Abstract
Pericytes in the central nervous system attract growing attention of neurobiologists because of obvious opportunities to use them as target cells in numerous brain diseases. Functional activity of pericytes includes control of integrity of the endothelial cell layer, regeneration of vascular cells, and regulation of microcirculation. Pericytes are well integrated in the so-called neurovascular unit (NVU) serving as a platform for effective communications of neurons, astrocytes, endothelial cells, and pericytes. Contribution of pericytes to the establishment and maintaining the structural and functional integrity of blood–brain barrier is confirmed in numerous experimental and clinical studies. The review covers current understandings on the role of pericytes in molecular pathogenesis of NVU/BBB dysfunction in Alzheimer’s disease with the special focus on the development of cerebral amyloid angiopathy, deregulation of cerebral angiogenesis, and progression of BBB breakdown seen in Alzheimer’s type neurodegeneration.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Pericytes represent one of the vascular cell populations located next to endothelial cells at their abluminal part. Their functioning covers a wide spectrum of activities, i.e., control of integrity of the endothelial cell layer, regeneration of vascular cells, and regulation of microcirculation (Trost et al. 2016). There is a growing evidence that intrinsic plasticity of pericytes is very important for their role on vascular remodeling since they are able to control endothelial cells proliferation, apoptosis, vascular sprouting, and corresponding regression (Simonavicius et al. 2012; Stapor et al. 2014). Pericytes express many surface antigens that are used for phenotyping of cells, i.e., nestin, angiopoietin, chondroitin sulfate proteoglycan 4 NG2, CD146, CD31 (PECAM-1), platelet-derived growth factor receptor-beta PDGFRβ, and desmin (Armulik et al. 2011). However, the majority of markers can also be detected on other cell types, i.e., CD31 on endothelial cells, NG2 on oligodendrocytes, PDGFRβ on fibroblasts. In the context of expression pattern, pericytes seem to be very close to mesenchymal stromal cells, and it was suggested that CD146(+)CD34(−) cells isolated from some (i.e., bone marrow, placenta) but not all the tissues may represent the population of pericytes (Blocki et al. 2013). Generation of pericytes occurs in embryonic and postnatal period throughout the life, but the origin of pericytes remains to be unresolved question in vascular biology, and current data suggest that even within one tissue pericytes seem to be heterogenous in their developmental story (Dias Moura Prazeres et al. 2017). Application of immunostaining for detection of surface antigens, using of transgenic markers (i.e., XlacZ4 and NG2 dsRED) and numerous functional assays allowed identifying several subtypes of pericytes, particularly, type-1 pericytes contribute to fibrogenesis and production of collagen (Birbrair et al. 2013, 2014a, b, c) whereas type-2 pericytes take part in angiogenesis (Birbrair et al. 2014a, b, c).
Pericytes in the central nervous system (CNS) attract the growing attention of neurobiologists because of obvious opportunities to use them as target cells in numerous brain diseases. Cerebral microvessels have higher pericytes/endothelial cells ratio (10–30-fold) than other tissues (Winkler et al. 2014), thereby the role of pericytes in controlling brain microvessel endothelial cell (BMECs) functional activity is rather significant. Brain pericytes are sparsely distributed and occupied the middle of the capillary bed, keep relative stability of their somata but demonstrate dynamic changes in their processes in the adult brain, and may effectively recover after damage (Berthiaume et al. 2018). Being in a close contact with endothelial cells, pericytes are surrounded by basement membrane and extend processes both along and around capillaries (Attwell et al. 2016), therefore, there are no doubts on the key role of pericytes in the control of endothelial cells. Thus, in the brain tissue, they are well integrated in the concept of neurovascular unit (NVU) as a platform for effective communications of neurons, astrocytes, endothelial cells, and pericytes which is required for the maintenance of metabolic coupling, gliovascular control as well as the integrity of the blood–brain barrier (BBB) in (patho)physiological conditions (Salmina et al. 2014). In addition, the activity of pericytes attributes to the regulation of cerebral angiogenesis (experience-induced and reparative), acquisition of specific phenotype of BMECs, establishment of neurovascular coupling providing adequate blood supply in active brain regions, promotion of neurogenesis within neurogenic niches or oligodendrogenesis within oligovascular niches (Hall et al. 2014; Trost et al. 2016). Moreover, pericytes are recognized as a key cellular component of BBB models in vitro suggested for BBB-on-chip or brain-on-chip microphysiological systems as well as a promising tool for nervous system regeneration (Yamamizu et al. 2017; Greenwood-Goodwin et al. 2016; Tian et al. 2017). Thus, pericyte dysfunction in neurodevelopmental and neurodegenerative diseases is gradually becoming a “hot topic” in neurosciences.
Pericyte Dysfunction in Cerebral Amyloid Angiopathy
Dysfunction of NVU is a well-recognized feature of Alzheimer’s disease (AD) (Salmina et al. 2010, 2015a, b). In addition to other hypothesis of AD development (amyloid, calcium, or gliocentric), for more than two recent decades, pathogenesis of AD has been discussed in the context of prominent vascular alterations culminating in the establishment of the so-called cerebral amyloid angiopathy (CAA) caused by the accumulation of beta-amyloid (Aβ) in small-sized and medium-sized vessels, mostly arterial (Biffi and Greenberg 2011) predominantly in leptomeningeal and cortical vessels of cerebral lobes and cerebellum (Yamada 2015). In severe angiopathy, amyloid deposits replace degenerating vessel smooth muscle cells leading to microaneurysms formation and hemorrhages (Yamada 2000; Jellinger 2002). In general, vascular nature of AD has been confirmed in numerous epidemiological, neuroimaging, pathological, experimental, and clinical studies (de la Torre 2004) (Fig. 7.1). The vascular hypothesis of AD underlies the initial role of chronic cerebral hypoperfusion, abnormal microvascular remodeling, BBB breakdown, development of ischemic lesions and microhemorrhages associated with Aβ deposition, neuroinflammation, NVU disorganization, loss of neuroplasticity, and synaptic plasticity, thereby resulting in progressive cognitive and behavioral deficits (Salmina et al. 2015a, b) (Fig. 7.2).
Mechanisms of neurovascular unit dysfunction in Alzheimer’s disease. NVU dysfunction is an obligatory component of Alzheimer’s disease pathogenesis. Compared with the normal brain, Alzheimer’s diseases brain is characterized by prominent neuronal loss, reactive gliosis, dysfunction and death of brain microvessel endothelial cells and pericytes caused by excessive accumulation of Aβ in brain parenchyma and perivascular region. As a result, structural and functional integrity of BBB is compromised, thereby supporting the establishment of circulus vitiosus
Molecular pathogenesis of cerebral amyloid angiopathy (CAA) development in Alzheimer’s disease. Various mechanisms contribute to initiation and progression of CAA in Alzheimer’s disease, including excessive Aβ production, inadequate Aβ clearance, insufficient blood supply in affected brain regions, and neuroinflammation
It is clear that all types of cells within the NVU could be affected in CAA; therefore, it is easy to observe endothelial alterations and smooth muscle cells degeneration in medium-sized cerebral vessels, as well as endothelial dysfunction, pathology of perivascular astroglia and prominent pericytes loss in cerebral microvessels along the time-course of AD progression. These changes affect the viability of neuronal cells (Grammas et al. 1999), alter microcirculation (Pluta et al. 2013), and lead to chronic ischemia and neurodegeneration. In a contrast to other NVU cells, pericytes have not been recognized as dramatically affected cells in the pathogenesis of AD, but recent findings suggest their significant role in the progression of AD-associated microvascular alterations and impairment of plasticity in AD brain.
