Abstract
Background
Functional dyspepsia is the main cause of upper abdominal discomfort affecting 5–10% of the world population. Despite various therapeutic approaches, up to 50% of patients with functional dyspepsia seek alternative treatments. In the present study we evaluated the effect of curcumin supplementation along with famotidine therapy on severity of functional dyspepsia. A total of 75 patients with functional dyspepsia according to Rome III criteria were allocated into intervention (N = 39) or control (N = 36) groups. The intervention group was treated with a combination of 500 mg curcumin and 40 mg famotidine daily for 1 month. The control group received placebo and 40 mg famotidine. Severity of dyspepsia symptoms was determined using the Hong Kong questionnaire at baseline, after the 1 month treatment and after a 1 month follow-up. The presence of H. pylori antigens in the stool samples was also investigated in all subjects. No significant difference was observed between intervention and control groups in biochemical indices, severity of dyspepsia and rate of H. pylori infection. A significant decrease was observed in severity of dyspepsia (p < 0.001) and rate of H. pylori infection (p = 0.004) immediately after the treatment and follow-up in the curcumin intervention group. This study indicated that curcumin therapy could be a favorable supplementation in the symptom management of functional dyspepsia. Moreover, curcumin could help efficient eradication of H. pylori in these patients.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
10.1 Introduction
Functional dyspepsia is the main cause of upper gastrointestinal tract discomfort manifested by abdominal fullness, heartburn, nausea, belching, acid taste and upper abdominal pain [1, 2]. It is a relapsing and remitting condition that affects 5–11% of population worldwide [1]. It is estimated that the financial burden of drugs in dyspepsia is about £450 million in the United Kingdom [3]. Based on Rome III criteria, functional dyspepsia is defined as chronic presence of early satiation, postprandial fullness, epigastric pain or epigastric burning, in endoscopy-negative patients [2, 4]. Genetic background and psychological distress are the most important factors related to this condition that evoke an inflammatory response and further clinical symptoms [1]. Some proposed treatments for functional dyspepsia are H. pylori eradication [5], acid-suppression [3], and administration of pro-kinetic agents [3], antidepressants [6,7,8,9] and herbal supplements [10, 11]. Due to increasing dissatisfaction with conventional therapeutic approaches in functional dyspepsia, almost 50% of patients seek alternative treatments [12]. Hence, more effective and yet safer agents are required to treat functional dyspepsia.
Curcumin is a yellow pigment derived from turmeric. A wide range of its pharmacological effects have so far been been reported for this dietary safe phytochemical [13,14,15,16,17,18,19,20,21]. Furthermore, clinical studies have indicated that curcumin supplementation could positively affect symptoms related to the gastrointestinal tract including epigastric pain, post-prandial fullness, bloating, belching and nausea. However, no improvement in H.pylori eradication has been shown thus far [22]. Similarly, curcumin treatment could efficiently improve some gastrointestinal diseases such as peptic ulcer, Barrett’s esophagus, non-alcoholic fatty liver disease, irritable bowel syndrome (IBS), pancreatitis, ulcerative colitis and gastrointestinal cancers [21,22,23,24,25,26,27,28,29,30,31,32,33].
Evidence of curcumin efficacy on treatment of functional dyspepsia is scarce. Here we aim to investigate the effect of curcumin on improving of functional dyspepsia according to Hong Kong score and H. pylori eradication rate.
