Abstract
Macroalgae commonly known as seaweeds are well-established natural resources of bioactive compounds responsible for various biological properties. There is a continuous search for new antiviral agents from natural sources, with a better activity and safety on resistant mutant strains and host cells respectively. Recently the focus has shifted towards the vast macroalgae resource and their antiviral potential; as they exhibited stronger activity than remdesivir. Their biochemical moieties are novel and distinct compared to the terrestrial plants. Some of them have shown lesser cytotoxicity. In this chapter, we aim to emphasize on their antiviral components and their mechanism of action. Studies have suggested their effectiveness against various viruses. Multiple antiviral activities from seaweeds towards the recent SARS-CoV-2 pandemic is also being discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Viral infections are responsible for causing severe acute respiratory syndrome (SARS), Ebola fever, influenza, hepatitis, and several other serious diseases in humans. They are one of the leading causes of death worldwide, among others like cardiovascular diseases and cancer. Enzootic and epizootic viral transmissions are the main sources of emerging human viral diseases. Parrish et al. (2008) emphasized an increase in epizootic transmissions, and which is also the cause of SARS CoV-2 pandemic that has taken its toll on the worldwide population. There is an immediate need for the development of new antiviral drugs as viral epidemics are estimated to increase in the future due to the increase in the interactions between humans and wildlife populations. Figure 25.1 represents a graph that indicates the number of global incidences of common viral infections in the last ten years. Various antiviral drugs have been developed and are being used but they are prone to drug resistance due to their extensive clinical use and side effects (Kim et al. 2011). Hence, exploring non-traditional resources for new compounds with a wide range of applications is being explored.
A graph depicting the number of global incidences of genital herpes, dengue, HIV/AIDS, acute hepatitis A, acute hepatitis B, acute hepatitis C, and measles. (Institute for Health Metrics and Evaluation 2021)
About 71% of the earth’s surface is covered in water; oceans consist of about 97% of the water volume available on earth (Charette and Walter 2010). Macroalgae are multicellular plants found in marine ecosystem and can belong to either eukaryotic or prokaryotic group of organisms. There is no particular definition of macroalgae as it is of polyphyletic origin. They are part of Asian culture in the form of food or traditional remedies and are consumed in dry or wet forms since earlier times. In the last few decades, there is a surge in the studies focused on obtaining biologically active metabolites from them (Kandale et al. 2011). They are a source of various primary and secondary metabolites, which have diverse chemical structures and a wide range of uses (Anil et al. 2017). The secondary metabolites profile is subjected to various physical and biological factors (Gallimore 2017); which can be used either in their natural form as an antiviral drug or their chemical structure can be evaluated to develop their semi-synthetic derivatives.
Various bioactive metabolites such as polysaccharides, tannins, phenolic acids, flavonoids, carotenoids, and bromophenols are derived from macroalgae. The amount of these metabolites in the macroalgae varies with the species. Some of the chemical compounds from these classes of metabolites have shown antiviral properties (Anil et al. 2017).
2 Antiviral Compounds from Macroalgae
2.1 Polysaccharides
Algal polysaccharides are economical, biodegradable and biocompatible natural non-toxic polymers found in abundance in nature. Figure 25.2 represents the life cycle of the virus and the general mechanism of action of antiviral polysaccharides derived from macroalgae.
(a) Life cycle of the virus in the host cell including (1) virus attachment, (2) penetration and uncoating, (3) biosynthesis (4) viral assembly and (5) viral release. (b) Mechanism of action of macroalgae derived antiviral polysaccharides which includes (1) blocking virus attachment to host cell, (2) inhibition of virus penetration (Hans et al. 2020)
2.1.1 Carrageenan
Carrageenan are natural anionic sulfated polysaccharides that are mostly found in the matrix of red algae such as Gigartina, Chondrus, Hypnea, and Eucheuma. They share structural and functional similarities with cellulose of higher order plants (Ahmadi et al. 2015).
They are classified based on the presence of 3,6-anhydrogalactopyranose and sulfated groups on the main structure into three types- namely- λ (lambda), κ (kappa), and ι (iota) carrageenan (Fig. 25.3). They selectively inhibit the binding of various enveloped and non-enveloped viruses on the host cells. They are an effective inhibitor of human papilloma virus (HPV). λ carrageenan can lead to the inactivation of herpes simplex virus (HSV). 1T1 is a λ carrageenan isolated from Gigartina skottsbegii which showed antiviral activity against HSV-2. The activity was displayed in mice due to the interference with the virus attachment stage to the host cells (Carlucci et al. 2004). Carrageenan extracted from Meristiella delirium was found to be effective against HSV-2 and Dengue virus-2 (DENV-2) with no cytotoxicity on Vero cells (Paula et al. 2006). ι carrageenan inhibited human rhinovirus (HRV) replication during its primary phase. It also inhibited the replication of the dengue virus in mosquitoes by affecting cell proliferation and protein synthesis, whereas the Vero cell line studies have shown early inhibitory activity due to the presence of some primary receptors (Talarico et al. 2011). κ-carrageenan on sulfation and acetylation inhibits the influenza virus. Molecular weight along with sulfonation groups are linked to the antiviral properties of the acetylated carrageenan against HIV (Yamada et al. 1997). It also binds to Enterovirus 71 (EV 71) and forms carrageenan-viruses complexes, disrupting the virus-receptor interaction which makes it an ideal candidate for the development of anti-EV 71 agents (Chiu et al. 2012).
