Abstract
This chapter presents the first attempt at a phytosociological analysis of forests in the upper Rio Negro (north-western Amazon) and adjacent south-western Orinoco basins, based on both floristic composition and species relative dominance. The region harbors unique plant communities that thrive under very wet climatic conditions on oligotrophic soils, drained by (mostly) black, white, or clear water rivers. The floristic composition and classification of the vegetation types were determined using data from 226 inventory plots where all individuals of diameter at breast height (DBH) ≥ 2.5 cm were measured. A set of 1368 species corresponding to 387 genera and 94 families of vascular plants was used to perform a two-way indicator species analysis (TWINSPAN). We defined the phytosociological class Eperuo leucanthae—Eperuetea purpureae, composed of seven forest communities at association level and a larger group integrated by one undetermined association. There are two orders at the highest rank. The first order (Heveo guianensis—Eperuetalia purpureae) includes three alliances and six associations, and contains the forests that grow on sandy, acidic, and extremely leached soil substrate. The Oenocarpodo batauae—Eperuion leucanthae alliance comprises two typical upper Rio Negro associations: the Amazonian caatinga and the “yévaro” forests (Micrandro elatae-Micrandretum spruceii and Oenocarpodo batauae-Eperuetum purpureae). The second order (Goupio glabrae—Minquartiion guianensis) corresponds to forests drained by white and clear waters, growing on clay soils. Data on the floristic diversity and the main environmental factors that determine the structure and floristic composition of these plant communities are presented.
“Sábado víspera de la Santísima Trinidad (3 de junio de 1542)… ..vimos una boca de otro río grande á la mano siniestra, que entraba en el que nosotros navegávamos, el agua del cual era negra como tinta, y por esto le pusimos nombre del Río Negro, el cual corría tanto y con tanta ferocidad que en más de veinte leguas hacia raya en la otra abu, sin resolver la una con la otra” (Fr. Gaspar de Carvajal. Relación del viaje de Francisco de Orellana al río Amazonas—1541–1542).
“On some black-water rivers, such as the Pacimoni, the Atabapo, and the Rio Negro in some parts of its course, the breadth of inundated land is entirely clad with bushes and small trees of very equable height, on the skirts of which the Virgin Forest rises abruptly to a height more than twice as great. This is called by the natives ‘caatinga-gapo.’ Besides these differences of aspect, the natives will tell you there are other more intrinsic ones.” (Notes of botanist on the Amazon and Andes, Richard Spruce, 1908)
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The entire Amazon watershed (sensu lato: 8, one million km2) includes 22 main tributaries, covering Bolivia, Brazil, Colombia, Ecuador, Peru, and Venezuela (including Amazon sensu stricto, Guayana Shield pro parte, Andean foothills and Gurupí basin; sensu ter Steege et al. 2013; Antonelli et al. 2018). These regions are home to a great diversity of ecosystems and to nearly 40% of the world’s tropical vegetation, with more than 50,000 plant species (Prance 2001; Morley 2011). The number of tree species inside the Amazon basin has been the subject of continuous debate during the last two decades (Hubbell et al. 2008; ter Steege et al. 2016, 2019a). Based on a comprehensive database of 1946 forest plots with up-to-date taxonomy, the most recent estimate is that over 15,000 tree species are expected to occur in Amazonia (ter Steege et al. 2020). In the exceptionally diverse Amazonian forests, between 1.4% (227 ssp.) and 4.03% (654 ssp.) of the tree species are extremely common and make up 50% of all trees over 10 cm DBH. This subset of disproportionately common trees has been dubbed the hyperdominants (ter Steege et al. 2013; Draper et al. 2021). The increase of hyperdominants is the result of a larger set of plots: for the first time, 1240 small 0.1 ha plots (that included all individuals with a minimum DBH cut-off of 2.5 cm) were assembled in a study that examined tree dominance across forest strata, from the understory to the tallest canopy, and emergent layers as well (Draper et al. 2021). These authors also found that although species belonging to a range of phylogenetically dispersed lineages have become hyperdominant in small size classes, hyperdominants in large size classes are restricted to a few lineages. In addition, some of the hyperdominants that dominate large forest areas are tree, treelet, and palm species with some evidence of domestication, especially in locations near archeological sites (Levis et al. 2017; Montoya et al. 2020). Moreover, the distribution range of only a few Amazonian tree species extends across the entire Amazon basin, while most tree species have restricted geographic coverage (Kristiansen et al. 2009).
A similar imbalance is observed in species-to-genus ratios: over half of all Amazonian tree species belong to genera with 100 or more species, while the majority of genera have ten or fewer species (Gentry 1993; Dexter and Chave 2016; ter Steege et al. 2019a).
Currently, there is a relatively good understanding of the structure and floristic composition of the forests in the Amazon basin, mainly the forests located in the western sector of the basin and in the Andes foothills (for reviews, see Pitman et al. 2008; Tuomisto et al. 2016, 2019; Alvez-Valles et al. 2018; Silva-Souza and Souza 2020).
Among the main rivers of the Amazon watersheds, the Rio Negro is remarkable: it is the second largest tributary of the Amazon river and the largest blackwater river in the world (Latrubesse and Stevaux 2015; Marengo et al. 2016). Its basin, together with the Amazon delta represents the wettest section of Amazonia (Espinoza et al. 2009; Nascimento et al. 2019). Rio Negro is the name that the Guainía river takes at its confluence with the Casiquiare channel. The botanical exploration of the Rio Negro basin was summarized in Huber and Wurdack (1984), Huber (1995a), Aymard et al. (2016), ter Steege et al. (2016), and in Appendix 3.2.
Here we present a phytosociological analysis of the forests in the upper Rio Negro (north-western Amazon) and adjacent south-western Orinoco basins, including terra-firme forests growing on clay soils, Amazonian white-sand forests, and flooded areas that occur along black, white, and clear-water rivers. This phytosociological classification provides vegetation information organized in hierarchical units to analyze and explain the floristic composition, vegetation structure, and diversity of the forests, as well as their relationship with environmental conditions, hitherto not presented in more conventional studies.
2 General Features of the Study Area
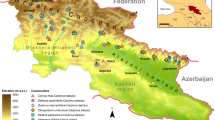
The study focuses on the Rio Negro (northern/north-western Amazon basin) and part of the adjacent south-western Orinoco basins. This region comprises the south-west of the Amazonas state of Venezuela, the south-east of the Guainía and Vaupés departments of Colombia, and the Roraima and north-west portion of the Amazonas states of Brazil. The Rio Negro basin has an area of ca. 750,652 km2 and a combined river length of ca 1600 km (Arellano-Peña et al. 2019). The two largest freshwater archipelagoes (Mariuá and Anavilhanas, with ca. 1200 and 400 islands, respectively), which also include the largest flooded igapó forest systems in the world with a highly diverse fauna (Latrubesse and Stevaux 2015), are located in the middle and lower portions of this river. Furthermore, in its upper course, the Rio Negro has the longest set of river rapids of the Amazon basin (ca. 16 km long, in São Gabriel de Cachoeira, Amazonas state, Brazil). This river originates in the headwaters of the Guianía river in Colombia, flows southward into north-western Brazil, and turns eastward north of the municipality of São Gabriel da Cachoeira, where it receives the waters of the Xié, Isana, and Vaupés rivers. It then continues in an east-south-east direction until it flows into the Solimões river, near Manaus (Brazil), together forming the main body of the Amazon river.
The geographic boundaries of the Rio Negro basin are defined in the north by the drainage divide that separates its watershed from the Guaviare, upper Atabapo, and Orinoco rivers; in the west by the upper watersheds of the Guainía and Vaupés rivers; to the south by the junction of the Japurá river with the Solimões river; and in the east by the southern foothills of the Guayana Shield drained primarily by the Rio Branco. The Serranía de la Neblina (Cerro de La Neblina: 2992 m and Pico de Neblina 3014 m), which divides Brazil from southern Venezuela, located in the Rio Negro basin, is part of the Guayana Shield highlands known as Tepuis (Huber 1987). The Serranía de la Neblina is formed by ancient rocks of the Serra dos Surucucus formation (1046 m) (Santos et al. 2003) and is rich in endemic species and unique habitats (Riina et al. 2019). The basin also holds other important ancient highlands such as: Serra da Aracá (ca. 2000 m), Serra de Curicuriari (1400 m, also known as Serra da Bela Adormecida), Serra do Pirapucu (2134 m), Pico Rondon (1189 m) in the Brazilian state of Amazonas; Serra do Apiaú (1222), Serra da Lua (532 m), and Serra do Tepequém (595 m) in Roraima state (Brazil); the Serranía de Naquén (ca. 900 m) which divides Brazil from southern Colombia and the Kanuku Mountains (1067 m) in southwestern Guyana; the Aracamuni and Avispa massifs (with a maximum elevation ca. 1600 m) in Southern Venezuela; and Pico Tamacuari (2349 m) in Sierra Curupira and SierraTapirapecó along the Brazilian-Venezuelan border (Huber 1995b).
The average annual rainfall in the Rio Negro basin varies between 3000 and 4095 mm (Espinoza et al. 2009), with the highest values in the upper stretch (≥ 3600 mm) and 0–2 months with less than 100 mm precipitation. Mean annual temperature fluctuates between 26 and 32 °C.
The upper Rio Negro and part of the adjacent south-western Orinoco region have a dense fluvial network, composed of numerous rivers the waters of which flow into the Amazon and Orinoco rivers (Goulding et al. 1988). The system is connected with the Orinoco basin through the Casiquiare channel (Fig. 3.1). The latter establishes a permanent water connection between the drainage basins of the Amazon and Orinoco (Stokes et al. 2018). The Rio Negro watershed is characterized predominantly by blackwater tributaries that originate from habitats encompassing large areas covered by white-sand soils, formed from the erosion of Precambrian Guayana Shield rocks (Klinge 1965, 1967; Junk et al. 2011). However, the Rio Branco is the exception, as it is the largest white water tributary of the Rio Negro basin. In contrast, the Orinoco basin distinguishes itself by having primarily clear and white water tributaries that drain Tertiary sediments of the Andes and the strongly weathered soils on the Guayana Shield (Ríos-Villamizar et al. 2020). The middle Orinoco river basin does have a few minor black water tributaries, such as the Atabapo, Inírida (vía the Guaviare), Ucata, and Sipapo rivers, among others, and a major one in its lower course, the Caroní river.
The basins show considerable variation in floristic composition and forest structure along local and regional environmental gradients. According to previous studies, such variation is strongly correlated with geomorphology, soils, geology, drainage, and climate (Rodrigues 1961; Takeuchi 1961, 1962a, 1962b, 1962c; Klinge et al. 1977; Anderson 1981; Klinge and Herrera 1983; Salamanca 1983; Medina et al. 1990; Ballesteros 1995; Schargel et al. 2000; Córdoba and Etter 2001; Boubli 2002; Rudas et al. 2002; Aymard et al. 2009; Stropp et al. 2011; Pombo de Souza 2012). The region also comprises a large, low-altitude peneplain of ca 165,000 km2, where hydromorphic Spodosols and Quartzipsamments are very frequent (Dubroeucq and Volkoff 1998; Schargel et al. 2000; Schargel and Marvez 2009). The peneplain landscape comprises a mosaic of unique vegetation types, particularly sclerophyllous forests on oligotrophic and acid soils, known as “caatinga Amazónica” (Colombia, Venezuela), “caatinga Amazónica baja,” “bosques de arena blanca” (Colombia), “campinarana forestada,” “campina alta” (Brazil), “varillales” (Colombia, Perú), “tastaboa” in Tucano, “parabcoha” in Desano (Ballesteros 1995), and “hamáliani” in Baniwa languages (Abraão et al. 2009).
The peneplain landscape also harbors small to extensive areas of savannas, shrubby savannas, and scrublands growing on white sands (Huber 1995c; Lleras 1997), locally called “caatinga gapó” by the natives of the upper Rio Negro (Spruce 1908), “sabanas de arena blanca,” “banas” (Klinge and Medina 1979), or low campinas, and further south “campina de solo arenoso” (Anderson 1981), “campinarana arbustiva” (Lisbôa 1975) or “campinarana graminea lenhosa” (Pombo de Souza 2012). A similar type of vegetation also occurs as far south as the Peruvian Amazon in the Jenaro Herrera district and the Allpahuayo-Mishana Reserve near Iquitos, where these plant communities are known as “varillales” and “chamizales” (Fine et al. 2010), as “varillales” in Leticia, Amazonas department in Colombia (Peñuela 2014), “muri bush” in Guyana (Richards 1952), and also in southern Brazil (Acre and Rondônia states), where they are known as “Chapada de Parecis” (Prance 2001).
The Rio Negro white-sand caatinga forests or campinaranas are not uniform in terms of floristic composition. Different white-sand forest types have been described and are summarized in Table 3.1. In addition, these white-sand forests are established through an ecological and floristic transition between terra-firme forests commonly found on peneplain red-yellow clay soils, and flooded forest communities on alluvial plains called igapó and varzéa (Prance 1980; Kubitzki 1989; Aymard et al. 2009; Wittmann et al. 2017; Luize et al. 2018). Chapters 5 and 6 in this book complement the results of the present chapter, as they describe the pattern of forest cover distribution within the Amazonas state in Venezuela.
3 Materials and Methods
3.1 Tree Inventory Data
The inventory data used in this study include all Angiosperm treelets and trees with diameter at breast height (DBH) ≥ 2.5 cm. The dataset contains 1368 species identified in inventory samples that were established across 226 localities (Appendices 3.1, 3.2, and Fig. 3.2; this information is available at the following link: https://1drv.ms/f/s!AsgLjs_JMencgZsGYB0kltyDnwkiIg
Location of the study plots in the Rio Negro basin of Brazil, Colombia, Guyana and Venezuela (blue area) and the Orinoco basin of Colombia and Venezuela (light green area). A high-resolution image is found in the following link: https://1drv.ms/f/s!AsgLjs_JMencgZsGYB0kltyDnwkiIg
The subset of 1368 species was selected from a database that includes 49,116 individuals and 2877 species and morphospecies; 1509 taxa were eliminated due to either too low representation in the region or difficulties in obtaining a good taxonomical resolution.
In Colombia, plots were located in Guainía (127), Guaviare (20), Vaupés (7), Amazonas (4), and Vichada (3) departments. In Venezuela, plots were concentrated in the Amazonas state (42) and, in Brazil, plots were located in the state of Amazonas (23) along the banks of the Rio Negro.
The tree identification names and taxonomic nomenclature were standardized and updated, using a dynamic list of Amazonian tree species (ter Steege et al. 2019 onward: http://atdn.myspecies.info/node/2466).
All maps were based on the reflectance data of a LANDSAT 5, 7, and 8 mosaic (58 tiles), and produced using a combination of supervised interpretation techniques. The final composition was built with algorithms from the software packages Octave (version 5.2.0; https://www.gnu.org/software/octave/) and Grass (version 7.8.2; https://grass.osgeo.org/). Using satellite mosaic classification and an artificial neural network, vegetation reflectance in areas without available plots was matched with the reflectance of plant communities already detected and identified in this study. Areas with unidentified vegetation types resulted from the lack of both plots and correlation with a known reflectance signal (Fig. 3.5).
Variation in floristic composition and forest structure across physical variables such as geomorphology, soils, geology, drainage, and climate were interpreted in the framework of previous studies including Schargel et al. (2000), Schargel and Marvez (2009), Quesada et al. (2011), IGAC (2014, 2018), and Zinck (Chaps. 6 and 7 this book).
3.2 Phytosociological Analysis
Information on species and field data was stored and managed using Microsoft Excel. The aboveground biomass for all species was calculated using a raw field and estimated data set with the Chave et al. (2005) allometric formula. Artificial neuronal networks (ANN) were employed to find some missing values of DBH (diameter at breast height), tree height, and wood density. Based on 11,100 complete tuple records (DBH, height, wood density; in a matrix m×n a tuple refers to a set of data in a complete row mi), a new matrix was constructed incorporating 39,211 new records (no tuples). The ANN outcomes showed a good fit for the missing values related to the known and raw field data behavior (Haykin 2009; Arellano-Peña and Rangel-Ch. 2015). According to the correlation coefficients (R), the R training describes the behavior between the existing variables within two matrices. The main matrix (X) has several columns that hold integer type data, where every number represents the classification of different taxa levels, besides some known vegetation variables (double type data) such as height and wood density.
In contrast, the second matrix (Y) has the known values of the dependent variables such as DBH. It is essential to emphasize the possibility of setting different configurations. As a consequence of the learning process with ANN, the training phase registered R values between 0.812 and 0.994. The R validation aims to compare some known and observed results within the model with some predicted results. The R values fluctuated between 0.792 and 0.994. The R test value proves the behavior of the predicted variables, their fit to the model with values between 0.805 and 0.994, and the whole process with R between 0.808 and 0.994; these results validate the ability of the model to estimate missing values (Fig. 3.3).
The plot data were analyzed using two-way indicator species analysis (TWINSPAN) (Hill 1979). The latter was carried out using PC-ORD Multivariate Analysis of Ecological Data for Windows, version 7.0 (McCune and Mefford 2016). TWINSPAN reveals clusters or groupings of plots similar in terms of species composition. Such clusters can indicate associations of tree communities or sub-communities.
The resulting TWINSPAN was interpreted in terms of syntaxonomical classification of the vegetation, based on floristic affinities, according to the Zürich-Montpellier approach (Braun-Blanquet 1979; Westhoff and van der Maarel 1973). TWINSPSAN classifies species and samples in a way that approaches the results of a Braun-Blanquet vegetation table. This was used to build a key to the classification of the samples by identifying one to several species, which were particularly diagnostic of each division in the classification. The pseudo-species analysis cuts were carried out in TWINSPAN with five levels or intervals. Five plots were chosen as the minimum group size for division, and five species as the maximum number of indicators per division.
