Abstract
Glucocorticoids (GC) are an integral component of multi-agent therapy regimens for a wide variety of lymphoid malignancies due to their potential effects to induce apoptosis in cells of the lymphoid lineage. Despite their clinical utility, de novo and acquired resistance to GC is a significant clinical problem that contributes to inferior outcomes for many of these diseases. This review summarizes what is currently known about mechanisms of GC resistance in lymphoid malignancies, with a particular focus on novel therapeutic strategies currently in preclinical or clinical development that are rationally-designed to overcome GC resistance and improve clinical outcomes.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
For decades, glucocorticoids (GCs) have been a key component of therapy for the treatment of lymphoid malignancies and are widely used in both frontline and salvage therapy regimens [1, 2]. In many of these cancers, the response to GC therapy is a strong prognostic indicator that is related to both overall and event-free survival (EFS) rates [1, 3, 4]. In particular, patients with acute lymphoblastic leukemia (ALL) treated on Berlin-Frankfurt-Munster (BFM) protocols can be classified as having a prednisone good response (PGR) or a prednisone poor response (PPR), defined based on the response to an upfront 1 week window of monotherapy consisting of the GC prednisone. In early ALL-BFM protocols, patients with a PPR had significantly inferior outcomes relative to patients with a PGR [1]. These data indicate that therapeutic strategies to overcome GC resistance may significantly improve patient outcomes. The objective of this review is to highlight key concepts regarding GC resistance in lymphoid malignancies, with a specific focus on therapeutic strategies designed to overcome GC resistance.
Mechanism of Glucocorticoid Action
GCs are a class of steroid hormones that bind to the GC receptor (GR). In the absence of endogenous or exogenous GC ligand, GR is largely retained in the cytoplasm through its association with a variety of molecular chaperone proteins, including HSP70 and HSP90 [5]. Upon ligand binding, GR undergoes a conformational change that promotes translocation of the GC-GR complex to the nucleus, where it associates with DNA sequences known as GC response elements (GREs) . These GREs function as enhancer elements to modulate the activity of associated gene promoters, which in turn mediate the activation or repression of target gene expression (Fig. 1.1) [6]. These effects of GCs are highly tissue-specific due to differences in GRE binding patterns and transcriptional activities in different cell types. Importantly, while GCs exert pro-survival effects in many tissues, they potently induce cell death in cells of the lymphoid lineage [7], underlying their importance in the treatment of lymphoid malignancies.
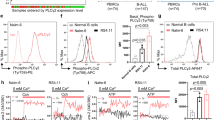
Mechanisms of GC Action. GCs bind to a cytoplasmic GR (1), which induces translocation of the GC/GR complex to the nucleus (2). This complex binds to GREs to induce or repress transcription (3). In cells of the lymphoid lineage, this transcriptional activity alters the expression of components of the intrinsic apoptotic pathway, resulting in apoptosis (4)
While there is little overall consensus regarding the specific components of the GR-associated transcriptome that mediate the effects of GCs on lymphoid cells, many groups have demonstrated that activation of the intrinsic apoptotic pathway is required for GC-induced cell death [7]. Consistent with this idea, the concept of a “BCL2 rheostat ” has been proposed whereby modulation of both the pro-and anti-apoptotic components of the intrinsic apoptotic pathway results in an altered apoptotic threshold that leads to cell death. Through an analysis of both basal and GC-induced expression of components of the intrinsic apoptotic pathway in primary ALL cells, Ploner et al. identified key expression patters that are associated with GC-induced apoptosis. Specifically, they noted potent induction of the pro-apoptotic family members BIM and BMF, and demonstrated that loss of expression of either of these proteins is sufficient to decrease GC sensitivity. Conversely, they demonstrated that overexpression of anti-apoptotic family members , including BCL2, BCL-XL, and MCL1, impairs GC-induced apoptosis, an effect that is reversed upon experimental silencing of these genes [8]. Taken together, these data suggest that coordinate modulation of both pro-and anti-apoptotic family members contribute to GC-induced apoptosis. Jing et al. further elucidated the importance of such a BCL2 rheostat through an analysis of ALL patient derived xenograft (PDXs). Using chromatin immunoprecipitation with sequencing (ChIP-seq) , this group identified a novel GR binding site within an intronic region of the BIM gene. When this region was mutated to abolish GR binding, GC-induced upregulation of BIM expression was lost and GC sensitivity was significantly decreased, providing further evidence that GR-mediated upregulation of BIM is required for GC sensitivity. Furthermore, this study elucidated a series of GR-mediated transcriptional events that lead to downregulation of BCL2 expression, and found that these events were also required for effective GC-induced apoptosis [9]. Given the importance of the intrinsic apoptotic pathway in mediating GC sensitivity in lymphoid cells, it is not surprising that while diverse mechanisms of GC resistance have been elucidated in lymphoid malignancies, these mechanisms largely converge on a failure to appropriately modulate the intrinsic apoptotic pathway.
Mechanisms of Glucocorticoid Resistance
GR Intrinsic Mechanisms of GC Resistance
NR3C1 Mutations
The GR protein , which is encoded by the NR3C1 gene , is comprised of three major functional domains: the ligand binding domain (LBD), the DNA biding domain (DBD), and the N-terminal transactivation domain (NTD), which interacts with the transcriptional machinery and the transcriptional coregulators to mediate the effects of GR on gene expression [5]. Mutations in each of these domains have been identified in the context of familial and sporadic generalized GC resistance, where they lead to complete or partial insensitivity to target tissues to both endogenous and exogenous GCs [10]. In addition to generalized GC resistance, localized GC resistance that is attributable to NR3C1 mutations has been reported in a number of disease contexts, including asthma and autoimmune diseases [11]. Given the precedent for GC resistance mediated by GR mutations, many groups have hypothesized that pre-existing mutations in the GR gene, or mutations acquired over the course of GC therapy, may contribute to de novo or acquired GC resistance in lymphoid malignancies. Much of this work has focused on CCRF-CEM cells, a cell line model of human T-cell ALL (T-ALL) that has been studied extensively in the context of GC sensitivity and resistance. Early studies involving CCRF-CEM identified considerable heterogeneity in the clonal composition of the cell line, leading to the subsequent isolation and characterization of a number of subclonal cell lines with varying degrees of GC sensitivity [12]. In an analysis of the parental CCRF-CEM cell line, a heterozygous mutation has since been identified in the LBD, and functional studies demonstrated impaired functionality of this mutant allele [13,14,15,16]. However, this same heterozygous mutation has since been identified in both GC sensitive and GC resistant subclones derived from the parental CCRF-CEM cell line, suggesting that additional events are required to confer GC resistance. Consistent with this idea, it has been shown that the GC resistant subclones derived form the GC sensitive parental cell line express this mutant allele in the absence of a wild-type allele, resulting in complete impairment of GR activity [13]. Interestingly, this LBD point mutation was identified in biopsy tissue taken after the initiation of treatment from the patient from whom CCRF-CEM cells were derived, suggesting that it was acquired in vivo and was likely selected for over the course of GC treatment [17].
Similar to CCRF-CEM cells , Jurkat cells, another human T-ALL cell line, are heterozygous for a mutation that impairs GR transcriptional activity. Unlike CCRF-CEM cells however, Jurkat cells also express low basal levels of GR and fail to induce expression of GR upon GC exposure, resulting in profound GC resistance [18].
Based on this evidence supporting a role for NR3C1 mutations as a cause of GC resistance in cultured cell lines, multiple groups have conducted studies to determine whether such mutations cause clinically relevant GC resistance in patients receiving GC therapy for the treatment of lymphoid malignancies. In an analysis of a panel of cell lines derived from paired diagnostic and relapsed samples taken from pediatric patients with ALL, Beesley et al. identified significant variability in GC sensitivity. Upon sequencing the NR3C1 gene in these cell lines, this group identified a number of polymorphisms, all of which had previously been shown to have a negligible effect on GC sensitivity [11], but no deleterious mutations. This finding led them to conclude that NR3C1 mutations are not a common mechanism of naturally-acquired GC resistance [19]. Consistent with these findings, sequencing of NR3C1 in a larger cohort of diagnostic pediatric ALL samples revealed a similar distribution of polymorphisms, but these polymorphisms failed to correlate with the clinical response to prednisone therapy and did not occur at a significantly higher rate than previously reported in the general population [20], supporting the conclusion that NR3C1 mutations are not a common cause of de novo GC resistance. However, there have been a number of reports demonstrating the presence of deleterious NR3C1 mutations that are undetectable at diagnosis but are significantly enriched at the time of disease relapse, suggesting that the acquisition of such mutations may confer acquired GC resistance [21,22,23]. Taken together, the existing data suggest that NR3C1 mutations are a relatively minor cause of GC resistance in human lymphoid malignancies, particularly at the time of diagnosis, but may be more important in the context of relapsed disease following the selective pressure of exposure to GC therapy.
Modulation of GR Expression and Function
In addition to GR mutations, expression levels of GR have been evaluated as a potential biomarker for GC sensitivity and resistance. Using large cohorts of diagnostic ALL samples, early clinical data suggested that the absolute number of GRs in lymphoblasts is positively correlated with the clinical response to GC monotherapy [24], the likelihood of disease remission [25], and with 5-year EFS rates [26]. More recently, GR expression has been shown to carry prognostic significance specifically in the context of pediatric B-cell ALL (B-ALL) harboring the ETV6/RUNX1 fusion oncogene. In these patients, deletions of NR3C1 resulting in loss of GR protein expression are associated with increased minimal residual disease (MRD) and with risk of relapse [27]. Despite these findings, other studies have failed to identify a clinically meaningful relationship between basal GR expression at diagnosis and the clinical response to GC therapy. In an analysis of GR protein expression in diagnostic samples taken from patients treated on ALL-BFM protocols, there was no significant difference in GR expression between PPR and PGR patient groups [28], suggesting that basal GR expression may be an unsuitable biomarker for predicting GC sensitivity. However, it has been shown that in lymphoid cells, exposure to GCs results in autoinduction of GR expression mediated by a direct transcriptional effect of GR [29], and multiple studies have demonstrated that expression levels of GR after autoinduction, rather than basal expression levels, are required for a GC response and may be a better predictor of GC sensitivity. Using a titratable expression system in human T-ALL cell line, Ramdas et al. demonstrated that basal levels of GR may be insufficient to confer GC sensitivity, but that levels comparable to those achieved following GC exposure and subsequent autoinduction of GR expression are sufficient to mediate GC-induced apoptosis [30]. Consistent with these data, a failure to autoinduce GR expression upon GC exposure has been implicated in GC resistance in Jurkat T-ALL cells [18] and in multiple myeloma cell lines [31].
