Abstract
Pest diseases in crop soils are likely to increase under the warming effects of climate change, calling for advanced practices to control pest and maintain food production. Biochar application, for example, is improving soil health by supplying nutrients, removing toxic compounds, increasing the population of mycorrhizal fungi, nutrient retention and influencing beneficial microorganisms which are known to enhance plant growth and resist phytopathogens. Few reports show that biochar protects to plants against soil borne diseases via induced systemic resistance and systemic acquired resistance. Here we review biochar uses with focus on properties, effects on plant-soil microflora interactions, plant health, plant growth improvement, and control of soilborne diseases.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Two most significant and tough challenges being faced by our society, are to feed the continuously rising populace and to evade the change of climate (Fahad et al. 2019, 2020, 2021, 2022). To diminish the effect of utilization of more land on various ecosystem amenities, the scientists must enormously focus on the eco-friendly and good approaches which are sustainable for our agriculture (Kolton et al. 2017; Shah et al. 2022; Al-Zahrani et al. 2022). The sustainability of agriculture is at stake due to various factors which are deteriorating the soil properties and soil health including soilborne diseases (Shaaban et al. 2018; Toju et al. 2018; Naz et al. 2021a; Bamagoos et al. 2021).
Healthy soil can efficiently improve the plant health and production while soilborne diseases are majorly affecting the soil health as well as quality production of food and feed (Yang et al. 2019; Riaz et al. 2021). The repeated cultivation of the same crop is known to deteriorate the soil physico-chemical properties and its nutrient status which can further worsen the soil for succeeding crop productivity and cause soil sickness (Wang et al. 2019; Liu et al. 2019; Zia et al. 2021). Seedling blight, damping-off, seed and root rots are the most communal soilborne diseases in plants which are particularly being instigated by Rhizoctonia solani and Pythium spp., causing significant crop yield losses for instance eggplant, cucumber, melon, pepper, corn, potato and tomato (Fischer and Glaser 2012; Nikraftar et al. 2013; Jaiswal et al. 2014).
Farmers usually rely on the use of certain chemicals and fungicides to eliminate the pathogens; however, these chemicals and fungicides may damage the plants and the beneficial microflora, besides being expensive (Naz et al. 2018; Jaiswal et al. 2019). Thus, control of soil borne pathogens is the essential step for preserving soil health and may be supportive for sustainable soil management to strengthen the agriculture (Xiang et al. 2019).
Consequently, there is a need to find the non-chemical, alternative approaches to reduce the incidence of soilborne diseases. One developing method that holds potential for eradicating the pathogens causing soilborne diseases is the addition of biochar which has fascinated extensive consideration owing to its key role in improving soil (Jaiswal et al. 2019). Biochar, the compact co-product of biomass pyrolysis, has increased significant research and profitable interest over the past time for a variety of reasons comprising increased soil fertility status (Frenkel et al. 2017; Ibad et al. 2022; Irfan et al. 2021; Khadim et al. 2021a, b; Khan et al. 2021; Khatun et al. 2021; Muhammad et al. 2022; Subhan et al. 2020; Tariq et al. 2018; Wiqar et al. 2022; Wu et al. 2020; Wu et al. 2019; Xue et al. 2022), pollutant fixation (Ho et al. 2017), improved plant efficiency (Ahmed et al. 2017). Soil amendments with biochar applications has been reported to increase tomato, maize, pepper, soybean and wheat plant growth and yield attributes (Graber et al. 2010; Islami et al. 2011; Alburquerque et al. 2013; Egamberdieva et al. 2016).
Moreover, the exogenous treatments of plants with biochar have also reduced the incidence of soilborne diseases by inducing ISR against fungal phytopathogens (Elad et al. 2011) including Botrytis cinerea (Mehari et al. 2015), Fusarium oxysporum in tomato (Akhter et al. 2016) R. solani in cucumber (Jaiswal et al. 2014). Hence, the biochar application has mitigated the harmful effects of soil reaction by adjusting the soil microflora (Wang et al. 2020) and have revealed the potential proficiency to subdue the soilborne plant diseases (Beesley et al. 2011). Meeting the twin challenges of rising food call and climate alteration, it is imperious to take environmental performs for maintainable farming.
Various studies have described that biochar applications also have impending role for modifying climate change by lasting reclamation of carbon and inducing greenhouse gas changes in soil, and biochar treatments has been recommended as an active countermeasure to lessen emissions of nitrous oxide and methane (Kolton et al. 2017). The biochar treatments have increased leaching of nutrients and supplementation for better plant growth (Xiang et al. 2019) and biochar adjusted soils had higher cation exchange, water holding, pH, larger surface area and lower soil bulk density, compared with the unamended soils (Enders et al. 2012).
2 Biochar to Improve Soil Health
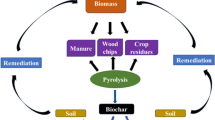
Growth and expansion into biochar, the addition of charcoal to soil, has been growing significantly over the last few decades. As a consequence of rising alarm over worldwide climate variation caused by synthetic, anthropogenic greenhouse gas releases, there is a global effort to move from a petro economy powered by fossil carbon to a budget driven by renewable energy assets, containing biomass. Biochar is a dense by-product of biomass formed by pyrolysis or by higher temperatures about 250 °C, under limited supply of or in the whole absenteeism of air (Mao et al. 2012). Being an exothermic procedure, pyrolysis of biomass gathers more energy than is keen in the heating procedure (Murakami et al. 2007). The gaseous and liquid co-products are used for energy or chemicals, while the biochar is useful to the soil.
Biochar is trumped up of vital elements for instance hydrogen, carbon, nitrogen, sulfur and oxygen as well as reserves in the ash portion. Biochar is highly spongy, black and finely grained, with light mass, enormous surface area and pH, all of which have a progressive influence on its use to soil. The raw material (biomass) used and handling parameters grasp the properties of the biochar.
Wood chips, cow manure, grass, wheat straw, casava rhizome and rice husk are being used as raw resources by pyrolysis technology to make biochar (Ronsse et al. 2013). Various other materials including agronomic wastes (husks, peels, bark, straw, sawdust, seeds bagasse, wood chunks, corn cobs and stalks, urban waste and industrial wastes and urban/civic wastes (Kameyama et al. 2016) have been expansively utilized, therefore also attaining waste managing through its manufacture and usage (Woolf et al. 2010). The biomass utilized for the manufacture of biochar is chiefly composed of hemicellulose, cellulose and lignin polymers (Sullivan and Ball 2012). Between these, cellulose has been originated to be the chief constituent of maximum plant-derived biomasses, but lignin is also imperative in woody biomass.