4 Results
TWINSPAN produced five levels and established 21 indicator species. These species classify the samples and plots under a unique class (two main orders, with five alliances, seven associations, and a larger group integrated by several indeterminate associations) (Figs. 3.4 and 3.5). The results of this phytosociological classification divided the forests into three main pedobiomes that occur in the study area. These pedobiomes reflect three forest types associated with different substrates and floristic and structure differences: the Amazonian caatinga (campinarana) on well-drained sandy soils on slightly higher peneplain surfaces, the tall terra-firme forests on deep well-drained clay soils, and forests in alluvial plains on soils with water-logging at different depths. Soil features (e.g., drainage, very low water retention capacity, and nutrient availability in lesser degree), and types of water are the factors that split the forests into three main pedobiomes in the study area. The influence of these factors is reflected in the floristic and structure differences among Amazonian caatinga, terra-firme forests, and forests in alluvial plains.
Map of the study region showing the phytosociological groups in the upper Rio Negro and Orinoco basins. A high-resolution imagen is found in the following link: https://1drv.ms/f/s!AsgLjs_JMencgZsGYB0kltyDnwkiIg
4.1 Phytosociological Classification
The procedure defined the phytosociological class Eperuo leucanthae—Eperuetea purpureae from which the first division level distinguished two orders.
4.1.1 Order I
The first order (Parahancornio surrogatae-Aldinion latifoliae) is composed of communities strongly associated with blackwater drainage (i.e., tall caatinga (campinarana) forest and Micrandra spruceana, Monopteryx uaucu, and Erisma japura forests), with the following indicator species: Hevea guianensis, Eperua purpurea, Mucoa duckei, Monopteryx uaucu, Oenocarpus batatua, Pouteria cuspidata, Caraipa longipedicellata, Calophylum brasiliense, Iryanthera elliptica, and Aldina kunhardtiana. This order was separated at the second level by alliance II with the indicator species Parahancarnia surrogata and Aldina latifolia. At the third level, alliance I was split into association I (Aldina latifolia, Terminalia ochroprumna, Swartzia sericea, Pachira nitida) and association II (Caraipa longipedicellata, Micrandra sprucei, Compsoneura debilis, Cyrilla recemiflora).
Association I also includes the flooded forest communities on blackwater floodplains, called igapó (Figs. 3.6 and 3.7) by Ducke (1954), Prance (1980), and Kubitzki (1989). In the Rio Negro region, about 119,000 km2 of the basin are covered by igapó with forest coverage >85% (Householder et al. 2021). The same authors are in agreement with the general hypothesis that the flood duration gradient is a major environmental driver of compositional turnover in floodplain forests, even among distant sites. This implies that species ecological distributions along the flood duration gradient are predictable and unlikely to be geographically distinctive between sites (Householder et al. 2021). The igapó in the Rio Negro also contains unique large areas made up of medium high trees with small dark leaves, very different from the exuberant Amazonian vegetation, even from the igapò forests. These communities are known as “boyales” or “selvas de boya” in Colombia and Venezuela (Vareschi 1963) and “formações do Molongó” in Brazil (Ducke 1938). “Boyales” are abundant in flooded areas of the northern Rio Negro basin in Colombia and Venezuela, and extend south to the river Urubú in Brazil (Ducke 1944). In general, they are made up of numerous small trees with extremely light wood (mainly at the base of the trunks), adapted to the blackwater habitats (Mägdefrau and Wutz 1961; Berry and Wiedenhoeft 2004). The unique ecological characteristics of the “boyales” make their flora predominantly endemic (e.g., Anaxagorea inundata, Malouetia molongo, Micrandra inundata, Pouteria pimichinensis), and well adapted to the extreme conditions of annual flooding (Fig. 3.8). These communities are dominated by taxa from Annonaceae, Apocynaceae, Malvaceae, and Euphorbiaceae (Aymard et al. 1989). This kind of vegetation represents unique and large riparian communities in the north-western Amazon basin and part of the Orinoquia (Romero-G et al. 2019). Nonetheless, recent studies and reviews of the vegetation associated with the rivers of both basins make no mention of “boyales” or “formações do Molongó” (Wittmann et al. 2017; Luize et al. 2018; Householder et al. 2021).
The third level also recognized alliance II (Clathrotropis glaucophylla, Eperua leucantha, Micrandra sprucei, Oenocarpus bataua) and association III (Macrolobium limbatum, Micrandra elata, M. sprucei, and Mucua duckei). The fourth level produced alliance III (Eperua leucantha, E. purpurea, Hevea guianensis, Monopteryx uaucu, Virola michelii) and association IV (Eperua purpurea, Oenocarpus bataua). The fifth level divided the rest of alliance III in associations V (Aldina heterophylla, E. leucantha) and VI (Eperua purpurea, Erisma japura). Association VI harbors the forests dominated by E. purpurea, a soil generalist species (Aymard et al. 2009), known as “aceitón” in Colombia, “copaibarama” in Brazil, “yevaro” in Venezuela, and “waapa” in Kuripako language (A. Calero-Cayopare, pers. com).
4.1.2 Order II
The second order (Goupio glabrae—Minquartiion guianensis) corresponds to forests associated with white and clear waters, growing mostly on clay soils. This syntaxon is named after the exclusive species Goupia glabra and Minquartia guianensis as the dominant species on disturbed forests. The Goupio glabrae—Minquartiion guianensis holds two alliances and two associations: an undefined Attalea maripa—Brosimum utile association, and the Eschweilero parviflorae—Mouririetum grandiflorae asociation. Forests dominated by palms are represented by the undefined alliance of Astrocaryo chambirae—Socrateion exorrhizae.
The second level of this order separated alliances IV (Goupia glabra, Minquartia guianensis, Mouriri grandiflora) and V (Astrocaryum chambira, Calycophyllum megistocaulum, Euterpe precatoria, Inga tessmannii, Psedolmedia laevis, Psychotria remota, Socratea exorrhiza), the latter being a complex group without defined forest associations in alluvial plains mixed with palm communities. The third level showed two different divisions, the association VII (Clathrotropis brachypetala, Erisma laurifolium, Eschweilera parviflora, Mouriri grandiflora, Pouteria baehniana, Protium divaricatum, Virola elongata, Zamia ulei), and another branch including a larger group without clear associations. The last cluster was characterized by the following indicator species: Attalea maripa, Brosimum utile, Eschweilera parviflora, Euterpe precatoria, Goupia glabra, Mayaba elengans, Mespilodaphne cymbarum, and Virola elongata. These communities extend from the alluvial plains to terra-firme forests on terraces such as the “sasafrás” forests, a community dominated by Mespilodaphne cymbarum (Fig. 3.9).
(a) “Sasafrás” forest, a community that extends from the alluvial plains to terra-firme forests on terraces drained by clear waters; (b) forest dominated by Mespilodaphne cymbarum (Kunth) Trofimov (Lauraceae), a valuable timber known as “Sasafrás del Orinoco”; Guaviare river, Guainía department, Colombia; photographs by Francisco Castro-Lima
4.2 Vegetation and Environmental Conditions
Soil and water types were related to the clustering of species dominance data into two orders. The first one comprises three alliances and six associations, mainly dominated by the high caatinga (campinarana) because of the abundance of Caraipa longipedicellata, Eperua leucantha, Micrandra sprucei, and Mucoa duckei growing on very poorly drained Spodosols (Figs. 3.10 and 3.11). Other communities, such as tall and medium forests on somewhat poorly drained Entisols on terraces, appeared in this order (i.e., Monopteryx uaucu and Erisma japura forests). These communities were separated from the high caatinga because their floristic composition shows that they share species well distributed over moderately and poorly drained Entisols, Inceptisols, Oxisols, and Ultisols. The tall and medium forests also contained taxa that were common in areas with Spodosols. The variety of habitats strongly suggests that the above-mentioned species are not white-sand specialists, as it has been proposed (García-Villacorta et al. 2016). Examples of taxa with a wide habitat preference and frequent in forests that grow on white sands in the upper Rio Negro are: Aldina kunhardtiana, Dendropanax neblinae, Eperua leucantha, E. purpurea, Erisma micranthum, Hebepetalum humiriifolium, Helianthostylis steyermarkii, Pentamerista neotropica, Sloanea floribunda, and Tetrameranthus duckei, all considered soil generalists by Aymard et al. (2009). Plant communities of the upper Rio Negro basin found on white sands and terra-firme forests growing on clay soils may have a common evolutionary history (Aymard et al. 2016).
The second order includes the communities associated with white and clear waters, growing on clayey soils. Under this physical condition, communities tolerate considerable differences in drainage, because in poorly drained soils root mats develop below the litter layer. These communities are composed of an array of medium to tall forests mixed with palm communities, with three to four vertical layers at heights of 25–35 m, 15–25 m, 7–12 m, and 3–7 m. The top layer included emergent trees reaching heights of 35–45 m, such as Erisma laurifolium and Goupia glabra, giving the canopy of this forest type a very irregular aspect. The vertical discontinuities are further accentuated by the frequent occurrence of gaps caused by tree falls. The third and fourth layers are not always easily distinguished because of the high density of species such as Anaxagorea brachycarpa, Clathrotropis glaucophylla, Heterostemon conjugatus, Iryanthera paradoxa, Matisia ochrocalyx, Pseudosenefeldera inclinata, Sagotia heterocalyx, and Zygia claviflora. Dense colonies of palm species such as Astrocaryum chambira, Bactris corosilla, Euterpe precatoria, Iriartella setigera, and Socratea exorrhiza, as well as the giant caulescent herb Phenakospermum guyannense (considered an indicator of disturbed forest) were present in the third layer. The phytosociological classification and description of the new alliances and associations of the forest communities of the Rio Negro region are outlined in Appendices 3.1, 3.3, and Figs. 3.4 and 3.5.
5 Discussion
5.1 General Aspects
The study shows that a relatively low number (226) of small to medium-sized transects and plots (mostly 0.1 ha transects) may be sufficient to set up a robust phytosociological analysis of vast regions (Figs. 3.4 and 3.5). This approach allows comparing local variations in forest structure and floristic composition by soil topography across areas with different environmental conditions. An important issue in evaluating the results is the degree to which site diversity (alpha diversity) is being measured vs. locally varying habitat diversity (beta diversity). Local diversity can be affected by the shape of the plot, with increasingly narrow rectangular plots generally showing higher diversity values than broader or square plots (Condit et al. 1996). Large plots (≥ 1 ha) have provided useful data to characterize forest structure and composition, and if properly tagged, protected, and monitored, they can provide long-term data on the growth, mortality, regeneration, dynamics of forest trees, and climate change (ForestPlots.net 2021). However, given the costs and labor they require to sample and maintain, such plots are relatively few and spatially scattered. A lower-cost and faster alternative, especially when permanent plots are not required or are not feasible to maintain, is 0.1 ha transects (Gentry 1982). With similar effort necessary to set up one-ha plots, many smaller plots can be established, yielding valuable information about variability in forest sites at a local scale (Gentry 1988a, 1988b; Clinebell et al. 1995; Aymard et al. 2009). Often, a lower diameter cut-off (usually 2.5 cm DBH) is used in small plots, which yields individual stem numbers of the same magnitude as those obtained from hectare plots sampled at ≥10 cm DBH. Gentry (1988a, b; Draper et al. 2021) used 980 transects of 0.1 ha, combining efficient ecological sampling with high-quality botanical identifications to describe large-scale patterns of alpha diversity and floristics, and then create highly distributed measurements of the world’s forests (for a review, see Phillips and Miller 2002; ForestPlots.net et al. 2021).
The phytosociological study presented here shows that the order level was most useful to characterize the alliances and their communities. At this level, the eigenvalues were as high as 0.5. High values determine a significant dispersion of the data in the analysis that allows displaying the species along environmental variables (ter Braak, 1987). The analysis with TWINSPAN generated end-groups of five blocks that represent vegetation units in five alliances and nine associations that form the dendrogram (Fig. 3.4).
Variations in floristic composition could be related to the particular drainage and water-holding capacity of soils where these forest types grow. For example, there are species that tend to occur on deep well-drained terra-firme soils (e.g., Allantoma lineata, Caryocar pallidum, Erisma japura, Eschweilera micrantha, Guarea trunciflora, Manilkara huberi, Mezilaurus itauba, Minquartia guianensis, Pseudosenefeldera inclinata, Scleronema micranthum), or on very poorly drained Spodosols in lowlands (e.g., Aspidosperma verruculosum, Caraipa longipedicellata, Compsoneura debilis, Couma catingae, Hevea rigidifolia, Mabea arenicola, Micrandra sprucei, Micropholis maguirei, Myrcia neoforsteri, Neocouma ternstroemiacea). In addition, a considerable amount of taxa appear only in riparian, swamp, and seasonally black water-flooded forests on alluvial plains growing on soils with water-logging at different depths (e.g., Eschweilera tenuifolia, Guatteria heteropetala, Handroanthus barbatus, Leopoldinia piassaba, Lissocarpa benthamii, Molongum laxum, Mouriri acutiflora, Myrcia argentigemma, Parahancornia negroensis, Spongiospermum riparium); individual species respond differently to the flood duration gradient (Householder et al. 2021).
This point is illustrated by Sabatier et al. (1997), who in French Guiana observed substantial changes in forest communities in the transition of soils with deep vertical drainage to superficial lateral drainage. At large scale, however, the forest structure and dynamics have been noted to vary across the Amazon basin in an east-west gradient in a pattern that coincides with variations in soil fertility, topography, and geology (Pitman et al. 2008; Quesada et al. 2011, 2012).
Therefore, large-scale variations in forest biomass could not be explained by any of the soil or climate properties analyzed here. A recent study using one-ha forest inventory plots in Costa Rica showed that, while plant species richness was controlled by climate and soil water availability, vegetation carbon storage was strongly related to wood density and soil phosphorus availability (Hofhansl et al. 2020). These results also suggest that local heterogeneity in resource availability and plant functional composition should be considered to improve projections of tropical forest ecosystem functioning under future climate scenarios.
Oligotrophic soils (either Spodosols or clay soils) throughout the Rio Negro basin (Herrera 1985; Dubroeucq and Volkoff 1998; Schargel and Marvez 2009; Quesada et al. 2011; IGAC 2014) influence in minor degree differences in forest types in this region (Schargel and Marvez 2009; Stropp et al. 2014). Moreover, several studies showed that soils, drainage, moisture retention, and water types (i.e., black, clear, and white waters) are the principal factors that separate terra-firme, caatinga, and alluvial plain forests in the study area (Medina et al. 1990; Franco and Dezzeo 1994; Coomes and Grubb 1996; Schargel et al. 2000; Stropp et al. 2011).
Besides, human intervention by way of shifting cultivation and the enrichment of the original forests with introduced, useful species, on the better-drained soils, cannot be discarded as a cause for creating floristic differences (Levis et al. 2017; Montoya et al. 2020). Total rainfall does not need to decrease drastically to favor vegetation change on these nutrient-poor soils, most of which have low water retention capacity. An increase in the length and severity of the dry season would be sufficient. It was observed that the geographical distribution of many non-generalist species is related to average annual rainfall and the duration of the dry season. For example, Aldina kunhardtiana, Caraipa longipedicellata, Chrysophyllum sanguilentum, Eperua purpurea, E. leucantha, Helianthostylis steyermarkii, Inga neblinensis, Leptobalanus cardiophyllus, Macrolobium limbatum, Mezilaurus itauba, Monopteryx uaucu, Ouratea clarkii, Protium carolense, P. crassipetalum, Roucheria punctata, Swartzia benthamiana, and Tachigali odoratissima represent taxa that belong to a larger group of species that is only found in areas within the basin with precipitations between 3000 and 3600 mm and 0–2 months with less than 180 mm precipitation, suggesting that the distribution pattern of many species is also directly related to annual precipitation patterns and the duration of the dry season. These climatic parameters, in conjunction with the gradient in soil fertility, are considered by ter Steege et al. (2003, 2006, 2010) as the two main variables that predict values of alpha diversity and stem density in Amazonian forests. In their proposed model, they found that the most diverse forests are located just south of the equatorial line (areas with 0–1 month with precipitation <100 mm); while the less diverse ones were found on the Guayana Shield and in the Amazon area of Bolivia (areas having 5–7 months with precipitation <100 mm). A comparison of this pattern with data provided by Aymard et al. (2009) shows that the duration of the dry season (DSL) was more useful in determining species distributions than predicting alpha diversity values, because the upper Rio Negro and part of the adjacent south-western Orinoco region, with higher rainfall and few months of little rain, also has low alpha diversity. This finding contradicts the assumption that predicts that high elevation and extremes of substrate-related factors underpin the floristic segregation of environmentally “marginal” vegetation types and “terra-firme” forests, rather than climatic factors, which in these case are relatively unimportant (Oliveira-Filho et al. 2021).
5.2 Forest Structural Characteristics
The upper Rio Negro (north-western Amazon) and adjacent south-western Orinoco basins, where forest structure varies across different soil types and local topography, holds a top layer that includes emergent trees reaching heights of 35–45 m, and a medium to short dynamic tree stratum like most northwestern Amazon forests (Quesada et al. 2012). According to Draper et al. (2021), smaller-statured species may be exposed to different biotic and abiotic filters across large spatial scales, and these variables develop greater local specialization associated with distinct functional characteristics.
Canopy and mean tree height decreased from soils with moderate to poor drainage to very poorly drained Spodosols. Very low moisture retention, due to shallow soil depth or coarse sandy saprolite, also determined a tree height decrease in the somewhat poorly drained Entisols (Schargel and Marvez 2009) together with an increase in high values of stems (Uhl and Murphy 1981; Aymard et al. 2009; Stropp et al. 2011).