At this point, it should be clarified that morphology and physiology of pericytes might be greatly compromised in aged brain, therefore, one should take care while talking about pericytes dysfunction in a time-course of Alzheimer’s type of dementia. As an example, aging is always associated with pericytes loss in various tissues and dramatic changes in their myogenic or angiogenic capacity, and decreased number of effective endothelial–pericyte interactions (Hughes et al. 2006; Birbrair et al. 2014a, b, c; Stefanska et al. 2015). Pericytes aging is also important in the brain tissue where reduction of pericytes number might be partially compensated by extension of their processes, presumably, due to the activity of PDGF/PDGFR signaling. Particularly, Berthiaume and colleagues demonstrated that pericytes can participate in vascular remodeling in the adult brain. The authors revealed pericytes’ plasticity in the adult brain by using elegant state-of-the-art techniques, including two-photon microscopy in combination with sophisticated Cre/loxP in vivo tracing technologies. They imaged at high resolution over several weeks cerebral pericytes in NG2-CreER/TdTomato, Myh11-CreER/TdTomato, and PDGFRβ-Cre/YFP mice. These experiments unveiled that pericytes compose a quasi-continuous, not overlapping, network along the entire length of blood vessels. Interestingly, the pericytes’ prolongations were not stable in length, extending or retracting during the period of analysis. Then, the authors explored the effect of pericyte’s death on its neighbor pericytes. After pericyte’ ablation, using targeted two-photon irradiation, Berthiaume and colleagues showed that adjacent pericytes extend their processes into the uncovered area, covering the exposed blood vessel. Strikingly, neighboring pericytes are able to inhibit vascular dilatation that happens after pericyte depletion (Berthiaume et al. 2018). Thus, one may assume that aging- or Alzheimer’s-associated loss of pericytes in cerebral microvessels could lead to compensatory extension of processes from neighboring pericytes in order to cover the most achievable area at the abluminal side of BMECs. However, since both aging and Alzheimer’s type of neurodegeneration are often accompanied by elevation in PDGF levels or dysfunction in PDGF/PDGFR signaling (Vazquez-Padron et al. 2004; Liu et al. 2018), remodeling of remaining pericytes might be inefficient to compensate for pericytes loss, thereby leading to incomplete coverage of BMECs and aberrant BBB structural and functional integrity.
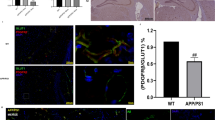
Indeed, Alzheimer’s type of neurodegeneration is marked with significant and progressive pericytes loss (Giannoni et al. 2016). Several recent reviews were focused on the role of pericytes in the pathogenesis of AD (Winkler et al. 2014; Kisler et al. 2017a, b). Aβ is toxic for cerebral pericytes, thereby, pericyte loss further contributes to amyloid neurotoxicity as it was confirmed in mice overexpressing the Swedish mutation of human Aβ-precursor protein (APPsw/0) and crossed with pericyte-deficient platelet-derived growth factor receptor-β (PDGFRβ+/−) mice (Sagare et al. 2013). Moreover, when C3H/10T1/2 mouse mesenchymal stem cells were differentiated into pericytes and stereotaxically injected into the brains of amyloid AD model APP/PS1 mice, local microcirculation was improved, whereas the levels of Aβ in the brain tissue were reduced 3 weeks later. Thus, it was found that functionally competent pericytes may contribute a lot to maintaining the adequate blood supply to the brain tissue and provide clearance of Aβ (Tachibana et al. 2018). Unfortunately, in a time-course of AD progression, pericytes are gradually losing their ability to control local blood flow and AD transport, therefore, clinical stages of AD are already associated with dramatic changes in pericytes quantity (due to apoptosis) and quality (due to intensification of pericyte dysfunction). The same effect might be achieved by high-fat diet provoking progressive pericytes loss and aberrant neurovascular coupling, Aβ accumulation and BBB breakdown (Thériault et al. 2016). Analogous changes in pericytes number could result from the accumulation of advanced glycation end products (AGEs) formed under the conditions of chronic hyperglycemia and non-enzymatic protein glycation (Lange et al. 2013) that are able to interact with their own receptors (RAGEs) expressed on endothelial cells and pericytes, thereby leading to apoptosis (Yamagishi et al. 2005). Even these data were obtained in diabetic pericytes, one may assume that the same mechanisms might be essential in Alzheimer’s disease characterized by local insulin resistance and alterations in glucose utilization in brain cells.
Thus, the main causes of pericytes loss in Alzheimer’s disease might be summarized as follows: (1) toxic action of supraphysiological concentrations of Aβ leading to pericytes injury and cell death; (2) toxic action of reactive oxygen species produced in the conditions of oxidative stress and excitotoxicity within the NVU; (3) pro-apoptotic action of AGEs; (4) aberrant signaling pathways that contribute to the control of pericytes’ functional activity and viability (i.e., PDGF-, TGFβ1-, or angiopoietin/Tie2-mediated); (5) metabolic alterations in pericytes; (6) excessive pericytes remodeling due to stimulation of angiogenesis leading to hypervascularity.
Pericytes take an active part in the translocation of Aβ through the BBB. In normal conditions, low-density lipoprotein receptor-related protein-1 (LRP1) acting as Aβ translocator is abundantly expressed in BMECs and pericytes being involved in the transfer of Aβ which is constantly produced in the brain tissue to the peripheral blood (Winkler et al. 2014). AD-associated loss of pericytes results in impaired clearance of Aβ in the brain, thereby leading to amyloid deposition and development of CAA. Moreover, incubation of cerebrovascular cells in vitro with toxic concentrations of Aβ results in the increase of LRP1 expression, probably, for better internalization of amyloid by pericytes, but excess of Aβ leads to loss of pericytes (Wilhelmus et al. 2007).
In general, pericytes appear as a nice model to study Alzheimer’s type of neurodegeneration in vitro. In very early studies, pericytes have been shown to produce and metabolize amyloid precursor protein (APP) as well as some other APP-associated molecules (ApoE, complement factor C1q (Verbeek et al. 1999)). At the same time, pericytes serve as a target for the cytotoxic action of Aβ but seem to be more resistant to its action comparing to other brain cells, i.e., in terms of Ca2+ signaling: basal levels of intracellular Ca2+ are greatly affected by the exposure of pericytes to Aβ in vitro, however, calcium machinery controlled by G-protein-coupled receptors remains unaffected (Piegsa et al. 2017).
In APPsw/0; Pdgfrβ+/−, pericyte deficiency leads to progression of signs of tau pathology and an early neuronal loss further resulting in accelerated cognitive decline due to complimentary harmful effects of Aβ accumulation and pericytes loss (Sagare et al. 2013).
Interesting properties of pericytes have been described in hypothalamus where these cells can specifically increase insulin sensitivity of hypothalamic neurons (Takahashi et al. 2015). Taking into consideration the current view on the pathogenesis of AD as a local insulin resistance and impairment of glucose metabolism in the brain tissue (An et al. 2018), one can suggest that pericytes loss in the defined brain regions could be responsible for reduced susceptibility of neurons to insulin action and glucose utilization. Pericytes serve as important regulators of insulin transport through the BBB (Banks et al. 2012), and, vice versa, insulin stabilizes BMECs-pericytes interactions and integrity of tight junctions within the BBB in a phosphoinositide-3 kinase/protein kinase B/glycogen synthase kinase-3β-dependent manner (Ito et al. 2017). Thus, dysfunction of pericytes in AD might lead to poor entry of peripherally produced insulin into the brain tissue and aggravation of local insulin resistance state. It is interesting that insulin may protect cerebral pericytes from Aβ cytotoxicity (Rensink et al. 2004a), whereas the treatment of pericytes with toxic concentrations of Aβ results in decreased expression of insulin-like growth factor-binding protein-2 mRNA (Rensink et al. 2004b).
Since functional insulin receptors are expressed in pericytes (Escudero et al. 2017), local insulin resistance in AD would have more pronounced effects on pericytes viability and functioning by limiting their glycolytic activity. It was shown that the inhibition of 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 (PFKFB3) activity (regulatory glycolytic enzyme) in pericytes resulted in dramatic changes such as suppression of motility and increasing adhesion to endothelial cells, thereby resulting in better coverage of endothelial layer with pericytes and, obviously, in the establishment of anti-angiogenic microenvironment (Cantelmo et al. 2016). However, no GluT4 expression has been detected in brain pericytes; therefore, insulin action on pericyte glycolytic metabolism needs in further careful evaluation.