10.2 Materials and Methods
10.2.1 Subjects
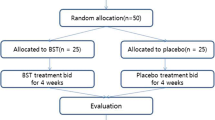
Adult subjects were selected from the outpatient unit of Gastroenterology clinic of Baqiyatallah Hospital (Tehran, Iran) with symptoms of dyspepsia and qualified as candidates for upper gastrointestinal endoscopy. Eligible patients diagnosed with functional dyspepsia according to Rome III criteria [2] were enrolled in the study. Gastrointestinal structural abnormalities were ruled out by upper gastrointestinal endoscopy. Exclusion criteria were liver and renal dysfunction, chronic disease, previous anti-helicobacter therapy, malignancies, deterioration of dyspepsia and other related complications during the study period. The study design was approved by the institutional ethics committee and was conducted as per the Helsinki declaration . Written informed consent was taken from all of participants. The Hong Kong index of dyspepsia severity was recorded prior to intervention, at the end of the study and after a follow-up for 1 month. Blood samples were obtained in order to measure the fasting blood sugar (FBS), hemoglobin (Hb), triglycerides (TG), cholesterol (Chol), low density lipoprotein (LDL), high density lipoprotein (HDL), aspartate aminotransferase (AST), alanine aminotransferase (ALT) and alkaline phosphatase (ALP). Stool samples were collected for assessment of H. pylori antigen prior to treatment, after the 1 month treatment and 1 month of follow-up periods. The patient flow chart is shown in Fig. 10.1.
10.2.2 Study Design
This study was performed as a randomized triple-blind placebo-controlled trial. Patients were randomly allocated into intervention or control groups. The intervention group received 40 mg famotidine and one capsule containing 500 mg curcuminoids (C3 Complex®, Sami Labs Ltd., Bangalore, India) after lunch for 30 days (n = 39). For the control group (n = 36), 40 mg famotidine and a placebo capsule were administered daily for the same period. The curcuminoid and placebo capsules were identical in size and shape. Each curcuminoid capsule also contained piperine (5 mg; Bioperine®, Sami Labs Ltd.), added for the purpose of enhancing bioavailability. The trial protocol was registered in the Iranian Registry of Clinical Trials (IRCT20080901001165N45).
10.2.3 Hong Kong Dyspepsia Index
Symptom severity of functional dyspepsia was determined using the Hong Kong dyspepsia index. The questionnaire consists of 12 items including stomach pain, upper abdominal dull pain, stomach pain before meals, stomach pain during anxiety, heartburn, upper abdominal bloating, vomiting, nausea, belching, acid regurgitation, feeling of acidity in the stomach and loss of appetite. Each item was scored from 1 (asymptomatic), 2 (mild symptoms that can be easily ignored), 3 (awareness of symptoms but easily tolerated), 4 (severe symptoms sufficient to interfere with normal daily activities) and 5 (incapacitating symptoms causing inability to perform daily activities and/or require days off work). Subsequently, all the above-mentioned scores were added together (from 12 to 60) to calculate the severity of functional dyspepsia [34].
10.2.4 Statistical Analysis
Data was analyzed using SPSS version 18 software. Data were reported as number and percent or median ± standard deviation. For analyzing categorical variables, the Chi-square test was used. Comparisons between groups were performed using independent Student’s T-tests. For comparison of quantitative variables prior to treatment, immediately after and 30 days later, repeated measures analysis of variance (ANOVA) was used. P-values <0.05 were considered significant.
10.3 Results
Total of 75 eligible subjects completed the study as shown in the patient flow diagram (Fig. 10.1). Baseline characteristics of participants are indicated in Table 10.1. Duration of functional dyspepsia as well as endoscopic findings, were not significantly different in the study groups. As shown in Table 10.2, none of the laboratory indices were different between two groups before or after intervention. The rate of H. pylori infection was decreased significantly in the intervention group (p = 0.004) in comparison to placebo, whereas no significant change in H. pylori was observed (p = 0.126) (Table 10.3). According to the Hong Kong questionnaire, dyspepsia severity index was decreased significantly in both groups (Table 10.4).