In the San Carlos de Río Negro area, Klinge and Medina (1979) and Bongers et al. (1985) found that the reduction in height of the caatinga (campinarana) community was related to a reduction in depth of the aerated soil layer above the water table. This forest community dominated by Micrandra sprucei and Eperua purpurea is similar to those of caatingas nearby Manaus (Takeuchi 1962a, 1962b). Moreover, the tallest layer of this forest is 15–25 m high, and dense colonies of Mauritia carana and Euterpe catinga also occurred in this layer (Aymard et al. 2009).
The frequency of tall trees (DBH > 80 cm, over 45 m high) is low in the upper Rio Negro basin compared with the adjacent south-western Orinoco basin. The most frequent trees which reached over 45 m high were: Allantoma lineata, Brosimum utile, Caryocar spp., Eperua purpurea, Ecclinusa ramiflora, Erisma bicolor, E. japura, Eschweilera spp., Goupia glabra, Mespilodaphe cymbarum, Micrandra spruceana, Micropholis brochidodroma, Minquartia guianensis, Monopteryx uaucu, Parahancornia negroensis, Pouteria spp., Swartzia floribunda, Terminalia (Buchenavia) spp., and Vochysia ssp. By contrast, all forest types had a larger number of medium and smaller trees located in the second and third strata that defined the forest structure.
With the minimum DBH cut-off of 2.5 cm used in this study, the number of stems sampled varied from 101 to 1017 per plot, whereas one-hectare plots with a minimum DBH of 10 cm often have around 500 stems (Valencia et al. 2005). The high density of individuals with lower average diameter in very nutrient-poor soils is due to increased tree longevity in the lower strata and understory levels, very slow growth, presence of sclerophyllous leaves with low nutrient content, and high levels of phenols and tannins that inhibit herbivory (Janzen 1974). Cuevas and Medina (1986, 1991), Medina et al. (1990), and Sanford and Cuevas (1996) observed in the upper Rio Negro region that plant individuals on soils with the lowest fertility invest more resources in the development of fine roots to penetrate the soil and obtain nutrients, than in photosynthetic tissues (discussed in Chap. 4). This reduction in photosynthetic tissue reduces the population of herbivores and eventually produces a positive result by developing plants with better defense mechanisms, representing an evolutionary response of species that grow on very nutrient-poor soils (Fine et al. 2006). However, Stropp et al. (2014) provide no evidence that an interaction between herbivory and soil nutrient availability drives habitat association of tree species in white-sand and terra-firme forests of the upper Rio Negro. Nutrient conservation depends on the structure of plant communities; forests on nutrient-poor soils, located in the upper Rio Negro in front of the Guayana Shield, have high wood density values, defense mechanisms, and more efficient nutrient conservation (Vitousek and Sanford 1986; Medina et al. 1990) than Amazonian forests located on more fertile soils on the foothills of the Andean Cordillera (Pitman et al. 2008), and in well-drained upland forests in north-western Amazonia (Duivenvoorden et al. 2005; Cano and Stevenson 2009).
The forests located in the south-western Orinoco basin have four vertical layers (35–45 m, 15–25 m, 7–12 m, and 3–7 m). The top layer included more emergent trees than in the Rio Negro basin, reaching heights of 45 m, giving the canopy of this forest a very irregular aspect. The second layer (15–25 m) is quite heterogeneous, and the third and fourth layers are not always easy to distinguish because of the high density of species such as Clathrotropis brachypetala, Matayba elegans, Mouriri grandiflora, Pseudolmedia laevis, and Virola elongata. These communities harbor numerous colonies of palms, of which stand out Euterpe precatoria, Iriartella setigera, Socratea exorrhiza, Oenocarpus bataua as well as the Strelitziaceae Phenakospermum guyannense. Palms perhaps represent the most characteristic physiognomic element of the terra-firme and riverine forests of the Amazon and Orinoco basins (for a review, see Alvez-Valles et al. 2018). In the tall forests studied here, seedlings of Oenocarpus bataua were abundant everywhere, and Astrocaryum gynacanthum and Bactris corosilla often formed a layer 3–6 m high with their long, divided leaves. Other small to medium-stature species, such as Astrocaryum chambira (“palma cumare”) with long spines along its trunks, were scattered in the forest. Individuals of Oenocarpus bataua were the tallest palms (to ca 30 m), and sometimes appeared as canopy emergent. The distribution of palm species in lowland forests is usually correlated with gradients from well to poorly drained soils (Alvez-Valles et al. 2018). In the study area, Mauritia carana, for example, occurs on poorly drained sites between the caatinga forest and white sand savannas, and Leopoldinia piassaba is generally associated with poorly drained Spodosols and low water retention soils (Vareschi 1963; Kubitzki 1991).
North-western Amazonian forests appear to be comparatively poor in climbers (Putz and Mooney 1991). The so-called “matas de cipós” (liana forests), found in other Amazonian regions especially between the Xingu and Tocantins rivers (Prance 1989) do not occur in the upper Rio Negro area (Putz 1983). In this study, lianas were represented by only 143 species (10.45%), mainly in terra-firme forests on moderately to poorly drained soils.
5.3 Plant Diversity
The upper Río Negro region is reportedly not a region rich in local tree diversity when compared to other Amazonian ecosystems (Uhl and Murphy 1981; Dezzeo et al. 2000; Boubli 2002; Cárdenas-López et al. 2007; Aymard et al. 2009; Stropp et al. 2011; Pombo de Souza 2012). The same pattern has been observed in other places with white-sand ecosystems in the basin (Capurucho et al. 2020). A study in a forest dominated by Eperua purpurea in San Carlos de Rio Negro (Venezuela) identified 183 species, detected by combining the results of a 0.10 ha transect and a 1-ha plot (Aymard et al. 2009). In this area, the highest number of species occurred in terra-firme forests because these are the largest communities, the terra-firme occupies 70–80% of Amazonia (ter Steege et al. 2003, 2019b).
Similarly, in the upper Rio Negro basin, terra-firme forests located in the upper Isana region are plant communities with high species diversity, recording between 108 and 162 species in three plots of 40 × 40 m (1600 m2) with DBH ≥ 5 cm (Arellano-Peña et al. 2019). In areas on podzolic soils and with high rainfall within the upper Rio Negro basin, the fewest species occur in sectors with drainage limitations, dominance of ectomycorrhiza in the topsoil layer, and lower nutrient retention capacity (Kubitzki 1990; Moyersoen 1993). This relationship is coherent with the high local species endemism reported by Steyermark (1982) and Kubitzki (1989) in the Rio Negro basin, including Duguetia aberrans, D. sancticaroli, Besleria yatuana, Chamaecrista ipanorensis, Eschweilera rionegrense, Mezilaurus caatingae Pseudephedranthus fragans, and Vochysia steyermarkiana. These forests consist of relatively few species (oligarchic) mixed with taxa represented by just one or a few individuals known as rare species (ter Steege et al. 2019b). The latter contribute enormously to regional diversity, have very low values of abundance in the oligarchic forests, and are usually the most difficult to identify at the level of species (Aymard et al. 2009).
The low diversity of species in the region is perhaps partially explained by factors related to the history and evolution of Neotropical forests, such as climatic events that the biota of the lowlands of northern South America lived through from Tertiary to Quaternary (Hooghiemstra et al. 2002, 2006; Hoorn et al. 2010, 2017; Wesselingh et al. 2010), including variations in precipitation patterns during the last glacial advance (Last Glacial Maximum, LGM) approximately 18,000 years ago (van der Hammen and Absy 1994; Ruiz-Pessenda et al. 2009). These events caused changes in forest vegetation in some sectors of the Amazon basin, which was replaced by savannas (Ruiz-Pessenda et al. 2009), shrubs or other characteristic communities of dry environments (Häggi et al. 2017). In other areas like the region that currently corresponds to the Rio Negro basin, vegetation may have been constantly subjected to changes through longer dry periods, with overall less rainfall, and higher sediment flow than in the western Amazon (Hooghiemstra et al. 2006), where forest cover was persistent (Bush et al. 2004). By responding to these changes in climate and significant landscape transformations, biomes expanded or contracted, becoming either connected or disjointed (Colinvaux et al. 2000; Pennington et al. 2004; Baker et al. 2020).
These environmental factors have produced significant alterations of the forest due to fires during the Holocene, a fact that is supported by the numerous samples of charcoal found in the region of San Carlos de Río Negro (Venezuela) and Lake Acarabixi (Brazil) dating from the Holocene (Saldarriaga and West 1986; Rodriguez-Zorro et al. 2018). Continuous changes in vegetation perhaps did not allow maintaining a previous flora or the formation of a flora as rich in species as that of western Amazonia (Stropp et al. 2009, 2011). Bush et al. (2004) and Cordeiro et al. (2011) conducted palynological and geochemical studies in the “Lagoa da Pata,” in the upper Rio Negro (Amazonas state, Brazil), that revealed significant changes in the environmental history of this portion of the Amazon basin during the late Quaternary. The authors found pollen from elements of Andean regions such as Alnus, Hedyosmum, Myrsine, Podocarpus, and Weinmannia, mixed with lowland genera (e.g., Cedrela). The sediment core (113.6 cm deep) indicated that the elements of the mountainous regions were very abundant between 45,000 and 12,000 yr. calBP.
Subsequently, in the range of 12,000 yr. calBP to the present, the pollen of the Andean genera disappeared completely from the sample studied (even Cedrela). Furthermore, the geochemical study from “Lagoa da Pata” matched perfectly with the results forwarded by Bush et al. (2004). This outcome revealed three hydrological and climatic regimes from 50,000 to 10,000 yr. calBP, characterized by a relatively wet climate (from 50,000 to 26,300 yr. calBP), a decrease in productivity (from 26,300 to 15,300 yr. calBP) that indicated a dry phase, and an increase in lacustrine productivity from approximately 15,300 to 10,000 yr. calBP (Cordeiro et al. 2011). In addition, two recent studies in the upper Rio Negro region (Lake Acarabixi) revealed that between 9000 and 4000 yr. calBP took place in the warmest and driest period of the last 100,000 years which coincided with changes in evaporation and precipitation that caused lake levels to drop over most of tropical South America (Rodriguez-Zorro et al. 2018; Nascimento et al. 2019). Furthermore, highland taxa such as Hedyosmum and Myrsine were found at that time together with igapó forest species like Astrocaryum, Eschweilera, Macrolobium, Myrtaceae, and Swartzia. During the late Holocene (1600 to 650 yr. calBP), no drastic changes in vegetation were observed, but the presence of pioneer species like Vismia and Cecropia, along with the evidence of fires, pointed to human disturbance. Currently, with the exception of Podocarpus (P. tepuinensis), Gordonia (G. spathulata), Cyathea (C. macrosora), two species of Otoba (O. glycycarpa, O. parviflora) and few species of Ilex (e.g., I. casiquiarensis, I. davidsei, and I. spruceana), none of the highland genera mentioned in the palynological study have been registered in the present-day flora of this region. Nowadays, Podocarpus is an almost exclusive genus of montane zones; nonetheless, P. tepuiensis was found in the watershed of the upper Rio Negro (Berry and Aymard 1997). The presence of these five genera in the lowlands indicates the remnants of a relict flora that existed in the region between 45,000 and 12,000 yr. calBP and was different in species than the current one. Therefore, other species may have disappeared, leading to very distinctive forests than we see now (Bush et al. 2004).
The occurrence of highlands taxa found in the study area, partially agree with the Oliveira-Filho et al. (2021) assumption that the lack of these taxa in lowland Amazonia is likely driven by temperature, an important environmental factor driving floristic differentiation between montane and terra firme forests in the Amazon basin.
All this evidence points out that perhaps the forest did not fragment, but suffered significant variations in floristic composition due to drastic changes in temperature and precipitation, which effected diversity during the late Quaternary.
Because of its important number of taxa and their endemic elements, Huber (1994) placed the upper Rio Negro basin in the Guayana phytogeographical region, rather than as part of the Amazonian phytogeographical region, where it has traditionally been placed by phytogeographers. Nevertheless, a recent study aimed to regionalize the Amazon tree flora identified the upper Rio Negro as a main subregion through 161 indicator taxa (Silva-Souza and Souza 2020).
At local scale, the diversity in the upper Rio Negro and south-western Orinoco basins is low, partially explained by factors related to the history, area and evolution of Neotropical forests. This condition is related to climatic events that affected the biota of the lowlands of northern South America from Tertiary to Quaternary. It is documented that a more seasonal dry-wet climate caused marginal forest retraction and, together with temperature decrease, rearranged forest composition to some extent. This is observed in pollen records across Amazonia, depicting the presence of taxa at glacial times in localities where they do not live presently (D’Apolito et al. 2017).
Most of the taxa recorded in this study show a wide distribution throughout much of the Amazon basin, while some of the dominant species are restricted to the upper Rio Negro area, such as Eperua purpurea, E. leucantha, Erisma japura, and Micrandra sprucei (Clark et al. 2000). However, sampling artifacts certainly contribute to distort species distributions in the Amazon basin due to the strong tendency for collection density to be high in very few localities (Nelson et al. 1990; Schulman et al. 2007), such as close to main towns (e.g., Barcelos, San Carlos de Rio Negro, São Gabriel de Cachoeira), and lower in more distant rural areas (Hopkins 2019). Furthermore, a recent study showed that ca 300,000 km2 of the Brazilian Amazon had been deforested by 2017, without having a single tree specimen recorded (Stropp et al. 2020).
6 Conclusions
This study provides new information on the floristic composition, structure, and diversity of the vegetation using a phytosociological classification. The analysis with TWISNPAN shows that the order level was the most useful variable to characterize vegetation. At this level, the classification developed notable resolution represented by five alliances and nine associations (Fig. 3.4). However, significant compositional differences may arise from chance alone, even among sites with identical environmental conditions, because small forest inventories yield a diverse pool of regional taxa (Gentry 1982; Ricklefs 1987).
Soil features (e.g., drainage, very low water retention capacity, and nutrient availability in lesser degree), and types of waters are the factors that split the forests in three main pedobiomes in the study area. The influence of these factors is reflected in the floristic and structure differences among Amazonian caatinga on well-drained sandy soils on slightly higher peneplain surfaces, tall terra-firme forests on deep well-drained clay soils, and forests in alluvial plains on soils with water-logging at different depths.
Although the Rio Negro basin harbors the largest blackwater river in the world, it is still one of the least studied regions of the Amazon (Hopkins 2019; Stropp et al. 2020). Lleras (1997) pointed out that the entire basin (ca 750,652 km2), at a regional level, holds an exceptionally high species diversity (over 15,000), with several genera (e.g., Asteranthos—Scytopetalaceae, Hylaea—Apocynaceae, Neblinaea—Asteraceae, Neblinantha—Gentianaceae, Neblinanthera—Melastomataceae, Pyrrorhiza—Haemodoraceae) and a significant number of species only known to occur in the Rio Negro basin and its tributaries.
The predominantly descriptive nature of this phytosociological study is justified considering the state of baseline knowledge on the northern and north-western Amazonian ecosystems, particularly concerning spatial components of soil, types of draining water, and forest variation. Nonetheless, our results support the hypothesis predicting that distinct forest categories are associated with soil and climate conditions. Moreover, our study detected a dominance of important tree species. In addition, we found that sites with similar vegetation types (i.e., tall caatinga forest) show high affinity (in terms of structure but not in floristical composition, see Table 3.1) between them regardless of their separation along a geographic gradient. This affinity was also pointed out by Oliveira-Filho et al. (2021) in the entire Amazon basin, at least in reference to forest structure.
7 Forest Conservation Issues
Despite the relatively low number of one-ha plots and transects (226) analyzed here, it was possible to report differences in species composition and structure of the forests, and their relationships with environmental conditions such as soil characteristics and drainage across the upper Rio Negro region. Some of the differences between the forest characteristics observed in this study may be easily detectable on satellite images (Tuomisto et al. 2019). Rapid diversity assessments in small plots and transects preceded by general physiographic surveys based on remote sensing would be an efficient tool to estimate the overall level of the vascular plant diversity and its variability over large regions, or else in separate physiographic subdivisions, such as floodplains, swamps, or well-drained areas. Such information would offer basic reference material to evaluate the desirability for conservation and protection of certain rainforest areas and to help interpret the effects of human intervention and fragmentation of forests upon local and regional levels of vascular plant diversity. Moreover, a larger number of smaller inventory sites, such as those established during rapid diversity assessments, is likely to capture more diverse and heterogeneous tropical forest habitats than a smaller set of larger transects (Clinebell et al. 1995).
One alternative approach to improve data collection, at least in the Rio Negro region, involves local inhabitants in academic research collaborations. While the deforestation rate inside indigenous territories and protected natural areas remains well below the rates outside, unsustainable forest clearing is on the rise across the Amazon basin (Walker et al. 2020). Indigenous rights are violated by changes in current legislation that threaten to weaken indigenous peoples’ constitutionally-guaranteed territorial rights. Regarding the Rio Negro indigenous territories, a total of 387 requests for mining concessions were pending in Brazil already in 2016 (Almeida et al. 2016) and 75 in Colombia by 2020 (ANLA 2020). The most severe threat may be in Venezuela, where the government overlooked illegal mining and deforestation. All these actions put at risk the forests of the indigenous people of the region, even though the Brazilian, Colombian, and Venezuelan constitutions recognize that indigenous people have rights to their traditional territories. Large protected areas of the Amazon basin north of the Equator are located in the Rio Negro region (Lleras 1997). These include the “Alto Orinoco-Casiquiare” Biosphere Reserve (82,662 km2, in Venezuela), a bi-national Park that features “Parque Nacional Serranía de la Neblina” (13,600 km2) in Venezuela, and “Parque Nacional del Pico de la Neblina” (22,200 km2) in adjacent Brazil, as well as “Monumento Natural Piedra del Cocuy” (0.15 km2) in Venezuela, a place close to which the frontiers of three countries merge. Lleras (1997) recommended giving high priority for the conservation of the large area along the Colombian-Venezuelan border (Rio Negro, Atabapo, and Vichada river basins), which would extend south to merge with existing conservation units in Brazil. Therefore, more conservation strategies and public policies are needed to respect the indigenous peoples’ rights, and at the same time, understand their ancestral role in accomplishing Amazonian forest sustainability for centuries.