Several studies have also identified other genetic and epigenetic events that lead to altered GR expression levels, and may therefore contribute to GC resistance. For example, loss-of-function mutations in the E3 ubiquitin ligase FBXW7 have been associated with a favorable prognosis and an early response to GC therapy in ALL [32, 33]. A later study demonstrated that FBXW7 mediates the ubiquitination and subsequent proteasomal degradation of GR, leading to insufficient GR levels to mediate GC-induced apoptosis. This same study found that inactivation of FBXW7 in an in vitro system was sufficient to restore GR expression and consequently, GC sensitivity [34]. The NALP3 inflammasome has also been implicated as a modulator of cellular GR levels. In an analysis of GC resistant primary ALL samples, it was found that decreased promoter methylation of CASP1 and NLRP3 resulted in increased expression of the NALP3 inflammasome, and that the associated increase in caspase 1 activity caused increased cleavage of GR protein, leading to an attenuated GC response mediated by a loss of GR protein expression [35]. Therefore, while basal GR expression has not proven to be a tractable biomarker with clinical utility for predicting GC sensitivity, altered levels of GR expression may nonetheless contribute to a poor GC response.
In addition to GR expression levels, post-transcriptional processing of the GR mRNA results in multiple GR isoforms, which may also play a role in dictating GC sensitivity. GRα is the most abundant GR isoform and has been shown to mediate the pro-apoptotic effects of GCs in lymphocytes. Exon 9 of NR3C1 encodes a portion of the LBD, and alternating splicing of this exon distinguishes the GRα isoform from the GRβ isoform [5]. The GRβ isoform does not bind GCs and does not have transcriptional activity, thereby impeding its pro-apoptotic activity [36]. Additionally, alternative splicing involving the intron between exons 3 and 4 gives rise to the GRγ isoform, which has an altered DBD. Therefore, GRγ retains ligand binding capacity but has limited transcriptional activity [5]. Finally, alternative splicing involving the LBD results in the production of the GR-A and GR-P isoforms, both of which fail to bind ligand [5]. As a result of the impaired activity of multiple GR isoforms, many groups have studied the relationship between GC sensitivity and the relative expression and distribution of these isoforms in a variety of lymphoid malignancies. One of the earliest such studies focused on a patient with chronic lymphocytic leukemia (CLL) who was found to have generalized GC resistance. An analysis of the expression pattern of GR isoforms in cells taken from this patient demonstrated decreased GRα expression and increased GRβ expression, resulting in an altered ratio between the two isoforms [37]. Given the dominant negative effect of GRβ on GRα, this group concluded that the altered ratio may contribute to the generalized GC resistance observed in this patient. Consistent with these findings, an analysis of 23 diagnostic ALL samples revealed an inverse correlation between the GRβ/GRα ratio and the number of apoptotic cells following in vitro exposure to prednisolone, further indicating that high expression of GRβ impairs GC sensitivity [38]. Relative to diagnostic samples, relapsed ALL samples have also been shown to have a decreased mRNA to protein ratio of GRα [39]. Similarly, GRγ expression has been shown to be increased in PPR patients relative to PGR patients, which is consistent with the idea that GRγ expression might impair the transcriptional activity of GRα, leading to an inferior GC response [40].
Further regulation of GR activity is mediated by the chaperone protein systems that interact with GR, the two most important of which are the HSP70 and HSP90 systems . These chaperones assist with maintaining GR in a conformation in which it is competent for ligand binding and they facilitate the subsequent nuclear translocation of ligand-bound GR [41]. Given the central role of chaperone proteins in modifying GR activity, several groups have hypothesized that aberrant expression or activity of these chaperone systems could contribute to GC resistance in lymphoid malignancies. However, in an analysis of PPR and PGHR patients treated on ALL–BFM trials, there was no correlation between in vivo GC sensitivity and HSP90 expression [42]. In a more in-depth analysis looking at mRNA expression of key chaperone proteins in GC sensitive versus GC resistant ALL cells, there were also no meaningful differences in transcript expression [43]. While this finding does not exclude the possibility that differences in protein expression of these chaperones may underlie differences in GC sensitivity, these studies suggest that chaperone proteins likely do not play a significant role in clinical GC resistance in lymphoid malignancies.
GR Extrinsic Mechanisms of GC Resistance
Epigenetic Regulation of GR Activity
Changes in GR target gene expression require the association of ligand-bound GR with a GRE [6]. Some of the cell-and tissue-specificity of GCs may be mediated by differences in chromatin accessibility, as GR binding has been shown to occur predominantly at accessible chromatin sites [44]. Given the requirement for pre-existing chromatin accessibility, a number of groups have assessed the role for an altered epigenetic landscape as a mediator of GC resistance. Chromatin accessibility is maintained in part through the activity of the SWI/SNF [44], and decreased expression of core components of this complex correlate with the occurrence of GC resistance in ALL cells [45]. In an analysis of gene expression and DNA methylation patterns in matched pairs of pediatric B-ALL samples obtained at the time of diagnosis and at relapse, Hogan et al. identified a distinct pattern of gene expression associated with relapse and found that this gene expression pattern co-occurred with increased promoter methylation [46]. With the addition of the DNA methyltransferase inhibitor decitabine, this relapse-specific gene expression pattern could be reverted, allowing for re-expression of hypermethylated genes. Exposure to decitabine, along with the histone deacetylase (HDAC) inhibitor vorinostat, resulted in significant potentiation of GC-induced apoptosis [47], suggesting that modification of the epigenetic landscape may facilitate GR-mediated changes in gene expression that lead to apoptosis. Similarly, it was shown that elevated expression of a number of HDAC genes is common in patients who have a PPR [48]. Consistent with these findings, Jones et al. reported a high frequency of deletions of TBL1XR1, a component of the nuclear receptor corepressor (NCoR) complex, in patients with B-ALL. These deletions stabilize NCoR, which represses GR activity by decreasing its recruitment to target gene loci and by recruiting HDAC3 to further promote inhibition of target gene expression. Treating these cells with an HDAC inhibitor was sufficient to restore GC sensitivity [49] Collectively, these data suggest that GC sensitivity is mediated in part by a permissive epigenetic landscape, and that the use of epigenetic modulators may represent a therapeutic strategy to enhance GC sensitivity in lymphoid malignancies that are associated with an altered epigenetic landscape.
Signal Transduction
Dysregulated signal transduction is a hallmark feature of many lymphoid malignancies including T-ALL [50], B-ALL [51], and non-Hodgkin lymphoma (NHL) [52]. Importantly, the downstream effectors of these signal transduction pathways exhibit known cross-talk with GR signaling and transcriptional activity [53]. As a result of these interactions, aberrant regulation of these signal transduction pathways is an important cause of GC resistance in lymphoid malignancies and significant attention has been devoted to the use of targeted signal transduction inhibitors as a strategy to overcome GC resistance.
Cyclic Adenosine Monophosphate (cAMP) Signaling
cAMP is a second messenger molecule that initiates signaling cascades responsible for mediating a variety of immune cell functions. cAMP is generated through the catalytic activity of adenylate cyclases and is degraded by a family of enzymes called phosphodiesterases (PDEs) [54]. It has long been known that in addition to GCs, activation of cAMP decreases lymphoid cell proliferation and induces apoptosis [55]. Furthermore, it has been shown in T-cell lines that stimulation of cAMP signaling has a synergistic effect to induce cell death when combined with the GC dexamethasone (DEX) [56], and that cAMP and GCs likely converge to promote the upregulation of BIM expression [57, 58], thereby facilitating the induction of apoptosis. Given the pro-apoptotic effects of cAMP and the effects of PDEs to decrease the cellular pool of cAMP, significant attention has been devoted to the development of PDE inhibitors [59], and a number of groups have evaluated the efficacy of PDE inhibitors as a means of overcoming GC resistance in lymphoid malignancies. In the CCRF-CEM cell line, both a non-specific PDE inhibitor and rolipram, a PDE4-specific inhibitor, significantly potentiated DEX-induced apoptosis [60]. In primary CLL cells, rolipram synergized with GCs to induce apoptosis, and this effect was associated with increased GR-mediated transcriptional activity [61]. Furthermore, in these same cells, it was found that rolipram exposure resulted in an increase in both transcript and protein expression of GRα [62]. In patients, PDE4 overexpression has been observed in a cohort of primary diffuse large B-cell lymphoma (DLBCL) samples. Consistent with the data in leukemia cell lines, inhibition of PDE4 in DLBCL cells was sufficient to restore GC sensitivity [63]. Finally, in a large-scale gene expression analysis of primary DLBCL samples obtained from patients who received treatment with cyclophosphamide, adriamycin, vincristine, and prednisone (CHOP), elevation of PDE4 expression was enriched in patients with fatal or refractory disease relative to patients who were cured with CHOP therapy [64]. Taken together, these data suggest that alterations in cAMP pathway signaling may contribute to GC resistance in lymphoid malignancies and that therapeutic targeting of this pathway may have clinical utility .
