Abstract
Wheat (Triticum spp. L.) has fed the humankind for centuries. It has affected human life economically and culturally as human has affected its evolution since its domestication 12,000 years ago. Wheat cultivars in use were mainly landraces till the end of the nineteenth century. After World War II, industrialized agriculture led to the selection of modern bread and durum wheat cultivars based upon their higher grain yields and technological characteristics. Nowadays, although wheat landraces generally have a lower grain yield, there is a renewed attention from farmers, consumers, and manufacturers for them, due to their lesser fertilizer input, higher genetic diversity and adaptability, and desired nutritional profile and flavor.
The wheat cultivars from “old” to “modern”, though, show significant technological quality increases over time, there are no extreme differences between landraces and modern wheat cultivars in terms of various nutritional components. Wheat landraces are consumed mostly as bread; however, they are also used for making pasta, bulgur, cookie, and other end-products. This chapter particularly reviews the compositional profile and technological properties of wheat landraces.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
Wheat has played a significant role as a main source of foodstuff since the early civilizations in the Fertile Crescent which includes some parts of Turkey. The domestication of wheat began about 12,000 years ago in Göbekli Tepe, southeastern Turkey, according to evidence from archaeological excavations (Schmidt 2007; Dietrich et al. 2012). During the migration, many traditional cultivars or landraces were chosen by farmers and nature to fit environmental and cultural niches (Hernández-Espinosa et al. 2019).
Until the beginning of the twentieth century, wheat cultivars were predominantly landraces, which were well adapted to their local environments. Since then, landraces have been used as a source of variability in the creation of modern wheat cultivars as breeding methods have developed. After World War II, intensive wheat breeding resulted in the entire replacement of landraces by new semi-dwarf and high-yielding wheat cultivars, resulting in a reduction in wheat genetic diversity (Bordes et al. 2008).
Although landraces were mostly displaced by the superior modern wheat cultivars, they have provided some opportunities for breeders, farmers, manufacturers, and consumers. Wheat landraces might behave as donors with significant features, such as drought and cold tolerance and grain quality. Wheat landraces generally represent considerably wider genetic diversity than modern wheat cultivars; therefore, they could lead to extending the genetic base of modern wheat cultivars (Azeez et al. 2018). Wheat landraces can precisely be adapted to their locality of origin and are frequently related with tolerance to biotic and abiotic stresses and higher grain yield under lower input management practices (Hernández-Espinosa et al. 2019). Wheat landraces and old cultivars could play a significant role in food safety not only as being an easily accessible gene source for breeders but also as being more resilient than modern wheat cultivars because they perform well in marginal areas (Migliorini et al. 2016). Besides, the landraces and old cultivars grown in marginal environments or under organic conditions can provide higher income to farmers as compared to modern wheat cultivars, as they are primarily used for the production of natural and healthy whole grain products (Kantor et al. 2001).
Healthy foods have been getting increasing popularity recently with a renewed interest in wheat landraces and old cultivars. Landraces cannot compete with modern cultivars for yield; however, they have generally higher desirable nutritional values (Marconi and Cubadda 2005). Landraces and old cultivars usually have higher protein contents (Dinelli et al. 2013; Giunta et al. 2019), minerals (Hussain et al. 2012a; Velu et al. 2019), and phenolic compounds (Dinelli et al. 2013) than modern wheat cultivars. In comparison with modern wheats, the landraces and old wheats tend to be richer in protein and linoleic acid, to be poorer in starch, and to have softer grains (Bordes et al. 2008). It is also reported that modern wheat cultivars possess the highest albumin and total starch contents, whereas landraces have the highest grain protein and gliadin contents, and old genotypes possess the highest glutenin and amylose contents (Boukid et al. 2018).
The local character of a landrace can derive from its grain quality features which are often appropriate for producing a particular local product according to the preferences of the consumer in a specified region (processing, baking, cooking, and tasting) (Hernández-Espinosa et al. 2019). The landraces are still being used in rustic areas worldwide to cook traditional foods. For instance, bulgur in different cities or regions of Turkey is produced with specific landraces including einkorn and emmer to obtain locally desired end-use product features.
Landraces have got increased popularity recently. Therefore, comprehensive studies comparing the nutritional values of landraces have recently been undertaken by scientists. However, limited studies have been carried out to screen the end-products’ quality characteristics of wheat landraces. The aim of this chapter is to review and compare the results of the nutritional and technological characteristics of wheat landraces accessible in the literature.
6.2 Physical Properties
Grain size is one of the main quality traits subjected to selection. Grain size has a clear effect on many compositional and qualitative characteristics since large and heavy kernels have higher amounts of starchy endosperm and lower levels of aleurone layers and external pericarp (Brandolini et al. 2011). Beside kernel size, uniform kernel size is important for effective milling. Out of specification, small grain proportions decrease the market value of the wheat and the advantage of the higher grain yield. Therefore, wheat breeders should screen the distribution of kernel size during the breeding in order to prevent small kernel problems (Hare 2017).
In a comprehensive study comparing kernel weights of 37 Iranian and 42 Mexican durum wheat landraces, none of the landraces came close to the bread wheat check samples, with an average kernel weight of Iranian accessions slightly higher than that of Mexican (Hernández-Espinosa et al. 2019). Dotlačil et al. (2010) studied two different sample sets of combining obsolete cultivars and winter wheat landraces and found that Set I (n = 122) and Set II (n = 101) had lower thousand kernel weight values as mean compared to bread wheat samples. Nazco et al. (2012) compared 154 durum wheat landraces, originating from 20 Mediterranean countries, as three groups (Eastern, North Balkan, and Western), to 18 modern wheat cultivars. Landraces from the Eastern Mediterranean countries showed the highest variability (28.9–54.9 g) with a mean of 44.6 g, but lower kernel weight than Western (49.4 g) and North Balkan (50.7 g) Mediterranean countries as well as modern cultivars (46.7 g).
The variation in kernel weight between both tetraploid and hexaploid landraces was large; however, tetraploid wheat landraces had heavier kernel weight than hexaploids (Blum et al. 1987). Beside wheat species and cultivars, environmental conditions and agronomic applications can also affect the kernel size and weight. A relatively low heritability value (51.27%) was reported for thousand kernel weight (Heidari et al. 2016). In the same study, thousand kernel weight of wheat landraces varied from 26.8 to 55.0 g.
Grain hardness is one of the most significant quality characteristics for milling and baking quality of wheat. The attachment between the protein and the starch in the endosperm is stronger in hard wheat than in soft one. During the milling, hard wheat has a higher amount of starch damage, flour extraction proportion, and energy consumption (Bedõ et al. 2010). Based on kernel hardness, wheat is classified as soft, medium-soft, medium-hard, hard, and extra-hard. This classification creates a fundamental basis for differentiating the world trade of wheat grain (Pasha et al. 2010).
The hardness is inherited and controlled by Ha hardness genes (Pin a and Pin b) residing on the short arm of chromosome 5D but is also impacted by other small-effect loci (Pasha et al. 2010). Pin a and Pin b have various alleles in hexaploid wheat (Morris and Bhave 2008). The variability in Pin function considerably impacts the quality features of the milling and end-product in wheat (Pasha et al. 2010). Grain hardness is a parameter strongly linked to wheat species. The durum wheat has no D-genome and represents a harder grain texture (Morris and Bhave 2008). The meaning of durum is “hard” in Latin and the species is the hardest of all wheats (Hare 2017).
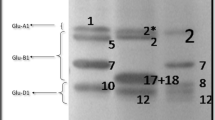
Einkorn kernels showed extra-soft texture (99–306 g) followed by spelt (205–214 g), bread (383–458 g), emmer (596–685 g), and durum (756–885 g) wheats, respectively (Brandolini et al. 2008). Migliorini et al. (2016) reported that grain hardness ranged from soft (34.0%) to hard (82.5%) among the landraces. Similarly, in a study conducted by Bordes et al. (2008), 372 hexaploid wheat accessions including 139 modern cultivars and 233 landraces and old cultivars were evaluated for grain hardness which showed a wide variation ranging from very soft (1.5%) to very hard (99.9%). No difference in grain hardness was reported between landraces and modern wheats in the same study. Cetiner et al. (2020) compared landraces and old and new bread wheat cultivars and stated that two of the three landraces were classified as hard. However, generally old cultivars had a softer grain structure compared to modern cultivars in the same study. The authors attributed this result to the harder wheat selection over the years to develop cultivars with better bread-making quality. The grain hardness (SKCS) scores of the 133 wheat landraces showed a wide range and varied between 28.0% and 99.3%, with an average of 67.9% (Black et al. 2000). Li et al. (2008) reported that frequencies of soft, mixed, and hard wheat samples were 3.9, 20.4, and 75.6%, respectively, in 431 landraces. In hard wheat landraces, distributions of Pin a-D1b, Pin b-D1b, and Pin b-D1p were determined as 38.0, 0.9, and 59.6%, respectively.
6.3 Protein Content and Quality
The functional characteristics of wheat for producing bread or pasta depend on protein content and quality (Cubadda and Marconi 1996). Therefore, improving grain protein content has been the main focus of wheat breeding (Mujeeb-Kazi et al. 2013). Grain protein contents of bread wheat landraces (13.8–16.7%, n = 42) were slightly higher compared to modern cultivars (13.9–15.2%, n = 7) (Akçura 2011). Raciti et al. (2003) reported that about 95% of accessions (n = 116) were characterized by grain protein content higher than (or equal to) the average protein content of control cultivars. It was also reported that the grain protein content of landraces ranged from 13% to 20%, and the variation was wider in tetraploid landraces than hexaploid landraces (Blum et al. 1987). Likewise, grain protein content displayed a declining trend over time of cultivar release, from ~18% in the old wheats to ~16.5% in the modern durum wheats (De Vita et al. 2007), comparable to results by Dinelli et al. (2013).
Landraces and old cultivars tend to have higher grain protein contents than modern cultivars at the same or even lower nitrogen (N) application levels and in soils with lower fertility (Nazco et al. 2012; Giunta et al. 2019). However, this does not justify the classification of them in common as protein-rich crops since their high protein content could be a result of lower grain yield (Čurná and Lacko-Bartošová 2017). An inverse relationship was reported between yield and grain protein content by some researchers (Akçura 2011; Shewry et al. 2013). Similarly, higher average protein content was obtained from 37 Iranian and 42 Mexican durum wheat landraces compared to bread wheat check with a higher thousand kernel weight (Hernández-Espinosa et al. 2019).
The characteristics of grain quality, including protein content, vary both within and across wheat species; however, they are highly affected by the environment (Arzani and Ashraf 2016). The variation on protein content strongly relies on the wheat cultivar, the growing circumstances, the fertility of the soil, and the fertilizer, especially nitrogen (Carson and Edwards 2009). The genotype effect was the strongest on protein content (Hidalgo and Brandolini 2017), and a high heritability value (82.3%) was calculated for grain protein content by Heidari et al. (2016). However, Shewry et al. (2013) reported that nitrogen fertilization had a greater effect on protein content compared to genotype. As a general rule, grain protein content increases with increased nitrogen fertilization rate. Besides, late nitrogen application has a positive effect on quality since it leads to increased protein content (Ottman et al. 2000). Migliorini et al. (2016) found about 30% lower protein content in the second year compared to the first year in the grains of wheat landraces because of the effect of the environment. Sowing time also affected the grain protein content and higher protein content was obtained from delayed sowing time (Fois et al. 2011). Melash et al. (2019) reported that increasing seed rate, from 100 to 175 kg ha−1, reduced the grain protein content, Zeleny sedimentation value, and wet gluten content by 8.7, 9.1, and 10.8%, respectively. It was also reported that the foliar application of FeSO4 in the anthesis stage tended to increase the grain protein content, Zeleny sedimentation value, and wet gluten content compared to the foliar application of ZnSO4.
