Abstract
Marine conservation biologists have identified mollusks as one of the appropriate surrogate taxa for characterizing marine benthic diversity. In turn, live/dead comparison studies have overwhelmingly demonstrated that mollusk remains are faithful proxies of the mollusk composition of the living communities from which they come, with positive consequences for the paleoecological evaluation of fossil assemblages. In this contribution, we evaluate the way in which mollusk biodiversity is distributed along the lower intertidal to supratidal (high water mark) dead shell assemblages accumulated on a northern Patagonian rocky shore, in order to explore the usefulness of these assemblages as paleontological proxies and potential surrogates of regional biodiversity . A diversity gradient from the lower intertidal to the supratidal was identified which is probably associated with vertical transport, although the influence of gradients of the living community should be tested to confirm this. The outstanding result of this study is the discovery of high levels of diversity among dead shells (31 bivalves and 39 gastropod species) in a single locality and with a moderate sampling effort. The supratidal death assemblage has higher species richness than expected, possibly caused by stranding of the fauna after storms. Nevertheless, this level shows the lowest level of evenness and a strong bias when samples are not sieved through a fine mesh. The record of marine benthic diversity in death assemblages is a promising area of research that deserves to be explored in depth.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Biological surrogates
- Conservation paleobiology
- Intertidal
- Death assemblage
- Rocky-bottom
- Patagonia
- Depth gradient
3.1 Introduction
Dead shells accumulated on the sea floor contain a wealth of information which is useful either for assessing important questions of the genesis of fossil deposits (taphonomy) or for studying living communities. In the search for evidence to determine how representative the fossil record is of communities that lived in the geological past, the discipline of taphonomy has developed tools which provide high-quality information of living ecosystems. This is achieved by allowing their features to be explored on longer timescales, beyond those typically used by ecologists (Kidwell and Tomašových 2017; Olszewski and Kidwell 2007; Tomašových and Kidwell 2009a; Archuby et al. 2015; De Francesco et al. 2013; Yanes et al. 2008; Hassan et al. 2018). Current developments go still further: it is now possible to identify the effect of human impact on ecosystems, by studying the differences between impacted living communities and time-averaged assemblages accumulated over the past decades or centuries (Erthal et al. 2011; Kidwell 2008; Yanes 2012; Dietl et al. 2015). The relevance of this paleobiological information, which offers us an otherwise inaccessible long-term perspective of biodiversity and community change, has given rise to a new discipline: conservation paleobiology (Barnosky et al. 2017; Louys 2012; Rick and Lockwood 2013; Dietl and Flessa 2011; Dietl et al. 2015; Kidwell 2009; Kidwell and Tomašových 2013).
Biodiversity is of fundamental importance to ecology because it is the consequence of how organisms in communities respond to biotic and abiotic factors (Olszewski and Kidwell 2007). The use of biological surrogates (i.e. estimators, such as polychaetes, crustaceans, mollusks , etc.) to evaluate marine biodiversity is a useful practice in conservation biology because it helps overcome the difficulties inherent in surveying benthic communities: time and cost, the occurrence of undescribed species and the problems of species identification (Tyler and Kowalewski 2017; Magierowski and Johnson 2006; Mellin et al. 2011; Warwick and Light 2002). Research focuses on finding appropriate surrogates for the different types of marine communities and their spatial and temporal variations. Mollusks, which are among the groups selected as appropriate surrogates of marine benthic communities, leave abundant mineralized dead remains, which have been proved to be good proxies of the communities from which they derive (Tyler and Kowalewski 2017; Kidwell 2008; Smith 2005).
Assessing how diversity transfers from living communities (life assemblages, LAs) to death assemblages (DAs) is a crucial step towards a better interpretation of diversity in fossil assemblages; this knowledge will also help us to evaluate death assemblages as faithful proxies of living communities. The step from LAs to DAs represents the first filter that modifies diversity measurements, through differential transport and destruction by waves, currents and wind and time-averaging (Archuby et al. 2015; Tomašových and Kidwell 2009a, 2010a).
In the absence of strong reworking of former beds, such as in the case of ravinement, marine beds encompass a short time span and their skeletal content is considered representative of the average composition of successions of communities along hundreds or, at the most, thousands of years (within-habitat time-averaging of Kidwell and Bosence 1991; see also Fürsich and Aberhan 1990). Recently, the quantitative knowledge of the differences between death assemblages and living communities, and the sources of these differences has been greatly improved (Olszewski and Kidwell 2007; Tomašových and Kidwell 2009a, b, 2010a, b, 2011; and many more).
3.2 Death Assemblages, Taxonomic Diversity, and Taphonomic Fidelity
Due to the time-averaged nature of DAs , their species composition is not particularly influenced by the short-term species composition fluctuations of living communities (Fürsich and Aberhan 1990; Tomašových and Kidwell 2010a; Archuby et al. 2015). These short-term fluctuations, such as the local extinction of the surf clam Mesodesma mactroides on the Atlantic coasts of Uruguay and Northern Argentina (Fiori and Cazzaniga 1999; Dadon 2005), might give totally different results in samples of living communities separated by only a few weeks. However, in this respect, DAs are highly informative due to their inertia in the face of such fluctuations. Compared with living assemblages, DAs which have accumulated over a few decades to several centuries are expected to have an increase in alpha diversity , a decrease in beta diversity (due to spatial mixing), reduced species dominance and increased frequency of rare species (Tomašových and Kidwell 2010a). Additionally, the ecological information of current ecosystems does not span more than a few decades into the past (Rick and Lockwood 2013). If we consider that human occupation of Patagonia dates from around 17,000–14,000 years BP (Perez et al. 2016), baseline ecological studies might fail to identify the non-impacted conditions when assessing anthropogenic influence, since the impacts were already there.