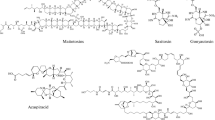
Antiviral compounds from macroalgae. The structure of protein griffithsin was generated by using PyMOL software and protein data bank accession number 2GTY (Ziółkowska et al. 2006)
2.1.2 Galactan
Galactans, also known as sulfated galactans, are found in red algae as the main extracellular polysaccharide. The main structure comprises of linear chains of galactose but there are few exceptions to this. There is an alternate chain of 3-β-D-galactopyranose (G units) and 4-α-D-galactopyranose residues or 4-3,6-anhydrogalactopyranose residues form the structural backbone with the presence of D-unit in carrageenans and L-units in agarans. In some exceptional galactans the DL-hybrids that enclose the G unit is attached to both D and L units (Estevez et al. 2001). Figure 25.3 shows the structure of galactan sulfate derived from red macroalgae. These varied structural forms of galactans are found to be effective against many enveloped viruses, namely HSV-1, HSV-2, DENV, hepatitis A, HIV-1, and HIV-2 (Witvrouw et al. 1994). Antiviral action of these galactans extracted from Callophyllis variegata has shown inhibitory effect with less cytotoxicity when tested for HSV-1, HSV-2, and DENV-2 (Rodríguez et al. 2005). Galactan sulfate isolated from Agardhiella tenera inhibited adhesion of virus to the cells in HIV-1 and HIV-2. Galactan isolated from Schizymenia binderi is highly selective against HSV-1 and HSV-2. (Matsuhiro et al. 2005). Structural hybrids of D, L-galactan, CS2-3 extracted from Cryptonemia crenulata has shown inhibition in the multiplication of three clinical strains of DENV-2 virus in a Vero cell line. It gave activity by inhibiting the adsorption of the virus into the host cells (Talarico et al. 2007).
2.1.3 Alginates
Alginates are acid polysaccharides primarily found in brown algae such as Ascophyllum nodusm, Macrocystis pyrifera, Laminaria hyperborea, Laminaria digitata, and Laminaria japonica. They are anionic linear polysaccharides with a backbone of poly-D-glucuronic acid(G blocks) and poly-D-mannuronic acid (M blocks), with D-guluronic acid and D-mannuronic acid (GM blocks) (Wang et al. 2012). Figure 25.3 shows the structures of M and G blocks. Another drug 911 is an alginate polysaccharide and showed promising anti-HIV-1 and anti-hepatitis B (HBV) results. In the case of HIV-1, it inhibits replication of the virus by decreasing the activity of reverse transcriptase (RTase) which in turn decreases viral adhesion to the host cells. It is effective in both chronic infection of H9 cells and acute infection of MT4 cells in vitro as well as in vivo (Xianliang et al. 2000). In the case of HBV, 911 inhibits viral replication by decreasing the activity of DNA polymerase (Jiang et al. 2003). A sulfated form of alginate namely sulfated polymannuroguluronate (SPMG) (Fig. 25.3) can be an anti-AIDS drug candidate as it inhibits gp120 from attaching to CD4 molecules on the surface of T cells in case of HIV-1 infection. There is a correlation between the size of SPMG oligosaccharides and their inhibitory action. The SPMG fragment must at least be a hexasaccharide to interact and inhibit gp120 and this property increases with an increase in the size of SPGM fragment (Meiyu et al. 2003; Liu et al. 2005). In vitro and in vivo studies suggests the effectiveness of SPMG against HPV infection. It is understood that it blocks HPV binding and entry by interacting with capsid L1 protein (Wang et al. 2020).
2.1.4 Fucan and Fucoidan
These are sulfated polysaccharides with high molecular weight in the range of 100–1600 kDa. They are found in the mucilaginous matrix or intercellular tissues of brown algae and are classified into three major groups i.e. glycuronogalactofucans, fucoidans, and xylofucoglycuronans. The structure of fucan is diverse and depends on the source of brown algae species used for its extraction. Sulfated fucans from Dictyota mertensii, Lobophora variegata, Fucus vesiculosus, and Spatoglossum schroederi block the reverse transcriptase activity and prevent HIV infections (Queiroz et al. 2008). They are also obtained from Cladosiphon okamuranus having sulfated fucose units and glucuronic acid, which inhibits DENV-2 infection in the BHK-21 cell line (Adhikari et al. 2006). MC26 is a fucose polysaccharide obtained from brown algae Sargassam piluliferm found to be effective in influenza with less cytotoxicity (Akamatsu et al. 2003). Fucans obtained from brown algae Cystoseira indica is effective against HSV-1 and HSV-2, showing no cytotoxicity for Vero cell culture with a proposed reason that it inhibits virus adsorption to the host cells. (Mandal et al. 2007). Fucoidans consist of L-fucose and less than 10% of other monosaccharides (Fig. 25.3). There is a higher proportion of fucose in the extracellular matrix of brown algae such as mozuku, komby, limu moui, bladderwrack, wakame, sea cucumber, and hijiki. The structure of fucoidans is diverse and repeating alternate sequence of α-1,3-linked sulfated L-fucose with an α-1,4-glycosidic bond which forms the backbone of fucoidans (Tanna and Mishra 2019). They are generally sulfated and acetylated and may contain uronic acid (Berteau and Mulloy 2003; Cumashi et al. 2007; Pomin and Mourão 2008). They are found to be effective against a few human RNA and DNA viruses both in vivo and in vitro (Witvrouw and De Clercq 1997). They are effective anti-HSV-1 and HSV-2 agents with no cytotoxicity for Vero cell lines. Fucoidans mainly block the adhesion of the virus to the host cells and inhibit viral-induced syncytium formation (Hidari et al. 2008). They exhibit better antiviral potency as compared with ribavirin in the Newcastle disease virus in the Vero cell studies (Elizondo-Gonzalez et al. 2012). They are capable of alternating the proteins of the extracellular matrix and can induce cell apoptosis by affecting cell proliferation (Haroun-Bouhedja et al. 2000; Koyanagi et al. 2003; Aisa et al. 2005; Moon et al. 2008).
2.1.5 Laminaran
Laminaran is a glucan found widely in brown algae such as Saccharina longicruris, F. vesiculosus, and Ascophyllum nodosum. Laminaran is a linear polysaccharide and has β(1 → 3)-linked glucose as the central chain along with β(1 → 6)-linked side-chain branching (Peat et al. 1958). There are two types of laminaran; one with glucose residues (G-series) and other with terminal D-mannitol residues (M-series) (Nelson and Lewis 1974). Their composition may vary depending on the species and the other physical and biological factors. Laminaran exhibits antiviral properties with low cellular toxicity in vivo; they were found to be useful in HIV by inhibiting replication and proliferation of the virus (O’Doherty et al. 2010).