Pericytes contribute a lot to the local control of cerebral blood flow, particularly, they are able to dilate in response to neuronal stimulation to provide adequate blood supply in active brain regions (Winkler et al. 2014). When functional activity of pericytes is compromised, i.e., in PDGFRβ+/− mice, such responses to neuronal stimulation is abolished within the NVU without obvious changes in the activity of other cells regulating functional hyperemia (BMEC , perivascular astroglia). As a result of hemodynamic alterations, NVU dysfunction develops and leads to neurodegeneration. PDGFRβ−/− also demonstrate delayed capillary but not arteriolar dilation to various stimuli corresponding to lack of pericyte coverage of BMECs (Kisler et al. 2017a, b). However, very recent data obtained with an optogenetic approach to pericytes stimulation suggest that pericyte-controlled diameter of small vessels in the brain tissue might not predominate over smooth-muscle actin (SMA)-regulated constriction of arterioles (Sweeney et al. 2018).
Other factors contributing to pericyte dysfunction and promotion of CAA are as follows: (1) oxidative stress due to Aβ-mediated cytotoxicity, overproduction of reactive oxygen species (ROS) and CAA-associated cerebrovascular deficits (Han et al. 2015), microglia-induced activation of pericytes leading to ROS production (Ding et al. 2017); (2) aberrant PDGFRβ expression and signaling induced by Aβ in neuronal cells (Liu et al. 2018) and in pericytes (Miners et al. 2018); (3) mitochondrial dysfunction caused by the deposition of Aβ and progression of CAA culminating in pericytes degeneration (Szpak et al. 2007); (4) hypoxia-induced HIF-1-mediated changes in pericytes motility and adhesion (Mayo and Bearden 2015) leading to excessive angiogenesis and hypervascularity of brain tissue.
Figure 7.3 summarizes current understandings of alterations of pericytes biology in CAA.
Molecular mechanisms leading to pericytes loss in cerebral amyloid angiopathy (CAA). Neurotoxic action of accumulated Aβ in Alzheimer’s disease leads to local insulin resistance which further promotes Aβ production and deposition. Chronic high levels of extracellular glucose result in non-enzymatic protein glycation, accumulation of advanced glycation end products (AGEs) and persistent activation of their receptors (RAGE) in pericytes. Then, pericytes respond to these changes by activation transcription of oxidative stress- and hypoxia-controlled transcription factors, thereby switching on the programmed cell death. Aberrant neoangiogenesis and neuroinflammation serve as mechanisms supporting non-reversible pericytes dysfunction
Pericyte Dysfunction in Cerebral Hypervascularity and BBB Breakdown Associated with Alzheimer’s Disease
Role of pericytes in controlling angiogenesis is well established. Particularly, they may participate in different stages of angiogenic process : (1) establishment of local pro-angiogenic microenvironment stimulating endothelial cells to proliferate and to migrate along the newly forming vascular tube; (2) detachment from the abluminal part of endothelial cells to ensure effective contribution of tip-cells to the vessel elongation; (3) regulation of final stages of angiogenesis when maturation of newly established vessels is of key importance for the integration of vessel in the pre-existing vascular network and acquisition of selective permeability; (4) controlling processes of microvascular rarefaction that are essential for adequate remodeling of vessels; (5) support of endothelial cells survival and functional competence (Benjamin et al. 1998; Franco et al. 2011; Ribatti et al. 2011; Simonavicius et al. 2012; Eilken et al. 2017). To do this, pericytes utilize many signaling pathways, including PDGF, VEGF, Angiopoietin/Tie, MMP9, Notch, Endosialin/CD248, purinergic signaling, and gap junction machinery.
Recently, it became clear that AD is characterized by paradoxical hypervascularity occurring due to excessive neoangiogenesis and establishment of newly formed small vessels with leaky BBB (Biron et al. 2011). Cessation of neoangiogenesis by Aβ immunotherapy may prevent further brain tissue degeneration (Biron et al. 2013). Disruption of PDGFRβ-mediated signaling in brain pericytes results in early and progressive loss of pericytes, microvascular rarefaction and alterations in BB structural and functional integrity predominantly in the cortex, hippocampus and striatum (Nikolakopoulou et al. 2017), thus providing new insights in the pathogenic role of pericyte-controlled vascular factor in AD.
Unstimulated pericytes support vessel wall integrity, however, they convert into cells with evident pro-angiogenic potential being stimulated by various regulatory and damaging factors. Recent data suggest that pericytes may contribute to excessive angiogenesis in AD via several mechanisms. First of all, as it was mentioned above, detachment of pericytes from the endothelial cell layer associated with CypA-MMP9-mediated basal membrane destruction is required for tip-cell movement and vascular tube formation. Secondly, stimulated pericytes are able to produce numerous pro-angiogenic factors, i.e., ligands of chemokine receptor CXCR3 (probably, CXCL4/platelet factor 4, CXCL9/MIG, CXCL10/IP-10, or CXCL11/IP-9) expressed on endothelial cells (Bodnar et al. 2013), or angiopoietin involved in angiopoietin/Tie signaling between endothelial cells and pericytes (Teichert et al. 2017), thereby affecting cerebral angiogenesis being damaged by accumulating Aβ. Thirdly, pericytes may contribute to cerebral microvascular rarefaction seen in normal aging and AD and correlating with dementia progression and BBB impairment (Tucsek et al. 2014). The same phenomenon—microvascular rarefaction and pericytes deficiency—is also well recognized in hypertensive individuals with AD (Toth et al. 2013), thereby leading to microthrombosis, inadequate blood supply in active brain regions. Fourthly, metabolic disturbances caused by the impairment of glucose metabolism in AD brain could lead to suppressed glycolytic flux in BMECs and pericytes, thereby resulting in abnormal vessel sprouting and disorganization, reduced pericyte coverage, and breakdown of BBB (Cruys et al. 2016). Finally, insulin signaling which is absolutely required for developmental angiogenesis and is rather specific for pericytes (Warmke et al. 2017).
Basal and stimulated production of lactate in glycolysis and its transport between the NVU cells is an important mechanism of angiogenesis and barriergenesis regulation in the brain tissue (Salmina et al. 2015a, b). Astrocytes serve a major source of lactate which is utilized by neurons (for energy production) and by endothelial cells (for brain-to-blood transfer). Within the NVU, lactate acts at target cells via specific lactate receptor (HCAR1/GPR81) which is known as metabolic sensor regulating several processes in carbohydrate and lipid metabolism, partially in the insulin-dependent manner in some peripheral tissues and in the NVU as well (Ahmed et al. 2010; Lauritzen et al. 2014). In this context, lactate acts as autocrine or paracrine regulator. Recently, we have shown that long-lasting stimulation of GPR81 receptors in BMECs in vitro could activates mitochondrial biogenesis but suppresses expression of monocarboxylate transporter-1 (MCT-1) and CD147 (Khilazheva et al. 2017). Taking into consideration that cerebral pericytes are equipped with GPR81 whose stimulation results in the elevation of local VEGFA levels and promotion of angiogenesis associated with the activation of extracellular signal-regulated kinase (ERK1/2)- and Akt (Morland et al. 2017), one may suggest that local production of lactate in active brain regions (due to neuron-astroglia metabolic coupling) could activate both pericytes and BMEC to provide microenvironment favoring angiogenesis. Moreover, it may also have some additional meaning in the context of vascular tone regulation as it was shown in retinal pericytes subjected to extracellular lactate (Yamanishi et al. 2006). High concentrations of lactate is a hallmark of aging and AD progression (Kapogiannis and Reiter 2014; Liguori et al. 2015); therefore, it is tempting to speculate that permanent elevated levels of lactate could degenerating brain to excessive angiogenesis (presumably, via mechanism involving BMECs and pericytes activation via GPR81 receptors) resulting in the establishment of disorganized and defective microvessels with impaired barrier function.