Protein quality is an important quality characteristic of wheat breeding. There are several physical and chemical tests to evaluate the protein quality of wheat (Hare 2017). Among various predictive tests, SDS sedimentation has been extensively used in wheat breeding programs, being particularly useful when only small flour samples are available. The sedimentation test depends on the swelling and flocculating properties of glutenin protein in dilute lactic acid solution, and the results are correlated to gluten strength and baking quality of wheat. A significant correlation between the sedimentation volume and Alveograph energy (W) value confirmed that the sedimentation test, simple and fast, is a valuable tool for predicting the Alveograph energy (W) value (Vázquez et al. 2012). The positive correlation between SDS sedimentation and gluten index revealed that high SDS sedimentation value was related to strong gluten strength (Dick and Quick 1983).
The variation is high among wheat genotypes in terms of the sedimentation volume. The variation among landraces for SDS sedimentation values has been identified by several researchers, ranging between 43 and 58 ml (n = 50) (Heidari et al. 2016), 22 and 36 ml (n = 20) (Akar et al. 2009), 16 and 24 ml (Blum et al. 1987), and 52 and 62 ml (n = 300) (Sezer et al. 2019). The SDS sedimentation range was 23–83 ml when a group of genotypes (n = 116) was analysed. Of 116 genotypes, 55 genotypes had higher or equal SDS sedimentation values than the control mean value (40 ml) (Raciti et al. 2003). Hernández-Espinosa et al. (2019) reported that average sedimentation volume was slightly higher in Iranian landraces than the Mexican group, comparing 37 Iranian and 42 Mexican durum wheat landraces. Genotype effect was found the strongest on SDS sedimentation value reported by Hidalgo and Brandolini (2017), and quite high heritability values (> 93%) were determined for both SDS and Zeleny sedimentation parameters (Heidari et al. 2016).
Wheat gluten was isolated from flour in 1728 by Beccari and wheat proteins were identified based on their extractability in different solvents: globulins (salt-soluble), albumins (water-soluble), prolamins (gliadin; alcohol-soluble), and glutelins (glutenin; dilute acid-soluble) by Osborne (Wrigley 2010; Kiszonas and Morris 2018). In wheat flour, gluten consisting of gliadins and glutenins accounts for about 80% of the total protein (Hoseney et al. 1969; Shewry et al. 2009). Glutenins, polymeric proteins, are responsible for the strength and elasticity of the dough, while gliadins, monomeric proteins, contribute to dough viscosity and extensibility. Gluten plays an important role in the bread-baking quality by supplying a network formation during the mixing and hydration in the dough. The gluten network provides distinctive features to the dough, such as air holding through the creation of an impermeable membrane around gas cells, resulting in a foamlike baked end-product (Delcour and Hoseney 2010).
Wet gluten can be obtained from wheat flour or meal using the automated gluten washer equipment. It is known that wet gluten content correlates positively with dry gluten content (Desheva et al. 2014). The gluten index value on wheat indicates gluten elasticity and strength and does not firmly rely on protein content. The values higher than 80% represent strong gluten (Migliorini et al. 2016). In a study, including 50 wheat landraces, carried out by Heidari et al. (2016), the dry gluten content had a high heritability value (84.8%) and ranged from 9.9% to 19.7% with a mean of 15.8%. The dry gluten contents and gluten index values of old durum wheat cultivars (n = 14) grown in Italy varied from 10.7% to 14.3% and from 4.5% to 60.6%, respectively (Mefleh et al. 2019). Konvalina and Moudrý (2008) reported that wet gluten and gluten index values of six emmer landraces were between 34.0% and 50.9% and 10% and 48%, respectively. Landraces and old cultivars possess lower gluten index values than modern durum wheat cultivars (De Vita et al. 2017). Breeding resulted in a significant increase in the gluten index, reflected by the development of protein quality (Motzo et al. 2004).
The wheat quality is influenced by the protein content and composition in the endosperm, particularly the contents and proportions of two gluten fractions, gliadins and glutenins, and of their low molecular weight (LMW) and high molecular weight (HMW) subunits (Mefleh et al. 2019). HMW glutenins have a greater effect on dough elasticity and strength than LMW glutenins (Gupta et al. 1991; Gupta and MacRitchie 1994). The gluten subunits come together to form a strong gluten network during dough mixing; however, with continuous dough mixing, LMW glutenins first disassociate from the gluten network, followed by HMW glutenins (Bonilla et al. 2019). Fois et al. (2011) reported that modern durum wheat cultivars with HMW-GS 6 + 8 and 7 + 8 showed superior gluten strength than old (before 1950) and intermediate (1950–1973) durum wheat cultivars with HMW-GS 20. The HMW/LMW ratio ranged from 0.54 to 1.03, from 0.61 to 0.68, and 0.54 to 0.89 for landraces, old, and modern genotypes, respectively. The gliadin to glutenin (Gli/Glu) ratio also varied between 0.59 and 1.18, 0.44 and 0.57, and 0.66 and 0.78, for the aforementioned genotype groups, respectively (Boukid et al. 2018). The presence of γ-45 gliadin and the absence of γ-42 gliadin bands are associated with strong gluten. It was reported that Mediterranean durum wheat landraces (n = 171) possessed 68.9% of γ-45 gliadin, 11.1% of γ-42 gliadin, and 20.0% of both gliadin bands (Nachit et al. 1995).
The time and rate of nitrogen fertilization can influence the gluten fraction proportions. Albumins and globulins were hardly affected by nitrogen application; however, gliadins were more affected than glutenins (Pechanek et al. 1997; Wieser and Seilmeier 1998). Mefleh et al. (2019) reported that additional nitrogen fertilization improved the gliadin fraction by 12.4% and unaffected the glutenin fraction. The gliadin to glutenin (Gli/Glu) ratio ranged from 3.46 to 4.74 among old cultivars, which was similar to the result of the modern cultivar in the same study. Also, sowing time significantly influenced Gli/Glu ratio (Fois et al. 2011).
6.4 Starch and Lipid Contents
Starch provides up to 90% of the average calorie intake in the diet of developing countries and more than 50% in the developed countries (Wang et al. 2015). High-yielding wheat cultivars with high starch content were selected to feed the increasing population by breeders (Boukid et al. 2018). Starch forms 65–70% of the dry matter in wheat grain (Bedõ et al. 2010). The total starch content ranged from 54.0% to 66.8%, from 55.6% to 63.3%, and from 65.1% to 67.8% for landraces, old genotypes, and modern wheats, respectively (Boukid et al. 2018). Alfeo et al. (2018) reported an inverse relationship between starch and protein contents. The starch content of durum wheat landraces ranged from 59.7% to 67.2% in the same study.
Starch, the main component of endosperm, is made up of 25% amylose (a mixture of linear and lightly branched) and 75% amylopectin (monodisperse and highly branched) (Maningat et al. 2009). Although both starch fractions have the same basic structure, their length and degree of branching are different, which influences the physicochemical properties of starch (Sofi et al. 2013). The gelatinization and pasting properties of starch are affected by the amylose content of wheat (Zeng et al. 1997). The proportion of amylose to amylopectin and amylopectin structure affect the processing, organoleptic characteristics, and digestibility in starch-based foodstuffs (Bao et al. 2006). The proportion of these two starch polymers within the starch granules differs, relying on the cereal and its cultivar (McKevith 2004).
The contents of amylose and amylopectin and amylose/amylopectin ratio of 33 lines, belonging to an old cultivar named Bánkúti 1201, ranged from 14.4% to 24.2%, 75.8% to 85.6%, and 0.16 to 0.32, respectively, suggesting that old cultivars are heterogeneous for starch contents (Rakszegi et al. 2003). In a study, carried out by Black et al. (2000), the amylose content of wheat landraces (n = 133) varied between 23.4% and 30.2% with a mean of 27.9%, while those of two commercial wheat cultivars were 27.9% and 29.9%.
Increased amylose is linked with increased resistant starch which is essential in the prevention of diabetes and obesity (Hazard et al. 2014). Therefore, healthy nutrition trends for enriched fibre consumption with low glycaemic food have pushed the growth of high amylose starch as a source of resistant starch acting like dietary fibre (Bertolini 2009). The wider variation was observed in landraces (28.0–60.1%) than old genotypes (51.5–65.9%) and modern genotypes (30.5–58.3%) in terms of resistant starch by Boukid et al. (2018). Dinelli et al. (2013) reported the cultivar and growing season effects on the resistant starch content of wheat. The higher resistant starch contents were obtained from a landrace and an old durum wheat cultivar (6.1–8.2 g/kg) compared to modern durum wheat cultivars (3.0–6.0 g/kg). The cultivars grown in the first year possessed approximately twofold resistant starch content than the ones grown in the second year of the study. Similarly, the higher resistant starch contents were determined at a landrace (39.8 ± 3.8 mg/kg dmb) and an old (44.6 ± 1.2 mg/kg dmb) durum wheat genotypes than modern durum wheat cultivars (12.0–37.0 mg/kg dmb) and Kamut (17.7 ± 1.2 mg/kg dmb) (Marotti et al. 2012).
Lipids are rather a minor component in wheat; however, they play a key role in nutrition, grain storage, and processing like dough mixing and baking. The lipids associate with the gluten proteins to form complexes, which contributes to the stabilization of the gas-cell structure. Therefore, they have important effects on bread volume and final texture of the baked products (Uthayakumaran and Wrigley 2010). Interaction between lipids and starch can influence gelatinization, retrogradation, and pasting properties of wheat starch and the vulnerability of starch to enzyme attack (Copeland et al. 2009).
In wheat, most of the lipids are concentrated in the germ (28.5%) and aleurone (8.0%), with only small amounts in the endosperm (1.5%) (Delcour and Hoseney 2010). The distribution of the lipids within the wheat species varies narrowly. Wheat lipids make up 2.03–2.85%, 1.80–2.85%, 1.88–1.93, and 1.96–2.82% of the weight of the whole einkorn, emmer, bread, and durum grains, respectively (Giambanelli et al. 2013). However, high variability, ranging from 22.4 to 33.7 g/kg, among durum wheat genotypes including a landrace and an old durum wheat cultivar was found by Dinelli et al. (2013). The crude fat content of 30 einkorn landraces grown in Kastamonu province in Turkey ranged from 1.62% to 2.72% with an average of 2.19% (dry matter basis) (Emeksizoğlu 2016). Lipid content was affected by sowing date, higher for spring (1.92–2.85%) compared to fall (1.80–2.65%) sowing (Giambanelli et al. 2013).
6.5 Vitamins and Minerals
Wheat is considered to be a significant source of vitamin B, particularly B1 (thiamine), B2 (riboflavin), B3 (niacin), B6 (pyridoxine), and B9 (folate) (Shewry and Hey 2015). Consuming products made by whole grain contributes to 40% of the suggested daily allowance for B1, 10% for B2, 22% for B3, 33% for B6, and 13% for B9 (Uthayakumaran and Wrigley 2010).
