Abstract
Asynchronies may appear at several points during the respiratory cycle. We can identify these asynchronies by ventilator waveform observation, through complex mathematical algorithms or using the electrical activity of the diaphragm or esophageal pressure. In routine clinical practice, waveform analysis is the most available and least invasive measure for patient-ventilator asynchronies interpretation at the bedside.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Clinical Cases in Non Invasive Ventilation: Pressure Waveform
1.1 Standard Clinical Case
A male patient, 72 years old, former smoker (90 pack-year) with a medical history of type II diabetes, arterial hypertension and chronic obstructive pulmonary disease with severe obstruction, multiple exacerbations (with hospital admissions, including one episode of invasive mechanical ventilation) and type 2 respiratory insufficiency, on supplementary oxygen and non invasive ventilation (NIV) at night, was brought to the emergency department with dyspnea at rest, cough with purulent sputum and fever (temperature of 39 °C maximum). At observation he was conscious and alert, tachycardic (120 bpm), hypertensive (140/90 mmHg) and had fever (38.5 °C). He was using his accessory muscles (intercostal, supraclavicular) with oxygen saturations of 87% under his domiciliary non invasive ventilator (PSV mode; IPAP 20; EPAP 6; respiratory backup rate 14; maximum inspiratory time 0.8–1.4 s; rise time 3; low ventilator trigger sensitivity) with adjuvant supplementary oxygen (2 L/min). At pulmonary auscultation he had crackles in the right basal hemithorax.
The exams performed at emergency department showed a respiratory acidosis (pH: 7187; PaCO2: 122.9 mmHg; PaO2: 69.3 mmHg; HCO3: 45.6 mmHg; SaO2: 87.2%), the blood tests leukocytosis and an elevated C-reactive protein and the chest radiograph showed an opacity at the base of the right lung, suggestive of pneumonia.
Under NIV, the patient was restless and anxious, due to poor patient–ventilator synchrony and the pattern on the pressure waveform was suggestive of ineffective triggering, leading to an increase in ventilator trigger sensitivity and a decrease in the maximum inspiratory time (to 0.5–1 s) with improvement in arterial blood gases (pH: 7338; PaCO2: 82.7 mmHg; PaO2: 58.2 mmHg; HCO3: 43.4 mmHg; SaO2: 86.8%). Despite that, the patient remained, somewhat, uncomfortable and, combined with the pressure waveform pattern, double triggering was detected leading to an increase of IPAP level to 22 mmHg, with achievement of patient ventilator synchrony.
In the following questions, choose the correct answer:
-
1.
One of the reasons to explain ineffective triggering in this case is:
-
(a)
High inspiratory trigger threshold
-
(b)
Low intrinsic positive end-expiratory pressure (PEEPi)
-
(c)
High respiratory drive
-
(a)
-
2.
When the patient had poor patient-ventilator synchrony and ineffective triggering was detected, we could identify in the pressure waveform:
-
(a)
Two cycles separated by a very short expiratory time
-
(b)
An increase in airway pressure near the end of the inspiratory phase
-
(c)
An airway pressure drop caused by the inspiratory effort of the patient without the delivery of pressure from the ventilator
-
(a)
-
3.
Double triggering can be identified:
-
(a)
In the pressure waveform by two mechanical cycles triggered by the patient, separated by a very short expiratory time
-
(b)
Frequently, in patients with low respiratory drive
-
(c)
Mostly in patients with a sufficient level of pressure support
-
(a)
-
4.
If auto-triggering occurred it could be because of (choose the incorrect answer):
-
(a)
Air leaks
-
(b)
Low trigger sensitivity
-
(c)
Changes of airway pressure not caused by patient effort
Answers: 1. (a); 2. (c); 3. (a); 4. (b)
-
(a)
2 Discussion
The most important aim when instituting noninvasive mechanical ventilation is to decrease patients’ work of breathing. The most effective unloading of the inspiratory muscles is obtained when the ventilator cycles are in synchrony with patient’s respiratory rhythm. Nevertheless, asynchronies may appear at several points during the respiratory cycle [1]. Patient ventilator asynchrony is a mismatch between patient and ventilator assisted breaths and the ventilator’s ability to meet the patient’s flow demand [2].
Poor patient–ventilator interaction and synchrony during non invasive ventilation (NIV) can cause discomfort, agitation, increased work of breathing and worsening of gas exchange.
We can identify these asynchronies trough ventilator waveform observation, complex mathematical algorithms or using the electrical activity of the diaphragm or esophageal pressure. The gold standard for measuring patient ventilator asynchronies is the phrenic neurogram and esophageal balloon catheter but it is less feasible in clinical practice, a source of discomfort to the patient and contributes to air leaks. The phrenic neurogram senses diaphragmatic muscle contraction (patient inspiration) through invasive sensory probes. The second measure is correlated with pleural pressure and is obtained from the esophageal balloon catheter. In routine clinical practice, visual inspection of the ventilator screen is the commonest method adopted. Waveform analysis is conducted by visually detecting particular morphological changes and is the most available and least invasive measure for patient-ventilator asynchronies interpretation at the bedside.