Essentially, biochar additions to soil have been displayed to expressively increase soil nutrient preservation and accessibility to vegetation, and crop output (McCormack et al. 2019). The modification of topsoil with biochar has been stated to increase plant growth and yield indices. Furthermore, biochar grips the ability as an appropriate carrier of microbial inoculants to expand plant growth. The biochar uses improved water holding capacity, soil cation exchange capacity and organic material.
3 Improvement of Soil Microflora and Plant Growth by Biochar Amendment
Various studies have been reported in displaying the ability of biochar to improve the soil microflora, resulting in greater accretion of carbon in soil. Also adsorbing nutrients organic constituents, and gases, biochar is expected to offer a locale for actinomycetes, bacteria and fungi (Thies and Rillig 2012). The improvement of water holding after biochar utilization in soil has been well known (Busscher et al. 2010) and this could disturb the microbial inhabitants of soil. Numerous interpretations stated that phosphate solubilizing fungi in combination with biochar improved growth and yield attributes of Glycine max and Vigna radiate plants, compared to untreated control (Saxena et al. 2017). The usage of biochar enhanced mycorrhizal growth because this association has provided the best conditions to plant roots for more colonization (Solaiman et al. 2010).
Biochar applications are also reported in Phaseolus vulgaris to improve the biological N2 fixation largely due to larger accessibility of micronutrients to plants. Moreover, biochar reduced leaching of NH4+ by supporting it in the apparent soil where it was existing for plant approval (Lehmann et al. 2003). Mycorrhizal fungi were frequently involved in crop administration approaches as they were broadly utilized as additions for soil inoculum (Schwartz et al. 2006). The bacterial and fungal hyphae that inhabit the biochar bits (or other porous materials) may be threatened from soil predators such as Collembola, mites, nematodes and protozoans (Ezawa et al. 2002). Biochar can upsurge the cost of unharvested crop yields and confirm the efficient plant growth (Oguntunde et al. 2004). Biochar applications to the soil, has significantly enhanced the rice yield with small P availability (Silber et al. 2010).
Several properties are interrelated and may turn synergistically to enlarge crop output. The straight helpful properties of biochar mixing for the suitability of nutrients are largely because of the higher content of phosphorus, zinc, and potassium accessibility and, to a reduced level of copper and calcium. Very few studies have examined the ability for altering biochar in soil to influence plant competition against pathogens. Alterations regarding charcoal additions reported to have negative impact on the proliferation of phytopathogens (Matsubara et al. 2002). It has been reported that the powdered hardwood when used to make biochar and supplemented to asparagus grown soil exhibited a prominent decrease in root lesions instigated by F. asparagi, F. oxysporum and F. proliferatum compared to control soil where biochar was not added (Novak et al. 2009).
4 Effects of Biochar Application on Plant Diseases
Few studies have described the strength of biochar soil adjustment against soilborne diseases to influence the level of plant resistance. The charcoal based biochar application has been documented for their suppressive effects against soilborne Fusarium sp. (Matsubara et al. 2002; Elmer and Pignatello 2011). The suppression of soilborne pathogens owing to biochar applications is dependent upon several mechanisms, including: (i) nutrient solubilization and distribution to plant for improving growth and resisting pathogenic microflora (ii) improving the defense system of soil microbes against phytopathogens via enhancing antibiotic production and parasitism (iii) presence of organic compounds in biochar compositions result in propagation of resilient communities of beneficial microbes; (iv) the elicitors released by biochar applications may persuade the systemic defense pathways (Atkinson et al. 2010; Frenkel et al. 2017).
Microorganisms which cause reduction in toxic organic pollutants are usually considered extra resistant to a diversity of lethal organic compounds as well as pathogenic attacks. Moreover, volatile compounds and antibiotic producers are also found to be resilient to an assembly of antibiotics (Ahmed et al. 2017). Microorganisms producing antibiotic compounds have been reported in biochar-amended soil for instance Pseudomonas aeruginosa and Pseudomonas mendocina (Graber et al. 2010). The prospect that biochar encourages plant systemic resistance responses against disease microorganisms has been thoughtful in numerous different systems linking foliar pathogens. The severity of diseases triggered by biotrophic (Oidiopsis sicula) and necrotrophic (Botrytis cinerea) pathogens in tomato and pepper (Graber et al. 2010) was sigificantly reduced in biochar-amended treatments. Biochar soil adjustments in strawberry plants additionally resulted in destruction of pathogens including Podosphaera aphanis, B. cinerea, and Colletotrichum acutatum (Meller Harel et al. 2012).
Induced resistance in plants, was found to be effective against a wide range of pathogens and parasites comprising fungi, viruses, bacteria and nematodes. ISR is a functional state of enhanced defensive capability provoked by exact stimuli, whereby the plant’s innate defenses are potentiated against succeeding diseases (Vallad and Goodman 2004).
4.1 Biochar to Control of Soilborne Phytopathogens
The concerns as food safety, decreasing soil richness, profitability and climate variation are the active components after the introduction of new skills or new agricultural schemes. The alteration of soils for their stress alleviation goals at dipping the danger of pollutant handover to entities in closeness. Biochar can aid as a standard select for this drive because its basis is biological and it might be applied directly as pretreatment to soils (Beesley et al. 2011). There are dual features which mark biochar adjustment higher to other organic supplies: the main is the high permanency against deterioration, with the aim of persisting in soil for lengthier times showing enduring helps to soil and the another is having extra competence to keep the nutrients available. Biochar adjustment increases the soil quality by increasing the number of beneficial microbes, improving pH, cation-exchange capacity and moisture-holding ability (Mensah and Frimpong 2018).