Mitogen Activated Protein Kinase (MAPK) Signaling
The three best studied MAPKs are p38, ERK, and JNK, all of which become activated downstream of a signaling cascade induced by cellular exposure to mitogenic stimuli [65]. Each of these MAPKs has been shown to modulate GC sensitivity, resulting in a considerable number of studies devoted to investigating the therapeutic potential of MAPK pathway signaling modulators as a means of enhancing GC sensitivity. In GC resistant clones derived from the parental CCRF-CEM cells, inhibition of p38 MAPK decreased DEX sensitivity, while inhibition of ERK activity increased sensitivity. These data implicate p38 as a positive regulator of GC activity and ERK as a negative regulator of GC activity [66], suggesting that distinct arms of the MAPK signaling cascade interact differently with the GR pathway. Consistent with these findings, it has been shown that exposing CCRF-CEM cells to DEX results in increased phosphorylation and activation of p38, one substrate of which is GR itself. Specifically, this study demonstrated that p38 mediates Ser-211 phosphorylation of GR [67], which has been shown to increase the transcriptional activity of GR [5], thereby providing a mechanistic explanation for the positive effect of p38 activity on GC sensitivity. Another study demonstrated that inhibition of p38 in CCRF-CEM cells resulted in decreased induction of BIM expression upon DEX exposure, leading to an attenuated apoptotic response and suggesting that p38 might further contribute to GC sensitivity by enabling the upregulation of BIM expression [68].
Other studies have focused on elucidating the molecular basis for the inhibitory effect of ERK signaling on GC sensitivity. Importantly, ERK has been shown to phosphorylate BIM, preventing it from interacting with other members of the intrinsic apoptotic pathway to induce apoptosis [69]. To determine whether this mechanism contributes to ERK-mediated GC resistance, Rambal et al. demonstrated in ALL cell lines and primary patient samples a synergistic interaction between a MEK inhibitor and DEX, with simultaneous exposure to both agents resulting in increased BIM expression due to a reduction in ERK-mediated BIM phosphorylation [70]. In addition to ERK, JNK activation has previously been implicated as a negative regulator of GC sensitivity. In contrast to p38, JNK is known to catalyze an inhibitory phosphorylation of GR, resulting in decreased transcriptional activity [71]. Jones et al. further established the role of ERK and JNK as negative regulators of GC sensitivity through an shRNA screen designed to identify genes that modify prednisolone sensitivity in B-ALL cell lines. Interestingly, this screen identified MEK2, which activates ERK, and MEK4, which activates JNK, as important candidate GC resistance genes. Through a variety of functional studies, the authors demonstrated that loss of MEK2 expression induced generalized chemosensitivity, including to GCs, through a p53-dependent mechanism and that loss of MEK4 increased expression of GR, leading to improved GC sensitivity. Furthermore, they demonstrated the clinical relevance of these findings by assessing ERK activity in paired diagnostic and relapse samples from patients with B-ALL and found increased levels of phosphorylated ERK in the relapsed samples [72], consistent with the idea that aberrant activation of ERK signaling may contribute to GC resistance. Given the large number of past and current clinical trials conducted in a wide variety of malignancies [73], the addition of small molecules that modulate MAPK pathway activity may be a feasible strategy for overcoming GC resistance in some lymphoid malignancies .
PI3K/AKT/mTOR Signaling
The PI3K/AKT/mTOR pathway is another signal transduction pathway that is commonly dysregulated in lymphoid malignancies and represents a potential therapeutic target for strategies aimed at overcoming GC resistance. In a recent study involving a large cohort of pediatric T-ALL samples, AKT1 and PTEN mutations were two of only a handful of genetic lesions that had a univariable association with relapse [50], suggesting that mutational activation of this pathway may play a role in therapy resistance, including to GCs. In another analysis of primary B-ALL samples, patients with increased phosphorylated AKT at diagnosis had a significantly inferior response to steroid-containing induction therapy and had decreased overall and relapse-free survival [74]. These studies provide correlative evidence for the role of aberrant PI3K/AKT/mTOR pathway activity in GC resistance. To more directly assess a mechanistic basis for this relationship, Piovan et al. demonstrated using co-immunoprecipitation that AKT1 binds to and phosphorylates GR on Ser-134, a phosphorylation event that impairs nuclear translocation of ligand-activated GR. Using the PTEN-null CCRF-CEM cell line, the authors demonstrated through both in vitro and in vivo studies that combined treatment with GCs and the AKT inhibitor MK2206 is sufficient to reverse GC resistance [75]. One class of proteins that has been found to cooperate with AKT to modulate GC activity is the 14–3–3 class of phospho-serine/threonine binding proteins, which regulate the subcellular localization of proteins with phosphorylated serine or threonine residues, including phosphorylated GR [76]. Consistent with this function, the 14–3–3σ protein interacts with GR upon AKT1-mediated Ser-134 phosphorylation, resulting in impaired nuclear translocation of ligand-bound GR and leading to reduced transcriptional activity in the presence of GCs [76, 77]. Similarly, it has been shown that more proximal inhibition of this pathway with a PI3K inhibitor results in synergy when combined with GCs, both in vitro and in an in vivo xenograft model [78]. In B-ALL cell lines and primary diagnostic patient samples, PI3K inhibition augmented nuclear translocation of ligand-activated GR through a reduction in Ser-134 phosphorylation [79], further confirming the effect of aberrant PI3K/AKT pathway inhibition to promote cytoplasmic retention of GR and prevent transcriptional activation.
One important downstream effector of PI3K/AKT pathway activation is mTOR [80], and many groups have studied the role of aberrant mTOR activation as a mediator of GC resistance in lymphoid malignancies. Using a chemical genomics approach, Wei et al. compared a large number of drug-associated gene expression profiles with the gene expression signature of GC sensitive and resistant ALL cells. Through this analysis, they determined that the changes in gene expression associated with exposure to the mTOR inhibitor rapamycin matched that associated with GC sensitive cells, suggesting that rapamycin may show efficacy by altering the gene expression pattern in GC resistant cells to better mimic that of GC sensitive cells. They further demonstrated that exposure to rapamycin sensitized cells to GCs through a mechanism involving downregulation of expression of the anti-apoptotic protein MCL1 [81]. Similarly, Gu et al. demonstrated a synergistic relationship between rapamycin and DEX in a panel of T-ALL cells, and further elucidated the mechanistic basis for this interaction by identifying a synergistic induction of expression of the pro-apoptotic BAX and BIM proteins in conjunction with downregulation of MCL1 [82]. In addition, it has been shown that simultaneous exposure to an mTOR inhibitor and GCs results in a synergistic induction of the cyclin-dependent kinase (CDK) inhibitor proteins p21 and p27 [82, 83], suggesting that mTOR inhibitors and GCs converge both to induce cell cycle arrest and activation of the intrinsic apoptotic pathway. This effect was further demonstrated in vivo using PDXs derived from primary patient T- and B-ALL samples, and the combinatorial effect of mTOR inhibition and GCs was found to be particularly effective in T-ALL samples with loss of PTEN expression [84], providing further evidence that aberrant regulation of upstream PI3K/AKT pathway activity results in altered mTOR activity that can be targeted therapeutically to augment the GC response. Finally, given the direct effects of both AKT and mTOR on GC sensitivity, several groups have investigated the efficacy of the dual PI3K and mTOR inhibitor BEZ235, reasoning that dual inhibition at two critical points in this pathway may have a more profound effect to induce GC sensitivity. Indeed, in ALL cell lines and primary patient samples both in vitro and in vivo, synergy has been demonstrated between BEZ235 and DEX [85, 86], suggesting that multiple nodes within this pathway are viable therapeutic targets for augmenting GC sensitivity .
JAK/STAT Signaling
The JAK/STAT signaling pathway is the critical effector pathway of cytokine receptor signaling, which plays a crucial role in mediating survival, proliferation, and differentiation of lymphoid cells [87]. Not surprisingly, aberrant activation of this pathway is common in lymphoid malignancies [50, 88], and significant attention has been devoted to assessing the role of JAK/STAT pathway inhibition as a novel treatment modality. Activation of cytokine receptors recruits JAK proteins to intracellular domains of cytokine receptors, and these activated JAK proteins recruit and phosphorylate STAT proteins, which translocate to the nucleus and function as transcription factors [87]. Interestingly, GR and one of these STAT proteins, STAT5, have been shown to physically interact at certain genomic loci. Specifically, STAT5 is known to inhibit the action of GR on GR target genes [89]. Consistent with this inhibitory role of JAK/STAT signaling on GR activity, inhibition of this pathway has been shown to overcome GC resistance in a number of lymphoid malignancies. In Philadelphia chromosome-like B-ALL, which is associated with aberrant JAK/STAT pathway activation, the combination of a JAK2 specific inhibitor and DEX demonstrated in vitro synergy and showed improved survival in an in vivo xenograft model [90]. Similarly, in primary diagnostic T-ALL samples, exposure to the cytokine interleukin-7 resulted in increased JAK/STAT pathway activity that induced GC resistance and could be overcome with the addition of the JAK1/2 inhibitor ruxolitinib [91]. Finally, in CLL cells, GC resistance was found to be associated with autocrine activation of another STAT protein, STAT3, and inhibition of STAT3 activation with ruxolitinib resulted in increased sensitivity to DEX in vitro [92].
NOTCH Signaling
NOTCH receptors are transmembrane receptors that, upon ligand binding, undergo a series of cleavage events to release the activated intracellular component of NOTCH from the membrane, allowing it to translocate to the nucleus and function as a transcription factor. The γ-secretase complex mediates the final step in this processing [93]. Due to the important role of NOTCH signaling in the pathogenesis of T-cell malignancies, inhibitors of this γ-secretase complex have been evaluated as potential therapeutic agents for the treatment of these diseases in combination with GCs. Specifically, in T-ALL cell lines, γ-secretase inhibitors have been shown to sensitize cells to the cytotoxic effects of DEX [94]. Several groups have demonstrated that the combination of γ-secretase inhibitors and GCs facilitates autoinduction of GR and potentiates the induction of BIM expression, leading to increased cell death in both in vitro and in vivo model systems [95, 96]. Despite these promising preclinical findings, the clinical utility of γ-secretase inhibitors has been limited by severe gastrointestinal toxicity [97]. However, in an elegant study conducted in a T-ALL xenograft model, it was shown that simultaneous exposure to a γ-secretase inhibitor and DEX not only overcame GC resistance, but also attenuated the toxicities associated with the γ-secretase inhibitor [95], suggesting that the combination of γ-secretase inhibitors and GCs may be a viable therapeutic strategy to enhance GC sensitivity. Finally, at least one study has evaluated the efficacy of an anti-NOTCH1 monoclonal antibody in a T-ALL PDX model and demonstrated potentiation of GC activity when given in combination [98].