The unresolved question remains whether such changes could have any relation to the mechanism of pro-angiogenic activity of hormones and growth factors (i.e., insulin or VEGF) which implies detachment of pericytes from endothelial cells as a prerequisite for effective tip-cell proliferation and migration along the growing vascular tube (Escudero et al. 2017). If so, Aβ-induced pericytes loss from the affected brain microvessels could serve as a mechanism of angiogenesis support. However, in a case of CAA, loss of pericytes would result in unbalanced pro- and anti-angiogenic activities in the perivascular space further leading to abnormal hypervascularity.
As expected, location of pericytes just next to the endothelial cell layer makes them ideal contributors to the control of BBB permeability. As an example, expression of CD146 which acts as a co-receptor for PDGFRβ is required for efficient pericyte–endothelial interactions and maturation of the BBB (Chen et al. 2017). Cerebrovascular pericytes produce lipidated forms of ApoE acting together with perivascular astroglial cells, and ApoE suppresses motility of pericytes and their adhesion in LRP1- and RhoA-dependent manner (Casey et al. 2015). Experimental data obtained in mice with targeted replacement of murine ApoE with each human ApoE isoform or in ApoE−/− mice demonstrate that ApoE is required for the cerebrovascular integrity by regulating cyclophilin A (CypA)–NF-κB–matrix metalloproteinase (MMP9) pathway in pericytes, whereas insufficiency of ApoE expression leads to elevated production of CypA in pericytes and disruption of tight junctions in BMECs (Bell et al. 2012). Individuals with ApoE4 gene (genetic risk factor for AD) display severe alterations of BBB structural and functional integrity that are related to the degree of pericytes loss in brain microvessels due to ApoE4 leads to excessive activation of LRP1-dependent cyclophilin A (CypA)– matrix metalloproteinase 9 (MMP-9) signaling in pericytes (Halliday et al. 2016).
Degree of pericytes loss correlates with the impairment of BBB permeability in individuals with Alzheimer’s type of neurodegeneration: high levels of sPDGFRβ originated from dysfunctional or destroyed pericytes have been detected in the cerebrovascular fluid of persons with mild cognitive impairment (MCI) compared to age-matched cognitively normal subjects, sPDGFRβ concentrations positively correlated with increased BBB permeability in the hippocampus of MCI patients (Montagne et al. 2015).
Elevated permeability of BBB caused (at least, partially) by functionally incompetent or damaged pericytes, and provides conditions for progression of neuroinflammation which is a key mechanism of AD pathogenesis. Contribution of glial cells and pericytes to the pathogenesis of neuroinflammation can be distinguished, i.e., in the 3D BBB-on-chip model in vitro (Herland et al. 2016). It should be noted that pericytes may act as macrophage-like cells to clean extracellular perivascular fluid in the brain tissue by means of phagocytosis and pinocytosis (Bergers and Song 2005). Besides, they have rather impressive secretome consisting of chemokines, interleukins (IL-9, -10, -12, -13), granulocyte-colony stimulating factor, granulocyte macrophage-colony stimulating factor, etc., particularly, being activated with pro-inflammatory stimuli (Kovac et al. 2011). On the other hand, neuroinflammation affects pericytes viability and functional activity, i.e., TGFβ1 whose dyscoordinated signaling has been detected in AD (von Bernhardi et al. 2015) may stimulate pericytes to release pro-inflammatory cytokines and extracellular matrix degrading enzymes. As a result, BBB integrity is compromised and microglia is attracted to the site of neuroinflammation (Rustenhoven et al. 2016). Some data suggest that due to multipotent stem cell activity of pericytes, they may serve as a source of microglial cells in brain ischemia (Sakuma et al. 2016), but whether this mechanism is active in chronic neurodegeneration is not clear yet.
In some clonogenic niches (i.e., in the bone marrow), CD146+ pericytes exist as multipotent cells (Mangialardi et al. 2016). In the brain, pericytes may differentiate to microglia in conditions with the demand for more immune cells, whereas neuronal cells could be also achieved from pericytes by means of genetic reprogramming procedure in vitro (Karow et al. 2012). Thus, the plasticity of pericytes allows achieving a phenotype which is mainly anticipated in the context of current conditions in the brain tissue. At the same time, pericyte-regulated BBB permeability may affect neuroplasticity as well. It is known that BBB serve as a platform for neurogenic and oligovascular niches providing optimal microenvironment for neurogenesis and oligodendrogenesis in (patho)physiological conditions. Partially compromised BBB may be rather important for the effective adjustment of niche microenvironment to the metabolic needs of neural stem cells and progenitors: leaky BBB could be an advantage for the population of actively proliferating cells with high intensity of metabolism. In a contrast to the structure of BBB in other brain regions or BBB in microvessels of hippocampal subgranular zone (SGZ), vascular scaffold in the subventricular zone (SVZ) is characterized by less expression of tight junction proteins and aquaporin-4 (AQP4), probably, for the direct contact of endothelium with stem or progenitors cells. Hence, BBB in this neurogenic niche might be functionally defective (Pozhilenkova et al. 2017), presumably, for maintaining the number of quiescent stem cells and preventing depletion of their pool (Ottone et al. 2014). Recent data reveal that pericytes play an important role in controlling stem cells proliferation within SVZ (Crouch et al. 2015). In SGZ, pericytes act in a coordination with astrocytes and endothelial cells being, probably, mainly involved in the regulation of stem cells adhesion (Ehret et al. 2015). However, how pericytes dysfunction may affect adult neurogenesis in Alzheimer’s disease is not clear yet and requires scrupulous investigation.
In oligovascular niches, pericytes contact to oligodendrocyte progenitor cells (OPCs) and these two cell populations mutually regulate proliferation and support survival of each other in the perivascular region (Maki et al. 2015). Thus, pericytes may control the process of oligodendrogenesis and myelinization of axons in newly formed neurons. Since oligodendrocyte pathology is a very early sign of AD (Desai et al. 2010), one may assume that oligovascular niches might be compromised due to insufficient pericyte support of OPCs development.
In sum, pericyte dysfunction in AD results in the induction of aberrant angiogenesis, pathological hypervascularity, disorganized microvasculature, and leaky BBB. AD-affected pericytes that should control neurogenesis and oligodendrogenesis lose their ability to support stem cells population dynamics, thereby contributing to progressive cognitive deficits. Figure 7.4 shows mechanisms of pericyte-mediated control of BBB integrity, angiogenesis and neurogenesis in Alzheimer’s disease.
Pericyte-mediated control of BBB integrity, angiogenesis and neurogenesis in Alzheimer’s disease. Alterations of BBB structural and functional integrity caused by endothelial and pericyte dysfunction result in the impairment of neurogenic and oligovascular niches activity. In sum, aberrant neurogenesis, oligodendrogenesis and angiogenesis underlie neuroplasticity alterations and progressive cognitive decline seen in chronic neurodegeneration
Summary and Future Prospects
Deciphering a role of pericytes in the regulation of key mechanisms within the NVU, incl. gliovascular control, BBB integrity, immune defense etc., suggests novel approaches to the treatment of CNS disorders associated with NVU dysfunction (neurodegeneration, stroke, trauma, neuroinfection). As an example, contractile pericytes in the brain vessels serve as cellular mediators of no-reflow phenomenon seen in ischemic brain tissue (O’Farrell et al. 2017); therefore, targeting pericytes in brain microvasculature could be helpful in restoring adequate blood supply in the ischemic regions. Such effects could be achieved by modulating pericytes response to neurotransmitters and gliotransmitters (serotonin, adenosine) (Li et al. 2017). Another intriguing possibility is to control PDGF/PDGFR2 and Angiopoietin-1/Tie2 signaling cascades in cerebral pericytes in a similar manner as was shown in retina (Arboleda-Velasquez et al. 2015) or in tumor tissue (Kang and Shin 2016) to stimulate or to reduce angiogenesis. This approach has not been tested in neurodegeneration, however, high degree of pericytes plasticity makes them very attractive tool for the promotion of CNS repair in neurodegenerative diseases (Lange et al. 2013). Finally, pericytes serve as a functional part of NVU/BBB models in vitro utilized for studying pharmacokinetics of novel drug candidates (Wang et al. 2016) or as a cell component for bioengineered constructs with a great potential in the regenerative medicine (Avolio et al. 2017).
