes which catalyze the oxidation of several aromatic substances (phenols) with the preferable reduction of oxygen to water (Robles et al. 2000). Phenols are oxidized by laccases to generate phenoxy radicals , which couple to form oligomeric and polymeric products . The laccases produced by the white-rot fungus during its secondary metabolic stage of growth are known to oxidize a wide variety of organic compounds. Laccase decolorizes some textile dyes without direct cleavage of the azo bond through a highly non-specific free radical mechanism, thereby avoiding the formation of toxic aromatic amines. Because of its wide range of substrates laccases can be used for biodegradation of textile dyes. Industrial dye decolorization by laccases from ligninolytic fungi (Pleurotus ostreatus, Pleurotus ostreiformis, Trametes versicolor , etc.) has been reported (Rodrıguez et al. 1999). Recently, laccase was isolated from the white-rot fungus Lenzites gibbosa and expressed in Pichia pastoris. This recombinant laccase efficiently decolorized alizarin red, neutral red, Congo red and crystal violet, without the addition of any redox mediators . This study strongly suggests that the decolorization capacity of recombinant laccase could be useful as biocatalyst for the treatment of dye-containing effluents (Zheng et al. 2014).
5 Mechanism and Pathways of Degradation
Biodegradation refers to chemical decomposition of organic substances by living organisms. In the decomposition or degradation, microbial enzyme systems act as biocatalysts to break down the original dye structure. There are several reports that microorganisms including bacteria, fungi, and yeasts can degrade textile dyes (Kolekar et al. 2008; Zucca et al. 2012). The reduction of azo linkage mediated is the initial step by the enzyme, azoreductase in the biodegradation of azo dyes (Levine 1991; Chung et al. 1992). The mineralization of dyes is usually achieved with two stage anaerobic/aerobic treatments. In this process, several environmental and physiological factors can influence the microbial activity and consequently the efficacy and effectiveness of complete biodegradation. The biodegradation process is depending on many factors, like concentration and structure of dyes, temperature, pH and the microorganisms involved in it. Considering all these factors, the degradation of dyes produced many small molecules which become very difficult to be degraded by the organism. These small molecules are aromatic amines and may cause more toxic and carcinogenic effect on the flora and fauna of water bodies, where aromatic amine containing waste water is getting discarded. In this scenario, it is very important to analyze the degradation of the dye as well as their intermediates, especially aromatic amines (Forss and Welander 2009). The study of the biodegradation products of synthetic dyes is very important in order to know about the environmental fate of these pollutants. Telke et al. (2010) reported the degradation of Congo red (1.0 g l−1) within 12–60 h duration by an isolated bacterium Pseudomonas sp. SU-EBT. As shown in Fig. 4, the complete mineralization of azo dyes without the release of aromatic amines has also been observed in Reactive Blue 59 degradation by Alishewanella sp. KMK6, where the single microorganism was able to degrade 2.5 g l−1 dye within 6 h (Kolekar and Kodam 2012).
Mechanism of degradation of reactive blue 59 by Alishewanella sp. strain KMK6 (Kolekar and Kodam 2012)
It is well known that several enzymes are present in the periplasmic and cytosolic fractions of the microorganisms where azoreductase cleaves an azo bond by reductive cleavage. The coordinated action of these enzymes give rise to low molecular weight molecules from complex dyes. The tracking of the various enzyme activities evidenced an involvement of various oxidative as well as reductive enzymes during the decolorization process. Induction of various enzymes during decolorization gives an additional insight of decolorization mechanism and also supports the active role of microorganisms in the biodegradation process (Phugare et al. 2010), mainly through oxidative enzymes , such as cytochrome P-450 and also many reductases, such as azoreductase, NADH–DCIP reductase , and malachite green reductase influence the status of biotransformation enzymes (Wang et al. 2012). The role of cytosolic enzymes, like oxidase, DCIP reductase, and azoreductase in dye decolorization is well documented (Phugare et al. 2010; Kolekar and Kodam 2012).