Src Family Kinase Signaling
In T-cells, the Src family kinases Lck and Fyn mediate critical signal transduction events downstream of the T-cell receptor (TCR) [99]. Through the use of reverse-phase protein arrays applied to PPR and PGR T-ALL samples, Lck was found to be aberrantly activated in PPR patients relative to PGR patients [100]. Consistent with these findings, inhibition of Lck with the Src family kinase inhibitor dasatinib has demonstrated in vitro efficacy to enhance GC sensitivity [101] and has been shown to impair the engraftment of T-ALL cells in vivo relative to treatment with either agent alone [100].
Metabolism
In addition to studies demonstrating the importance of GR expression levels as a mediator of GC sensitivity, many groups have demonstrated that metabolic processes that limit the availability of GC ligand can similarly contribute to GC resistance. In normal physiology, the 11β-hydroxysteroid dehydrogenase (HSD) class of enzymes mediates the conversion between cortisol, the active endogenous hormone, and cortisone, the inert form of the hormone. Specifically, 11β-HSD1 regenerates cortisol from cortisone while 11β-HSD2 inactivates cortisol [102]. In an analysis of primary patient ALL samples, basal 11β-HSD1 expression was found to be higher in GC sensitive samples relative to GC resistant samples. Furthermore, 11β-HSD1 expression was upregulated in response to DEX exposure specifically in the GC sensitive samples but not in the GC resistant samples, suggesting that 11β-HSD1 may participate in a GC-regulated feedback loop to maintain the availability of ligand for GR binding [103]. The same group similarly analyzed 11β-HSD2 expression in the GC resistant T-ALL cell line MOLT4F and the GC sensitive CCRF-CEM cell line and demonstrated that 11β-HSD2 expression was higher in the setting of GC resistance. They further demonstrated that pharmacologic inhibition of 11β-HSD2 was sufficient to potentiate GC-induced apoptosis [104]. Consistent with these findings, 11β-HSD2 expression was compared between GC resistant T-ALL cell lines, GC sensitive NHL cell lines, and normal peripheral T-cells. In the GC resistant cell lines, 11β-HSD2 expression was found to be significantly elevated relative to the GC sensitive cell lines or normal T-cells [105]. To determine how 11β-HSD2 is dynamically regulated in the presence of GCs, transcript and protein expression as well as enzymatic activity were assessed in the GC sensitive CEM-C7 cell line after exposure to DEX. This analysis demonstrated a reduction in expression and enzymatic activity upon DEX exposure, suggesting that, in contrast to 11β-HSD1, GC-induced downregulation of 11β-HSD2 may be important for maintaining GC sensitivity [106].
In addition to HSDs, glutathione S-transferases (GSTs) are a class of enzymes involved in the metabolism of a wide variety of drugs, including steroids [107]. In an analysis of PGR and PPR patient samples from children treated on an ALL-BFM protocol, deletion of the GST family member GSTT1 was enriched in the PPR patient group and was associated with an increased risk of relapse [108]. These data suggest that genetic lesions involving GST genes might contribute to differences in clinical GC response, though further studies are needed to determine whether aberrant GST activity plays a significant role in altering the availability of GC ligand and whether this contributes to GC resistance.
While metabolism of GCs themselves may play a role in modulating GC sensitivity, GC resistance has also been attributed to the aberrant activity of key bioenergetic metabolic pathways. Specifically, it has been shown in both ALL cell lines and in primary patient ALL samples that GC resistance is associated with increased rates of glycolysis, oxidative phosphorylation, and cholesterol biosynthesis. In a gene expression profiling study using ALL cell lines, pathways involved in these metabolic processes emerged as the top biological pathways associated with GC resistance. Furthermore, when these gene sets were studied in the context of primary patient samples, enrichment for these gene sets was a strong predictor of relapse [109], suggesting that activation of these bioenergetic pathways may promote chemoresistance. The same group went on to demonstrate that inhibition of glycolysis, oxidative phosphorylation, or cholesterol biosynthesis was sufficient to sensitize GC resistant T-ALL cells to GCs [110], further supporting the idea that aberrant activation of cellular metabolic processes may confer GC resistance.
Based on these findings, significant attention has been devoted to studying the role of glucose metabolism as a modulator of GC sensitivity. In an analysis of a large cohort of primary B-ALL samples with varying degrees of in vitro prednisolone sensitivity, genes associated with carbohydrate metabolism were found to be differentially expressed between GC sensitive and GC resistant samples [111]. Furthermore, in ALL cell lines and primary patient samples, prednisolone resistance was found to correlate with increased glucose consumption, and inhibition of glycolysis with the metabolite 2-deoxy-D-glucose (2-DG) sensitized cells to GCs, supporting the idea that excessive metabolic activity may impair GC-induced apoptosis [112]. To further assess the relationship between glucose consumption and GC sensitivity, GC sensitive ALL cell lines and primary patient samples were exposed to DEX, which was found to inhibit glycolysis, leading to decreased glucose consumption that was mediated by a reduction in the expression of the glucose transporter GLUT1. This group went on to demonstrate that culturing cells in low glucose conditions resulted in increased DEX-induced apoptosis [113]. Taken together, these data suggest that a reduction in glucose metabolism may be required for optimal GC-induced apoptosis. Consistent with this idea, it was shown that in prednisolone sensitive primary B-ALL samples, MCL1 expression decreased upon exposure to prednisolone, while it did not decrease in prednisolone resistant samples. Genetic silencing of MCL1 was found to be associated with an increase in glucose consumption, and simultaneous inhibition of glycolysis and silencing of MCL1 resulted in further sensitization to prednisolone [114]. The importance of excessive glucose metabolism as a mediator of GC resistance has also been studied in the context of NHL cell lines and primary patient samples. In these cells, inhibition of glycolysis was found to synergize with methylprednisolone to induce cell cycle arrest and apoptosis [115]. Providing a genetic explanation for the relationship between altered glucose metabolism and GC sensitivity, Chan et al. recently performed ChIP-seq to assess the binding pattern of transcription factors that are commonly inactivated in B-ALL, including PAX5 and IKZF1. They demonstrated that in B-ALL, these transcription factors are recruited to genetic loci that encode positive and negative regulators of glucose uptake. Re-expression of PAX5 and IKZF1 in B-ALL cells resulted in decreased glucose uptake and was sufficient to overcome prednisolone resistance [116]. The authors speculated that the hypermetabolic state associated with the deletion of these transcription factors facilitates leukemogenesis and simultaneously facilitates resistance to GC therapy.
In addition to glucose metabolism, aberrant lipid metabolism has also been shown to contribute to GC resistance. Specifically, lymphoid cells have been shown to have a unique dependency on exogenously synthesized cholesterol, and similar to glycolysis, GCs may exert their pro-apoptotic effects in part through inhibiting this cholesterol synthesis pathway. Indeed, in GC sensitive CEM-C7 cells, DEX was found to inhibit cholesterol synthesis, while this did not occur effectively in GC resistant CEM-C1 cells. Furthermore, exposure of CEM-C7 cells to exogenous cholesterol decreased DEX sensitivity, suggesting that DEX resistance may be mediated in part by increased cholesterol metabolism [117]. Further supporting these data, T-ALL PDXs treated with a GC-containing four-drug induction regimen that acquired in vivo drug resistance were found to have altered cholesterol metabolism. In these samples, exposure to DEX and simvastatin, an inhibitor of cholesterol biosynthesis, demonstrated ex vivo synergy [118]. These data suggest that additional preclinical studies may be warranted to evaluate the use of drugs that modulate bioenergetic pathways as a means of overcoming GC resistance.
MicroRNAs
MicroRNAs (miRNAs) are short non-coding RNAs that are most commonly contained within introns. Once transcribed, they bind to complementary sequences within the 3’ untranslated region (3’UTR) of target gene mRNAs. Through this activity, miRNAs function primarily as negative regulators of translation, though they may have other repressive and activating roles [119]. Dysfunctional expression of miRNAs is a common feature of many cancers, including hematologic malignancies. In ALL samples, a miRNA microarray analysis of paired diagnostic and relapse samples identified a distinct miRNA profile in the relapse samples relative to the diagnostic samples [120]. Similarly, in a study involving miRNA sequencing of samples from patients with Burkitt lymphoma , DLBCL, and follicular lymphoma , many miRNAs were found to be aberrantly expressed in lymphoma cells relative to normal lymphoid cells. Functionally, these miRNAs were found to be associated with altered regulation of key signal transduction pathways, including the Ras/MAPK and PI3K/AKT signaling pathways, suggesting that these miRNAs may play a role both in lymphomagenesis and in chemoresistance, including GC resistance [121].
MiR-17
In B-ALL cell lines, DEX exposure downregulated expression of miR-17 in GC sensitive but not in GC resistant cells. ChIP-seq analysis demonstrated that this is mediated by direct GR binding to the miR-17 locus specifically in GC sensitive cells. Functionally, miR-17 was found to target the BIM transcript for silencing, and pharmacologic inhibition of miR-17 increased DEX sensitivity with an associated increase in BIM expression [122].
MiR-100/99a
The miRNA species miR-100/99a has also been implicated in GC resistance and is known to be downregulated in samples from ALL patients with clinically high risk features. Specifically, low expression has been associated with inferior leukemia-free and overall survival [123]. In cell lines, ectopic expression of miR-100/99a promoted DEX-induced apoptosis through a reduction in expression of the miR-100/99a target FKBP51. The reduction in FKBP51 expression was associated with increased nuclear localization of ligand-bound GR and decreased expression of mTOR, subsequently leading to a reduction in MCL1 expression which further potentiated apoptosis [123].
MiR-124
The role of MiR-124 as a mediator of GC resistance was first appreciated in the context of sepsis, where it was found that miR-24 represses the GRα transcript [124], suggesting that its overexpression might mediate GC resistance by decreasing the availability of GR protein for ligand binding. Indeed, miR-124 expression was found to be increased in prednisolone resistant ALL cell lines and in PPR patient samples and overexpression of miR-124 in ALL cells was associated with a reduction in GR protein expression [125]. However, at least one study has suggested the opposite effect of miR-124 in GC sensitivity. In DLBCL cells, miR-124 expression was found to decrease expression of PDE4B, thereby relieving the inhibitory effect on cAMP signaling and increasing GC sensitivity [126]. Further studies are therefore needed to elucidate the role of miR-124 in modulating GC sensitivity in distinct lymphoid malignancies.
