Abstract
This chapter describes the application of genomic, transcriptomic, proteomic, and metabolomic methods in the study of SARS-CoV-2 variants of concern. We also describe the important role of machine learning tools to identify the most significant biomarker signatures and discuss the latest point-of-care devices that can be used to translate these findings to the physician’s office or to bedside care. The main emphasis is placed on increasing our diagnostic capacity and predictability of disease outcomes to guide the most appropriate treatment strategies.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
According to databases such as Worldometer [1] and the Johns Hopkins Institute [2], more than 648 million people have been infected by the SARS-CoV-2 virus which causes COVID-19 disease and more than 6.6 million of these individuals have died (as of December 1, 2022). However, the actual number of infected people is likely to be much higher, with some studies estimating that almost 50% of the world population has been infected [3]. From early on in the pandemic, it was deemed that early, rapid, and accurate detection of COVID-19 disease was critical for better management of the crisis, as well as for facilitating better therapeutic outcomes, and a lower damaging effect on healthcare and financial systems [4,5,6,7]. However, at that time, most of the testing for such infectious diseases was performed in centralized laboratories by trained personnel, and it could take up to several days for the results of these tests. Given the urgency evidenced by the COVID-19 pandemic and the threat of future outbreaks, it is clear that there is a need for more user-friendly and diagnostic tests that can be used in a point-of-care (POC) capacity. Advances made in the areas of microfluidics, miniaturization, and integration have now enabled the application of these devices in standard laboratory and clinical environments as well as in emergency use scenarios [8,9,10,11,12].
A major obstacle in the use of POC devices occurs at the sample stage. Importantly, this should involve as little human interaction as possible as this is where errors or biases can be introduced. If there is a rush of infected persons to get to the site of testing, as occurred during the early stages of the COVID-19 pandemic [13], there is also the chance of cross-infections. One solution to this is that the testers visit the prospective patients in their homes or places of work and carry out the testing there. Of course this would require trained professionals and for the testing kit to be portable, with a sample-sealing capability to avoid cross-contaminations. This would help to minimize the number of false positives and false negatives.
If the test is polymerase chain reaction (PCR)-based, the sample acquisition and preparation steps are critical. This is due to the presence of constituents in body fluids such as blood serum/plasma [14] and saliva [15] that can inhibit the amplification step in PCR [16]. However, there have been advances in overcoming these potential effects and making the sample preparation step more PCR-friendly. For example, we recently described the use of a commercially available inhibitor-tolerant PCR mix which circumvents the need for extraction, allowing for a faster and more accurate identification of the infective agent and determination of viral load [17, 18]. Multiplex PCR platforms offer a number of advantages of single assay systems as they can significantly lower test times, conserve samples, lower costs, while allowing for simultaneous analysis of multiple pathogens such as influenza types A and B [17, 19, 20] and different SARS-CoV-2 variants [18, 21, 22]. The correct identification of a pathogen using such systems would also allow patient stratification or triage for the most appropriate treatment and also provide a means of correctly determining across a suspected group of pathogens in the different waves of an outbreak and/or the emergence of a new pandemic.
The COVID-19 pandemic has enhanced the need and drive of researchers around the world to develop POC devices to enable early diagnosis of SARS-CoV-2 infection, variant subtyping, and to lay the groundwork in the advent of future pandemics. In this chapter, we describe some of the major developments which have served to advance these efforts.
2 The Omicron Variant
The B.1.1.529/BA.1 SARS-CoV-2 variant (termed Omicron by the WHO) was first reported on 24 November 2021 [23, 24], with cases appearing in Botswana and then South Africa (Fig. 27.1) [23]. By 10 Jan 2022, it had been reported in 89 countries and reached a peak infection rate of more than four million cases per day on 21 Jan 2022 [1, 2, 25]. After this, several Omicron sub-variants evolved which led to further smaller waves and perpetuation of the pandemic [26]. Because of the increase in diversity and highly infectious nature of this variant, the WHO updated their tracking system with a new arm called ‘Omicron sub-variants under monitoring’ to help identify the sub-variants which may need to be prioritised in public health warnings (Table 27.1) [27].
Molecular evolution of SARS-COV-2 with a focus on the omicron strain (phylogeny maintained by Nextstrain, enabled by data from GISAID, Image courtesy: https://nextstrain.org/ncov/open/global/6m)
With 31 or more mutations, Omicron and its sub-variants have the largest number of spike protein amino acid substitutions compared to the preceding Alpha (B.1.1.7), Beta (B.1.351), Gamma (P.1), and Delta (B.1.617.2) variants of concern [28, 29]. Approximately half of these mutations in the spike protein occur within the receptor-binding domain (RBD) which binds to the angiotensin converting enzyme 2 (ACE2) receptor on host cells in the infection process [28,29,30]. In addition, many of these mutations are known to alter the binding of antibodies produced by the existing vaccines or from previous SARS-CoV-2 infections [31,32,33].
These changes in properties have led to increasing concerns about the potential emergence of newer variants with increased virulence and capacity to escape the vaccines. However, as the virus adapts to us, we can also adapt to the virus and help to prepare ourselves for a future pandemic like this one or one that is potentially even worse. For example, since the virus has evolved to evade the existing vaccines, we must follow suit and learn to efficiently and effectively update the vaccination programmes to keep pace with these changes. In line with this, Pfizer/BioNTech has released two different bivalent vaccines which target both the original Wuhan spike protein and either the Omicron BA.1 or BA.4–5 spike proteins, and both of these were authorized for use in the European Union in September 2022 [34]. Moderna has also released a bivalent vaccine against the Wuhan and the omicron BA.1 spike proteins which was also approved in September 2022, as well as one that targets the BA.4–5 spike protein, and this is currently under evaluation.