References
Abraão MB, Shepard GH Jr, Nelson BW, Baniwa JC, Andrello G, Yu DW (2009) Baniwa vegetation classification in the white-sand Campinarana habitat of the Northwest Amazon. In: Johnson LM, Hunn E (eds) Landscape ethnoecology: concepts of biotic and physical space. Berghahn Books, New York and Oxford, pp 83–115
Almeida A, Futada S, Klein T (2016) Protected areas and indigenous lands in the Amazon region are affected by more than 17,500 mining processes. https://www.socioambiental.org/en/node/5044
Alvez-Valles CM, Balslev H, Garcia-Villacorta R, Carvalho FA, Menini Neto L (2018) Palm species richness, latitudinal gradients, sampling effort, and deforestation in the Amazon region. Acta Bot Bras 32(4):527–539
Anderson AB (1981) White-sand vegetation of Brazilian Amazonia. Biotropica 13(3):199–210
ANLA (2020) Autoridad Nacional de Licencias Ambientales, Minambiente, Bogotá, Colombia. http://www.anla.gov.co/. (Accessed 05 Octuber 2020)
Antonelli A, Zizka A, Antunes Carvalho F, Scharn R, Bacon CD, Silvestro C, Condamine FL (2018) Amazonia is the primary source of neotropical biodiversity. Proc Nat Acad Sci (USA) 115:6034–6039
Arellano-Peña H, Rangel-Ch JO (2015) A solution to the high bias in estimates of carbon held in tropical forest above-ground biomass. arXiv:1508.03667v1 [q-bio.QM]
Arellano-Peña H, Bernal-Gutiérrez G, Calero-Cayopare A, Castro-L F, Lozano A, Bernal-Linares DS, Méndez-R C, Aymard G (2019) The first botanical exploration to the upper Cuiarí (Cuyarí) and Isana rivers, upper Río Negro basin, Guainía department. Colombia Harvard Papers in Botany 24(2):83–102
Aymard G (2001) Estructura y composición florística en bosques de tierra firme del alto Río Orinoco, sector La Esmeralda. Estado Amazonas Venezuela Acta Botánica Venezuelica 23(2):123–156
Aymard, G (in preparation) “Proyecto Inventario de los Recursos Naturales de la Guayana Venezolana” (PIRNRG): Nine years of botanical expeditions (1985–1994). Boletín de Historia de las Geociencias en Venezuela
Aymard G, Stergios B, Cuello N (1989) Informe sobre la vegetación del Interfluvio Orinoco-Atabapo, sector “Los Pozos” (03°10' N; 67°17' O). Departamento Atabapo. Territorio Federal Amazonas, Venezuela. Boletín Técnico (Programa de R. N. R. UNELLEZ-Guanare. Venezuela) 15:170–219
Aymard G, Schargel R, Berry PE, Stergios B (2009) Estudio de los suelos y la vegetación (estructura, composición florística y diversidad) en bosques macrotérmicos no-inundables, estado Amazonas Venezuela (aprox. 01° 30′ - 05° 55′ N; 66° 00′ - 67° 50′ O). Biollania ed esp 9:6–251
Aymard G, Arellano-Peña H, Minorta-C V, Castro-Lima F (2016) First report of Rhabdodendraceae for the vascular flora of Colombia and the upper Río Negro basin, with comments on phytogeography, habitats, and distribution of Rhabdodendron amazonicum. Harv Pap Bot 21:5–21
Baker PA, Fritz SC, Battisti DS, Dick CW, Vargas OM, Asner GP, Martin RE, Wheatley A, Prates I (2020) Beyond refugia: new insights on quaternary climate variation and the evolution of biotic diversity in tropical South America. In: Rull V, Carnaval A (eds) Neotropical diversification. Springer, Berlin, pp 51–70
Ballesteros MM (1995) Estructura, biomasa e inventario de nutrientes de la Caatinga baja Amazónica y su comparación con el bosque de tierra firme (Departamento del Vaupés, Colombia), Tesis MSc Universidad de los Andes, Facultad de Ciencias, Postgrado de Ecología Tropical. Mérida, Venezuela
Berry P, Aymard G (1997) “A historic Portage” revisited. Biollania ed esp 6:263–273
Berry P, Wiedenhoeft AC (2004) Micrandra inundata (Euphorbiaceae), a new species with unusual wood anatomy from black-water river banks in southern Venezuela. Syst Bot 29:125–133
Bongers F, Engelen D, Klinge H (1985) Phytomass structure of natural plant communities on spodosols in southern Venezuela: the bana woodland. Vegetatio 63(1):13–34
Boubli JP (2002) Lowland floristic assessment of Pico da Neblina National Park, Brazil. Plant Ecol 160:149–167
Braun-Blanquet J (1979) Plant sociology, the study of plant communities. Transl by Fueller GD and Conard HS. Mc Graw-Hill, New York
Brewer-Carías C (1988) Cerro de la Neblina: Resultados de la Expedición 1986-1987. In: Fundación para el Desarrollo de las Ciencias Físicas, Matemáticas y Naturales, Caracas. Venezuela
Bush MB, McMichael CN (2016) Holocene variability of an Amazonian hyperdominant. J Ecol 104(5):1370–1378
Bush MB, de Oliveira BPE, Colinvaux PA, Miller MC, Moreno JE (2004) Amazonian paleoecological histories: one hill, three watersheds. Paleoegeography, Palaeoclimatology, Palaeoecology 214:359–393
Cano A, Stevenson PR (2009) Diversidad y composición florística de tres tipos de bosque en la estación biológica Caparú, Vaupés. Revista Colombia Forestal 12:63–80
Capurucho JMG, Borges SH, Cornelius C, Prata EMB, Costa FM, Campos P, Sawakuchi AO, Rodrigues Zular A, Aleixo A, Bates JM, Ribas CC (2020) Patterns and processes of diversification in Amazonian white sand ecosystems: insights from birds and plants. In: Rull V, Carnaval A (eds) Neotropical diversification: patterns and processes. Fascinating Life Sciences, Springer, Cham, pp 245–270
Cárdenas-López D, Barreto Silva JS, Arias García JC, Murcia García UG, Salazar Cardona CA, Méndez Quevedo O (2007) Caracterización ytipificación forestal de ecosistemas en el municipio de Inírida y el corregimiento de Cacahual (Guainía); una zonificación forestall para la ordenación de los recursos. Sinchi y Corporación para el Desarrollo Sostenible del norte y el oriente Amazónico, Bogotá, 252 p
Carvajal de Fr. G. (1848) Descubrimiento del río de las Amazonas según la relación hasta ahora inédita del viaje de Francisco de Orellana. (Edited by J. T. Medina.) Imp. E. Rasco, Sevilla, España
Chave J, Andalo C, Brown S, Cairns MA, Chambers JQ, Eamus D, Fölster H, Fromard F, Higuchi N, Kira T, Lescure JP, Nelson BW, Ogawa H, Puig H, Riéra B, Yamakura T (2005) Tree allometry and improved estimation of carbon stocks and balance in tropical forests. Oecologia 145:87–99
Cintra JP (2011) Magni Amazoni Fluvii: o mapa do Conde de Pagan. Anais do 1° Simpósio Brasileiro de Cartografia Histórica. Belo Horizonte, Brasil 1:1–20
Cintra JP (2012a) O mapa das cortes e as fronteiras do Brasil. Boletim Ciências Geodésicas, sec Artigos (Curitiba) 8(3):421–445
Cintra JP (2012b) Cartography and historical maps: techniques, applications and peculiarities. Revista Brasileira de Cartografia (Rio de Janeiro) 64(6):901–918
Cintra JP, de Oliveira RH (2014) Nicolas Sanson and his map: the course of the Amazon River. Acta Amazon 44(3):353–366
Cintra JP, Freitas JC (2011) Sailing down the Amazon River: La Condamine’s map. Surv Rev 43:550–566
Cintra JP, Furtado JF (2011) A Carte de l’Amérique Méridionale de Bourguignon D’Anville: eixo perspectivo de uma cartografia amazónica comparada. Revista Brasileira de História 31:273–316
Clark H, Liesner R, Berry P, Fernández A, Aymard G, Maquirino P (2000) Catálogo anotado de la flora del área de San Carlos de Río Negro, Venezuela. Scientia Guaianae 11:101–316
Clinebell RR, Phillips OL, Gentry AH, Stark N, Zuurihg H (1995) Prediction of neotropical tree and liana species richness from soil and climatic data. Biodivers Conserv 4:56–90
Colinvaux PA, de Oliveira PE, Bush MB (2000) Amazonian and neotropical plant communities on glacial time-scales: the failure of the aridity and refuge hypotheses. Quat Sci Rev 19:141–169
Condit R, Hubbell SP, Lafrankie JV, Sukumar R, Manokaran N, Foster BR, Ashton PS (1996) Species-area and species-individual relationships for tropical trees: a comparison of three 50-ha plots. J Ecol 84(4):549–562
Coomes DA, Grubb PJ (1996) Amazonian caatinga and related communities at La Esmeralda, Venezuela: forest structure, physiognomy and floristics, and control by soils factors. Vegetatio 122:167–191
Cordeiro RC, Turcq BJ, Sifeddine A, Lacerda LD, Silva Filho EV, Gueiros BB, Cunha YPP, Santelli RE, Pádua EO, Pachinelam SR (2011) Biogeochemical indicators of enviromnental changes from 50 ka to 10 ka. Palaeogeogr Palaeoclimatol Palaeoecol 299:426–436
Córdoba MP, Etter A (2001) Flora Puinawai. In: Etter A, Baptsite LG, Córdoba M, Muñoz Y, Repizzo A, Romero M, Álvarez M, Escobar F, Fernández F, Mendoza H, Rojas A (eds) Puinawai y Nukak. Caracterización ecológica de dos reservas nacionales naturales de la Amazonia Colombiana. Pontificia Universidad Javeriana, Colciencias, Inst A von Humboldt y Fundación FES, Bogotá, Colombia, pp 102–107
Cortesão J (1965) História do Brasil nos velhos mapas. Tomos 1 e 2. Ministério das Relações Exteriores, Instituto Rio Branco, Rio de Janeiro, Brasil
Cuevas E, Medina E (1986) Nutrient dynamics within Amazonian forest ecosystems I. nutrient flux in fine litter fall and efficiency of nutrient utilization. Oecologia 68:466–472
Cuevas E, Medina E (1991) Phosphorus/nitrogen interactions in adjacent Amazon forests with contrasting soils and water availability. In: Tiessen H, López-Hernández D, Salcedo TH (eds) Regional workshop 3: south and Central America. Saskatchewan Institute of Pedology, pp 84–94
D'Apolito C, Absy ML, Latrubesse EM (2017) The movement of pre-adapted cool taxa in north-Central Amazonia during the last glacial. Quat Sci Rev 169:1–12
de La Condamine CM (1745) Relation Abrégée d’un Voyage fait dans l’Intériéur de l’Amérique Méridionale, Depuis la Côtes de la Mer du Sud, jusqu’aux Côtes du Brésil & de la Guyane, en Descendant la Riviere des Amazones. Veuve Pissot, Paris, France
Dexter K, Chave J (2016) Evolutionary patterns of range size, abundance and species richness in Amazonian angiosperm trees. PeerJ 4:e2402. https://doi.org/10.7717/peerj.2402
Dezzeo N, Maquirino P, Berry PE, Aymard G (2000) Principales tipos de bosques en el área de San Carlos de Río Negro, Venezuela. Scientia Guaianae 11:15–36
Draper F, Costa FRC, Arellano G, Phillips OL, Duque A, Macía MJ, ter Steege H, Asner GP, Aymard G et al (2021) Amazon tree dominance across forest strata. Nature Ecol Evol. https://doi.org/10.1038/s41559-021-01418-y
Dubroeucq D, Volkoff B (1998) From Oxisols to spodosols and histosols: evolution of the soil mantles in the Rio Negro basin (Amazonia). Catena 32:245–280
Ducke A (1938) A Flora do Curicuriarí, afluente do Rio Negro, observada em viagens com a comissão demarcadora das fronteiras do setor Oeste. In: Anais da Primeira Reuniao Sul-Americana de Botanica 3 (1):389-398. Rio de Janeiro
Ducke A (1944) Flora do Rio Urubú (observações realizadas durante as viagens da comissão que escolheria as tierras para a colônia agricola nacional do Amazonas). Boletim Ministerio da Agricultura, Servico Florestal do Brasil, Rio de Janeiro
Ducke A (1954) Notas sobre a fitogeografia Amazônia Brasileira. Boletim técnico do Instituto Agronômico do Norte 29:1–62
Duivenvoorden JF, Cavelier J, García A, Grandez C, Macia M, Romero-Saltos H, Sánchez M, Valencia R (2005) Density and diversity of plants in relation to soil nutrient reserves in well-drained upland forests in NW Amazonia. Biologiske Skrifter 55:25–35
Espinoza JC, Ronchail J, Guyot JL, Cochonneau G, Naziano F, Lavado W, de Oliveira E, Pombosa R, Vauchel P (2009) Spatio-temporal rainfall variability in the Amazon basin countries (Brazil, Peru, Bolivia, Colombia, and Ecuador). Int J Climatol 29(11):1574–1594
Fine PV, Miller ZJ, Mesones I, Irazuzta S, Appel HM, Stevens MH, Sääksjärvi I, Schultz JC, Coley PD (2006) The growth-defense trade-off and habitat specialization by plants in Amazonian forests. Ecology 87(7):S150–S162
Fine PV, García-Villacorta R, Pitman NCA, Mesones I, Kembel SW (2010) A floristic study of the white-sand forests of Peru. Ann Mo Bot Gard 97:283–305
ForestPlots.net, Blundo C, Carilla J, Grau R, Malizia A, Malizia L, Osinaga-Acosta O et al (554 co-authors) (2021) Taking the pulse of Earth’s tropical forests using networks of highly distributed plots. Biol Conserv. https://doi.org/10.1016/j.biocon.2020.108849
Franco W, Dezzeo N (1994) Soils and water regime in the terra firme-caatinga forest complex near San Carlos de Río Negro, state of Amazonas. Venezuela Interciencia 19(6):305–316
Fritz S (1717) Description abrégée du Maragnon, et des missions établies aux environs de ce grand fleuve, tirée d’un mémoire espagnole du père Samuel Fritz, missionaire de la Compagnie de Jésus. Lettres Édifiantes et Curieuses, Écrites des Missions Etrangeres, par Quelques Missionaires de la Compagnie de Jesus 12:212–231
Fritz S (1726) Beschreibung des Fluß Maragnon und deren Missionen. Der Neuer Welt-Bott 5(111):59–61
Fritz S (1755) Descripcion abreviada del rio Marañón, y de las missions establecidas en sus contornos—Sacada de una memoria española del padre Samuèl Fritz, Missionero de la Compañia de Jesus. Cartas edificantes, y Curiosas, Escritas de las Missiones Estrangeras, por Algunos Missioneros de la Compañia de Jesus [Traducidas del idioma francés por el padre Diego Davin, de la Compañia de Jesus] 8:42-50
Fritz S (1819) Description abrégée du Maragnon, et des missions établies aux environs de ce grand fleuve, tirée d’un mémoire espagnole du père Samuel Fritz, missionaire de la Compagnie de Jésus. Lettres édifiantes et curieuses, écrites des Missions Étrangères Nouvelle Édition. Mémoires d’Amérique [Lyon] 5:172–180
García-Villacorta R, Dexter KG, Pennington T (2016) Amazonian white-sand forests show strong floristic links with surrounding oligotrophic habitats and the Guiana shield. Biotropica 48:47–57
Gentry AH (1982) Neotropical floristic diversity: phytogeographical connections between central and South America: Pleistocene climatic fluctuations or an accident of the Andean orogeny? Ann Mo Bot Gard 69:557–593
Gentry AH (1988a) Tree species richness of upper Amazonian forests. Proceedings of the National Academy of Sciences USA 85:156–159
Gentry AH (1988b) Changes in plant community diversity and floristic composition on environmental and geographical gradients. Ann Mo Bot Gard 75:1–34
Gentry AH (1993) Diversity and floristic composition of lowland forest in Africa and South America. In: Goldblatt P (ed) Biological relationships between Africa and South America. Yale University Press, New Haven, pp 500–547
Goulding M, Leal Carvalho M, Ferreira EG (1988) Rio Negro, rich life in poor water (Amazonian diversity and foodchain ecology as seen through fish communities). SPB Academic Publishing, The Netherlands
Häggi C, Chiessi CM, Merkel U, Mulitza S, Prange M, Schulz M, Schefuß E (2017) Response of the Amazon rainforest to late Pleistocene climate variability. Earth Planet Sci Lett 479:50–59
Haykin S (2009) Neural networks and learning machines, 3rd edn. Pearson Prentice Hall, Upper Saddle River, New Jersey, USA
Herrera R (1979) Nutrient distribution and cycling in an Amazon Caatinga rainforest on Spodosols in southern Venezuela. PhD. thesis. University of Reading
Herrera R (1985) Nutrient cycling in Amazonian forests. In: Prance GT, Lovejoy TE (eds) Key environments: Amazonia. Pergamon Press, Oxford, pp 95–105
Hill MO (1979) TWINSPAN-a Fortran program for arranging multivariate data in an ordered two-way table of classification of individuals and attributes. Cornell University, Ithaca, NY
Hofhansl F, Chacón-Madrigal E, Fuchslueger L, Jenking D, Morera-Beita A, Plutzar C, Silla F, Andersen KM, Buchs DM, Dullinger S, Fiedler K, Franklin O, Hietz P, Huber W, Quesada CA, Rammig A, Schrodt F, Vincent AG, Weissenhofer A, Wanek W (2020) Climatic and edaphic controls over tropical forest diversity and vegetation carbon storage. Sci Rep 10:5066. https://doi.org/10.1038/s41598-020-61868-5
Hooghiemstra HT, van der Hammen T, Cleef AM (2002) Paleoecología de la flora boscosa. In: Guariguata MR, Kattan GH (eds) Ecología y conservación de bosques Neotropicales. Editorial Libro Universitario Regional, Costa Rica, pp 43–58
Hooghiemstra HT, Wijninga VM, Cleef AM (2006) The paleobotanical record of Colombia; implications for biogeography and biodiversity. Annals of Missouri Botanical Garden 93:297–325
Hoorn C, Wesselingh FP, ter Steege H, Bermudez MA, Mora A, Sevink J, Sanmartín I, Sánchez-M A, Anderson CL, Figueiredo JP, Jaramillo C, Riff D, Negri RF, Hooghiemstra HT, Lundberg J, Stadler T, Särkinen T, Antonelli A (2010) Amazonia through time: Andean uplift, climate change, landscape evolution, and biodiversity. Science 330:927–931
Hoorn C, Bogotá-A GR, Romero-Báez M, Lammertsma E, Flantua SGA, Dantas EL, Dino R, do Carmo DA, Chemale F Jr (2017) The Amazon at sea: onset and stages of the Amazon river from a marine record, with special reference to Neogene plant turnover in the drainage basin. Glob Planet Chang 153:51–65
Hopkins MJG (2019) Are we close to knowing the plant diversity of the Amazon? An Acad Bras Cienc 91(3):1–7. https://doi.org/10.1590/0001-3765201920190396
Householder JE, Schöngart J, Piedade MTF, Junk WJ, ter Steege H, Montero JC, de Assis RL, de Aguiar DPP, Pombo MM, Quaresma AC, Demarchi LO, Parolin P, Lopes A, Feitoza GV, Durgante FM, Albuquerque BW, Chu A, Enßlin D, Fabian T, Fettweiß K, Hirsch M, Hombach M, Hubbuch A, Hutter B, Jäger T, Kober-Moritz R, Lindner MKR, Maier F, Nowak J, Petridis Z, Schierling L, Snjaric E, Egger G, Schneider E, Damm C, Wittmann F (2021) Modeling the ecological responses of tree species to the flood pulse of the Amazon Negro River floodplains. Front Ecol Evol 9:628606. https://doi.org/10.3389/fevo.2021.628606
