Abstract
Gastrointestinal stromal tumor is the most common mesenchymal neoplasm arising the gastrointestinal tract. The primary tumor ist most common in the stomach (60–70%), followed by the small intestine (20–25%), colon and rectum (5%), and esophagus (less than 5%). The median age at diagnosis is between 60 and 65 years. Histologically, GIST is characterized by its immunopositivity for CD117 (KIT). Clinically, there is a paucity of specific symptoms and a majority of cases becomes symptomatic after local compression caused by tumor mass. Surgery is the main treatment for localized disease. The indication for adjuvant imatinib is based upon risk factors such as primary tumor site, tumor size and number of mitosis. KIT-targeted tyrosine kinase inhibitors (TKI) are the cornerstone for the treatment of metastatic disease. Imatinib is the drug of choice in the first-line setting. Sunitinib, regorafenib, and pazopanib are studied further-line treatment optionse. Immunotherapy studies are ongoing for TKI-refractory patients.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Definition
GIST is the most common mesenchymal tumour in the gastrointestinal tract. GIST is generally characterised by immunopositivity for CD117 (KIT) and arises from interstitial cells of Cajal that are normally part of the autonomic nervous system of the intestine.
2 Epidemiology
GIST represents the most frequent mesenchymal tumour in the gastrointestinal tract, representing 1–3% of gastrointestinal malignancies [1, 2]. The annual incidence of GIST is approximately 15 per million per year [3]. The incidence has dramatically increased in the last decade mostly due to improved histopathologic detection and greater awareness, although the true incidence may also be increasing [4]. More recent data suggest that the frequency of incidentally detected subcentimetre gastric GIST lesions may be much higher than expected [5].
The median age is approximately 60–65 years [6, 7]. However, GIST has been reported in all age groups but is extremely rare in children. In the young subpopulation, GIST represents a distinct subtype, characterised by female predominance and the absence of KIT/platelet-derived growth factor alpha (PDGFRA) mutations [8].
There is no clear predilection for either gender, but some data have suggested a slight male predominance [6].
Although most GISTs appear to be sporadic, less than 5% occur as part of hereditary familial syndromes either with mutations in the KIT gene or in the form of idiopathic multitumour syndromes such as neurofibromatosis type I (NF-1), the Carney triad (GIST, paraganglioma and pulmonary chordomas) and the Carney-Stratakis-syndrome (dyad of GIST and paraganglioma) [9,10,11] (Table 37.1).
In adult patients, approximately 60% of GISTs occur in the stomach and 30% in the small intestine. Other sites of origin are the colon, including the rectum, in approximately 5% and the oesophagus in approximately 1% of adult patients. Rarely, GISTs develop outside the gastrointestinal tract in the mesentery, omentum or retroperitoneum. However, most of those extragastrointestinal GISTs are metastatic or may be detached from a gastrointestinal primary source [13, 14].
3 Histology
3.1 Cellular Origin
Based on their histology, GISTs were originally considered to be derived from smooth muscle. However, they rarely showed clear-cut features of complete muscle differentiation. Additionally, in many cases, their immunophenotypic profile differed from that of leiomyomas arising from other sites (e.g., the uterus or soft tissue). The understanding of GIST biology changed significantly with the identification of the near-universal expression of the CD117 antigen, also known as proto-oncogene c-kit, in GISTs in the late 1980s [15]. At that time, it was shown that the interstitial cells of Cajal that are part of the autonomic nervous system of the intestine and that serve a pacemaker function in controlling motility express the KIT receptor [16]. Interstitial cells of Cajal have immunophenotypic and ultrastructural features of both smooth muscle and neuronal differentiation. Because GISTs, like interstitial cells of Cajal, express KIT, interstitial cells of Cajal are thought to be the cell of origin. Additionally, as two-thirds of GISTs express CD34, it is postulated that GISTs originate from CD34-positive stem cells within the gut wall differentiating toward the pacemaker cell phenotype with time [17, 18].
3.2 Histopathology
The differential diagnosis of a subepithelial tumor arising in the gastrointestinal tract is broad, and histologic findings observed on haematoxylin and eosin-stained sections are not specific for GIST. The cellular morphology of GISTs is mainly divided into three categories, namely the spindle cell type (70%), epithelioid type (20%) and mixed type (10%) [14, 19]. Whereas gastric, small intestinal and colonic GISTs are mostly composed of spindle cell tumours, KIT-negative GISTs are more often of the epithelioid type [20]. The epithelioid variant may show discohesive, hypercellular, sarcomatous morphology with significant atypia and mitotic activity [21].
3.3 Immunohistochemical Features
KIT-positive GIST:
A significant breakthrough was the discovery that most GISTs show strong positivity for CD117 (KIT) in contrast to leiomyomas, true leiomyosarcomas and other spindle-cell tumors of the GI tract, which were typically CD117 negative [22]. CD117 is an antigen that is part of the KIT transmembrane receptor tyrosine kinase (RTK) family and is the product of the KIT proto-oncogene (also denoted c-kit). In more than 80% of GISTs, a mutation in the KIT gene leads to a structural variant of the KIT protein, which is abnormally activated and plays an essential role in cell survival, proliferation and differentiation. When KIT binds to its ligand, it forms a dimer that activates its intrinsic tyrosine kinase activity that, in turn, phosphorylates and activates signal transduction molecules that propagate the signal in the cell (Fig. 37.1).