References
Ahmed, K., Tunaru, S., Tang, C., Muller, M., Gille, A., Sassmann, A., Hanson, J., & Offermanns, S. (2010). An autocrine lactate loop mediates insulin-dependent inhibition of lipolysis through GPR81. Cell Metabolism, 11, 311–319.
An, Y., Varma, V. R., Varma, S., Casanova, R., Dammer, E., Pletnikova, O., Chia, C. W., Egan, J. M., Ferrucci, L., Troncoso, J., Levey, A. I., Lah, J., Seyfried, N. T., Legido-Quigley, C., O’Brien, R., & Thambisetty, M. (2018). Evidence for brain glucose dysregulation in Alzheimer’s disease. Alzheimer’s & Dementia, 14, 318–329.
Arboleda-Velasquez, J. F., Valdez, C., Marko, C. K., & D’Amore, P. A. (2015). From pathobiology to the targeting of pericytes for the treatment of diabetic retinopathy. Current Diabetes Reports, 15, 573–573.
Armulik, A., Genové, G., & Betsholtz, C. (2011). Pericytes: Developmental, physiological, and pathological perspectives, problems, and promises. Developmental Cell, 21, 193–215.
Attwell, D., Mishra, A., Hall, C. N., O’Farrell, F. M., & Dalkara, T. (2016). What is a pericyte? Journal of Cerebral Blood Flow & Metabolism, 36, 451–455.
Avolio, E., Alvino, V. V., Ghorbel, M. T., & Campagnolo, P. (2017). Perivascular cells and tissue engineering: Current applications and untapped potential. Pharmacology & Therapeutics, 171, 83–92.
Banks, W. A., Owen, J. B., & Erickson, M. A. (2012). Insulin in the brain: There and back again. Pharmacology & Therapeutics, 136, 82–93.
Bell, R. D., Winkler, E. A., Singh, I., Sagare, A. P., Deane, R., Wu, Z., Holtzman, D. M., Betsholtz, C., Armulik, A., Sallstrom, J., Berk, B. C., & Zlokovic, B. V. (2012). Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature, 485, 512–516.
Benjamin, L. E., Hemo, I., & Keshet, E. (1998). A plasticity window for blood vessel remodelling is defined by pericyte coverage of the preformed endothelial network and is regulated by PDGF-B and VEGF. Development, 125, 1591–1598.
Bergers, G., & Song, S. (2005). The role of pericytes in blood-vessel formation and maintenance. Neuro-Oncology, 7, 452–464.
Berthiaume, A. A., Grant, R. I., McDowell, K. P., Underly, R. G., Hartmann, D. A., Levy, M., Bhat, N. R., & Shih, A. Y. (2018). Dynamic remodeling of pericytes in vivo maintains capillary coverage in the adult mouse brain. Cell Reports, 22, 8–16.
Biffi, A., & Greenberg, S. M. (2011). Cerebral amyloid angiopathy: A systematic review. Journal of Clinical Neurology, 7, 1–9.
Birbrair, A., Zhang, T., Wang, Z. M., Messi, M. L., Mintz, A., & Delbono, O. (2013). Type-1 pericytes participate in fibrous tissue deposition in aged skeletal muscle. American Journal of Physiology. Cell Physiology, 305, C1098–C1113.
Birbrair, A., Zhang, T., Files, D. C., Mannava, S., Smith, T., Wang, Z. M., Messi, M. L., Mintz, A., & Delbono, O. (2014a). Type-1 pericytes accumulate after tissue injury and produce collagen in an organ-dependent manner. Stem Cell Research & Therapy, 5, 122.
Birbrair, A., Zhang, T., Wang, Z.-M., Messi, M. L., Mintz, A., & Delbono, O. (2014b). Pericytes: Multitasking cells in the regeneration of injured, diseased, and aged skeletal muscle. Frontiers in Aging Neuroscience, 6, 245.
Birbrair, A., Zhang, T., Wang, Z. M., Messi, M. L., Olson, J. D., Mintz, A., & Delbono, O. (2014c). Type-2 pericytes participate in normal and tumoral angiogenesis. American Journal of Physiology. Cell Physiology, 307, C25–C38.
Biron, K. E., Dickstein, D. L., Gopaul, R., & Jefferies, W. A. (2011). Amyloid triggers extensive cerebral angiogenesis causing blood brain barrier permeability and hypervascularity in Alzheimer’s disease. PLoS One, 6, e23789.
Biron, K. E., Dickstein, D. L., Gopaul, R., Fenninger, F., & Jefferies, W. A. (2013). Cessation of neoangiogenesis in Alzheimer’s disease follows amyloid-beta immunization. Scientific Reports, 3, 1354.
Blocki, A., Wang, Y., Koch, M., Peh, P., Beyer, S., Law, P., Hui, J., & Raghunath, M. (2013). Not all MSCs can act as pericytes: Functional in vitro assays to distinguish pericytes from other mesenchymal stem cells in angiogenesis. Stem Cells and Development, 22, 2347–2355.
Bodnar, R. J., Rodgers, M. E., Chen, W. C. W., & Wells, A. (2013). Pericyte regulation of vascular remodeling through the CXC receptor 3. Arteriosclerosis, Thrombosis, and Vascular Biology, 33, 2818–2829.
Cantelmo, A. R., Conradi, L.-C., Brajic, A., Goveia, J., Kalucka, J., Pircher, A., Chaturvedi, P., Hol, J., Thienpont, B., Teuwen, L.-A., Schoors, S., Boeckx, B., Vriens, J., Kuchnio, A., Veys, K., Cruys, B., Finotto, L., Treps, L., Stav-Noraas, T. E., Bifari, F., Stapor, P., Decimo, I., Kampen, K., De Bock, K., Haraldsen, G., Schoonjans, L., Rabelink, T., Eelen, G., Ghesquière, B., Rehman, J., Lambrechts, D., Malik, A. B., Dewerchin, M., & Carmeliet, P. (2016). Inhibition of the glycolytic activator pfkfb3 in endothelial cells induces tumor vessel normalization, impairs metastasis and improves chemotherapy. Cancer Cell, 30, 968–985.
Casey, C. S., Atagi, Y., Yamazaki, Y., Shinohara, M., Tachibana, M., Fu, Y., Bu, G., & Kanekiyo, T. (2015). Apolipoprotein E inhibits cerebrovascular pericyte mobility through a RhoA protein-mediated pathway. The Journal of Biological Chemistry, 290, 14208–14217.
Chen, J., Luo, Y., Hui, H., Cai, T., Huang, H., Yang, F., Feng, J., Zhang, J., & Yan, X. (2017). CD146 coordinates brain endothelial cell–pericyte communication for blood–brain barrier development. Proceedings of the National Academy of Sciences, 114, E7622–E7631.
Crouch, E. E., Liu, C., Silva-Vargas, V., & Doetsch, F. (2015). Regional and stage-specific effects of prospectively purified vascular cells on the adult V-SVZ neural stem cell lineage. The Journal of Neuroscience, 35, 4528–4539.
Cruys, B., Wong, B. W., Kuchnio, A., Verdegem, D., Cantelmo, A. R., Conradi, L.-C., Vandekeere, S., Bouché, A., Cornelissen, I., Vinckier, S., Merks, R. M. H., Dejana, E., Gerhardt, H., Dewerchin, M., Bentley, K., & Carmeliet, P. (2016). Glycolytic regulation of cell rearrangement in angiogenesis. Nature Communications, 7, 12240.
de la Torre, J. C. (2004). Is Alzheimer’s disease a neurodegenerative or a vascular disorder? Data, dogma, and dialectics. Lancet Neurology, 3, 184–190.