In turn, death assemblages are used to characterise not only the average species compositions of source communities, but also biotic interactions such as local level predator-prey relationships (e.g., Visaggi and Kelley 2007; Yanes and Tyler 2009; Gordillo and Archuby 2012, 2014; Martinelli et al. 2013; Tyler et al. 2014; Archuby and Gordillo 2018), and to compare these along geographical gradients (e.g., Kelley and Hansen 2007; Visaggi and Kelley 2015; Martinelli et al. 2013). Quantifying predator-prey interactions in living communities implies sampling strategies that are complex and expensive, while the records from death assemblages are a significant source of information.
Studies on taphonomic fidelity (correlation of living and death assemblages ) have been developed in marine, freshwater and terrestrial environments (e.g., Fürsich and Flessa 1987; Kidwell and Bosence 1991; Yanes et al. 2008; Tietze and De Francesco 2012; Terry 2010; more references in Archuby et al. 2015). Studies of marine death assemblages are abundant, although they are mostly based on soft-bottom ecosystems (Olszewski and Kidwell 2007; Kidwell 2013), and there are few studies of communities inhabiting rocky bottoms (Zuschin et al. 2000; Zuschin and Oliver 2003; Zuschin and Stachowitsch 2007). Recently, Archuby et al. (2015) assessed the taphonomic fidelity of rocky -bottom communities along 1500 km of the Patagonian Atlantic coast, from death assemblages collected at the high-water mark. These authors found a general agreement between life and death assemblages at the biogeographical province level, working with non-sieved, representative samples (hand-picked along transects). Besides the regional agreement, on smaller geographical scales DAs tended to cluster together and separated from LAs . So far, there are no detailed studies on the nature of DAs on rocky shores. A better understanding of the provenance of the diversity differences between life and death assemblages in modern environments is also crucial for correctly interpreting fossil assemblages and ecosystems (Olszewski and Kidwell 2007).
3.3 Purpose of This Study
In this study, we evaluate the way in which species richness and evenness of mollusk death assemblages is distributed along the depth gradient, from the lower intertidal to accumulations at the high-water mark, in Punta Mejillón, Northern Patagonia , Argentina . Our two goals are to improve the understanding of DAs as paleontological proxies and to evaluate their usefulness as surrogates of shallow benthic living communities. Punta Mejillón has little human impact due to its distance from the nearest city (the town of San Antonio Oeste, 105 km away), the difficulty getting there (sand dunes often cover the route), the need for a four-wheel drive vehicle to reach the beach, and also because it is located in a natural protected area (see The Study Area, below). We aim to determine whether the DAs coming from the same habitat but accumulated at different depths include specimens of different species in different proportions (i.e., there is diversity partitioning of DAs along the gradient). We also test the effect of sieving versus non sieving on species richness and evenness. Specifically, we aim to answer the following questions: (i) How does DA species composition vary along the lower intertidal to supratidal gradient? Is there diversity partitioning along the depth gradient in the rocky intertidal belt of northern Patagonia ? (ii) Are death assemblages from rocky shores appropriate surrogates of benthic biodiversity in northern Patagonian shallow marine communities? Is there a horizon along the lower intertidal to supratidal belts that collects most of the information on the death assemblages ? In other words, where is it best to sample? (iii) What is the effect of sieving on the biodiversity record?
3.4 Methodology
3.4.1 The Study Area
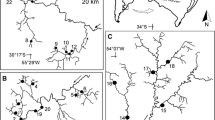
The study was carried out in Punta Mejillón (PM), located in the Caleta de los Loros natural protected area in Río Negro Province, Argentine Patagonia . The place is difficult to access, which minimizes the impact of tourism and human activities on living communities and death assemblages (Fig. 3.1). Punta Mejillón is on the Atlantic coast (41° 00′ 37″) in the San Matías Gulf. The coastline runs approximately from SE to NW, and the intertidal belt is exposed for more than 300 m during low tides (Fig. 3.2). Biogeographically, PM is in the transition zone between the Argentine and Magellanic Provinces and is characterized by a mixture of species from both biogeographical entities (Balech and Ehrlich 2008). In a recent article, Güller and Zelaya (2017) mention a surprisingly high level of mollusk diversity in the San Matías Gulf, which they describe as a hot-spot of diversity.
The northern part of San Matías Gulf, where Punta Mejillón is located, is subject to high levels of physical disturbance, consisting of strong winds, high tidal amplitudes (up to around 9 m) which leave large areas of the intertidal belt exposed, high-energy flows during high tide and low temperatures (sea surface temperatures 10.1–18.9 °C) (Bertness et al. 2006; Archuby et al. 2015). Due to the high levels of desiccation stress caused by winds, the region is considered an extremely harsh intertidal rocky ecosystem (Bertness et al. 2006), which results in intertidal communities which are strongly organized by physical stress.
3.4.2 Sampling
Sampling was carried out on 29 November 2013 during low tide, between latitudes S 41° 00′ 32″ and 41° 00′ 54″. Samples were collected at four levels: 1. accumulation of shells at the high-water mark (supratidal or “Supra”); 2. upper intertidal belt (UI); 3. middle intertidal belt (MI); and 4. lower intertidal belt (LI). At each level, several replicates were extracted from the upper 15 cm using a shovel and were pooled together, until completing 15 L of sediment. The replicates were extracted every 10 m along a transect parallel to the coastline. Since the substrate is mostly hard, samples were taken from depressions filled with sediments in the area surrounding the sampling point. In the absence of a suitable place to extract the replicate, the point was skipped, and the sample was taken at the next point. Samples were sieved in the field with a 10 × 10 mm aperture mesh (coarse) above and a 1 mm × 1 mm aperture mesh (fine) below so that large shells were captured separately from small shells (Fig. 3.3). The coarse mesh sieve retains shells that are visible and was considered as a proxy “hand-collecting method”, that was compared with “whole” samples per level (made by the pooling of coarse and fine samples). The 1 mm sieve was used to explore a suitable sampling strategy for rocky -bottom dominated intertidal DAs from the Patagonian Atlantic coast. Kidwell (2002) suggested that sampling with mesh sizes lower than 1 mm might collect a non-representative high amount of larvae and juveniles.
