Abstract
Plastic pollution has become a major environmental concern of the globe. The increasing plastic pollution has captured the attention of many researchers. Disposal of plastic on land and in water, in due course, leads to the production of various plastic particles like micro (1–5 mm) and nano (1–100 nm) plastics by diverse physico-chemical processes. These micro- and nanoplastics are ubiquitous and have become the major contributors of pollution to aquatic and terrestrial ecosystems. Plants, which are considered as the main producers of the terrestrial and aquatic ecosystems, are vulnerable to the plastic pollutants. Terrestrial and aquatic plants are exposed to different types of plastics, leading to altered physiological and metabolic functions. This chapter has highlighted the impact of micro- and nanoplastics on plants during seed germination and growth. The altered responses of plants are because of the imbalances in soil microbial community, anti-oxidative enzymes and photosynthetic and metabolic activities. Various characteristics of autotrophic macrophytes on exposure to plastic pollutants are also being focussed on. Many of these studies have shown the inhibitory effects of these particles, which are dependent on their size, shape, charge and concentration used. Based on the size, they get adsorbed or internalised by the plant, which reduces its growth and photosynthetic activity primarily by inducing oxidative stress. The accumulated particles block the pores on the seed and root surface thereby affecting seed germination and also nutrient uptake by roots. This chapter covers the major research topics that investigated the effects of micro- and nanoplastics on various parameters of plant growth and functions. The effects of micro- and nanoplastics on plant functionalities are being discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Microplastics
- Nanoplastics
- Functionalities
- Polyethylene
- High-density polyethylene
- Low-density polyethylene
- Polystyrene
- Oxidative stress
1 Introduction
We live in a world surrounded by plastic. Usage of plastic has become convenient as it is economical and reliable (Boucher & Friot, 2017). The usage of plastic has increased by 25 folds over last 40 years (Sutherland et al., 2014). Because of its extensive and uncontrollable use, plastic pollution has become a global issue. Plastic production was estimated to be more than 359 million tons as of 2019 (Europe, 2017). The estimated plastic production would reach 12,000 million tons by 2050 (Geyer et al., 2017). In spite of recycle and reuse, 32% of the plastic waste still remains in the natural habitat (Geyer et al., 2017; de Souza Machado et al., 2018).
Plastic contamination is omnipresent, on land and in water, namely, oceans, lakes, estuaries and shores (Alomar et al., 2016; Browne et al., 2011; Naidoo et al., 2015). Plastic gets degraded in course of time by physical and biological processes. Plastic particles that are synthesised primarily for certain purposes and those that originated from degradative process will eventually lead to marine water with runoff, which is the main source of plastic in marine water (Andrady, 2011). So, most of the focus was on marine plastic pollution but terrestrial environment is also a major recipient of plastic pollutants (Khalid et al., 2020; Zhou et al., 2021). Plastic pollution generates particles of various sizes, ranging from micro to nano size in course of time.
Soil is the source of many plastic particles in terrestrial ecosystem, which hints us of the safety of agricultural crops and food. Soil polluted with various plastic particles like micro and nanoplastics will affect plants negatively or positively (de Souza Machado et al., 2018). Micro and nanoplastic pollution is widespread. Microplastics (MPs) are found in various terrestrial systems like agricultural fields and industrial areas (Piehl et al., 2018; Fuller & Gautam, 2016). Microplastics are becoming a threat to terrestrial ecosystem (de souza Machado et al., 2018). They alter the soil structure and plant performance (de souza Machado et al., 2019). They change the physical and chemical properties of the soil and affect microbial activities and plant performance (Xu et al., 2020). These effects are dependent on their shape and size rather than on their chemical composition (Rillig & Lehmann, 2020). Khalid et al. (2020), have described various direct and indirect ways of microplastic effects on terrestrial plants. Microplastics show direct effects on plant growth and functionalities; and indirectly affect plants by altering soil properties and soil-microbe interactions. Zhou et al. (2021) have focussed on the effects of microplastics on soil properties and microbial communities. Microplastics are found to alter soil properties like aggregation (Rillig et al., 2017) and soil pH (Boots et al., 2019). Because of the altered properties, plant performance would be affected.
Various kinds of microplastics have shown variable effects on plant growth, as noticed in Triticum aestivum, Allium fistulosum, Lactuca sativa and Phaseolus vulgaris, under different conditions (de Souza Machado et al., 2019; Jiang et al., 2019; Meng et al., 2021; Qi et al., 2018).
Nanoparticles are inherently difficult to detect and analyse as the minimum size limits of most of the commonly used sampling and analysis techniques fall in the range of 10–100 μm (Li et al., 2020c, 2021a; vanWeert et al., 2019). This creates the additional challenge of detecting and tracing nanoplastics in human food items and animal feeds. The nanoplastics should therefore be evaluated for presence in food production chain, mode of entry of the plastic into the production chain, and effect of the particles on production process and potential risks that may develop from it (Yin et al., 2021; Mateos-Cárdenas et al., 2021). Various effects of MPs and NPs on agro-ecosystems were reported by Ng et al. (2018). As plants are primary producers and main living constituents of the environment, much attention is needed to understand the effects of micro plastics (MPs) and nanoplastics (NPs) on them.
2 Types of Plastics
There are various classes of plastic depending on their physicochemical properties, namely, polyethylene terephthalate (PET), high-density polyethylene (HDPE), low-density polyethylene (LDPE), Polystyrene (PS), polypropylene (PP), polyvinyl chloride (PVC) and others (Fig. 11.1). These are produced to a great extent and commonly found in the environment. Plastic span over different shapes and sizes, Microplastics (MPs) are those plastic particles ranging from 1 to 5 mm (Frias & Nash, 2019) and particles ranging from 1 to 100 nm that exhibit colloidal behaviour are Nanoplastics (NP) (Gigault et al., 2018). Because of their extensive usage, plastic particles are widespread across the world. They have many effects on living beings, including human beings.
Different types of MPs have been detected in the environment (Mintenig et al., 2019; Sharma & Chatterjee, 2017) these plastic particles cause serious environmental problems. Primary microplastics are those that are synthesised for some purposes like fabrication, as cosmetics and in the form of microbeads. Secondary microplastics emerge from the fragmentation of larger plastics by physical, chemical and biological processes (Duis & Coors, 2016; De Falco et al., 2019). These microplastics are of various shapes, like beads, fibres, films and fragments, which are carriers of various toxic substances (Wang et al., 2018). Depending on their shape, they show various effects on plants, directly or indirectly (Rillig et al., 2019).
2.1 Sources of MPs and NPs
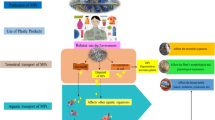
Microplastics/Nanoplastics enter into plants from the soil, which acts as the medium of transfer. An ample focus was drawn to microplastics pollution because of the reports that stated much of microplastics that were released were from personal care products (Duis & Coors, 2016). Soil polluted with microplastics will affect plant performance (de Souza Machado et al., 2019). Main sources of microplastics in soil arise from municipal sludge, irrigation water and plastic mulching. Plastic used in agriculture (agroplastic), is considered to be an important source of plastic in terrestrial ecosystem, whose usage is expected to increase 69% from 2012 to 2019 (Sintim & Flury, 2017). Wastewater treatment plants remove waste from water and prevent them from entering water sources. The remaining sludge is used as fertiliser in agricultural fields, which is the main source of plastic particles in agricultural fields (Alimi et al., 2018; Hurley & Nizzetto, 2018). Corradini et al. (2019) have emphasised on the role of municipal sludge in contaminating agricultural soil with plastic particles. While wastewater treatment methods can remove up to 90% of the micro and nanoplastics that exist in the wastewater, this amount is not satisfactory and the use of this treated water in agriculture for irrigation has effectively introduced micro and nanoplastic from the water into the agricultural ecosystem (Fig. 11.2).