The mixing of biochar to the soil has exposed the increase in accessibility of prime cations as well as in absorptions of nitrogen and phosphorus (Lehmann et al. 2003). Various studies have revealed the dominance of biochar in controlling phytopathogens. Biochar has been found to be very suppressive against soilborne (R. solani and species of Phytophthora and Fusarium) as well as airborne pathogens (powdery mildew and B. cinerea) (Bonanomi et al. 2015). The claim of the biochar resulting from citrus wood was proficient of decreasing the incidence of air-borne gray mold in chili caused by B. cinerea. However, the available data is very scarce regarding the disease suppressive potential of biochar against soilborne pathogens (Elmer and Pignatello 2011).
Additionally, biochar application was established to lessen plant diseases by influencing systemic resistance in plants in contradiction of different fungal pathogens, containing R. solani in cucumber, F. oxysporum and B. cinerea in tomato (Azeem et al. 2021). Biochar applications alone as well as in combination with mycorrhizal fungi to asparagus soils, has not only increased the asparagus biomass but also decreased the root rot infections caused by Fusarium (Elmer and Pignatello 2011; Thies and Rillig 2012; Akhter et al. 2016); Ogawa (2009) stated the usage of biochar and biochar edited manures for monitoring the diseases caused by fungi and bacteria in topsoil.
4.2 Role of Biochar in Induced Resistance Against Soilborne Phytopathogens
Generally, there are two well defined systems of induced resistance which are termed as are induced systemic resistance (ISR) and systemic acquired resistance (SAR). The chemical composition of elicitors as well as controlling pathways for both of these systems are prominently different from each other. SAR is connected with the production of pathogenesis-related (PR) proteins and arbitrated through a salicylic acid dependent procedure (Naz et al. 2018, 2021a). The hypersensitive reaction is known as the initiative response of SAR against pathogenic. However, certain fungal and bacterial species particularly PGPR colonization with plant roots develop systemically the ISR mechanism (Van der Ent et al. 2009; Ullah et al. 2020).
The ISR resistance mechanism is arbitrated by jasmonic acid and ethylene signaling however, the induction of PR-proteins is not included in such type of resistance mechanism (Van der Ent et al. 2009). The biological as well as chemical elicitors which can be released from nonpathogenic or pathogenic microorganisms, can elicit SAR (Ali et al. 2018; Naz et al. 2021b). For instance, the compounds released from Trichoderma spp. can influence SAR as much as they stimulate ISR (Nawrocka and Małolepsza 2013). Chemical stimulators of systemic resistance comprise the synthetic SA-analogues acibenzolar-S-methyl and 2,6-dichloroisoniciotinic acid, methyl jasmonate, chitin and chitosan, β-aminobutyric acid and laminarin, silicon, fatty acids, amino acids, and phosphate salts, remains can also produce systemic resistance, as can ecological agents such as moisture, osmotic, temperature stresses and mechanical wounding (Romero-Puertas et al. 2008).
Primed plants show sooner and sturdier instigation of cellular defense following pathogen challenge relative to the un-primed or untreated plants (Zimmerman et al. 2011; Naz et al. 2021a, 2022), comprising earlier oxidative eruption and strongly up-regulating the expression of defense genes (Zimmerman et al. 2011; Meller Harel et al. 2012; Naz et al. 2014; Butt et al. 2019). While the molecular and physiological mechanisms underlying well-informed responses are widely unidentified, priming has been detected to be an essential part of both ISR and SAR (Yasmin et al. 2020). Molecular indication for the induction of plant defenses systemically via both ISR and SAR paths by biochar was observed (Meller Harel et al. 2012; Jaiswal et al. 2020). Biochar addition to the hitting medium of strawberry plants repressed fungal diseases produced B. cinerea, C. acutatum, and P. apahanis.
The biochar amendments to plant roots confirmed the ethylene and SA-induced expression by increasing the expression of defense-related genes including FaWRKY1, FaPR1, Falox, Faolp2 and Fra a3 (Meller Harel et al. 2012). The question increases, which mechanism(s) are employed by biochar to induce ISR and SAR defense systems, PGPF and PGPR root colonization is known to develop ISR systemically in plants (Hossain et al. 2017). The Bacteroidetes associated Flavobacterium was found to be the most intensely tempted by the biochar. Adherents of the Flavobacterium, usually own a storage of extracellular enzymes for example chitinases and proteinases with having the potential to damage fungi, insects, nematode and bacteria residents (Bernardet and Bowman 2006). Also, many other species of genus Flavobacterium are commonly known to release secondary metabolites including antibiotics (Enisoglu-Atalay et al. 2018).
In addition, some Flavobacterium strains were proficient of instigating a fighting response of plants to diverse diseases (Kolton et al. 2011; Enisoglu-Atalay et al. 2018). Further, hydrolytic enzyme-producing genera including Cellvibrio (Betaproteobacteria) were also persuaded in the rhizosphere of the biochar-altered pepper plants (Kolton et al. 2011). Stimulatingly, biochar alteration was found to antagonize the Pseudoxanthomonas genus (Rajkumar et al. 2008; Kolton et al. 2011). It rests to be grasped what types of biochar can persuade conflict responses, seeing the very big inconsistency in chemical and chemical properties that biochar display, contingent on original pyrolysis and conditions (Sohi et al. 2009). Yet, we imagine that disease control efficiency will differ with other biochar production, biomass sources, temperatures, plant growth systems, plant species and diseases (Table 10.1).
5 Status
Biochar as an important constituent of soil-less substrates has been tried in several experiments; which were focused with numerous types of biochars, and several studies intricate mixtures of biochar with other additions for instance fertilizers and mycorrhiza (Costell et al. 2012) and humic acid harvests (Vickers 2017). The studies verified elevated percentages of biochar: growing media frequently reaching very high biochar percentages (>60%) (Dumroese et al. 2011). Analyses encompassed chemical properties and several parameters of plant development and additional measurements for instance photosynthetic pigments (Fascella 2015). Mostly, biochar had an impartial or helpful influence on plant growth paralleled with peat media when present in absorptions lower than 30%, and in some works even an abundant concentration was found to be not injurious (Méndez et al. 2015; Nieto et al. 2016).
A helpful impact of biochar on reducing plant fungal diseases was first reported about 170 years ago and described consideration in the last decade where numerous pathosystems particularly soilborne pathogens, were considered by different crowds globally (Elad et al. 2011; Postma et al. 2013; Iyyer et al. 2014). Later, Bonanomi et al. 2015 summarized the data from 13 pathosystems that shown the result of biochar on plant diseases. In their study, they described that 85% of the studies exposed a helpful effect of biochar in reducing the severity of plant disease, 12% showed no result, and only 3% presented that biochar mixing up were favorable to plant disease (Zhang and Lin 2014). Though, their analysis did not deliberate the detail that many of these revisions exposed that plant resistance and(or) vulnerability to disease was reliant on the critical aspect of the biochar dosage (Conversa et al. 2015).