Activation of KIT. Two KIT receptors normally dimerise in the presence of the ligand stem cell factor (SCF) to initiate downstream signalling (left). Mutations in the receptor cause abnormal constitutive signalling without stimulation from the SCF ligand (right). Hornick JL, MD PhD, Harvard Medical School, Department of Pathology, Boston, MA, and Lazar AJF, MD PhD, Sarcoma Research Center, M. D. Anderson Cancer Center Houston, Texas, reproduced with permission of GIST Support International
Immunohistochemically, most GISTs (>90%) show strong positivity for CD117 and usually negativity for desmin and S-100, which are positive in smooth muscle and neural tumors [23]. Although KIT positivity is a major defining feature for GIST, its expression may not be sufficient for diagnosis. KIT-positive malignancies include metastatic melanoma, angiosarcoma, the Ewing’s sarcoma family of tumours, seminoma, and others [24]. Other commonly expressed markers of GIST include CD34 antigen (70%), smooth muscle actin (SMA; 30–40%), desmin (<5%), and S100 protein (~5%) [25]. In contrast to GIST, leiomyoma and leiomyosarcoma are positive for SMA and desmin and negative for KIT and CD34. Malignant melanoma exhibits diffuse immunoreactivity for S100 protein but can be focally positive for KIT. Schwannomas are strongly and diffusely immunoreactive for S100 protein and negative for KIT [26] (Figs. 37.2 and 37.3).
-
KIT-negative GISTs:
A small subset of GISTs lacks the characteristic KIT mutations [20, 27]. In a proportion of these tumours, activating mutations in the related RTK, PDGFRA, were detected [28]. Many of these PDGFRA-mutant GISTs have an epithelioid morphology. Immunostaining with PDGFRA was shown to be helpful in discriminating between KIT-negative GISTs and other gastrointestinal mesenchymal tumors [29, 30].
DOG1, a calcium-dependent, chloride channel protein, is another highly sensitive and specific marker that often reacts with CD117-negative GISTs [31]. DOG1 expression does not appear to be different between the KIT/PDGFRA mutant or wild-type GISTs. Hence, this marker can be used to diagnose KIT-negative tumour variants.
Inactivation of the succinate dehydrogenase (SDH) complex appears to be an event shared by sporadic and syndromic GISTs that lack mutations in KIT and PDGFRA [32]. Immunohistochemical loss of succinate dehydrogenase subunit B (SDHB) has been shown to be a practical marker to identify SDH-deficient GISTs [33].
The experience with these novel immunomarkers (other than KIT) is currently limited, and problems exist concerning the quality and availability of the commercial antibodies used to stain for them.
3.4 Molecular Pathology
Mutational analysis is an essential diagnostic tool in GIST and plays a key role in the confirmation of the diagnosis and in getting prognostic and predictive, hence treatment-relevant—information.
As noted previously, 95% of adult GISTs overexpress KIT, and approximately one-third of KIT-negative GISTs express DOG1. Therefore, the diagnosis of GIST can be made in most of the cases by observing the macroscopic, microscopic and immunophenotypic characteristics. In cases where the diagnosis of GIST cannot be made based on these features, mutational analysis can be helpful to confirm the diagnosis.
Approximately 80–90% of GISTs have oncogenic mutations, most of them in KIT and approximately 6–8% in the PDGFR oncogene. Both of these genes are located on the 4q12 chromosome and encode receptor tyrosine kinases. These oncogenic mutations are the reason for the constitutive activation (“gain of function”) of the respective proteins, leading to uncontrolled stimulation of KIT- and PDGFR-dependent signalling pathways [22].
KIT mutations mostly affect exon 11 and, less commonly, exon 9, 13, or 17 [34] (Fig. 37.4).
Oncogenic mutations in GISTs include in-frame deletions, missense mutations and tandem duplications. Notably, different mutations are associated with specific tumour locations and maybe clinically more relevant. The prognosis and treatment response correlate with the underlying kinase genotype. Whereas exon 11 mutations are found in virtually every anatomic region, exon 9 mutations are almost exclusively found in intestinal tumours. Tandem duplications are associated with a gastric origin and favourable prognosis. Gastric GISTs with exon 11 deletions have a worse prognosis than those with missense mutations [35, 36]. In terms of the response to systemic therapy, patients with exon 11 mutations are more likely to respond to imatinib than those with other mutations (e.g., in exon 9) or those who lack mutations altogether [37].
PDGFR mutations are mainly located in exons 12, 14, and 18 (Fig. 37.3) [38]. A subset of gastric GISTs, particularly tumours with epithelioid morphology, has these types of mutations. The most common mutation is the point mutation D842V, which is relatively insensitive to imatinib although other GIST subtypes confer sensitivity to this agent [28].
GISTs without KIT and PDGFR mutations have been called “wild-type” GISTs, suggesting that these tumours do not have any mutations.