6 Microbial Toxicity of Dyes and Their Degradation Products
The color in wastewater is highly visible and affects aesthetics, water transparency and gas solubility in water bodies, The toxicity (i.e. mortality, genotoxicity, mutagenicity and carcinogenicity) studies include the tests with aquatic organisms (fish, algae, bacteria, etc.), mammals and plants. The acute toxicity of dyestuffs is generally low. Algal growth in presence of different commercial dyestuffs was not inhibited at dye concentrations below 1 ppm, whereas the growth was severely inhibited at higher concentrations. The most acutely toxic dyes for algae are cationic basic dyes (Greene and Baughman 1996). Fish mortality tests showed that 2 % out of 3,000 commercial dyestuffs tested had LC50 values below 1 ppm. Basic dyes are found to be most acutely toxic to fish, especially those with a triphenylmethane structure. Fish seems to be relatively sensitive to many acid dyes. Mortality tests with rats showed that only 1 % out of 4,461 commercial dyestuffs tested had LD50 values below 250 mg kg−1 body weight (Clarke and Anliker 1980). Therefore, the chance of human mortality due to acute dyestuff toxicity is probably very low. However, acute sensitization reactions by humans to dyestuffs often occur. Especially, some disperse dyestuffs have been found to cause allergic reactions, i.e. eczema or contact dermatitis (Specht and Platzek 1995).
Chronic effects of dyestuffs, especially of azo dyes, have been studied for several decades. Researchers had traditionally focused on the effects of food colorants, usually azo compounds . Furthermore, the effects of occupational exposure of human workers to dyestuffs in dye manufacturing and dye utilizing industries have received little attention. Azo dyes in purified form are seldom mutagenic or carcinogenic, except for some azo dyes with free amino groups (Brown and Devito 1993). However, some of the degradation products of azo dyes are known to be mutagenic and/or carcinogenic. In mammals, metabolic activation (or reduction) of azo dyes is mainly due to bacterial activity in the anaerobic parts of the lower gastrointestinal tract. Various human other organs, especially liver and kidneys, can reduce azo dyes. After azo dye reduction in the intestinal tract, the released aromatic amines are absorbed by the intestine and excreted in the urine. The acute toxic hazard of aromatic amines is carcinogenesis, especially bladder cancer. The carcinogenicity mechanism probably includes the formation of acyloxy amines through N-hydroxylation and N-acetylation of the aromatic amines, followed by O-acylation. These acyloxy amines can be converted to nitrenium and carbonium ions that bind to DNA and RNA and induce mutations and tumor formation (Brown and Devito 1993). The mutagenic activity of aromatic amines is strongly related to molecular structure. Recently, mutagenicity and phytotoxicity tests were performed on different biological sequencing batch reactor (SBR) systems, which studied for efficiency of effectiveness in removing toxic substances from textile wastewaters and a comparison was made to evaluate the ecotoxicity results observed before and after wastewater treatment (Giorgetti et al. 2011). The biodegradation products of Congo red and textile industry effluent by Pseudomonas sp. SU-EBT were found to be non-phytotoxic to Sorghum bicolor, Vigna radiata, Lens culinaris and Oryza sativa plants as compared to Congo red and textile industry effluent (Telke et al. 2010).
7 Conclusions
Presence of color and its causative compounds has always been undesirable in water used for either industrial or domestic purposes. Amongst complex industrial wastewater with various types of coloring agents, dye wastes are predominant. The color in wastewater is highly visible and affects aesthetics, water transparency and gas solubility in water bodies and their toxicity has been investigated in numerous researches. Dyes are difficult to be decolorized due to their complex structure, synthetic origin and recalcitrant nature. Hence, it becomes obligatory to remove them from industrial effluents before being disposed into water bodies. The degradation and decolorization of textile dyes within safer limits prior to release from treatment plant is essential for environmental safety. In this context, several workers have reported degradation and decolorization of dyes by using bacteria and fungus under different environmental conditions (pH, temperature, salinity, anaerobic/aerobic, nutrients, etc.). Besides, many researchers have studied the use of bacterial consortium for dye degradation. In addition to the bacterial consortium, a detailed study is needed for the use of bacterial granules in bioremediation of dye wastewaters.