6 Conclusion
Biochar can be potentially amended to soil for improving the plant growth, performance and alleviating the negative impacts of soilborne diseases which eventually can reduce the crop yield losses. Biochar amendments cause such changes of controlling pathogens and enhancing the community of beneficial microbes by making adjustments in the soil microflora. Biochar alterations has been reported in this chapter to significantly enhance the beneficial bacterial community which is known to improve the soil and plant health by improving the physico-chemical properties of soil. Moreover, the biochar treatment should be taken as ecofriendly and very efficient practice as it can effectively suppress the pathogenic growth and, applied as a sustainable approach in agriculture systems for soil management.
Therefore, it can be decided by this inclusive review in this chapter that biochar has the potential to increase the soil properties, microbial abundance, plant growth, inhibiting soilborne pathogens and biological nitrogen fixation. Consequently, it is suggested to practice biochar as a soil adjustment for long-term carbon sink renovation.
References
Ahmed F, Islam MS, Iqbal MT (2017) Biochar amendment improves soil fertility and productivity of mulberry plant. Eurasian J Soil Sci 6:226–237. https://doi.org/10.18393/ejss.291945
Akhter A, Hage-Ahmed K, Soja G, Steinkellner S (2016) Potential of fusarium wilt-inducing chlamydospores, in vitro behaviour in root exudates and physiology of tomato in biochar and compost amended soil. Plant Soil 406:425–440. https://doi.org/10.1007/s11104-016-2948-4
Alburquerque JA, Salazar P, Barrón V, Torrent J, del Campillo MC, Gallardo A, Villar R (2013) Enhanced wheat yield by biochar addition under different mineral fertilization levels. Agronom Sustain Dev 33:475–484. https://doi.org/10.1007/s13593-012-0128-3
Ali A, Shah L, Rahman S, Riaz MW, Yahya M, Xu YJ, Liu F, Si W, Jiang H, Cheng B (2018) Plant defense mechanism and current understanding of salicylic acid and NPRs in activating SAR. Physiol Mol Plant Pathol 104:15–22. https://doi.org/10.1016/j.pmpp.2018.08.001
Al-Zahrani HS, Alharby HF, Fahad S (2022) Antioxidative defense system, hormones, and metabolite accumulation in different plant parts of two contrasting rice cultivars as influenced by plant growth regulators under heat stress. Front Plant Sci 13:911846. https://doi.org/10.3389/fpls.2022.911846
Atkinson CJ, Fitzgerald JD, Hipps NA (2010) Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: a review. Plant Soil 337:1–18. https://doi.org/10.1007/s11104-010-0464-5
Azeem M, Hassan TU, Tahir MI, Ali A, Jeyasundar PGSA, Hussain Q, Bashir S, Mehmood S, Zhang Z (2021) Tea leaves biochar as a carrier of Bacillus cereus improves the soil function and crop productivity. App Soil Ecol 157:103732. https://doi.org/10.1016/j.apsoil.2020.103732
Bamagoos A, Alharby H, Fahad S (2021) Biochar coupling with phosphorus fertilization modifies antioxidant activity, osmolyte accumulation and reactive oxygen species synthesis in the leaves and xylem sap of rice cultivars under high-temperature stress. Physiol Mol Biol Plants 27:2083–2100. https://doi.org/10.1007/s12298-021-01062-7
Beesley L, Moreno-Jiménez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T (2011) A review of biochars’ potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut 159:3269–3282. https://doi.org/10.1016/j.envpol.2011.07.023
Bernardet J-F, Bowman JP (2006) The genus flavobacterium. Prokaryotes 7:481–531. https://doi.org/10.1007/0-387-30747-8_17
Bonanomi G, Ippolito F, Scala F (2015) A “black” future for plant pathology? Biochar as a new soil amendment for controlling plant diseases. J Plant Pathol 97(2):223–234. https://doi.org/10.4454/jpp.v97i2.3381
Busscher WJ, Novak JM, Evans DE, Watts DW, Niandou M, Ahmedna M (2010) Influence of pecan biochar on physical properties of a Norfolk loamy sand. Soil Sci 175:10–14. https://doi.org/10.1097/SS.0b013e3181cb7f46
Butt UR, Naz R, Nosheen A, Yasmin H, Keyani R, Hussain I, Hassan MN (2019) Changes in pathogenesis-related gene expression in response to bioformulations in the apoplast of maize leaves against fusarium oxysporum. J Plant Interact 14:61–72. https://doi.org/10.1080/17429145.2018.1550217
Conversa G, Bonasia A, Lazzizera C, Elia A (2015) Influence of biochar, mycorrhizal inoculation, and fertilizer rate on growth and flowering of Pelargonium (Pelargonium zonale L.) plants. Front Plant Sci 6:429. https://doi.org/10.3389/fpls.2015.00429
Costell MH, Ancellin N, Bernard RE, Zhao S, Upson JJ, Morgan LA, Hauk K, Olzinski AR, Ballard VL, Herry K (2012) Comparison of soluble guanylate cyclase stimulators and activators in models of cardiovascular disease associated with oxidative stress. Front Pharmacol 3:128. https://doi.org/10.3389/fphar.2012.00128
Dumroese RK, Heiskanen J, Englund K, Tervahauta A (2011) Pelleted biochar: chemical and physical properties show potential use as a substrate in container nurseries. Biomass Bioenergy 35:2018–2027. https://doi.org/10.1016/j.biombioe.2011.01.053
Egamberdieva D, Wirth S, Behrendt U, Abd Allah EF, Berg G (2016) Biochar treatment resulted in a combined effect on soybean growth promotion and a shift in plant growth promoting rhizobacteria. Front Microbiol 7:209. https://doi.org/10.3389/fmicb.2016.00209