3 Genomic Surveillance
3.1 Next-Generation Sequencing
At the beginning of the pandemic, the SARS-CoV-2 genome was sequenced using a metagenomics approach, which basically allowed determination of the full genome without any prior knowledge of the sequence. After this, it became possible to use more targeted and efficient approaches which involved the design of primers for amplification of multiple overlapping sequences to cover the whole SARS-CoV-2 genome. At this stage, the identification of specific variants required the use of various bioinformatics pipelines. New lineages are usually assigned using the Nextclade or Pangolin algorithms [35, 36].
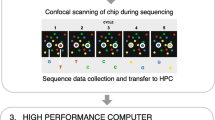
Whole genome analysis by next-generation sequencing (NGS) is currently the gold standard technique used for identification and monitoring of new SARS-CoV-2 variants and sub-variants [37]. The method essentially allows parallel sequencing of billions of DNA fragments which are combined afterwards by read assembly [38,39,40]. This method is typically performed using four basic steps (Fig. 27.2):
-
1.
Library preparation through random fragmentation of the genome and ligation of adapters
-
2.
Generation of clusters by loading the library into a flow cell for capture of the fragments on bound oligonucleotides complementary to the adapters, flowed by bridge amplification of each fragment
-
3.
Reversible terminator sequencing for detection of each nucleotide as it is incorporated into a new strand
-
4.
Data analysis and alignment for detection of single nucleotide polymorphisms, mutations, recombination events, and/or phylogenic tree construction
The basic steps of the NGS method. A double-stranded cDNA library is produced by reverse transcription of SARS-CoV-2 single-stranded RNA. Two distinct oligonucleotide adapters are ligated to the cDNA sequences. The adapters allow binding to complementary oligonucleotides linked covalently within the flow cell. Covalently attached cDNA fragments are amplified complementary to the hybridized cDNA templates. Denaturation leaves the new cDNA strands covalently bound to the flow cell and is used to generate multiple copies bridge amplification. This generates DNA clusters reading in forward and reverse directions. Removal of the reverse strands leave only forward DNA strands which are used for sequencing. Primers hybridized to the cDNA strands and fluorescently labelled terminator nucleotides are passed through the cell for sequencing. Finally, all sequencing reads are aligned and mapped to the reference genome
There are also nanopore sequencing methods that allow maximum coverage of the SARS-CoV-2 RNA genome via PCR tiling [41, 42]. A method called Midnight works through amplification of the genome in overlapping segments of 1000–1200 base pairs which makes it resistant to amplification dropouts due to mutations. The ARTIC method is similar but amplifies the genome in shorter segments of approximately 400 base pairs. This helps to improve coverage of samples that may be partly degraded.
3.2 Real-Time PCR
Once the main viral sequence has been established, there are more rapid and simpler techniques for detecting variants of concern. One of the most useful methods for this is real-time reverse transcription PCR, which has also been the mainstay in COVID-19 screening, diagnostics, and epidemiology [43,44,45]. This method works through the use of sequence-specific primers and fluorescent reporter probes in repeated cycles of cDNA amplification. The increase in the fluorescent signal with each round of amplification is then related to the amount of viral nucleic acid present in the sample. In addition, different primer/probe sets can be used to detect the presence or absence of specific variant sequences. For example, we described a method which can be used for simultaneous real-time quantitation of the United Kingdom, South Africa, and Brazil SARS-CoV-2 variants, which were prominent during the first year of the pandemic [18]. Figure 27.3a shows the location of the primer and probe sets used to detect these variants in multiplex PCR analyses. In the example shown, target failure by both primer/probe sets 1 and 2 would suggest the presence of the Alpha variant in the sample. Sole failure of primer/probe set 1 indicates potential presence of Beta and Gamma variants in the sample. Finally, target success with primer/probe sets 1–3 indicates that none of these variants are present but cannot rule out the presence of the other SARS-CoV-2 strains.
Similar approaches have been used to detect the Omicron variant. For example, Ayadi et al. described a multiplex PCR screen to distinguish the Delta variant from the Omicron BA.1 and BA.2 sub-variants [46]. This was based on the presence or absence of unique sequences in the spike protein in each of these lineages. Delta has a deletion of the glutamate and phenylalanine residues at amino acid position 156–157 (ΔEF156–157), Omicron BA.1 has a glutamate-phenylalanine-glutamate insert at position 214 (InsEPE-214) and BA.2 has a leucine-proline-proline deletion at amino acids 24–26 (ΔLPP24–26). In the scheme shown in Fig. 27.3b, Delta can be distinguished from Omicron BA.1 using forward wild type and variant primers with a common reverse primer. In a separate reaction, Omicron BA.1 can be distinguished from Omicron BA.2 using a similar strategy.
4 Machine Learning
In contrast with classical statistics, machine learning techniques employ algorithms which can learn from data to enable predictions using pattern recognition and apply this to new datasets. In case of SARS-CoV-2, this could be used to determine how specific features such as molecular biomarker patterns in the host are related to a specific disease status or outcome, as well as response to therapeutics. These relationships can be developed in a training set and then deployed to predict outcomes in new datasets. One big advantage of these approaches is that the algorithms can be retrained in an on-going manner with new input information so that it can be refined and adjusted to enhance predictive accuracy.
Deep learning methods have a complex multi-layered structure and require large datasets as input, but this allows the prediction of outcomes with high accuracy. They are generally constructed of input, hidden, and output layers, with the nodes in each layer representing the conversion of input data into a calculated output weight in connected nodes in the next layer (Fig. 27.4). The data is passed on from layer to layer by an activation function. The hidden layers carry out complex decisions and make changes to the data during this transit, and ultimately relay this information to the output layer. This final layer represents a convergence point for all data from the previous layers and a final predictive value is made [47]. The learning stage comes from a process called back propagation which involves assigning random weights to the input features and performing several more rounds of training until the most robust combination of input data with the lowest error rate arrives at the correct answer [48]. Following this stage, the model is tested to determine generalizability to new datasets. For this, the study sample can be partitioned into several folds and all but one of these is used in the same iterative way as above to train the model. Next, the model is applied repeatedly to each fold that was not included to assess overall performance. Higher generalizability can be achieved by applying the model to a completely new validation dataset [49, 50].