Clinically relevant asynchronies are present in up to 60% of patients doing noninvasive ventilation. Pressure/time and flow/time waveform analyses require a trained eye. Since asynchronies are common and complex, clinicians need to be knowledgeable regarding waveform analysis to detect ineffective patient ventilator interaction [2]. We can use the asynchrony index (AI%) to evaluate the rate of asynchrony. This is calculated by dividing the asynchronous breaths by the overall breath count. Values of AI% ≥10 are associated with worsened patients’ outcomes (for example high levels of inspiratory pressure can be associated with and increased index).
In this chapter we will learn how to interpret asynchronies through pressure waveform analysis.
We can classify patient–ventilator asynchronies as major (ineffective triggering, auto-triggering, and double triggering) and minor (premature cycling, prolonged cycling, triggering delay).
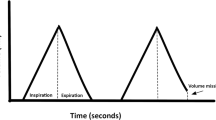
Ineffective triggering (wasted efforts) is the most common asynchrony and occurs because an inspiratory effort is not assisted by the ventilator. There are various factors that can contribute to this event, particularly low respiratory drive, low ventilator trigger sensitivity or high intrinsic positive end-expiratory pressure (PEEPi). A high incidence of ineffective efforts during long term non invasive ventilation is associated with a poorer nocturnal oxygen gas exchange [3]. Ramsay et al. used parasternal electromyography to assess types and prevalence of asynchronies during long term NIV and nearly 80% of patients had an AI% ≥10. Ineffective efforts were the most common asynchrony [4]. Lower levels of inspiratory support decrease the rate of ineffective triggering (increases patient respiratory drive). In fact, in patients with airway obstruction, reducing tidal volume and minute ventilation improves patient–ventilator synchrony because it decreases auto or PEEPi that can also contribute to ineffective triggering. Due to a high PEEPi and when pressure triggering is used, the patient cannot decrease his airway pressure below PEEPi and the inspiratory effort is ineffective. High inspiratory trigger thresholds may also result in ineffective triggering. In the presence of ineffective efforts we will have an airway pressure drop in the pressure waveform (Fig. 19.1) caused by the inspiratory effort of the patient (decreasing the airway pressure) and, a change in the expiratory flow (which tends to return to zero due to the inspiratory effort of the patient) without the delivery of pressure from the ventilator [1, 5].
Waveforms of airway pressure (Paw) and flow for each type of patient–ventilator asynchronies. Arrows highlight the asynchronous events. (From: Eugenio Garofalo, Andrea Bruni, Corrado Pelaia, Luisa Liparota, et al. (2018). Recognizing, quantifying and managing patient-ventilator asynchrony in invasive and noninvasive ventilation. Expert Review of Respiratory Medicine, 12(7):557–567)
Auto-triggering occurs when the assistance that the ventilator delivers is not related to the patient’s spontaneous effort. During mechanical ventilation triggering of the ventilator should result from inspiratory muscles contraction. However, changes in airway flow/pressure because of cardiac oscillations, water in the circuit, air leaks can be erroneously interpreted as triggering efforts. Auto-triggering, most often, occurs with low respiratory drive and frequency and when dynamic hyperinflation is absent. Zero flow can occur for some time during expiration, just before the next inspiration, making the system vulnerable to triggering from changes of airway pressure not caused by patient effort. Therefore, a greater sensitivity of the triggering system programmed on the ventilator also increases the risk of auto-triggering. Auto-triggering shows in the pressure waveform, a lack of airway pressure drop or variations in the flow waveform at the beginning of the inspiratory phase (Fig. 19.1).
Double triggering is characterized by two mechanical cycles triggered by the patient, separated by a very short expiratory time. It is more common in patients with low respiratory compliance receiving pressure support ventilation or patients with high respiratory drive. It is associated with an insufficient level of pressure support, and results in an inspiratory effort retriggering the ventilator after it has discontinued pressurization [5, 6]. We can identify this asynchrony in pressure waveform in the presence of two cycles separated by a very short expiratory time (Fig. 19.1).
Premature cycling is related to the end of the ventilator insufflation before a patient’s effort has finished (patients with low compliance) and delayed or prolonged cycling is the opposite, which means it happens when the patient’s inspiration has ceased and mechanical assistance continues (patients with higher resistance and normal or elevated lung compliance, like in patients with chronic obstructive pulmonary disease or in presence of air leaks). Premature cycling produces a decrease in airway pressure, which can be seen immediately after the end of the inspiratory phase and delayed cycling is evidenced in the pressure waveform as an increase in airway pressure near the end of the inspiratory phase (Fig. 19.1).
Finally, triggering delay corresponds to a prolonged time between patient’s respiratory effort and ventilator support initiation [5]. In pressure waveform before ventilator support there is a pressure drop caused by the inspiratory effort of the patient (Fig. 19.1).
In NIV, air leaks represent the most important contributors to asynchrony. The development of software capable of detecting and compensating for air leaks has promoted a better patient–ventilator interaction during NIV, lowering the occurrence of auto-triggering, ineffective efforts, and late cycling. It is also crucial to choose the right interface to avoid excessive air leaks. Interestingly, the study conducted by Carlucci et al. suggested that the occurrence of asynchrony does not differ between patients with obstructive or restrictive disease [7].