Abdel-Aal et al. (1995) reported that thiamine contents among wheat species were not large, ranging from 0.50 mg/100 g to 0.60 mg/100 g. Riboflavin content was relatively high in einkorn (0.45 mg/100 g) and bread (0.55 mg/100 g) wheat, but was low in spelt (0.14–0.17 mg/100 g). However, spelt had higher content of niacin (2.0–5.7 mg/100 g) compared to einkorn (3.1 mg/100 g) and bread wheat (2.3 mg/100 g). The amount of pyridoxine varied from 0.35 mg/100 g to 0.49 mg/100 g among species. In another study carried out by Stehno et al. (2011), variability ranges were as follows: thiamine (0.29–0.44 mg/100 g), riboflavin (0.108–0.135 mg/100 g), niacin (8.4–10.6 g/100 g), and pyridoxine (0.27–0.45 mg/100 g) contents for eight emmer genotypes compared to bread wheat, with 0.36 mg/100 g thiamine, 0.071 mg/100 g riboflavin, 6.8 mg/100 g niacin, and 0.37 mg/100 g pyridoxine (dry matter basis).
Cereal and cereal products are important sources of folate, a water-soluble form of vitamin B9 and also known as folic acid or folacin. Folate is important for the prevention of neural tube defects, anemia, and cardiovascular disease (Scott et al. 2000; De Wals et al. 2007). In the HEALTHGRAIN project, only the folate content of ancient and modern wheat species was determined. The folate concentrations were found higher in durum (0.74 μg/g dmb) and emmer (0.69 μg/g dmb) wheats compared to einkorn, spelt, and bread wheats (0.58, 0.58, and 0.56 μg/g dmb, respectively) (Piironen et al. 2008). Heritabilities of these vitamins in a G × E study including 26 lines were found quite low, with the highest values being for thiamine (31%) followed by folate (24%), riboflavin (16%), pyridoxine (12%), and niacin (7%) (Shewry et al. 2013).
Major micronutrients in wheat are vitamin E, some B vitamins, and several minerals. These minerals are distributed unequally in the seed and are mostly localized in germ and bran (Uthayakumaran and Wrigley 2010). The recommended daily intake of vitamin E, an important antioxidant, is 10 mg/day according to the European Union Council (EC). Comparing genotypic groups, the highest vitamin E activity was determined as 12.3 mg/kg from landraces, followed by old cultivars (10.8 mg/kg), modern cultivars (10.6 mg/kg), spelt wheat (7.7 mg/kg), and primitive wheat (6.1%). Wheat contributed 12.2–24.5% vitamin E of the daily intake based on EC. Vitamin E activity is known to diminish by heating (Hussain et al. 2012b).
Functional and nutritional properties of wheat are important components of grain quality; however, increasing mineral concentration in grain did not have a priority in genetic improvements. Therefore, modern wheat cultivars have generally lower mineral contents than older cultivars (Fan et al. 2008; Hussain et al. 2012a) because landraces and old cultivars have generally lower thousand kernel weight and grain yield. In a study comparing Zn, Ca, and Fe contents of bread wheat cultivars from obsolete to current, cultivars released between 1965 and 1976, compared with the current cultivars (2001–2008), contained significantly more Zn (18%) and Ca (14%) but similar Fe content (Hussain et al. 2012a). Some mineral element contents of 86 bread wheat landraces grown in Turkey varied from 35.53 to 53.08 mg/kg for Fe, from 22.66 to 38.57 mg/kg for Zn, from 30.92 to 48.58 mg/kg for Mn, from 8.63 to 15.77 mg/kg for B, from 4.12 to 6.69 mg/kg for Cu, from 0.85 to 1.78 mg/kg for Mo, from 2.25 to 5.41 g/kg for K, from 1.02 to 1.69 g/kg for Mg, and from 0.34 to 0.55 g/kg for Ca (dry matter basis). Landraces had especially higher Zn, Fe, and Mn contents than the bread wheat cultivars in the same study (Akcura and Kokten 2017). In a study conducted by Manickavelu et al. (2017), the landraces (n = 267) were more variable and contained higher average values (Fe, Zn, Mn, Mg, P, and K) except for Fe than check cultivars of Japan and Afghanistan. Similarly, Kondou et al. (2016) observed that the landraces showed greater variability than the check cultivars regarding K, P, Mg, and Fe.
Humans require more than 22 mineral elements to meet their metabolic needs. Some of them are needed in large amounts such as Na, Ca, K, and Mg, but others, like Zn, Fe, Cu, Mn, I, and Se are needed in trace amounts (Welch and Graham 2004). Cereals and cereal products provide on average >40% of the daily intake of Fe, >10% of K, 27% of Mg, 30% of Ca, 25% of Zn, and 33% of Cu (Swan 2004). Recommended daily intakes of Zn, Fe, and Ca are usually not achieved in the developing countries (Brown et al. 2001; Gibson 2006). Among the micronutrient insufficiencies, deficiencies of Fe and Zn are mainly important for affecting human health (Ozkan et al. 2007). As a sustainable solution, biofortifying grains with essential minerals that are insufficient in peoples’ diets are recommended (Bouis and Welch 2010; Ficco et al. 2009). Biofortification depends on agronomic and genetic methods to increase the bioavailable amount of minerals in cereals (Hawkesford and Zhao 2007; Hussain et al. 2010). Breeding for biofortification, genetic engineering for more uptake from the soil, and fertilizer application are the main methodologies to increase the contents of mineral elements in grain (Cakmak 2008; Waters et al. 2009; Wang et al. 2008). For instance, there is large genotypic variation in contents of Fe and Zn among 54 einkorn wheat genotypes ranged from 0.21 to 2.16 μg seed−1 for zinc and from 0.54 to 3.09 μg seed−1 for iron, and these variations might be used for biofortification in wheat breeding (Ozkan et al. 2007). Wild emmer is also a significant genetic resource for increasing the concentration of Fe and Zn in modern wheats (Cakmak et al. 2004). Selenium fortification provided more Se accumulation in landraces and obsolete cultivars than in modern cultivars. Besides, Se content in the durum wheat was raised by up to 35-fold that of the untreated application (De Vita et al. 2017). It was also reported that wheat accumulated more Fe than Zn (Manickavelu et al. 2017).
The milling process affects mineral content because the outer layer of the grain is removed. Milling of durum wheat grain into semolina can cause a 40–80% loss of Fe, Zn, Cu, and Mg (Cubadda et al., 2009; Lyons et al., 2005). De Vita et al. (2017) reported that the Se concentration diminished during the milling (11%), while the processing of pasta did not display significant decreases.
There is a strong genetic effect on Zn and Fe accumulation in the grain although there is a significant genotype × environment (G × E) interaction effect on Zn and Fe contents. Further research also showed that there was not an inverse linkage between yield and Zn and Fe contents in the grain. Therefore, it should be possible to increase Zn and Fe contents in wheat grain by breeding (Welch and Graham 2004). High zinc wheat lines were obtained from the biofortification breeding program, using wild relatives and landraces (Velu et al. 2014). Di Silvestro et al. (2012) reported that old bread wheat cultivars had higher mineral contents than modern cultivars when grown under low input management practices. Beside breeding, the biosynthesis and accumulation of minerals are affected by genotype and environment (Migliorini et al. 2016) and farming practices (Rizzello et al. 2015).
It is also possible to increase the bioavailability of minerals in wheat grain by reducing phytic acid (White and Broadley 2009), because phytic acid is an antinutrient and binds positively charged mineral cations such as iron, zinc, and calcium to create insoluble complexes, which inhibits the absorption of the minerals into the body (Weaver and Kannan 2002). Most of the total phosphorus present in wheat grain (75%) is stored as phytic acid, particularly in the germ and aleurone layers of the wheat kernel (Lott and Spitzer, 1980). Phytase activity decreases the phytic acid breakdown in wheat. Hence, the mineral bioavailability is attached to mineral and phytase concentrations. These should be taken into consideration in wheat improvement for biofortification (Mujeeb-Kazi et al. 2013). The phosphorus content is controlled to a large extent by the environment (62%). Wider ranges in inorganic phosphorus content were reported among the modern durum wheat genotypes (0.47–0.76 mg/g), compared to landraces (0.48–0.69 mg/g) and advanced lines (0.46–0.66 mg/g) (Ficco et al. 2009). Çetiner et al. (2018) reported that bread wheat cultivar Tosunbey possessed lower phytic acid content (797 mg/100 g) than old cultivars and landraces, ranging from 1125 to 1606 mg/100 g.
6.6 Phytochemicals and Antioxidants
Wheat is an important source of health-promoting components, particularly phytochemicals and antioxidants as well as the main components of protein, carbohydrate, and lipid (Arzani 2019).
Ferulic acid is the main phenolic component of both the insoluble-bound and the soluble-conjugated fractions in different wheat species (Yilmaz et al. 2015). According to Li et al. (2008), the average total ferulic acid concentrations were similar for spelt, durum, and bread wheat samples (about 400 μg/g dmb), but higher in emmer wheat samples (476 μg/g dmb) and lower in einkorn wheat samples (298 μg/g dmb). This finding is corroborated by the results of Serpen et al. (2008): the ferulic acid content of einkorn wheat was about twofold lower than that of emmer wheat. Ferulic acid is a distinctive trait of old and modern wheat genotypes that the landraces possessed the lower ferulic acid content (0.64–0.85 g/kg) than modern wheat cultivars (1.21–1.36 g/kg) (Piergiovanni 2013).
Alkylresorcinols, one of the main groups of phenolic compounds, are mainly located in the external layers of the wheat grain with high levels (Landberg et al. 2008). A comprehensive study carried out by Ziegler et al. (2016) using whole grain flour of 15 genotypes each of five species grown at four environments showed that the contents of alkylresorcinol varied greatly among the genotypes within each species, and the overall average concentrations of the species were 761 ± 92.3, 743 ± 56.7, 654 ± 47.9, 697 ± 93.6, and 737 ± 90.9 μg/g dmb in bread, spelt, durum, emmer, and einkorn wheat samples, respectively. Ciccoritti et al. (2013) reported that alkylresorcinol mean values were 344 ± 8, 377 ± 17, 321 ± 18, and 286 ± 11 μg g−1 dmb for einkorn, emmer, bread, and durum wheats, respectively. Similarly, the results of the HEALTHGRAIN project displayed significant variations in the total alkylresorcinol content in wheat species, and alkylresorcinol contents were higher in ancient wheats (emmer, einkorn, and spelt) compared to modern wheats (bread and durum) (Andersson et al. 2008).
High heritability values were found for tocochromanols (h2 = 0.88–0.97), steryl ferulates (h2 = 0.88–0.94), and alkylresorcinols (h2 = 0.69–0.97) regarding five wheat species. These results demonstrated that lipophilic antioxidant contents in einkorn, emmer, spelt, bread, and durum wheats were under strong genetic control (Ziegler et al. 2016).
Tocols are a class of lipid-soluble liquids, viscous, synthesized only by photosynthetic plants, and classified as tocopherols and tocotrienols. They both occur as a polar chromanol ring and a hydrophobic 16-carbon side chain. In tocopherols, the side chain is a saturated isoprenoid group, while in tocotrienols, it has three double bonds. Tocopherols and tocotrienols both consist of four derivatives: α-, β-, γ-, and δ-tocols and collectively known as tocochromanols (Hidalgo et al. 2006; Lampi et al. 2008; Okarter et al. 2010; Lachman et al. 2013; Ziegler et al. 2016). But α- and β-tocols are the major derivatives present (Lampi et al. 2008). There are more tocotrienols than tocopherols, and β-tocotrienol is the predominant tocol followed by α-tocotrienol, α-tocopherol, and β-tocopherol in wheat (Hidalgo et al. 2006; Hidalgo and Brandolini 2017).