Increasing microplastics are an alarm to indicate the threat imposed on environment. Leaching of plasticisers and polymers from municipal waste disposal sites into ground water impose threat to terrestrial ecosystem (Teuten et al., 2009). Huang et al. (2020) have described plastic mulching as the source of microplastics in the soil. They are also commonly found in the aquatic ecosystem like oceans, lakes and estuaries, which have been accumulating over last four decades (Thompson et al., 2004). Runoff and weathering are the main sources of microplastics in marine waters (Andrady, 2011).
There are various sources, through which NPs enter the terrestrial environment. Nanoplastic sources can be broadly classified as point sources and non-point sources. Point sources include sources from which free nanoparticles are directly dispersed into the surrounding environment. This includes direct release of nanoplastics into the environment as a result of untreated release and undesired leakage or spill out during the production and transport of nanoplastics; and products that use micro and nanoplastics (Yano et al., 2021). The advancing trend in the consumer industry (Yoshino et al., 2012) to lean towards nanoparticles has resulted in the production of nano particulate polymers which can add to the pool of nanoplastic in the environment thus acting as non-point sources of nanoplastic pollution into the environment.
2.2 Accumulation of MPs and NPs in Plants
MPs and NPs can adsorb on the surface of the plants and enter into plant system. Some of these particles are usually trapped on the root surface and enter root cells (Li et al., 2019). Various fluorescently labelled tags were used to track their transport, NPs and micro-sized MPs were found to travel from root to leaf (Li et al., 2020a, b). They travel to the upper parts of the plant by transpirational pull (Li et al., 2020a). Depending on their size and shape, these MPs and NPs can affect plants. They may have a positive or negative effect on plants depending on the species (Rillig et al., 2019). Vascular plant tissues have been observed to adsorb nanoplastics under specific conditions. Studies using scanning electron microscopy (SEM) and laser scanning confocal microscopy (LSCM) have observed the location specificity and internalisation process of nanoplastics in plants (Khalid et al., 2020; Lian et al., 2020; Yan et al., 2021a). These studies have shown that absorption of nanoplastics depends on the plant species and characteristic properties of the plastic.
Positively and negatively charged NPs accumulated differently in A. thaliana (Sun et al., 2020). PS beads have been found to accumulate on the root surface but are not absorbed in floating plants like Spirodela polyrhiza (Mateos-Cárdenas et al., 2019). MPs were detected on seagrass and macroalgae by Seng et al. (2020), these macrophytes act as glue for various plastic particles and serve as temporary sink for MPs (Sfriso et al., 2021), which hold MPs in aquatic environment. Fibres are more predominant in MPs, retained by filamentous algae (Esiukova et al., 2021). Internalisation of PS microbeads was studied using fluorescently labelled particles. These MPs (80 and 1 μm) are being absorbed by roots and translocated to the aerial parts through the vascular system in rice. This study highlights the accumulation of MPs in crop plants and their probable transfer in food chain (Liu et al., 2022; Li et al., 2020c). Physical deformations or damages in the plant roots can provide pathways for the entry of plant nanoparticles into the plant. A study conducted by Li et al. (2020a) revealed that cracks in the lateral roots of lettuce (Lactuca sativa) and wheat (Triticum aestivum) provided a pathway for the entry of nanoparticles of PMMA (polymethylmethacrylate) and PS (polystyrene). Another pathway that can lead to nanoplastic internalisation is the endocytosis through a liquid phase. Zhou et al. (2021) showed that aquaporins can assist in the nanoplastic intake.
Airborne MPs are gaining attention for being a threat to human health (Cox et al., 2019). Terrestrial plants act as a sink for various particulate matter, including MPs (Rindy et al., 2019). Depending on the leaf trait (leaf area and hairiness), particulate matter is being deposited on the urban trees, which are usually considered to reduce the pollutants in the air (Chiam et al., 2019). So, terrestrial plants act as an important sink for MPs and influence their accumulation in plants and soil (Bi et al., 2020). Airborne transport and accumulation of NPs have also been demonstrated in Zea mays L. by Sun et al. (2021). They exposed leaf of Zea mays L. seedlings to both positively and negatively charged PS particles. Positively charged PS particles aggregation was more notable and travelled from leaf to the roots through vascular bundle, which induced inhibitory effect on photosynthesis. MPs in combination with other chemical compounds have more adverse effects on plants (Prata et al., 2018). MPs/NPs in the soil serve as reservoirs of many pollutants. Cyanotoxins (CTX) released by bacteria would accumulate on MPs/NPs and serve as co-contaminants. Maity et al. (2021) have presumed the importance of these co-contaminants as phytotoxic. MPs, especially the abundant polystyrene (PS) was found to be accumulated in plants using fluorescent dyes by confocal laser scanning microscope (Li et al., 2020b). These MPs and NPs accumulate in plants by various ways and pose health risks to humans. A study on risk assessment of MPs on edible fruits and vegetables was carried out by Conti et al. (2020). Apple and carrot are the most contaminated fruit and vegetable respectively. There are various studies that concentrated on effects of MPs and NPs on plant performance, like seed germination, photosynthesis, growth and others. The present chapter explains the effect of these MPs/NPs on plant functionalities.
3 Effects of MPs and NPs on Plant Functionalities
MPs/NPs affect plants negatively, by reducing growth and photosynthetic activity in terrestrial and aquatic primary producers. Majority of MPs/NPs induce oxidative stress, thereby increasing anti-oxidative enzyme levels. Various effects of MPs/NPs include inhibition of seed germination, reduced or inhibited photosynthetic activity, reduction of root and shoot lengths, plant biomass and others (Fig. 11.3). The direct and indirect effects of MPs/NPs on plants based on the published literature are discussed below:
-
(i)
Effects of MPs and NPs on Seed Germination:
MPs and NPs have shown to affect seed germination in various plant species. These effects are dependent on their size and concentration (De Silva et al., 2021) as shown in Table 11.1. Soil is the main sink for these particles. MPs (PLA, polylactic acid) accumulated in soil and affected germination in L. perenne (Boots et al., 2019). Many of the studies report that MPs adsorb on the plant surface, whereas NPs are being absorbed into the plants. The accumulated particles on the seed surface reduce the germination rate (De Silva et al., 2021). Recent studies have highlighted that seed germination and growth of plants are dependent on the age of decaying plastic. Based on the age of microplastic, its toxicity also varied on the germination, growth and photosynthesis in L. Sativum (Pflugmacher et al., 2021). Bosker et al. (2019) studied the effects of differently sized MPs (50, 500 and 4800 nm) of different concentrations on seed germination of L. sativum, using 72 h germination assay. Germination was reduced due to the blockage of the seed capsule by accumulated MPs. The impact of PS-MPs (2 μm and 80 nm) on germination of ornamental plants was studied by Guo et al. (2022). It was found that these MPs could inhibit germination in these species depending on their concentration (Table 11.1). Zhang et al. (2021b) studied the effect of PS-MPs (200 nm) on the seed germination of rice. These particles showed no significant effect on seed germination but reduced the activity of anti-oxidative enzymes. Reactive oxygen species (ROS) levels increased in roots and transcriptome analysis revealed that PS-MPs increased the activity of anti-oxidative enzymes in roots.
The composition of MPs also affects the toxicity. Various MPs like PP, PE and PVC were analysed on different growth parameters like seed germination, plant height, biomass production and oxidative stress levels in Lepidium sativum, by Pignattelli et al. (2020). The evaluated toxicity was found to be due to PVC on L. Sativum. These particles alone or in combination with other toxic elements will affect the plant performance, as witnessed in L. sativum. PET alone or with acid rain was found to negatively affect seed germination, plant growth and increased chlb production (Pignattelli et al., 2021). Ethylene vinyl acetate (EVA), LLDPE (Linear low-density polyethylene) and poly methyl methacrylate (PMMA) have shown to inhibit seed germination in Triticum aestivum, at lower concentrations (Lian et al., 2019). MPs derived from fertilisers have exhibited differential effects on wheat growth and soil properties (Lian et al., 2021b).