All gastropod and bivalve shells and shell fragments were analyzed and identified to the species level with some exceptions that were unidentifiable due to preservation issues. Other skeletal elements not included in the study were: crab fragments, serpulid tubes, abundant cirriped plates, sea-urchin spines, fragments of bryozoan colonies, oyster recruits on large valves and polyplacophoran plates. Cirripeds and cirriped plates, although very abundant, were excluded from analysis due to the difficulty in identifying the plates. Gastropod shells and articulated bivalves were assigned one count. Left and right valves of bivalve species were counted separately. The count per species resulted from the sum of articulated specimens plus the most abundant valves (left or right). Ostrea puelchana and Pododesmus rudis shells that could not be identified were counted together and divided by 2, and then assigned to the respective species. Bivalve fragments were counted if the umbo and at least one-third of the valve were preserved (very small fragments were discarded). Gastropod fragments were counted when they contained the apex and at least half of the shell.
3.4.3 Statistical Methods
Counts were made per level (LI, MI, UI, and Supra). The coarse mesh size fraction of samples was also registered separately for each level. Diversity was estimated using different indices: species richness (S, the raw number of species and by rarefaction), the Shannon-Wiener (H’) index, the equitability J index (Hammer and Harper 2006) and the probability of an interspecific encounter (PIE), an evenness index (Hurlbert 1971). Rarefaction to the lowest sample size was calculated in order to evaluate species richness without the effect of sample size. The H’ index summarises information on species richness and evenness and correlates with S and sample size, as does the J index. The PIE index was added to obtain an estimation of evenness which was not affected by sample size (Olszewski and Kidwell 2007). Data management and calculation of the PIE index according to Hurlbert’s formula was carried out using standard spreadsheet software. Other diversity indices were calculated using PAST v 3.15 (Hammer et al. 2001).
Samples (levels) were plotted using a non-metric multidimensional scaling (NMDS) ordination analysis to evaluate their similarity. The database was first transformed to percentages per sample, then square root transformed, and then a similarity matrix was calculated based on the Bray-Curtis index (Clarke 1993; Clarke and Warwick 2001; Clarke et al. 2006). NMDS was carried out using R software, version 3.4.3 (R Core Team 2017).
To test the effect of using samples sieved with coarse mesh (as proxies for collecting by hand), we compared these with the results obtained for whole samples (coarse + fine mesh) by using diversity indices and an ordination plot (NMDS).
Beta diversity was quantified in order to assess both the existence of a gradient along the coastal profile for the four levels (directional turnover) and non-directional variation for comparing the coarse mesh subsample with the whole sample (whole = coarse plus fine mesh subsamples) (Anderson et al. 2011). To evaluate the gradient in beta diversity , the similarity between the supratidal sample and the samples from all other levels was calculated with the Jaccard similarity index on a presence/absence matrix. The results were plotted in their position on the coastal profile, from Supra to LI. If species turnover along the gradient existed, then a pattern of similarity decrease would be expected from left to right. To determine the differences in species presence/absence in coarse and fine samples, Whittaker’s beta diversity index (βw) was calculated between pairs of coarse and whole samples per level, and then plotted in their position on the coastal profile. Higher levels of βw imply a greater mismatch between the coarse mesh samples and the whole samples (Koleff et al. 2003).
3.5 Results
A total of 12,790 mollusk specimens belonging to 31 bivalve and 39 gastropod species were collected (Table 3.1, Supp Appendix A and B ). The sample size was uneven between levels due to the variable densities of shells in sediments from the different samples (Fig. 3.4a). Lower and middle intertidal samples contained less than half the specimens of the upper and supratidal samples. The coarse fraction per level fluctuated between 17 and 38% (Supra and MI respectively. Figure 3.4b). MI, with the smallest sample size (1520 specimens), has the highest percentage of the coarse fraction (38%).
3.5.1 Alpha and Beta Diversities Across the Intertidal Gradient
The 70 species identified in this study are distributed differently across samples (Tables 3.1 and 3.2). The S index is highest in Supra, followed by LI, UI, and MI. However, when standardizing to n = 1520 by rarefaction, the highest diversity is found in LI (46), followed by Supra, MI and UI, which have between 36 and 39 species (Fig. 3.5a, b). The rarefaction curves show that none of the samples have a stabilizing size pattern (Fig. 3.5c), suggesting that larger sample sizes are necessary to accurately document the kind of study.
a Plot of species richness (S) per level. The bar represents a bootstrap 95% confidence interval. b Rarefaction species richness to n = 1520 per sample. The bar includes 2 standard errors. c Rarefaction curves per level with 95% bootstrap confidence interval. Species richness on the y axis; sample size on the x axis
Evenness differs between levels, and is consistently lowest in Supra, followed by UI, and then LI and MI with higher values (Table 3.2 and Fig. 3.6a, b and c). The H’ index is highest for LI, while the J index has MI as the evenest sample. The PIE index, which is more reliable for studies with different sample sizes, is highest for LI, followed by MI, UI, and Supra, coinciding with the H’ index.
Multivariate ordination using an NMDS plot indicates a similarity between LI and MI, while UI and Supra remain separate (Fig. 3.7a). The analysis of beta diversity allows the identification of a pattern of decrease along the supratidal to the lower intertidal gradient (Fig. 3.9a).
3.5.2 Effect of Mesh Aperture Size
The samples sieved with coarse mesh have richness and equitability values which are lower than estimations for whole (coarse + fine) samples (Table 3.2 and 3.3). Coarse mesh samples consistently underestimate the species richness of the death assemblage (Fig. 3.8a), and are less even for all four levels (PIE index, Fig. 3.8b). On the NMDS ordination plot, coarse mesh samples cluster together and are separate from the whole samples (Fig. 3.7b). The comparison of Whittaker’s beta diversity indices (βw) shows a large mismatch between the coarse mesh and whole samples at the supratidal level (Fig. 3.9b), suggesting that at this level the species composition of the coarse sample is the least similar to the whole sample.