2.1.6 Ulvan
Ulvan is a gelling polysaccharide obtained from Ulva species, an edible green seaweed. Ulvans are polyanionic heteropolysaccharide and their sugar composition mainly consist of rhamnose, glucuronic acid, and xylose (Fig. 25.3). However, it contains a wide range of other monosaccharides. α- and β-(1,4)-linked monosaccharides with repeating disaccharide units form the backbone of the ulvan structure. Type A ulvanobiuronic acid and type B ulvanobiuronic acid are the major disaccharide repeating units found in ulvan whereas, ulvanobioses (type U) is the minor disaccharide present (Lahaye and Robic 2007).
2.1.7 Naviculan
It is a high molecular weight polysaccharide made of various sugar moieties like fructose, xylose, rhamnose, mannose, fucose, and sulfate groups. Naviculan is obtained from a diatom Navicula directa . It is found to be effective against HSV-1, HSV-2, and influenza virus and works by inhibiting the initial stages of virus replication. Studies suggest its effectiveness against enveloped viruses (Lee et al. 2006).
2.1.8 Calcium Spirulan (Ca-SP)
It is obtained from the marine blue-green algae Arthrospira plantensis , and is a novel sulfated polysaccharide. It comprises of ribose, fructose, mannose, glucose, xylose, galactose, rhamnose, galacturonic acid, glucuronic acid, calcium and sulfate. It was found to inhibit virus entry into the host cell during in vitro and Vero studies. It is a selective inhibitor of viruses like HSV-1, HCMV, influenza A, measles, HIV-1, polio, mumps, and Coxsackie virus. It also exhibits mild anticoagulant properties (Hayashi et al. 1996). Ca-SP can be a promising new anti-HIV drug candidate.
2.1.9 Nostoflan (NSF)
It is found in edible blue-green algae; Nostoc flagelliforme . On hydrolysis NSF yields two types of oligosaccharides namely PA-1 [β-D-GlcAp-(1 → 4)-Xyl-PA] and PA-2 [β-D-GlcAp-(1 → 6)-β-D-Glcp-(1 → 4)-Gal-PA] (Fig. 25.3). It is proposed to be effective against various enveloped viruses by inhibiting their binding with the host cells. It can be a great candidate for the newer antiherpes drug (Kanekiyo et al. 2005; Thuan et al. 2019).
2.1.10 Xylomannan Sulfate
Xylomannan is a novel anti-freeze agent, that was first isolated from an Alaskan beetle Upis ceramboides . Its structure consists of β-D-mannopyranosyl-(1 → 4)- β-D-xylopyranose-disaccharide-repeating units. Some seaweed-derived xylomannan has antiviral properties (Table 25.1). Sulphated polysaccharides can be an option for the development of an anti-COVID drug, since the early symptoms of COVID-19 manifests common cold and flu, with similarities in the mechanism of action of the virus. Carrageenans in the form of nasal spray is an effective treatment for the common cold in adults and children (Ron Eccles et al. 2010). Iota carrageenan co-administered with Zanamivir in the form of nasal spray relieves upper respiratory symptoms in the patients suffering from influenza A (R Eccles et al. 2015). Such polysaccharides can be evaluated for their effectiveness against SARS COVID-2 virus. The ability of various microalgal polysaccharides especially ulvans, fucoidans, and carrageenan to inhibit virus adhesion and replication can be tested for the current pandemic (Pereira and Critchley 2020).
2.2 Lectin
Lectins are proteins or glycoproteins that are found naturally in cells that bind reversibly to glycans of glycoproteins, glycolipids, and polysaccharides. They are responsible for cell-cell interaction and protein folding and poses bioactivity; they are used as probes to determine cell surface structure and function. Currently, they are used to develop chemotherapeutic and antiviral agents. They are currently found to be effective against HIV. Griffithsin (GRFT), cyanovirin (CV-N), and banana lectin (Banlec) are some of the promising lectins for the development of antiviral drugs (Lusvarghi and Bewley 2016).
2.2.1 Griffithsin (GRFT)
Griffithsin was first isolated from Griffithsia sp., a type of red algae. GRFT is a protein and shows no similarity with any other known protein. GRFT exists as a stable homodimer and every subunit consist of 121 amino acids. GRFT interacts with the terminal sugar moiety of oligosaccharides (Sanchez 2013) (Fig. 25.3). A study on mice infected with the SARS-CoV virus has shown a 100% recovery when administered with GRFT by intranasal route (Ishag et al. 2013). It also showed potential to be a good candidate for the development of topical antiviral agents. (O’Keefe et al. 2009; Girard et al. 2018). GRFT binds with the glycoprotein enveloped by the virus and prevents CD4 and other antibodies from binding to the virus (Alexandre et al. 2010). Its immediate antiviral action is a great advantage compared to other antiviral agents which are being evaluated for the same purpose in HIV-1 (Emau et al. 2007). GRFT inhibits HCV, besides it can be effective against enveloped viruses such as the Japanese Encephalitis Virus (JEV), HSV-2, and HPV (Lusvarghi and Bewley 2016). It is also effective for the inhibition of different strains of coronavirus from replicating without cell proliferation. It can be a good candidate to test against respiratory infection for SARS COV-2 pandemic. The antiviral property of GRFT can be in synergism with other lectins (Ziółkowska et al. 2006; O’Keefe et al. 2010).