Hubbell SP, He F, Condit R, Borda-de Agua L, Kellner J, ter Steege H (2008) How many tree species are there in the Amazon and how many of them will go extinct? Proc Nat Acad Sci USA 105:11498–11504
Huber O (1987) Consideraciones sobre el concepto de Pantepui. Pantepui 2:2–10
Huber O (1994) Recent advances in the phytogeography of the Guayana region, South America. Mémoires de la Société de Biogéographie 4:53–63
Huber O (1995a) History of botanical exploration. In: Berry PE, Holst B, Yatskievych K (eds) Flora of Venezuelan Guayana, vol I. Timber Press, Portland, Oregon, pp 63–95
Huber O (1995b) Geographical and physical features. In: Berry PE, Holst B, Yatskievych K (eds) Flora of Venezuelan Guayana, vol I. Timber Press, Portland, Oregon, pp 1–62
Huber O (1995c) Vegetation. In: Berry PE, Holst B, Yatskievych K (eds) Flora of Venezuelan Guayana, vol I. Timber Press, Portland, Oregon, pp 97–192
Huber O, Wurdack JJ (1984) History of botanical exploration in Territorio Federal Amazonas, Venezuela. Smithson Contrib Bot 56:1–86
IGAC (2014) Departamento del Guainía: Estudio general de suelos y zonificación de la tierra, escala 1:100.000/Instituto Geográfico Agustín Codazzi. Subdirección de Agrología. Imprenta Nacional de Colombia, Bogotá
IGAC (2018) Estudio general de suelos y zonificación de tierras departamento de Vichada, escala 1.100.000/Instituto Geográfico Agustín Codazzi. Subdirección de Agrología. Imprenta Nacional de Colombia, Bogotá
Janzen DH (1974) Tropical Blackwater rivers, animals and mass fruiting by the Dipterocarpaceae. Biotropica 6:69–103
Junk WJ, Piedade MTF, Schöngart J, Cohn-Haft M, Adeney JM, Wittmann FA (2011) Classification of major naturally-occurring Amazonian lowland wetlands. Wetlands 31:623–640
Klinge H (1965) Podzol soils in the Amazon basin. J Soil Sci 16(1):96–103
Klinge H (1967) Podzol soils: a source of Blackwater rivers in Amazonia. Atas do Simpósio sobre a Biota Amazônica 3:117–125
Klinge H, Herrera R (1983) Phytomass structure of natural plant communities on spodosols in southern Venezuela: the tall Amazon caatinga forest. Plant Ecol 53:65–84
Klinge H, Medina E (1979) Rio Negro caatingas and Campinas, Amazonas states of Venezuela and Brazil. In: Specht RL (ed) Heathlands and related shrublands. Elsevier Scientific Publications, New York, pp 483–488
Klinge H, Medina E, Herrera R (1977) Studies on the ecology of Amazon caatinga forest in southern Venezuela. Part 1: general features. Acta Cientifica Venezolana 28:270–276
Kristiansen T, Svenning JC, Grandez C, Salo J, Balslev H (2009) Commonness of Amazonian palm (Arecaceae) species: cross-scale links and potential determinants. Acta Oecol 35(4):554–562
Kubitzki K (1989) The ecogeographical differentiation of Amazonian inundation forests. Plant Syst Evol 162:285–304
Kubitzki K (1990) The psammophilous flora of northern South America. Mem N Y Bot Gard 64:248–253
Kubitzki K (1991) Dispersal and distribution in Leopoldinia (Palmae). Nord J Bot 11:429–432
Landrum LR (1986) The life and botanical accomplishments of Boris Alexander Krukoff. Adv Econ Bot 2:1–96
Latrubesse EM, Stevaux JC (2015) The Anavilhanas and Mariuá archipelagos: fluvial wonders from the Negro River, Amazon Basin. In: Carvalho-Viera B, Rodrigues-Salgado AA, Cordeiro-Santos L (eds) Landscapes and landforms of Brazil. World geomorphological landscapes series. Springer, Dordrecht, Heidelberg, New York, London, pp 157–170
Levis C, Costa FRC, Bongers F, Peña-Claros M, Clement CR, Junqueira AB, Neves EG, Tamanaha EK, Figueiredo FOG, ter Steege ter H et al. (2017) Persistent effects of pre-Columbian plant domestication on Amazonian forest composition. Science 355(6328):925–931
Lisbôa PL (1975) Estudos da vegetação das Campinas Amazonica - II. Observaçôes gerais e revisão bibliográfica sobre as campinas amazónicas de areia branca. Acta Amazon 5:211–223
Lleras E (1997) Upper Rio Negro region (Brazil, Colombia, Venezuela). In: Davies SD, Heywood VH, Herrera-MacBryde O, Villa-Lobos J, Hamilton AC (eds) Centres of plant biodiversity. Volumen 3: The Americas. World Wildlife Fund for Nature & The World Conservation Union, Cambridge, UK, pp 333-337
Lopes de Sousa B (1955) Indios e exploraçoes geográficas. Publicação no. 110. Conselho Nacional de Proteção aos Índios, Ministério da Agricultura, Rio de Janeiro, Brasil
Lopes de Sousa B (1959) Do Rio Negro ao Orenoco (A Terra-O Homen). Publica no. 111. Conselho Nacional de Proteção aos Índios, Ministério da Agricultura, Rio de Janeiro, Brasil
Luize BG, Lima Magalhães JÃL, Queiroz H, Lopes MA, Venticinque EM, de Moraes Novo EML, Silva TSF (2018) The tree species pool of Amazonian wetland forests: which species can assemble in periodically waterlogged habitats? PLosOne 13(5):1–13
Mägdefrau K, Wutz A (1961) Leichthölzer und Tonnenstämme in Schwarzwassergebieten und Dornbuschwäldern des tropischen Südamerika. Forstwissenschaftliches Centralblatt 80:17–28
Marengo JA, Williams RE, Alves LM, Soares WR, Rodriguez DA (2016) Extreme seasonal climate variations in the Amazon basin: droughts and floods. In: Nagy L, Forsberg BR, Artaxo P (eds) Interactions between biosphere, atmosphere and human land use in the Amazon basin. Ecological studies 227. Springer, Heidelberg, Berlin, pp 54–76
McCune B, Mefford MJ (2016) PC-ORD multivariate analysis of ecological data, Version 7.0. Wild Blueberry Media, Corvalis, Oregon, USA
Medina E (2000) El Proyecto Amazonas del Instituto Venezolano de Investigaciones Científicas: Origen y desarrollo. Scientia Guaianae 11:1–6
Medina E, Garcia V, Cuevas E (1990) Sclerophylly and oligotrophic environments: relationships between leaf structure, mineral nutrient content, and drought resistance in tropical rain forests of the upper Río Negro region. Biotropica 22:51–64
Meggers BJ (1979) Climatic oscillation as a factor in the prehistory of Amazonia. Am Antiq 44(2):252–266
Meggers BJ (1994) Archeological evidence for the impact of mega-Niño events on Amazonia during the past two millennia. Climate Change 28:321–338
Montoya E, Lombardo U, Levis C, Aymard G, Mayle FE (2020) Human contribution to Amazonian diversity: pre-Columbian legacy to current plant communities. In: Rull V, Carnaval A (eds) Neotropical diversification. Springer, Berlin, pp 495–520
Morley RJ (2011) Cretaceous and tertiary climate change and the past distribution of megathermal rainforests. In: Bush MB, Flenley JR, Gosling WD (eds) Tropical rainforest responses to climatic change, 2nd edn. Springer-Verlag, Berlin-Heidelberg, pp 1–34
Moyersoen B (1993) Ectomicorrizas y micorrizas vesículo-arbuscalares en caatinga Amazónica del sur de Venezuela. Scientia Guianeae 3:1–83
Nascimento MN, Renato-C GSM, Turcq CB, Moreira LS, Bush MB (2019) Vegetation response to climatic changes in western Amazonia over the last 7,600 years. J Biogeogr 46(11):2389–2406
Nelson WB, Ferreira CAC, da Silva MF, Kawasaki ML (1990) Endemism centres, refugia and botanical collection density in Brazilian Amazonia. Nature 345:714–716
Neves EG (1998) Paths in dark waters: archaeology as indigenous history in the upper Rio Negro basin, Northwest Amazon. PhD thesis, Indiana University, USA
Olivares I, Svenning JC, van Bodegom PM, Balslev H (2015) Effects of warming and drought on the vegetation and plant diversity in the Amazon Basin. Bot Rev 81(1):1–28
Oliveira-Filho AT, Dexter KG, Pennington RT, Simon MF, Bueno ML, Neves DM (2021) On the floristic identity of Amazonian vegetation types. Biotropica. https://doi.org/10.1111/btp.12932
Pagan BF (1655) Relation Historique et Géographique de la Grande Rivière des Amazones dans l’ Amérique. Gardin Bensogne, Paris, France
Pennington RT, Lavin L, Prado DE, Pendry CA, Pell SK, Butterworth CA (2004) Historical climatic change and speciation: neotropical seasonally dry forest plants shows pattern of both tertiary and quaternary diversifications. Philos Trans Royal Soc B London 359:515–538
Peñuela MC (2014) Understanding Colombian Amazonian white sand forests. PhD Thesis, Utrecht University, The Netherlands
Pereira da Silva J (2008) Carta e inventario enviados per A. Rodrigues-Ferreiradesde Barcelos el 17 de abril de 1786. In: Monteiro Soares JP Ferrão C (ed) Viagem ao Brasil de Alexander Rodrigues Ferreira volume 3. [Correspondência 1a parte (1779–1788); “Transcrição, Establecimiento do Texto e Notas” de J. Pereira da Silva] Kapa Editorial, Petrópolis, Brasil, pp 80–86
Phillips O, Miller JS (2002) Global patterns of plant diversity: Alwyn H Gentry’s forest 980 transect data set Monographs Systematic Botany 89:1-319. Missouri Botanical Press
Pitman NC, Mogollón H, Dávila N, Ríos M, García-Villacorta R, Guevara JE, Baker T, Monteagudo A, Phillips O, Vásquez-Martínez R, Ahuite M, Aulestia M, Cárdenas-López D, Cerón C, Loizeau PA, Neill D, Nuñez P, Palacios W, Spichiger R, Valderrama R (2008) Tree community change across 700 km of lowland Amazonian forest from the Andean foothills to Brazil. Biotropica 40:525–535
Pombo de Souza MM (2012) Estrutura e composicao das comunidades de arvores na bacia do alto rio Negro. Master’s Thesis, Instituto Nacional de Pesquisas da Amazônia-INPA, Manaus, Brasil
Prance GT (1980) A terminologia dos tipos de florestas amazonicas sujeitas a inundaçao. Acta Amazon 10:495–504
Prance GT (1989) American tropical forests. In: Lieth H, Werger MJA (eds) Tropical rain Forest ecosystems, ecosystems of the world 14B. Elsevier, Amsterdam, The Nerderlands, pp 99–132
Prance GT (2001) Amazon ecosystems. In: Asher-Levin S (ed) Encyclopedia of biodiversity, A-C, vol 1. Academic Press, New York, pp 145–157
Prance GT, Nelson BW, Freitas da Silva M, Daly DC (1984) Projeto Flora Amazônica: eight years of binational botanical expeditions. Acta Amazonica 14(Supl. 1/2):5–29
Putz FE (1983) Liana biomass and leaf area of a “terra firme” forest in the Rio Negro basin, Venezuela. Biotropica 15:185–189
Putz FE, Mooney HA (1991) The biology of vines. Cambridge University Press, Cambridge
Quesada CA, Lloyd J, Anderson LO, Fyllas NM, Schwarz M, Czimczik CI (2011) Soils of Amazonia with particular reference to the RAINFOR sites. Biogeosciences 8:1415–1440
Quesada CA, Phillips OL, Schwarz M, Czimczik CI, Baker TR, Patino S, Fyllas NM, Hodnett MG, Herrera R, Almeida S, Alvarez-Dávila E, Arneth A, Arroyo L, Chao KJ, Dezzeo N, Erwin T, di Fiore A, Higuchi N, Honorio Coronado E, Jimenez EM, Killeen T, Torres-Lezama A, Lloyd G, Lopez-González G, Luizão FJ, Malhi Y, Monteagudo A, Neill DA, Nuñez Vargas P, Paiva R, Peacock J, Peñuela MC, Peña Cruz A, Pitman N, Priante Filho N, Prieto A, Ramírez H, Rudas A, Salomão R, AJB S, Schmerler J, Silva N, Silveira M, Vásquez R, Vieira I, Terborgh J, Lloyd J (2012) Basin-wide variations in Amazon forest structure and function are mediated by both soils and climate. Biogeosciences 9:2203–2246
Rice HA (1910) The river Uaupés. Geogr J 35(6):682–700
Rice HA (1914) Further explorations in the North-Western Amazon Valley. Geogr J 44(2):137–168
Rice HA (1918) Notes on the Rio Negro (Amazonas). Geogr J 52(4):205–218. [“The journey… was made during the first three months of 1917”]
Rice HA (1921) The Río Negro, the Casiquiare Canal, and the upper Orinoco, September 1919-April 1920. Geogr J 63(5):321–244
Rice HA (1928) The Rio Branco, Uraricuera, and Parima. Geogr J 71(2):113–143
Richards PW (1952) The tropical rain forest. Cambridge University Press, Cambridge
Ricklefs RE (1987) Community diversity: relative roles of local and regional processes. Science 235:167–171
Riina R, Berry PE, Huber O, Michelangeli FA (2019) Pantepui plant diversity and biogeography. In: Rull V, Vegas-Vilarrúbia T, Huber O, Señaris J (eds) Biodiversity of Pantepui: the pristine “lost world” of the neotropical Guiana highlands. Academic Press, Cambridge, pp 121–147
Ríos-Villamizar EA, Adeney JM, Junk WJ, Piedade MTF (2020) Physicochemical features of Amazonian water typologies for water resources management. IOP Conf Series: Earth and Environmental Science 427:012003. https://doi.org/10.1088/1755-1315/427/1/012003
Rodrigues WA (1961) Aspectos fitosociológicos das caatingas do rio Negro. Boletim do Museu Paraense Emílio Goeldi (Botânica) 15:3–41
Rodriguez-Zorro PA, Turcq B, Cordeiro RC, Moreira LS, Costa RL, McMichael CH, Behling H (2018) Forest stability during the early and late Holocene in the igapó floodplains of the Rio Negro, northwestern Brazil. Quat Res 89(1):75–89
Romero-G GA, Gómez-D CA, González-Ñañez AG (2019) Las cuatro “Yavitas”. Boletín de historia de las Geociencias en Venezuela 131:1–133
Rudas A, Prieto A, Rangel JO (2002) Principales tipos de vegetación de La Ceiba (Guainía), Guayana Colombiana. Caldasia 24:243–365
Ruiz-Pessenda LC, de Oliveira PE, Mofatto M, Brito de Medeiros V, Francischetti- García RJ, Aravena R, Bendassoli JA, Zuniga-Leite A, Saad AR, Etchebehere ML (2009) The evolution of a tropical rainforest/grassland mosaic in southeastern Brazil since 28,000 14C yr BP based on carbon isotopes and pollen records. Quat Res 71:437–452
Sabatier D, Grimaldi M, Prévost MF, Guillaume J, Gedron M, Desso M, Curmi P (1997) The influence of soil cover organization on the floristic and structural heterogeneity of a Guianan rain forest. Plant Ecol 131:81–108
Salamanca S (1983) La vegetación de la Orinoquia-Amazonia: fisiografía y formaciones vegetales. Colombia Geográfica 10:5–31
Saldarriaga JG, West DC (1986) Holocene fires in the northern Amazon basin. Quat Res 26:358–366
Sanford RL, Cuevas E (1996) Root growth and rhizosphere interactions in tropical forests. In: Mulkey SS, Chazdon RL, Smith AP (eds) Tropical forest plant ecophysiology. Chapman & Hall, USA, pp 268–300
Sanford RL, Saldarriaga JG, Clark KE, Uhl C, Herrera R (1985) Amazon rain-forest fires. Science 227:53–55
Santos JOS, Potter PE, Reis NJ, Hartmann LA, Fletcher IR, McNaughton NJ (2003) Age, source, and regional stratigraphy of the Roraima supergroup and Roraima-like outliers in northern South America based on U-pb geochronology. Geol Soc Am Bull 115(3):331–348
Schargel R, Marvez P (2009) Suelos. En: Aymard G (ed) Estudio de los suelos y la vegetación (estructura, composición florística y diversidad) en bosques macrotérmicos no-inundables, estado Amazonas Venezuela (aprox. 01° 30′ - 05° 55’ N; 66° 00′ -- 67° 50’ O). Biollania ed esp 9:99–108
Schargel R, Aymard A, Berry P (2000) Características y factores formadores de Spodosoles en el sector Maroa-Yavita, Amazonía Venezolana. Revista UNELLEZ de Ciencia y Tecnología 18(1):85–96
Schultes RE (1983) Richard Spruce: an early ethnobotanist and explorer of the Northwest Amazon and northern Andes. J Ethnobiol 3(2):139–147
Schulman L, Toivonen T, Ruokolainen K (2007) Analysing botanical effort in Amazonia and correcting for it in species range estimation. J Biogeogr 34:1388–1399
Silva-Souza KJP, Souza AF (2020) Woody plant subregions of the Amazon forest. J Ecol 108(6):2321–2335. https://doi.org/10.1111/1365-2745.13406
Spruce R (1908) Notes of botanist on the Amazon and Andes (being records of travel on the Amazon and its tributaries, the Trombetas, Rio Negro, Uaupés, Casiquiare, Pacimoni, Huallaga and Pastasa; as also to the cataracts of the Orinoco along the eastern side of the Andes of Peru and Ecuador, and the shores of the Pacific, during the years 1849–1864). Volume edited by AR Wallace. The Macmillan Company, London
Steyermark JA (1982) Relationships of some Venezuelan forest refuges with lowland tropical floras. In: Prance DT (ed) Biological diversification in the tropics. Columbia University Press, New York, pp 182–220
Stokes MF, Goldberg SL, Perron JT (2018) Ongoing river capture in the Amazon. Geophys Res Lett 45:5545–5552
Stropp J, ter Steege H, Malhi Y, ATDN and RAINFOR (2009) Disentangling regional and local tree diversity in the Amazon. Ecography 32:46–54
Stropp J, van der Sleen P, Assunção PA, Lopes da Silva A, ter Steege H (2011) Tree communities of white-sand and terra-firme forests of the upper Rio Negro. Acta Amazon 41:521–544
Stropp J, van der Sleen P, Quesada CA, ter Steege H (2014) Herbivory and habitat association of tree seedlings in lowland evergreen rainforest on white-sand and terra-firme in the upper Rio Negro. Plant Ecol Diversity 7(1–2):255–265
Stropp J, Umbelino B, Correia BRA, Campos-Silva JV, Ladle RJ, Malhado ACM (2020) The ghosts of forests past and future: deforestation and botanical sampling in the Brazilian Amazon. Ecography 43:1–11
Takeuchi M (1961) The structure of the Amazonian vegetation. III Campina forest in the Río Negro region. J Fac Sci, University of Tokyo (Sect Bot 3) 8(2):27–35
Takeuchi M (1962a) The structure of the Amazonian vegetation. IV High Campina forest in the upper Rio Negro. J Fac Sci, University of Tokyo (Sect Bot 3) 8(5):279–288
Takeuchi M (1962b) The structure of the Amazonian vegetation V. Tropical rain forest near Uaupés. J Fac Sci: University of Tokyo (Sect Bot 3) 8(6):289–296
Takeuchi M (1962c) The structure of the Amazonian vegetation. VI igapo. J Fac Sci, University of Tokyo (Sect Bot 3) 8(7):297–304
Tenreiro-Aranha B de F (1906) As exploraçoes e os exploradores do Rio Uaupés. Arquivo do Amazonas, año I, I (2):23–54 (23 de octubre), Manaos, Brasil
Ter Braak CJE (1987) The analysis of vegetation-environment relationships by canonical correspondence analysis. Vegetatio 69:69–77