Recently, some GISTs that lack mutations in KIT/PDGFRA have been shown to have inactivation or a deficiency in the SDH complex. Somatic and germline mutations in the genes encoding for the B, C, and D subunits of the SDH enzyme have been described in children and adults with sporadic GISTs that are wild-type for KIT and PDGFRA and those arising in the setting of the inherited Carney-Stratakis syndrome [32, 39].
In a very small population of “wild-type” GISTs, activating oncogenic mutations in BRAF and KRAS have been detected. The clinical relevance of those subentities is unknown, although few data suggest the activity of BRAF inhibitors [40, 41].
Hence, the definition of “wild-type” GIST is changing, and the presence of different new molecular markers has been confirmed. A new definition of “wild-type” GIST was proposed at the ESMO Sarcoma Conference 2014, defining this cohort as lacking KIT exon 9,11,13, and 17 and PDGFR exon 12,14, and 18 mutations.
4 Clinical Presentation
GISTs are associated with a broad range of symptoms. Although many smaller GISTs are detected incidentally during endoscopy, surgery or radiologic imaging, others present with various symptoms. Symptoms and signs are not disease specific but are related more to the site of disease. The most common clinical features are the following:
-
Vague abdominal complaints (early satiety, bloating, loss of appetite, nausea, vomiting)
-
Fatigue secondary to anaemia
-
Gastrointestinal bleeding
-
Intraperitoneal haemorrhage
-
Symptoms of obstruction
-
Symptoms of tumour perforation
-
Rarely severe hypoglycaemia due to paraneoplastic tumour production of insulin-like growth factor-2 [42].
Recurrence after primary local treatment is mainly intra-abdominal. The most common site of metastasis is the liver, whereas bone, peripheral skin, soft-tissue and pulmonary metastasis occur much less frequently. Similarly, lymph node metastasis is a very rare condition [43].
5 Diagnosis and Staging
The primary investigations before the diagnosis of GIST is made are usually upper or lower endoscopy, abdominal ultrasound or CT. In addition to rectal and liver lesions, where local MRI is much more precise in providing diagnostic and preoperative staging information, the initial modality of choice for staging work-up should include contrast-enhanced abdominal and pelvic CT. The initial work-up should be completed using patient history, routine laboratory testing and chest CT or X-ray [44]. The usual CT appearance of GIST is quite specific and is characterised by a solid, smoothly contoured, soft-tissue mass with heterogeneous enhancement. Larger tumors may include varying degrees of necrosis and haemorrhage [45].
GISTs are positron emission tomography (PET)-avid tumors. Although routine PET for staging and follow-up is not yet recommended, it could be useful to differentiate an active tumor from necrotic or inactive scar tissue, to reveal a small metastasis that would have been missed otherwise and to determine when early detection of the tumor response to tyrosine kinase therapy is of special concern [46, 47].
Obtaining adequate tumor tissue material for definitive diagnosis before surgical resection has been challenging. Because these tumors tend to be soft and friable, biopsy may cause tumour rupture and may be associated with an increased risk for tumor dissemination. Therefore, preoperative biopsy is not generally recommended if the appearance on CT is highly suspicious of GIST, the tumor is resectable tumour, and the patient is operable. Conversely, biopsy might be needed if radiologic characteristics are atypical, and if preoperative therapy is being considered for unresectable or marginally resectable tumors. As percutaneous biopsy carries the theoretical risk of tumor rupture with peritoneal spread of disease, endoscopic ultrasound-guided biopsy is preferred over a percutaneous one [48, 49].
6 Risk Stratification and Stage Classification
Based on three large retrospective trials performed at the Armed Forced Institute of Pathology (AFIP), the tumor size and mitotic rate were identified as the most important prognostic factors [1, 21, 50]. Because this series represents the largest published GIST cohort with long-term follow-up in the preimatinib era, the data formed the foundation for the National Institutes of Health (NIH) consensus approach to risk stratification of GISTs published in 2002 [25].
Subsequently, evaluating long-term follow-up of even more patients, Miettinen et al. suggested new guidelines for the risk stratification, including the primary tumour site as a relevant prognostic factor considering that anatomic location affects the risk for disease recurrence and progression. When using these tools, it is important to appreciate that the mitotic index and tumor size are non-linear continuous variables, so thresholds should be interpreted wisely (Table 37.2).
According to these guidelines, gastric GISTs that are 2 cm or smaller with a mitotic index of 5 or less per 50 HPF can be regarded as essentially benign, but gastric lesions larger than 2 cm with the same mitotic index have a risk for recurrence. Data are lacking on the prognosis of patients with GISTs smaller than 2 cm with a mitotic count of more than 5 per 50 HPF. Additionally, these data confirmed that small intestinal GISTs are more aggressive than gastric GISTs of equal size This risk classification is an accepted and widely used tool and mainly serves to discriminate patients benefiting from adjuvant systemic therapy [13, 51].
A nomogram was recently published by the Memorial Sloan-Kettering Cancer Center that can be used as an alternative to the risk stratification schema described above. The nomogram can quantify the risk of disease recurrence after complete resection as a continuous variable [52].