Comparison between different items of Hong Kong questionnaire is summarized in Fig. 10.2. Abdominal pain, belching and acid regurgitation were decreased in both the intervention (p < 0.001 for all three symptom scores) and control (p = 0.033, <0.001 and 0.022, for the respective symptom scores) groups. Upper abdominal dull pain (p < 0.001), stomach pain before meals (p < 0.001), stomach pain when anxious (p < 0.001), heartburn (p < 0.001), upper abdominal bloating (p < 0.001), nausea (p = 0.033) and feeling of acidity in stomach (p < 0.001) were decreased significantly in the curcumin group. Vomiting and loss of appetite were the only symptoms that did not change after treatment in both groups.
Comparison between different items of hong kong questionarre in case and control group (H1: abdominal pain, H2: upper abdominal dull pain, H3: stomach pain before meals, H4: stomach pain when anxious, H5: heartburn, H6: upper abdominal bloating, H7: vomiting, H8: nausea, H9: belching, H10: acid regurgitation, H11: feeling of acidity in stomach, H12: loss of appetite)
10.4 Discussion
This study was designed to evaluate the efficacy of curcumin supplementation on improving the severity of symptoms in patients with functional dyspepsia. This study indicated that 500 mg of curcumin as adjunct therapy per day could improve the severity of functional dyspepsia according to Hong Kong score over a 30 day period and after 30 days of follow-up. Curcumin administration could efficiently ameliorate upper abdominal dull pain, stomach pain before meals, stomach pain when anxious, heartburn, upper abdominal bloating, nausea and feeling of acidity in the stomach. Furthermore, the current study indicated that curcumin administration could efficiently eradicate the H. pylori infection rate.
Curcumin is a natural polyphenol with anti-inflammatory properties comparable with some steroidal and non-steroidal compounds. It modulates inflammation through inhibition of cyclooxygenase-2 (COX-2), lipoxygenase (LOX), inducible nitric oxide synthase (iNOS), production of cytokines such as interleukin (IL)-6, IL-8, interferon-gamma (IFN-γ) and tumor necrosis factor-alpha (TNF-α), and through inactivation of transcription factors like nuclear factor kappa B (NF-κB), and activator protein 1 (AP-1) [35]. Experimental studies have supported the concept of curcumin effectiveness in H. pylori eradication [36, 37]. H. pylori adheres to the gastric epithelium and secretes virulence factors which, in turn, activate the pro-inflammatory pathways causing gastric mucosal damage. Curcumin supplementation could significantly downregulate NF-κB and matrix metallopeptidase (MMP)-3 and MMP-9 activities which play roles in H. pylori infection [38,39,40]. Furthermore, an experimental study indicated that curcumin could suppress gastric inflammation caused by H. pylori-infection in mouse mucosa [31]. A comprehensive review by Sarkar et al. introduced curcumin as a potential treatment for H. pylori and its associated diseases [35]. On the contrary, some studies have found inconsistent results. Di Mario et al. studied 25 H. pylori-positive subjects with functional dyspepsia. The patients were treated with a combination of pantoprazol, N-acetylcystein, lactoferrin and 30 mg curcumin two times a day for 7 days. These therapies were not effective for H. pylori eradication [22]. In another study, the efficacy of curcumin was evaluated on H. pylori eradication in patients with H. pylori-positive peptic ulcers through a parallel-group design. Subjects were treated with 500 mg of curcumin daily plus a standard H. pylori medication (clarithromycin, amoxicillin, pantoprazole) for four weeks. In contrast with the current study, no significant effect was observed for H. pylori eradication [23]. These inconsistencies may be due to differences in curcumin dosage, duration of intervention, combination therapies and comorbidities across the studies. In addition, the source of curcumin also plays a role in its antibacterial activity. As shown by Vetvica et al. only some curcumin preparations have shown a good activity against H. pylori infection [40].
In contrast to the conflicting results related to the effects of curcumin on the rate of H. pylori infection, to our knowledge all available studies have found that curcumin attenuates the symptoms of dyspepsia. Di Mario et al. reported that curcumin decreased the overall severity of dyspepsia and some symptoms of dyspepsia including stomach pain, post-prandial fullness, bloating, belching and nausea up to 2 months after intervention [22]. Khonche et al. also reported significant alleviation of symptoms including upper abdominal dull pain, stomach pain before meals and belching following curcuminoid supplementation [23].