8 Future Prospectives
Biodegradation of textile dyes using different microbes, like bacteria, fungi, and algae has become a promising approach for the treatment of different dyes from industrial wastewaters . To improve the degradation potential of microorganisms, they can be exposed gradually to higher concentration of dyes which will ultimately make them acclimatize and evolve. It has been observed that the microbial communities adapting to higher concentrations of dyes showed better decolorization ability. In this process, the up regulated genes and proteins can be explored further for designing the microbes with enhanced degradation capabilities. Development of aerobic granules is a promising technique for the treatment of wastewater with high strength of textile dye. This granular system may be helpful in many conditions and can be operated for the treatment of larger organic loads. The granular system has good settling capacity which can treat the effluents better than any conventional systems. The formation of these granules and mechanisms of degradation of high organic loads and their application for different industrial effluent needs to be explored in depth for their application at a large scale for removal of dyes from wastewater.
References
Adav SS, Lee DJ, Lai JY (2009) Functional consortium from aerobic granules under high organic loading rates. Bioresour Technol 100:3465–3470
Adav SS, Lee DJ, Show KY, Tay JH (2008) Aerobic granular sludge: recent advances. Biotechnol Adv 26:411–423
An H, Qian Y, Gu XS, Tang WZ (1996) Biological treatment of dye wastewaters using an anaerobic-oxic system. Chemosphere 33:2533–2542
Anjaneyulu Y, Chary NS, Raj DSS (2005) Decolorization of industrial effluents—available methods and emerging technologies—a review. Rev Environ Sci Biotechnol 4:245–273
Bernhardt R (2006) Cytochromes P450 as versatile biocatalysts. J Biotechnol 124:128–145
Bidoia ED, Montagnolli RN, Lopes PRM (2010) Microbial biodegradation potential of hydrocarbons evaluated by colorimetric technique: a case study. In: Mendez-Vilas A (ed) Current research, technology and education topics in applied microbiology and microbial biotechnology. Formatex, Spain, pp 1277–1288
Blumel S, Contzen M, Lutz M, Stolz M, Knackmuss HJ (1998) Isolation of a bacterial strain with the ability to utilize the sulfonated azo compound 4-carboxy-4′- sulfoazobenzene as sole source of carbon and energy. Appl Microbiol Biotechnol 64:2315–2317
Boopathy R (2000) Factors limiting bioremediation technologies. Bioresour Technol 74:63–67
Brohm K, Frohwein E (1937) Nachweis von durch Säueringentfarbten künstlichen Eigelbfrabstoffen in Milchspeiseeis. Zbl Lebensmitt Forsch 73:30
Brown D, Laboureur P (1983) The degradation of dyestuffs: part I—primary biodegradation under anaerobic conditions. Chemosphere 12:397–404
Brown MA, Devito SC (1993) Predicting azo dye toxicity. Crit Rev Environ Sci Technol 23:249–324
Carliell CM, Barclay SJ, Buckely CA (1996) Treatment of exhausted reactive dye bath effluent using anaerobic digestion: laboratory and full scale trails. Water SA 22:225–233
Chang JS, Chou C, Lin PJ, Ho JY, Hu TL (2001) Kinetic characteristics of bacterial azo-dye decolorization by Pseudomonas luteola. Water Res 35:2841–2850
Chaudhari AU, Tapase SR, Markad VL, Kodam KM (2013) Simultaneous decolorization of reactive Orange M2R dye and reduction of chromate by Lysinibacillus sp. KMK-A. J Hazard Mater 262:580–588
Chen KC, Wu JY, Huang CC, Liang YM, Hwang SCJ (2003) Decolorization of azo dye using PVA-immobilized microorganisms. J Biotechnol 101:241–252