Tumor rupture, either at surgery or spontaneously, should be regarded an independent risk factor affecting prognosis negatively [53]. Considering this additional risk factor, Joensuu et al. recently proposed a novel, modified risk classification system by generating prognostic heat and contour maps [54] (Fig. 37.5).
Contour maps for estimating the risk of GIST recurrence after surgery. The upper-row maps are used when the tumor rupture status is unknown (a–c), the middle-row maps are used when the tumor has not ruptured (d–f), and the bottom-row maps are used when tumor rupture has occurred (g–i). Red areas depict high risk, blue areas depict low risk, and white areas indicate a lack of data. The percentages associated with each colour (key) indicate the probability of GIST recurrence within the first 10 years of follow-up after surgery. For example, the middle map of the far left column (d) shows that the 10-year risk of GIST recurrence of a patient diagnosed with a 10-cm gastric GIST with five mitoses per 50 high power fields (HPFs) of the microscope and no rupture is 20–40%. The 10-year risk associated with a similar tumour when the mitosis count is ten per 50 HPFs increases to 40–60%. E-GIST extragastrointestinal stromal tumour (arising outside the gastrointestinal tract). (Reprinted with permission from Ref. [54])
Thus far, mutational status has not been incorporated in any risk classification, although some genotypes have a distinct natural history [44, 55] (Fig. 37.6).
Recurrence-free survival in 127 patients with completely resected localized gastrointestinal stromal tumor (GIST) based on the type of mutation. (Reprinted with permission from Ref. [55])
Although the TNM classification was published recently, it does not have a clinical impact due to several limitations and, thus, is not recommended [56].
7 Management of GIST
For optimal management of GIST patients, it is essential to discuss all relevant information, including medical history and laboratory and radiologic findings, within a multidisciplinary team. Pathologists, radiologists, surgeons, and clinical and medical oncologists should be involved in the decision making to ensure the best treatment strategy for each individual with this disease.
7.1 Primary Local Treatment
Complete surgical removal (R0 excision) of localised GISTs is the mainstay of treatment for potentially resectable tumours with a size ≥2 cm [57]. Routine lymph node dissection should not be performed because lymph node metastasis is an extremely rare event [58]. Nevertheless, approximately 50% of GISTs will recur [43]. Resection can be performed by traditional open surgery or laparoscopic surgery, although the latter approach should only be performed by surgeons with expertise in the laparoscopic management of cancer and mainly for gastric primaries [59]. The importance of achieving negative microscopic margins is a controversially discussed issue because a negative impact on OS in patients treated with adjuvant imatinib is lacking. However, R1 resection may be associated with a greater risk for recurrence [60]. A re-resection in a R1 situation is not mandatory but may be carried out if functional sequelae are not expected. Depending on the primary tumour site (oesophago-gastric junction, small intestine, rectum), neoadjuvant treatment with imatinib should be considered (see Sect. 37.7.2).
The natural history of small oesophago-gastric and duodenal lesions smaller than 2 cm in size regarding the growth rate and metastatic potential is difficult to anticipate. Many of these lesions will have a very low risk of tumour progression and a low metastatic potential. Endoscopic biopsy may be difficult, and tumour spillage remains a relevant risk. Hence, endoscopic ultrasound assessment and regular follow-up are reasonable in these cases. Should there be any feature of malignant behaviour on ultrasound a resection should also be performed. An algorithmic approach to the management of gastric GISTs based on size and endoscopic ultrasound (EUS) appearance has been proposed [49] (Fig. 36.7).
Proposed algorithm for the management of localized gastrointestinal stromal cell tumors. (Reprinted with permission from Ref. [49])
7.2 Neoadjuvant Systemic Therapy
The aim of neoadjuvant systemic therapy is to reduce the size of a locally advanced GIST to increase the likelihood of complete resection, reduce surgical morbidity and eventually limit the risk of tumour rupture. Because there are no prospective randomised data, the recommendations on neoadjuvant imatinib therapy are largely based on a few prospective, non-randomised and mainly retrospective studies [61,62,63,64].
Eisenberg and colleagues published a prospective phase II RTOG0132/ACRIN6665 trial investigating the feasibility of neoadjuvant imatinib in KIT-positive, resectable ≥5-cm primary GIST, or resectable, recurrent GIST. Sixty-three patients received 600 mg/day of imatinib for 8–12 weeks prior to surgery and then continued imatinib for 2 additional years. Among the patients with localised primary disease, only two (7%) had an objective response to preoperative imatinib, but stable disease was achieved in 25 (83%) patients. In 77% of these patients, complete resection could be performed. The present study confirmed the safety of administering imatinib neoadjuvantly, although the treatment period was quite short [61]. Another open-label, single-arm phase II study from Canada investigated neoadjuvant imatinib treatment with 400–600 mg daily in patients with locally advanced or metastatic GIST that was potentially resectable. Imatinib was administered for a maximum of 12 months to a maximal tumour response. Six of 14 patients showed a partial response, and eight showed stable disease; no progressive disease was documented. The median treatment duration was 9 months. Therefore, the authors concluded that the optimal preoperative treatment duration should be between 6 months and 12 months [64].