10.5 Conclusions
The findings of this study revealed that adding curcumin as an adjunct therapy not only improves clinical symptoms of dyspepsia but also eradicates the rate of H. pylori infection. Considering the good safety profile of curcumin and its pleiotropic actions, it could be used as an efficacious agent in functional dyspepsia. However, additional larger and long-term investigations are needed to confirm these promising results and the potential role of adjunctive curcumin therapy in the management of functional dyspepsia. Finally, it seems that the presence of H. pylori infection is a confounding factor in functional dyspepsia and the efficacy of curcumin on H. pylori eradication was not fully established. The impact of the presence H. pylori infection on the efficacy of curcumin in ameliorating the symptoms of functional dyspepsia needs to be further scrutinized.
References
Talley NJ, Ford AC (2015) Functional dyspepsia. N Engl J Med 373(19):1853–1863
Imthon AK, Moeller ME, Drewes AM, Juel J, Aziz Q (2015) Functional gastroduodenal disorders. Hamdan Med J 212(2374):1–11
Moayyedi P, Soo S, Deeks J, Delaney B, Innes M, Forman D (2006) Pharmacological interventions for non-ulcer dyspepsia. Cochrane Database Syst Rev 4:CD001960. https://doi.org/10.1002/14651858.CD001960.pub3
Drossman DA, Dumitrascu DL (2006) Rome III: new standard for functional gastrointestinal disorders. J Gastrointestin Liver Dis 15(3):237–241
Moayyedi P, Soo S, Deeks J, Delaney B, Harris A, Innes M et al (2005) Eradication of Helicobacter pylori for non-ulcer dyspepsia. Cochrane Database Syst Rev 1:CD002096. https://doi.org/10.1002/14651858.CD002096.pub2
Van Kerkhoven LA, Laheij RJ, Aparicio N, De Boer WA, Van Den Hazel S, Tan AC et al (2008) Effect of the antidepressant venlafaxine in functional dyspepsia: a randomized, double-blind, placebo-controlled trial. Clin Gastroenterol Hepatol 6(7):746–752
Ly HG, Carbone F, Holvoet L, Bisschops R, Caenepeel P, Arts J et al (2013) 161 mirtazapine improves early satiation, nutrient intake, weight recovery and quality of life in functional dyspepsia with weight loss: a double-blind, randomized, Placebo-Controlled Pilot Study. American Gastroenterological Association | American ...www.gastro.org
The American Gastroenterological Association (AGA) abstract. Gastroenterology 144(5):S–37
Talley NJ, Locke GR, Saito YA, Almazar AE, Bouras EP, Howden CW et al (2015) Effect of amitriptyline and escitalopram on functional dyspepsia: a multicenter, randomized controlled study. Gastroenterology 149(2):340–349.e2
Tan VP, Cheung TK, Wong WM, Pang R, Wong BC (2012) Treatment of functional dyspepsia with sertraline: a double-blind randomized placebo-controlled pilot study. World J Gastroenterol 18(42):6127–6133
Bortolotti M, Coccia G, Grossi G, Miglioli M (2002) The treatment of functional dyspepsia with red pepper. Aliment Pharmacol Ther 16(6):1075–1082
Pilichiewicz AN, Horowitz M, Russo A, Maddox AF, Jones KL, Schemann M et al (2007) Effects of Iberogast on proximal gastric volume, antropyloroduodenal motility and gastric emptying in healthy men. Am J Gastroenterol 102(6):1276–1283
Lahner E, Bellentani S, Bastiani RD, Tosetti C, Cicala M, Esposito G et al (2013) A survey of pharmacological and nonpharmacological treatment of functional gastrointestinal disorders. United European Gastroenterol J 1(5):385–393