Though all tocopherols and tocotrienols have antioxidant activity, α-tocopherol is the most effective antioxidant for the breaking free radical driven-chain reactions (Packer 1995). Besides having antioxidant activity, only α-tocopherol has vitamin E activity (Schneider 2005). In addition to their antioxidant properties, the tocochromanols of cereals could have positive health effects such as lowering LDL cholesterol in the blood and the risks of cancer and cardiovascular diseases (Tiwari and Cummins 2009). Furthermore, tocotrienols have potential as neuroprotective dietary factors (Frank et al. 2012). In a study comparing the amount of total tocochromanol among the wheat groups, the landraces with 32.9 ± 3.37 mg/kg was followed by modern cultivars (32.5 ± 0.99 mg/kg), old cultivars (30.3 ± 4.41 mg/kg), spelt wheat (28.9 ± 3.47 mg/kg), and primitive wheat (28.0 ± 5.39 mg/kg) (Hussain et al. 2012b).
Wheat has antioxidant activity because of its lipophilic (carotenoids, tocopherols) and hydrophilic (phenolics, selenium) antioxidant contents (Konvalina et al. 2017). Lachman et al. (2012) reported that spring wheat genotypes possessed lower antioxidant activity (195.8–210.0 mg Trolox/kg dmb) than einkorn (149.8–255.8 mg Trolox/kg dmb) and emmer (215.4–257.6 mg Trolox/kg dmb) wheats. In a study conducted with 26 genotypes of einkorn, emmer, spelt, bread wheat landraces, and spring wheat in three growing seasons, the average antioxidant activity ranged from 225.45 to 400.83 mg Trolox/kg dmb, displaying a broad range among wheat species and genotypes (Konvalina et al. 2017). These results are about twofold higher than the findings of Lachman et al. (2012). This difference in antioxidant activity content is explained by the weather or stress conditions during the growing season and genotype effects by Konvalina et al. (2017). A linear relationship (r = 0.74, p < 0.05) existed between antioxidant activity and total polyphenols (Lachman et al. 2012).
Polyphenols are the most indicative antioxidant compounds in wheat kernel (Migliorini et al. 2016). Polyphenols consist of flavonoids and phenolic acids, and they might be found in the bound insoluble and the free soluble forms (Dinelli et al. 2009; Migliorini et al. 2016). In a study in which five bread wheat landraces grown at two different years and locations, significant differences were found between years and cultivars, showing that the second year and cultivar Gentil Rosso possessed higher amounts of the total, free, and bound polyphenols. These results demonstrated that polyphenols were affected by wheat cultivars and environmental conditions such as abiotic and biotic stresses (Migliorini et al. 2016). It was also reported that the environment was the main source of variation in the total soluble phenolic content although genotype, environment, and their interaction had significant effects on the parameter (Bellato et al. 2013). Dinelli et al. (2009) found that there were no significant differences between the mean values of old and modern wheat cultivars in terms of phenolic and flavonoid compounds. However, old cultivars had slightly higher free (181.8 ± 37.8 μmol GAE/100 g), bound (696.4 ± 53.5 μmol GAE/100 g), and total (878.2 ± 19.0 μmol GAE/100 g) phenolic compounds than modern cultivars. These values were 178.4 ± 51.9, 687.4 ± 91.0, and 865.9 ± 128.9 μmol GAE/100 g for the modern cultivars, respectively. In terms of flavonoid compounds, old cultivar had higher free flavonoid compounds (52.5 ± 22.7 μmol CE/100 g), while modern cultivars possessed higher bound (80.0 ± 15.9 μmol CE/100 g) and total (122.6 ± 25.4 μmol CE/100 g) flavonoid compounds on average. Though the range in mean values of bound, free, and total phenolic contents between modern and old cultivars did not vary significantly, differences between the cultivars were significant.
Phytosterols, consumed with the diet, may have a role in preventing colon cancer (Rao and Janezic 1992) and cardiovascular diseases (Piironen et al. 2000). In a screening study of 175 genotypes of different wheat types, which represent current, uncommon, and obsolete, the highest average total phytosterol content was obtained from einkorn (1054 μg/g dmb), followed by durum (987 μg/g dmb), spelt (928 μg/g dmb), spring (864 μg/g dmb), emmer (857 μg/g dmb), and winter (841 μg/g dmb) wheats. The difference between the lowest (670 μg/g dmb) and highest (1187 μg/g dmb) total phytosterol contents in all wheat genotypes was determined as 77% by Nurmi et al. (2008). The total sterol contents of einkorn and emmer wheats were found similar (554.3–828.5 and 500.8–816.4 mg kg−1 dmb, respectively), higher than bread wheat (440.8–661.8 mg kg−1 dmb) and lower than durum wheat (614.8–929.0 mg kg−1 dmb) by Giambanelli et al. (2013). The most abundant phytosterol in wheat is β-sitosterol (34.2–42.7% of phytosterols) followed by campesterol, sitostanol, and campestanol (Giambanelli et al. 2016). It was also confirmed that the most plentiful phytosterol in wheat types, which contain current, uncommon, and obsolete, was sitosterol (40–61% of total phytosterols), while the highest variation was shown in total stanols (7–31% of total phytosterols) (Nurmi et al. 2008).
Carotenoids, lipid-soluble antioxidants, are produced by most photosynthetic organisms and are accountable for the orange, red, and yellow colours in numerous fruits, flowers, and bird feathers. Lutein is the predominant component of carotenoids followed by zeaxanthin in wheat; however, other carotenoids like α-carotene and β-carotene only present in minor amounts (Hidalgo et al. 2006; Abdel-Aal et al. 2007; Digesù et al. 2009). The total yellow pigment content is a widely used test in durum wheat breeding programs. Significant positive relations were observed between total yellow pigment content and lutein (r = 0.94, p < 0.01) and total carotenoid (r = 0.99, p < 0.01) contents. This shows that the total yellow pigment content or colorimetric method is a reliable test to predict the lutein and total carotenoid contents in wheat (Abdel-Aal et al. 2007). It is also reported that the ratio of carotenoids was 33.2% of the yellow pigment concentration by Digesù et al. (2009).
Since yellow or amber pasta colour is usually desired by consumers, yellow pigment content is an important quality parameter in the evaluation of semolina colour, especially in determining the end-product quality of durum wheat (Digesù et al. 2009). For this reason, wheat breeders have focused on high yellow pigment content during the selection of new wheat cultivars for the last two decades. Therefore, modern durum wheat cultivars have generally higher yellow pigment content than older cultivars (Digesù et al. 2009). The total carotenoid contents of 102 tetraploid wheat accessions ranged from 1.18 to 4.42 μg/g dmb, with a mean of 2.46 μg/g dmb. Durum wheat cultivars (released after 1991) possessed a higher average value (3.11 μg/g dmb) than the older ones (released in the period 1971–1991) (2.56 μg/g dmb) and landraces (before 1971) (2.33 μg/g dmb) (Digesù et al. 2009). Dinelli et al. (2013) observed significant differences between durum wheat genotypes (n = 8), including a landrace and an old durum wheat cultivar, in terms of lutein and total carotenoid contents. Total carotenoid contents of genotypes varied between 3.28 and 6.09 μg/g, while lutein contents of those changed from 1.50 to 3.23 μg/g which was almost half of the total carotenoid amount. Nazco et al. (2012) compared the yellow colour indexes of 154 durum wheat landraces, originating from 20 different Mediterranean countries, as three groups (Eastern, North Balkan, and Western), to those of 18 modern durum wheat cultivars. The landraces from the Eastern Mediterranean countries showed the widest variability (11.5–17.6) with a mean of 15.3, but lower than modern cultivars (15.9). North Balkan (14.6) and Western (14.8) Mediterranean countries possessed similar yellow index values determined at whole grain flour. Large and significant variation was determined in carotene contents of tetraploid landraces, ranging from 1.5 to 5.5 ppm by Blum et al. (1987). Similarly, Akar et al. (2009) reported that there was a large variation among the tetraploid landraces (n = 20) in terms of semolina b colour value, varying between 22 and 29.
Carotenoid concentration and lutein content were under genetic control (Lachman et al. 2013; Ziegler et al. 2016). High heritability values were calculated for total carotenoid (0.94), lutein (0.93), and yellow pigment (>0.91) concentrations, intermediate values for α-carotene (0.79) and β-cryptoxanthin (0.72) concentrations, and relatively low values for β-carotene (0.57) and zeaxanthin (0.48) concentrations by Digesù et al. (2009). Yellow pigment content is present at different levels in wheat cultivars and species. Einkorn had the highest lutein content ranging from 6.37 to 8.46 μg/kg dmb in whole flour with an average value of 7.41 μg/kg dmb, higher than durum (5.41 μg/kg dmb), emmer (3.97 μg/kg dmb), bread (2.11 μg/kg dmb), and spelt (1.47 μg/kg dmb) wheat species (Abdel-Aal et al. 2007).
Starr et al. (2015) investigated the volatile compound profiles of 64 wheat cultivars and 17 landraces. A large diversity in volatile profiles happened among wheat samples that landraces had higher levels of alcohols, esters, and some furans, while modern cultivars possessed higher levels of pyrazines, terpenes, and straight-chained aldehydes.
6.7 Dietary Fibre and β-Glucan
Dietary fibre is described as the edible part of plants or similar carbohydrates which resist digestion and absorption in the small intestine while they are completely or partially fermented in the large intestine (Gebruers et al. 2008).
In the HEALTHGRAIN cereal diversity screening program, among wheat species, bread wheat genotypes with 11.5–18.3 g/100 g contained the highest level of dietary fibre compared to other wheat species, such as durum (10.7–15.5 g/100 g) and spelt (10.7–13.9 g/100 g) wheats, whereas wild wheats such as einkorn (9.3–12.8 g/100 g) and emmer (7.2–12.0 g/100 g) had the lowest values (Gebruers et al. 2008). Similarly, the highest total dietary fibre content was found in bread wheat (12.3% dmb), followed by spelt, einkorn, and emmer wheats with mean values of 10.3, 8.7, and 7.9% dmb, respectively (Løje et al. 2003). The total, soluble, and insoluble dietary fibre contents of durum wheat genotypes, including two landraces and one old wheat, ranged between 127.4 and 199.7, 18.1 and 37.1, and 102.3 and 180.8 g/kg dmb, respectively (Marotti et al. 2012). The result of total dietary fibre obtained from the previous study was about 42% higher on average than that reported by Gebruers et al. (2008), where the range in total dietary fibre content was 107.0–155.0 g/kg dmb. This variation was explained by the effects of both genotype and environment on dietary fibre content (Gebruers et al. 2010; Shewry et al. 2010). Beside genotype and environment effects, the cultivar × year interaction effect was also reported on the total dietary fibre contents of durum wheat genotypes including one landrace and old cultivar by Dinelli et al. (2013).
The most essential dietary fibre components are the non-starch polysaccharide arabinoxylans (AX), which are the most plentiful dietary fibre, mixed-linkage β-glucans, cellulose, and the non-polysaccharide compound lignin, which are all cell wall components (Gebruers et al. 2008; Bedõ et al. 2010). AX have many health benefits such as immunomodulatory activity, attenuate type II diabetes, cholesterol-lowering activity, faecal bulking effect, enhanced absorption of certain minerals, and prebiotics effect (Mendis and Simsek 2014). Beside health benefits, AX affect water-binding capacity, rheology, starch retrogradation, and gas retention in dough (Simsek et al. 2011).