Taken all together, the data reveal that there is no consensus regarding the indications for neoadjuvant therapy because a particularly treatment benefit was not proven. However, preoperative therapy is a widely accepted concept, particularly in large, bulky tumours of any origin and notably in GIST arising in the oesophagus, oesophago-gastric junction, duodenum and distal rectum, to reduce significant surgical morbidity. Importantly, a biopsy to confirm the diagnosis and exclude imatinib-resistant mutations is mandatory. The treatment response to imatinib should be evaluated early during the treatment course to exclude tumour progression and prepone resection.
To date, questions regarding the imatinib dose in patients with exon 9 mutation and the duration of additive adjuvant treatment in this specific situation remain unanswered, but a total duration of 3 years appears reasonable.
7.3 Adjuvant Systemic Therapy
Although surgery remains the therapeutic modality of choice for localised GIST, the risk of recurrence following complete excision is still eminent. In a recently published analysis of a pool of 2560 patients, including 10 different population-based published series, the estimated 5-, 10-, and 15-year relapse-free survival [RFS] rates were 71%, 63%, and 60%, respectively [54]. This meaningful risk of recurrence is likely due to persistent microscopic disease following surgery. Therefore, the effect of adjuvant systemic treatment with imatinib has been explored subsequently to improve the likelihood of survival in patients with a high risk of recurrence. However, there is no clear consensus from expert groups regarding the level or cutoff of recurrence risk that would justify the use of adjuvant imatinib [44].
After a few phase II trials with very promising results, the benefit of adjuvant imatinib therapy has been evaluated in at least 3 randomised studies.
In the multicentre, randomised, double-blind and placebo-controlled US trial Z9001, 713 patients with a resected GIST and a tumour ≥3 cm in size were included and patients were randomly assigned to imatinib 400 mg/day or placebo for 1 year. The study was closed after the first interim analysis, which confirmed a significant reduction in recurrence-free survival that was subsequently the primary endpoint. After a median follow-up of 19.7 months, the 1-year RFS rate was 98 versus 83% favouring imatinib, with a hazard ratio for RFS of 0.35 and a 95% CI of 0.22 to 0.53. A benefit in terms of OS could not be confirmed most likely due to cross-over to active treatment and the short duration of follow-up. Imatinib was well tolerated and showed the known toxicity profile (see below) [65]. That pivotal study led to the accelerated approval of imatinib for the adjuvant treatment of completely resected GISTs ≥3 cm in size. Notably, patients were not stratified according to tumour site and mitotic rate.
The second practise-changing phase III trial was performed by the Scandinavian Sarcoma Group (SSG) XVIII comparing 12 versus 36 months of adjuvant imatinib treatment. Eligible patients were of high risk defined according to the modified consensus criteria as having at least one of the following: a tumor size >10 cm, a mitotic count >10/50 high-power fields (hpf), a tumor size >5 cm with a mitotic rate >5/hpf, or tumour rupture. After recruitment of 400 patients with a median follow-up of 54 months, patients in the 3-year arm showed a significant improvement in RFS, the primary endpoint (5-year RFS, 66 versus 48%; HR, 0.46; 95% CI, 0.32–0.65) as well as overall survival (OS, 92 versus 82%; HR, 0.45; 95% CI, 0.22–0.89). Subgroup analysis demonstrated that patients with exon 9 or PDGFRA mutation did not show a treatment benefit. In summary, these data established at least 36 months of adjuvant imatinib as a new standard for patients with high-risk GIST [66].
Recently, an abstract of the EORTC 62024 study randomising GIST patients between 2 years of adjuvant imatinib and no adjuvant treatment was presented and showed no significant benefit in the primary endpoint, which was imatinib-free survival, under the intermediate- and high-risk scenario [67]. These results per se implicate that progression of GIST may be delayed but survival might not be improved with the available TKIs.
A few outstanding questions need further investigation. First, whereas there is a consensus that PDGFRA D842V-mutated GISTs should not be treated with adjuvant therapy due to their lack of imatinib-sensitivity, the treatment dose in patients with exon 9 mutation is a matter of debate and 800 mg/day of imatinib may be used analogous to the evidence in the metastatic tumour stage. However, there are often regulatory problems limiting this practise. Additionally, we could not confirm whether “wild-type” GISTs also benefit from adjuvant therapy considering their lower sensitivity to imatinib and more indolent natural history [37, 38, 68].
Second, the question remains concerning the optimal treatment duration and whether treatment should be continued for longer than 3 years. In the Scandinavian trial from Joensuu et al., in both groups, within 6–12 months of discontinuation of adjuvant imatinib, the rates of disease recurrence were similarly increased [66]. Similarly, we know from the BFR-14 trial, in patients with advanced GIST, that some patients who had a complete response to imatinib relapsed even after 5 years of treatment when therapy was interrupted therapy [62]. Hence, the latter findings raises questions as to whether recurrences are truly being prevented or just delayed and whether the duration of adjuvant therapy should be beyond 3 years. Currently, a phase II, non-randomised, open-label multicentre study is investigating 5 years of adjuvant imatinib therapy in patients at significant risk for recurrence following complete resection of primary GISTs (NCT00867113).