Mollazadeh H, Cicero AFG, Blesso CN, Pirro M, Majeed M et al (2019) Immune modulation by curcumin: the role of interleukin-10. Crit Rev Food Sci Nutr 59(1):89–101
Soleimani V, Sahebkar A, Hosseinzadeh H (2018) Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances: Review. Phytother Res 32(6):985–995
Teymouri M, Pirro M, Johnston TP, Sahebkar A (2017) Curcumin as a multifaceted compound against human papilloma virus infection and cervical cancers: a review of chemistry, cellular, molecular, and preclinical features. Biofactors 43(3):331–346
Momtazi AA, Derosa G, Maffioli P, Banach M, Sahebkar A (2016) Role of microRNAs in the therapeutic effects of curcumin in non-cancer diseases. Mol Diagn Ther 20(4):335–345
Ghandadi M, Sahebkar A (2017) Curcumin: An effective inhibitor of interleukin-6. Curr Pharm Des 23(6):921–931
Panahi Y, Khalili N, Sahebi E, Namazi S, Simental-Mendía LE, Majeed M, Sahebkar A. Effects of Curcuminoids Plus Piperine on Glycemic, Hepatic and Inflammatory Biomarkers in Patients with Type 2 Diabetes Mellitus: A Randomized Double-Blind Placebo-Controlled Trial. Drug Res (Stuttg). 2018 Jul;68(7):403-409. https://doi.org/10.1055/s-0044-101752.
Panahi Y, Ahmadi Y, Teymouri M, Johnston TP, Sahebkar A (2018) Curcumin as a potential candidate for treating hyperlipidemia: a review of cellular and metabolic mechanisms. J Cell Physiol 233(1):141−152
Panahi Y, Khalili N, Sahebi E, Namazi S, Simental-Mendía LE, Majeed M, et al (2018) Effects of curcuminoids plus piperine on glycemic, hepatic and inflammatory biomarkers in patients with type 2 diabetes mellitus: a randomized double-blind placebo-controlled trial. Drug Res (Stuttg) 68(7):403−409
Di Mario F, Cavallaro LG, Nouvenne A, Stefani N, Cavestro GM, Iori V et al (2007) A curcumin-based 1-week triple therapy for eradication of Helicobacter pylori infection: something to learn from failure? Helicobacter 12(3):238–243
Khonche A, Biglarian O, Panahi Y, Valizadegan G, Soflaei S, Ghamarchehreh M et al (2016) Adjunctive therapy with curcumin for peptic ulcer: a randomized controlled trial. Drug Res 66(08):444–448
Chemnitzer O, Götzel K, Maurer L, Dietrich A, Eichfeld U, Lyros O et al (2017) Response to TNF-α is increasing along with the progression in Barrett’s esophagus. Dig Dis Sci 62(12):3391–3401
Czekaj R, Majka J, Ptak-Belowska A, Szlachcic A, Targosz A, Magierowska K et al (2016) Role of curcumin in protection of gastric mucosa against stress-induced gastric mucosal damage. Involvement of hypoacidity, vasoactive mediators and sensory neuropeptides. J Physiol Pharmacol 67(2):261–275
Haider S, Naqvi F, Tabassum S, Saleem S, Batool Z, Sadir S et al (2013) Preventive effects of curcumin against drug-and starvation-induced gastric erosions in rats. Sci Pharm 81(2):549–558
He P, Zhou R, Hu G, Liu Z, Jin Y, Yang G et al (2015) Curcumin-induced histone acetylation inhibition improves stress-induced gastric ulcer disease in rats. Mol Med Rep 11(3):1911–1916
Kerdsakundee N, Mahattanadul S, Wiwattanapatapee R (2015) Development and evaluation of gastroretentive raft forming systems incorporating curcumin-Eudragit® EPO solid dispersions for gastric ulcer treatment. Eur J Pharm Biopharm 94:513–520