Elad Y, Cytryn E, Harel YM, Lew B, Graber ER (2011) The biochar effect: plant resistance to biotic stresses. Phytopathol Mediterr 50:335–349. https://doi.org/10.14601/Phytopathol_Mediterr-9807
Elmer WH, Pignatello JJ (2011) Effect of biochar amendments on mycorrhizal associations and fusarium crown and root rot of asparagus in replant soils. Plant Dis 95:960–966. https://doi.org/10.1094/PDIS-10-10-0741
Enders A, Hanley K, Whitman T, Joseph S, Lehmann J (2012) Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour Technol 114:644–653. https://doi.org/10.1016/j.biortech.2012.03.022
Enisoglu-Atalay V, Atasever-Arslan B, Yaman B, Cebecioglu R, Kul A, Ozilhan S, Ozen F, Catal T (2018) Chemical and molecular characterization of metabolites from Flavobacterium sp. PLoS One 13:0205817. https://doi.org/10.1371/journal.pone.0205817
Ezawa T, Yamamoto K, Yoshida S (2002) Enhancement of the effectiveness of indigenous arbuscular mycorrhizal fungi by inorganic soil amendments. Soil Sci Plant Nutr 48:897–900. https://doi.org/10.1080/00380768.2002.10408718
Fahad S, Adnan M, Hassan S, Saud S, Hussain S, Wu C, Wang D, Hakeem KR, Alharby HF, Turan V, Khan MA, Huang J (2019) Rice responses and tolerance to high temperature. In: Hasanuzzaman M, Fujita M, Nahar K, Biswas JK (eds) Advances in rice research for abiotic stress tolerance. Woodhead Publishing Ltd, Abington/Cambridge, pp 201–224. https://doi.org/10.1016/B978-0-12-814332-2.00010-1
Fahad S, Hasanuzzaman M, Alam M, Ullah H, Saeed M, Ali Khan I, Adnan M (eds) (2020) Environment, climate, plant and vegetation growth. Spring, Switzerland. https://doi.org/10.1007/978-3-030-49732-3
Fahad S, Saud S, Yajun C, Chao W, Depeng W (eds) (2021) Abiotic stress in plants. IntechOpen, London. https://doi.org/10.5772/intechopen.91549
Fahad S, Adnan M, Saud S (eds) (2022) Improvement of plant production in the era of climate change. In: Footprints of climate variability on plant diversity, 1st edn. CRC Press, Boca Raton. https://doi.org/10.1201/9781003286417
Fascella G (2015) Growing substrates alternative to peat for ornamental plants. In: Soilless culture-Use of substrates for the production of quality horticultural crops, pp 47–67. https://doi.org/10.5772/59596
Fischer D, Glaser B (2012) Synergisms between compost and biochar for sustainable soil amelioration. Management of organic waste 1:167–198. http://www.intechopen.com/books/management-of-organic-waste/synergism-between-biocharand-compost-for-sustainable-soil-amelioration
Frenkel O, Jaiswal AK, Elad Y, Lew B, Kammann C, Graber ER (2017) The effect of biochar on plant diseases: what should we learn while designing biochar substrates? J Environ Eng Landsc 25:105–113. https://doi.org/10.3846/16486897.2017.1307202
Graber ER, Meller Harel Y, Kolton M, Cytryn E, Silber A, Rav David D, Tsechansky L, Borenshtein M, Elad Y (2010) Biochar impact on development and productivity of pepper and tomato grown in fertigated soilless media. Plant Soil 337:481–496. https://doi.org/10.1007/s11104-010-0544-6
Graber E, Frenkel O, Jaiswal A, Elad Y (2014) How may biochar influence severity of diseases caused by soilborne pathogens? Carbon Manag 5:169–183. https://doi.org/10.1080/17583004.2014.913360
Ho S-H, Zhu S, Chang J-S (2017) Recent advances in nanoscale-metal assisted biochar derived from waste biomass used for heavy metals removal. Bioresour Technol 246:123–134. https://doi.org/10.1016/j.biortech.2017.08.061
Hossain M, Sultana F, Islam S (2017) Plant growth-promoting fungi (PGPF): phytostimulation and induced systemic resistance. In: Plant-microbe interact in agro-ecol pers. pp 135–191
Huang W-K, Ji H-l, Gheysen G, Debode J, Kyndt T (2015) Biochar-amended potting medium reduces the susceptibility of rice to root-knot nematode infections. BMC Plant Biol 15:1–15. https://doi.org/10.1186/s12870-015-0654-7
Ibad U, Dost M, Maria M, Shadman K, Muhammad A, Fahad S, Muhammad I, Ishaq AM, Aizaz A, Muhammad HS, Muhammad S, Farhana G, Muhammad I, Muhammad ASR, Hafiz MH, Wajid N, Shah S, Jabar ZKK, Masood A, Naushad A, AkbarM R, Shah MK, Jan B (2022) Comparative effects of biochar and NPK on wheat crops under different management systems. Crop Pasture Sci 74(40):31. https://doi.org/10.1071/CP21146
Irfan M, Muhammad M, Muhammad JK, Khadim MD, Dost M, Ishaq AM, Waqas A, Fahad S, Saud S et al (2021) Heavy metals immobilization and improvement in maize (Zea mays L.) growth amended with biochar and compost. Sci Rep 11:18416. https://doi.org/10.1038/s41598-021-97525-8
Islami T, Guritno B, Basuki N, Suryanto A (2011) Maize yield and associated soil quality changes in cassava+ maize intercropping system after 3 years of biochar application. J Agri Food Technol 1:112–115. Corpus ID: 130733835
Iyyer, M., Boyd-Graber, J., Claudino, L., Socher, R., Daumé III, H. (2014). A neural network for factoid question answering over paragraphs. In: Proceedings of the 2014 conference on (EMNLP), pp 633–644
Jaiswal AK, Elad Y, Graber ER, Frenkel O (2014) Rhizoctonia solani suppression and plant growth promotion in cucumber as affected by biochar pyrolysis temperature, feedstock and concentration. Soil Biol Biochem 69:110–118. https://doi.org/10.1016/j.soilbio.2013.10.051
Jaiswal AK, Graber ER, Elad Y, Frenkel O (2019) Biochar as a management tool for soilborne diseases affecting early stage nursery seedling production. J Crop Prot 120:34–42. https://doi.org/10.1016/j.cropro.2019.02.014