Additionally, the optimal treatment duration in the case of tumour rupture is unknown given the uncertainty concerning whether these patients should be viewed as virtually metastatic.
Finally, there is no consensus concerning the definition of high-risk GIST, which depends on different risk classifications.
7.4 Systemic Treatment in the Palliative Setting
7.4.1 Cytotoxic Chemotherapy
Until 2000, the diagnosis of GIST was not well defined. Therefore, trials published before that time included a mixture of so-called GISTs, leiomyosarcoma and different other sarcoma subtypes, indicating meaningless clinical activity in these patients. Since then, a few trials have investigated the efficacy of cytotoxic chemotherapy in specific GISTs, confirming a very low response rate of 0–5% [69,70,71]. As such, overall, the data strongly support the lack of benefit of cytotoxic agents for the treatment of GISTs. Hence, the use of cytotoxic agents is not recommended in daily practise.
7.4.2 First-Line Treatment: Imatinib
Imatinib mesylate is a pyrimidine derivative that functions as a specific inhibitor of several tyrosine kinase enzymes, mainly ABL, BCR-ABL, KIT and PDGFR. Imatinib works by binding close to the ATP binding site, locking it and thereby preventing substrate phosphorylation, subsequently leading to the inhibition of signalling pathways involved in proliferation and survival [72, 73].
Many studies have confirmed the impressing benefit of imatinib in metastatic GISTs [74, 75]. The standard dose of imatinib is 400 mg daily. A higher dose level of 600 or 800 mg daily was studied in different randomised trials and have failed to show significantly greater efficacy for higher imatinib doses. Trial data are indicative of more side effects from higher-dose therapy [76,77,78]. One possible explanation for the failure to demonstrate a benefit from higher imatinib doses is interpatient variability in pharmacokinetic exposure. In a study including 73 patients who were randomly assigned to 400 or 600 mg of imatinib daily, there was a tenfold variance in trough levels with either dose. Clinical outcomes were correlated with steady state trough levels. Trough values below 1100 ng/mL were associated with a significantly shorter time to tumor progression and a lower rate of clinical benefit compared with higher trough levels [79, 80].
Another finding in different imatinib trials was the influence of mutations on the treatment response. For example, in the US Intergroup trial comparing 400 with 800 mg of daily imatinib, patients whose tumors expressed an exon 11 mutant isoform were more likely to have an objective response to imatinib compared with those with an exon 9 isoform or those who had no kinase mutations (72 versus 44 and 45%, respectively). Patients with an exon 11 mutation also had a significantly longer time to disease progression (25 versus 17 and 13 months, respectively) and median overall survival (median 60 versus 38 and 49 months, respectively). However, improved response rates were documented for patients with exon 9–mutant tumors treated with imatinib 800 mg versus 400 mg (CR/PR, 67% v 17%; p 0.02) [81].
Additionally, considering PDGFRA mutations, the D842V subtype was shown to be imatinib resistant, whereas other PDGFRA mutations appear to be imatinib sensitive [82].
In summary, most of the international guidelines (NCCN, ESMO) recommend a treatment start of 400 mg of imatinib. Should mutational analysis be available and exon 9 mutation is found, a starting dose of 800 mg is reasonable if covered by the health insurance. Treatment should be continued indefinitely because treatment interruption is generally associated with an early relapse [62]. The median time to progression on imatinib is approximately 2–3 years [76, 77].
The most common side effects of imatinib include the following (Table 37.3):
Most of these side effects are manageable conservatively. For example, nausea can be mitigated by taking the drug with food, which does not seem to interfere with absorption. Diarrhoea can be managed with loperamide. Rashes are often resolved spontaneously with time. Muscle cramps can be reduced by increased oral fluid intake and electrolyte substitution. Fluid retention represents a very common symptom and can be associated with pleural effusion and ascites. Should supportive treatment of this condition be successful, such as a low-salt diet and/or diuretics, no dose reduction is needed. Nevertheless, the latter can potentially lead to severe congestive cardiac failure, which is an uncommon but still a severe side effect [83]. Notably, the toxicity profile may improve with prolonged treatment; importantly, all of these toxicities abate if imatinib is withheld.
The most common haematologic side effects include haematotoxicity and elevated liver function tests. Therefore, regular clinical and laboratory follow-ups are recommended to check the liver parameters. Imatinib is metabolised in the liver by the CYP3A4 enzymatic system. Hence, co-medication with CYP3A4 inhibitors should be avoided, or the imatinib dose should be adapted.
7.4.3 Second-Line Treatment: Imatinib and Sunitinib
Before altering first-line treatment, it is essential to assess patient compliance to imatinib therapy. Any reasons for noncompliance (i.e., depression, asymptomatic disease, side-effects, or cost) should be evaluated carefully, and a solution should be sought to ameliorate regular imatinib intake [84].
In patients with progressing GISTs and manageable side effects, one therapeutic option is to escalate the dose of imatinib to 800 mg. The efficacy of this approach was investigated in the follow-up reports of different trials. Roughly, one-third of patients who were crossed over to the high-dose imatinib regimen achieved either an objective response or stable disease [85].