Desai, M. K., Mastrangelo, M. A., Ryan, D. A., Sudol, K. L., Narrow, W. C., & Bowers, W. J. (2010). Early oligodendrocyte/myelin pathology in Alzheimer’s disease mice constitutes a novel therapeutic target. The American Journal of Pathology, 177, 1422–1435.
Dias Moura Prazeres, P. H., Sena, I. F. G., Borges, I. D. T., de Azevedo, P. O., Andreotti, J. P., de Paiva, A. E., de Almeida, V. M., de Paula Guerra, D. A., Pinheiro Dos Santos, G. S., Mintz, A., Delbono, O., & Birbrair, A. (2017). Pericytes are heterogeneous in their origin within the same tissue. Developmental Biology, 427, 6–11.
Ding, X., Zhang, M., Gu, R., Xu, G., & Wu, H. (2017). Activated microglia induce the production of reactive oxygen species and promote apoptosis of co-cultured retinal microvascular pericytes. Graefe’s Archive for Clinical and Experimental Ophthalmology, 255, 777–788.
Ehret, F., Vogler, S., & Kempermann, G. (2015). A co-culture model of the hippocampal neurogenic niche reveals differential effects of astrocytes, endothelial cells and pericytes on proliferation and differentiation of adult murine precursor cells. Stem Cell Research, 15, 514–521.
Eilken, H. M., Dieguez-Hurtado, R., Schmidt, I., Nakayama, M., Jeong, H. W., Arf, H., Adams, S., Ferrara, N., & Adams, R. H. (2017). Pericytes regulate VEGF-induced endothelial sprouting through VEGFR1. Nature Communications, 8, 1574.
Escudero, C. A., Herlitz, K., Troncoso, F., Guevara, K., Acurio, J., Aguayo, C., Godoy, A. S., & Gonzalez, M. (2017). Pro-angiogenic role of insulin: From physiology to pathology. Frontiers in Physiology, 8, 204.
Franco, M., Roswall, P., Cortez, E., Hanahan, D., & Pietras, K. (2011). Pericytes promote endothelial cell survival through induction of autocrine VEGF-A signaling and Bcl-w expression. Blood, 118, 2906–2917.
Giannoni, P., Arango-Lievano, M., Neves, I. D., Rousset, M. C., Baranger, K., Rivera, S., Jeanneteau, F., Claeysen, S., & Marchi, N. (2016). Cerebrovascular pathology during the progression of experimental Alzheimer’s disease. Neurobiology of Disease, 88, 107–117.
Grammas, P., Moore, P., & Weigel, P. H. (1999). Microvessels from Alzheimer’s disease brains kill neurons in vitro. The American Journal of Pathology, 154, 337–342.
Greenwood-Goodwin, M., Yang, J., Hassanipour, M., & Larocca, D. (2016). A novel lineage restricted, pericyte-like cell line isolated from human embryonic stem cells. Scientific Reports, 6, 24403.
Hall, C. N., Reynell, C., Gesslein, B., Hamilton, N. B., Mishra, A., Sutherland, B. A., O’Farrell, F. M., Buchan, A. M., Lauritzen, M., & Attwell, D. (2014). Capillary pericytes regulate cerebral blood flow in health and disease. Nature, 508, 55–60.
Halliday, M. R., Rege, S. V., Ma, Q., Zhao, Z., Miller, C. A., Winkler, E. A., & Zlokovic, B. V. (2016). Accelerated pericyte degeneration and blood-brain barrier breakdown in apolipoprotein E4 carriers with Alzheimer’s disease. Journal of Cerebral Blood Flow and Metabolism, 36, 216–227.
Han, B. H., Zhou, M. L., Johnson, A. W., Singh, I., Liao, F., Vellimana, A. K., Nelson, J. W., Milner, E., Cirrito, J. R., Basak, J., Yoo, M., Dietrich, H. H., Holtzman, D. M., & Zipfel, G. J. (2015). Contribution of reactive oxygen species to cerebral amyloid angiopathy, vasomotor dysfunction, and microhemorrhage in aged Tg2576 mice. Proceedings of the National Academy of Sciences of the United States of America, 112, E881–E890.
Herland, A., van der Meer, A. D., FitzGerald, E. A., Park, T.-E., Sleeboom, J. J. F., & Ingber, D. E. (2016). Distinct contributions of astrocytes and pericytes to neuroinflammation identified in a 3D human blood-brain barrier on a chip. PLoS One, 11, e0150360.
Hughes, S., Gardiner, T., Hu, P., Baxter, L., Rosinova, E., & Chan-Ling, T. (2006). Altered pericyte-endothelial relations in the rat retina during aging: Implications for vessel stability. Neurobiology of Aging, 27, 1838–1847.
Ito, S., Yanai, M., Yamaguchi, S., Couraud, P. O., & Ohtsuki, S. (2017). Regulation of tight-junction integrity by insulin in an in vitro model of human blood-brain barrier. Journal of Pharmaceutical Sciences, 106, 2599–2605.
Jellinger K. A. (2002). Alzheimer disease and cerebrovascular pathology: an update. Journal of Neural Transmission (Vienna), 109(5–6), 813–836.
Kang, E., & Shin, J. W. (2016). Pericyte-targeting drug delivery and tissue engineering. International Journal of Nanomedicine, 11, 2397–2406.
Kapogiannis, D., & Reiter, D. (2014). Low glucose utilization and high lactate production in the Alzheimer’s disease brain. Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, 10, P62.
Karow, M., Sanchez, R., Schichor, C., Masserdotti, G., Ortega, F., Heinrich, C., Gascon, S., Khan, M. A., Lie, D. C., Dellavalle, A., Cossu, G., Goldbrunner, R., Gotz, M., & Berninger, B. (2012). Reprogramming of pericyte-derived cells of the adult human brain into induced neuronal cells. Cell Stem Cell, 11, 471–476.
Khilazheva, E. D., Pisareva, N. V., Morgun, A. V., Boytsova, E. B., Taranushenko, T. E., Frolova, O. V., & Salmina, A. B. (2017). Activation of GPR81 lactate receptors stimulates mitochondrial biogenesis in cerebral microvessel endothelial cells. Annals of Clinical and Experimental Neurology, 11, 34–39.
Kisler, K., Nelson, A. R., Montagne, A., & Zlokovic, B. V. (2017a). Cerebral blood flow regulation and neurovascular dysfunction in Alzheimer disease. Nature Reviews Neuroscience, 18, 419–434.
Kisler, K., Nelson, A. R., Rege, S. V., Ramanathan, A., Wang, Y., Ahuja, A., Lazic, D., Tsai, P. S., Zhao, Z., Zhou, Y., Boas, D. A., Sakadžić, S., & Zlokovic, B. V. (2017b). Pericyte degeneration leads to neurovascular uncoupling and limits oxygen supply to brain. Nature Neuroscience, 20, 406–416.
Kovac, A., Erickson, M. A., & Banks, W. A. (2011). Brain microvascular pericytes are immunoactive in culture: Cytokine, chemokine, nitric oxide, and LRP-1 expression in response to lipopolysaccharide. Journal of Neuroinflammation, 8, 139–139.
Lange, S., Trost, A., Tempfer, H., Bauer, H.-C., Bauer, H., Rohde, E., Reitsamer, H. A., Franklin, R. J. M., Aigner, L., & Rivera, F. J. (2013). Brain pericyte plasticity as a potential drug target in CNS repair. Drug Discovery Today, 18, 456–463.
Lauritzen, K. H., Morland, C., Puchades, M., Holm-Hansen, S., Hagelin, E. M., Lauritzen, F., Attramadal, H., Storm-Mathisen, J., Gjedde, A., & Bergersen, L. H. (2014). Lactate receptor sites link neurotransmission, neurovascular coupling, and brain energy metabolism. Cerebral Cortex, 24, 2784–2795.
Li, Y., Lucas-Osma, A. M., Black, S., Bandet, M. V., Stephens, M. J., Vavrek, R., Sanelli, L., Fenrich, K. K., Di Narzo, A. F., Dracheva, S., Winship, I. R., Fouad, K., & Bennett, D. J. (2017). Pericytes impair capillary blood flow and motor function after chronic spinal cord injury. Nature Medicine, 23, 733–741.