Jaiswal AK, Alkan N, Elad Y, Sela N, Philosoph AM, Graber ER, Frenkel O (2020) Molecular insights into biochar-mediated plant growth promotion and systemic resistance in tomato against Fusarium crown and root rot disease. Sci Rep 10(1):13934. https://doi.org/10.1038/s41598-020-70882-6
Kameyama K, Miyamoto T, Iwata Y, Shiono T (2016) Influences of feedstock and pyrolysis temperature onthenitrate adsorption of biochar. Soil Sci Plant Nutr 62:180–184. https://doi.org/10.1080/00380768.2015.1136553
Khadim D, Fahad S, Jahangir MMR, Iqbal M, Syed SA, Shah AK, Ishaq AM, Rahul D et al (2021a) Biochar and urease inhibitor mitigate NH3 and N2O emissions and improve wheat yield in a urea fertilized alkaline soil. Sci Rep 11:17413. https://doi.org/10.1038/s41598-021-96771-0
Khadim D, Saif-ur-R FS, Syed SA, Shah AK et al (2021b) Influence of variable biochar concentration on yield-scaled nitrous oxide emissions, wheat yield and nitrogen use efficiency. Sci Rep 11:16774. https://doi.org/10.1038/s41598-021-96309-4
Khan MMH, Niaz A, Umber G, Muqarrab A, Muhammad AA, Muhammad I, Shabir H, Shah F, Vibhor A, Shams HA-H, Reham A, Syed MBA, Nadiyah MA, Ali TKZ, Subhan D, Rahul D (2021) Synchronization of boron application methods and rates is environmentally friendly approach to improve quality attributes of Mangifera indica L, vol 29. Saudi J Bio Sci, On sustainable basis, pp 1869–1880. https://doi.org/10.1016/j.sjbs.2021.10.036
Khatun M, Sarkar S, Era FM, Islam AKMM, Anwar MP, Fahad S, Datta R, Islam AKMA (2021) Drought stress in grain legumes: effects, tolerance mechanisms and management. Agronomy 11:2374. https://doi.org/10.3390/agronomy11122374
Kolton M, Meller Harel Y, Pasternak Z, Graber ER, Elad Y, Cytryn E (2011) Impact of biochar application to soil on the root-associated bacterial community structure of fully developed greenhouse pepper plants. J App Environ Microbiol 77:4924–4930. https://doi.org/10.1128/AEM.00148-11
Kolton M, Graber ER, Tsehansky L, Elad Y, Cytryn E (2017) Biochar-stimulated plant performance is strongly linked to microbial diversity and metabolic potential in the rhizosphere. New Phytol 213:1393–1404. https://doi.org/10.1111/nph.14253
Lehmann J, Pereira da Silva J, Steiner C, Nehls T, Zech W, Glaser B (2003) Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: fertilizer, manure and charcoal amendments. Plant Soil 249:343–357. https://doi.org/10.1023/A:1022833116184
Liu H, Yang M, Zhu S (2019) Strategies to solve the problem of soil sickness of Panax notoginseng (family: Araliaceae). Allelopathy J 47:37–56. https://doi.org/10.26651/allelo.j/2019-47-1-1218
Mao J-D, Johnson R, Lehmann J, Olk D, Neves E, Thompson M, Schmidt-Rohr K (2012) Abundant and stable char residues in soils: implications for soil fertility and carbon sequestration. Environ Sci Technol 46:9571–9576. https://doi.org/10.1021/es301107c
Matsubara Y, Hasegawa N, Fukui H (2002) Incidence of Fusarium root rot in asparagus seedlings infected with arbuscular mycorrhizal fungus as affected by several soil amendments. J Jpn Soc Hortic Sci 71:370–374. https://doi.org/10.2503/jjshs.71.370
McCormack SA, Ostle N, Bardgett RD, Hopkins DW, Pereira MG, Vanbergen AJ (2019) Soil biota, carbon cycling and crop plant biomass responses to biochar in a temperate mesocosm experiment. Plant Soil 440:341–356. https://doi.org/10.1007/s11104-019-04062-5
Mehari ZH, Elad Y, Rav-David D, Graber ER, Meller Harel Y (2015) Induced systemic resistance in tomato (Solanum lycopersicum) against Botrytis cinerea by biochar amendment involves jasmonic acid signaling. Plant Soil 395:31–44. https://doi.org/10.1007/s11104-015-2445-1
Meller Harel Y, Elad Y, Rav-David D, Borenstein M, Shulchani R, Lew B, Graber ER (2012) Biochar mediates systemic response of strawberry to foliar fungal pathogens. Plant Soil 357:245–257. https://doi.org/10.1007/s11104-012-1129-3
Méndez A, Paz-Ferreiro J, Gil E, Gascó G (2015) The effect of paper sludge and biochar addition on brown peat and coir based growing media properties. Sci Hortic 193:225–230. https://doi.org/10.1016/j.scienta.2015.07.032
Mensah AK, Frimpong KA (2018) Biochar and/or compost applications improve soil properties, growth, and yield of maize grown in acidic rainforest and coastal savannah soils in Ghana. Intern J Agronom 2018:1–8. https://doi.org/10.1155/2018/6837404
Muhammad I, Khadim D, Fahad S, Imran M, Saud A, Manzer HS, Shah S, Jabar ZKK et al (2022) Exploring the potential effect of Achnatherum splendens L.–derived biochar treated with phosphoric acid on bioavailability of cadmium and wheat growth in contaminated soil. Environ Sci Pollut Res 29:37676–37684. https://doi.org/10.1007/s11356-021-17950-0
Murakami T, Xu G, Suda T, Matsuzawa Y, Tani H, Fujimori T (2007) Some process fundamentals of biomass gasification in dual fluidized bed. Fuel 86(1–2):244–255. https://doi.org/10.1016/j.fuel.2006.05.025
Nawrocka J, Małolepsza U (2013) Diversity in plant systemic resistance induced by Trichoderma. Biol Control 67:149–156. https://doi.org/10.1016/j.biocontrol.2013.07.005
Naz R, Bano A, Wilson NL, Guest D, Roberts TH (2014) Pathogenesis-related protein expression in the apoplast of wheat leaves protected against leaf rust following application of plant extracts. Phytopathology 104:933–944. https://doi.org/10.1094/PHYTO-11-13-0317-R