A systemic study done on the effect of polystyrene nanoplastics (PSNPs) on crop plant Triticum aestivum L. (Lian et al., 2020) and on Allium cepa (Giorgetti et al., 2020), has shown that PSNPs exhibited no visible effect on seed germination. But there was an increase in root length and decrease in shoot-to-root biomass ratio in T.aestivum. PSNPs’ effect on seed germination is species dependent. Among the four food crops (Italian lettuce, radish, wheat and corn) exposed to PSNPs and microspheres (micro PS), seed germination in Italian lettuce was inhibited at a very early stage of development. It was also observed that these toxic effects are because of the oxidative stress imposed on plants (Gong et al., 2021). A pot experiment conducted by Li et al. (2021a) on effects of LDPE and bio mulch on germination and growth of Glycine max has shown that bio-mulch has negatively affected germination viability and LDPE affected plant height.
-
(ii)
Effects of Accumulated MPs and NPs on Plant Growth:
MPs and NPs, when accumulated in plants, affect their growth directly in various ways, like reduction in biomass and by induction of oxidative stress. These plastic pollutants are ubiquitous; they are toxic alone or in combination with other organic or metallic pollutants. They even affect plant performance indirectly by altering soil properties like soil pH or soil microbial community (Rillig et al., 2019). Foliar application of PSNPs have shown to reduce the growth and induce oxidative stress in lettuce. These NPs got absorbed at the leaf and transported to the root part. They reduced nutritional quality, biomass and leaf size of lettuce (Lian et al., 2021a).
-
(a)
Direct Effects of MPs and NPs on Plant Growth:
Much of the scientific attention is being driven by these pollutants and their effects on agricultural crops, which were studied using hydroponic cultures in certain crop plants. Accumulation of MPs (PE microbeads) in the rhizosphere and reduction in water and nutrient uptake were observed in hydroponic maize. Upward transport of PE beads to shoot was not observed (Urbina et al., 2020). In hydroponic wheat seedlings, there was no effect of PSMPs on photosynthesis and oxidative stress. Instead, PSMPs reduced the toxicity of cadmium and copper on wheat seedlings (Zong et al., 2021). Rice (Oryza sativa) seedlings were exposed to 1 μm and 100 nm PSMPs, to study their toxic effects in hydroponic conditions. 1 μm PS were more toxic than 100 nm particles in rice seedlings (Wu et al., 2021). Differently sized particles would affect plants differently. MPs of 1 μm size accumulated in the intercellular space of carrot roots. But larger MPs would enter roots and leaves in the presence of arsenic. They reduced the quality of carrots and eventually led to health risks (Dong et al., 2021b).
Molecular and physiological effects of differently sized MPs were studied in maize seedlings by Pehlivan and Gedik (2021). They analysed various combinations of MPs differing in their sizes, for their toxicity. They induced xenobiotic stress in the maize. It was observed that the bigger the particle size, the quicker they restore to the normal condition. Their research insists on the toxicity of pollutants on agricultural crops. Likewise, the toxicity of MPs on the leguminous plant soybean was also investigated by Xu et al. (2021). PSMPs decreased the availability of organic pollutants like phenanthrene in soybean seedlings. It was shown that PSMPs along with phenanthrene are harmful to higher plants. Ren et al. (2021) studied the effect of PS beads on the growth of Chinese cabbage. They found that these plastic particles have soil-microbial community that would affect the plant growth.
Nanoplastics can get internalised in plants and affect them adversely. Exposure of corn to PSNPs has altered the plant’s metabolic rates because of the elevated anti-oxidative enzymes. But photosynthetic activity was not altered (Zhang et al., 2021a). The effect of PSNPs was also studied in cucumber by exposing it to different sized particles. It was found that they first accumulated at root but later transported to fruit, flower and leaves. They increased the protein content of fruit and also root activity (Li et al., 2021b). Differently charged nanoplastics accumulated differentially in Arabidopsis thaliana. Positively charged particles, though accumulated slowly, could reduce the growth of the plant. Negatively charged particles were found in apoplast and xylem (Sun et al., 2020). Giorgetti et al. (2020) studied the interaction of PSNPs and Allium cepa. Various concentrations of PSNPs (0.01, 0.1 and 1 mg/L) were used during germination. All concentrations were shown to inhibit root length. Higher concentrations were found to induce oxidative stress. The internalisation and movement of particles was visualised using TEM. Internalised nanoplastic particles in food crops raise concerns for food safety, as they are transferred in trophic levels. Foliar application of positively and negatively charged PSNPs on Zeamays L. was carried out to observe the absorption tendency of air transported NPs carrying different charges. They got internalised and transported to the root leading to photosynthesis inhibition by PS-NH2 (Sun et al., 2021).
In higher plants like Vicia faba, the root tips were exposed to 5 and 100 nm PSMPs of various concentrations. It was observed that 100-nm particles accumulated in roots and blocked intercellular connections. There was a decrease in the growth with 100-nm particles, which have also proven to be genotoxic and imposed oxidative damage (Jiang et al., 2019). In lettuce, MP (PS) under di-n-butyl phthalate (DBP) stress conditions induced toxicity by blocking root pores. PS in lettuce decreased biomass by inducing oxidative stress and damaging leaves and root. (Gao et al., 2021a). Similar research was carried to study the effect of PS and DBP on green and purple lettuce. Toxicity induced was dependent on the variety; purple variety was more sensitive to PS. The toxicity induced was by reducing growth of root, soluble protein and sugar in leaves (Gao et al., 2021b). PE MPs effect on cadmium (Cd) bioavailability was also investigated in lettuce by Wang et al. (2021). They increased the availability of Cd and accumulation in plants. This study suggests that MPs increase the toxicity of heavy metals in contaminated soils. Toxicological effects of MPs on farmland were researched on crops like lettuce. Physiological responses of lettuce in presence of PVC microplastics were studied by applying different sizes of PVC (100 nm–18 μm and 18–150 μm). Various contents were analysed on the root activity, which had no effect. But, root length, diameter and surface were increased. Photosynthetic activity was altered with 100 nm–18 μm particles and 18–150 μm sized particles were correlated to root morphology (Li et al., 2020b).
-
(b)
Indirect Effects of MPs on Plants:
Microplastics show altered effects on various plants indirectly by changing soil structure, properties or soil microbial interaction. Boots et al. (2019) have reported the change in soil structure in the presence of HDPE and PLA, which results in altered soil ecosystem. Different MPs show different effects on soil property and plant biomass, based on their concentration (Lozano et al., 2021). These altered structure and physicochemical properties of soil will affect the plant functionalities. de souza Machado et al. (2019) conducted an experiment to study the effects of six different MPs on Allium fistulosum. Depending on the type and size, they altered the soil properties, which affected the plant’s performance. MPs get integrated into the soil when they reach the soil surface (Rillig et al., 2017). They contain a lot of organic carbon, so they add to the organic part of the soil, which will affect the carbon cycle in the soil and the plant’s performance (Rillig et al., 2021). Hydroponic cultures of maize were employed to study the effect of MPs. Isotopic analysis was used to study the effects of MPs and it was noticed that 30% of the carbon in the rhizosphere was derived from microplastic (Urbina et al., 2020). Biodegradable plastic is being employed in the place of plastic mulching, which releases various compounds on degradation. An in vitro toxicity test was conducted to study the effects of biodegradable plastic on crop plants. Among the compounds released from bioplastic, adipic acid was shown to inhibit growth of tomato and lettuce (Martin-Closas et al., 2014). The influence of biodegradable plastics on soil microbial communities and agro-ecosystem was reported by Bandopadhyay et al. (2018).
-
(iii)
Effects of MPs on Plant Community:
The effects of microplastics are mainly dealt with in an individual or in plant species. But the effects of microplastics vary with plant species within a community. MPs have been found to show allelopathic effects, which affect plant community structure. In allelopathy, they promote the growth of same species and inhibit the same of neighbour. Lozano and Rillig (2020) have demonstrated this in Hieracium and its neighbour Festuca. They affect the community structure and plant productivity (Rillig et al., 2019), which is witnessed in root length of Plantago lanceolata (forb) and A. fistulosum. They have shown opposite responses to microplastic exposure (de Souza machado et al., 2019; Van Kleunen et al., 2020). So, this indicates that microplastics affect varyingly on different plant species within a community. Yu et al. (2021b), highlighted the importance of research on the effects of microplastics on plant biomass and community structure. Lozano and Rillig (2020) have shown the effect of microfibres on productivity and structure of plant community. They witnessed the dominance of an allelopathic species in the presence of microplastics and reduced biomass of the other species in a community.