Machine learning approaches have been used recently to identify robust molecular signatures comprised of transcriptomic [51], proteomic [51, 52], metabolomic [51], and laboratory blood test results [53], for prediction of COVID-19 disease severity and outcomes with excellent sensitivity and specificity scores. Along the same lines, Sardar et al. developed an artificial intelligence algorithm based on a combination of proteomic and clinical biomarkers which had a good overall accuracy for prediction of survival outcomes in COVID-19 patents [54]. Also, another study used machine learning to construct an algorithm from metabolomics data collected from COVID-19 patients at different time points during the disease course, which revealed that a model developed during the earliest phase of the disease was successful in determining disease severity in the later stages [55].
Machine learning algorithms have also been used to identify mutations across the SARS-CoV-2 variants of concern associated with higher infectivity [56, 57] and escape from neutralizing antibodies or the antibodies produced by some of the vaccines [58]. Thus, these approaches could be used to assess current and future variants which would help healthcare workers to manage the disease more effectively.
5 Lab-on-a-Chip Devices
Although miniaturization of the working components is the key to POC devices, this can also cause a number of problems such as issues arising from use outside a designated laboratory and operation by untrained technicians. However, a number of commercialized products have emerged which go some way to overcoming some of these issues. One of these was aimed at detection of the Ebola virus during the 2014–2016 outbreak in West Africa [59, 60]. After the World Health Organization (WHO) declared this outbreak a public health emergency of international concern, an emergency use scheme was put in place to drive research and development of new medical devices for use in public health emergencies [61]. One early success was the GeneXpert Ebola PCR assay which took approximately 5 months to develop and deploy [62]. This was an automated assay which required application of the patient sample into a well on a cartridge, inserting this into a compact reader and retrieving the result within 2 h. Another early example was the FilmArray BioThreat-E device which was also based on PCR and had a sample application to readout time of 1 h [63].
The standard lateral flow device, as applied by the National Health Service in the United Kingdom [64, 65], consists of a 7 × 2 cm cassette comprising a sample well and an enclosed membrane containing: (1) SARS-CoV-2 nucleocapsid protein antibodies conjugated with colour particles; (2) SARS-CoV-2 nucleocapsid protein antibodies bound on a test (T) line; and (3) secondary antibodies which target the primary antibodies bound on a control (C) line (Fig. 27.5). If virus is present in the sample, this is bound by the detector antibody. The virus-detector antibody complex is carried along the membrane by capillary action to the T line, where it is captured by nucleocapsid protein antibody. Unbound detector antibody also binds to the secondary antibody on the C line. This leads to generation of a colour on both the T and C lines as an indicator of a positive result. If the virus is not present in the sample, the detector antibodies will flow past the T line without binding to be captured by the secondary antibodies on the C line. This results in formation of a coloured line in the C region only as an indication of a negative result.
In 2012, Schumacher et al. reported on the development of a marketable, multi-parameter LOC system that could be used for POC diagnostics [66]. The system consisted of a microfluidic credit card-sized cartridge containing reagent reservoirs, integrated pumping and temperature control mechanisms, and an optical transducer. After the sample(s) are applied to the appropriate wells, the cartridge is inserted into a base unit that contains the essential controlling electronics and an optical system with a touch screen for user-friendly control of the assay and analysis of the results.
5.1 LOC Devices for Diagnosis of COVID-19
Early in the pandemic, Cojocaru reported on the development of microchip real-time PCR assay for detection of SARS-CoV-2 from nasopharyngeal swab samples [67]. This chip contained the primer/probe sets for the SARS-CoV-2 nucleocapsid protein gene in a 1.2-μL reaction volume. They validated the assay using reference and clinical samples and found a detection limit of one RNA copy per reaction. Cui et al. presented a proof-of-concept study of a microfluidic microwave sensing method for diagnosis of COVID-19 [68]. The method employs an immobilized antibody on the sensor to immunoprecipitate the virus which results in a detectable resonance frequency shift. The device showed 4000 copies/mL sensitivity for SARS-CoV-2 virus, and this could be distinguished from the CD4 antigen, MERS-CoV, and CoV-HKU1. Another PCR-based LOC device for COVID-19 detection and quantitation was described by Yin et al. [69]. This was a droplet microfluidic chip capable of multiplex analysis of nine samples with a detection limit of 10 nucleic acid copies per test and a total run time of 15 min. Zai et al. described development of a gravity-driven LOC device for viral nucleic acid diagnosis with extraction-free amplification [70]. They validated this by successful detection of SARS-CoV-2, influenza A/B, and papillomavirus 16/18 viruses. Parker et al. described the use of an optofluidic lab-in-a-fibre device which combines droplet microfluidics with laser-induced fluorescence detection of reverse transcription loop-mediated isothermal amplification (RT-LAMP) products for SARS-CoV-2 diagnostics [71]. The device offers advantages over other LOC systems as fibre technology is ideal for enhanced optical coupling. For monitoring and surveillance purposes, Donia et al. described the use of a LAMP-based LOC device that they used in the detection of SARS-CoV-2 in wastewater samples in COVID-19 hotspots [72].