Naz R, Nosheen A, Yasmin H, Bano A, Keyani R (2018) Botanical-chemical formulations enhanced yield and protection against Bipolaris sorokiniana in wheat by inducing the expression of pathogenesis-related proteins. PLoS One 13:0196194. https://doi.org/10.1371/journal.pone.0196194
Naz R, Bano A, Nosheen A, Yasmin H, Keyani R, Shah STA, Anwar Z, Roberts TH (2021a) Induction of defense-related enzymes and enhanced disease resistance in maize against Fusarium verticillioides by seed treatment with Jacaranda mimosifolia formulations. Sci Rep 11:1–15. https://doi.org/10.1038/s41598-020-79306-x
Naz R, Batool S, Shahid M, Keyani R, Yasmin H, Nosheen A, Hassan MN, Mumtaz S, Siddiqui MH (2021b) Exogenous silicon and hydrogen sulfide alleviates the simultaneously occurring drought stress and leaf rust infection in wheat. Plant Physiol Biochem 166:558–571. https://doi.org/10.1016/j.plaphy.2021.06.034
Naz R, Asif T, Mubeen S, Khushhal S (2022) Seed application with microbial inoculants for enhanced plant growth. In: Sustain hortic. Elsevier, pp 333–368 https://doi.org/10.1016/B978-0-323-91861-9.00008-2
Nieto A, Gasco G, Paz-Ferreiro J, Fernández J, Plaza C, Mendez A (2016) The effect of pruning waste and biochar addition on brown peat based growing media properties. Sci Hortic 199:142–148. https://doi.org/10.1016/j.scienta.2015.12.012
Nikraftar F, Taheri P, Rastegar MF, Tarighi S (2013) Tomato partial resistance to Rhizoctonia solani involves antioxidative defense mechanisms. Physiol Mol Plant Pathol 81:74–83. https://doi.org/10.1016/j.pmpp.2012.11.004
Novak JM, Lima I, Xing B, Gaskin JW, Steiner C, Das K, Ahmedna M, Rehrah D, Watts DW, Busscher WJ (2009) Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann Environ Sci 3:195–206
Ogawa M (2009) Charcoal use in agriculture in Japan. Keynote address. In: 1st Asia Pacific biochar conference, Gold Coast, Australia, May 17–20, 2009
Oguntunde PG, Fosu M, Ajayi AE, Van De Giesen N (2004) Effects of charcoal production on maize yield, chemical properties and texture of soil. Biol Fertil Soils 39:295–299. https://doi.org/10.1007/s00374-003-0707-1
Postma J, Clematis F, Nijhuis EH, Someus E (2013) Efficacy of four phosphate-mobilizing bacteria applied with an animal bone charcoal formulation in controlling Pythium aphanidermatum and fusarium oxysporum f. sp. radicis lycopersici in tomato. Biol Control 67:284–291. https://doi.org/10.1016/j.biocontrol.2013.07.002
Rajkumar M, Lee K, Freitas H (2008) Effects of chitin and salicylic acid on biological control activity of Pseudomonas spp. against damping off of pepper. S Afr J Bot 74:268–273. https://doi.org/10.1016/j.sajb.2007.11.014
Riaz R, Khan A, Khan WJ, Jabeen Z, Yasmin H, Naz R, Nosheen A, Hassan MN (2021) Vegetable associated Bacillus spp. suppress the pea (Pisum sativum L.) root rot caused by Fusarium solani. Biol Control 158:104610. https://doi.org/10.1016/j.biocontrol.2021.104610
Romero-Puertas MC, Campostrini N, Mattè A, Righetti PG, Perazzolli M, Zolla L, Roepstorff P, Delledonne M (2008) Proteomic analysis of S-nitrosylated proteins in Arabidopsis thaliana undergoing hypersensitive response. Proteomics 8:1459–1469. https://doi.org/10.1002/pmic.200700536
Ronsse F, Van Hecke S, Dickinson D, Prins W (2013) Production and characterization of slow pyrolysis biochar: influence of feedstock type and pyrolysis conditions. GCB Bioenergy 5:104–115. https://doi.org/10.1111/gcbb.12018
Saxena J, Rawat J, Kumar R (2017) Conversion of biomass waste into biochar and the effect on mung bean crop production. Clean Soil Air Water 45:1501020. https://doi.org/10.1002/clen.201501020
Schwartz MW, Hoeksema JD, Gehring CA, Johnson NC, Klironomos JN, Abbott LK, Pringle A (2006) The promise and the potential consequences of the global transport of mycorrhizal fungal inoculum. Ecol Lett 9:501–515. https://doi.org/10.1111/j.1461-0248.2006.00910
Shaaban M, Van Zwieten L, Bashir S, Younas A, Núñez-Delgado A, Chhajro MA, Kubar KA, Ali U, Rana MS, Mehmood MA (2018) A concise review of biochar application to agricultural soils to improve soil conditions and fight pollution. J Environ Manag 228:429–440. https://doi.org/10.1016/j.jenvman.2018.09.006
Shah S, Li X, Jiang Z, Fahad S, Hassan S (2022) Exploration of the phytohormone regulation of energy storage compound accumulation in microalgae. Food Energy Secur 11(4):e418. https://doi.org/10.1002/fes3.418
Silber A, Levkovitch I, Graber ER (2010) pH-dependent mineral release and surface properties of cornstraw biochar: agronomic implications. Environ Sci Technol 44:9318–9323. https://doi.org/10.1021/es101283d
Sohi S, Lopez-Capel E, Krull E, Bol R (2009) Biochar, climate change and soil: a review to guide future research. CSIRO Land Water Sci Rep 5:17–31
Solaiman ZM, Blackwell P, Abbott LK, Storer P (2010) Direct and residual effect of biochar application on mycorrhizal root colonisation, growth and nutrition of wheat. Soil Res 48:546–554. https://doi.org/10.1071/SR10002
Subhan D, Zafar-ul-Hye M, Fahad S, Saud S, Martin B, Tereza H, Rahul D (2020) Drought stress alleviation by ACC deaminase producing achromobacter xylosoxidans and enterobacter cloacae, with and without timber waste biochar in maize. Sustain 12:6286. https://doi.org/10.3390/su12156286