-
(iv)
Effects of MPs and NPs on Growth of Aquatic Photosynthetic Organisms:
The aquatic system receives much of plastic particles by runoff. They adversely affect many aquatic plants in various ways. The toxicity exhibited by these plastic particles is dependent on their properties like charge, type and size (Lagarde et al., 2016). Charge dependent toxicity was witnessed in microalgae, Dunaliella tertiolecta. Positively charged nano-PS particles have inhibited the growth when compared to negatively charged PS particles (Bergami et al., 2017). Schiavo et al. (2021) found the toxic effects of PS, PP and PE leachates on growth and DNA of microalgae. As the macrophytes exposed to various concentrations of MPs and NPs, NPs were found to be more effective in reducing shoot to root length compared to Mps (vanWeert et al., 2019). In a comparative study of the effects of MPs and NPs, PSNPs were found to impose a more inhibitory effect on the growth of Chlamydomonas reinhardtii. The treated algal species was shown to have increased levels of reactive oxygen species and malonaldehyde. Internalised NPs were visualised in vacuoles. They mainly imposed oxidative stress in the algae (Yan et al., 2021b). Yang et al. (2020) proved the toxic effects of MPs (PS, PE, PA) on growth due to oxidative stress in Chlorella pyrenoidosa. Similar effect of PSMPs was observed on the growth of Chlorella pyrenoidosa; there was reduction in the growth due to oxidative stress and physical damage. Later, it could regain its growth in late logarithmic phase (Mao et al., 2018). In the floating plant Salvinia cucullata, the combined effect of MPs and herbicide (glyphosate) has reduced the growth by activating antioxidative enzymes. PSMP (1 μm) could reduce relative growth and root morphology. These findings indicate the toxic effects of contaminants in aquatic ecosystems (Yu et al., 2021a). A growth inhibition rate of 39.7% was also observed in marine microalgae Skeletonema costatum, on 96 h exposure to MPs. The toxicity was dependent on particle size and concentration of MPs on interaction with microalgae (Zhang et al., 2017). The toxicity of MP in combination with Cu nanoparticles and triclosan was also studied in Skeletonema costatum (Zhu et al., 2019; Zhu et al., 2020), shown in Table 11.2. Chlamydomonas rheinhardtii was also affected by an inhibition rate of 45.8%, on exposure to PSMPs (Li et al., 2020d). Kalčíková et al. (2017) studied the effect of PE microbeads on freshwater duckweed, Lemna minor. It was observed that these particles reduced the root length, but the photosynthetic activity was not affected (Table 11.2).
-
(v)
Effects of MPs and NPs on Photosynthetic Activity of Plants:
Photosynthetic organisms are present in aquatic and terrestrial systems. They are the primary producers and sinks for CO2. MPs and NPs have shown to affect photosynthesis by altering chlorophyll content in photosynthetic organisms (Table 11.3). Photosynthetic activity was reduced in cucumber plant when it was exposed to various sizes of PSNPs (100, 300, 500 and 700 nm). Among them 100 nm particles reduced chlorophyll-a and chlorophyll-b content, along with fluorescence in leaves. Exposure to 300 nm particles reduced biomass, while 700 nm particles induced oxidative stress in leaves (Li et al., 2020a). An antagonistic response was observed in wheat seedlings on exposure to PSNPs. The photosynthetic activity was increased at 0.1 mg/L concentration and shoot-to-root ratio was decreased at 0.01 mg/L concentration (Lian et al., 2020).
MPs act as carriers of many pollutants and exert their effects on crop plants. Effects of MPs in combination of other pollutants were being investigated in some of the researches. Growth and photosynthetic activity were reduced in lettuce, on exposure to MP (PE) and di-n-butyl phthalate (DBP). Exogenous MP has further increased the effect of DBP on photosynthesis (Gao et al., 2019).The integrated effect of PS and di-butyl phthalate (DBP) on photosynthesis of red lettuce were studied Dong et al. (2021a). It was noted that PS particles reduced the uptake of DBP; jointly, they could reduce photosynthetic activity and also the quality of red lettuce. This explains the possible risk of microplastics on vegetable crops.
-
(a)
Effects of MPs and NPs on Photosynthesis of Aquatic Photosynthetic Organisms:
Microalgae are primary producers in the aquatic ecosystem; study of plastic pollutants on these organisms will give us an insight into how MPs/NPs would affect photosynthetic process (Table 11.3). Small size and positive surface charge on MPs will have more adverse effects on microalgae (Prata et al., 2019). Small-sized PSMPs (0.05 μm) were proven to have adverse effects at certain concentrations, on microalgae by inhibiting growth, but photosynthesis was not affected (Sjollema et al., 2016). MPs have proven to be toxic to the freshwater algae by negatively affecting the photosynthesis, which was dependent on the size, in a study dealing with the interaction of MPs and microalgae Skeletonema costatum (Zhang et al., 2017). Toxicity of PVC was greater than PP at certain concentrations by affecting chlorophyll-a content in Chlorella pyrenoidosa and Microcystis (M.) flosaquae. These results indicate the risk of MPs on growth of algae (Wu et al., 2019). Similar effects of PSMPs were observed in Chlamydomonas rheinhardtii, in all different concentrations tested (Li et al., 2020d). It could recover from the toxic effect of MPs thereafter. The effects of MPs on the entire growth cycle of Chlorella pyrenoidosa was studied by Mao et al. (2018). It was observed that the photosynthetic activity was decreased initially but later, after logarithmic phase, an increase in the photosynthetic activity was observed. MPs/NPs do not necessarily reduce photosynthetic activity in all, some plants like Spirodela polyrhiza are not affected by plastic (Dovidat et al., 2020). PE MPs adhered to duckweed species, Lemma minor have imposed no effect on photosynthesis and growth of the weed (Mateos-Cárdenas et al., 2019). Reduced chla and growth were noticed in case of Chlorella vulgaris on exposure to PSMP and metals (Cu, Zn and Mn). Combined effect showed more inhibition on growth and chla content (Tunali et al., 2020). Long-term exposure of differently sized PSMP and PSNP on Chlorella vulgaris were proven to reduce cell viability, chla and also induced stress. PS particles of varying sizes 20, 50 and 500 nm were used along with PS-COOH. Smaller-sized particles declined the amount of chla and cell viability. 20 nm particles induced stress and small-sized PSNPs were responsible for cell wall damage, which was visualised by SEM and TEM (Hazeem et al., 2020). A similar study was conducted by Khoshnamvand et al. (2021) using PSNP (PS-NH2) with diameter ranging 90, 200 and 300 nm. Diameters of 90 and 200 nm decreased chla and algal biomass. These studies convey the need to focus on the adverse effects of various plastic particles on the photosynthesis of phytoplankton, which is the primary producer and major source of oxygen in aquatic ecosystems.