2.3 Phlorotannins
Phlorotannins are derivatives of a water-soluble polyphenolic compounds called tannins. Phlorotannins consist of polymer-forming phloroglucinol units which are biosynthesized by the acetate-malonate pathway. Phlorotannins are mainly obtained from brown and red algae (Nagayama et al. 2002; Kim et al. 2006). They are effective against the HIV-1 virus and exhibit inhibition of reverse transcriptase, protease, and integrase enzymes which play a vital role in virus replication inside the host cells. When obtained from various sources, they may elicit different inhibitory properties to these target enzymes (Kim and Karadeniz 2011). Phlorotannins derivatives obtained from brown alga Ecklonia cava demonstrated inhibition of protease and reverse transcriptase (RT) enzyme. Out of the four phlorotannins derivatives tested, 8,8 ′-bieckol and 8,4 ‴-dieckol (Fig. 25.3) inhibited HIV-1 RT efficiently while showing moderate inhibition towards HIV-1 protease enzyme (Ahn et al. 2004).
3 Conclusion and Future Perspective
Viral infection and re-infections are responsible for deadly diseases in human history and can be highly contagious causing an outbreak of epidemic. It is a lasting challenge for the healthcare sector, as with the rising human population and frequent travel throughout the globe has increased the contact between humans and animals. Countries with higher human-wildlife interactions are more prone to an enzootic and epizootic viral transmission that can be fatal for both humans and wildlife species. Moreover, viruses are a marvel of nature with a complex life cycle and ability to undergo mutations which makes it difficult for the development of antiviral drugs. Developing a vaccine for some old known viruses such as HIV or dengue has been challenging enough. The timely outbreaks of SARS, MERS, Ebola in recent years have called for immediate action for revolutionary discovery and development of antiviral drugs. Viral outbreaks are sudden as we can see in the case of the current SARS COV-2 pandemic; prior knowledge of viruses and the antiviral agents has helped for screening the candidates and for the development of vaccine against SARS-2. Viruses are diverse in their structure and function, thus there is a need for development of antivirals that can target a large group of viruses. Macroalgae are a great source of sulphated and halogenated polysaccharides, lectins, and phlorotannins which are promising candidates for the research and development of new antiviral agents. Most of these moieties are at early phases of development and more extensive investigations are required to develop an effective antiviral drug. The compounds belonging to these groups of metabolites can lead to groundbreaking discovery of new antivirals. They can be evaluated to be used as direct-acting antivirals or in a combination to enhance the effectiveness of already available antivirals. Moreover, they can be useful to cope up with the increasing cases of antiviral resistant strains and new viruses.
Abbreviations
- ASFV:
-
African swine fever virus
- Banlec:
-
Banana lectin
- BHK-21:
-
Baby hamster kidney (strain-21)
- BVDV:
-
Bovine viral diarrhea virus
- Ca-SP:
-
Calcium spirulan
- CV-N:
-
Cyanovirin
- DENV:
-
Dengue Virus
- DENV-2:
-
Dengue virus-2
- EV 71:
-
Enterovirus 71
- gp120 :
-
Glycoprotein 120
- GRFT:
-
Griffithsin
- H5N1:
-
Avian influenza virus
- HBV:
-
Hepatitis B virus
- HCMV:
-
Human cytomegalovirus
- HCV:
-
Hepatitis C virus
- HIV:
-
Human immunodeficiency virus
- HIV-1 :
-
Human immunodeficiency virus-1
- HIV-2:
-
Human immunodeficiency virus-2
- HPV:
-
Human papilloma virus
- HRV:
-
Human rhinovirus
- HSV:
-
Herpes simplex virus
- HSV-1:
-
Herpes simplex virus-1
- HSV-2:
-
Herpes simplex virus-2
- JEV:
-
Japanese Encephalitis Virus
- MERS:
-
Middle east respiratory syndrome
- NSF:
-
Nostoflan
- RTase:
-
Reverse transcriptase
- SARS:
-
Severe acute respiratory syndrome
- SPMG:
-
Sulfated polymannuroguluronate
References
Adhikari U, Mateu CG, Chattopadhyay K, Pujol CA, Damonte EB, Ray B (2006) Structure and antiviral activity of sulfated fucans from Stoechospermum marginatum. Phytochemistry. https://doi.org/10.1016/j.phytochem.2006.05.024
Ahmadi A, Moghadamtousi SZ, Abubakar S, Zandi K (2015) Antiviral potential of algae polysaccharides isolated from marine sources: a review. Biomed Res Int 2015. https://doi.org/10.1155/2015/825203
Ahn MJ, Yoon KD, Min SY, Lee JS, Kim JH, Kim TG, Kim SH, Kim NG, Huh H, Kim J (2004) Inhibition of HIV-1 reverse transcriptase and protease by phlorotannins from the brown alga Ecklonia cava. Biol Pharm Bull 27:544–547. https://doi.org/10.1248/bpb.27.544
Aisa Y, Miyakawa Y, Nakazato T, Shibata H, Saito K, Ikeda Y, Kizaki M (2005) Fucoidan induces apoptosis of human HS-sultan cells accompanied by activation of caspase-3 and down-regulation of ERK pathways. Am J Hematol 78:7–14. https://doi.org/10.1002/ajh.20182
Akamatsu E, Shimanaga M, Kamei Y (2003) Isolation of an anti-influenza virus substance, MC26 from a marine brown alga, Sargassum piluliferum and its antiviral activity against influenza virus. Coastal Bioenvironment - Saga University, Saga, Japan