ter Steege H, Pitman NC, Sabatier D, Castellanos H, van der Hout P, Daly DC, Silveira M, Phillips O, Vasquez R, van Andel T, Duivenvoorden J, de Oliveira AA, Ek R, Lilah R, Thomas R, van Essen J, Baider C, Maas P, Mori S, Terborgh J, Nuñez P, Mogollón H, Morawetz W (2003) A spatial model of tree α-diversity and tree density for the Amazon. Biodivers Conserv 12:2255–2277
ter Steege H, Pitman NC, Phillips O, Chave J, Sabatier D, Duque A, Molino DF, Prévost MF, Spichiger R, Castellanos H, von Hildebrand P, Vasquez R (2006) Continental-scale patterns of canopy tree composition and function across Amazonia. Nature 443(28):444–447
ter Steege H, ATDN (Amazon Tree Diversity Network: collective author) and RAINFOR (The Amazon Forest Inventory Network: collective author) (2010) Contribution of current and historical processes to patterns of tree diversity and composition of the Amazon. In: Hoorn C, Wesseling FP (eds) Amazonia landscape and species evolution: a look into the past. Wiley-Blackwell Publishing Ltd, pp 347–359
ter Steege H, Pitman NC, Sabatier D, Baraloto C, Salomão RP, Guevara JE, Phillips OL, Castilho CV, Magnusson WE, Molino JF, Monteagudo A, Núñez P, Montero JC, Feldpausch et al (2013) Hyper-dominance in the Amazonian tree flora. Science 342:325–335
ter Steege H, Vaessen RW, Cárdenas-López D, Sabatier D, Antonelli A, Mota de Oliveira S, Pitman NCA, Jørgensen PM, Salomão RP (2016) The discovery of the Amazonian tree flora with an updated checklist of all known tree taxa. Sci Rep 6:29549. https://doi.org/10.1038/srep29549
ter Steege H, Mota de Oliveira S, Pitman NCA, Sabatier D, Antonelli A, Guevara Andino JE, Aymard G, Salomão RP (2019a) Towards a dynamic list of Amazonian tree species. Sci Rep 9:3501. https://doi.org/10.1038/s41598-019-40101-y
ter Steege H, Henkel TW, Marimon BS, Pitman NCA, Phillips O, Aymard G, Salomão AR et al (2019b) Rarity of monodominance in hyperdiverse Amazonian forests. Sci Rep 9:13822. https://doi.org/10.1038/s41598-019-50323-9
ter Steege H, Prado HPI, Lima RF, Pos E, de Souza CL, de Andrade Lima Filho D, Salomão RP, Phillips OL, Aymard G et al (2020) Biased-corrected richness estimates for the Amazonian tree flora. Sci Rep 10:10130. https://doi.org/10.1038/s41598-020-66686-3
Tuomisto H, Moulatlet GM, Balslev H, Emilio T, Figueiredo FOG, Pedersen D, Ruokolainen K (2016) A compositional turnover zone of biogeographical magnitude within lowland Amazonia. J Biogeogr 43:2400–2411
Tuomisto H, Van Doninck J, Ruokolainen K, Moulatlet GM, FOG F, Sirén A, Zuquim G (2019) Discovering floristic and geoecological gradients across Amazonia. J Biogeogr 46(8):1734–1748
Uhl C, Murphy P (1981) A comparison of productivities and energy values between slash and burn agriculture and secondary succession in the upper Rio Negro region of the Amazon Basin. Agro-Ecosystems 7(1):63–83
Valencia R, Foster R, Villa G, Condit R, Svenning JC, Hernández C, Romeleroux R, Losos E, Magaard E, Balslev H (2005) Trees species distributions and local habitat variation in the Amazon: a large forest plot in eastern Ecuador. J Ecol 92:214–229
van der Hammen T (1972) Changes in vegetation and climate in the Amazon basin and Sur-rounding areas during the Pleistocene. Geologie Mijnbouw 51:641–643
van der Hammen T (2006) Bases para una prehistoria ecológica amazónica y el caso Chiribiquete. In: Morcote-Ríos G, Mora-Camargo S, Franky-Calvo C (eds) Pueblos y paisajes antiguos de la selva amazónica. Universidad Nacional de Colombia, Facultad de Ciencias, Bogotá, Taraxacum, Smithsonian Institution, Washington DC, pp 29–48
van der Hammen T, Absy ML (1994) Amazonia during the last glacial. Palaeogeogr Palaeoclimatol Palaeoecol 109:247–261
Vareschi V (1963) La bifurcación del Orinoco. Observaciones hidrográficas y ecológicas de la expedición conmemorativa de Humboldt del año 1958. Acta Cient Venez 14(4):98–103
Vazquez F (1881) Relación de lo que Sucedió en la Jornada de Omagua y Dorado hecha por el Gobernador Pedro de Orsúa [“Advertencia Preliminar” de Feliciano Ramírez de Arellano, Marqués de la Fuensanta del Valle]. Sociedad de Bibliófilos Españoles, Madrid
Vitousek PM, Sanford RL (1986) Nutrient cycling in moist tropical forest. Annu Rev Ecol Syst 17:137–167
von Bauer PP (1919) NW-Amazonien: Ein Beitrag zur Geographie Äquatorial-Amerikas. Rudolf M, Rohrer, Brünn
Walker WS, Gorelika SR, Baccinia A, Aragon-Osejo JL, Josse C, Meyerd C, Macedo MN, Augusto C, Rios S, Katan T, Almeida de Souza A, Cuellar S, Llanos A, Zager I, Díaz Mirabal G, Solvika KK, Farina MK, Moutinho P, Schwartzmand S (2020) The role of forest conversion, degradation, and disturbance in the carbon dynamics of Amazon indigenous territories and protected areas. Proc Nat Acad Sci USA 117(6):3015–3025
Wesselingh FP, Hoorn C, Kroonenberg SB, Antonelli A, Lundberg JG, Vonhof HB, Hooghiemstra H (2010) On the origin of Amazonian landscapes and biodiversity: a synthesis. In: Hoorn C, Wesselingh F (eds) Amazonia, landscape and species evolution. Wiley-Blackwell, Oxford, pp 421–432
Westhoff V, van der Maarel E (1973) The Braun-Blanquet approach. In: Whittaker RH (ed) Handbook of vegetation science, vol 5. Junk, The Hague, pp 617–726
Wittmann F, Marques MC, Júnior GD, Budke JC, Piedade MTF, Wittmann de Oliveira A, Montero JC, de Assis RL, Targhetta N, Parolin P, Junk WJ, Householder JE (2017) The Brazilian freshwater wetscape: changes in tree community diversity and composition on climatic and geographic gradients. PLoS One 12(4):1–18
Wurdack JJ (1971) The Melastomataceae collections of A. Rodrigues Ferreira Taxon 20:595–596
Zinck A (1986) El inventario de los recursos naturales de la Guayana en marcha. Pantepui 1:2–16
Zucchi A (2006) Ríos de aguas blancas y negras, asentamientos, organización social y patrones migratorios de grupos Arawacos del alto negro venezolano. In: Morcote-Ríos G, Mora-Camargo S, Franky-Calvo C (eds) Pueblos y paisajes antiguos de la selva amazónica. Universidad Nacional de Colombia. Facultad de Ciencias, Bogotá, Taraxacum, Smithsonian Institution, Washington DC, pp 157–170
Acknowledgments
We gratefully acknowledge the support provided by indigenous communities with particular reference to the Baniwa and Kuripako nations, who have contributed directly and indirectly to build and collect information and scientific field material used in this contribution. The first and last authors also thank Germán Bernal-Gutiérrez, a CEO of –Greenlife– for providing logistic and economic support to set up 12 permanent plots located in faraway and unexplored botanical places in the upper Guianía river basin, Guainía department, Colombia, which made this undertaking possible. Finally, we thank Ernesto Medina and Alfred Zinck for their comments on earlier versions of this text.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
1 Electronic Supplementary Material
Appendices
Appendix 3.1: Phytosociological Classification and Description of the New Alliances and Associations of the Forest Communities in the Rio Negro Region
Phytosociological classification | Physiognomy and composition | Syntaxonomy | Ecology and distribution |
|---|---|---|---|
ORDEN I 1. Parahancornio surrogatae Aldinion latifoliae all. Nov. (Figs. 3.5 and 3.6) Typus: Aldino latifolia - Terminalietum ochroprumnae (this study; Appendix 3.1 (this information is available in the following link: https://1drv.ms/f/s!AsgLjs_JMencgZsGYB0kltyDnwkilg). Lowlands forests of the Parahancornia surrogata - Aldina latifolia alliance | The forest communities of this alliance are of medium to high stature (25–35 m tall), characterized by the presence of trees of: Apocynaceae, Euphorbiaceae, Lauraceae, Fabaceae, Malvaceae, Moraceae, Myristicaceae, and Sapotaceae | This alliance is defined on the basis of 24 samples that included 279 species, 166 genera in 62 families. This syntaxon is named with the elective species Parahancornia surrogata and the dominant legume species Aldina latifolia. Within the elective species are: Calophyllum brasiliense, Glandonia williamsii, Caraipa longipedicellata, Sloanea laurifolia, Hevea nitida, Mollia speciosa, Pachira nitida and Licania mollis. Other species can be found in Appendix 1. This alliance includes two new associations: Aldino latifoliae - Terminalietum ochroprumnae and Caraipo longipedicellatae - Micrandretum spruceii | The Parahancornia surrogata - Aldina latifolia alliance is found on slightly elevated positions in depressions, and on a sandy, somewhat poorly drained Entisols and spodosols. It is exposed to water-logging and also more susceptible to short droughts than other poorly or very poorly drained sandy soils, due to the shallow depth to the coarse sandy saprolite. This alliance is compound by forests on white sands, “terra firme,” and seasonally flooded forests with or without palm dominance. |
1.1 Aldino latifoliae - Terminalietum ochroprumnae assoc. nov. (Figs. 3.5 and 3.6). Typus: Plot no. CC_130, TWINSPAN ID 69. Coordinates datum WGS84 LAT 3.6877, LON -67.4550. Altitude 89 m. Colombia. (Appendix 2; this information is available in the following link: https://1drv.ms/f/s!AsgLjs_JMencgZsGYB0kltyDnwkilg) Forests of terminalia ochroprumna and Aldina latifolia | The forests of terminalia ochroprumna and Pachira nitida are medium to high in stature and density, composed by trees with a DBH average greater than 10 cm. In this association, the following species registered the largest height: Vochysia catingae (26.1 m), Parkia discolor (25.7 m), terminalia ochroprumna (21.3 m), Elaeoluma crispa (18 m), and Aldina latifolia (16 m). The species with the highest physiognomic expression (relativized units) are: Aldina latifolia (5.7%), Calophyllum brasiliense (5.6%), Hevea guianensis (5.1%), Pachira nitida (4.9%), Humiriastrum excelsum (3.3%), Leptabalanus wurdackii (3.1%), Elaeoluma crispa (2.9%) and Mollia lepidota (2.8%). | This association is defined on the basis of 7 samples that included 90 species, 61 genera in 27 families. The name of this syntaxon was based on the elective species, the legume Aldina latifolia and terminalia ochroprumna, the most dominant and exclusive species. In this association, Elaeoluma crispa was registered as exclusive species as well. Within the elective species are: Pachira nitida, Macrolobium multijugum, Mollia lepidota, Peltogyne paniculata, Swartzia sericea, Tachigali paniculata, Humiriastrum excelsum, and Leptobalanus apetalus. Other species can be found in the Appendix 3.1. | This vegetation community consists of low caatingas on white sand and seasonally flooded forests in black water. The forest of the association Aldino latifoliae Terminalietum ochroprumnae was determined based on seven (7) samples located in Colombia (i.g., along the Atabapo river in the border with Venezuela). Detailed information about these localities are found in Appendix 3.2. |
1.2. Caraipo longipedicellatae -Micrandretum spruceii assoc. nov. (Figs. 3.5 and 3.6) Typus: Plot CC_153, TWINSPAN ID 92. Coordinates datum WGS84 LAT 3.0986, LON -67.7889. Altitude 110 m. Colombia. (Appendix 3.1) Forests of Caraipa longipedicellata and Micrandra sprucei | The forests of Caraipa longipedicellata and Micrandra sprucei exhibit medium statures and densities. These are composed of trees with a DBH average greater than 10 cm. In some areas there are some emergent trees of up to 35 m. The highest heights were recorded to Parahancornia negroensis (35 m), P. surrogata (28 m), Discophora guianensis (26.5 m), Caraipa llanorum (21.5 m), and Micrandra sprucei (20 m). Some of the species that present a high physiognomic expression in this syntaxon are: Micrandra sprucei (18.7%), Aspidosperma verruculosum (8.9%), Henriquezia nitida (6.6%), Caraipa longipedicellata (5.4%), Parahancornia surrogata (3.2%), Pradosia schomburgkiana (1.8%), Chrysophyllum amazonicum (1.8%), and Doliocarpus novogranatensis (1.5%). | This association is defined on the basis of 17 samples that included 222 species, 145 genera and 60 families. This syntaxon is named with the elective species Caraipa longipedicellata and the dominant species Micrandra sprucei; Humiria crassifolia and Cyrilla racemiflora were recorded as exclusive species. Micrandra sprucei, Compsoneura debilis, and Caraipa longipedicellata are among the elective species | This association includes caatingas on white sands, “terra firme” forests, and seasonally flooded forests in black water with or without palms dominance over poorly drained sandy soils. A substrate that tolerate considerable differences in drainage classes, probably by the root mat develops in the litter layer. The forest of the association Caraipo longipedicellatae Micrandretum spruceii was described based on 17 samples located in Colombia (e.g., Atabapo and Guasacavi rivers, near of Inirida, Mirití, Mitú towns, and the “Serranía de Naquén”) and Venezuela (e.g., along San Carlos de Río Negro-Solano road). Detailed information about these localities is found in Appendix 3.2. |
2. Oenocarpodo batauae - Eperuion leucanthae all. Nov. (Figs. 3.5 and 3.6) Typus: Micrandro elatae - Micrandretum spruceii (this study; Appendix 3.1) Lowlands forests of the Oenocarpus bataua and Eperua leucantha | The forest communities of this alliance are forests of medium to high stature, reaching to 40 m height. These are characterized by the presence of trees of Apocynaceae, Arecaceae, Lauraceae, Fabaceae, Malvaceae, Moraceae, and Myristicaceae. | This alliance is defined on the basis of 61 samples that included 852 species, 323 genera in 87 families. Oenocarpus bataua (as the elective species) and Eperua leucantha (as the dominant) were chosen to coined the name of this syntaxon. The exclusive species are: Erisma micranthum, Retiniphyllum concolor, and Hevea rigidifolia. Eperua leucantha, Clathrotropis glaucophylla, Macrolobium limbatum, and Micrandra sprucei were registered as elective species. This alliance comprises two new upper Rio Negro typical associations such as the Amazonian caatinga, and the “Yévaro” forests (Micrandrum elatum - Micrandrum spruceae and Oenocarpodo batauae Eperuetum purpureae). | This alliance contains low and high Caatingas on white sands, “terra firme” forests and seasonally flooded forests in black water with or without palms dominance. The Oenocarpus bataua—Eperua leucantha alliance is located on elevated positions in depressions, very poorly drained sandy Spodosols on plains, with similar characteristics to the Spodosols located in the San Carlos de Rio Negro (Herrera 1979, 1985; Schargel and Marvez 2009). |
2.1. Micrandro elatae - Micrandretum spruceii assoc. nov. (Figs. 3.5 and 3.6) Typus: Plot CC_154, TWINSPAN ID 93. Coordinates datum WGS84 LAT 3.0833, LON -67.7833. Altitude 108 m. Colombia. (Appendix 1) Forests of Micrandra elata and Micrandra sprucei | The forests of Micrandra elata and M. sprucei are of medium stature (28 and 25 m) and high density composed of trees with a DBH greater than 10 cm. Calophyllum brasiliense (30 m), Hevea benthamiana (30 m), H. guianensis (30 m), and Sloanea floribunda (30 m) recorded the highest heights in this formation. The species with the biggest dominance expression (relativized units) are: Micrandra sprucei (25.4%), M. elata (6.9%), Caraipa longipedicellata (4.1%), Hevea benthamiana (3.1%), Eperua leucantha (3.0%), E. obtusata (2.8%), Hevea pauciflora (2.7%), Micrandra sprucei (2.7%), and Macrolobium limbatum (2.1%). | This association is defined on the basis of 18 samples that included 255 species, 136 genera in 52 families. This syntaxon was named used Micrandra elata (the elective species) and Micrandra sprucei as the dominant. Chrysophyllum bombycinum and Hevea benthamiana were recorded as exclusive species. Among the elective species are: Micrandra sprucei, M. elata, Macrolobium limbatum, Ficus guianensis, Caraipa longipedicellata, and Mucoa duckei. | This kind of vegetation is composed of low and high caatingas on white sands, “terra firme” forests and seasonally flooded forests over black and white water with or without palms dominance. The forest of the association Micrandrum elatum-Micrandrum spruceanum can be found on areas in upper Rio Negro over poorly drained sandy soils. This community tolerates considerable differences in drainage classes. This association was determined based on 18 samples located in Colombia (e.g., near Inírida, Almidón/La Ceiba area, Chorro Bocón at Inírida river, Campo Alegre at Cuiarí river) and Venezuela (e.g., La Esmeralda, upper Orinoco river, along Maroa-Yavita road). Detailed information on these localities is found in Appendix 3.2. |