MiR-128b and miR-221
In MLL-AF4 ALL , miR-128b and miR-221 were found to be downregulated relative to other types of ALL. Overexpression of these miRNAs in MLL-AF4 ALL cell lines resulted in increased sensitivity to GCs, which was accompanied by downregulation of MLL, AF4, and their associated fusion genes [127]. Further implicating low miR-128b as a mediator of GC resistance in MLL-AF4 ALL, miR-128b mutations found in both cell lines and primary patient samples were shown to impair the appropriate processing of miR-128b. This resulted in GC resistance mediated by a failure to downregulate the expression of fusion oncogenes involving MLL and AF4, though the mechanisms by which MLL and AF4 themselves contribute to GC resistance are currently unknown [128].
MiR-142-3p
MiR-142-3p was initially shown in T-regulatory cells to target adenylyl cyclase 9 mRNA for silencing, resulting in a reduction in the cellular pool of cAMP due to the loss of adenylyl cyclase enzymatic activity [129]. Elevated expression of miR-142-3p in primary T-ALL samples was found to be associated with an increased risk of relapse and decreased leukemia-free survival relative to patients with lower miR-142-3p expression. Consistent with its known effects on the adenylyl cyclase 9 transcript, high miR-142-3p expression was associated with increased cAMP pathway activity. Furthermore, miR-142-3p was found to target the GRα transcript for repression via direct binding to the 3’UTR. In this context, inhibition of miR-142-3p overcame GC resistance both by facilitating an increase in cAMP pathway activity and an increase in GRα expression [130].
MiR-182
In an analysis of a variety of murine and human malignant lymphoid cell lines, miR-182 expression was higher in GC resistant cells relative to GC sensitive cells, and high expression was associated with decreased FOXO3A expression. One important downstream target of FOXO3A is BIM, and high expression of miR-182 was also associated with a reduction in BIM expression. Consistent with this activity, overexpression of miR-182 restored BIM expression, thereby overcoming GC resistance [131].
MiR-185-5p
Finally, miR-185-5p was found to be overexpressed in GC sensitive ALL cell lines. One target of miR-185-5p is the mTORC2 mRNA. Forced overexpression of miR-185-5p in GC resistant ALL cells restored GC sensitivity with a concomitant reduction in mTORC2 activity [132].
Conclusion
Given the pleiotropic effects of GCs and the innumerable interactions between GR and a wide variety of cellular processes, it is not surprising that the mechanisms of GC resistance are complex and that our understanding of these mechanisms is constantly evolving (Fig. 1.2). However, despite a well-justified concern for GC resistance and its associated clinical implications, GCs are profoundly efficacious in the treatment of lymphoid malignancies and will undoubtedly remain an integral component of therapy. Therefore, there is an urgent need to translate the findings from the numerous preclinical and clinical studies highlighted in this review into standard clinical practice for the treatment of these diseases. With the application of large-scale sequencing and epigenetic profiling technologies, the development of small molecule and biologic therapeutics, and increasing access to patient-derived tissue samples, there is significant potential for the elucidation of additional causes of GC resistance and the identification and implementation of novel therapeutic strategies to overcome them.
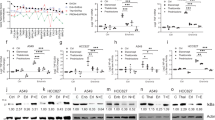
Mechanisms of GC Resistance. GC resistance may arise due to processes that impact any component of normal GR signaling, including the availability of GC ligand or GR (A), nuclear translocation of the activated GC/GR complex (B), transcriptional activity of ligand-bound (C), or the induction of apoptosis (D)
Abbreviations
- 2-DG:
-
2-Deoxy-D-Glucose
- 3’UTR:
-
3’ Untranslated Region
- B-CLL:
-
B-Cell Chronic Lymphocytic Leukemia
- BFM:
-
Berlin-Frankfurt-Munster
- cAMP:
-
Cyclic Adenosine Monophosphate
- CDK:
-
Cyclin Dependent Kinase
- ChIP-Seq:
-
Chromatin Immunoprecipitation with Sequencing
- CHOP:
-
Cyclophosphamide, Adriamycin, Vincristine, and Prednisone
- DBD:
-
DNA Binding Domain
- DEX:
-
Dexamethasone
- DLBCL:
-
Diffuse Large B-Cell Lymphoma
- EFS:
-
Event-Free Survival
- GC:
-
Glucocorticoid(s)
- GR:
-
Glucocorticoid Receptor
- GRE:
-
Glucocorticoid Response Element
- GST:
-
Glutathione S-Transferase
- HDAC:
-
Histone Deacetylase
- HSD:
-
Hydroxysteroid Dehydrogenase
- LBD:
-
Ligand Binding Domain
- LOH:
-
Loss of Heterozygosity
- MAPK:
-
Mitogen Activated Protein Kinase
- miR:
-
MicroRNA
- MRD:
-
Minimal Residual Disease
- NCoR:
-
Nuclear Co-Receptor
- NHL:
-
Non-Hodgkin Lymphoma
- NTD:
-
N-Terminal Transactivation Domain
- PDE:
-
Phosphodiesterase
- PDX:
-
Patient-Derived Xenograft
- PGR:
-
Prednisone Good Responder
- PKA:
-
Protein Kinase A
- PPR:
-
Prednisone Poor Responder
- RT-PCR:
-
Real-Time Polymerase Chain Reaction
- T-ALL:
-
T-Cell Acute Lymphoblastic Leukemia
- TCR:
-
T-Cell Receptor
- WBC:
-
White Blood Cell
References
Schrappe M, Reiter A, Zimmermann M, Harbott J, Ludwig WD, Henze G, et al. Long-term results of four consecutive trials in childhood ALL performed by the ALL-BFM study group from 1981 to 1995. Berlin-Frankfurt-Munster. Leukemia. 2000;14(12):2205–22.
Fisher RI, Gaynor ER, Dahlberg S, Oken MM, Grogan TM, Mize EM, et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma. N Engl J Med. 1993;328(14):1002–6.
Dordelmann M, Reiter A, Borkhardt A, Ludwig WD, Gotz N, Viehmann S, et al. Prednisone response is the strongest predictor of treatment outcome in infant acute lymphoblastic leukemia. Blood. 1999;94(4):1209–17.
Mathew BS, Carson KA, Grossman SA. Initial response to glucocorticoids. Cancer. 2006;106(2):383–7.
Oakley RH, Cidlowski JA. The biology of the glucocorticoid receptor: new signaling mechanisms in health and disease. J Allergy Clin Immunol. 2013;132(5):1033–44.
Yamamoto KR. Steroid receptor regulated transcription of specific genes and gene networks. Annu Rev Genet. 1985;19:209–52.
Schmidt S, Rainer J, Ploner C, Presul E, Riml S, Kofler R. Glucocorticoid-induced apoptosis and glucocorticoid resistance: molecular mechanisms and clinical relevance. Cell Death Differ. 2004;11(Suppl 1):S45–55.
Ploner C, Rainer J, Niederegger H, Eduardoff M, Villunger A, Geley S, et al. The BCL2 rheostat in glucocorticoid-induced apoptosis of acute lymphoblastic leukemia. Leukemia. 2008;22(2):370–7.
Jing D, Bhadri VA, Beck D, Thoms JA, Yakob NA, Wong JW, et al. Opposing regulation of BIM and BCL2 controls glucocorticoid-induced apoptosis of pediatric acute lymphoblastic leukemia cells. Blood. 2015;125(2):273–83.
Charmandari E, Kino T, Chrousos GP. Primary generalized familial and sporadic glucocorticoid resistance (Chrousos syndrome) and hypersensitivity. Endocr Dev. 2013;24:67–85.
Bray PJ, Cotton RG. Variations of the human glucocorticoid receptor gene (NR3C1): pathological and in vitro mutations and polymorphisms. Hum Mutat. 2003;21(6):557–68.
Harmon JM, Thompson EB. Isolation and characterization of dexamethasone-resistant mutants from human lymphoid cell line CEM-C7. Mol Cell Biol. 1981;1(6):512–21.
Ashraf J, Thompson EB. Identification of the activation-labile gene: a single point mutation in the human glucocorticoid receptor presents as two distinct receptor phenotypes. Mol Endocrinol. 1993;7(5):631–42.
Powers JH, Hillmann AG, Tang DC, Harmon JM. Cloning and expression of mutant glucocorticoid receptors from glucocorticoid-sensitive and -resistant human leukemic cells. Cancer Res. 1993;53(17):4059–65.
Hala M, Hartmann BL, Bock G, Geley S, Kofler R. Glucocorticoid-receptor-gene defects and resistance to glucocorticoid-induced apoptosis in human leukemic cell lines. Int J Cancer. 1996;68(5):663–8.
Strasser-Wozak EM, Hattmannstorfer R, Hala M, Hartmann BL, Fiegl M, Geley S, et al. Splice site mutation in the glucocorticoid receptor gene causes resistance to glucocorticoid-induced apoptosis in a human acute leukemic cell line. Cancer Res. 1995;55(2):348–53.
Hillmann AG, Ramdas J, Multanen K, Norman MR, Harmon JM. Glucocorticoid receptor gene mutations in leukemic cells acquired in vitro and in vivo. Cancer Res. 2000;60(7):2056–62.
Riml S, Schmidt S, Ausserlechner MJ, Geley S, Kofler R. Glucocorticoid receptor heterozygosity combined with lack of receptor auto-induction causes glucocorticoid resistance in Jurkat acute lymphoblastic leukemia cells. Cell Death Differ. 2004;11(Suppl 1):S65–72.
Beesley AH, Weller RE, Senanayake S, Welch M, Kees UR. Receptor mutation is not a common mechanism of naturally occurring glucocorticoid resistance in leukaemia cell lines. Leuk Res. 2009;33(2):321–5.
Tissing WJ, Meijerink JP, den Boer ML, Brinkhof B, van Rossum EF, van Wering ER, et al. Genetic variations in the glucocorticoid receptor gene are not related to glucocorticoid resistance in childhood acute lymphoblastic leukemia. Clin Cancer Res. 2005;11(16):6050–6.