Alexandre KB, Gray ES, Lambson BE, Moore PL, Choge IA, Mlisana K, Abdool Karim SS et al (2010) Mannose-rich glycosylation patterns on HIV-1 subtype C gp120 and sensitivity to the lectins, Griffithsin, Cyanovirin-N and Scytovirin. Virology 402:187–196. https://doi.org/10.1016/j.virol.2010.03.021
Ana P, Nathalie B, Gilles B, Daniel R, Tomás MS, Yolanda FP (2021) Anti-Herpes simplex virus (HSV-1) activity and antioxidant capacity of carrageenan-rich enzymatic extracts from Solieria filiformis (Gigartinales, Rhodophyta). Int J Biol Macromol 168:322–330. https://doi.org/10.1016/j.ijbiomac.2020.12.064
Anil S, Venkatesan J, Chalisserry EP, Nam SY, Kim S-K (2017) Applications of seaweed polysaccharides in dentistry. Seaweed Polysaccharides 331–340:12. https://doi.org/10.1016/B978-0-12-809816-5.00017-7
Berteau O, Mulloy B (2003) Sulfated fucans, fresh perspectives: structures, functions, and biological properties of sulfated fucans and an overview of enzymes active toward this class of polysaccharide. Glycobiology 13:29R–40R. https://doi.org/10.1093/glycob/cwg058
Carlucci MJ, Scolaro LA, Noseda MD, Cerezo AS, Damonte EB (2004) Protective effect of a natural carrageenan on genital herpes simplex virus infection in mice. Antivir Res 64:137–141. https://doi.org/10.1016/j.antiviral.2004.07.001
Charette M, Walter S (2010) The volume of earth’ s ocean. J Oceanogr Soc 23:112–114
Chiu YH, Chan YL, Tsai LW, Li TL, Wu CJ (2012) Prevention of human enterovirus 71 infection by kappa carrageenan. Antiviral Res 95:128–134. https://doi.org/10.1016/j.antiviral.2012.05.009
Cumashi A, Ushakova NA, Preobrazhenskaya ME, D’Incecco A, Piccoli A, Totani L, Tinari N et al (2007) A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 17:541–552. https://doi.org/10.1093/glycob/cwm014
Eccles R, Meier C, Jawad M, Weinmüllner R, Grassauer A, Prieschl-Grassauer E (2010) Efficacy and safety of an antiviral iota-carrageenan nasal spray: a randomized, double-blind, placebo-controlled exploratory study in volunteers with early symptoms of the common cold. Respir Res 11:108. https://doi.org/10.1186/1465-9921-11-108
Eccles R, Winther B, Johnston SL, Robinson P, Trampisch M, Koelsch S (2015) Efficacy and safety of iota-carrageenan nasal spray versus placebo in early treatment of the common cold in adults: the ICICC trial. Respir Res 16:121. https://doi.org/10.1186/s12931-015-0281-8
Elizondo-Gonzalez R, Elizabeth Cruz-Suarez L, Ricque-Marie D, Mendoza-Gamboa E, Rodriguez-Padilla C, Trejo-Avila LM (2012) In vitro characterization of the antiviral activity of fucoidan from Cladosiphon okamuranus against Newcastle disease virus. Virol J 9:1–9. https://doi.org/10.1186/1743-422X-9-307
Emau P, Tian B, O’keefe BR, Mori T, McMahon JB, Palmer KE, Jiang Y, Bekele G, Tsai CC (2007) Griffithsin, a potent HIV entry inhibitor, is an excellent candidate for anti-HIV microbicide. J Med Primatol 36:244–253. https://doi.org/10.1111/j.1600-0684.2007.00242.x
Estevez JM, Ciancia M, Cerezo AS (2001) DL-Galactan hybrids and agarans from gametophytes of the red seaweed Gymnogongrus torulosus. Carbohydr Res 331:27–41. https://doi.org/10.1016/S0008-6215(01)00015-5
Gallimore W (2017) Marine metabolites: oceans of opportunity. Pharmacognosy: fundamentals, applications and strategy. Elsevier Inc. doi:https://doi.org/10.1016/B978-0-12-802104-0.00018-4
García-Villalón D, Gil-Fernández C (1991) Antiviral activity of sulfated polysaccharides against African swine fever virus. Antivir Res. https://doi.org/10.1016/0166-3542(91)90031-L
Ghosh T, Pujol CA, Damonte EB, Sinha S, Ray B (2009) Sulfated xylomannans from the red seaweed Sebdenia polydactyla: structural features, chemical modification and antiviral activity. Antivir Chem Chemother 19:235–242. https://doi.org/10.1177/095632020901900603
Girard L, Birse K, Holm JB, Gajer P, Humphrys MS, Garber D, Guenthner P et al (2018) Impact of the griffithsin anti-HIV microbicide and placebo gels on the rectal mucosal proteome and microbiome in non-human primates. Sci Rep 8:8059. https://doi.org/10.1038/s41598-018-26313-8
Grassauer A, Weinmuellner R, Meier C, Pretsch A, Prieschl-Grassauer E, Unger H (2008) Iota-carrageenan is a potent inhibitor of rhinovirus infection. Virol J 5:107. https://doi.org/10.1186/1743-422X-5-107
Güven KC, Coban B, Özdemir O (2020) Pharmacology of marine macroalgae. Encycl Mar Biotechnol 10:585–615. https://doi.org/10.1002/9781119143802.ch20
Hans N, Malik A, Naik S (2020) Antiviral activity of sulfated polysaccharides from marine algae and its application in combating COVID-19: mini review. Bioresour Technol Rep 13:100623. https://doi.org/10.1016/j.biteb.2020.100623
Hardouin K, Bedoux G, Burlot AS, Donnay-Moreno C, Bergé JP, Nyvall-Collén P, Bourgougnon N (2016) Enzyme-assisted extraction (EAE) for the production of antiviral and antioxidant extracts from the green seaweed Ulva armoricana (Ulvales, Ulvophyceae). Algal Res 16:233–239. https://doi.org/10.1016/j.algal.2016.03.013