Chen Y, Jiang W, Liang DT, Tay JH (2007) Structure and stability of aerobic granules cultivated under different shear force in sequencing batch reactors. Appl Microbiol Biotechnol 76:1199–1208
Chinwetkitvanich S, Tuntoolvest M, Panswad T (2000) Anaerobic decolorization of reactive dye bath effluents by a two stage UASB system with Tapioca as co-substrate. Water Res 34:2223–2232
Chung KT, Stevens SE, Cerniglia CE (1992) The reduction of azo dyes by the intestinal microflora. Crit Rev Microbiol 18:175–190
Clarke A, Anliker R (1980) Organic dyes and pigments. In: Hutzinger O (ed) The handbook of environmental chemistry, vol 3., Part A—anthropogenic compoundsSpringer, Berlin, Heidelberg, New York, pp 181–215
Clarke EA, Steinle D (1995) Health and environmental safety aspects of organic colorants. Rev Prog Color 25:1–10
Coughlin MF, Kinkle BK, Bishop PL (1999) Degradation of azo dyes containing aminonaphthol by Sphingomonas sp. strain 1CX. J Ind Microbiol Biotechnol 23:341–346
Cui D, Li G, Zhao D, Gu X, Wang C, Zhao M (2012) Purification and characterization of an azoreductase from Escherichia coli CD-2 possessing quinone reductase activity. Process Biochem 47:544–549
David PB, Steven DA, Barr DP, Aust SD (1994) Mechanisms white rot fungi use to degrade pollutants. Environ Sci Technol 28:78A–87A
Dos Santos AB, Cervantes FJ, Van Lier JB (2007) Review paper on current technologies for decolorization of textile wastewaters: perspectives for anaerobic biotechnology. Bioresour Technol 98:2369–2385
Ekici P, Leupold G, Parlar H (2001) Degradability of selected azo dye metabolites in activated sludge systems. Chemosphere 44:721–728
Erden E, Kaymaz Y, Pazarlioglu NK (2011) Biosorption kinetics of a direct azo dye Sirius Blue K-CFN by Trametes versicolor. Electron J Biotechnol 14:1–10
Field JA, Stams AJM, Kato M, Schraa G (1995) Enhanced biodegradation of aromatic pollutants in co-cultures of anaerobic and aerobic bacterial consortia. Antonie Van Leeuwenhoek 67:47–77
Forss J, Welander U (2009) Decolorization of reactive azo dyes with microorganisms growing on soft wood chips. Intl Biodeter Biodegr 63:752–758
Gahr F, Hermanutz F, Oppermann W (1994) Ozonation- an important technique to comply with new German laws for textile wastewater treatment. Water Sci Technol 30:255–263
Gao D, Wen X, Zeng Y, Qian Y (2006) Decolorization of a textile-reactive dye with Phanerochaete chrysosporium incubated in different ways under non-sterile conditions. Water Pract Technol 1:10801–10806
Garner DL, Johnson LA, Lake S, Chaney N, Stephenson D, Pinkel D, Gledhill BL (1984) Morphological and ultrastructural characterization of mammalian spermatozoa processed for flow cytometric DNA analysis. Gamete Res 10:339–352
Giorgetti L, Talouizte H, Merzouki M, Caltavuturo L, Geri C, Frassinetti S (2011) Genotoxicity evaluation of effluents from textile industries of the region Fez-Boulmane, Morocco: a case study. Ecotoxicol Environ Saf 74:2275–2283
Glenn JK, Gold MH (1983) Decolorization of several polymeric dyes by the lignin degrading basidiomycete Phanerochaete chrysosporium. Appl Environ Microbiol 45:1741–1747
Goszczynski S, Paszczynski A, Pasti-Grigsby MB, Crawford RL, Crawford DL (1994) New pathway for degradation of sulfonated azo dyes by microbial peroxidases of by Phanerochaete chrysosporium and Streptomyces chromofuscus. J Bacteriol 176:1339–1347
Greene JC, Baughman GL (1996) Effects of 46 dyes on population growth of freshwater green alga Selenastrum capricornutum. Text Chem Color 28:23–30
Haug W, Schmidt A, Nortemann B, Hempel DC, Stolz A, Knackmuss HJ (1991) Mineralization of the sulfonated azo dye mordant yellow 3 by a 6-aminonaphthalene-2-sulfonate-degrading bacterial consortium. Appl Environ Microbiol 57:3144–3149