Liang Z, Wu R, Xie W, Geng H, Zhao L, Xie C et al (2015) Curcumin suppresses MAPK pathways to reverse tobacco smoke-induced gastric epithelial–mesenchymal transition in mice. Phytother Res 29(10):1665–1671
Martin RC, Locatelli E, Li Y, Zhang W, Li S, Monaco I et al (2015) Gold nanorods and curcumin-loaded nanomicelles for efficient in vivo photothermal therapy of Barrett’s esophagus. Nanomedicine 10(11):1723–1733
Panahi Y, Ghanei M, Hajhashemi A, Sahebkar A (2016) Effects of curcuminoids-piperine combination on systemic oxidative stress, clinical symptoms and quality of life in subjects with chronic pulmonary complications due to sulfur mustard: a randomized controlled trial. J Diet Suppl 13(1):93–105
Santos A, Lopes T, Oleastro M, Gato I, Floch P, Benejat L et al (2015) Curcumin inhibits gastric inflammation induced by Helicobacter pylori infection in a mouse model. Nutrients 7(1):306–320
Panahi Y, Kianpour P, Mohtashami R, Soflaei SS, Sahebkar A (2019) Efficacy of phospholipidated curcumin in nonalcoholic fatty liver disease: a clinical study. J Asian Nat Prod Res 21(8):798–805
Hu WH, Lam KF, Wong YH, Lam CL, WM HU, Lai KC et al (2002) The Hong Kong index of dyspepsia: a validated symptom severity questionnaire for patients with dyspepsia. J Gastroenterol Hepatol 17(5):545–551
Sarkar A, De R, Mukhopadhyay AK (2016) Curcumin as a potential therapeutic candidate for Helicobacter pylori associated diseases. World J Gastroenterol 22(9):2736–2748
De R, Kundu P, Swarnakar S, Ramamurthy T, Chowdhury A, Nair GB et al (2009) Antimicrobial activity of curcumin against Helicobacter pylori isolates from India and during infections in mice. Antimicrob Agents Chemother 53(4):1592–1597
Sintara K, Thong-Ngam D, Patumraj S, Klaikeaw N, Chatsuwan T (2010) Curcumin suppresses gastric NF-kappaB activation and macromolecular leakage in Helicobacter pylori-infected rats. World J Gastroenterol 16(32):4039–4046
Foryst-Ludwig A, Neumann M, Schneider-Brachert W, Naumann M (2004) Curcumin blocks NF-κB and the motogenic response in Helicobacter pylori-infected epithelial cells. Biochem Biophys Res Commun 316(4):1065–1072
Kundu P, De R, Pal I, Mukhopadhyay AK, Saha DR, Swarnakar S (2011) Curcumin alleviates matrix metalloproteinase-3 and-9 activities during eradication of Helicobacter pylori infection in cultured cells and mice. PLoS One 6(1):e16306. https://doi.org/10.1371/journal.pone.0016306
Vetvicka V, Vetvickova J, Fernandez-Botran R (2016) Effects of curcumin on Helicobacter pylori infection. Ann Transl Med 4(24):479. https://doi.org/10.21037/atm.2016.12.52
Conflict of Interest
Muhammed Majeed is the founder of Sabinsa Corp. and Sami Labs Ltd. The other authors declare no competing interests.
Funding
This study was supported by the Research Council at the Baqiyatallah University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Panahi, Y. et al. (2021). Effect of Curcumin on Severity of Functional Dyspepsia: a Triple Blinded Clinical Trial. In: Barreto, G.E., Sahebkar, A. (eds) Pharmacological Properties of Plant-Derived Natural Products and Implications for Human Health. Advances in Experimental Medicine and Biology, vol 1308. Springer, Cham. https://doi.org/10.1007/978-3-030-64872-5_10
Download citation
DOI: https://doi.org/10.1007/978-3-030-64872-5_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-64871-8
Online ISBN: 978-3-030-64872-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)