Yang et al. (2021) studied the interaction between nanoplastics (80 nm) and Chlorella pyrenoidosa. They found the inhibitory effect of nanoplastics was greater than microplastics. Nanoplastics inhibited the growth by blocking gene expression of aminoacyl t-RNA synthetase enzyme at low concentrations. At high concentrations, photosynthesis was affected. The combined effect of PSNP (nPS-NH2) and glyphosate was studied in blue-green algae, Mycrocystis aeruginosa. PSNP at 5 mg/L was not as toxic as glyphosate at the same concentration. But after 96 h of exposure, chla content decreased, indicating a decrease in photosynthetic activity. The integrated effect on growth indicated an antagonistic effect of PS-NH2 and glyphosate (Zhang et al., 2018). The most widespread photosynthetic organism, Prochlorococcus, was used to study the effect of plastic leachates, HDPE and PVC. A rapid decline in the photosynthetic activity was observed at various concentrations used. Growth and oxygen production were also hindered (Tetu et al., 2019). In another research conducted by Sarker et al. (2020) on the impact of weathering on toxicity of these leachates, it was observed that toxicity declined gradually on weathering. But leachates produced even after weathering had shown to affect the growth and photosynthesis negatively. The toxicity of weathered and unweathered leachates varied on two strains of prochlorococcus tested. Zinc, which is the common additive in many plastics, was found to leach after 112 days of its entry into the environment. The impact of varying concentrations of zinc on growth of Prochlorococuus and Synechococcus was also focussed on (Sarker et al., 2021).
4 Conclusion
Plants, both terrestrial and aquatic, are the primary producers and act as sinks for these plastic particles, which in turn are severely affected by them. The outcomes of the exposure of Micro and nanoplastics on photosynthetic autotrophs are focussed on in the present chapter. Most of the research claimed the toxic effects of MPs/NPs on plants. Their toxicity is dependent on the type, size and concentration being employed. MPs/NPs exhibit direct effects by altering plant growth or indirectly by altering soil properties or soil microbial interaction, which will impair plant performance. The positive impact of these particles on plant growth is also witnessed in certain cases. Phytoplankton, which is an important primary producer and source of oxygen, is also negatively affected by the MPs/NPs that get accumulated in aquatic ecosystems in various ways. The effects of weathering on toxicity of plastic leachates on phytoplankton are also being investigated. MPs/NPs act as carriers of many toxic compounds that exhibit harmful effects on plants. The researches focussed on the effects of micro and nanoplastics on agricultural crops recommend focusing on safety of food and human health, as the internalised particles might get transferred at trophic levels. So, there is a need to encourage and focus our research on the impact of plastic pollution on plant functionalities.
References
Alimi, O. S., FarnerBudarz, J., Hernandez, L. M., & Tufenkji, N. (2018). Microplastics and nanoplastics in aquatic environments: Aggregation, deposition, and enhanced contaminant transport. Environmental Science & Technology, 52(4), 1704–1724.
Alomar, C., Estarellas, F., & Deudero, S. (2016). Microplastics in the Mediterranean Sea: Deposition in coastal shallow sediments, spatial variation and preferential grain size. Marine Environmental Research, 115, 1–10.
Andrady, A. L. (2011). Microplastics in the marine environment. Marine Pollution Bulletin, 62(8), 1596–1605.
Bandopadhyay, S., Martin-Closas, L., Pelacho, A. M., & DeBruyn, J. M. (2018). Biodegradable plastic mulch films: Impacts on soil microbial communities and ecosystem functions. Frontiers in Microbiology, 9, 819.
Bergami, E., Pugnalini, S., Vannuccini, M. L., Manfra, L., Faleri, C., Savorelli, F., Dawson, K. A., & Corsi, I. (2017). Long-term toxicity of surface-charged polystyrene nanoplastics to marine planktonic species Dunaliellatertiolecta and Artemiafranciscana. Aquatic Toxicology, 189, 159–169.
Bi, M., He, Q., & Chen, Y. (2020). What roles are terrestrial plants playing in global microplastic cycling? (Vol. 54, pp. 5325–5327).
Boots, B., Russell, C. W., & Green, D. S. (2019). Effects of microplastics in soil ecosystems: Above and below ground. Environmental Science & Technology, 53(19), 11496–11506.
Bosker, T., Bouwman, L. J., Brun, N. R., Behrens, P., & Vijver, M. G. (2019). Microplastics accumulate on pores in seed capsule and delay germination and root growth of the terrestrial vascular plant Lepidiumsativum. Chemosphere, 226, 774–781.
Boucher, J., & Friot, D. (2017). Primary microplastics in the oceans: A global evaluation of sources (Vol. 43). IUCN.
Browne, M. A., Crump, P., Niven, S. J., Teuten, E., Tonkin, A., Galloway, T., & Thompson, R. (2011). Accumulation of microplastic on shorelines woldwide: Sources and sinks. Environmental Science & Technology, 45(21), 9175–9179.
Chiam, Z., Song, X. P., Lai, H. R., & Tan, H. T. W. (2019). Particulate matter mitigation via plants: Understanding complex relationships with leaf traits. Science of the Total Environment, 688, 398–408.
Conti, G. O., Ferrante, M., Banni, M., Favara, C., Nicolosi, I., Cristaldi, A., Fiore, M., & Zuccarello, P. (2020). Micro-and nano-plastics in edible fruit and vegetables. The first diet risks assessment for the general population. Environmental Research, 187, 109677.
Corradini, F., Meza, P., Eguiluz, R., Casado, F., Huerta-Lwanga, E., & Geissen, V. (2019). Evidence of microplastic accumulation in agricultural soils from sewage sludge disposal. Science of the Total Environment, 671, 411–420.
Cox, K. D., Covernton, G. A., Davies, H. L., Dower, J. F., Juanes, F., & Dudas, S. E. (2019). Human consumption of microplastics. Environmental Science & Technology, 53(12), 7068–7074.
De Falco, F., Di Pace, E., Cocca, M., & Avella, M. (2019). The contribution of washing processes of synthetic clothes to microplastic pollution. Scientific Reports, 9(1), 1–11.
De Silva, Y. S. K., Rajagopalan, U. M., & Kadono, H. (2021). Microplastics on the growth of plants and seed germination in aquatic and terrestrial ecosystems. Global Journal of Environmental Science and Management, 7, 347–368.
de Souza Machado, A. A., Kloas, W., Zarfl, C., Hempel, S., & Rillig, M. C. (2018). Microplastics as an emerging threat to terrestrial ecosystems. Global Change Biology, 24(4), 1405–1416.
de Souza Machado, A. A., Lau, C. W., Kloas, W., Bergmann, J., Bachelier, J. B., Faltin, E., Becker, R., Görlich, A. S., & Rillig, M. C. (2019). Microplastics can change soil properties and affect plant performance. Environmental Science & Technology, 53(10), 6044–6052.
Dong, Y., Gao, M., Qiu, W., & Song, Z. (2021a). Uptake of microplastics by carrots in presence of As (III): Combined toxic effects. Journal of Hazardous Materials, 411, 125055.
Dong, Y., Song, Z., Liu, Y., & Gao, M. (2021b). Polystyrene particles combined with di-butyl phthalate cause significant decrease in photosynthesis and red lettuce quality. Environmental Pollution, 278, 116871.
Dovidat, L. C., Brinkmann, B. W., Vijver, M. G., & Bosker, T. (2020). Plastic particles adsorb to the roots of freshwater vascular plant Spirodelapolyrhiza but do not impair growth. Limnology and Oceanography Letters, 5(1), 37–45.
Duis, K., & Coors, A. (2016). Microplastics in the aquatic and terrestrial environment: Sources (with a specific focus on personal care products), fate and effects. Environmental Sciences Europe, 28(1), 1–25.
Esiukova, E. E., Lobchuk, O. I., Volodina, A. A., & Chubarenko, I. P. (2021). Marine macrophytes retain microplastics. Marine Pollution Bulletin, 171, 112738.
Europe, P. (2017). Plastics-the facts 2017 an analysis of European plastics production, demand and waste data. Available on the website: http://www.plasticseurope.org
Frias, J. P. G. L., & Nash, R. (2019). Microplastics: Finding a consensus on the definition. Marine Pollution Bulletin, 138, 145–147.
Fuller, S., & Gautam, A. (2016). A procedure for measuring microplastics using pressurized fluid extraction. Environmental Science & Technology, 50(11), 5774–5780.
Gao, M., Liu, Y., & Song, Z. (2019). Effects of polyethylene microplastic on the phytotoxicity of di-n-butyl phthalate in lettuce (Lactucasativa L. var. ramosaHort). Chemosphere, 237, 124482.