2.2. Oenocarpodo batauae - Eperuetum purpureae assoc. nov. (Figs. 3.5 and 3.6) Typus: Plot CC_115, TWINSPAN ID 54. Coordinates datum WGS84 LAT 3.3822, LON -67.3386. Altitude 101 m. Colombia. (Appendix 3.1) Forests of Oenocarpus bataua and Eperua purpurea | The forests of Oenocarpus bataua and Eperua purpurea are medium to tall stature and high density. These are composed of trees with a DBH average diameter greater than 10 cm. The canopy of the forest is composed of trees of between 20 and 40 m with a dense cover and great number of palms. In this association, the following species registered the highest heights: Eperua purpurea (46 m), Erisma japura (40 m), Eschweilera tessmannii (40 m), Monopteryx uaucu (40 m), Pseudoxandra leiophylla (37 m), Vochysia grandis (37 m), and Oenocarpus bataua (25 m). The species with the most physiognomic expression are Eperua purpurea (10.3%), Micrandra sprucei (6.3%), Swartzia parvifolia (4.4%), Eperua leucantha (3.6%), Leopoldinia piassaba (3.2%), Monopteryx uaucu (3.1%), Erisma japura (3.0%), Oenocarpus bataua (2.4%), and Aldina kunhardtiana (2.0%). | This association is defined on the basis of 44 samples that included 761 species, 297 genera in 82 families. The elective species Oenocarpus bataua and the dominant species Eperua purpurea, were chosen to coined the name of this syntaxon. Within the elective species are: Brosimum utile, Minquartia guianensis, Virola elongata, Pseudolmedia laevigata, Clathrotropis glaucophylla, Iryanthera crassifolia, Erisma splendens, Ocotea aciphylla, Roucheria Columbiana, and Leopoldinia piassaba. | This association harbors the low and high caatingas on white sands, “terra firme” forests and seasonally flooded forests over mixed water with palms dominance. The forest of the association Oenocarpodo batauae Eperuetum purpureae can be found on areas in the upper Rio Negro over poorly drained sandy soils. This type of vegetation was described based on samples located in Colombia (e.g., along Atabapo river, Nabuquén at Inírida river, upper Isana river) and Venezuela (Casiquiare channel, near San Carlos de Río Negro). Detailed information on these localities is found in Appendices 3.2, 3.3 and 3.4. |
3. Monopterygo uaucus - Eperuion purpureae all. Nov. (Figs. 3.5 and 3.6) Typus: Aldino heterophyllae Eperuetum leucanthae (this study; Appendix 3.1) Lowlands forests of Monopteryx uaucu and Eperua purpurea | The communities of this alliance are composed of forests of medium to high stature (up to 30–45 m tall), characterized by the presence of emergent trees. According to the abundance, frequency and basal area values, the most important families are Apocynaceae, Arecaceae, Lauraceae, Fabaceae, Malvaceae, Moraceae, and Myristicaceae. | This alliance is defined on the basis of 27 samples, that included 919 species, corresponding to 314 genera in 78 families. Monopteryx uaucu as the elective species, and Eperua purpurea as the dominant were chosen to coined the name of this syntaxon. Within the elective species are: Eperua leucantha, Hevea guianensis, Micrandra sprucei, M. spruceana, Brosimum guianense, Erisma japura, Eschweilera pedicellata, Monopteryx uaucu, Iryanthera laevis, Zygia claviflora, Micropholis guyanensis, and Abarema jupunba. Other species can be shown in Appendix 3.1. | This kind of vegetation assembles low and high Caatingas on white sands and “terra firme” forests. The Monopteryx uaucu—Eperua purpurea alliance hold forests with four vertical layers. This forest was located on a poorly drained sandy Entisol in depression, on a moderately drained Ultisol on a hill, and a poorly drained Inceptisol on a foot-slope of a low hill. Both the Ultisol and the Inceptisol have sandy loam, and sandy clay loam textures below the sandy surface horizon (Schargel and Marvez 2009). |
3.1. Aldino heterophyllae Eperuetum leucanthae assoc. nov. (Figs. 3.5 and 3.6) Typus: Plot S_6, TWINSPAN ID 218. Coordinates datum WGS84 LAT −0.1007, LON -66.8804. Brazil. (Appendix 3.1) Forests of Aldina heterophylla and Eperua leucantha | The forests of Aldina heterophylla and Eperua leucantha are of medium stature and high density. These are composed of trees with a DBH average greater than 10 cm. In some areas there are some emergent trees of up to 46 m; the highest heights were recorded to Scleronema micranthum (46.4 m), Brosimum utile (35 m), Erisma laurifolium (35 m), protium alvarezianum (35 m), Swartzia tomentifera (34.4 m), Eperua purpurea (30 m), E. leucantha (30 m), and Aldina heterophylla (28.1 m). Several species presented higher physiognomic expression in this syntaxon (relativized units) such as: Eperua leucantha (13.1%), E. purpurea (9.4%), Monopteryx uaucu (7.3%), Aldina heterophylla (4.7%), Clathrotropis macrocarpa (3.6%), Micrandra spruceana (3.0%), Scleronema micranthum (2.2%), Swartzia polyphylla (1.6%), and Swartzia tomentifera (1.3%). | This association is defined on the basis of 19 samples that included 716 species, 262 genera in 70 families. This syntaxon is named used Aldina heterophylla (the exclusive species) and Eperua leucantha (the dominant species). In this vegetation pattern, Chamaecrista adiantifolia, Sloanea obtusifolia, Himatanthus obovatus, Pradosia schomburkiana, and Vitex sprucei were identified as exclusive species. Within the elective species are: Swartzia tomentifera, Trattinnickia glaziovii, Ocotea rhynchophylla, Sandwithia guyanensis, Brosimum rubescens, Pouteria cuspidata, Bocageopsis pleiosperma, Virola calophylla, Erisma calcaratum, Micrandra spruceana, Hevea guianensis, Chimarrhis duckeana, Couma guianensis, and Cyrillopsis paraensis. | This kind of vegetation harbors low caatingas on white sands and “terra firme” forests. The forest of the association Aldino heterophyllae - Eperuetum leucanthae can be found on areas in the upper Rio Negro growing on poorly drained sandy soils; it tolerates considerable differences in drainage class. This association was determined based on 19 samples located in Brazil (e.g., São Gabriel da Cachoeira, middle Içana river, Pico da Neblina National Park), Colombia (e.g., Punta de Tigre, upper Isana river) and Venezuela (i.g. Mawarinuma river at the base of “Sierra de la Neblina”) as well. Detailed information on these localities is found in Appendix 3.2. |
3.2. Eperuo purpureae - Erismetum japurae assoc. nov. (Figs. 3.5 and 3.6) Typus: Plot P6, TWINSPAN ID 191. Coordinates datum WGS84 LAT 2.2071, LON -68.2781. Colombia. (Appendix 3.1) Forests of Eperua purpurea and Erisma japura | The forests of Eperua purpurea and Erisma japura have a medium to tall stature, high density and great number of palms. The trees have a DBH average greater than 10 cm. In this association the following species have the highest heights: Pouteria ucuqui (45 m), Eschweilera collina (44 m), Brosimum utile (40 m), Micropholis brochidodroma (40 m), Swartzia floribunda (40 m), Allantoma lineata (35 m), Ecclinusa ramiflora (35 m), and Goupia glabra (35 m). The species with most physiognomic expression (relativized units) are: Erisma japura (18.3%), Eperua purpurea (13.9%), Goupia glabra (3.3%), Heterostemon conjugatus (2.9%), Ecclinusa ramiflora (2.9%), Swartzia pinnata (2.5%), Sandwithia heterocalyx (2.5%), Pseudosenefeldera inclinata (2.1%), Hevea guianensis (2.1%), and Eschweilera pedicellata (2.0%). | This association is defined on the basis of 8 samples, that included 332 species, 169 genera in 55 families. Eperua purpurea (as elective species) and Erisma japura (as the dominant) were chosen to name this taxon. Within the elective species are: Protium crassipetalum, Aldina kunhardtiana, Heterostemon conjugatus, Swartzia pinnata, Clathrotropis glaucophylla, Oenocarpus bacaba, Ecclinusa bullata, Helianthostylis steyermarkii, and Pouteria guianensis. Other species can be shown in the Appendix 3.1. | This association assembles the “terra firme” forests and the tall caatingas. The forest of the association Eperuo purpureae Erismetum japurae can be found on areas in the upper Rio Negro growing on poorly drained sandy and clay soils; it tolerates considerable differences in drainage classes. This kind of vegetation was described based on eight (8) samples located in Colombia (e.g., mouth of Naquén river, Puerto Colombia - Guainía) and Venezuela (e.g., Maroa-Yavita road). Detailed information on these localities is found in Appendix 3.2. |
ORDER II 1. Goupio glabrae - Minquartiion guianensis all. Nov. (Figs. 3.5 and 3.6) Typus: Eschweilero parviflorae - Mouririetum grandiflorae (this study; Appendix 1) Lowlands forests of the Eschweilera parviflora and Mouriri grandiflora | The forest communities of this alliance are medium to high in stature, up to 45 m tall. These communities are characterized by the presence of trees of Arecaceae, Burseraceae, Lauraceae, Lecythidaceae, Fabaceae, Melastomataceae, Moraceae, Myristicaceae, Olacaceae, Rubiaceae, Sapindaceae, and Sapotaceae. | This alliance is defined on the basis of 95 samples that included 1983 species, 532 genera in 120 families. This syntaxon is named with the exclusive species Goupia glabra and the dominant species Minquartia guianensis. In this vegetation alliance others exclusive species were: Abuta grandifolia, Cynometra marginata, Mabea nitida, Clarisia racemosa, Oenocarpus minor, and Richeria grandis. The elective species are: Dendrobangia boliviana, Siparuna guianensis, Mouriri grandiflora, Virola elongata, Heterostemon mimosoides, Trichilia micrantha, and Xylopia nervosa. This alliance includes two associations: An undefined named compound by Attalea Maripa and Brosimum utile, and the Eschweilero parviflorae - Mouririetum grandiflorae. | This alliance is compound by “terra firme” forests, forests with palm dominance and seasonally flooded forests in white and clear waters with or without palm dominance. The Eschweilea parviflora—Mouriri grandiflora order is located on in the transition between alluvial plains and the hills (“lomeríos”) over Ultisol and poorly drained Entisol. |
1.1 Attalea Maripa - Brosimum utile (Figs. 3.5 and 3.6). Representative plot no. P2, TWINSPAN ID 187. Coordinates datum WGS84 LAT 1.7449, LON -69.7660. Altitude 193 m. Colombia, Venezuela and Brazil (Appendix 3.1) Forests of Attalea Maripa and Brosimum utile | The forests of Attalea Maripa and Mespilodaphe cymbarum are communities of medium to high stature and density, mainly composed by trees with a DBH average greater than 15 cm. This analysis was outline such as an undefined association. The canopy of the forest is composed of trees with heights between 20 and 40 m. Brosimum utile, Euterpe precatoria, Matayba elegans, Eschweilera parviflora, and Virola elongata being the most abundant species. Some of the highest heights were recorded in Minquartia guianensis (58 m), Aldina latifolia (40 m), Brosimum utile (40 m), Caryocar glabrum (40 m), Eschweilera parviflora (40 m), Mespilodaphe cymbarum (40 m), Vochysia assua (40 m), V. grandis (40 m), and Attalea Maripa (20 m).To this syntaxon the species with the biggest dominance expression are Eschweilera parviflora (3.0%), Attalea Maripa (1.9%), Goupia glabra (1.7%), Allantoma lineata (1.6%), Swartzia tomentifera (1.5%), Euterpe precatoria (1.4%), Minquartia guianensis (1.3%), Chaetocarpus schomburgkianus (1.3%), and Brosimum utile (1.2%). | This kind of vegetation was defined on the basis of 68 samples, that included 1905 species, 516 genera in 117 families. This group of undefined associations registered Attalea Maripa as the elective species, and Brosimum utile as the dominant. As exclusive species were registered: Brosimum utile, Zygia cataractae, Maprounea guianensis, Mouriri acutiflora, Garcinia madruno, Piper arboreum, protium laxiflorum, Zygia inaequalis, and Homalolepis cedron. Other exclusive species can be shown in the Appendix 3.1. Among the elective species are: Gustavia augusta, Myrcia fallax, protium stevensonii, Inga acrocephala, Macrosamanea amplissima, Maquira calophylla, Duguetia quitarensis, and Gustavia acuminata. Other species can be shown in Appendix 1. | This vegetation is represented by “terra firme” forests, flooded forests with palm dominance, and seasonally flooded forests with or without palm dominance. The forest of Attalea Maripa and Brosimum utile assembles 68 samples located in Brazil (e.g., near Manaus city, Uatuma river, Pico da Neblina National Park), Colombia (i.g. Cumaribo-Vichada, Mitú, middle Caquetá river, “El Retiro” (“La Lindosa”), Inírida river, near mouth of “caño Bocón,” Guaviare river, upper Isana river) and Venezuela (e.g., along Casiquiare river, middle and lower Ventuari river). Detailed information on these localities is found in Appendix 3.2. |
1.2 Eschweilero parviflorae - Mouririetum grandiflorae assoc. nov. (Figs. 3.5 and 3.6). Typus: Plot CC_173, TWINSPAN ID 112. Coordinates datum WGS84 LAT 3.7917, LON -67.8197. Altitude 108 m. Colombia. (Appendix 3.1). Forests of Eschweilera parviflora and Erisma laurifolium | The forests of Eschweilera parviflora and Erisma laurifolium are communities of medium to high stature and density, mainly composed of trees with a DBH average greater than 15 cm. Protium divaricatum, Eschweilera parviflora, Brosimum utile, Pouteria baehniana, Mouriri grandiflora, Clathrotropis brachypetala, and Virola elongata being the most abundant species. In this association following species registered the highest heights Clathrotropis glaucophylla (35 m), Eperua purpurea (35 m), Erisma laurifolium (35 m) Goupia glabra (35 m), Vochysia splendens (33 m), Caryocar glabrum (32 m), Qualea ingens (32 m), Eschweilera parviflora (30 m), and Mouriri grandiflora (30 m). Some of the species that present a high physiognomic expression in this syntaxon (relativized units) are: Eperua purpurea (5.2%), Caryocar glabrum (4.2%), Mouriri nigra (3.4%), Goupia glabra (2.9%), Leopoldinia piassaba (2.7%), Leptobalanus apetalus (2.6%), Eschweilera parviflora (2.6%), protium divaricatum (2.4%), Virola elongata (2.2%), and Mouriri grandiflora (1.8%). | This association is defined on the basis of 28 samples that included 373 species, 176 genera and 60 families. This syntaxon is named with the exclusive species Eschweilera parviflora and the dominant species Mouriri grandiflora. In this vegetation pattern Pouteria baehniana was registered as exclusive species. Within the elective species are: Clathrotropis brachypetala, Mouriri grandiflora, Eschweilera parviflora, Virola elongata, protium divaricatum, Erisma laurifolium, and Aptandra tubicina. Other species can be shown in the Appendix 1. | This association is compound by “terra firme” forests and seasonally flooded forests with or without palms dominance. The forest of the association Eschweilero parviflorae Mouririetum grandiflorae was determined based on 28 samples located in Colombia (e.g., Guaviare river at mouth Inírida river, near Inírida, Nukak reservation). Detailed information on these localities is found in Appendix 3.2. |
2. Astrocaryo chambirae - Socrateion exorrhizae all. Nov. (Figs. 3.5 and 3.6). Representative plot. GB_8, TWINSPAN ID 172. Coordinates datum WGS84 LAT 2.4232, LON -72.3699. Colombia. (Appendix 3.1) Lowlands forests of the Astrocaryum chambira and Socratea exorrhiza without indefinite associations | The forest communities of this alliance are medium to high stature, up to 25–30 m tall, characterized by the high presence of palms and trees of Burseraceae, Lauraceae, Lecythidaceae, Fabaceae, Melastomataceae, Moraceae, Myristicaceae, Rubiaceae, Sapindaceae, and Sapotaceae. Some of the highest heights were recorded in Vochysia splendens (26.5 m), Virola marleneae (22.4 m), Clathrotropis macrocarpa (21.4 m), Socratea exorrhiza (13.9 m), and Astrocaryum chambira (9.3 m). The species with most physiognomic expression are Erythroxylum macrophyllum (6.8%), Psychotria remota (6.0%), Palicourea raveniana (6.0%), Sorocea muriculata (4.8%), Theobroma subincanum (2.5%), Socratea exorrhiza (2.3%), Astrocaryum chambira (2.1%), Miconia punctata (2.0%), Pseudolmedia laevis (1.8%), and Clathrotropis macrocarpa (1.8%). | This alliance is defined on the basis of 16 samples that included 102 species, 79 genera and 39 families. Astrocaryum chambira (as the elective species) and Socratea exorrhiza (as the dominant) were chosen to name this syntaxon. As exclusive species were registered: Inga tessmannii, Psychotria remota and Jacaratia spinosa. The elective species are: Calycophyllum megistocaulum, Astrocaryum chambira, Pseudolmedia laevis, Euterpe precatoria, and Socratea exorrhiza. | This alliance is composed of “terra firme” forests with palms dominance, located in the transitions between the alluvial plains and the terraces over Entisol and Ultisol soils. This kind of vegetation was described based on 16 samples located in Colombia (e.g., San José del Guaviare. Nabuquén creek, Puinawai natural reserve). Detailed information on these localities is found in Appendix 3.2. |
Appendix 3.2 Botanical Explorations in the Rio Negro Basin: A Review
1.1 Introduction
The record of human occupation in the upper Rio Negro basin, based on ceramic shards, dates back to between 3750 (Sanford et al. 1985) and 3570 (Neves 1998) years B.P. Other data, based on soil charcoal samples, dates human occupation to between 640 and 250 years B.P. for the top 20 cm soil layers, and between 6260 and 530 years B.P for the lower 20–90 cm soil layers (Saldarriaga and West 1986). It is interesting that the dates reported by these authors correspond with dry episodes in the Amazon basin and surrounding areas (van der Hammen 1972; Bush and McMichael 2016). The authors suggest that dry and humid periods alternated from 6000 to 400 B.P. The age estimated from charcoal and shards confirms that the region has been subjected to fires during extreme dry periods, and indicates periods of human disturbance (e.g., shifting cultivation) for the last 3750 years.