Heinfling A, Berghauer M, Szewzyk U (1997) Biodegradation of azo and phthalocyanine dyes by Trametes versicolor and Bjerkandera adusta. Appl Microbiol Biotechnol 48:261–266
Husain Q (2006) Potential applications of the oxidoreductive enzymes in the decolorization and detoxification of textile and other synthetic dyes from polluted water: a review. Crit Rev Biotechnol 26:201–221
Jadhav SU, Jadhav MU, Kagalkar AN, Govindwar SP (2008) Decolorization of brilliant blue G Dye mediated by degradation of the microbial consortium of Galactomyces geotrichum and Bacillus sp. J Chin Inst Chem Engrs 39:563–570
Jamal F, Singh S, Qidwai T, Pandey PK, Singh D (2012) Optimization of internal conditions for biocatalytic dye color removal and a comparison of redox mediator’s efficiency on partially purified Trichosanthes dioica peroxidase. J Mol Catal B: Enzym 74:116–124
Khan R, Bhawana P, Fulekar MH (2013) Microbial decolorization and degradation of synthetic dyes: a review. Rev Environ Sci Biotechnol 12:75–97
Khehra MS, Saini HS, Sharma DK, Chadha BS, Chimni SS (2005) Decolorization of various azo dyes by bacterial consortia. Dyes Pigm 67:55–61
Kirk TK, Farrell RL (1987) Enzymatic “combustion”: the microbial degradation of lignin. Ann Rev Microbiol 41:465–505
Kodam KM, Soojhawon I, Lokhande PD, Gawai KR (2005) Microbial decolorization of reactive azo dyes under aerobic conditions. World J Microbiol Biotechnol 21:367–370
Kolekar YM (2010) Isolation, characterization and evaluation of dye degradation potential of the novel bacterial species, Alishewanella soli. Ph.D. thesis, University of Pune, India (http://shodhganga.inflibnet.ac.in/handle/10603/3221)
Kolekar YM, Kodam KM (2012) Decolorization of textile dyes by Alishewanella sp. KMK6. Appl Microbiol Biotechnol 95:521–529
Kolekar YM, Konde PD, Markad VL, Kulkarni SV, Chaudhari AU, Kodam KM (2013) Effective bioremoval and detoxification of textile dye mixture by Alishewanella sp. KMK6. Appl Microbiol Biotechnol 97:881–889
Kolekar YM, Nemade HN, Markad VL, Adav SS, Patole MS, Kodam KM (2012) Decolorization and biodegradation of azo dye, reactive blue 59 by aerobic granules. Bioresour Technol 104:818–822
Kolekar YM, Pawar SP, Gawai KR, Lokhande PD, Shouche YS, Kodam KM (2008) Decolorization and degradation of disperse blue 79 and acid orange 10, by Bacillus fusiformis KMK5 isolated from the textile dye contaminated soil. Bioresour Technol 99:8999–9003
Konopka A (1993) Isolation and characterization of a subsurface bacterium that degrades aniline and methylanilines. FEMS Microbiol Lett 111:93–99
Kudlich M, Hetheridge MJ, Knackmuss HJ, Stolz A (1999) Autoxidation reactions of different aromatic o-aminohydroxynaphthalenes that are formed during the anaerobic reduction of sulfonated azo dyes. Environ Sci Technol 33:896–901
Lens PN, De PMP, Cronenberg CC, Verstraete WH (1995) Sulfate reducing and methane producing bacteria in aerobic wastewater treatment systems. Water Res 29:871–880
Lettinga G, van Velsen AFM, Hobma SW, de Zeeuw W, Klapwijk A (1980) Use of the upflow sludge blanket (USB) reactor concept for biological wastewater treatment, especially for anaerobic treatment. Biotechnol Bioeng 22:699–734
Levine WG (1991) Metabolism of azo dyes: implication for detoxification and activation. Drug Metab Rev 23:253–309
Lim SL, Chu WL, Phang SM (2010) Use of Chlorella vulgaris for bioremediation of textile wastewater. Bioresour Technol 101:7314–7322
Lin HL, Jianlong W, Xianghua W, Yi Q (2005) The formation and characteristics of aerobic granules in sequencing batch reactor (SBR) by seeding anaerobic granules. Process Biochem 40:5–11