Another study described a nanoplasmonic LOC device for rapid and quantitative PCR diagnostics [73]. The device consisted of a plasmofluidic chip with glass nanopillar arrays with gold islands, gas-permeable microfluidic channels, reaction arrays, vacuum cell, and a vapour barrier. This allowed sample loading in less than 3 min, and PCR results for the SARS-CoV-2 envelope protein in approximately 5 min. Stambaugh et al. described an LOC device comprising a bead-based solid extraction with sandwich antibody configuration and a fluorescent reporter probe, which they validated in detection of both SARS-CoV-2 and influenza A viruses from nasopharyngeal swab samples [74]. The multiplexing capability was conferred by multispot excitation on a multimode interference waveguide platform, with a sensitivity of 30 ng/mL. Another variation on the LOC concept was described by Kim et al. to enable detection of antigens at low concentrations [75]. This leveraged a rotationally focused flow approach for enhanced sensitivity by wavelength shift of optical sensors upon antigen detection in the module. This worked by addition of a low-density fluid to focus the target fluid into a microchannel and yielded a sensitivity of 0.19 fM, which is more sensitive than single flow methods.
5.2 LOC Devices for Detection of SARS-CoV-2 Antibodies
To aid in determinations of immune protection against new SARS-COV-2 variants, Rajsri et al. described a rapid quantitative POC assay in an injection-moulded polymethyl methacrylate cassette capable of quantifying circulating SARS-CoV-2 antibodies in less than 15 min [76]. Another study reported on the development of a 3D-printed LOC device with multiplexed electrochemical outputs which allows simultaneous detection of SARS-CoV-2 RNA and SARS-CoV-2 immunoglobulins in saliva in less than 2 h [77]. Thus, this could be used for both SARS-CoV-2 diagnosis and for monitoring antibody responses in immunized or infected persons. Along the same lines, Mandal et al. constructed an ultrasonic-guided wave sensor designed in a multi-threaded comb shape with cantilever beams for multiplexing capability [78]. This showed selectivity and sensitivity for detection of SARS-COV-2 antibodies and could be easily adapted for detection or other antibodies or antigens, simultaneously.
5.3 SARS-CoV-2 Disease-associated Effects
We recently described the use of an antibody microarray in combination with an LOC system to automate and increase the speed of multiplex immunoassays for detection of the SARS-CoV-2 cytokine storm effect [79]. For this, we carried out a fully automated LOC immunoassay for detection of C-reactive protein (CRP) in blood samples with pumping of all of the usual assay steps within the cartridge and data analysis using the base unit. The total assay time after application of the sample was 15 min. This is important as most existing multiplex immunoassay protocols are impractical in routine laboratory and clinical tests, as these typically involve long experimental times with the need for sophisticated laboratory equipment and procedures, as well as trained operators. LOC systems have no such limitations as their user-friendly automated platforms incorporate many of the above steps.
Other LOC devices have also been developed to detect changes in biomarkers-associated COVID-19 disease effects. Recktenwald et al. developed a LOC device called Erysense which can evaluate red blood cell flow properties in samples less than 1 μL [80]. Haghayegh et al. described development of a self-powered automated microfluidic chip which included controls for sample delivery and an electrochemical immune-based biosensor, which allowed detection of the SARS-CoV-2 nucleocapsid protein in phosphate buffer within 15 min [81]. The linear detection range was 10–1000 pg/mL with a limit of detection of 3.1 pg/mL. McRae et al. described the use of ‘smart diagnostics’ which is powered by the combination of miniaturised electronics, cloud-based computing, and machine learning approaches in the identification and validation of disease signatures [82]. This method also includes deep learning–based inference and clinical decision support, with reporting and integration with healthcare records. In line with this, an Internet of Diseases (IOD) platform has been developed which links an LOC device for SARS-CoV-2 diagnosis using saliva samples to diagnostic data in a cloud-based system for disease control and prevention in a regional manor [83]. Choi et al. demonstrated a similar multiplexed LOC PCR device with a linked smartphone application for automatic processing and cloud storage [84]. Using this, they were able to carry out analysis of nine RNA viruses simultaneously, which included the OC43, 229E, and NL63 human coronaviruses, with high linearity and sensitivity. Also, Heithoff et al. demonstrated a smartphone-based LAMP assay called smaRT-LAMP for detection of SARS-CoV-2 infection, and this showed high concordance with standard RT-PCR tests [85].
5.4 LOC Devices for Detection of SARS-CoV-2 Variants
Based on their capability of identifying specific sequences, PCR-based LOC devices can be used for identification of SARS-CoV-2 variants. Applying this idea in combination with the system described by Schumacher et al. [66], we recently described the development of a microarray LOC device which could be used for diagnosis of COVID-19 infections or for sub-typing of SARS-CoV-2 variants (Fig. 27.6) [86]. We demonstrated this principle through detection of signal nucleotide polymorphisms in methicillin-resistant Staphylococcus aureus (MRSA) using the LOC system. Following a PCR stage of 60 min, this resulted in hybridization, washing and readout times of less than 15 min. For analyses of the SARS-CoV-2 virus, we suggest use of inhibitor-tolerant PCR mix such as that developed by Meridian Bioscience to bypass the RNA extraction step. This step is normally rate limiting and may lead to poor recovery and performance of the assay [87]. Kumar et al. described the development of an FnCas9-based CRISPR LOC device for detection of SARS-CoV-2 infections as well as the presence of the N501Y mutation present in multiple SARS-CoV-2 variants [88]. Another report described the development of a microfluidic device capable of discriminating the SARS-CoV-2 Alpha variant from both the SARS-CoV-2 original isolate and negative controls in saliva samples [89]. The assay was based on RT-LAMP PCR in the detection of spike gene target failure as a way of distinguishing the SARS-CoV-2 Alpha variant from the original SARS-CoV-2 strain at least 10 copies/μL within 30 min. They validated the performance of the test by analysis of 38 saliva specimens, which yielded a sensitivity greater than 90% and a specificity of 100%. Another study described the development of a similar device that was used successfully to detect SARS-CoV-2 in clinical samples [90].