It is well known that the region comprising the upper Río Negro and Orinoco basins was traveled, explored, and inhabited for several millennia by ancestral groups such as the Makú-Puinave, the Arawak, and the Tucano (Zucchi 2006). Migrations or movements of these ancestral groups came from the Central Amazon region to the Rio Negro basin approximately 4500–3500 B.P. (Meggers 1979), perhaps escaping the devastating droughts or Mega-Niño events that took place in Central Amazonia during this time (Meggers 1994; van der Hammen 2006; Olivares et al. 2015). No doubt, the Río Negro basin was relatively well explored by the earliest inhabitants of the region, who were able to classify vegetation types and its most important plant species before Europeans arrived (Abraão et al. 2009). Also, Pre-Columbian populations categorized Amazonian rivers by the color of their water, and they knew that water color was related with fish richness or soil fertility (Junk et al. 2011).
1.2 The Journeys
1.2.1 Fifteenth to Nineteenth Centuries
The first Iberian journeys down the Amazon river, from the Andes to the Atlantic Ocean, were undertaken by two groups of Spaniards, one commanded by Francisco de Orellana (1541–1542) and later by Pedro de Ursúa (1560–1561) accompanied by, among others, the infamous Lope de Aguirre. Chronicles of these travels, written by Fray G. de Carvajal in the case of Orellana’s saga (Carvajal 1848), and by several witnesses and second-hand accounts in the case of Ursúa and Aguirre’s (e.g., Vazquez 1881), spoke of large areas of forest along the Amazon river and numerous, well-populated native villages. During Orellana’s journey a mighty black water river was sighted flowing from “El Poniente,” and was called “Río Negro” (Carvajal 1848).
After the two Iberian voyages down the Amazon river, the interest in knowing the Amazon basin in greater detail took a remarkable impact at the cartographic level, especially by the French, Portuguese, and Jesuits and Franciscans priests (Cintra 2012a). The first accurate map of the Amazon River was drawn by French cartographers, including one by Count of Blaise François Pagan (1655) and several by Nicolas Sanson (1656, 1657, 1680, 1698, and 1699), drawn and engraved during the golden age of French cartography (Cortesão 1965). Pagan’s map, Magni Amazoni Fluvii (Pagan 1655), is considered the most remarkable of all the charts, not just of the Amazonas river, but of the whole of Amazon basin (Cortesão 1965; Cintra 2011). It was based mainly on the account of Father Cristóbal de Acuña, who descended the river with Pedro Teixeira in 1639, and determined some latitudes and estimated distance in leagues between consecutive locations. This chart was the first established canvas of the meridians and parallels that scientifically situated the Amazon and took full advantage of the geographic data supplied by the discoverers. Nicolas Sanson (the royal cartographer of France) published his first Amazonas map in 1656. He improved his first map with several versions published in 1657, 1680, 1698, and 1699. The maps of the Amazon River traced by Sanson present precise geographic coordinates considering its time, shows a well-determined prime meridian, and also employs a creative methodology to deduce longitudes from latitudes and distances that had been covered (Cintra and de Oliveira 2014). After Pagan’s and Sanson’s maps, the Jesuit priest Samuel Fritz drew a map of the Amazon in 1691 and later had one engraved in 1707. Much simplified versions of Fritz’ map were first published in 1717 (engraved in French), 1726 (in German), 1755 (in Spanish), and in 1819 (a second version in French), accompanying an extract of his description of the “Maragnon.” Of the map engraved in 1707, apparently, only a few copies circulated are fewer are extant; the other four maps were published in “Lettres Édifiantes et Curieuses” (in Fritz 1717 and 1819, for the first and second versions in French, respectively), in Der Neüer Welt-Bott (Fritz 1726, in German), and “Cartas edificantes, y Curiosas” (Fritz 1755), journals then little read outside religious circles. It is evident that the scientific cartography of Amazonas begins with the map of Count of Pagan (for a review see: Cintra 2011, 2012b; Cintra and Freitas 2011; Cintra and Furtado 2011; Cintra and de Oliveira 2014).
Some years later, researchers started collecting plants and studying the vegetation of the Amazon river. Charles de La Condamine, who navigated this river in 1743, wrote the first biological report (de La Condamine 1745). European scientists considered his expedition the beginning of the great era of Amazonian travel (ter Steege et al. 2016). La Condamine remarked on numerous plant products such as curare, the arrow poison, derived from Strychnos spp. (Loganiaceae); he also documented for the first time the quinine tree, Cinchona officinalis L. (Rubiaceae) and the rubber tree, Hevea spp. (Euphorbiaceae). However, the first known large collection of Amazon plants was made by Alexandre Rodrigues Ferreira (1756–1815) during his voyage of 1783–1792 (Wurdack 1971). He explored the Amazon river and its main tributaries, including the Rio Negro and Rio Branco between 1785 and 1786, where his itinerary notably included the Isana and Vaupés rivers (Pereira da Silva 2008). His group collected and drew numerous plants and animals during this expedition. Asteranthos brasiliensis Desf. (Scytopetalaceae), with showy yellow, fused petals, and an actinomorphic androecium, is an endemic genus of the Rio Negro basin, and perhaps one of the most extraordinary plants they documented (see Fig. 3.12 on the text of Chap. 3).
Asteranthos brasiliensis Desf. Lecythidaceae. Predominantly occupies a geographical range from Eastern Colombia and stretches to incorporate Southern Venezuela and Northern Brazil, with a specific concentration in the Upper Río Negro region. This species is classified as arboreal and grows best primarily in hydric or moisture-rich environments—image captured by H. ter Steege
Later, “caño” Pimichín and the rivers Guianía and upper reaches of the Río Negro were explored by the famous naturalists F. H. A. von Humboldt and A. J. A. Bonpland (in April 1800), gathering the first biological collections of that section of the Río Negro basin. They were followed by C. F. P. von Martius and J. B. von Spix (1819–1820), A. Plée (1821), L. Reidel (1828), J. B. Natterer (1830–1832), R. H. Schomburgk (1839; 1855), P. J. Ayres (1842–1844), A. R. Wallace (1850–1851), and R. Spruce (1850–1854), at a time when scientists in Europe were fascinated by the tremendous diversity of fishes, insects, mammals and plants being discovered, and before modern scientific research on ecology and evolutionary biology.
Richard Spruce (1817–1893) was remarkable among all personalities above mentioned. He was a pioneer botanical explorer of the north-west Amazon and the northern Andes in the middle of the last century. He collected ca 7000 botanical specimens and made numerous important botanical discoveries. This British botanist, who opened up the Rio Negro region to science between 1850 and 1854, must be counted amongst the greatest naturalists ever to have engaged in collecting and studies anywhere in unexplored Neotropical territories (Schultes 1983). As a result of his meticulous observation and insatiable curiosity, a basis for our understanding of great areas of the Amazon was early and most firmly laid. Not only did Spruce advance taxonomy and floristics, but he was also a notable bryologist and made many important observations in ethnology, ethnobotany, linguistics, and geology (Spruce 1908).
1.2.2 Nineteenth to twentieth Centuries
The Portuguese and later Brazilians started an ambitious plan of exploration in the Rio Negro during the nineteenth century, shortly after the consolidation of Amazonas province in 1850. Several explorers visited the watersheds of main affluents (for a review see: Tenreiro-Aranha 1906). After this period, the exploration of the Rio Negro basin and its most important rivers continued with the work of, among others, G. Wallis (1863–1864), J. W. H. Traill (1874), J. Barbosa Rodrigues (1884–1885), P. H. W. Taubert (1896), E. H. G. Ule (1901–1902), É. Bommechaux (1903), G. A. E. Hübner (1903–1907, 1914), C. T. Koch-Grünberg (1903–1905), J. Huber (1904), H. Schmidt and L. Weiss (1907–1908), W. A. Ducke (1910–1932, 1933–1936, 1941–1942), J. G. Kuhlmann (1918), and D. E. Melin (1924). Also, it is important to point out that large sections of the Rio Negro and some of its tributaries, as well as the Casiquiare and the upper Orinoco, were extensively explored in the first quarter of the twentieth century by H. A. Rice and P. P. von Bauer. Their emphasis was geographical exploration; unfortunately, little biological material was collected, but important cartographical material resulted from their travels (Rice 1910, 1914, 1918, 1921, 1928; von Bauer 1919).
To this list of explorers, we should also mention the many plant collectors who worked on behalf of commercial horticultural houses in Europe and the United States who, although remaining largely anonymous, were behind the discovery and introduction of many plant species, particularly orchids, bromeliads, and other ornamental plants.
After the Treaty of Bogotá between Colombia and Brazil was signed in 1907, the Brazilian government started another program of exploration in the Rio Negro basin in 1927–1929, under the charge of Marshall Boaberges Lopes de Sousa. The botanist on this expedition, F. von Luetzelburg made significant botanical collections and annotations of types of vegetation of the upper Rio Negro (Lopes de Sousa 1955, 1959); he also visited the Casiquiare channel (Huber and Wurdack 1984). Attalea luetzelburgii (Burret) Wess. Boer (Arecaceae), a palm with subterranean stems represents a notable species among the numerous plants collected during his expedition.
The exploration of the Río Negro basin continued with the field work of G. H. H. Tate (1928–1929), E. G. Holt, W. Gehriger and E. R. Blake (1930–1931), B. A. Krukoff (1936), J. Cuatrecasas (1939), R. de Lemos Fróes (1941–1952), Ll. Williams (1942), J. A. Steyermark (1944; 1970), P. H. Allen (1943–1945), F. Cardona-Puig (1946), R. E. Schultes and F. López (1947–1948), J. Murça Pires (1947–1952), G. A. Black (1947–1950), B. Maguire et al. (1950–1966), J. J. Wurdack et al. (1951–1959), H. García-Barriga (1951), R. Romero-Castañeda (1952), A. Fernández-Pérez (1953), W. A. Rodrigues (1954–1968), V. Vareschi and K. Mägdefrau (1958–1962), L. A. Garay (1960), G. Eiten (1963), J. Ewel (1964), N. T. da Silva and U. Brazâo (1966), E. Medina (1968), L. Ruiz-Terán and J. Bautista (1968), M. Fariñas et al. (1969), J. A. Steyermark, C. Brewer-Carias and G. C. K. Dunsterville (1970), P. Maas (1971), B. Manara (1971), and G. T. Prance (1971). As shown here, numerous botanists, anthropologists, and ecologists have visited the Rio Negro basin to study the flora, vegetation, ethnography, and inhabitants in the last two hundred years. In the last five decades, fieldwork was carried out primarily by botanists, ecologists, and naturalists from the three countries that share the Rio Negro basin. Multiple studies have been done, too numerous to cited them all here.
Besides this remarkable amount of fieldwork, thousands of plant collections from Amazonia (including some from the Rio Negro) were products of the 8 expeditions to the Amazon basin conducted by B. A. Krukoff in 1923–1950 (Landrum 1986), and the 25 expeditions sponsored by the bi-national plant collecting program “Projeto Flora Amazônica” (Prance et al. 1984).
In addition, thousands of botanical specimens were collected in Brazil during the execution of the “Biological Dynamics of Forest Fragments Project (BDFFP)” set up near Manaus, and resulting from the field work conducted by “Instituto Amazónico de Investigaciones Científicas SINCHI” for nearly three decades in the Colombian Amazon. Likewise, in Venezuela, many botanical collections and publications resulted from: the interdisciplinary and multi-national project conducted by the “Instituto Nacional de Investigaciones Científicas (IVIC),” perhaps the most detailed study of Amazon caatinga and terra-firme forests ever conducted in the Upper Rio Negro (Medina 2000); the “Proyecto inventario de los recursos naturales de la región Guayana-PIRNRG-” (Zinck 1986); and the expeditions to “Serranía de la Neblina” (Brewer-Carías 1988). Finally, we must cite the collections generated by “Proyecto inventario de los recursos naturales de la región Guayana-PIRNRG-”, conducted by “CVG-Tecmin” (Corporación Venezolana de Guayana-Técnica Minera, C.A.) and other national and international institutions, a major effort to inventory the natural resources of the Venezuelan Guayana. As part of this project, a multidisciplinary team studied the upper Rio Negro and Orinoco basins in 1990–1992, which gathered large sets of plant specimens, ecological data (Aymard 2001), as well as soils and rocks samples. This field work resulted in the discovery of numerous new plant species and the gathering of data for a chorological report for this region (Aymard, in preparation).
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Arellano-Peña, H. et al. (2023). The Forests of the Upper Rio Negro (North-Western Amazon) and Adjacent South-Western Orinoco Basins: A Phytosociological Classification. In: Zinck, J.A., Huber, O., García Montero, P., Medina, E. (eds) Psammic Peinobiomes. Ecological Studies, vol 247. Springer, Cham. https://doi.org/10.1007/978-3-031-20799-0_3
Download citation
DOI: https://doi.org/10.1007/978-3-031-20799-0_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-20798-3
Online ISBN: 978-3-031-20799-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)