Irving JA, Minto L, Bailey S, Hall AG. Loss of heterozygosity and somatic mutations of the glucocorticoid receptor gene are rarely found at relapse in pediatric acute lymphoblastic leukemia but may occur in a subpopulation early in the disease course. Cancer Res. 2005;65(21):9712–8.
Mullighan CG, Phillips LA, Su X, Ma J, Miller CB, Shurtleff SA, et al. Genomic analysis of the clonal origins of relapsed acute lymphoblastic leukemia. Science. 2008;322(5906):1377–80.
Oshima K, Khiabanian H, da Silva-Almeida AC, Tzoneva G, Abate F, Ambesi-Impiombato A, et al. Mutational landscape, clonal evolution patterns, and role of RAS mutations in relapsed acute lymphoblastic leukemia. Proc Natl Acad Sci USA. 2016;113(40):11306–11.
Pui CH, Dahl GV, Rivera G, Murphy SB, Costlow ME. The relationship of blast cell glucocorticoid receptor levels to response to single-agent steroid trial and remission response in children with acute lymphoblastic leukemia. Leuk Res. 1984;8(4):579–85.
Quddus FF, Leventhal BG, Boyett JM, Pullen DJ, Crist WM, Borowitz MJ. Glucocorticoid receptors in immunological subtypes of childhood acute lymphocytic leukemia cells: a pediatric oncology group study. Cancer Res. 1985;45(12 Pt 1):6482–6.
Kato GJ, Quddus FF, Shuster JJ, Boyett J, Pullen JD, Borowitz MJ, et al. High glucocorticoid receptor content of leukemic blasts is a favorable prognostic factor in childhood acute lymphoblastic leukemia. Blood. 1993;82(8):2304–9.
Grausenburger R, Bastelberger S, Eckert C, Kauer M, Stanulla M, Frech C, et al. Genetic alterations in glucocorticoid signaling pathway components are associated with adverse prognosis in children with relapsed ETV6/RUNX1-positive acute lymphoblastic leukemia. Leuk Lymphoma. 2016;57(5):1163–73.
Lauten M, Cario G, Asgedom G, Welte K, Schrappe M. Protein expression of the glucocorticoid receptor in childhood acute lymphoblastic leukemia. Haematologica. 2003;88(11):1253–8.
Denton RR, Eisen LP, Elsasser MS, Harmon JM. Differential autoregulation of glucocorticoid receptor expression in human T- and B-cell lines. Endocrinology. 1993;133(1):248–56.
Ramdas J, Liu W, Harmon JM. Glucocorticoid-induced cell death requires autoinduction of glucocorticoid receptor expression in human leukemic T cells. Cancer Res. 1999;59(6):1378–85.
Gomi M, Moriwaki K, Katagiri S, Kurata Y, Thompson EB. Glucocorticoid effects on myeloma cells in culture: correlation of growth inhibition with induction of glucocorticoid receptor messenger RNA. Cancer Res. 1990;50(6):1873–8.
Asnafi V, Buzyn A, Le Noir S, Baleydier F, Simon A, Beldjord K, et al. NOTCH1/FBXW7 mutation identifies a large subgroup with favorable outcome in adult T-cell acute lymphoblastic leukemia (T-ALL): a Group for Research on Adult Acute Lymphoblastic Leukemia (GRAALL) study. Blood. 2009;113(17):3918–24.
Clappier E, Collette S, Grardel N, Girard S, Suarez L, Brunie G, et al. NOTCH1 and FBXW7 mutations have a favorable impact on early response to treatment, but not on outcome, in children with T-cell acute lymphoblastic leukemia (T-ALL) treated on EORTC trials 58881 and 58951. Leukemia. 2010;24(12):2023–31.
Malyukova A, Brown S, Papa R, O’Brien R, Giles J, Trahair TN, et al. FBXW7 regulates glucocorticoid response in T-cell acute lymphoblastic leukaemia by targeting the glucocorticoid receptor for degradation. Leukemia. 2013;27(5):1053–62.
Paugh SW, Bonten EJ, Savic D, Ramsey LB, Thierfelder WE, Gurung P, et al. NALP3 inflammasome upregulation and CASP1 cleavage of the glucocorticoid receptor cause glucocorticoid resistance in leukemia cells. Nat Genet. 2015;47(6):607–14.
Oakley RH, Sar M, Cidlowski JA. The human glucocorticoid receptor beta isoform. Expression, biochemical properties, and putative function. J Biol Chem. 1996;271(16):9550–9.
Shahidi H, Vottero A, Stratakis CA, Taymans SE, Karl M, Longui CA, et al. Imbalanced expression of the glucocorticoid receptor isoforms in cultured lymphocytes from a patient with systemic glucocorticoid resistance and chronic lymphocytic leukemia. Biochem Biophys Res Commun. 1999;254(3):559–65.
Koga Y, Matsuzaki A, Suminoe A, Hattori H, Kanemitsu S, Hara T. Differential mRNA expression of glucocorticoid receptor alpha and beta is associated with glucocorticoid sensitivity of acute lymphoblastic leukemia in children. Pediatr Blood Cancer. 2005;45(2):121–7.
Haarman EG, Kaspers GJ, Pieters R, Rottier MM, Veerman AJ. Glucocorticoid receptor alpha, beta and gamma expression vs in vitro glucocorticod resistance in childhood leukemia. Leukemia. 2004;18(3):530–7.
Beger C, Gerdes K, Lauten M, Tissing WJ, Fernandez-Munoz I, Schrappe M, et al. Expression and structural analysis of glucocorticoid receptor isoform gamma in human leukaemia cells using an isoform-specific real-time polymerase chain reaction approach. Br J Haematol. 2003;122(2):245–52.
Grad I, Picard D. The glucocorticoid responses are shaped by molecular chaperones. Mol Cell Endocrinol. 2007;275(1–2):2–12.
Lauten M, Beger C, Gerdes K, Asgedom G, Kardinal C, Welte K, et al. Expression of heat-shock protein 90 in glucocorticoid-sensitive and -resistant childhood acute lymphoblastic leukaemia. Leukemia. 2003;17(8):1551–6.
Tissing WJ, Meijerink JP, den Boer ML, Brinkhof B, Pieters R. mRNA expression levels of (co)chaperone molecules of the glucocorticoid receptor are not involved in glucocorticoid resistance in pediatric ALL. Leukemia. 2005;19(5):727–33.
John S, Sabo PJ, Johnson TA, Sung MH, Biddie SC, Lightman SL, et al. Interaction of the glucocorticoid receptor with the chromatin landscape. Mol Cell. 2008;29(5):611–24.
Pottier N, Yang W, Assem M, Panetta JC, Pei D, Paugh SW, et al. The SWI/SNF chromatin-remodeling complex and glucocorticoid resistance in acute lymphoblastic leukemia. J Natl Cancer Inst. 2008;100(24):1792–803.
Hogan LE, Meyer JA, Yang J, Wang J, Wong N, Yang W, et al. Integrated genomic analysis of relapsed childhood acute lymphoblastic leukemia reveals therapeutic strategies. Blood. 2011;118(19):5218–26.
Bhatla T, Wang J, Morrison DJ, Raetz EA, Burke MJ, Brown P, et al. Epigenetic reprogramming reverses the relapse-specific gene expression signature and restores chemosensitivity in childhood B-lymphoblastic leukemia. Blood. 2012;119(22):5201–10.
Gruhn B, Naumann T, Gruner D, Walther M, Wittig S, Becker S, et al. The expression of histone deacetylase 4 is associated with prednisone poor-response in childhood acute lymphoblastic leukemia. Leuk Res. 2013;37(10):1200–7.
Jones CL, Bhatla T, Blum R, Wang J, Paugh SW, Wen X, et al. Loss of TBL1XR1 disrupts glucocorticoid receptor recruitment to chromatin and results in glucocorticoid resistance in a B-lymphoblastic leukemia model. J Biol Chem. 2014;289(30):20502–15.
Liu Y, Easton J, Shao Y, Maciaszek J, Wang Z, Wilkinson MR, et al. The genomic landscape of pediatric and young adult T-lineage acute lymphoblastic leukemia. Nat Genet. 2017;49(8):1211–8.
Zhang J, Mullighan CG, Harvey RC, Wu G, Chen X, Edmonson M, et al. Key pathways are frequently mutated in high-risk childhood acute lymphoblastic leukemia: a report from the Children’s Oncology Group. Blood. 2011;118(11):3080–7.
Moffitt AB, Dave SS. Clinical applications of the genomic landscape of aggressive non-hodgkin lymphoma. J Clin Oncol. 2017;35(9):955–62.
Revollo JR, Cidlowski JA. Mechanisms generating diversity in glucocorticoid receptor signaling. Ann NY Acad Sci. 2009;1179:167–78.
Raker VK, Becker C, Steinbrink K. The cAMP pathway as therapeutic target in autoimmune and inflammatory diseases. Front Immunol. 2016;7:123.
Gruol DJ, Campbell NF, Bourgeois S. Cyclic AMP-dependent protein kinase promotes glucocorticoid receptor function. J Biol Chem. 1986;261(11):4909–14.
Medh RD, Saeed MF, Johnson BH, Thompson EB. Resistance of human leukemic CEM-C1 cells is overcome by synergism between glucocorticoid and protein kinase A pathways: correlation with c-Myc suppression. Cancer Res. 1998;58(16):3684–93.
Zhang L, Insel PA. The pro-apoptotic protein Bim is a convergence point for cAMP/protein kinase A- and glucocorticoid-promoted apoptosis of lymphoid cells. J Biol Chem. 2004;279(20):20858–65.
Dong H, Carlton ME, Lerner A, Epstein PM. Effect of cAMP signaling on expression of glucocorticoid receptor, Bim and Bad in glucocorticoid-sensitive and resistant leukemic and multiple myeloma cells. Front Pharmacol. 2015;6:230.
Lerner A, Epstein PM. Cyclic nucleotide phosphodiesterases as targets for treatment of haematological malignancies. Biochem J. 2006;393(Pt 1):21–41.