6 Conclusions and Future Perspectives
In this chapter, we described attempts to control the COVID-19 pandemic through application of surveillance methods aimed at detection of new SARS-CoV-2 variants of concern and prediction of how specific mutational changes alter the transmissibility, virulence, and immune evasion capabilities of the virus. Other steps that should be taken to prepare us for the next pandemic should include the surveillance and early detection of SARS-CoV-2 and other coronavirus strains and variants in domesticated and wild animals, considering the zoonotic nature of this virus [91]. Although detection of new viral sequences requires whole genome sequencing, once this has been achieved, more targeted methods can be applied for monitoring variants such as real-time PCR. In addition, omic techniques such as multiplex cytokine screening should be used to determine the effects of new viral strains on the host. This would enable development of biomarker testing for prediction of disease severity and outcomes to guide the most appropriate treatment course. Future efforts should also be directed towards translating these methods onto user-friendly platforms, and even handheld devices enabled by smart technologies, for POC testing so that therapeutics could be administered in personalized medicine approach. With this infrastructure in place, we should be able to curtail any future catastrophic waves caused by emergence of new SARS-CoV-2 variants and other deadly zoonotic viruses.
References
Worldometer; COVID-19 CORONAVIRUS PANDEMIC. https://www.worldometers.info/coronavirus/. Accessed December 1, 2022
Johns Hopkins University; Coronavirus Dasboard. https://coronavirus.jhu.edu/map.html. Accessed December 1 13, 2022
Center for Infectious Disease Research and Policy; University of Minnesota, USA; Estimate: Less than half the world has had COVID-19. https://www.cidrap.umn.edu/news-perspective/2022/04/estimate-less-half-world-has-had-covid-19. Accessed October 13, 2022
Cakir B (2020) COVID-19 in Turkey: Lessons Learned. J Epidemiol Glob Health 10(2):115–117
Minni A, Ralli M, Candelori F, et al (2021) Lessons learned from COVID-19 pandemic in Italy – A commentary. Bosn J Basic Med Sci 21(1):117–119
Pletcher MJ, Olgin JE, Peyser ND, et al (2021) Factors Associated With Access to and Timing of Coronavirus Testing Among US Adults After Onset of Febrile Illness. JAMA Netw Open 4(5):e218500. https://doi.org/10.1001/jamanetworkopen.2021.8500
Chen YJ, Jian WH, Liang ZY, et al (2021) Earlier diagnosis improves COVID-19 prognosis: a nationwide retrospective cohort analysis. Ann Transl Med 9(11):941. https://doi.org/10.21037/atm-20-7210
Herr AE, Hatch AV, Giannobile WV, et al (2007) Integrated microfluidic platform for oral diagnostics. Ann N Y Acad Sci 1098:362–374
Myers FB, Lee LP (2008) Innovations in optical microfluidic technologies for point-of-care diagnostics. Lab Chip 8(12):2015–2031
Park S, Zhang Y, Lin S, et al (2011) Advances in microfluidic PCR for point-of-care infectious disease diagnostics. Biotechnol Adv 29(6):830–839
Christodoulides N, Pierre FN, Sanchez X, et al (2012) Programmable bio-nanochip technology for the diagnosis of cardiovascular disease at the point-of-care. Methodist Debakey Cardiovasc J 8(1):6–12
Harpaldas H, Arumugam S, Campillo Rodriguez C, et al (2021) Point-of-care diagnostics: recent developments in a pandemic age. Lab Chip 21(23):4517–4548
Zhu P, Qian M, Wu Q, Liu X (2022) Challenges Faced in Large-Scale Nucleic Acid Testing during the Sudden Outbreak of the B.1.617.2 (Delta). Int J Environ Res Public Health 19(3):1573. https://doi.org/10.3390/ijerph19031573
Frickhofen N, Young NS (1991) A rapid method of sample preparation for detection of DNA viruses in human serum by polymerase chain reaction. J Viral Methods 35(1):65–72.
Ochert AS, Boulter AW, Birnbaum W, et al (1994) Inhibitory effect of salivary fluids on PCR: potency and removal. Genome Res 3(6):365–368
Morais OM, Azevedo Alves MR, Fernandes PADC (2022) Impact of Thermal Pretreatment of Saliva on the RT-PCR Detection of SARS-CoV-2. Adv Virol 2022:7442907. https://doi.org/10.1155/2022/7442907
Hawkins SFC, Guest PC (2022) Multiplex Quantitative Polymerase Chain Reaction Diagnostic Test for SARS-CoV-2 and Influenza A/B Viruses. Methods Mol Biol 2511:53–65
Hawkins SFC, Guest PC (2022) Multiplex Quantitative Polymerase Chain Reaction Test to Identify SARS-CoV-2 Variants. Methods Mol Biol 2511:67–78
Daum LT, Canas LC, Arulanandam BP, et al (2007) Real-time RT-PCR assays for type and subtype detection of influenza A and B viruses. Influenza Other Respir Viruses 1(4):167–175
Shisong F, Jianxiong L, Xiaowen C, et al (2011) Simultaneous detection of influenza virus type B and influenza A virus subtypes H1N1, H3N2, and H5N1 using multiplex real-time RT-PCR. Appl Microbiol Biotechnol 90(4):1463–1470