3.6 Discussion
3.6.1 Alpha and Beta Diversity Trends
There is a general trend in decreasing diversity from the lower intertidal to the supratidal belt, both for species richness and evenness. The pattern is more evident in estimations not dependant on sample size (Figs. 3.5b and 3.6c) than in measurements associated with sample size (Figs. 3.5a and 3.6a, b). In the case of species richness, its estimation via rarefaction to the lowest sample size (n = 1520) suggests a trend from LI to UI, although the Supra sample is slightly more diverse than MI and UI (Fig. 3.5b). The PIE index shows a decrease in evenness from LI to Supra, and a similar situation can be observed in the J and H’ indices, despite the effect of sample size (Fig. 3.8a, b and c). NMDS ordination does not reflect a clear pattern. However, the values of Bray-Curtis similarity indices between levels follow the LI to Supra gradient (Table 3.4): contiguous samples are more similar to one another than non-contiguous ones. Distance between samples was coded as 1 to 3 (1 for contiguous samples, 2 for LI to UI and Mi to Supra; and 3 for LI to Supra), and the Spearman rank correlation index was calculated between the Bray-Curtis index and distance, thereby obtaining a significant negative correlation of −0.93 (p = 0.033). Additional evidence of the influence of the depth gradient in the species composition of samples comes from the evaluation of beta diversity : compared with the Supra level, there is a decrease in similarity from UI to LI. This is interpreted as a consequence of the depth gradient, whether due to a taphonomic gradient explained by biostratinomic factors (transport by waves and wind, selective destruction), the species composition gradient of the living community, or both. In turn, harsh environmental conditions (high levels of desiccation, strong winds, and wave energy) suggest that the upper intertidal , and particularly the supratidal , belts should have poorer living community diversity; however, this is not seen in the death assemblages , thus inferring vertical transport from the middle and lower intertidal and shallow subtidal. The higher than expected richness in the Supra sample might be a consequence of the trapping (stranding) of shells above the high water mark during energetic storms; shells and live specimens are stranded above the high water mark, and are no longer reached by usual or normal storm waves (López et al. 2008).
Finally, Bray-Curtis similarity was calculated to compare each level with the whole sample (pooled), and it was found that the Supra and UI levels are the most similar to the total sample (Table 3.5). Although in every case abundances were standardized to percentages and square root transformed, the most abundant samples, Supra and UI, might still influence the result and cause this similarity. The best sampling strategy would still be to collect material from every level, but sampling death assemblages accumulated on the high water mark (Supra) level would not lead to important biases. However, it must be considered that the Supra level has the highest bias when fine meshes are not used, as seen below.
Whether diversity along the lower intertidal to supratidal areas of this study follows a gradient of a biological (species composition of the living community), taphonomic (differential transport, destruction and shell production among species) or mixed nature, will hopefully be answered in future studies. As for soft-bottom studies, there is still a need for more actualistic updated research (Olszewski and Kidwell 2007; Kidwell 2015; Tyler and Kowalewski 2017). Our investigation is particularly relevant because it helps to fill the need for studies of this kind on hard-bottom environments (Smith 2008; Archuby et al. 2015).
3.6.2 Mesh Size Matters: The Effect of Sieving on Biodiversity Assessment
There are relevant differences between the coarse fraction and the whole sample (whole samples are composed of coarse and fine mesh samples. For an explanation, see Sampling in Methodology). Coarse samples are less even (Fig. 3.8b) and have lower species richness (as expected, since “coarse” samples are part of the “whole” samples of each level; Fig. 3.8a). Species richness in coarse samples ranges from 80 to 62% of the values obtained for the whole samples, with equivalent sampling efforts. The differences observed are also reflected in the ordination plot, in which all coarse samples are clustered together and separated from whole samples (NMDS, Fig. 3.7b). Discordance in terms of beta diversity is more marked in Supra than in the rest of the samples. This means that in Supra, the mismatch between coarse and whole samples is highest. This is still more relevant if we consider that many minute species, such as Phlyctiderma semiaspera, Turbonilla macaensis or Anachis isabellei, are present in the coarse samples because they were stuck to mytilid byssus, which would not otherwise have been sampled by hand. Without these queue-jumper species, the biases of coarse samples would have been even larger. Olszewski and Kidwell (2007) detected that the evenness of death assemblages is more similar to live assemblages when samples are sieved with a mesh size finer than 2 mm, which was also the case for species richness. A positive bias in evenness and species richness in coarse mesh samples with respect to fine mesh samples (which are in turn more similar to live assemblages), can be caused by the greater durability of large mollusk shells, lower temporal volatility of adult specimens in living communities, or both (Kidwell 2002; Olszewski and Kidwell 2007).
The positive effect of sieving with fine meshes in diversity studies based on DAs is supported by evidence from living communities. In a compilation of mollusk diversity (including bivalves , gastropods , polyplacophorans, scaphopods and cephalopods) in the San Matías and San José gulfs, Güller and Zelaya (2017) noted that out of the total 196 species described for the whole area, 61 (31.1%) have a maximum size smaller than 10 mm. As a consequence, almost one-third of the species in the assemblage have a lower probability of being collected, and would perhaps be neglected if fine mesh size had not been used. The most abundant gastropod and bivalve species found by these authors, respectively Parvanachis isabellei and Crenella divaricata , fit this condition.