In a comprehensive study carried out by Gebruers et al. (2008), beside total dietary fibre, bread wheat genotypes had the widest variation in total arabinoxylan content (TO-AX) varying between 1.35 and 2.75% dmb. TO-AX ranging from 1.70% to 2.35% dmb, from 1.60% to 2.15% dmb, from 1.45% to 2.35% dmb, and from 1.40% to 1.95% dmb were reported for durum, spelt, einkorn, and emmer wheat flours, respectively. According to Marotti et al. (2012), the TO-AX of durum wheat genotypes, including two landraces and an old wheat, varied between 26.9 and 35.6 g/kg dmb, with a mean value of 32.7 g/kg dmb. In a study including old and modern bread wheats and landraces, the modern wheats possessed the highest mean TO-AX value (8.03%) compared to old wheats (7.60%) and the landraces (6.41%). It was also stated that modern breeding had no negative effects on the contents of AX components when comparing the wheat groups (Cetiner et al. 2020). Based on the literature, it is obvious that there is a significant variation in TO-AX of wheat genotypes. This phenomenon was explained by the effects of genotype and environment on TO-AX (Li et al. 2009; Gebruers et al. 2010; Simsek et al. 2011). Finnie et al. (2006) concluded that cultivar was the main source of variability for TO-AX although the effects of cultivar and environment were statistically significant on TO-AX.
The water-extractable arabinoxylan (WE-AX) accounts for 25–30% of TO-AX, while water-unextractable arabinoxylan (WU-AX) accounts for the rest of TO-AX (Meuser and Suckow 1986). The variation for WE-AX in wheat species was wide. The largest variation in WE-AX in wheat flour was observed for bread wheat (from 0.30% to 1.40% dmb), while narrow variation (from 0.50% to 0.65% dmb) was found in einkorn. Durum and spelt wheats had a similar range from 0.25% to 0.55% dmb and from 0.30% to 0.45% dmb, respectively. The lowest value (0.15–0.55% dmb) was obtained from emmer wheat in the same study (Gebruers et al. 2008). In a study, comparing the landraces and old and modern wheats in terms of WE-AX and WU-AX contents, old wheats possessed the highest mean WE-AX value (0.81%), followed by modern wheats (0.79%) and landraces (0.67%). WU-AX mean values were 7.24, 6.79, and 5.74% for modern wheats, old wheats, and landraces, respectively (Cetiner et al. 2020). Finnie et al. (2006) reported that WE-AX content was more greatly affected by genotype, while WU-AX content was primarily influenced by the environment.
Along with arabinoxylans, β-glucan is one of the most important dietary fibre components in wheat (Marotti et al. 2012). High β-glucan content is desirable to increase the health benefits by lowering blood cholesterol levels (Lia et al. 1997). β-Glucans are mostly located in the cell wall of the endosperm (Laroche and Michaud 2007). In a study carried out by Biel et al. (2016), the levels of β-glucans were found dependent on cultivar only. However, the contents of β-glucans in wheat grain varied between species, cultivars, and environmental conditions, ranging from 0.25% to 1.40% of dry weight (Marconi et al. 1999; Løje et al. 2003; Gebruers et al. 2008; Biel et al. 2016). In a comprehensive study, Gebruers et al. (2008) pointed out significant variations in β-glucan content of five wheat species. On average, the species of einkorn, emmer, and durum wheat possessed half of the β-glucan amount, noted for the spelt and bread wheat species. The ranges of variation were 0.25–0.35%, 0.30–0.40%, 0.55–0.70%, 0.50–0.95%, and 0.25–0.45% of dry weight in einkorn, emmer, spelt, bread, and durum whole meals, respectively. Marotti et al. (2012) reported significant differences in β-glucan contents of durum wheat genotypes, including two landraces and an old durum wheat cultivar. However, β-glucan content, ranging from 2.4 to 4.1 g/kg dmb, in all wheat genotypes was lower than those determined in other cereal grains such as oat and barley.
6.8 Rheological Properties
Rheology of dough plays an important role in determining the quality of wheat-based products (Kundu et al. 2017). Physical dough analyses such as Farinograph, Alveograph, and Mixograph are used to predict the dough mixing features in the world (Marchylo and Dexter 2001). Farinograph analysis is used widely to determine particularly water absorption of flour. Water absorption has a key role in baked products, affecting each step of the process, yield, and end-product quality. Water absorption is the amount of water which is needed to produce a dough of suitable consistency. Amount of protein, damaged starch, and non-starch polysaccharide (in particular pentosans) contents affect water absorption of flour. Protein can absorb water about twice its weight when the dough is mixing. Some polysaccharides can absorb even more water. Water absorptions of flour can vary between 50% and 70%, depending on grain hardness, milling, and the desired flour specifications (Miskelly et al. 2010).
Comparing breeding periods, the lowest Farinograph water absorption value (WA) was 50.4% at initial cultivars (released from the mid-1940s until the Green Revolution), followed by landraces (released before 1940) with 51.5% WA, and modern wheat cultivars (released from 1970 to 2001) with 53.1% WA (Sanchez-Garcia et al. 2015). Evaluating 330 Chinese wheat cultivars as four groups based on released year, development time, stability, and Farinograph quality number of cultivars released after 2000 were 17.9, 71.1, and 44.3% higher than those of cultivars released between 1949 and 1976. The results showed that these Farinograph characteristics increased significantly over time (Yang et al. 2014). Farinograph water absorption was associated positively with protein content (Corbellini et al. 1999). Sanchez-Garcia et al. (2015) reported that water absorption was strongly under genotype effects which accounted for 73.8% of the total variation.
Parameters obtained from Alveograph, one of the rheological analyses, are P (the pressure, related to the height of the curve), L (the length, extensibility), P/L (tenacity/extensibility ratio), and W (the work, related to the area of the curve). Especially, the Alveograph W value determines the strength of gluten by measuring the force needed to blow the bubble of dough until it ruptures. De Vita et al. (2007) compared the Alveograph parameters of Italian landraces and durum wheat genotypes. Landraces possessed the lowest baking strength (W values: 30–99 10–4 J) and dough-gluten properties. In another study, modern cultivars showed about three times and twice more Alveograph W and P values of the landraces, respectively (Sanchez-Garcia et al. 2015). Similarly, 35 lines, derived from landraces, possessed lower Alveograph W values (ranging from 37 to 253 10–4 J) than bread wheat checks (ranging from 135 to 431 10–4 J) (Guzmán et al. 2014). Significant differences were observed between landraces, ranging from 58 to 161 10–4 J, for Alveograph W values (Migliorini et al. 2016). Comparing between 37 Iranian and 42 Mexican durum wheat landraces, Hernández-Espinosa et al. (2019) found that Mexican landraces had a higher average Alveograph W value than the Iranian group. Although genotype, environment (year), and their interaction had significant effects on the Alveograph W (Migliorini et al. 2016), the genotype effect was the strongest on the Alveograph W and P parameters (Sanchez-Garcia et al. 2015). A relatively high heritability value (0.61) was determined for dough strength (W), while Alveograph tenacity (P), extensibility (L), and P/L ratio possessed lower heritability values (ranging from 0.14 to 0.31) (Igrejas et al. 2002).
Mixograph is also a useful instrument for determining the gluten strength of wheat. For instance, midline peak time, a parameter of Mixograph, shows the highest correlation with gluten strength. A high midline peak time value shows strong gluten strength (Beta et al. 2019). The landraces and old cultivars showed a wider range of variation than modern wheat cultivars in terms of almost all traits of Alveograph and Mixograph (Bordes et al. 2008). Similarly, a large variation was determined among the Mixograph scores of landraces, ranging from 2 to 9. However, modern wheat cultivars possessed a higher mean Mixograph score than landraces (Blum et al. 1987). Guzmán et al. (2014) also reported that bread wheat check samples possessed about twice higher Mixograph dough development time and dough strength values on average than the lines derived from landraces. Likewise, the modern durum wheat cultivars showed the strongest dough properties, having higher work input to peak (WIP) and time-bandwidth (ETBW) values, compared to older cultivars. Sowing time affected WIP but not ETBW (Fois et al. 2011). Although both genotype and year had significant effects on almost all 11 Mixograph parameters, belonging to 150 lines of a landrace population, quite low heritability values ranging from 0.08 to 0.40 were reported by Igrejas et al. (2002).
The use of Rapid Visco Analyser (RVA) has been increased as an instrument in wheat breeding in the past two decades. The RVA instrument is used to determine various parameters associated with the starch pasting characteristics of wheat (e.g. viscosity, pasting temperature) (Cozzolino 2016). The range of peak viscosity was 175–295 Rapid Visco Analyser units (RVU) with an average of 254 RVU among the wheat landraces (n = 133) (Black et al. 2000). Bhattacharya et al. (1997) reported that the pasting characteristics of 242 hexaploid wheat landraces showed wide diversity in all RVA parameters. The average peak viscosity of modern wheats was 260 RVU and varied between 185 and 355 RVU, while it ranged from 139 to 305 RVU for landraces, and positively correlated (r = 0.73, p < 0.001) with flour swelling volume. High peak viscosity, low setback, high breakdown, and low final viscosity are the properties concerned with high eating quality of Japanese white salted noodles (Black et al. 2000).
6.9 Wheat Landrace-Based Foodstuffs
Limited studies have been carried out to determine the end-product quality of wheat landraces. In a study comparing bread volumes of 37 Iranian and 42 Mexican durum wheat landraces, bread volumes of both landrace groups were on average lower than bread wheat control; however, 14 Mexican and 6 Iranian landraces possessed similar or higher bread volumes than check (Hernández-Espinosa et al. 2019). Bread volumes of 35 lines derived from landraces, ranging from 495 to 745 ml with a mean of 698.9 ± 42.9 ml, were lower than bread wheat checks ranging between 670 and 900 ml (Guzmán et al. 2014). A landrace named Tir has moderate baking quality. Although it is not preferred by the bread wheat industry, local people mostly use it for making the lavash (flatbread) baked in tandoor (Ülker et al. 2019). Regarding the sensory analysis, breads produced from five landraces were preferred by consumers (Migliorini et al. 2016). In terms of aroma profile, landraces and old bread wheat cultivars had a softer aroma, while modern cultivars possessed a much stronger aroma (Starr et al. 2013).
Bulgur is a whole grain product that is commonly produced from Triticum durum. However, bulgur in different cities or regions of Turkey is produced with specific landraces such as einkorn and emmer to achieve regionally desired end-use product characteristics. On average, 55.7% of wheat landrace production is used in bread-making such as lavash, 35.8% is used in bulgur-making, and 2.6% is in pasta-making such as erişte in Turkey. However, about half of wheat landrace production is used in bulgur-making in the Mediterranean and Southeastern Anatolian regions of Turkey (Kan et al. 2015).
Protein content, rather than gluten strength, is the main determinant of high temperature dried pasta cooking quality (Dexter and Matsuo 1977; D’Egidio et al. 1990). Some old durum wheat cultivars such as Senatore Cappelli possessed good pasta texture compared to modern durum wheat cultivars (Fois et al. 2011). Protein quality and quantity, as well as starch gel properties, are the most important factors responsible for the oil content of instant noodles. Instant noodles made from Iranian wheat landraces possessed intermediate oil contents, compared to commercial wheat flours and US/Canadian samples (Wu et al. 2006).