Haroun-Bouhedja F, Ellouali M, Sinquin C, Boisson-Vidal C (2000) Relationship between sulfate groups and biological activities of Fucans. Thromb Res 100:453–459. https://doi.org/10.1016/S0049-3848(00)00338-8
Hayashi T, Hayashi K, Maeda M, Kojima I (1996) Calcium Spirulan, an inhibitor of enveloped virus replication, from a blue-green alga Spirulina platensis. J Nat Prod 59:83–87. https://doi.org/10.1021/np960017o
Hemmingson JA, Falshaw R, Furneaux RH, Thompson K (2006) Structure and antiviral activity of the Galactofucan sulfates extracted from UndariaPinnatifida (Phaeophyta). J Appl Phycol 18:185. https://doi.org/10.1007/s10811-006-9096-9
Hidari KIPJ, Takahashi N, Arihara M, Nagaoka M, Morita K, Suzuki T (2008) Structure and anti-dengue virus activity of sulfated polysaccharide from a marine alga. Biochem Biophys Res Commun 376:91–95. https://doi.org/10.1016/j.bbrc.2008.08.100
Institute for Health Metrics and Evaluation (2021) Global health data exchange. http://ghdx.healthdata.org. Accessed 12 Feb 2021
Ishag HZA, Li C, Huang L, Sun MX, Wang F, Ni B, Malik T, Chen PY, Mao X (2013) Griffithsin inhibits Japanese encephalitis virus infection in vitro and in vivo. Arch Virol 158:349–358. https://doi.org/10.1007/s00705-012-1489-2
Jiang B-f, Xiao-fei X, Li L, Yuan W (2003) Study on—911‖ anti-HBV effect in HepG2. 2.15 cells culture. Modern Preventive Medicine 30:517–518
Jönsson M, Allahgholi L, Sardari RRR, Hreggviosson GO, Karlsson EN (2020) Extraction and modification of macroalgal polysaccharides for current and next-generation applications. Molecules 25. https://doi.org/10.3390/molecules25040930
Kandale A, Meena AK, Rao MM, Panda P, Mangal AK, Reddy G, Babu R (2011) Marine algae: An introduction, food value and medicinal uses. J Pharm Res 17
Kanekiyo K, Lee JB, Hayashi K, Takenaka H, Hayakawa Y, Endo S, Hayashi T (2005) Isolation of an antiviral polysaccharide, nostoflan, from a terrestrial cyanobacterium, Nostoc flagelliforme. J Nat Prod 68:1037–1041. https://doi.org/10.1021/np050056c
Kim SK, Karadeniz F (2011) Anti-HIV activity of extracts and compounds from marine algae. In: Advances in food and nutrition research, vol 64, 1st edn. Elsevier, London. https://doi.org/10.1016/B978-0-12-387669-0.00020-X
Kim M-M, Van Ta Q, Mendis E, Rajapakse N, Jung W-K, Byun H-G, Jeon Y-J, Kim S-K (2006) Phlorotannins in Ecklonia cava extract inhibit matrix metalloproteinase activity. Life Sci 79:1436–1443. https://doi.org/10.1016/j.lfs.2006.04.022
Kim SK, Vo TS, Ngo DH (2011) Potential application of marine algae as antiviral agents in medicinal foods. In: Advances in food and nutrition research, vol 64, 1st edn. Elsevier, London. https://doi.org/10.1016/B978-0-12-387669-0.00019-3
Koyanagi S, Tanigawa N, Nakagawa H, Soeda S, Shimeno H (2003) Oversulfation of fucoidan enhances its anti-angiogenic and antitumor activities. Biochem Pharmacol 65:173–179. https://doi.org/10.1016/S0006-2952(02)01478-8
Lahaye M, Robic A (2007) Structure and function properties of Ulvan, a polysaccharide from green seaweeds. Biomacromolecules 8:1765–1774. https://doi.org/10.1021/bm061185q
Lee JB, Hayashi K, Maeda M, Hayashi T (2004) Antiherpetic activities of sulfated polysaccharides from green algae. Planta Med 70:813–817. https://doi.org/10.1055/s-2004-827228
Lee JB, Hayashi K, Hirata M, Kuroda E, Suzuki E, Kubo Y, Hayashi T (2006) Antiviral sulfated polysaccharide from Navicula directa, a diatom collected from deep-sea water in Toyama Bay. Biol Pharm Bull 29:2135–2139. https://doi.org/10.1248/bpb.29.2135
Lee JB, Takeshita A, Hayashi K, Hayashi T (2011) Structures and antiviral activities of polysaccharides from Sargassum trichophyllum. Carbohydr Polym 86:995–999. https://doi.org/10.1016/j.carbpol.2011.05.059
Liu H, Geng M, Xin X, Li F, Zhang Z, Li J, Ding J (2005) Multiple and multivalent interactions of novel anti-AIDS drug candidates, sulfated polymannuronate (SPMG)-derived oligosaccharides, with gp120 and their anti-HIV activities. Glycobiology 15:501–510. https://doi.org/10.1093/glycob/cwi031
Lopes N, Ray S, Espada SF, Bomfim WA, Ray B, Faccin-Galhardi LC, Linhares REC, Nozawa C (2017) Green seaweed Enteromorpha compressa (Chlorophyta, Ulvaceae) derived sulphated polysaccharides inhibit herpes simplex virus. Int J Biol Macromol 102:605–612. https://doi.org/10.1016/j.ijbiomac.2017.04.043
Lusvarghi S, Bewley CA (2016) Griffithsin: An antiviral lectin with outstanding therapeutic potential. Viruses 8. https://doi.org/10.3390/v8100296
Makarenkova ID, Deriabin PG, Lvov DK, Zviagintseva TN, Besednova NN (2010) Antiviral activity of sulfated polysaccharide from the brown algae Laminaria japonica against avian influenza a (H5N1) virus infection in the cultured cells. Voprosy virusologii 55:41–45
Mandal P, Mateu CG, Chattopadhyay K, Pujol CA, Damonte EB, Ray B (2007) Structural features and antiviral activity of sulphated fucans from the brown seaweed Cystoseira indica. Antivir Chem Chemother 18:153–162. https://doi.org/10.1177/095632020701800305