3.6.3 Sampling Issues: Features of Death Assemblages Along the Intertidal Belt
Dead shell assemblages differ from living communities due to time-averaging and differential transport and destruction (Kidwell 2001). The effect of tidal regime together with the action of waves on the bottom differ in intensity along the intertidal belt. As a consequence of these differences in the intensity of transport, destruction, and sorting of shells, some variation might be expected in shell density, bioclast size, and vertical transport, which would affect the results of sampling. Shell density varies along the intertidal belt, as evidenced in the individuals counted per sample (Fig. 3.4a and Table 3.1). The rarefaction curves indicated that none of the samples were large enough to be representative of species diversity, so more sampling is therefore needed, especially from the lower and middle intertidal levels. The proportion of coarse to fine mesh shells varies little; the exception is MI, but the fact that this level has the smallest sample size could explain the difference (Fig. 3.4b). Strong vertical transport of shells in DAs is also evident since the species composition of all the samples includes at least some taxa that are characteristic of subtidal to lower intertidal belts (e.g. Aulacomya atra , Venus antiqua , some Buccinanops species, etc.). On the other hand, these rocky Patagonian shores have little diversity of living fauna in the upper intertidal belt, and almost no marine life higher up, in the supratidal fringe (Bertness et al. 2006 and personal observations).
3.6.4 Mollusk Shell Dead Shell Assemblages as Samples of Living Marine Biodiversity
Two theoretical frameworks have come together in this study: 1. conservation biology has supplied information on the use of biological surrogates of marine benthic communities (Magierowski and Johnson 2006; Smith 2005, 2008; Mellin et al. 2011; Tyler and Kowalewski 2017); and 2. taphonomy, and the new, related discipline conservation paleobiology, have contributed with the assessment of the processes that operate between living communities and accumulations of their remains, for those taxa that bear mineralized or highly durable tissues (Kidwell 2001; Tomašových and Kidwell 2009a; Dietl and Flessa 2011). Put simply: if a particular taxon is an appropriate surrogate for a living community, and the accumulation of its durable remains or death assemblages are good proxies of the living counterpart of the taxon, then the death assemblages are highly valuable tools as rapid and faithful proxies of the living communities.
Different studies coincide on the point that exhaustively sampling living marine diversity is almost impossible, very expensive and particularly time-consuming, mainly due to the difficulties involved in accessing study sites, poor taxonomic knowledge and the high diversity of marine communities (Warwick and Light 2002; Magierowski and Johnson 2006; Smith 2008; Mellin et al. 2011; Tyler and Kowalewski 2017). Besides, results show that at least in some cases (depending on habitat type and spatial scale), mollusks are appropriate surrogates of marine communities (e.g., Smith 2008; Tyler and Kowalewski 2017). Dead shell assemblages represent time-averaged relics of the communities they come from. Their differences from living assemblages are explained mainly by their time-averaged nature: they are composed of a mixture of successive communities that lived in the same area and are modified by vertical and lateral transport and other biostratinomic agents (Kidwell 2001, 2013; Archuby et al. 2015). One of the expectations with respect to the features of death assemblages is an increase in species richness and evenness (Tomašových and Kidwell 2010a). Olszewski and Kidwell (2007) demonstrated that on average death assemblages surpass living communities in species richness and evenness, although particular examples might have a different pattern (with little frequency). The only study of live/dead comparisons along the Patagonian Atlantic coast detected a systematic increase in both diversity measurements in every single comparison (Archuby et al. 2015), even when sediments were not sieved (just hand collected) and live and dead samples did not coincide in time or extent (living communities were only sampled from the middle intertidal a few years before the collection of death assemblages ).
In order to evaluate to what degree our death assemblages can provide relevant information on regional biodiversity , we compared them with data from surveys of benthic communities. Relevant studies on living mollusk diversity in the San Matías and San José gulfs are summarised in Table 3.6, including species richness (discriminating between bivalves and gastropods ), the nature of the sample (life or death assemblage ), the extent of the sampling area, the sampling effort (in terms of cost and time of the sampling process) and, when available, sample size. Archuby et al. (2015) studied a series of live and dead mollusk assemblages, spanning 1500 km from Punta Mejillón to Puerto Deseado in the South of Patagonia . Their case studies from the San Matías Gulf were numbered 2, 4, 5 (LAs) , 6 and 7 (DAs) . Avaca et al. (2008) provided two databases of living communities, sampled in the North and Northwest margins of the San Matías Gulf. Case study 9 corresponds to an extensive study in the San José Gulf and the closest part of the San Matías Gulf (Zaixso et al. 1998). Recently, Güller and Zelaya (2017) published a highly-qualified study with information on mollusk species from the San Matías and San José gulfs, which includes results from their own samples and from an exhaustive bibliographic compilation. The authors used 85 sampling points from the intertidal to a depth of 25 m in the subtidal, and also took some (not detailed) samples from deeper bottoms, obtaining a total of 30,481 mollusk specimens, including empty shells and valves. Their database, and most of their study focused on four main areas which together account for 119 species of bivalves (49) and gastropods (70) for both gulfs (when considering only live species found in their samples). When other studies are added, the species count for the whole area reaches 141 species (60 bivalves and 81 gastropods ).
Death assemblages offer relatively high levels of diversity compared to life assemblages (Fig. 3.10). Individual (single place) samples of living communities such as 2, 4 and 5 represent low effort sampling but with very little diversity (up to 6 species). Several-point samples of living communities demand high levels of effort (availability of vessels, complex sampling devices, diving). Case studies 3 and 8 are samples of living communities with 122 and 32 sampling points respectively, where only 21 (case study 3) and 23 (case study 8) species were collected. In these two cases, the sampling area was considered of medium size (narrow fringes parallel to the coast). Case study 9 (Zaixso et al. 1998) is a 120 sample point survey in which a species richness of 61 was collected. Case study 12, taken from Güller and Zelaya (2017), is based on Zaxso’s data plus additional information. Case studies 10, 11 and 13 are based on multi-point sampling of living communities across wide areas of the San Matías Gulf. Case study 14 represents a synthesis of multipoint sampling and all available published information on mollusk diversity in the San Matías and San José gulfs. Güller and Zelaya (2017) and Zaixso et al. (1998) sieved their samples with less than 2 mm aperture meshes, while Avaca et al. (2008) used 40 mm aperture nets. According to Güller and Zelaya (2017), sampling without fine meshes drastically reduces diversity, since they detected that more than 45% of species have shells with a maximum size smaller than 15 mm. This difference in sampling strategy might have caused the reduced diversity record in Avaca’s samples. Güller and Zelaya (2017) consider that the lower than expected diversity found in the San José Gulf compared with their results in other areas is due to the different sampling methods used (they did not actually sample this gulf, but instead summarised information from other studies).