Lourenço ND, Novais JM, Pinheiro HM (2000) Reactive textile dye color removal in a sequencing batch reactor. Water Sci Technol 42:321–328
McMullan G, Meehan C, Conneely A, Nirby N, Robinson T, Nigam P, Banat IM, Marchant R, Smyth WF (2001) Mini review: microbial decolorization and degradation of textile dyes. Appl Microbiol Biotechnol 56:81–87
Mielgo I, Lopez C, Moreira MT, Feijoo G, Lema JM (2003) Oxidative degradation of azo dyes by manganese peroxidase under optimized conditions. Biotechnol Prog 19:325–331
Mielgo I, Moreira MT, Feijoo G, Lema JM (2001) A packed-bed fungal bioreactor for the continuous decolorization of azo-dyes (Orange II). J Biotechnol 89:99–106
Mohana SV, Shrivastav S, Divecha J, Madamwar D (2008) Response surface methodology for optimization of medium for decolorization of textile dye Direct Black 22 by a novel bacterial consortium. Bioresour Technol 99:562–569
Moosvi S, Keharia H, Madamwar D (2005) Decolorization of textile dye reactive violet by a newly isolated bacterial consortium. RVM 11.1. World J Microbiol Biotechnol 21:667–672
Nachiyar CV, Rajkumar GS (2003) Degradation of tannery and textile dye, Navitan Fast Blue S5R by Pseudomonas aeruginosa. World J Microbiol Biotechnol 19:609–614
Nachiyar CV, Rajkumar GS (2005) Purification and characterization of an oxygen insensitive azoreductase from Pseudomonas aeruginosa. Enzym Microb Technol 36:503–509
Nigam P, Armour G, Banat IM, Singh D, Marchant R (2000) Physical removal of textile dyes from effluents and solid-state fermentation of dye-adsorbed agricultural residues. Bioresour Technol 72:219–226
Nigam P, Banat IM, Singh D, Marchant R (1996) Microbial process for the decolorization of textile effluent containing azo, diazo and reactive dye. Process Biochem 31:435–442
Omura T, Sato R (1962) A new cytochrome in liver microsomes. J Biol Chem 237:1375–1376
O’Neill C, Hawkes FR, Hawkes DL, Lourenco ND, Pinheiro HM, Delée W (1999) Color in textile effluents—sources, measurement, discharge consents and simulation: a review. J Chem Technol Biotechnol 74:1009–1018
Ooi T, Shibata T, Matsumoto K, Kinoshita S, Taguchi S (2009) Comparative enzymatic analysis of azoreductases from Bacillus sp. B29. Biosci Biotechnol Biochem 73:1209–1211
Ouzman S (1998) Towards a mindscape of landscape: rock art as expression of world-understanding. In: Chippindale C, Taçon PSC (eds) The archaeology of rock art. Cambridge University Press, Cambridge, UK, pp 30–41
Pandey A, Singh P, Iyengar L (2007) Bacterial decolorization and degradation of azo dyes. Int Biodeter Biodegr 59:73–84
Pazarlioglu NK, Akkaya A, Akdogan HA, Gungor B (2010) Biodegradation of direct blue 15 by free and immobilized Trametes versicolor. Water Environ Res 82:579–585
Phugare SS, Kalyani DC, Patil AV, Jadhav JP (2010) Textile dye degradation by bacterial consortium and subsequent toxicological analysis of dye and dye metabolites using cytotoxicity, genotoxicity and oxidative stress studies. J Hazard Mater 186:713–723
Priya B, Uma L, Ahamed AK, Subramanian G, Prabaharan D (2011) Ability to use the diazo dye C. I. Acid Black 1 as a nitrogen source by the marine cyanobacterium Oscillatoria curviceps BDU92191. Bioresour Technol 102:7218–7223
Raghavacharya C (1997) Color removal from industrial effluents: a comparative review of available technologies. Chem Eng World 32:53–54
Robles A, Lucas R, De Cienfuegos AG, Galvez A (2000) Phenoloxidase (laccase) activity in strain of the hyphomycete Chalara paradoxa isolated from olive mill wastewater disposal ponds. Enz Microb Technol 26:484–490
Rodriguez E, Pickard MA, Vazquez-Duhalt R (1999) Industrial dye decolorization by peroxidase from Geotrichum candidum Dec 1 involved in decolorization of dyes. Appl Environ Microbiol 65:1029–1035