Liguori, C., Stefani, A., Sancesario, G., Sancesario, G. M., Marciani, M. G., & Pierantozzi, M. (2015). CSF lactate levels, tau proteins, cognitive decline: A dynamic relationship in Alzheimer’s disease. Journal of Neurology, Neurosurgery, and Psychiatry, 86, 655–659.
Liu, H., Saffi, G. T., Vasefi, M. S., Choi, Y., Kruk, J. S., Ahmed, N., Gondora, N., Mielke, J., Leonenko, Z., & Beazely, M. A. (2018). Amyloid-beta inhibits PDGFbeta receptor activation and prevents PDGF-BB-induced neuroprotection. Current Alzheimer Research, 15, 618–627.
Maki, T., Maeda, M., Uemura, M., Lo, E. K., Terasaki, Y., Liang, A. C., Shindo, A., Choi, Y. K., Taguchi, A., Matsuyama, T., Takahashi, R., Ihara, M., & Arai, K. (2015). Potential interactions between pericytes and oligodendrocyte precursor cells in perivascular regions of cerebral white matter. Neuroscience Letters, 597, 164–169.
Mangialardi, G., Cordaro, A., & Madeddu, P. (2016). The bone marrow pericyte: An orchestrator of vascular niche. Regenerative Medicine, 11, 883–895.
Mayo, J. N., & Bearden, S. E. (2015). Driving the hypoxia inducible pathway in human pericytes promotes vascular density in an exosome dependent manner. Microcirculation (New York, NY), 22, 711–723.
Miners, J. S., Schulz, I., & Love, S. (2018). Differing associations between Abeta accumulation, hypoperfusion, blood-brain barrier dysfunction and loss of PDGFRB pericyte marker in the precuneus and parietal white matter in Alzheimer’s disease. Journal of Cerebral Blood Flow and Metabolism, 38, 103–115.
Montagne, A., Barnes, S. R., Sweeney, M. D., Halliday, M. R., Sagare, A. P., Zhao, Z., Toga, A. W., Jacobs, R. E., Liu, C. Y., Amezcua, L., Harrington, M. G., Chui, H. C., Law, M., & Zlokovic, B. V. (2015). Blood-brain barrier breakdown in the aging human hippocampus. Neuron, 85, 296–302.
Morland, C., Andersson, K. A., Haugen, Ø. P., Hadzic, A., Kleppa, L., Gille, A., Rinholm, J. E., Palibrk, V., Diget, E. H., Kennedy, L. H., Stølen, T., Hennestad, E., Moldestad, O., Cai, Y., Puchades, M., Offermanns, S., Vervaeke, K., Bjørås, M., Wisløff, U., Storm-Mathisen, J., & Bergersen, L. H. (2017). Exercise induces cerebral VEGF and angiogenesis via the lactate receptor HCAR1. Nature Communications, 8, 15557.
Nikolakopoulou, A. M., Zhao, Z., Montagne, A., & Zlokovic, B. V. (2017). Regional early and progressive loss of brain pericytes but not vascular smooth muscle cells in adult mice with disrupted platelet-derived growth factor receptor-β signaling. PLoS One, 12, e0176225.
O’Farrell, F. M., Mastitskaya, S., Hammond-Haley, M., Freitas, F., Wah, W. R., & Attwell, D. (2017). Capillary pericytes mediate coronary no-reflow after myocardial ischaemia. eLife, 6, e29280.
Ottone, C., Krusche, B., Whitby, A., Clements, M., Quadrato, G., Pitulescu, M. E., Adams, R. H., & Parrinello, S. (2014). Direct cell-cell contact with the vascular niche maintains quiescent neural stem cells. Nature Cell Biology, 16, 1045–1056.
Piegsa, J., Miners, S., Love, S., Shore, A., Tamagnini, F., & Randall, A. (2017). Ca2+ signalling in human brain pericytes: Investigation of effects of amyloid beta treatment. Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, 13, P331.
Pluta, R., Jabłoński, M., Ułamek-Kozioł, M., Kocki, J., Brzozowska, J., Januszewski, S., Furmaga-Jabłońska, W., Bogucka-Kocka, A., Maciejewski, R., & Czuczwar, S. J. (2013). Sporadic Alzheimer’s disease begins as episodes of brain ischemia and ischemically dysregulated Alzheimer’s disease genes. Molecular Neurobiology, 48, 500–515.
Pozhilenkova, E. A., Lopatina, O. L., Komleva, Y. K., Salmin, V. V., & Salmina, A. B. (2017). Blood-brain barrier-supported neurogenesis in healthy and diseased brain. Reviews in the Neurosciences, 28, 397–415.
Rensink, A. A., Otte-Holler, I., de Boer, R., Bosch, R. R., ten Donkelaar, H. J., de Waal, R. M., Verbeek, M. M., & Kremer, B. (2004a). Insulin inhibits amyloid beta-induced cell death in cultured human brain pericytes. Neurobiology of Aging, 25, 93–103.
Rensink, A. A., Otte-Holler, I., ten Donkelaar, H. J., De Waal, R. M., Kremer, B., & Verbeek, M. M. (2004b). Differential gene expression in human brain pericytes induced by amyloid-beta protein. Neuropathology and Applied Neurobiology, 30, 279–291.
Ribatti, D., Nico, B., & Crivellato, E. (2011). The role of pericytes in angiogenesis. The International Journal of Developmental Biology, 55, 261–268.
Rustenhoven, J., Aalderink, M., Scotter, E. L., Oldfield, R. L., Bergin, P. S., Mee, E. W., Graham, E. S., Faull, R. L. M., Curtis, M. A., Park, T. I. H., & Dragunow, M. (2016). TGF-beta1 regulates human brain pericyte inflammatory processes involved in neurovasculature function. Journal of Neuroinflammation, 13, 37.
Sagare, A. P., Bell, R. D., Zhao, Z., Ma, Q., Winkler, E. A., Ramanathan, A., & Zlokovic, B. V. (2013). Pericyte loss influences Alzheimer-like neurodegeneration in mice. Nature Communications, 4, 2932.
Sakuma, R., Kawahara, M., Nakano-Doi, A., Takahashi, A., Tanaka, Y., Narita, A., Kuwahara-Otani, S., Hayakawa, T., Yagi, H., Matsuyama, T., & Nakagomi, T. (2016). Brain pericytes serve as microglia-generating multipotent vascular stem cells following ischemic stroke. Journal of Neuroinflammation, 13, 57.
Salmina, A. B., Inzhutova, A. I., Malinovskaya, N. A., & Petrova, M. M. (2010). Endothelial dysfunction and repair in Alzheimer-type neurodegeneration: Neuronal and glial control. Journal of Alzheimer’s Disease, 22, 17–36.
Salmina, A. B., Morgun, A. V., Kuvacheva, N. V., Lopatina, O. L., Komleva, Y. K., Malinovskaya, N. A., & Pozhilenkova, E. A. (2014). Establishment of neurogenic microenvironment in the neurovascular unit: The connexin 43 story. Reviews in the Neurosciences, 25, 97–111.
Salmina, A. B., Komleva, Y. K., Szijártó, I. A., Gorina, Y. V., Lopatina, O. L., Gertsog, G. E., Filipovic, M. R., & Gollasch, M. (2015a). H(2)S- and NO-signaling pathways in Alzheimer’s amyloid vasculopathy: Synergism or antagonism? Frontiers in Physiology, 6, 361.
Salmina, A. B., Kuvacheva, N. V., Morgun, A. V., Komleva, Y. K., Pozhilenkova, E. A., Lopatina, O. L., Gorina, Y. V., Taranushenko, T. E., & Petrova, L. L. (2015b). Glycolysis-mediated control of blood-brain barrier development and function. The International Journal of Biochemistry & Cell Biology, 64, 174–184.