Mandal P, Pujol CA, Carlucci MJ, Chattopadhyay K, Damonte EB, Ray B (2008) Anti-herpetic activity of a sulfated xylomannan from Scinaia hatei. Phytochemistry 69:2193–2199. https://doi.org/10.1016/j.phytochem.2008.05.004
Matsuhiro B, Conte AF, Damonte EB, Kolender AA, Matulewicz MC, Mejías EG, Pujol CA, Zúñiga EA (2005) Structural analysis and antiviral activity of a sulfated galactan from the red seaweed Schizymenia binderi (Gigartinales, Rhodophyta). Carbohydr Res 340:2392–2402. https://doi.org/10.1016/j.carres.2005.08.004
Mazumder S, Ghosal PK, Pujol CA, Carlucci MJ, Damonte EB, Ray B (2002) Isolation, chemical investigation and antiviral activity of polysaccharides from Gracilaria corticata (Gracilariaceae, Rhodophyta). Int J Biol Macromol 31:87–95. https://doi.org/10.1016/S0141-8130(02)00070-3
Meiyu G, Fuchuan L, Xianliang X, Jing L, Zuowei Y, Huashi G (2003) The potential molecular targets of marine sulfated polymannuroguluronate interfering with HIV-1 entry: interaction between SPMG and HIV-1 rgp120 and CD4 molecule. Antivir Res 59:127–135. https://doi.org/10.1016/S0166-3542(03)00068-8
Moon HJ, Lee SR, Shim SN, Jeong SH, Stonik VA, Rasskazov VA, Zvyagintseva T, Lee YH (2008) Fucoidan inhibits UVB-induced MMP-1 expression in human skin fibroblasts. Biol Pharm Bull 31:284–289. https://doi.org/10.1248/bpb.31.284
Morán-Santibañez K, Cruz-Suárez LE, Ricque-Marie D, Robledo D, Freile-Pelegrín Y, Peña-Hernández MA, Rodríguez-Padilla C, Trejo-Avila LM (2016) Synergistic effects of sulfated polysaccharides from Mexican seaweeds against measles virus. Biomed Res Int 2016. https://doi.org/10.1155/2016/8502123
Nagayama K, Iwamura Y, Shibata T, Hirayama I, Nakamura T (2002) Bactericidal activity of phlorotannins from the brown alga Ecklonia kurome. J Antimicrob Chemother 50:889–893. https://doi.org/10.1093/jac/dkf222
Nelson TE, Lewis BA (1974) Separation and characterization of the soluble and insoluble components of insoluble laminaran. Carbohydr Res 33:63–74. https://doi.org/10.1016/S0008-6215(00)82940-7
O’Doherty JV, Dillon S, Figat S, Callan JJ, Sweeney T (2010) The effects of lactose inclusion and seaweed extract derived from Laminaria spp. on performance, digestibility of diet components and microbial populations in newly weaned pigs. Anim Feed Sci Technol 157:173–180. https://doi.org/10.1016/j.anifeedsci.2010.03.004
O’Keefe BR, Vojdani F, Buffa V, Shattock RJ, Montefiori DC, Bakke J, Mirsalis J et al (2009) Scaleable manufacture of HIV-1 entry inhibitor griffithsin and validation of its safety and efficacy as a topical microbicide component. Proc Natl Acad Sci U S A 106:6099–6104. https://doi.org/10.1073/pnas.0901506106
O’Keefe BR, Giomarelli B, Barnard DL, Shenoy SR, Chan PKS, McMahon JB, Palmer KE et al (2010) Broad-Spectrum and Activity and Efficacy of the Antiviral Protein Griffithsin against Emerging Viruses of the Family Coronaviridae. J Virol 84:2511–2521. https://doi.org/10.1128/JVI.02322-09
Parrish CR, Holmes EC, Morens DM, Park E-C, Burke DS, Calisher CH, Laughlin CA, Saif LJ, Daszak P (2008) Cross-species virus transmission and the emergence of new epidemic diseases. Microbiol Mol Biol Rev 72:457–470. https://doi.org/10.1128/mmbr.00004-08
Paula PC, Talarico LB, Noseda MD, Silvia SM, Damonte EB, Maria ER, Duarte. (2006) Chemical structure and antiviral activity of carrageenans from Meristiella gelidium against herpes simplex and dengue virus. Carbohydr Polym 63:459–465. https://doi.org/10.1016/j.carbpol.2005.09.020
Peat S, Whelan WJ, Lawley HG (1958) The structure of laminarin. Part I. the main polymeric linkage. J Chem Soc 141:724–728. https://doi.org/10.1039/JR9580000724
Pereira L, Critchley AT (2020) The COVID 19 novel coronavirus pandemic 2020: seaweeds to the rescue? Why does substantial, supporting research about the antiviral properties of seaweed polysaccharides seem to go unrecognized by the pharmaceutical community in these desperate times? doi:https://doi.org/10.1007/s10811-020-02143-y/Published
Piccini LE, Carro AC, Quintana VM, Damonte EB (2020) Antibody-independent and dependent infection of human myeloid cells with dengue virus is inhibited by carrageenan. Virus Res 290. https://doi.org/10.1016/j.virusres.2020.198150
Pomin VH, Mourão PAS (2008) Structure, biology, evolution, and medical importance of sulfated fucans and galactans. Glycobiology 18:1016–1027. https://doi.org/10.1093/glycob/cwn085
Ponce NMA, Pujol CA, Damonte EB, Flores ML, Stortz CA (2003) Fucoidans from the brown seaweed Adenocystis utricularis: extraction methods, antiviral activity and structural studies. Carbohydr Res 338:153–165. https://doi.org/10.1016/S0008-6215(02)00403-2
Pujol CA, Estevez JM, Carlucci MJ, Ciancia M, Cerezo AS, Damonte EB (2002) Novel DL-galactan hybrids from the red seaweed Gymnogongrus torulosus are potent inhibitors of herpes simplex virus and dengue virus. Antivir Chem Chemother 13:83–89. https://doi.org/10.1177/095632020201300202
Queiroz KCS, Medeiros VP, Queiroz LS, Abreu LRD, Rocha HAO, Ferreira CV, Jucá MB, Aoyama H, Leite EL (2008) Inhibition of reverse transcriptase activity of HIV by polysaccharides of brown algae. Biomed Pharmacother 62:303–307. https://doi.org/10.1016/j.biopha.2008.03.006