Key Teaching Points
-
Asynchronies may appear at several points during the respiratory cycle.
-
We can identify these asynchronies by ventilator waveform observation.
-
We can classify patient–ventilator asynchronies as major (ineffective triggering, auto-triggering and double triggering) and minor (premature cycling, prolonged cycling, triggering delay).
-
Ineffective triggering is the most common asynchrony and occurs because an inspiratory effort is not assisted by the ventilator.
-
In NIV, air leaks represent the most important contributors to asynchrony.
Abbreviations
- AI%:
-
Asynchrony index
- COPD:
-
Chronic obstructive pulmonary disease
- NIV:
-
Non invasive ventilation
- PEEPi:
-
Intrinsic positive end-expiratory pressure
References
Achoura L, Letelliera C, Cuvelierb A, Vérinc E, et al. Asynchrony and cyclic variability in pressure support noninvasive ventilation. Comput Biol Med. 2007;37:1308–20.
Mellott KG, Grap MJ, Munro CL, Sessler CN, et al. Patient ventilator asynchrony in critically ill adults: frequency and types. Heart Lung. 2014;43(3):231–43.
Fanfulla F, Delmastro M, Berardinelli A, D’Artavilla N, et al. Effects of different ventilator settings on sleep and inspiratory effort in patients with neuromuscular disease. Am J Respir Crit Care Med. 2005;172:620–4.
Ramsaym M, Mandal S, Suh E-S, Steier J. Parasternal electromyography to determine the relationship between patient-ventilator asynchrony and nocturnal gas exchange during home mechanical ventilation set-up. Thorax. 2015;70:946–52.
Garofalo E, Bruni A, Pelaia C, Liparota L, et al. Recognizing, quantifying and managing patient-ventilator asynchrony in invasive and noninvasive ventilation. Expert Rev Respir Med. 2018;12(7):557–67.
Vignaux L, Vargas F, Roeseler J, Tassaux D, et al. Patient–ventilator asynchrony during non-invasive ventilation for acute respiratory failure: a multicenter study. Intensive Care Med. 2009;35:840–6.
Carlucci A, Pisani L, Ceriana P, Malovini A, et al. Patient-ventilator asynchronies: may the respiratory mechanics play a role? Crit Care. 2013;17:R54.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
1 Electronic Supplementary Material
Basic physiology breathing patterns: tachipnea (MOV 31128 kb)
Basic physiology breathing patterns: apnea—bradipnea (MOV 10257 kb)
Pressure-flow volume waveforms relationships_1 (MOV 9994 kb)
Pressure-flow volume waveforms relationships_2 (MOV 4530 kb)
Obstructive patterns: COPD Exacerbation_Auto PEEP (MOV 15654 kb)
Expiratory flow asynchrony waveform_1 (MOV 115217 kb)
Expiratory flow asynchrony waveform_2 (MOV 34291 kb)
Expiratory flow asynchrony waveform_3 (MOV 72938 kb)
Expiratory flow asynchrony waveform_6 (MOV 28864 kb)
Expiratory flow asynchrony waveform_7 (MOV 14034 kb)
Expiratory flow asynchrony waveform_8 (MOV 27398 kb)
Expiratory flow asynchrony waveform_9 (MOV 16698 kb)
Expiratory flow asynchrony waveform_10 (MOV 35518 kb)
Expiratory flow asynchrony waveform_11 (MOV 14481 kb)
Expiratory flow asynchrony waveform_12 (MOV 34772 kb)
Expiratory flow asynchrony waveform_13 (MOV 30802 kb)
Expiratory flow asynchrony waveform_14 (MP4 2216 kb)
Expiratory flow asynchrony waveform_15 (MOV 12775 kb)
Auto cycling. Asynchrony (MOV 45556 kb)
Inspiratory expiratory time relationships. Asynchrony (MOV 66269 kb)
Leakages effects. Asynchrony severe patterns_1 (MOV 34291 kb)
Leakages effects. Asynchrony severe patterns_2 (MOV 11903 kb)
Leakages effects. Asynchrony severe patterns_3 (MOV 72938 kb)
Leakages effects. Asynchrony severe patterns_4 (MOV 115217 kb)
Leakages effects. Asynchrony leak effects_1 (MP4 2948 kb)
Leakages effects. Asynchrony leak effects_2 (MOV 66269 kb)
Leakages effects. Asynchrony leak effects_3 (MP4 2948 kb)
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Vieira, A.C., Silveira, A.M. (2022). Clinical Cases in Non Invasive Ventilation: Pressure Waveform. In: Esquinas, A.M. (eds) Teaching Pearls in Noninvasive Mechanical Ventilation. Springer, Cham. https://doi.org/10.1007/978-3-030-71298-3_19
Download citation
DOI: https://doi.org/10.1007/978-3-030-71298-3_19
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-71297-6
Online ISBN: 978-3-030-71298-3
eBook Packages: MedicineMedicine (R0)