Gao, M., Liu, Y., Dong, Y., & Song, Z. (2021a). Effect of polyethylene particles on dibutyl phthalate toxicity in lettuce (Lactucasativa L.). Journal of Hazardous Materials, 401, 123422.
Gao, M., Xu, Y., Liu, Y., Wang, S., Wang, C., Dong, Y., & Song, Z. (2021b). Effect of polystyrene on di-butyl phthalate (DBP) bioavailability and DBP-induced phytotoxicity in lettuce. Environmental Pollution, 268, 115870.
Geyer, R., Jambeck, J. R., & Law, K. L. (2017). Production, use, and fate of all plastics ever made. Science Advances, 3(7), e1700782.
Gigault, J., Ter Halle, A., Baudrimont, M., Pascal, P. Y., Gauffre, F., Phi, T. L., El Hadri, H., Grassl, B., & Reynaud, S. (2018). Current opinion: What is a nanoplastic? Environmental Pollution, 235, 1030–1034.
Giorgetti, L., Spanò, C., Muccifora, S., Bottega, S., Barbieri, F., Bellani, L., & Castiglione, M. R. (2020). Exploring the interaction between polystyrene nanoplastics and Allium cepa during germination: Internalization in root cells, induction of toxicity and oxidative stress. Plant Physiology and Biochemistry, 149, 170–177.
Gong, W., Zhang, W., Jiang, M., Li, S., Liang, G., Bu, Q., Xu, L., Zhu, H., & Lu, A. (2021). Species-dependent response of food crops to polystyrene nanoplastics and microplastics. Science of the Total Environment, 796, 148750.
Guo, M., Zhao, F., Tian, L., Ni, K., Lu, Y., & Borah, P. (2022). Effects of polystyrene microplastics on the seed germination of herbaceous ornamental plants. Science of the Total Environment, 809, 151100.
Hazeem, L. J., Yesilay, G., Bououdina, M., Perna, S., Cetin, D., Suludere, Z., Barras, A., & Boukherroub, R. (2020). Investigation of the toxic effects of different polystyrene micro-and nanoplastics on microalgae Chlorella vulgaris by analysis of cell viability, pigment content, oxidative stress and ultrastructural changes. Marine Pollution Bulletin, 156, 111278.
Huang, Y., Liu, Q., Jia, W., Yan, C., & Wang, J. (2020). Agricultural plastic mulching as a source of microplastics in the terrestrial environment. Environmental Pollution, 260, 114096.
Hurley, R. R., & Nizzetto, L. (2018). Fate and occurrence of micro (nano) plastics in soils: Knowledge gaps and possible risks. Current Opinion in Environmental Science & Health, 1, 6–11.
Jiang, X., Chen, H., Liao, Y., Ye, Z., Li, M., & Klobučar, G. (2019). Ecotoxicity and genotoxicity of polystyrene microplastics on higher plant Viciafaba. Environmental Pollution, 250, 831–838.
Kalčíková, G., Gotvajn, A. Ž., Kladnik, A., & Jemec, A. (2017). Impact of polyethylene microbeads on the floating freshwater plant duckweed Lemna minor. Environmental Pollution, 230, 1108–1115.
Khalid, N., Aqeel, M., & Noman, A. (2020). Microplastics could be a threat to plants in terrestrial systems directly or indirectly. Environmental Pollution, 267, 115653.
Khoshnamvand, M., Hanachi, P., Ashtiani, S., & Walker, T. R. (2021). Toxic effects of polystyrene nanoplastics on microalgae Chlorella vulgaris: Changes in biomass, photosynthetic pigments and morphology. Chemosphere, 280, 130725.
Lagarde, F., Olivier, O., Zanella, M., Daniel, P., Hiard, S., & Caruso, A. (2016). Microplastic interactions with freshwater microalgae: Hetero-aggregation and changes in plastic density appear strongly dependent on polymer type. Environmental Pollution, 215, 331–339.
Li, L., Zhou, Q., Yin, N., Tu, C., & Luo, Y. (2019). Uptake and accumulation of microplastics in an edible plant. Chinese Science Bulletin, 64(9), 928–934.
Li, Z., Li, Q., Li, R., Zhao, Y., Geng, J., & Wang, G. (2020a). Physiological responses of lettuce (Lactucasativa L.) to microplastic pollution. Environmental Science and Pollution Research, 27, 30306–30314.
Li, Z., Li, R., Li, Q., Zhou, J., & Wang, G. (2020b). Physiological response of cucumber (Cucumissativus L.) leaves to polystyrene nanoplastics pollution. Chemosphere, 255, 127041.
Li, L., Luo, Y., Li, R., Zhou, Q., Peijnenburg, W. J., Yin, N., Yang, J., Tu, C., & Zhang, Y. (2020c). Effective uptake of submicrometre plastics by crop plants via a crack-entry mode. Nature sustainability, 3(11), 929–937.
Li, S., Wang, P., Zhang, C., Zhou, X., Yin, Z., Hu, T., Hu, D., Liu, C., & Zhu, L. (2020d). Influence of polystyrene microplastics on the growth, photosynthetic efficiency and aggregation of freshwater microalgae Chlamydomonasreinhardtii. Science of the Total Environment, 714, 136767.
Li, B., Huang, S., Wang, H., Liu, M., Xue, S., Tang, D., Cheng, W., Fan, T., & Yang, X. (2021a). Effects of plastic particles on germination and growth of soybean (Glycine max): A pot experiment under field condition. Environmental Pollution, 272, 116418.
Li, Z., Li, Q., Li, R., Zhou, J., & Wang, G. (2021b). The distribution and impact of polystyrene nanoplastics on cucumber plants. Environmental Science and Pollution Research, 28(13), 16042–16053.
Lian, J., Shen, M., & Liu, W. (2019). Effects of microplastics on wheat seed germination and seedling growth. Journal of Agro-Environment Science, 38(4), 737–745.
Lian, J., Wu, J., Xiong, H., Zeb, A., Yang, T., Su, X., Su, L., & Liu, W. (2020). Impact of polystyrene nanoplastics (PSNPs) on seed germination and seedling growth of wheat (Triticum aestivum L.). Journal of Hazardous Materials, 385, 121620.
Lian, J., Liu, W., Meng, L., Wu, J., Chao, L., Zeb, A., & Sun, Y. (2021a). Foliar-applied polystyrene nanoplastics (PSNPs) reduce the growth and nutritional quality of lettuce (Lactucasativa L.). Environmental Pollution, 280, 116978.
Lian, J., Liu, W., Meng, L., Wu, J., Zeb, A., Cheng, L., Lian, Y., & Sun, H. (2021b). Effects of microplastics derived from polymer-coated fertilizer on maize growth, rhizosphere, and soil properties. Journal of Cleaner Production, 318, 128571.
Liu, Y., Guo, R., Zhang, S., Sun, Y., & Wang, F. (2022). Uptake and translocation of nano/microplastics by rice seedlings: Evidence from a hydroponic experiment. Journal of Hazardous Materials, 421, 126700.
Lozano, Y. M., & Rillig, M. C. (2020). Effects of microplasticfibers and drought on plant communities. Environmental Science & Technology, 54(10), 6166–6173.
Lozano, Y. M., Lehnert, T., Linck, L. T., Lehmann, A., & Rillig, M. C. (2021). Microplastic shape, polymer type, and concentration affect soil properties and plant biomass. Frontiers in Plant Science, 12, 169.
Maity, S., Guchhait, R., Chatterjee, A., & Pramanick, K. (2021). Co-occurrence of co-contaminants: Cyanotoxins and microplastics, in soil system and their health impacts on plant–A comprehensive review. Science of the Total Environment, 794, 148752.
Mao, Y., Ai, H., Chen, Y., Zhang, Z., Zeng, P., Kang, L., Li, W., Gu, W., He, Q., & Li, H. (2018). Phytoplankton response to polystyrene microplastics: Perspective from an entire growth period. Chemosphere, 208, 59–68.
Martin-Closas, L., Botet, R., & Pelacho, A. M. (2014). An in vitro crop plant ecotoxicity test for agricultural bioplastic constituents. Polymer Degradation and Stability, 108, 250–256.