Patients who are intolerant of imatinib, progress after a very short time on imatinib (a few months) or progress after long-term imatinib therapy should be switched to sunitinib.
Sunitinib malate is another orally administered multi-targeted receptor tyrosine kinase inhibitor of all PDGFR and VEGF receptors and KIT, among a few others. The evidence for its efficacy comes from an international phase III trial of sunitinib versus placebo. This landmark trial included 312 patients with refractory disease, and the median follow-up was 42 months. Despite a low objective response rate in the sunitinib group (7% partial response), the median time to tumour progression, the primary endpoint, was four fold higher than that in the placebo group (27 versus 6 weeks, respectively). The allowance of cross-over for the placebo group was based on the lack of significant difference in overall survival. The median number of weeks on treatment was 22 [86, 87]. Not surprisingly, the clinical activity of sunitinib is significantly influenced by the specific mutational subtype. Clinical benefit (partial response or stable disease for longer than 6 months) was significantly higher for those with a primary KIT 9 exon (58%) or “wild-type” GIST (56%) than for those with a KIT exon 11 mutation (34%) [81].
Therefore, sunitinib was approved for the treatment of imatinib-refractory or intolerant advanced GISTs.
The main side effects are listed in the following table (Table 37.4).
Most of the sunitinib-related side effects are manageable with temporary withdrawal or dose reductions (37.4 or 25 mg/d). Mucositis can usually be treated with supportive measures and avoiding irritating food. With the routine application of emollient lotions, hand-foot-syndrome can be improved or even prevented. Additionally, at follow-ups, the focus should be on the close monitoring of hypertension, heart failure, haematotoxicity, proteinuria, hypothyroidism, gastrointestinal bleeding, bowel perforation and delayed wound healing. In patients with a high cardiovascular risk profile, a baseline echocardiogram should be considered excluding left ventricular dysfunction, which was recorded in approximately 8%. In patients with a history of QT interval prolongation, sunitinib should be used cautiously, and electrolytes should be monitored and substituted if necessary. Hypothyroidism is a very common toxicity recently documented in 62% of GIST patients [88]. Its risk increases with treatment duration. Therefore, TSH levels should be checked every 3–6 months. For planned surgical procedures, sunitinib treatment should be interrupted roughly 1 week before surgery and continued after adequate wound healing has occurred. As sunitinib is also metabolised by CYP3A4, concomitant drug interactions should be evaluated.
7.4.4 Mechanism of Resistance to Imatinib and Sunitinib
The development of drug resistance belongs to the natural history of neoplastic diseases. The armamentarium of tumour cells to survive is immense. Intrinsic (or primary) imatinib resistance is defined as an absence of objective response or disease stabilisation lasting less than 3–6 months. Resistance is most commonly related to the primary GIST genotype and is clinically present in approximately 10–15% of patients. Most of these patients will have imatinib-resistant KIT exon 9 or PDGFRA exon 18 D842V mutations or no detectable mutation [27, 38, 81].
Acquired (or secondary) resistance is observed in initially responding or stable GIST and develops at a median time of 18–24 months. The most commonly identified mechanism is the emergence or acquisition of secondary KIT mutations in exons 13, 14 or 17. These sites represent the ATP binding pocket and kinase activation loop of KIT [81].
Secondary mutations have been identified in 40–80% of tumour biopsy samples obtained from patients progressing on imatinib and are more common when the patient has a primary KIT exon 11 mutation [89,90,91]. Polyclonal resistance mechanisms are commonly identified. Coexisting distinct resistance mutations at an inter-lesional and intra-lesional level have been demonstrated to occur in as many as two-thirds of tested patients [92]. Other identified mechanisms of acquired resistance have included amplification of KIT and pharmacokinetic resistance that may involve altered activity of drug transporters, induction of the cytochrome P450 CYP3A4 isoenzyme, and poor patient compliance [93,94,95].
Resistance to sunitinib shares similar pathogenetic mechanisms to those identified in imatinib failure, with acquisition of secondary mutations after an extended initial response to the drug [96].
7.4.5 Third-Line Treatment: Regorafenib
Regorafenib is another oral TKI targeting a similar spectrum of kinases, including KIT, PDGFR and VEGF receptors. In a phase III trial (GRID trial) including 199 patients, its efficacy was proven. Regorafenib (160 mg once daily for 3 of 4 weeks) was compared with best supportive care (BSC) in patients with advanced GIST following progression or intolerance on imatinib and sunitinib treatment. Regorafenib was shown to improve PFS significantly, 4.8 versus 0.8 months, respectively. Crossover was allowed after progression on placebo (85%). Hence, an OS benefit could not be confirmed. The most common grade 3 side effects were hypertension, hand-foot skin reaction and diarrhoea; however, generally, the toxicities have been shown to be similar to those of other TKIs [97]. Information concerning the potential difference in efficacy regarding mutational status is sparse and very much awaited.
7.4.6 Further-Line Treatment
Various other systemic treatment options showing beneficial efficacy have been tested in recent years. Due to low study evidence, which is based on prospective trials with a small sample size but mainly retrospective data, these other treatment options are rarely available because of regulatory issues.