Sullivan AL, Ball R (2012) Thermal decomposition and combustion chemistry of cellulosic biomass. Atmos Environ 47:133–141. https://doi.org/10.1016/j.atmosenv.2011.11.022
Tariq M, Ahmad S, Fahad S, Abbas G, Hussain S, Fatima Z, Nasim W, Mubeen M, Ur Rehman MH, Khan MA, Adnan M (2018) The impact of climate warming and crop management on phenology of sunflower-based cropping systems in Punjab. Pakistan Agri Forest Met 15(256):270–282. https://doi.org/10.1016/j.agrformet.2018.03.015
Thies JE, Rillig MC (2012) Characteristics of biochar: biological properties. In: Biochar environ manage. Routledge, pp 117–138
Toju H, Peay KG, Yamamichi M, Narisawa K, Hiruma K, Naito K, Fukuda S, Ushio M, Nakaoka S, Onoda Y (2018) Core microbiomes for sustainable agroecosystems. Nat Plants 4:247–257. https://doi.org/10.1038/s41477-018-0139-4
Ullah H, Yasmin H, Mumtaz S, Jabeen Z, Naz R, Nosheen A, Hassan MN (2020) Multitrait Pseudomonas spp. isolated from monocropped wheat (Triticum aestivum) suppress Fusarium root and crown rot. Phytopathology 110:582–592. https://doi.org/10.1094/PHYTO-10-19-0383-R
Vallad GE, Goodman RM (2004) Systemic acquired resistance and induced systemic resistance in conventional agriculture. Crop Sci 44:1920–1934. https://doi.org/10.2135/cropsci2004.1920
Van der Ent S, Van Wees SC, Pieterse CM (2009) Jasmonate signaling in plant interactions with resistance-inducing beneficial microbes. Phytochem Lett 70:1581–1588. https://doi.org/10.1016/j.phytochem.2009.06.009
Vickers NJ (2017) Animal communication: when i’m calling you, will you answer too? Curr Biol 27:713–715. https://doi.org/10.1016/j.cub.2017.05.064
Wang H, Guo W, Liu B, Wu Q, Luo H, Zhao Q, Si Q, Sseguya F, Ren N (2019) Edge-nitrogenated biochar for efficient peroxydisulfate activation: an electron transfer mechanism. Water Res 160:405–414. https://doi.org/10.1016/j.watres.2019.05.059
Wang L, Chen L, Tsang DC, Guo B, Yang J, Shen Z, Hou D, Ok YS, Poon CS (2020) Biochar as green additives in cement-based composites with carbon dioxide curing. J Clean Prod 258:120678. https://doi.org/10.1016/j.jclepro.2020.120678
Wiqar A, Arbaz K, Muhammad Z, Ijaz A, Muhammad A, Fahad S (2022) Relative efficiency of biochar particles of different sizes for immobilising heavy metals and improving soil properties. Crop Pasture Sci 74:112–120. https://doi.org/10.1071/CP20453
Woolf D, Amonette JE, Street-Perrott FA, Lehmann J, Joseph S (2010) Sustainable biochar to mitigate global climate change. Nat Commun 1:1–9. https://doi.org/10.1038/ncomms1053. www.nature.com/naturecommunications
Wu C, Tang S, Li G, Wang S, Fahad S, Ding Y (2019) Roles of phytohormone changes in the grain yield of rice plants exposed to heat: a review. PeerJ 7:e7792. https://doi.org/10.7717/peerj.7792
Wu C, Kehui C, She T, Ganghua L, Shaohua W, Fahad S, Lixiao N, Jianliang H, Shaobing P, Yanfeng D (2020) Intensified pollination and fertilization ameliorate heat injury in rice (Oryza sativa L.) during the flowering stage. Field Crops Res 252:107e795
Xiang Y, Xu Z, Zhou Y, Wei Y, Long X, He Y, Zhi D, Yang J, Luo L (2019) A sustainable ferromanganese biochar adsorbent for effective levofloxacin removal from aqueous medium. Chemosphere 237:124464. https://doi.org/10.1016/j.chemosphere.2019.124464
Xue B, Huang L, Li X, Lu J, Gao R, Kamran M, Fahad S (2022) Effect of clay mineralogy and soil organic carbon in aggregates under straw incorporation. Agronomy 12:534. https://doi.org/10.3390/agronomy12020534
Yang M, Yuan Y, Huang H, Ye C, Guo C, Xu Y, Wang W, He X, Liu Y, Zhu S (2019) Steaming combined with biochar application eliminates negative plant-soil feedback for sanqi cultivation. Soil Tillage Res 189:189–198. https://doi.org/10.1016/j.still.2019.02.006
Yasmin H, Naz R, Nosheen A, Hassan MN, Ilyas N, Sajjad M, Anjum S, Gao X, Geng Z (2020) Identification of new biocontrol agent against charcoal rot disease caused by Macrophomina phaseolina in soybean (Glycine max L.). Sustainability 12:6856. https://doi.org/10.3390/su12176856
Zhang T, Lin W (2014) Metal–organic frameworks for artificial photosynthesis and photocatalysis. Chem Soc Rev 43:5982–5993. https://doi.org/10.1039/C4CS00103F
Zia MA, Riaz R, Batool A, Yasmin H, Nosheen A, Naz R, Hassan MN (2021) Glucanolytic rhizobacteria associated with wheat-maize cropping system suppress the Fusarium wilt of tomato (Lycopersicum esculentum L). Sci Hortic 287:110275. https://doi.org/10.1016/j.scienta.2021.110275
Zimmerman AR, Gao B, Ahn M-Y (2011) Positive and negative carbon mineralization priming effects among a variety of biochar-amended soils. Soil Biol Biochem 43:1169–1179. https://doi.org/10.1016/j.soilbio.2011.02.005
Zwart DC, Kim S-H (2012) Biochar amendment increases resistance to stem lesions caused by Phytophthora spp. in tree seedlings. Hort Sci 47:1736–1740. https://doi.org/10.21273/HORTSCI.47.12.1736
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Abid, F., Naz, R., Asif, T. (2023). Biochar for Crop Protection from Soil Borne Diseases. In: Fahad, S., Danish, S., Datta, R., Saud, S., Lichtfouse, E. (eds) Sustainable Agriculture Reviews 61. Sustainable Agriculture Reviews, vol 61. Springer, Cham. https://doi.org/10.1007/978-3-031-26983-7_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-26983-7_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-26982-0
Online ISBN: 978-3-031-26983-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)