Ogawa R, Streiff MB, Bugayenko A, Kato GJ. Inhibition of PDE4 phosphodiesterase activity induces growth suppression, apoptosis, glucocorticoid sensitivity, p53, and p21(WAF1/CIP1) proteins in human acute lymphoblastic leukemia cells. Blood. 2002;99(9):3390–7.
Tiwari S, Dong H, Kim EJ, Weintraub L, Epstein PM, Lerner A. Type 4 cAMP phosphodiesterase (PDE4) inhibitors augment glucocorticoid-mediated apoptosis in B cell chronic lymphocytic leukemia (B-CLL) in the absence of exogenous adenylyl cyclase stimulation. Biochem Pharmacol. 2005;69(3):473–83.
Meyers JA, Taverna J, Chaves J, Makkinje A, Lerner A. Phosphodiesterase 4 inhibitors augment levels of glucocorticoid receptor in B cell chronic lymphocytic leukemia but not in normal circulating hematopoietic cells. Clin Cancer Res. 2007;13(16):4920–7.
Kim SW, Rai D, Aguiar RC. Gene set enrichment analysis unveils the mechanism for the phosphodiesterase 4B control of glucocorticoid response in B-cell lymphoma. Clin Cancer Res. 2011;17(21):6723–32.
Shipp MA, Ross KN, Tamayo P, Weng AP, Kutok JL, Aguiar RC, et al. Diffuse large B-cell lymphoma outcome prediction by gene-expression profiling and supervised machine learning. Nat Med. 2002;8(1):68–74.
Pearson G, Robinson F, Beers Gibson T, Xu BE, Karandikar M, Berman K, et al. Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev. 2001;22(2):153–83.
Tanaka T, Okabe T, Gondo S, Fukuda M, Yamamoto M, Umemura T, et al. Modification of glucocorticoid sensitivity by MAP kinase signaling pathways in glucocorticoid-induced T-cell apoptosis. Exp Hematol. 2006;34(11):1542–52.
Miller AL, Webb MS, Copik AJ, Wang Y, Johnson BH, Kumar R, et al. p38 Mitogen-activated protein kinase (MAPK) is a key mediator in glucocorticoid-induced apoptosis of lymphoid cells: correlation between p38 MAPK activation and site-specific phosphorylation of the human glucocorticoid receptor at serine 211. Mol Endocrinol. 2005;19(6):1569–83.
Lu J, Quearry B, Harada H. p38-MAP kinase activation followed by BIM induction is essential for glucocorticoid-induced apoptosis in lymphoblastic leukemia cells. FEBS Lett. 2006;580(14):3539–44.
Harada H, Quearry B, Ruiz-Vela A, Korsmeyer SJ. Survival factor-induced extracellular signal-regulated kinase phosphorylates BIM, inhibiting its association with BAX and proapoptotic activity. Proc Natl Acad Sci USA. 2004;101(43):15313–7.
Rambal AA, Panaguiton ZL, Kramer L, Grant S, Harada H. MEK inhibitors potentiate dexamethasone lethality in acute lymphoblastic leukemia cells through the pro-apoptotic molecule BIM. Leukemia. 2009;23(10):1744–54.
Rogatsky I, Logan SK, Garabedian MJ. Antagonism of glucocorticoid receptor transcriptional activation by the c-Jun N-terminal kinase. Proc Natl Acad Sci USA. 1998;95(5):2050–5.
Jones CL, Gearheart CM, Fosmire S, Delgado-Martin C, Evensen NA, Bride K, et al. MAPK signaling cascades mediate distinct glucocorticoid resistance mechanisms in pediatric leukemia. Blood. 2015;126(19):2202–12.
Burotto M, Chiou VL, Lee JM, Kohn EC. The MAPK pathway across different malignancies: a new perspective. Cancer. 2014;120(22):3446–56.
Morishita N, Tsukahara H, Chayama K, Ishida T, Washio K, Miyamura T, et al. Activation of Akt is associated with poor prognosis and chemotherapeutic resistance in pediatric B-precursor acute lymphoblastic leukemia. Pediatr Blood Cancer. 2012;59(1):83–9.
Piovan E, Yu J, Tosello V, Herranz D, Ambesi-Impiombato A, Da Silva AC, et al. Direct reversal of glucocorticoid resistance by AKT inhibition in acute lymphoblastic leukemia. Cancer Cell. 2013;24(6):766–76.
Kino T, Souvatzoglou E, De Martino MU, Tsopanomihalu M, Wan Y, Chrousos GP. Protein 14-3-3sigma interacts with and favors cytoplasmic subcellular localization of the glucocorticoid receptor, acting as a negative regulator of the glucocorticoid signaling pathway. J Biol Chem. 2003;278(28):25651–6.
Habib T, Sadoun A, Nader N, Suzuki S, Liu W, Jithesh PV, et al. AKT1 has dual actions on the glucocorticoid receptor by cooperating with 14-3-3. Mol Cell Endocrinol. 2017;439:431–43.
Silveira AB, Laranjeira AB, Rodrigues GO, Leal PC, Cardoso BA, Barata JT, et al. PI3K inhibition synergizes with glucocorticoids but antagonizes with methotrexate in T-cell acute lymphoblastic leukemia. Oncotarget. 2015;6(15):13105–18.
Evangelisti C, Cappellini A, Oliveira M, Fragoso R, Barata JT, Bertaina A, et al. Phosphatidylinositol 3-kinase inhibition potentiates glucocorticoid response in B-cell acute lymphoblastic leukemia. J Cell Physiol. 2018;233(3):1796–811.
Hay N, Sonenberg N. Upstream and downstream of mTOR. Genes Dev. 2004;18(16):1926–45.
Wei G, Twomey D, Lamb J, Schlis K, Agarwal J, Stam RW, et al. Gene expression-based chemical genomics identifies rapamycin as a modulator of MCL1 and glucocorticoid resistance. Cancer Cell. 2006;10(4):331–42.
Gu L, Zhou C, Liu H, Gao J, Li Q, Mu D, et al. Rapamycin sensitizes T-ALL cells to dexamethasone-induced apoptosis. J Exp Clin Cancer Res. 2010;29:150.
Batista A, Barata JT, Raderschall E, Sallan SE, Carlesso N, Nadler LM, et al. Targeting of active mTOR inhibits primary leukemia T cells and synergizes with cytotoxic drugs and signaling inhibitors. Exp Hematol. 2011;39(4):457–72.. e3
Zhang C, Ryu YK, Chen TZ, Hall CP, Webster DR, Kang MH. Synergistic activity of rapamycin and dexamethasone in vitro and in vivo in acute lymphoblastic leukemia via cell-cycle arrest and apoptosis. Leuk Res. 2012;36(3):342–9.
Schult C, Dahlhaus M, Glass A, Fischer K, Lange S, Freund M, et al. The dual kinase inhibitor NVP-BEZ235 in combination with cytotoxic drugs exerts anti-proliferative activity towards acute lymphoblastic leukemia cells. Anticancer Res. 2012;32(2):463–74.
Hall CP, Reynolds CP, Kang MH. Modulation of glucocorticoid resistance in pediatric T-cell acute lymphoblastic leukemia by increasing BIM expression with the PI3K/mTOR inhibitor BEZ235. Clin Cancer Res. 2016;22(3):621–32.
Murray PJ. The JAK-STAT signaling pathway: input and output integration. J Immunol. 2007;178(5):2623–9.
Roberts KG, Morin RD, Zhang J, Hirst M, Zhao Y, Su X, et al. Genetic alterations activating kinase and cytokine receptor signaling in high-risk acute lymphoblastic leukemia. Cancer Cell. 2012;22(2):153–66.
Stocklin E, Wissler M, Gouilleux F, Groner B. Functional interactions between Stat5 and the glucocorticoid receptor. Nature. 1996;383(6602):726–8.
Wu SC, Li LS, Kopp N, Montero J, Chapuy B, Yoda A, et al. Activity of the type II JAK2 inhibitor CHZ868 in B cell acute lymphoblastic leukemia. Cancer Cell. 2015;28(1):29–41.
Delgado-Martin C, Meyer LK, Huang BJ, Shimano KA, Zinter MS, Nguyen JV, et al. JAK/STAT pathway inhibition overcomes IL7-induced glucocorticoid resistance in a subset of human T-cell acute lymphoblastic leukemias. Leukemia. 2017;31(12):2568–76.
Oppermann S, Lam AJ, Tung S, Shi Y, McCaw L, Wang G, et al. Janus and PI3-kinases mediate glucocorticoid resistance in activated chronic leukemia cells. Oncotarget. 2016;7(45):72608–21.
Tzoneva G, Ferrando AA. Recent advances on NOTCH signaling in T-ALL. Curr Top Microbiol Immunol. 2012;360:163–82.
De Keersmaecker K, Lahortiga I, Mentens N, Folens C, Van Neste L, Bekaert S, et al. In vitro validation of gamma-secretase inhibitors alone or in combination with other anti-cancer drugs for the treatment of T-cell acute lymphoblastic leukemia. Haematologica. 2008;93(4):533–42.
Real PJ, Tosello V, Palomero T, Castillo M, Hernando E, de Stanchina E, et al. Gamma-secretase inhibitors reverse glucocorticoid resistance in T cell acute lymphoblastic leukemia. Nat Med. 2009;15(1):50–8.
Samon JB, Castillo-Martin M, Hadler M, Ambesi-Impiobato A, Paietta E, Racevskis J, et al. Preclinical analysis of the gamma-secretase inhibitor PF-03084014 in combination with glucocorticoids in T-cell acute lymphoblastic leukemia. Mol Cancer Ther. 2012;11(7):1565–75.
Milano J, McKay J, Dagenais C, Foster-Brown L, Pognan F, Gadient R, et al. Modulation of notch processing by gamma-secretase inhibitors causes intestinal goblet cell metaplasia and induction of genes known to specify gut secretory lineage differentiation. Toxicol Sci. 2004;82(1):341–58.
Agnusdei V, Minuzzo S, Frasson C, Grassi A, Axelrod F, Satyal S, et al. Therapeutic antibody targeting of Notch1 in T-acute lymphoblastic leukemia xenografts. Leukemia. 2014;28(2):278–88.