Hernandez MM, Banu R, Shrestha P, et al (2022) A Robust, Highly Multiplexed Mass Spectrometry Assay to Identify SARS-CoV-2 Variants. Microbiol Spectr; Sep 7:e0173622. https://doi.org/10.1128/spectrum.01736-22
Imaizumi Y, Ishige T, Fujikawa T, et al (2022) Development of multiplex S-gene-targeted RT-PCR for rapid identification of SARS-CoV-2 variants by extended S-gene target failure. Clin Chim Acta 536:6–11
Callaway E (2021) Heavily mutated Omicron variant puts scientists on alert. Nature 600(7887):21. https://doi.org/10.1038/d41586-021-03552-w
World Health Organization; Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern. Accessed October 24, 2022
The Guardian (Dec 18, 2021); WHO says Omicron in 89 countries and spreading rapidly. https://www.theguardian.com/world/2021/dec/18/who-says-omicron-in-89-countries-and-spreading-rapidly
Rahman S, Hossain MJ, Nahar Z, et al (2022) Emerging SARS-CoV-2 Variants and Subvariants: Challenges and Opportunities in the Context of COVID-19 Pandemic. Environ Health Insights 16:11786302221129396. https://doi.org/10.1177/11786302221129396
World Health Organization; Omicron subvariants under monitoring (as of 12 October 2022). https://www.who.int/activities/tracking-SARS-CoV-2-variants. Accessed October 29, 2022
Ou J, Lan W, Wu X, et al (2022) Tracking SARS-CoV-2 Omicron diverse spike gene mutations identifies multiple inter-variant recombination events. Signal Transduct Target Ther 7(1):138. https://doi.org/10.1038/s41392-022-00992-2
Kumar S, Karuppanan K, Subramaniam G (2022) Omicron (BA.1) and sub-variants (BA.1.1, BA.2, and BA.3) of SARS-CoV-2 spike infectivity and pathogenicity: A comparative sequence and structural-based computational assessment. J Med Virol 94(10):4780–4791
Mannar D, Saville JW, Zhu X, et al (2022) SARS-CoV-2 Omicron variant: Antibody evasion and cryo-EM structure of spike protein-ACE2 complex. Science 375(6582):760–764
Li Q, Nie J, Wu J, et al (2021) SARS-CoV-2 501Y.V2 variants lack higher infectivity but do have immune escape. Cell 184(9):2362–2371.e9. https://doi.org/10.1016/j.cell.2021.02.042
Garcia-Beltran WF, Lam EC, St. Dennis K, et al (2021) Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell 184(9):2372–2383.e9. https://doi.org/10.1016/j.cell.2021.03.013
Wang Y, Zhang L, Li Q, et al (2022) The significant immune escape of pseudotyped SARS-CoV-2 variant Omicron. Emerg Microbes Infect 11(1):1–5
European Medicines Agency; Adapted COVID-19 vaccines. https://www.ema.europa.eu/en/human-regulatory/overview/public-health-threats/coronavirus-disease-covid-19/treatments-vaccines/vaccines-covid-19/covid-19-vaccines-authorised. Accessed October 20, 2022
Nextclade; Clade assignment, mutation calling, and sequence quality checks. https://clades.nextstrain.org/. Accessed November 19, 2022
GISAID; Clade and lineage nomenclature, March 2, 2021; Clade and lineage nomenclature aids in genomic epidemiology studies of active hCoV-19 viruses https://gisaid.org/resources/statements-clarifications/clade-and-lineage-nomenclature-aids-in-genomic-epidemiology-of-active-hcov-19-viruses/. Accessed November 19, 2022
World Health Organization; Genomic sequencing of SARS-CoV-2: a guide to implementation for maximum impact on public health. https://www.who.int/publications/i/item/9789240018440. Accessed October 24, 2022
Fedurco M, Romieu A, Williams S, et al (2006) BTA, a novel reagent for DNA attachment on glass and efficient generation of solid-phase amplified DNA colonies. Nucleic Acids Res 34(3):e22. https://doi.org/10.1093/nar/gnj023
Turcatti G, Romieu A, Fedurco M, et al (2008) A new class of cleavable fluorescent nucleotides: synthesis and optimization as reversible terminators for DNA sequencing by synthesis. Nucleic Acids Res 36(4):e25. https://doi.org/10.1093/nar/gkn021
Voelkerding KV, Dames SA, Durtschi J (2009) Next-generation sequencing: from basic research to diagnostics. ClinChem 55(4):641–658
Pembaur A, Sallard E, Weil PP, et al (2021) Simplified Point-of-Care Full SARS-CoV-2 Genome Sequencing Using Nanopore Technology. Microorganisms 9(12):2598. https://doi.org/10.3390/microorganisms9122598
Tyson JR, James P, Stoddart D, et al (2020) Improvements to the ARTIC multiplex PCR method for SARS-CoV-2 genome sequencing using nanopore. bioRxiv. 2020.09.04.283077. https://doi.org/10.1101/2020.09.04.283077
Jain A, Rophina M, Mahajan S, et al (2021) Analysis of the potential impact of genomic variants in global SARS-CoV-2 genomes on molecular diagnostic assays. Int J Infect Dis 102:460–462
Joyce N, Seim L, Smerina M (2021) The Dynamics of SARS-CoV-2 (RT-PCR) Testing. Case Rep Med 2021:6688303. https://doi.org/10.1155/2021/6688303
Tombuloglu H, Sabit H, Al-Khallaf H, et al (2022) Multiplex real-time RT-PCR method for the diagnosis of SARS-CoV-2 by targeting viral N, RdRP and human RP genes. Sci Rep 12(1):2853. https://doi.org/10.1038/s41598-022-06977-z
Ayadi W, Taktak A, Gargouri S, et al (2022) Development of a simple genotyping method based on indel mutations to rapidly screen SARS-CoV-2 circulating variants: Delta, Omicron BA.1 and BA.2. J Virol Methods 307:114570. https://doi.org/10.1016/j.jviromet.2022.114570