Plot of calculated species richness in different case studies in the San Matías and San José gulfs. Order of case studies and acronyms of location sampling areas, as in Table 3.6. (1: PM (this study). 2: PM (Archuby et al. 2015) (2). 3: SMG (Avaca et al. 2008. NOR). 4: LG (Archuby et al. 2015) (2). 5: PD (Archuby et al. 2015) (2). 6: PD (Archuby et al. 2015). (1). 7: PL (Archuby et al. 2015) (1). 8: SMG (Avaca et al. 2008. NOE). 9: SJG, SMG. (Zaixo et al. 1998). 10: SAB Güller and Zelaya 2017 (3). 11: PL Güller and Zelaya 2017 (3). 12: SJG Güller and Zelaya 2017 (3). 13: PD Güller and Zelaya 2017 (3). 14: SMG Güller and Zelaya 2017 (3). Empty symbols: life assemblages . Filled symbols: death assemblages . Squares: low sampling effort. Triangles: moderate sampling effort. Circles: high sampling effort. Slashed lines join samples from comparable localities
The three death assemblage case studies, 1 (this study), 6 and 7 (Archuby et al. 2015), are single point samples that represent moderate or low effort but offer a relatively high number of specimens and high diversity. Case studies 6 and 7 sampled 24 and 21 species respectively, which is similar to the 21 and 23 species in Avaca et al. (2008) which required 122 and 32 samples for a similar result. However, those samples were taken without sieving. In our study, a single point in Punta Mejillón, sampled using 1 × 1 mm aperture mesh, detected higher species richness than 122 samples throughout the entire San José Gulf.
This study has confirmed that sampling species richness requires sieving with fine mesh sieves for both living communities (Güller and Zelaya 2017) and their associated death assemblages (Kidwell 2002). In turn, death assemblages give excellent results at equivalent levels of sampling effort, if compared with the sampling of life assemblages . This can be explained by the time-averaging and spatial homogenization of successive communities which accumulated to constitute the death assemblage (Tomašových and Kidwell 2010a).
A word of caution is needed here: the case studies of living communities used for comparisons are mainly based on samples taken from subtidal soft bottoms, while our study was carried out in a rocky -bottom intertidal belt. More quantitative research is needed in order to compare life and death assemblages from equivalent habitats and to evaluate the partitioning of biodiversity along depth gradients and in different types of seafloor in the San Matías and San José gulfs.
3.7 Conclusions
One of the outstanding conclusions of this work is that death assemblages accumulated in rocky -bottom coastal environments are highly informative of the regional biodiversity . In this study, we showed that a single point sample contains more species than almost every study of living communities based on dozens of samples for the same region.
The lower intertidal to supratidal (high-water mark) depth gradient contains dead shell accumulations that reflect a gradient in diversity (a decrease in species richness and evenness), as well as a pattern of species turnover. The gradient in the death assemblages is mainly explained by differential transport upwards, and also by a gradient in species turnover in the living community.
Representative samples of death assemblages must be obtained with the use of fine (up to 2 mm aperture) meshes. Supratidal death assemblages , i.e., shells accumulated in the high water mark, are an acceptable proxy of the whole intertidal to subtidal assemblage if a horizon is to be chosen for sampling. The supratidal is more diverse than expected in terms of species richness, probably due to the supply of shells by strong storms, which were then trapped, out of the reach of normal waves or normal storm waves. In turn, supratidal samples show the highest bias in coase-mesh sieved samples (considered as equivalent to hand-collecting). Results from this study suggest that none of the levels is fully representative, and a sample which pools the different levels is recommended. A live/dead comparison is needed to assess this question in more depth. Exploring the live/dead mismatch in rocky -bottom intertidal environments is useful for improving our knowledge of benthic marine life, and for filling a void in the studies of rocky bottoms, as well as for evaluating the novel idea of conservation paleobiology with respect to the detection of diversity-altered, human-impacted ecosystems.