Low protein content and hardness are the crucial parameters for predicting biscuit-making quality. Beside these parameters, some particular storage proteins such as HMW-GS 13 + 16 glutenin subunits are particularly useful in evaluating biscuit quality. In a comprehensive study comparing 98 lines from a landrace population named Barbela, biscuit mass, cooking time, length, width, thickness, density, and surface appearance of lines varied between 10.9 and 14.3 g, 6.58 and 10.12 min, 6.4 and 6.9 cm, 4.6 and 6.0 cm, 9.2 and 13.0 mm, 0.13 and 0.38 g/cm3, and 1.0 and 4.5, respectively (Igrejas et al. 2002).
6.10 Conclusions
Wheat is the second most important grain after maize in the world. In terms of wheat production, it is not appropriate to make comparisons of the modern wheat cultivars and the landraces with their negative aspects, making each other useless. Instead of negative approaches, it should be emphasized which one should be preferred according to the conditions. Under conditions where wheat has the potential to provide high grain yield, choosing landraces instead of modern wheat cultivars will be risky in terms of food security. On the other hand, landraces have the opportunity to be produced under low yield, low input, and high-stress conditions. The landraces are naturally suitable for organic agriculture and environmental friendly practices. The landraces also provide some opportunities for breeders since they generally represent considerably wider genetic diversity than modern cultivars. Therefore, they could lead to extending the genetic base of modern wheat cultivars. The landraces are also suitable for regions with high local demands. The use of landraces could represent a strategy for local communities in the production of traditional niche food products.
Wheat is an important source of health-promoting components, particularly phytochemicals and antioxidants as well as the main components of protein, carbohydrate, and lipid. There are no extreme differences between landraces and modern wheat cultivars in terms of various nutritional components. Most of the components which are beneficial for health are mainly concentrated in the germ and aleurone layers of the wheat kernel. Maximum benefit from these components of wheat could be possible by consuming it as whole grain products. For this reason, consuming wheat as whole grain instead of separating it as a landrace or modern wheat will be more beneficial for health.
From the technological point of view, limited studies were carried out to screen the end-product quality characteristics of wheat landraces. Therefore, further detailed studies are required for the determination of the end-product quality of wheat landraces.
References
Abdel-Aal ES, Hucl P, Sosulski FW (1995) Compositional and nutritional characteristics of spring einkorn and spelt wheats. Cereal Chem 72:621–624
Abdel-Aal ESM, Young JC, Rabalski I et al (2007) Identification and quantification of seed carotenoids in selected wheat species. J Agric Food Chem 55:787–794
Akar T, Mert Z, Yazar S et al (2009) Sustainable use of winter durum wheat landraces under Mediterranean conditions. Afr J Biotechnol 8:4108–4116
Akçura M (2011) The relationships of some traits in Turkish winter bread wheat landraces. Turk J Agric For 35:115–125
Akcura M, Kokten K (2017) Variations in grain mineral concentrations of Turkish wheat landraces germplasm. Qual Assur Saf Crop Foods 9:153–159
Alfeo V, Jaskula-Goiris B, Venora G et al (2018) Screening of durum wheat landraces (Triticum turgidum subsp. durum) for the malting suitability. J Cereal Sci 83:101–109
Andersson AAM, Kamal-Eldin A, Fras A et al (2008) Alkylresorcinols in wheat varieties in the HEALTHGRAIN diversity screen. J Agric Food Chem 56:9722–9725
Arzani A (2019) Emmer (Triticum turgidum ssp. dicoccum) flour and bread. In: Preedy V, Watson R (eds) Flour and breads and their fortification in health and disease prevention, 2nd edn. Elsevier, Amsterdam, pp 89–98
Arzani A, Ashraf A (2016) Smart engineering of genetic resources for enhanced salinity tolerance in crop plants. Crit Rev Plant Sci 35:146–189
Azeez MA, Adubi AO, Durodola FA (2018) Landraces and crop genetic improvement. In: Grillo O (ed) Rediscovery of landraces as a resource for the future. InTechOpen. https://doi.org/10.5772/intechopen.75944
Bao J, Shen Y, Jin L (2006) Determination of thermal and retrogradation properties of rice starch using near-infrared spectroscopy. J Cereal Sci 46:75–81
Bedõ Z, Láng L, Rakszegi M (2010) Breeding for grain-quality traits. In: Wrigley CW, Batey IL (eds) Cereal grains assessing and managing quality, 1st edn. Woodhead Publishing, Oxford, pp 369–392
Bellato S, Ciccoritti R, Del Frate V et al (2013) Influence of genotype and environment on the content of 5-n alkylresorcinols, total phenols and on the antiradical activity of whole durum wheat grains. J Cereal Sci 57:162–169
Bertolini AC (2009) Trends in starch applications. In: Bertolini AC (ed) Starches characterization, properties, and applications, 1st edn. CRC Press, Boca Raton, pp 1–19
Beta T, Li W, Apea-Bah FB (2019) Flour and bread from black, purple, and blue-colored wheats. In: Preedy V, Watson R (eds) Flour and breads and their fortification in health and disease prevention, 2nd edn. Elsevier, pp 75–88
Bhattacharya M, Jafari-Shabestari J, Qualset CO, Corke H (1997) Diversity of starch pasting properties in Iranian hexaploid wheat landraces. Cereal Chem 74:417–423
Biel W, Stankowski S, Jaroszewska A et al (2016) The influence of selected agronomic factors on the chemical composition of spelt wheat (Triticum aestivum ssp. spelta L.) grain. J Integr Agric 15:1763–1769
Black CK, Panozzo JF, Wright CL, Lim PC (2000) Survey of white salted noodle quality characteristics in wheat landraces. Cereal Chem 77:468–472
Blum A, Sinmena B, Golan G, Mayer J (1987) The grain quality of landraces of wheat as compared with modern cultivars. Plant Breed 99:226–233
Bonilla JC, Schaber JA, Bhunia AK, Kokini JL (2019) Mixing dynamics and molecular interactions of HMW glutenins, LMW glutenins, and gliadins analyzed by fluorescent co-localization and protein network quantification. J Cereal Sci 89:102792
Bordes J, Branlard G, Oury FX et al (2008) Agronomic characteristics, grain quality and flour rheology of 372 bread wheats in a worldwide core collection. J Cereal Sci 48:569–579
Bouis HE, Welch RM (2010) Biofortification: a sustainable agricultural strategy for reducing micronutrient malnutrition in the global south. Crop Sci 50:20–32
Boukid F, Vittadini E, Prandi B et al (2018) Insights into a century of breeding of durum wheat in Tunisia: the properties of flours and starches isolated from landraces, old and modern genotypes. LWT 97:743–751
Brandolini A, Hidalgo A, Moscaritoloa S (2008) Chemical composition and pasting properties of einkorn (Triticum monococcum L. subsp. monococcum) whole meal flour. J Cereal Sci 47:599–609
Brandolini A, Hidalgo A, Plizzari L et al (2011) Impact of genetic and environmental factors on einkorn wheat (Triticum monococcum L. subsp. monococcum) polysaccharides. J Cereal Sci 53:65–72
Brown KH, Wuehler SE, Peerson JM (2001) The importance of zinc in human nutrition and estimation of the global prevalence of zinc deficiency. Food Nutr Bull 22:113–125
Cakmak I (2008) Enrichment of cereal grains with zinc: agronomic or genetic biofortification. Plant Soil 302:1–17
Cakmak I, Torun A, Millet E et al (2004) Triticum dicoccoides: an important genetic resource for increasing zinc and iron concentration in modern cultivated wheat. Soil Sci Plant Nutr 50:1047–1054
Carson GR, Edwards NM (2009) Criteria of wheat and flour quality. In: Khan K, Shewry PR (eds) Wheat: chemistry and technology, 4th edn. American Association of Cereal Chemists, Minnesota, pp 97–118
Çetiner B, Tömösközi S, Schall E et al (2018) Comparison of some landraces and a modern wheat in terms of quality, functional properties and gluten/gliadin content. Paper presented at the LACC4 – 4th ICC Latin American Cereals Conference, Mexico City, Mexico, 11–14 March 2018
Cetiner B, Tömösközi S, Török K et al (2020) Comparison of the arabinoxylan composition and physical properties of old and modern bread wheat (Triticum aestivum L.) and landraces genotypes. Cereal Chem 97:505–514
Ciccoritti R, Carbone K, Bellato S et al (2013) Content and relative composition of some phytochemicals in diploid, tetraploid and hexaploid Triticum species with potential nutraceutical properties. J Cereal Sci 57:200–206
Copeland L, Blazek J, Salman H, Tang MC (2009) Form and functionality of starch. Food Hydrocoll 23:1527–1534
Corbellini M, Empilli S, Vaccino P et al (1999) Einkorn characterization for bread and cookie production in relation to protein subunit composition. Cereal Chem 76:727–733
Cozzolino D (2016) The use of the rapid visco analyser (RVA) in breeding and selection of cereals. J Cereal Sci 70:282–290
Cubadda R, Marconi E (1996) Technological and nutritional aspects in emmer and spelt. In: Padulosi S, Hammer K, Heller J (eds) Hulled wheat, proceedings of the first international workshop on hulled wheats, 21–22 July 1995, Pascoli, Tuscany, Italy, p 203–211
Cubadda F, Aureli F, Raggi A et al (2009) Effect of milling, pasta making and cooking on minerals in durum wheat. J Cereal Sci 49:92–97
Čurná V, Lacko-Bartošová M (2017) Chemical composition and nutritional value of emmer wheat (Triticum dicoccon Schrank): a review. J Cent Eur Agric 18:117–134
D’Egidio MG, Mariani BM, Nardi S et al (1990) Chemical and technological variables and their relationships: a predictive equation for pasta cooking quality. Cereal Chem 67:275–281
De Vita P, Nicosia OLD, Nigro F et al (2007) Breeding progress in morpho-physiological, agronomical and qualitative traits of durum wheat cultivars released in Italy during the 20th century. Eur J Agron 26:39–53
De Vita P, Platani C, Fragasso M et al (2017) Selenium-enriched durum wheat improves the nutritional profile of pasta without altering its organoleptic properties. Food Chem 214:374–382
De Wals P, Tairou F, Van Allen MI et al (2007) Reduction in neural-tube defects after folic acid fortification in Canada. N Engl J Med 357:135–142
Delcour JA, Hoseney RC (2010) Principles of cereal science and technology. American Association of Cereal Chemists, Minnesota
Desheva G, Valchinova E, Kyosev B et al (2014) Grain physical characteristics of and bread-making quality of alternative wheat cereals towards common and durum wheat. Emirates J Food Agric 26:418–424
Dexter JE, Matsuo RR (1977) Influence of protein content on some durum wheat quality parameters. Can J Plant Sci 57:717–727