Ghods A, Cook DJ (2021) A Survey of Deep Network Techniques All Classifiers Can Adopt. Data Min Knowl Discov 35(1):46–87
Yu CS, Chang SS, Chang TH, et al (2021) A COVID-19 Pandemic Artificial Intelligence-Based System With Deep Learning Forecasting and Automatic Statistical Data Acquisition: Development and Implementation Study. J Med Internet Res 23(5):e27806. https://doi.org/10.2196/27806
Cortes C, Vapnik V (1995) Support-vector networks. Machine learning 20:273–297
Ho TK (1995) Random Decision Forest. Proceedings of the 3rd International Conference on Document Analysis and Recognition, Montreal, 14–16 August 1995, 278–282
Papoutsoglou G, Karaglani M, Lagani V, et al (2021) Automated machine learning optimizes and accelerates predictive modeling from COVID-19 high throughput datasets. Sci Rep 11(1):15107. https://doi.org/10.1038/s41598-021-94501-0
Lazari LC, Ghilardi FR, Rosa-Fernandes L, et al (2021) Prognostic accuracy of MALDI-TOF mass spectrometric analysis of plasma in COVID-19. Life Sci Alliance 4(8):e202000946. https://doi.org/10.26508/lsa.202000946
Krysko O, Kondakova E, Vershinina O, et al (2021) Artificial Intelligence Predicts Severity of COVID-19 Based on Correlation of Exaggerated Monocyte Activation, Excessive Organ Damage and Hyperinflammatory Syndrome: A Prospective Clinical Study. Front Immunol 12:715072. https://doi.org/10.3389/fimmu.2021.715072
Karami H, Derakhshani A, Ghasemigol M, et al (2021) Weighted Gene Co-Expression Network Analysis Combined with Machine Learning Validation to Identify Key Modules and Hub Genes Associated with SARS-CoV-2 Infection. J Clin Med 10(16):3567. https://doi.org/10.3390/jcm10163567
Sindelar M, Stancliffe E, Schwaiger-Haber M, et al (2021) Longitudinal metabolomics of human plasma reveals prognostic markers of COVID-19 disease severity. Cell Rep Med 2(8):100369. https://doi.org/10.1016/j.xcrm.2021.100369
Huang F, Chen L, Guo W, et al (2022) Identifying COVID-19 Severity-Related SARS-CoV-2 Mutation Using a Machine Learning Method. Life (Basel) 12(6):806. https://doi.org/10.3390/life12060806
Zhao LP, Lybrand TP, Gilbert PB, et al (2022) Rapidly identifying new coronavirus mutations of potential concern in the Omicron variant using an unsupervised learning strategy. Sci Rep 12(1):19089. https://doi.org/10.1038/s41598-022-23342-2
Taft JM, Weber CR, Gao B, et al (2022) Deep mutational learning predicts ACE2 binding and antibody escape to combinatorial mutations in the SARS-CoV-2 receptor-binding domain. Cell 185(21):4008–4022.e14
Malvy D, McElroy AK, de Clerck H, et al (2019) Ebola virus disease. Lancet 393(10174):936–948
Park C (2022) Lessons learned from the World Health Organization’s late initial response to the 2014–2016 Ebola outbreak in West Africa. J Public Health Afr 13(1):1254. https://doi.org/10.4081/jphia.2022.1254
World Health Organization (2015) Norms and standards: assessing new medical products in health emergencies: the EUAL procedures. https://apps.who.int/iris/bitstream/handle/10665/331062/DI293-305-323-eng.pdf. Accessed October 13, 2022
Semper AE, Broadhurst MJ, Richards J, et al (2016) Performance of the GeneXpert Ebola Assay for Diagnosis of Ebola Virus Disease in Sierra Leone: A Field Evaluation Study. PLoS Med 13(3):e1001980. https://doi.org/10.1371/journal.pmed.1001980
Gay-Andrieu F, Magassouba N, Picot V, et al (2017) Clinical evaluation of the BioFire FilmArray((R)) BioThreat-E test for the diagnosis of Ebola virus disease in Guinea. J Clin Virol 92:20. https://doi.org/10.1016/j.jcv.2017.04.015
Peto T; UK COVID-19 Lateral Flow Oversight Team (2021) COVID-19: Rapid antigen detection for SARS-CoV-2 by lateral flow assay: A national systematic evaluation of sensitivity and specificity for mass-testing. EClinicalMedicine 36:100924. https://doi.org/10.1016/j.eclinm.2021.100924
Guest PC, Rahmoune H (2022) COVID-19 Detection Using the NHS Lateral Flow Test Kit. Methods Mol Biol 2511:297–305
Schumacher S, Nestler J, Otto T, Wegener M, et al (2012) Highly-integrated lab-on-chip system for point-of-care multiparameter analysis. Lab Chip 12(3):464–473
Cojocaru R, Yaseen I, Unrau PJ, et al (2021) Microchip RT-PCR Detection of Nasopharyngeal SARS-CoV-2 Samples. J Mol Diagn 23(6):683–690
Cui W, Zhao P, Wang J, et al (2022) Reagent free detection of SARS-CoV-2 using an antibody-based microwave sensor in a microfluidic platform. Lab Chip 22(12):2307–2314
Yin H, Tong Z, Shen C, et al (2022) Micro-PCR chip-based multifunctional ultrafast SARS-CoV-2 detection platform. Lab Chip 22(14):2671–2681
Zai Y, Min C, Wang Z, et al (2022) A sample-to-answer, quantitative real-time PCR system with low-cost, gravity-driven microfluidic cartridge for rapid detection of SARS-CoV-2, influenza A/B, and human papillomavirus 16/18. Lab Chip 22(18):3436–3452