References
Anderson MJ, Crist TO, Chase JM, Vellend M, Inouye BD, Freestone AL, Sanders NJ, Cornell HV, Comita LS, Davies KF, Harrison SP, Kraft NJ, Stegen JC, Swenson NG (2011) Navigating the multiple meanings of b diversity: a roadmap for the practicing ecologist. Ecol Lett 14(19):19–28
Archuby FM, Adami M, Martinelli J, Gordillo S, Boretto GM, Malve ME (2015) Regional-scale compositional and size fidelity of rocky intertidal communities from the Patagonian Atlantic coast. Palaios 30:627–643
Archuby FM, Gordillo S (2018, September) Drilling predation traces on recent limpets from northern Patagonia, Argentina. Pal Elect 21.3.36A:1–23
Avaca MS, Narvarte MA, Gonzaléz R (2008) Asociaciones macrobentónicas en la zona norte del Golfo San Matías (Río Negro, Argentina). IBMP—Serie Publicaciones, VII:39–58
Balech E, Ehrlich MD (2008) Esquema biogeográfico del mar Argentino. Rev INIDEP 19:45–75
Barnosky AD, Hadly EA, Gonzalez P, Head J, Polly PD, Lawing AM, Eronen JT, Ackerly DD, Alex K, Biber E, Blois J, Brashares J, Ceballos G, Davis E, Dietl GP, Dirzo R, Doremus H, Fortelius M, Greene HW, Hellmann J, Hickler T, Jackson ST, Kemp M, Koch PL, Kremen C, Lindsey EL, Looy C, Marshall CR, Mendenhall C, Mulch A, Mychajliw AM, Nowak C, Ramakrishnan U, Schnitzler J, Das Shrestha K, Solari K, Stegner L, Stegner MA, Stenseth NC, Wake MH, Zhang Z (2017) Merging paleobiology with conservation biology to guide the future of terrestrial ecosystems. Science, 355(6325):eaah4787
Bertness MD, Crain CM, Silliman BR, Bazterrica MC, Reyna MV, Hildago F, Farina JK (2006) The community structure of Western Atlantic Patagonian rocky shores. Ecol Mon 76(3):439–460
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Austral J E 18(1):117–143
Clarke KR, Somerfield PJ, Chapman MG (2006) On resemblance measures for ecological studies, including taxonomic dissimilarities and a zero-adjusted Bray-Curtis coefficient for denuded assemblages. J Exp Mar Biol Ecol 330(1):55–80
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edn. PRIMER-E, Plymouth
Dadon J (2005) Changes in the intertidal community structure after a mass mortality event in sandy beaches of Argentina. Cont Zool 74(1–2):27–39
De Francesco CG, Tietze E, Cristini PA (2013) Mollusk Successions of Holocene Shallow-Lake Deposits from the Southeastern Pampa Plain. Argentina Palaios 28(12):851–862
Dietl GP, Kidwell SM, Brenner M, Burney DA, Flessa KW, Jackson ST, Koch PL (2015) Conservation paleobiology: leveraging knowledge of the past to inform conservation and restoration. Ann Rev Earth Planet Sci 43:79–103
Dietl GP, Flessa KW (2011) Conservation paleobiology: putting the dead to work. Trends Ecol Evol 26(1):30–37
Erthal F, Kotzian CB, Simoes MG (2011) Fidelity of Molluscan Assemblages from the Touro Passo formation (Pleistocene-Holocene), Southern Brazil: Taphonomy as a tool for discovering natural baselines for freshwater communities. Palaios 26(7):433–446
Fiori SM, Cazzaniga NJ (1999) Mass mortality of the yellow clam, Mesodesma mactroides (Bivalvia: Mactracea) in Monte Hermoso beach. Argentina Biol Cons 89(3):305–309
Fürsich FT, Aberhan M (1990) Significance of time averaging for palaeocommunity analysis. Lethaia 23(2):143–152
Fürsich FT, Flessa K (1987) Taphonomy of tidal flat molluscs in the northern Gulf of California: paleoenvironmental analysis despite the perils of preservation. Palaios 2(6):543–559
Gordillo S, Archuby FM (2012) Predation by drilling gastropods and asteroids upon mussels in rocky shallow shores of Southernmost South America: paleontological implications. Act Palaeont Polonica 57(3):633–646
Gordillo S, Archuby FM (2014) Live-live and live-dead interactions in marine death assemblages: The case of the patagonian clam venus antiqua. Act Palaeont Polonica 59(2):429–442
Güller M, Zelaya DG (2017) A hot-spot of biodiversity in Northern Patagonia, Argentina. Biodiv Cons 14:1–14
Hammer Ø, Harper DAT (2006) Paleontological data analysis. Blackwell Publishing, Oxford
Hammer Ø, Harper DAT, Ryan PD (2001) Paleontological statistics software package for education and data analysis. Palaeont Elect 4(1):9–18
Hassan GS, Rojas LA, De Francesco CG (2018) Incorporating taphonomy into community-based paleoenvironmental reconstructions: can diatom preservation discriminate among shallow lake sub-environments? Palaios 33:376–392
Hurlbert SHS (1971) The nonconcept of species diversity: a critique and alternative parameters. Ecology 52(4):577–586
Kelley PH, Hansen TA (2007) Latitudinal patterns in naticid gastropod predation along the east coast of the United States: a modern baseline for interpreting temporal patterns in the fossil record. In Sediment-organism interactions: a multifaceted ichnology. SPEM Special Publication. SEPM, pp 287–299
Kidwell SM (2001) Preservation of species abundance in marine death assemblages. Science 294(November):1091–1094
Kidwell SM (2002) Mesh-size effects on the ecological fidelity of death assemblages: a meta-analysis of molluscan live-dead studies. Geobios 35(suppl):107–119
Kidwell SM (2008) Ecological fidelity of open marine molluscan death assemblages: Effects of post-mortem transportation, shelf health, and taphonomic inertia. Lethaia 41(3):199–217
Kidwell SM (2009) Evaluating human modification of shallow marine ecosystems: mismatch in composition of molluscan living and time-averaged death assemblages. In: Dietl GP, Flessa KW (eds) Conservation paleobiology: using the past to manage for the future. pp 113–139
Kidwell SM (2013) Time-averaging and fidelity of modern death assemblages: building a taphonomic foundation for conservation palaeobiology. Palaeontology 56(3):487–522
Kidwell SM (2015) Biology in the Anthropocene: challenges and insights from young fossil records. Proc Nal Acad Sci 112(16):4922–4929
Kidwell SM, Bosence DWJ (1991) Taphonomy and time-averaging of marine shelly faunas. In: Allison PA, Briggs DEG (eds) Taphonomy: releasing the data locked in the fossil record. pp 115–209
Kidwell SM, Tomašových A (2013) Implications of time-averaged death assemblages for ecology and conservation biology. Ann Rev Ecol Evol Syst 44(1):539–563