Mateos-Cárdenas, A., Scott, D. T., Seitmaganbetova, G., van Pelt Frank, N. A. M., & AK, J. M. (2019). Polyethylene microplastics adhere to Lemna minor (L.), yet have no effects on plant growth or feeding by Gammarusduebeni (Lillj.). Science of the Total Environment, 689, 413–421.
Mateos-Cárdenas, A., van Pelt, F. N., O’Halloran, J., & Jansen, M. A. (2021). Adsorption, uptake and toxicity of micro-and nanoplastics: Effects on terrestrial plants and aquatic macrophytes. Environmental Pollution, 284, 117183.
Meng, F., Yang, X., Riksen, M., Xu, M., & Geissen, V. (2021). Response of common bean (Phaseolus vulgaris L.) growth to soil contaminated with microplastics. Science of the Total Environment, 755, 142516.
Mintenig, S. M., Löder, M. G. J., Primpke, S., & Gerdts, G. (2019). Low numbers of microplastics detected in drinking water from ground water sources. Science of the Total Environment, 648, 631–635.
Naidoo, T., Glassom, D., & Smit, A. J. (2015). Plastic pollution in five urban estuaries of KwaZulu-Natal, South Africa. Marine pollution bulletin, 101(1), 473–480.
Ng, E. L., Lwanga, E. H., Eldridge, S. M., Johnston, P., Hu, H. W., Geissen, V., & Chen, D. (2018). An overview of microplastic and nanoplastic pollution in agroecosystems. Science of the Total Environment, 627, 1377–1388.
Pehlivan, N., & Gedik, K. (2021). Particle size-dependent biomolecular footprints of interactive microplastics in maize. Environmental Pollution, 277, 116772.
Pflugmacher, S., Tallinen, S., Kim, Y. J., Kim, S., & Esterhuizen, M. (2021). Ageing affects microplastic toxicity over time: Effects of aged polycarbonate on germination, growth, and oxidative stress of Lepidium sativum. Science of the Total Environment, 790, 148166.
Piehl, S., Leibner, A., Löder, M. G., Dris, R., Bogner, C., & Laforsch, C. (2018). Identification and quantification of macro-and microplastics on an agricultural farmland. Scientific Reports, 8(1), 1–9.
Pignattelli, S., Broccoli, A., & Renzi, M. (2020). Physiological responses of garden cress (L. sativum) to different types of microplastics. Science of The Total Environment, 727, 138609.
Pignattelli, S., Broccoli, A., Piccardo, M., Terlizzi, A., & Renzi, M. (2021). Effects of polyethylene terephthalate (PET) microplastics and acid rain on physiology and growth of Lepidium sativum. Environmental Pollution, 282, 116997.
Prata, J. C., Lavorante, B. R., Maria da Conceição, B. S. M., & Guilhermino, L. (2018). Influence of microplastics on the toxicity of the pharmaceuticals procainamide and doxycycline on the marine microalgae Tetraselmis chuii. Aquatic Toxicology, 197, 143–152.
Prata, J. C., da Costa, J. P., Lopes, I., Duarte, A. C., & Rocha-Santos, T. (2019). Effects of microplastics on microalgae populations: A critical review. Science of the Total Environment, 665, 400–405.
Qi, Y., Yang, X., Pelaez, A. M., Lwanga, E. H., Beriot, N., Gertsen, H., Garbeva, P., & Geissen, V. (2018). Macro-and micro-plastics in soil-plant system: Effects of plastic mulch film residues on wheat (Triticum aestivum) growth. Science of the Total Environment, 645, 1048–1056.
Ren, X., Tang, J., Wang, L., & Liu, Q. (2021). Microplastics in soil-plant system: Effects of nano/microplastics on plant photosynthesis, rhizosphere microbes and soil properties in soil with different residues. Plant and Soil, 462(1), 561–576.
Rillig, M. C., & Lehmann, A. (2020). Microplastic in terrestrial ecosystems. Science, 368(6498), 1430–1431.
Rillig, M. C., Ingraffia, R., & de Souza Machado, A. A. (2017). Microplastic incorporation into soil in agroecosystems. Frontiers in Plant Science, 8, 1805.
Rillig, M. C., Lehmann, A., de Souza Machado, A. A., & Yang, G. (2019). Microplastic effects on plants. New Phytologist, 223(3), 1066–1070.
Rillig, M. C., Leifheit, E., & Lehmann, J. (2021). Microplastic effects on carbon cycling processes in soils. PLoS Biology, 19(3), e3001130.
Rindy, J. E., Ponette-González, A. G., Barrett, T. E., Sheesley, R. J., & Weathers, K. C. (2019). Urban trees are sinks for soot: Elemental carbon accumulation by two widespread oak species. Environmental Science & Technology, 53(17), 10092–10101.
Sarker, I., Moore, L. R., Paulsen, I. T., & Tetu, S. G. (2020). Assessing the toxicity of leachates from weathered plastics on photosynthetic marine bacteria Prochlorococcus. Frontiers in Marine Science, 7, 777.
Sarker, I., Moore, L. R., & Tetu, S. G. (2021). Investigating zinc toxicity responses in marine Prochlorococcus and Synechococcus. Microbiology, 167(6), 001064.
Schiavo, S., Oliviero, M., Chiavarini, S., Dumontet, S., & Manzo, S. (2021). Polyethylene, Polystyrene, and Polypropylene leachate impact upon marine microalgae Dunaliella tertiolecta. Journal of Toxicology and Environmental Health, Part A, 84(6), 249–260.
Seng, N., Lai, S., Fong, J., Saleh, M. F., Cheng, C., Cheok, Z. Y., & Todd, P. A. (2020). Early evidence of microplastics on seagrass and macroalgae. Marine and Freshwater Research, 71(8), 922–928.
Sfriso, A. A., Tomio, Y., Juhmani, A. S., Sfriso, A., Munari, C., & Mistri, M. (2021). Macrophytes: A temporary sink for microplastics in transitional water systems. Water, 13(21), 3032.
Sharma, S., & Chatterjee, S. (2017). Microplastic pollution, a threat to marine ecosystem and human health: A short review. Environmental Science and Pollution Research, 24(27), 21530–21547.
Sintim, H. Y., & Flury, M. (2017). Is biodegradable plastic mulch the solution to agriculture’s plastic problem? Environmental Science and Technology, 51, 1068–1069.
Sjollema, S. B., Redondo-Hasselerharm, P., Leslie, H. A., Kraak, M. H., & Vethaak, A. D. (2016). Do plastic particles affect microalgal photosynthesis and growth? Aquatic Toxicology, 170, 259–261.
Sun, X. D., Yuan, X. Z., Jia, Y., Feng, L. J., Zhu, F. P., Dong, S. S., Liu, J., Kong, X., Tian, H., Duan, J. L., & Ding, Z. (2020). Differentially charged nanoplastics demonstrate distinct accumulation in Arabidopsis thaliana. Nature Nanotechnology, 15(9), 755–760.
Sun, H., Lei, C., Xu, J., & Li, R. (2021). Foliar uptake and leaf-to-root translocation of nanoplastics with different coating charge in maize plants. Journal of Hazardous Materials, 416, 125854.
Sutherland, W. J., Aveling, R., Brooks, T. M., Clout, M., Dicks, L. V., Fellman, L., Fleishman, E., Gibbons, D. W., Keim, B., Lickorish, F., & Monk, K. A. (2014). A horizon scan of global conservation issues for 2014. Trends in Ecology & Evolution, 29(1), 15–22.
Tetu, S. G., Sarker, I., Schrameyer, V., Pickford, R., Elbourne, L. D., Moore, L. R., & Paulsen, I. T. (2019). Plastic leachates impair growth and oxygen production in Prochlorococcus, the ocean’s most abundant photosynthetic bacteria. Communications biology, 2(1), 1–9.