Di Silvestro R, Marotti I, Bosi S et al (2012) Health-promoting phytochemicals of Italian common wheat varieties grown under low-input agricultural management. J Sci Food Agric 92:2800–2810
Dick JW, Quick JS (1983) A modified screening test for rapid estimation of gluten strength in early-generation durum wheat breeding lines. Cereal Chem 60:315–318
Dietrich O, Heun M, Notroff J et al (2012) The role of cult and feasting in the emergence of Neolithic communities. New evidence from Göbekli Tepe, south-eastern Turkey. Antiquity 86:674–695
Digesù AM, Platani C, Cattivelli L et al (2009) Genetic variability in yellow pigment components in cultivated and wild tetraploid wheats. J Cereal Sci 50:210–218
Dinelli G, Segura Carretero A, Di Silvestro R et al (2009) Determination of phenolic compounds in modern and old varieties of durum wheat using liquid chromatography coupled with time-of-flight mass spectrometry. J Chromatogr A 1216:7229–7240
Dinelli G, Marotti I, Di Silvestro R et al (2013) Agronomic, nutritional and nutraceutical aspects of durum wheat (Triticum durum Desf.) cultivars under low input agricultural management. Ital J Agron 8:85–93
Dotlačil L, Hermuth J, Stehno Z et al (2010) How can wheat landraces contribute to present breeding? Czech J Genet Plant Breed 46:70–74
Emeksizoğlu B (2016) Kastamonu yöresinde yetiştirilen siyez (Triticum monococcum L.) buğdayının bazı kalite özellikleri ile bazlama ve erişte yapımında kullanımının araştırılması. Dissertation, University of Ondokuz Mayıs (in Türkish)
Fan MS, Zhao FJ, Fairweather-Tait SJ et al (2008) Evidence of decreasing mineral density in wheat grain over the last 160 years. J Trace Elem Med Biol 22:315–324
Ficco DBM, Riefolo C, Nicastro G et al (2009) Phytate and mineral elements concentration in a collection of Italian durum wheat cultivars. Field Crops Res 111:235–242
Finnie SM, Bettge AD, Morris CF (2006) Influence of cultivar and environment on water-soluble and water-insoluble arabinoxylans in soft wheat. Cereal Chem 83:617–623
Fois S, Schlichting L, Marchylo B et al (2011) Environmental conditions affect semolina quality in durum wheat (Triticum turgidum ssp. durum L.) cultivars with different gluten strength and gluten protein composition. J Sci Food Agric 91:2664–2673
Frank J, Chin XWD, Schrader C et al (2012) Do tocotrienols have potential as neuroprotective dietary factors? Ageing Res Rev 11:163–180
Gebruers K, Dornez E, Boros D et al (2008) Variation in the content of dietary fiber and components thereof in wheats in the HEALTHGRAIN diversity screen. J Agric Food Chem 56:9740–9749
Gebruers K, Dornez E, Bedõ Z et al (2010) Environment and genotype effects on the content of dietary fiber and its components in wheat in the HEALTHGRAIN diversity screen. J Agric Food Chem 58:9353–9361
Giambanelli E, Ferioli F, Koçaoglu B et al (2013) A comparative study of bioactive compounds in primitive wheat populations from Italy, Turkey, Georgia, Bulgaria and Armenia. J Sci Food Agric 93:3490–3501
Giambanelli E, Ferioli F, D’Antuono LF (2016) Assessing the effect of traditional hulled wheat processing on bioactive compounds retention. J Cereal Sci 72:60–68
Gibson RS (2006) Zinc: the missing link in combating micronutrient malnutrition in developing countries. Proc Nutr Soc 65:51–60
Giunta F, Pruneddu G, Motzo R (2019) Grain yield and grain protein of old and modern durum wheat cultivars grown under different cropping systems. Field Crops Res 230:107–120
Gupta RB, MacRitchie F (1994) Allelic variation at glutenin subunit and gliadin loci, glu-1, glu-3 and gli-1 of common wheats. II. Biochemical basis of the allelic effects on dough properties. J Cereal Sci 19:19–29
Gupta RB, MacRitchie F, Shepherd KW, Ellison F (1991) Relative contributions of LMW and HMW glutenin subunits to dough strength and dough stickiness of bread wheat. In: Bushuk W, Tkachuk R (eds) Proc 4th Int workshop gluten proteins. St. Paul, Minnesota, pp 71–80
Guzmán C, Medina-Larqué AS, Velu G et al (2014) Use of wheat genetic resources to develop biofortified wheat with enhanced grain zinc and iron concentrations and desirable processing quality. J Cereal Sci 60:617–622
Hare R (2017) Durum wheat: grain-quality characteristics and management of quality requirements. In: Wrigley C, Batey I, Miskelly D (eds) Cereal grains: assessing and managing quality, 2nd edn. Elsevier, Duxford, pp 135–152
Hawkesford MJ, Zhao FJ (2007) Strategies for increasing the selenium content of wheat. J Cereal Sci 46:282–292
Hazard B, Zhang X, Naemeh M, Dubcovsky J (2014) Registration of durum wheat germplasm lines with combined mutations in SBEIIa and SBEIIb genes conferring increased amylose and resistant starch. J Plant Regist 8:334–338
Heidari B, Padash S, Dadkhodaie A (2016) Variations in micronutrients, bread quality and agronomic traits of wheat landrace varieties and commercial cultivars. Aust J Crop Sci 10:377–384
Hernández-Espinosa N, Payne T, Huerta-Espino J et al (2019) Preliminary characterization for grain quality traits and high and low molecular weight glutenins subunits composition of durum wheat landraces from Iran and Mexico. J Cereal Sci 88:47–56
Hidalgo A, Brandolini A (2017) Nitrogen fertilization effects on technological parameters and carotenoid, tocol and phenolic acid content of einkorn (Triticum monococcum L. subsp. monococcum): a two-year evaluation. J Cereal Sci 73:18–24
Hidalgo A, Brandolini A, Pompei C et al (2006) Carotenoids and tocols of einkorn wheat (Triticum monococcum ssp. monococcum L.). J Cereal Sci 44:182–193
Hoseney RC, Finney KF, Shogren MD et al (1969) Functional (breadmaking) and biochemical properties of wheat flour components. III. Characterisation of gluten protein fractions obtained by ultracentrifugation. Cereal Chem 46:126–135
Hussain S, Maqsood MA, Rahmatullah M (2010) Increasing grain zinc and yield of wheat for the developing world: a review. Emir J Food Agric 22:326–339
Hussain S, Maqsood MA, Rengel Z, Khan MK (2012a) Mineral bioavailability in grains of Pakistani bread wheat declines from old to current cultivars. Euphytica 186:153–163
Hussain A, Larsson H, Olsson ME et al (2012b) Is organically produced wheat a source of tocopherols and tocotrienols for health food? Food Chem 132:1789–1795
Igrejas G, Guedes-Pinto H, Carnide V et al (2002) Genetical, biochemical and technological parameters associated with biscuit quality. II. Prediction using storage proteins and quality characteristics in a soft wheat population. J Cereal Sci 36:187–197
Kan M, Küçükçongar M, Keser M et al (2015) Wheat landraces in farmers’ fields in Turkey: national survey, collection and conservation, 2009-2014. Food and Agriculture Organization of the United Nations (FAO), Ankara
Kantor LS, Variyam JN, Allshouse JE et al (2001) Choose a variety of grain daily, especially whole grains: a challenge for consumers. J Nutr 131:473–486
Kiszonas AM, Morris CF (2018) Wheat breeding for quality: a historical review. Cereal Chem 95:17–34
Kondou Y, Manickavelu A, Komatsu K et al (2016) Analysis of grain elements and identification of best genotypes for Fe and P in Afghan wheat landraces. Breed Sci 66:676–682
Konvalina P, Moudrý J (2008) Quality parameters of emmer wheat landraces. J Cent Eur Agric 9:539–546
Konvalina P, Grausgruber H, Dang KT et al (2017) Rheological and technological quality of minor wheat species and common wheat. In: Wanyera R, Owuoche J (eds) Wheat improvement, management and utilization, 1st edn. InTechOpen, Rijeka, pp 255–273
Kundu M, Khatkar BS, Gulia N (2017) Assessment of chapatti quality of wheat varieties based on physicochemical, rheological and sensory traits. Food Chem 226:95–101
Lachman J, Orsák M, Pivec V et al (2012) Antioxidant activity of grain of einkorn (Triticum monococcum L.), emmer (Triticum dicoccum Schuebl [Schrank]) and spring wheat (Triticum aestivum L.) varieties. Plant Soil Environ 58:15–21
Lachman J, Hejtmánková K, Kotíková Z (2013) Tocols and carotenoids of einkorn, emmer and spring wheat varieties: selection for breeding and production. J Cereal Sci 57:207–214
Lampi AM, Nurmi T, Ollilainen V et al (2008) Tocopherols and tocotrienols in wheat genotypes in the HEALTHGRAIN diversity screen. J Agric Food Chem 56:9716–9721
Landberg R, Kamal-Eldin A, Salmenkallio-Marttila M et al (2008) Localization of alkylresorcinols in wheat, rye and barley kernels. J Cereal Sci 48:401–406
Laroche C, Michaud P (2007) New developments and prospective for β (1, 3) glucans. Recent Pat Biotechnol 1:59–73
Li G, He Z, Lillemo M et al (2008) Molecular characterization of allelic variations at Pina and Pinb loci in Shandong wheat landraces, historical and current cultivars. J Cereal Sci 47:510–517
Li S, Morris CF, Bettge AD (2009) Genotype and environment variation for arabinoxylans in hard winter and spring wheats of the U.S. Pacific Northwest. Cereal Chem 86:88–95
Lia A, Anderson H, Mekki N et al (1997) Postprandial lipidemia in relation to sterol and fat excretion in ileostomy subjects given oat bran and wheat test meals. Am J Clin Nutr 66:357–365
Løje H, Møller B, Laustsen AM et al (2003) Chemical composition, functional propreties and sensory profiling of einkorn (Triticum monococcum L.). J Cereal Sci 37:231–240
Lott JNA, Spitzer E (1980) X-ray analysis studies of elements stored in protein body globid crystals of Triticum grains. Plant Physiol 66:494–499
Lyons G, Ortiz-Monasterio I, Stangoulis J, Graham R (2005) Selenium concentration in wheat grain: is there sufficient genotypic variation to use in breeding? Plant Soil 269:269–380
Manickavelu A, Hattori T, Yamaoka S et al (2017) Genetic nature of elemental contents in wheat grains and its genomic prediction: toward the effective use of wheat landraces from Afghanistan. PLoS One 12:0169416
Maningat CC, Seib PA, Bassi SD et al (2009) Wheat starch: production, properties, modification and uses. In: BeMiller J, Whistler R (eds) Starch: chemistry and technology, a volume in food science and technology, 3rd edn. Elsevier, New York, pp 441–510
Marchylo BA, Dexter JE (2001) Pasta production. In: Owens G (ed) Cereals processing technology, 1st edn. Woodhead Publishing, pp 109–130
Marconi M, Cubadda R (2005) Emmer wheat. In: Abdel-Aal ESM, Wood PJ (eds) Speciality grains for food and feed, 1st edn. American Association of Cereal Chemists, St. Paul, pp 63–108
Marconi E, Carcea M, Graziano M et al (1999) Kernel properties and pasta-making quality of five European spelt wheat (Triticum spelta L.) cultivars. Cereal Chem 76:25–29
Marotti I, Bregola V, Aloisio I et al (2012) Prebiotic effect of soluble fibres from modern and old durum-type wheat varieties on Lactobacillus and Bifidobacterium strains. J Sci Food Agric 92:2133–2140
McKevith B (2004) Nutritional aspects of cereals. Nutr Bull 29:111–142