Kidwell SM, Tomašových A (2017) Nineteenth-century collapse of a benthic marine ecosystem on the open continental shelf. Proc Roy Soc B 284:1–9
Koleff P, Gaston KJ, Lennon JJ (2003) Measuring beta diversity for presence—absence data. JAnimal Ecol 72:367–382
López RA, Penchaszadeh PE, Marcomini SC (2008) Storm-related strandings of mollusks on the Northeast Coast of Buenos Aires, Argentina. J Coast Res 244:925–935
Louys J (2012) Paleontology in ecology and conservation: an introduction. In: Louys J (ed) Paleontology in ecology and conservation. Springer, Berlin-Heidelberg, pp 1–7
Magierowski RH, Johnson CR (2006) Robustness of surrogates of biodiversity in marine benthic communities. Ecol Appl 16(6):2264–2275
Martinelli JC, Gordillo S, Archuby FM (2013) Muricid drilling predation at high latitudes: insights from the Southernmost Atlantic. Palaios 28(1):33–41
Mellin C, Delean S, Caley J, Edgar G, Meekan M, Pitcher R, Przeslawski R, Williams A, Bradshaw C (2011) Effectiveness of biological surrogates for predicting patterns of marine biodiversity: a global meta-analysis. PLoS ONE 6(6):e20141
Olszewski TD, Kidwell SM (2007) The preservational fidelity of evenness in molluscan death assemblages. Paleobiology 33(1):1–23
Perez SI, Postillone MB, Rindel D, Gobbo D, Gonzalez PN, Bernalet V (2016) Peopling time, spatial occupation and demography of Late Pleistocene-Holocene human population from Patagonia. Quat Int 425:214–223
R Core Team (2017) R: a language and environment for statistical computing. Available at: https://www.r-project.org
Rick TC, Lockwood R (2013) Integrating paleobiology, archeology, and history to inform biological conservation. Cons Biol 27(1):45–54
Smith SDA (2005) Rapid assessment of invertebrate biodiversity on rocky shores: where there’s a whelk there’s a way. Biodivers Conserv 14:3565–3576
Smith SDA (2008) Interpreting molluscan death assemblages on rocky shores: are they representative of the regional fauna? J Exp Mar Biol Ecol 366(1–2):151–159
Terry RC (2010) The dead do not lie: using skeletal remains for rapid assessment of historical small-mammal community baselines. Proceedings R Soc2 B 77(1685):1193–1201
Tietze E, De Francesco CG (2012) Compositional fidelity of subfossil mollusk assemblages in streams and lakes of the Southeastern Pampas. Argentina Palaios 27(6):401–413
Tomašových A, Kidwell SM (2009a) Fidelity of variation in species composition and diversity partitioning by death assemblages: time-averaging transfers diversity from beta to alpha levels. Paleobiology 35(1):94–118
Tomašových A, Kidwell SM (2009b) Preservation of spatial and environmental gradients by death assemblages. Paleobiology 35(1):119–145
Tomašových A, Kidwell SM (2010a) Predicting the effects of increasing temporal scale on species composition, diversity, and rank-abundance distributions. Paleobiology 36(4):672–695
Tomašových A, Kidwell SM (2010b) The effects of temporal resolution on species turnover and on testing metacommunity models. Am Nat 175(5):587–606
Tomašových A, Kidwell SM (2011) Accounting for the effects of biological variability and temporal autocorrelation in assessing the preservation of species abundance. Paleobiology 37(2):332–354
Tyler CL, Kowalewski M (2017) Surrogate taxa and fossils as reliable proxies of spatial biodiversity patterns in marine benthic communities. Proc R Society B 284(1850):20162839
Tyler CL, Leighton LR, Kowalewski M (2014) The effects of limpet morphology on predation by adult cancrid crabs. J Exp Mar Biol Ecol 451:9–15
Visaggi CC, Kelley PH (2007) Relationship between drilling predation and shell morphology of patellid limpets from Southwestern England. Denver, Colorado, USA, GSA Anual Meeting, p 19813
Visaggi CC, Kelley PH (2015) Equatorward increase in naticid gastropod drilling predation on infaunal bivalves from Brazil with paleontological implications. Palaeogeogr Palaeoclimatol Palaeoecol 438:285–299
Warwick RM, Light J (2002) Death assemblages of molluscs on St Martin’s Flats, Isles of Scilly: a surrogate for regional biodiversity? Biodiv Cons 11:99–112
Yanes Y (2012) Anthropogenic effect recorded in the live-dead compositional fidelity of land snail assemblages from San Salvador Island. Bahamas. Biodiv Cons 21(13):3445–3466
Yanes Y, Tyler CL (2009) Drilling predation intensity and feeding preferences by Nucella (Muricidae) on limpets inferred from a dead-shell assemblage. Palaios 24(5):280–289
Yanes Y, Tomašových A, Kowalewski M, Castillo C, Aguirre J, Alonso MR, Ibáñez M (2008) Taphonomy and compositional fidelity of Quaternary fossil assemblages of terrestrial gastropods from carbonate-rich environments of the Canary Islands. Lethaia 41(3):235–256
Zaixso HE, Lizarralde Z, Pastor C, Gomes-Simes E, Romanello E, Pagnoni G (1998) Distribución espacial del macrozoobentos submareal del golfo San José (Chubut, Argentina). Rev Biol Mar Oceanogr 33(1):43–72
Zuschin M, Hohenegger J, Steininger FF (2000) A comparison of living and dead molluscs on coral reef associated hard substrata in the northern Red Sea—implications for the fossil record. Palaeogeogr Palaeoclimat Palaeoecol 159:167–190
Zuschin M, Oliver PG (2003) Fidelity of molluscan life and death assemblages on sublittoral hard substrata around granitic islands of the Seychelles. Lethaia 36(2):133–149
Zuschin M, Stachowitsch M (2007) The distribution of molluscan assemblages and their postmortem fate on coral reefs in the Gulf of Aqaba (Northern Red Sea). Mar Biol 151(6):2217–2230
Acknowledgements
This work was supported by the research project PI 40-A-383 of Universidad Nacional de Río Negro.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
3.1 Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Archuby, F.M., Roche, A. (2020). Intertidal Death Assemblages as Proxies of Marine Biodiversity. An Example from Northern Patagonia, Argentina. In: Martínez, S., Rojas, A., Cabrera, F. (eds) Actualistic Taphonomy in South America. Topics in Geobiology, vol 48. Springer, Cham. https://doi.org/10.1007/978-3-030-20625-3_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-20625-3_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-20624-6
Online ISBN: 978-3-030-20625-3
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)