Teuten, E. L., Saquing, J. M., Knappe, D. R., Barlaz, M. A., Jonsson, S., Björn, A., Rowland, S. J., Thompson, R. C., Galloway, T. S., Yamashita, R., & Ochi, D. (2009). Transport and release of chemicals from plastics to the environment and to wildlife. Philosophical Transactions of the Royal Society B: Biological Sciences, 364(1526), 2027–2045.
Thompson, R. C., Olsen, Y., Mitchell, R. P., Davis, A., Rowland, S. J., John, A. W., McGonigle, D., & Russell, A. E. (2004). Lost at sea: Where is all the plastic? Science, 304(5672), 838–838.
Tunali, M., Uzoefuna, E. N., Tunali, M. M., & Yenigun, O. (2020). Effect of microplastics and microplastic-metal combinations on growth and chlorophyll a concentration of Chlorella vulgaris. Science of the Total Environment, 743, 140479.
Urbina, M. A., Correa, F., Aburto, F., & Ferrio, J. P. (2020). Adsorption of polyethylene microbeads and physiological effects on hydroponic maize. Science of the Total Environment, 741, 140216.
van Kleunen, M., Brumer, A., Gutbrod, L., & Zhang, Z. (2020). A microplastic used as infill material in artificial sport turfs reduces plant growth. Plants, People, Planet, 2(2), 157–166.
vanWeert, S., Redondo-Hasselerharm, P. E., Diepens, N. J., & Koelmans, A. A. (2019). Effects of nanoplastics and microplastics on the growth of sediment-rooted macrophytes. Science of the Total Environment, 654, 1040–1047.
Wang, F., Wong, C. S., Chen, D., Lu, X., Wang, F., & Zeng, E. Y. (2018). Interaction of toxic chemicals with microplastics: A critical review. Water Research, 139, 208–219.
Wang, F., Wang, X., & Song, N. (2021). Polyethylene microplastics increase cadmium uptake in lettuce (Lactucasativa L.) by altering the soil microenvironment. Science of the Total Environment, 784, 147133.
Wu, Y., Guo, P., Zhang, X., Zhang, Y., Xie, S., & Deng, J. (2019). Effect of microplastics exposure on the photosynthesis system of freshwater algae. Journal of Hazardous Materials, 374, 219–227.
Wu, J., Liu, W., Zeb, A., Lian, J., Sun, Y., & Sun, H. (2021). Polystyrene microplastic interaction with Oryzasativa: Toxicity and metabolic mechanism. Environmental Science: Nano, 8(12), 3699–3710.
Xu, B., Liu, F., Cryder, Z., Huang, D., Lu, Z., He, Y., Wang, H., Lu, Z., Brookes, P. C., Tang, C., & Gan, J. (2020). Microplastics in the soil environment: Occurrence, risks, interactions and fate–a review. Critical Reviews in Environmental Science and Technology, 50(21), 2175–2222.
Xu, G., Liu, Y., & Yu, Y. (2021). Effects of polystyrene microplastics on uptake and toxicity of phenanthrene in soybean. Science of the Total Environment, 783, 147016.
Yan, N., Tang, B. Z., & Wang, W. X. (2021a). Cell cycle control of nanoplastics internalization in phytoplankton. ACS Nano, 15, 12237–12248.
Yan, Z., Xu, L., Zhang, W., Yang, G., Zhao, Z., Wang, Y., & Li, X. (2021b). Comparative toxic effects of microplastics and nanoplastics on Chlamydomonasreinhardtii: Growth inhibition, oxidative stress, and cell morphology. Journal of Water Process Engineering, 43, 102291.
Yang, W., Gao, X., Wu, Y., Wan, L., Tan, L., Yuan, S., Ding, H., & Zhang, W. (2020). The combined toxicity influence of microplastics and nonylphenol on microalgae Chlorella pyrenoidosa. Ecotoxicology and Environmental Safety, 195, 110484.
Yang, W., Gao, P., Li, H., Huang, J., Zhang, Y., Ding, H., & Zhang, W. (2021). Mechanism of the inhibition and detoxification effects of the interaction between nanoplastics and microalgae Chlorella pyrenoidosa. Science of the Total Environment, 783, 146919.
Yano, K. A., Geronimo, F. K., Reyes, N. J., & Kim, L. H. (2021). Characterization and comparison of microplastic occurrence in point and non-point pollution sources. Science of the Total Environment, 797, 148939.
Yin, L., Wen, X., Huang, D., Du, C., Deng, R., Zhou, Z., Tao, J., Li, R., Zhou, W., Wang, Z., & Chen, H. (2021). Interactions between microplastics/nanoplastics and vascular plants. Environmental Pollution, 290, 117999.
Yoshino, M., Osawa, H., & Yamanaka, A. (2012). Effects of process conditions on nano-dot array formation by thermal dewetting. Journal of Manufacturing Processes, 14(4), 478–486.
Yu, H., Peng, J., Cao, X., Wang, Y., Zhang, Z., Xu, Y., & Qi, W. (2021a). Effects of microplastics and glyphosate on growth rate, morphological plasticity, photosynthesis, and oxidative stress in the aquatic species Salviniacucullata. Environmental Pollution, 279, 116900.
Yu, Z. F., Song, S., Xu, X. L., Ma, Q., & Lu, Y. (2021b). Sources, migration, accumulation and influence of microplastics in terrestrial plant communities. Environmental and Experimental Botany, 192, 104635.
Zhang, C., Chen, X., Wang, J., & Tan, L. (2017). Toxic effects of microplastic on marine microalgae Skeletonema costatum: Interactions between microplastic and algae. Environmental Pollution, 220, 1282–1288.
Zhang, Q., Qu, Q., Lu, T., Ke, M., Zhu, Y., Zhang, M., Zhang, Z., Du, B., Pan, X., Sun, L., & Qian, H. (2018). The combined toxicity effect of nanoplastics and glyphosate on Microcystis aeruginosa growth. Environmental Pollution, 243, 1106–1112.
Zhang, Y., Yang, X., Luo, Z. X., Lai, J. L., Li, C., & Luo, X. G. (2021a). Effects of polystyrene nanoplastics (PSNPs) on the physiology and molecular metabolism of corn (Zea mays L.) seedlings. Science of the Total Environment, 806, 150895.
Zhang, Q., Zhao, M., Meng, F., Xiao, Y., Dai, W., & Luan, Y. (2021b). Effect of polystyrene microplastics on Rice seed germination and antioxidant enzyme activity. Toxics, 9(8), 179.
Zhou, J., Wen, Y., Marshall, M. R., Zhao, J., Gui, H., Yang, Y., Zeng, Z., Jones, D. L., & Zang, H. (2021). Microplastics as an emerging threat to plant and soil health in agroecosystems. Science of the Total Environment, 787, 147444.
Zhu, Z. L., Wang, S. C., Zhao, F. F., Wang, S. G., Liu, F. F., & Liu, G. Z. (2019). Joint toxicity of microplastics with triclosan to marine microalgae Skeletonema costatum. Environmental Pollution, 246, 509–517.
Zhu, X., Zhao, W., Chen, X., Zhao, T., Tan, L., & Wang, J. (2020). Growth inhibition of the microalgae Skeletonema costatum under copper nanoparticles with microplastic exposure. Marine Environmental Research, 158, 105005.
Zong, X., Zhang, J., Zhu, J., Zhang, L., Jiang, L., Yin, Y., & Guo, H. (2021). Effects of polystyrene microplastic on uptake and toxicity of copper and cadmium in hydroponic wheat seedlings (Triticumaestivum L.). Ecotoxicology and Environmental Safety, 217, 112217.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Srividya, A., Ganesh, A., Rathnaprabha, D. (2023). Micro- and Nanoplastics on Plant Functionalities. In: Maddela, N.R., Reddy, K.V., Ranjit, P. (eds) Micro and Nanoplastics in Soil. Springer, Cham. https://doi.org/10.1007/978-3-031-21195-9_11
Download citation
DOI: https://doi.org/10.1007/978-3-031-21195-9_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-21194-2
Online ISBN: 978-3-031-21195-9
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)