Parker HE, Sengupta S, Harish AV, et al (2022) A Lab-in-a-Fiber optofluidic device using droplet microfluidics and laser-induced fluorescence for virus detection. Sci Rep 12(1):3539. https://doi.org/10.1038/s41598-022-07306-0
Donia A, Furqan Shahid M, Hassan SU, et al (2022) Integration of RT-LAMP and Microfluidic Technology for Detection of SARS-CoV-2 in Wastewater as an Advanced Point-of-Care Platform. Food Environ Virol; May 4:1–10. https://doi.org/10.1007/s12560-022-09522-3
Kang BH, Lee Y, Yu ES, et al (2021) Ultrafast and Real-Time Nanoplasmonic On-Chip Polymerase Chain Reaction for Rapid and Quantitative Molecular Diagnostics. ACS Nano 15(6):10194–10202
Stambaugh A, Parks JW, Stott MA, et al (2021) Optofluidic multiplex detection of single SARS-CoV-2 and influenza A antigens using a novel bright fluorescent probe assay. Proc Natl Acad Sci USA 118(20):e2103480118. https://doi.org/10.1073/pnas.2103480118
Kim N, Han K, Su PC, et al (2021) A rotationally focused flow (RFF) microfluidic biosensor by density difference for early-stage detectable diagnosis. Sci Rep 11(1):9277. https://doi.org/10.1038/s41598-021-88647-0
Rajsri KS, McRae MP, Simmons GW, et al (2022) A Rapid and Sensitive Microfluidics-Based Tool for Seroprevalence Immunity Assessment of COVID-19 and Vaccination-Induced Humoral Antibody Response at the Point of Care. Biosensors (Basel) 12(8):621. https://doi.org/10.3390/bios12080621
Najjar D, Rainbow J, Sharma Timilsina S, et al (2022) A lab-on-a-chip for the concurrent electrochemical detection of SARS-CoV-2 RNA and anti-SARS-CoV-2 antibodies in saliva and plasma. Nat Biomed Eng 6(8):968–978
Mandal D, Indaleeb MM, Younan A, Banerjee S (2022) Piezoelectric point-of-care biosensor for the detection of SARS-COV-2 (COVID-19) antibodies. Sens Biosensing Res 37:100510. https://doi.org/10.1016/j.sbsr.2022.100510
Peter H, Mattig E, Guest PC, Bier FF (2022) Lab-on-a-Chip Immunoassay for Prediction of Severe COVID-19 Disease. Methods Mol Biol 2511:235–244
Recktenwald SM, Lopes MGM, Peter S, et al (2022) Erysense, a Lab-on-a-Chip-Based Point-of-Care Device to Evaluate Red Blood Cell Flow Properties With Multiple Clinical Applications. Front Physiol 13:884690. https://doi.org/10.3389/fphys.2022.884690
Haghayegh F, Salahandish R, Zare A, et al (2021) Immuno-biosensor on a chip: a self-powered microfluidic-based electrochemical biosensing platform for point-of-care quantification of proteins. Lab Chip 22(1):108–120
McRae MP, Rajsri KS, Alcorn TM, McDevitt JT (2022) Smart Diagnostics: Combining Artificial Intelligence and In Vitro Diagnostics. Sensors (Basel) 22(17):6355. https://doi.org/10.3390/s22176355
Bhuiyan NH, Uddin MJ, Lee J, et al (2022) An Internet-of-Disease System for COVID-19 Testing Using Saliva by an AI-Controlled Microfluidic ELISA Device. Adv Mater Technol 7(9):2101690. https://doi.org/10.1002/admt.202101690
Choi JW, Seo WH, Lee YS, et al (2022) Development of an IoT-integrated multiplexed digital PCR system for quantitative detection of infectious diseases. Lab Chip 22(20):3933–3941
Heithoff DM, Barnes L 5th, Mahan SP, et al (2022) Assessment of a Smartphone-Based Loop-Mediated Isothermal Amplification Assay for Detection of SARS-CoV-2 and Influenza Viruses. JAMA Netw Open 5(1):e2145669. https://doi.org/10.1001/jamanetworkopen.2021.45669
Mattig E, Guest PC, Peter H (2022) A Rapid User-Friendly Lab-on-a-Chip Microarray Platform for Detection of SARS-CoV-2 Variants. Methods Mol Biol 2511:117–131
Meridian Bioscience; Inhibitor-Tolerant RT-qPCR Mix Product Handling Guide. https://www.meridianbioscience.com/uploads/MDX016-Inhibitor-Tolerant-RT-qPCR-Mix-Product-Handling-Guide-V1.pdf. Accessed October 14, 2022
Kumar M, Gulati S, Ansari AH, et al (2021) FnCas9-based CRISPR diagnostic for rapid and accurate detection of major SARS-CoV-2 variants on a paper strip. Elife 10:e67130. https://doi.org/10.7554/eLife.67130
Lim J, Stavins R, Kindratenko V, et al (2022) Microfluidic point-of-care device for detection of early strains and B.1.1.7 variant of SARS-CoV-2 virus. Lab Chip 22(7):1297–1309
Torezin Mendonça G, Cassaboni Stracke M, de Oliveira Coelho B, et al (2022) A new RT-LAMP-on-a-Chip Instrument for SARS-CoV-2 diagnostics. Microchem J 180:107600. https://doi.org/10.1016/j.microc.2022.107600
Saied AA, Metwally AA (2022) SARS-CoV-2 variants of concerns in animals: An unmonitored rising health threat. Virusdisease 33(4):466–476
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Guest, P.C., Hawkins, S.F.C., Rahmoune, H. (2023). Rapid Detection of SARS-CoV-2 Variants of Concern by Genomic Surveillance Techniques. In: Guest , P.C. (eds) Application of Omic Techniques to Identify New Biomarkers and Drug Targets for COVID-19. Advances in Experimental Medicine and Biology(), vol 1412. Springer, Cham. https://doi.org/10.1007/978-3-031-28012-2_27
Download citation
DOI: https://doi.org/10.1007/978-3-031-28012-2_27
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-28011-5
Online ISBN: 978-3-031-28012-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)