Saratale RG, Saratale GD, Kalyani DC, Chang JS, Govindwar SP (2009) Enhanced decolorization and biodegradation of textile azo dye Scarlet R by using developed microbial consortium GR. Bioresour Technol 100:2493–2500
Solís M, Solís A, Pérez HI, Manjarrez N, Flores M (2012) Microbial decoloration of azo dyes: a review. Process Biochem 47:1723–1748
Specht K, Platzek T (1995) Textile dyes and finishes—remarks to toxicological and analytical aspects. Deut Lebensm Rundsch 91:352–359
Stolz A (2001) Basic and applied aspects in the microbial degradation of azo dyes. Appl Microbiol Biotechnol 56:69–80
Strickland AF, Perkins WS (1995) Decoloration of continuous dyeing wastewater by ozonation. Text Chemist Colorist 27:11–16
Telke AA, Joshi SM, Jadhav SU, Tamboli DP, Govindwar SP (2010) Decolorization and detoxification of Congo red and textile industry effluent by an isolated bacterium Pseudomonas sp. SU-EBT. Biodegradation 21:283–296
Tien M, Kirk TK (1983) Lignin degrading enzyme from Phanerochaete chrysosporium: purification, characterization and catalytic properties of unique H2O2-requiring oxygenase. Proc Nat Acad Sci USA 81:2280–2284
Uddin MS, Zhou J, Qu Y, Guo J, Wang P, Zhao LH (2007) Biodecolorization of azo dye acid red b under high salinity condition. Bull Environ Contam Toxicol 79:440–444
van der Zee FP, Lettinga G, Field JA (2001) Azo dye decolorization by anaerobic granular sludge. Chemosphere 44:1169–1176
van der Zee FP, Villaverde S (2005) Combined anaerobic-aerobic treatment of azo dyes—a short review of bioreactor studies. Water Res 39:1425–1440
Walker R (1970) The metabolism of azo compounds: a review of the literature. Food Cosmet Toxicol 8:659–676
Wang J, Gao F, Liu Z, Qiao M, Niu X, Zhang KQ, Huang X (2012) Pathway and molecular mechanisms for malachite green biodegradation in Exiguobacterium sp. MG2. PLoS One 7:e51808
Wesenberg D, Kyriakides I, Agathos SN (2003) White-rot fungi and their enzymes for the treatment of industrial dye effluents. Biotechnol Adv 22(1–2):161–187
Wuhrmann K, Mechsner K, Kappeler T (1980) Investigation on rate-determining factors in the microbial reduction of azo dyes. Eur J Appl Microbiol Biotechnol 9:325–338
Yoo ES, Libra J, Adrian L (2001) Mechanism of decolorization of azo dyes in anaerobic mixed culture. J Environ Eng 127:844–849
Yu J, Wang X, Yue PL (2001) Optimal decolorization and kinetic modeling of synthetic dyes by Pseudomonas strains. Water Res 35:3579–3586
Zheng M, Chi Y, Yi H, Shao S (2014) Decolorization of alizarin red and other synthetic dyes by a recombinant laccase from Pichia pastoris. Biotechnol Lett 36:39–45
Zimmermann T, Kulla HG, Leisinger T (1982) Properties of purified orange II azoreductase, the enzyme initiating azo dye degradation by Pseudomonas KF 46. Eur J Biochem 129:197–203
Zollinger H (2003) Color chemistry: syntheses, properties, and applications of organic dyes and pigments, 3rd edn. Wiley, Weinheim
Zucca P, Rescigno A, Pintus M, Rinaldi AC, Sanjust E (2012) Degradation of textile dyes using immobilized lignin peroxidase-like metalloporphines under mild experimental conditions. Chem Cent J 6:1–8
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Kodam, K.M., Kolekar, Y.M. (2015). Bacterial Degradation of Textile Dyes. In: Singh, S. (eds) Microbial Degradation of Synthetic Dyes in Wastewaters. Environmental Science and Engineering(). Springer, Cham. https://doi.org/10.1007/978-3-319-10942-8_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-10942-8_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-10941-1
Online ISBN: 978-3-319-10942-8
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)