Simonavicius, N., Ashenden, M., van Weverwijk, A., Lax, S., Huso, D. L., Buckley, C. D., Huijbers, I. J., Yarwood, H., & Isacke, C. M. (2012). Pericytes promote selective vessel regression to regulate vascular patterning. Blood, 120, 1516–1527.
Stapor, P. C., Sweat, R. S., Dashti, D. C., Betancourt, A. M., & Murfee, W. L. (2014). Pericyte dynamics during angiogenesis: New insights from new identities. Journal of Vascular Research, 51, 163–174.
Stefanska, A., Eng, D., Kaverina, N., Duffield, J. S., Pippin, J. W., Rabinovitch, P., & Shankland, S. J. (2015). Interstitial pericytes decrease in aged mouse kidneys. Aging, 7, 370–382.
Sweeney, P. W., Walker-Samuel, S., & Shipley, R. J. (2018). Insights into cerebral haemodynamics and oxygenation utilising in vivo mural cell imaging and mathematical modelling. Scientific Reports, 8, 15.
Szpak, G. M., Lewandowska, E., Wierzba-Bobrowicz, T., Bertrand, E., Pasennik, E., Mendel, T., Stepień, T., Leszczyńska, A., & Rafałowska, J. (2007). Small cerebral vessel disease in familial amyloid and non-amyloid angiopathies: FAD-PS-1 (P117L) mutation and CADASIL. Immunohistochemical and ultrastructural studies. Folia Neuropathologica, 45, 192–204.
Tachibana, M., Yamazaki, Y., Liu, C. C., Bu, G., & Kanekiyo, T. (2018). Pericyte implantation in the brain enhances cerebral blood flow and reduces amyloid-beta pathology in amyloid model mice. Experimental Neurology, 300, 13–21.
Takahashi, H., Takata, F., Matsumoto, J., Machida, T., Yamauchi, A., Dohgu, S., & Kataoka, Y. (2015). Brain pericyte-derived soluble factors enhance insulin sensitivity in GT1-7 hypothalamic neurons. Biochemical and Biophysical Research Communications, 457, 532–537.
Teichert, M., Milde, L., Holm, A., Stanicek, L., Gengenbacher, N., Savant, S., Ruckdeschel, T., Hasanov, Z., Srivastava, K., Hu, J., Hertel, S., Bartol, A., Schlereth, K., & Augustin, H. G. (2017). Pericyte-expressed Tie2 controls angiogenesis and vessel maturation. Nature Communications, 8, 16106.
Thériault, P., ElAli, A., & Rivest, S. (2016). High fat diet exacerbates Alzheimer’s disease-related pathology in APPswe/PS1 mice. Oncotarget, 7, 67808–67827.
Tian, X., Brookes, O., & Battaglia, G. (2017). Pericytes from mesenchymal stem cells as a model for the blood-brain barrier. Scientific Reports, 7, 39676.
Toth, P., Tucsek, Z., Sosnowska, D., Gautam, T., Mitschelen, M., Tarantini, S., Deak, F., Koller, A., Sonntag, W. E., Csiszar, A., & Ungvari, Z. (2013). Age-related autoregulatory dysfunction and cerebromicrovascular injury in mice with angiotensin II-induced hypertension. Journal of Cerebral Blood Flow and Metabolism, 33, 1732–1742.
Trost, A., Lange, S., Schroedl, F., Bruckner, D., Motloch, K. A., Bogner, B., Kaser-Eichberger, A., Strohmaier, C., Runge, C., Aigner, L., Rivera, F. J., & Reitsamer, H. A. (2016). Brain and retinal pericytes: Origin, function and role. Frontiers in Cellular Neuroscience, 10, 20.
Tucsek, Z., Toth, P., Tarantini, S., Sosnowska, D., Gautam, T., Warrington, J. P., Giles, C. B., Wren, J. D., Koller, A., Ballabh, P., Sonntag, W. E., Ungvari, Z., & Csiszar, A. (2014). Aging exacerbates obesity-induced cerebromicrovascular rarefaction, neurovascular uncoupling, and cognitive decline in mice. The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences, 69, 1339–1352.
von Bernhardi, R., Cornejo, F., Parada, G. E., & Eugenín, J. (2015). Role of TGFβ signaling in the pathogenesis of Alzheimer’s disease. Frontiers in Cellular Neuroscience, 9, 426.
Vazquez-Padron, R. I., Lasko, D., Li, S., Louis, L., Pestana, I. A., Pang, M., Liotta, C., Fornoni, A., Aitouche, A., & Pham, S. M. (2004). Aging exacerbates neointimal formation, and increases proliferation and reduces susceptibility to apoptosis of vascular smooth muscle cells in mice. Journal of Vascular Surgery, 40, 1199–1207.
Verbeek, M. M., Otte-Holler, I., Ruiter, D. J., & de Waal, R. M. (1999). Human brain pericytes as a model system to study the pathogenesis of cerebrovascular amyloidosis in Alzheimer’s disease. Cellular and Molecular Biology (Noisy-le-Grand, France), 45, 37–46.
Wang, J. D., Khafagy el, S., Khanafer, K., Takayama, S., & ElSayed, M. E. (2016). Organization of endothelial cells, pericytes, and astrocytes into a 3D microfluidic in vitro model of the -assod-brain barrier. Molecular Pharmaceutics, 13, 895–906.
Warmke, N., Griffin, K. J., Slater, C. I., Walker, A. M. N., Yuldasheva, N. Y., & Cubbon, R. M. (2017). Reduced pericyte insulin signalling causes abnormal developmental angiogenesis. Heart, 103, A127–A128.
Wilhelmus, M. M., Otte-Holler, I., van Triel, J. J., Veerhuis, R., Maat-Schieman, M. L., Bu, G., de Waal, R. M., & Verbeek, M. M. (2007). Lipoprotein receptor-related protein-1 mediates amyloid-beta-mediated cell death of cerebrovascular cells. The American Journal of Pathology, 171, 1989–1999.
Winkler, E. A., Sagare, A. P., & Zlokovic, B. V. (2014). The pericyte: A forgotten cell type with important implications for Alzheimer’s disease? Brain Pathology (Zurich, Switzerland), 24, 371–386.
Yamada, M. (2000). Cerebral amyloid angiopathy: an overview. Neuropathology, 20(1), 8–22.
Yamada, M. (2015). Cerebral amyloid angiopathy: emerging concepts. Journal of Stroke, 17, 17–30.
Yamagishi, S., Takeuchi, M., Matsui, T., Nakamura, K., Imaizumi, T., & Inoue, H. (2005). Angiotensin II augments advanced glycation end product-induced pericyte apoptosis through RAGE overexpression. FEBS Letters, 579, 4265–4270.
Yamamizu, K., Iwasaki, M., Takakubo, H., Sakamoto, T., Ikuno, T., Miyoshi, M., Kondo, T., Nakao, Y., Nakagawa, M., Inoue, H., & Yamashita, J. K. (2017). In vitro modeling of blood-brain barrier with human iPSC-derived endothelial cells, pericytes, neurons, and astrocytes via notch signaling. Stem Cell Reports, 8, 634–647.
Yamanishi, S., Katsumura, K., Kobayashi, T., & Puro, D. G. (2006). Extracellular lactate as a dynamic vasoactive signal in the rat retinal microvasculature. American Journal of Physiology. Heart and Circulatory Physiology, 290, H925–H934.
Acknowledgment
A.B.S., Y.K.K., and O.L.L. are supported by the grant given by the President of Russian Federation for the Leading Scientific Teams (N 6240.2018.7).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Salmina, A.B., Komleva, Y.K., Lopatina, O.L., Birbrair, A. (2019). Pericytes in Alzheimer’s Disease: Novel Clues to Cerebral Amyloid Angiopathy Pathogenesis. In: Birbrair, A. (eds) Pericyte Biology in Disease. Advances in Experimental Medicine and Biology, vol 1147. Springer, Cham. https://doi.org/10.1007/978-3-030-16908-4_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-16908-4_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-16907-7
Online ISBN: 978-3-030-16908-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)