Rabanal M, Ponce NMA, Navarro DA, Gómez RM, Stortz CA (2014) The system of fucoidans from the brown seaweed Dictyota dichotoma: chemical analysis and antiviral activity. Carbohydr Polym 101:804–811. https://doi.org/10.1016/j.carbpol.2013.10.019
Rodríguez MC, Merino ER, Pujol CA, Damonte EB, Cerezo AS, Matulewicz MC (2005) Galactans from cystocarpic plants of the red seaweed Callophyllis variegata (Kallymeniaceae, Gigartinales). Carbohydr Res 340:2742–2751. https://doi.org/10.1016/j.carres.2005.10.001
Rudke AR, de Andrade CJ, Ferreira SRS (2020) Kappaphycus alvarezii macroalgae: An unexplored and valuable biomass for green biorefinery conversion. Trends Food Sci Technol 103:214–224. https://doi.org/10.1016/j.tifs.2020.07.018
Sanchez G (2013) Las instituciones de ciencia y tecnología en los procesos de aprendizaje de la producción agroalimentaria en Argentina. El sistema argentino de innovación: instituciones, empresas y redes. El desafío de la creación y apropiación de conocimiento 670:661–670. https://doi.org/10.1002/prot
Talarico LB, Zibetti RGM, Faria PCS, Scolaro LA, Duarte MER, Noseda MD, Pujol CA, Damonte EB (2004) Anti-herpes simplex virus activity of sulfated galactans from the red seaweeds Gymnogongrus griffithsiae and Cryptonemia crenulata. Int J Biol Macromol 34:63–71. https://doi.org/10.1016/j.ijbiomac.2004.03.002
Talarico LB, Duarte MER, Zibetti RGM, Noseda MD, Damonte EB (2007) An algal-derived DL-galactan hybrid is an efficient preventing agent for in vitro dengue virus infection. Planta Med 73:1464–1468. https://doi.org/10.1055/s-2007-990241
Talarico LB, Noseda MD, Ducatti DRB, Duarte MER, Damonte EB (2011) Differential inhibition of dengue virus infection in mammalian and mosquito cells by iota-carrageenan. J Gen Virol 92:1332–1342. https://doi.org/10.1099/vir.0.028522-0
Tanna B, Mishra A (2019) Nutraceutical potential of seaweed polysaccharides: structure, bioactivity, safety, and toxicity. Compr Rev Food Sci Food Saf 18:817–831. https://doi.org/10.1111/1541-4337.12441
Thuan NH, An TT, Shrestha A, Canh NX, Sohng JK, Dhakal D (eds) (2019) Recent advances in exploration and biotechnological production of bioactive compounds in three cyanobacterial genera: Nostoc, Lyngbya, and Microcystis. Front Chem. https://doi.org/10.3389/fchem.2019.00604
Thuy TTT, Ly BM, Van TTT, Van Quang N, Tu HC, Zheng Y, Seguin-Devaux C, Mi B, Ai U (2015) Anti-HIV activity of fucoidans from three brown seaweed species. Carbohydr Polym 115:122–128. https://doi.org/10.1016/j.carbpol.2014.08.068
Wang W, Wang SX, Guan HS (2012) The antiviral activities and mechanisms of marine polysaccharides: An overview. Mar Drugs 10:2795–2816. https://doi.org/10.3390/md10122795
Wang S, Lu Z, Wang S, Liu W, Gao J, Tian L, Wang L et al (2020) The inhibitory effects and mechanisms of polymannuroguluronate sulfate against human papillomavirus infection in vitro and in vivo. Carbohydr Polym 241:116365. https://doi.org/10.1016/j.carbpol.2020.116365
Witvrouw M, De Clercq E (1997) Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen Pharmacol Vasc S 29:497–511. https://doi.org/10.1016/S0306-3623(96)00563-0
Witvrouw M, Este JA, Mateu MQ, Reymen D, Andrei G, Snoeck R, Ikeda S et al (1994) Activity of a sulfated polysaccharide extracted from the red seaweed Aghardhiella tenera against human immunodeficiency virus and other enveloped viruses. Antivir Chem Chemother 5:297–303. https://doi.org/10.1177/095632029400500503
Xianliang X, Meiyu G, Huashi G, Zelin L (2000) Study on the mechanism of inhibitory action of 911 on replication of HIV-1 in vitro. Chin J Marine Drugs 19:15–18
Yamada T, Ogamo A, Saito T, Watanabe J, Uchiyama H, Nakagawa Y (1997) Preparation and anti-HIV activity of low-molecular-weight carrageenans and their sulfated derivatives. Carbohydr Polym 32:51–55. https://doi.org/10.1016/S0144-8617(96)00128-2
Zhu B, Ni F, Sun Y, Zhu X, Yin H, Yao Z, Yuguang D (2018) Insight into carrageenases: major review of sources, category, property, purification method, structure, and applications. Crit Rev Biotechnol 38:1261–1276. https://doi.org/10.1080/07388551.2018.1472550
Ziółkowska NE, O’Keefe BR, Mori T, Zhu C, Giomarelli B, Vojdani F, Palmer KE, McMahon JB, Wlodawer A (2006) Domain-swapped structure of the potent antiviral protein Griffithsin and its mode of carbohydrate binding. Structure 14:1127–1135. https://doi.org/10.1016/j.str.2006.05.017
Acknowledgments
We thank SVKM’S NMIMS for all the support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Singh, S., Kumar, M.S. (2022). Antiviral Applications of Macroalgae. In: Ranga Rao, A., Ravishankar, G.A. (eds) Sustainable Global Resources of Seaweeds Volume 2. Springer, Cham. https://doi.org/10.1007/978-3-030-92174-3_25
Download citation
DOI: https://doi.org/10.1007/978-3-030-92174-3_25
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-92173-6
Online ISBN: 978-3-030-92174-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)