Palacios EH, Weiss A. Function of the Src-family kinases, Lck and Fyn, in T-cell development and activation. Oncogene. 2004;23(48):7990–8000.
Serafin V, Capuzzo G, Milani G, Minuzzo SA, Pinazza M, Bortolozzi R, et al. Glucocorticoid resistance is reverted by LCK inhibition in pediatric T-cell acute lymphoblastic leukemia. Blood. 2017;130(25):2750–61.
Harr MW, Caimi PF, McColl KS, Zhong F, Patel SN, Barr PM, et al. Inhibition of Lck enhances glucocorticoid sensitivity and apoptosis in lymphoid cell lines and in chronic lymphocytic leukemia. Cell Death Differ. 2010;17(9):1381–91.
Seckl JR. 11beta-hydroxysteroid dehydrogenases: changing glucocorticoid action. Curr Opin Pharmacol. 2004;4(6):597–602.
Sai S, Nakagawa Y, Sakaguchi K, Okada S, Takahashi H, Hongo T, et al. Differential regulation of 11beta-hydroxysteroid dehydrogenase-1 by dexamethasone in glucocorticoid-sensitive and -resistant childhood lymphoblastic leukemia. Leuk Res. 2009;33(12):1696–8.
Sai S, Nakagawa Y, Yamaguchi R, Suzuki M, Sakaguchi K, Okada S, et al. Expression of 11beta-hydroxysteroid dehydrogenase 2 contributes to glucocorticoid resistance in lymphoblastic leukemia cells. Leuk Res. 2011;35(12):1644–8.
Tao Y, Gao L, Wu X, Wang H, Yang G, Zhan F, et al. Down-regulation of 11beta-hydroxysteroid dehydrogenase type 2 by bortezomib sensitizes Jurkat leukemia T cells against glucocorticoid-induced apoptosis. PLoS One. 2013;8(6):e67067.
Garbrecht MR, Schmidt TJ. Expression and regulation of 11- beta hydroxysteroid dehydrogenase type 2 enzyme activity in the glucocorticoid-sensitive CEM-C7 human leukemic cell line. ISRN Oncol. 2013;2013:245246.
Homma H, Maruyama H, Niitsu Y, Listowsky I. A subclass of glutathione S-transferases as intracellular high-capacity and high-affinity steroid-binding proteins. Biochem J. 1986;235(3):763–8.
Anderer G, Schrappe M, Brechlin AM, Lauten M, Muti P, Welte K, et al. Polymorphisms within glutathione S-transferase genes and initial response to glucocorticoids in childhood acute lymphoblastic leukaemia. Pharmacogenetics. 2000;10(8):715–26.
Beesley AH, Firth MJ, Ford J, Weller RE, Freitas JR, Perera KU, et al. Glucocorticoid resistance in T-lineage acute lymphoblastic leukaemia is associated with a proliferative metabolism. Br J Cancer. 2009;100(12):1926–36.
Samuels AL, Heng JY, Beesley AH, Kees UR. Bioenergetic modulation overcomes glucocorticoid resistance in T-lineage acute lymphoblastic leukaemia. Br J Haematol. 2014;165(1):57–66.
Holleman A, Cheok MH, den Boer ML, Yang W, Veerman AJ, Kazemier KM, et al. Gene-expression patterns in drug-resistant acute lymphoblastic leukemia cells and response to treatment. N Engl J Med. 2004;351(6):533–42.
Hulleman E, Kazemier KM, Holleman A, Vander Weele DJ, Rudin CM, Broekhuis MJ, et al. Inhibition of glycolysis modulates prednisolone resistance in acute lymphoblastic leukemia cells. Blood. 2009;113(9):2014–21.
Buentke E, Nordstrom A, Lin H, Bjorklund AC, Laane E, Harada M, et al. Glucocorticoid-induced cell death is mediated through reduced glucose metabolism in lymphoid leukemia cells. Blood Cancer J. 2011;1(7):e31.
Aries IM, Hansen BR, Koch T, van den Dungen R, Evans WE, Pieters R, et al. The synergism of MCL1 and glycolysis on pediatric acute lymphoblastic leukemia cell survival and prednisolone resistance. Haematologica. 2013;98(12):1905–11.
Pang YY, Wang T, Chen FY, Wu YL, Shao X, Xiao F, et al. Glycolytic inhibitor 2-deoxy-d-glucose suppresses cell proliferation and enhances methylprednisolone sensitivity in non-Hodgkin lymphoma cells through down-regulation of HIF-1alpha and c-MYC. Leuk Lymphoma. 2015;56(6):1821–30.
Chan LN, Chen Z, Braas D, Lee JW, Xiao G, Geng H, et al. Metabolic gatekeeper function of B-lymphoid transcription factors. Nature. 2017;542(7642):479–83.
Madden EA, Bishop EJ, Fiskin AM, Melnykovych G. Possible role of cholesterol in the susceptibility of a human acute lymphoblastic leukemia cell line to dexamethasone. Cancer Res. 1986;46(2):617–22.
Samuels AL, Beesley AH, Yadav BD, Papa RA, Sutton R, Anderson D, et al. A pre-clinical model of resistance to induction therapy in pediatric acute lymphoblastic leukemia. Blood Cancer J. 2014;4:e232.
Lawrie CH. MicroRNAs in hematological malignancies. Blood Rev. 2013;27(3):143–54.
Han BW, Feng DD, Li ZG, Luo XQ, Zhang H, Li XJ, et al. A set of miRNAs that involve in the pathways of drug resistance and leukemic stem-cell differentiation is associated with the risk of relapse and glucocorticoid response in childhood ALL. Hum Mol Genet. 2011;20(24):4903–15.
Hezaveh K, Kloetgen A, Bernhart SH, Mahapatra KD, Lenze D, Richter J, et al. Alterations of microRNA and microRNA-regulated messenger RNA expression in germinal center B-cell lymphomas determined by integrative sequencing analysis. Haematologica. 2016;101(11):1380–9.
Harada M, Pokrovskaja-Tamm K, Soderhall S, Heyman M, Grander D, Corcoran M. Involvement of miR17 pathway in glucocorticoid-induced cell death in pediatric acute lymphoblastic leukemia. Leuk Lymphoma. 2012;53(10):2041–50.
Li XJ, Luo XQ, Han BW, Duan FT, Wei PP, Chen YQ. MicroRNA-100/99a, deregulated in acute lymphoblastic leukaemia, suppress proliferation and promote apoptosis by regulating the FKBP51 and IGF1R/mTOR signalling pathways. Br J Cancer. 2013;109(8):2189–98.
Ledderose C, Mohnle P, Limbeck E, Schutz S, Weis F, Rink J, et al. Corticosteroid resistance in sepsis is influenced by microRNA-124—induced downregulation of glucocorticoid receptor-alpha. Crit Care Med. 2012;40(10):2745–53.
Liang YN, Tang YL, Ke ZY, Chen YQ, Luo XQ, Zhang H, et al. MiR-124 contributes to glucocorticoid resistance in acute lymphoblastic leukemia by promoting proliferation, inhibiting apoptosis and targeting the glucocorticoid receptor. J Steroid Biochem Mol Biol. 2017;172:62–8.
Kim J, Jeong D, Nam J, Aung TN, Gim JA, Park KU, et al. MicroRNA-124 regulates glucocorticoid sensitivity by targeting phosphodiesterase 4B in diffuse large B cell lymphoma. Gene. 2015;558(1):173–80.
Kotani A, Ha D, Hsieh J, Rao PK, Schotte D, den Boer ML, et al. miR-128b is a potent glucocorticoid sensitizer in MLL-AF4 acute lymphocytic leukemia cells and exerts cooperative effects with miR-221. Blood. 2009;114(19):4169–78.
Kotani A, Ha D, Schotte D, den Boer ML, Armstrong SA, Lodish HF. A novel mutation in the miR-128b gene reduces miRNA processing and leads to glucocorticoid resistance of MLL-AF4 acute lymphocytic leukemia cells. Cell Cycle. 2010;9(6):1037–42.
Huang B, Zhao J, Lei Z, Shen S, Li D, Shen GX, et al. miR-142-3p restricts cAMP production in CD4+CD25- T cells and CD4+CD25+ TREG cells by targeting AC9 mRNA. EMBO Rep. 2009;10(2):180–5.
Lv M, Zhang X, Jia H, Li D, Zhang B, Zhang H, et al. An oncogenic role of miR-142-3p in human T-cell acute lymphoblastic leukemia (T-ALL) by targeting glucocorticoid receptor-alpha and cAMP/PKA pathways. Leukemia. 2012;26(4):769–77.
Yang A, Ma J, Wu M, Qin W, Zhao B, Shi Y, et al. Aberrant microRNA-182 expression is associated with glucocorticoid resistance in lymphoblastic malignancies. Leuk Lymphoma. 2012;53(12):2465–73.
Chen P, Shen T, Wang H, Ke Z, Liang Y, Ouyang J, et al. MicroRNA-185-5p restores glucocorticoid sensitivity by suppressing the mammalian target of rapamycin complex (mTORC) signaling pathway to enhance glucocorticoid receptor autoregulation. Leuk Lymphoma. 2017:1–11.
Acknowledgements
L.K.M. is supported by the UCSF Medical Scientist Training Program Grant T32 GM007618 and by a Genentech Foundation Award. M.L.H is supported by the National Cancer Institute Grant R01 CA193776, The Campini Foundation, The Buster Posey Family Foundation, and The Pepp Family Foundation. The authors thank Kevin Shannon and Anica Wandler for their critical reading of the manuscript.
Disclosure of Conflict of Interest
No potential conflicts of interest were disclosed.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Meyer, L.K., Hermiston, M.L. (2019). Mechanisms of Glucocorticoid Response and Resistance in Lymphoid Malignancies. In: Xavier, A., Cairo, M. (eds) Resistance to Targeted Therapies in Lymphomas . Resistance to Targeted Anti-Cancer Therapeutics, vol 21. Springer, Cham. https://doi.org/10.1007/978-3-030-24424-8_1
Download citation
DOI: https://doi.org/10.1007/978-3-030-24424-8_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-24423-1
Online ISBN: 978-3-030-24424-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)