Mefleh M, Conte P, Fadda C et al (2019) From seed to bread: variation in quality in a set of old durum wheat cultivars. J Sci Food Agric. https://doi.org/10.1002/jsfa.9745
Melash AA, Mengistu DK, Aberra DA, Tsegay A (2019) The influence of seeding rate and micronutrients foliar application on grain yield and quality traits and micronutrients of durum wheat. J Cereal Sci 85:221–227
Mendis M, Simsek S (2014) Arabinoxylans and human health. Food Hydrocoll 42:239–243
Meuser F, Suckow P (1986) Nonstarch polysaccharides. In: Blanshard JMV, Frazier PJ, Galliard T (eds) Chemistry and physics of baking materials, processes, and product, 1st edn. The Royal Society of Chemistry, London, pp 42–61
Migliorini P, Spagnolo S, Torri L et al (2016) Agronomic and quality characteristics of old, modern and mixture wheat varieties and landraces for organic bread chain in diverse environments of northern Italy. Eur J Agron 79:131–141
Miskelly D, Batey IL, Suter DAI (2010) Processing wheat to optimize product quality. In: Wrigley CW, Batey IL (eds) Cereal grains assessing and managing quality, 1st edn. Woodhead Publishing, Duxford, pp 431–457
Morris CF, Bhave M (2008) Reconciliation of D-genome puroindoline allele designations with current DNA sequence data. J Cereal Sci 48:277–287
Motzo R, Fois S, Giunta F (2004) Relationship between grain yield and quality of durum wheats from different eras of breeding. Euphytica 140:147–154
Mujeeb-Kazi A, Kazi AG, Dundas I et al (2013) Genetic diversity for wheat improvement as a conduit to food security. In: Sparks DL (ed) Advances in agronomy. Elsevier, Burlington, pp 179–258
Nachit MM, Baum M, Impiglia A (1995) Studies on some grain quality traits in durum wheat grown in Mediterranean environments. CIHEAM, Options Méditerranéennes: Série A Séminaires Méditerranéens 2:181–187
Nazco R, Villegas D, Ammar K et al (2012) Can Mediterranean durum wheat landraces contribute to improved grain quality attributes in modern cultivars? Euphytica 185:1–17
Nurmi T, Nyström L, Edelmann M et al (2008) Phytosterols in wheat genotypes in the HEALTHGRAIN diversity screen. J Agric Food Chem 56:9710–9715
Okarter N, Liu CS, Sorrells ME et al (2010) Phytochemical content and antioxidant activity of six diverse varieties of whole wheat. Food Chem 119:249–257
Ottman MJ, Doerge TA, Martin EC (2000) Durum grain quality as affected by nitrogen fertilization near anthesis and irrigation during grain fill. Agron J 92:1035–1041
Ozkan H, Brandolini A, Torun A et al (2007) Natural variation and identification of microelements content in seeds of einkorn wheat (Triticum monococcum). In: Buck HT, Nisi JE, Salomón N (eds) Wheat production in stressed environments, 1st edn. Springer, Dordrecht, pp 455–462
Packer L (1995) Nutrition and biochemistry of the lipophilic antioxidants vitamin E and carotenoids. In: Ong ASH, Niki E, Packer L (eds) Nutrition, lipids, health, and disease, 1st edn. AOCS Press, Champaign, pp 8–35
Pasha I, Anjum FM, Morris CF (2010) Grain hardness: a major determinant of wheat quality. Food Sci Technol Int 16:511–522
Pechanek U, Karger A, Gröger S et al (1997) Effect of nitrogen fertilization on quantity of flour protein components, dough properties, and breadmaking quality of wheat. Cereal Chem 74:800–805
Piergiovanni AR (2013) Evaluation of genetic variation and grain quality of old bread wheat varieties introduced in north-western Italian environments. Genet Resour Crop Evol 60:325–333
Piironen V, Lindsay DG, Miettinen TA et al (2000) Plant sterols: biosynthesis, biological function and their importance to human nutrition. J Sci Food Agric 80:939–966
Piironen V, Edelmann M, Kariluoto S et al (2008) Folate in wheat genotypes in the HEALTHGRAIN diversity screen. J Agric Food Chem 56:9726–9731
Raciti CN, Doust MA, Lombardo GM et al (2003) Characterization of durum wheat Mediterranean germplasm for high and low molecular weight glutenin subunits in relation with quality. Eur J Agron 19:373–382
Rakszegi M, Batey IL, Vida GY et al (2003) Starch properties in different lines of an old Hungarian wheat variety, Bánkúti 1201. Starch 55:397–402
Rao AV, Janezic SA (1992) The role of dietary phytosterols in colon carcinogenesis. Nutr Cancer 18:43–52
Rizzello CG, Cavoski I, Turk J et al (2015) Organic cultivation of Triticum turgidum subsp. durum is reflected in the flour-sourdough fermentation-bread axis. Appl Environ Microbiol 81:3192–3204
Sanchez-Garcia M, Álvaro F, Peremarti A et al (2015) Changes in bread-making quality attributes of bread wheat varieties cultivated in Spain during the 20th century. Eur J Agron 63:79–88
Schmidt K (2007) Göbekli Tepe en eski tapınağı yapanlar/Taş çağı avcılarının gizemli anıtsal alanı (trans: Aslan R). Arkeoloji ve Sanat Yayınları, İstanbul
Schneider C (2005) Chemistry and biology of vitamin E. Mol Nutr Food Res 49:7–30
Scott J, Rébeillé F, Fletcher J (2000) Folic acid and folates: the feasibility for nutritional enhancement in plant foods. J Sci Food Agric 80:795–824
Serpen A, Gökmen V, Karagöz A et al (2008) Phytochemical quantification and total antioxidant capacities of emmer (Triticum dicoccon Schrank) and einkorn (Triticum monococcum L.) wheat landraces. J Agric Food Chem 56:7285–7292
Sezer B, Morgounov A, Ozdemir F et al (2019) Chemical composition of wheat landraces in Turkey. Paper presented at the international conference on wheat diversity & human health, İstanbul, 22–24 October 2019
Shewry RP, Hey S (2015) Do “ancient” wheat species differ from modern bread wheat in their contents of bioactive components? J Cereal Sci 65:236–243
Shewry PR, D’ovidio R, Lafiandra D et al (2009) Wheat grain proteins. In: Khan K, Shewry PR (eds) Wheat: chemistry and technology, 4th edn. American Association of Cereal Chemists, Minnesota, pp 223–298
Shewry PR, Piironen V, Lampi AM et al (2010) The HEALTHGRAIN wheat diversity screen: effects of genotype and environment on phytochemicals and dietary fiber components. J Agric Food Chem 58:9291–9298
Shewry PR, Hawkesford MJ, Piironen V et al (2013) Natural variation in grain composition of wheat and related cereals. J Agric Food Chem 61:8295–8303
Simsek S, Whitney K, Ohm JB, Mergoum M (2011) Refrigerated dough quality of hard red spring wheat: effect of genotype and environment on dough syruping and arabinoxylan production. Cereal Chem 88:445–450
Sofi BA, Wani IA, Masoodi FA et al (2013) Effect of gamma irradiation on physicochemical properties of broad bean (Vicia faba L.) starch. LWT 54:63–72
Starr G, Bredie WLP, Hansen AS (2013) Sensory profiles of cooked grains from wheat species and varieties. J Cereal Sci 57:295–303
Starr G, Petersen MA, Jespersen BM, Hansen AS (2015) Variation of volatile compounds among wheat varieties and landraces. Food Chem 174:527–537
Stehno Z, Paulickova I, Bradová J et al (2011) Evaluation of emmer wheat genetics resources aimed at dietary food production. J Life Sci 5:207–212
Swan G (2004) Findings from the latest national diet and nutrition survey. Proc Nutr Soc 63:505–512
Tiwari U, Cummins E (2009) Nutritional importance and effect of processing on tocols in cereals. Trends Food Sci Technol 20:511–520
Ülker M, Altuner F, Oral E et al (2019) Tir as a landrace and a sowing method. Paper presented at the international conference on wheat diversity & human health, İstanbul, 22–24 October 2019
Uthayakumaran S, Wrigley CW (2010) Wheat: characteristics and quality requirements. In: Wrigley CW, Batey IL (eds) Cereal grains assessing and managing quality, 1st edn. Woodhead Publishing, Duxford, pp 59–111
Vázquez D, Berger AG, Cuniberti M et al (2012) Influence of cultivar and environment on quality of Latin American wheats. J Cereal Sci 56:196–203
Velu G, Ortiz-Monasterio I, Cakmak I et al (2014) Biofortification strategies to increase grain zinc and iron concentrations in wheat. J Cereal Sci 59:365–372
Velu G, Crespo Herrera L, Guzman C et al (2019) Assessing genetic diversity to breed competitive biofortified wheat with enhanced grain Zn and Fe concentrations. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01971
Wang ZH, Li SX, Malhi S (2008) Effects of fertilization and other agronomic measures on nutritional quality of crops. J Sci Food Agric 88:7–23
Wang S, Li C, Copeland L et al (2015) Starch retrogradation: a comprehensive review. Compr Rev Food Sci Food Saf 14:568–585
Waters BM, Uauy C, Dubcovsky J, Grusak MA (2009) Wheat (Triticum aestivum) NAM proteins regulate the translocation of iron, zinc, and nitrogen compounds from vegetative tissues to grain. J Exp Bot 60:4263–4274
Weaver CM, Kannan S (2002) Phytate and mineral bioavailability. In: Reddy NR, Sathe SK (eds) Food phytates, 1st edn. CRC Press, Boca Raton, pp 211–223
Welch RM, Graham RD (2004) Breeding for micronutrients in staple food crops from a human nutrition perspective. J Exp Bot 55:353–364
White PJ, Broadley MR (2009) Biofortification of crops with seven mineral elements often lacking in human diets-iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol 182:49–84
Wieser H, Seilmeier W (1998) The influence of nitrogen fertilization on quantities and proportions of different protein types in wheat flour. J Sci Food Agric 76:49–55
Wrigley CW (2010) Cereal-grain morphology and composition. In: Wrigley CW, Batey IL (eds) Cereal grains assessing and managing quality, 1st edn. Woodhead Publishing, Duxford, pp 24–44
Wu J, Aluko RE, Corke H (2006) Partial least-squares regression study of the effects of wheat flour composition, protein and starch quality characteristics on oil content of steamed-and-fried instant noodles. J Cereal Sci 44:117–126
Yang X, Wu L, Zhu Z et al (2014) Variation and trends in dough rheological properties and flour quality in 330 Chinese wheat varieties. Crop J 2:195–200
Yilmaz VA, Brandolini A, Hidalgo A (2015) Phenolic acids and antioxidant activity of wild, feral and domesticated diploid wheats. J Cereal Sci 64:168–175
Zeng M, Morris CF, Batey IL, Wrigley CW (1997) Sources of variation for starch gelatinization, pasting, and gelation properties in wheat. Cereal Chem 74:63–71
Ziegler JU, Schweiggert RM, Würschum T et al (2016) Lipophilic antioxidants in wheat (Triticum spp.): a target for breeding new varieties for future functional cereal products. J Funct Foods 20:594–605
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kaplan Evlice, A. (2021). Nutritional and Technological Properties of Wheat Landraces. In: Zencirci, N., Baloch, F.S., Habyarimana, E., Chung, G. (eds) Wheat Landraces. Springer, Cham. https://doi.org/10.1007/978-3-030-77388-5_6
Download citation
DOI: https://doi.org/10.1007/978-3-030-77388-5_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-77387-8
Online ISBN: 978-3-030-77388-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)