Nilotinib, another second-generation TKI, was investigated in a randomised phase III trial (400 mg b.i.d.) versus BSC, BSC with imatinib and BSC with sunitinib. In the centrally reviewed intention-to-treat analysis (ITT), no difference in PFS could be noted. Because approximately 20% of the patients had more than two lines of previous treatment, a post-hoc analysis was performed through the third-line setting. Although not powered for this analysis, a significant OS benefit of more than 4 months could be documented for the nilotinib group of patients [98].
Sorafenib, a TKI that inhibits KIT, VEGFR and PDGFR-beta, was shown to be beneficial in terms of the disease control rate (68%) in a phase II trial with either imatinib or imatinib and sunitinib-refractory patients [99]. Additionally, a beneficial effect was also documented in a retrospective cohort in the third and fourth-line settings [100]. Therefore, sorafenib should be suggested as an active drug in further-line treatment.
Pazopanib was investigated in a randomized phase II trial. Eighty-one patients were enrolled. Pazopanib plus best supportive care was compared to best supportive care alone for patients previously treated with imatinib and sunitinib [101]. An improvement in progression-free survival for patients treated with pazopanib could be documented (3.4 months versus 2.3 months; HR 0.59, 95% CI 0.37–0.96). Despite the statistically significant result, the clinical benefit is questionable.
Dewaele and colleagues published in vitro results of dasatinib being remarkably effective for the imatinib-resistant PDGFRA(D842V) mutant isoform [101].
Finally, the question was raised whether imatinib rechallenge after therapy with different TKIs should be supported with the goal to target disease clones that retained sensitivity to imatinib again. The results of a phase III trial showed a significantly greater median PFS for those patients who received imatinib (1.8 versus 0.9 months in the placebo group). Most of the patients were crossed over; hence, the median overall survival was similar in both groups. Although this trial was statistically significantly positive, the results question the clinical relevance of this tiny difference in PFS [102, 103].
7.4.7 Future Perspectives: Immunotherapy (New Section Sugested)
GIST contain tumor-infiltrating lymphocytes and other immune cells that provide an opportunity for developing GIST immunotherapy [105]. The most common tumor-infiltrating inflammatory cells are tumor-associated macrophages (TAMs) and CD3+ T cells. This immune environment should be modulated by imatinib therapy [105]. A study conducted by Balachandran et al. combined imatinib and CTLA-4 inhibitor to treat GIST-bearing mice and found that the therapy significantly decreases tumor size when compared with either treatment alone [106].
The binding of PD-1 on immune cells with PD-L1 on tumor cells inhibits the lymphocytes and it is crucial to the immune escape of neoplasm. Bertucci et al. studied PD-L1 mRNA expression using DNA microarray in 139 untreated localized GISTs and found heterogeneous PD-L1 expression across tumors [107]. PD-L1 expression is higher in low-risk tumors than that in high-risk tumors. As expected, patients with low PD- L1 expression have a higher metastatic risk. Pembrolizumab is a monoclonal antibody against PD-1. A clinical trial of Axitinib, a tyrosine kinase inhibitor, plus pembrolizumab is enrolling patients with advanced sarcomas including GIST (NCT02636725).
7.5 Local Treatment in the Palliative Setting
The role of surgery in metastatic GIST is a controversial issue. There is no randomised data providing a response to whether survival may be lengthened with this approach. However, single-institution retrospective studies document improved long-term disease control compared with historical controls following resection for selected patients with limited metastatic disease and a favourable response to systemic therapy. Additionally, patients with localised progression on systemic treatment seem to benefit from surgery. The rationale behind this approach is to overcome drug resistance and, hence, to eliminate malignant cells with secondary mutations and malignant cells that no longer respond to systemic treatment [104,105,106].
In addition to surgery, other local treatment options to consider, particularly for liver metastasis, are arterial embolisation, chemoembolisation and radiofrequency ablation [107, 108]. Surgery has little to offer in the setting of generalised progression [109, 110].
In summary, lacking clear evidence, surgical treatment in metastatic GIST may be well considered investigational, and a decision should be made by a multidisciplinary team on a case-by-case basis. Furthermore, resection, even if complete, does not eliminate the need for continued treatment with TKI therapy. Progression-free survival is significantly shorter in patients who discontinue treatment than in those who continue the drug after resection.
7.6 Role of Radiotherapy
Until recently, GISTs were indicated to be radioresistant tumour entities. Very little was known concerning the efficacy of radiotherapy in this patient cohort. Several case reports have indicated that radiation can reduce the tumour burden and produce durable local control in locally advanced and metastatic tumours [111]. This impression was confirmed by the reported institutional experience of the Memorial Sloan Kettering Cancer Center and a few others. Heavily pretreated patients with symptomatic tumour manifestations were treated with radiotherapy. At least partial palliation of symptoms was achieved in 94.4% of the tumours, whereas complete disappearance of symptoms was achieved in 44.4% of the tumors. A partial response according to RECIST criteria was observed in 35.3% of tumors, and the response was not assessed using Choi criteria. Stable disease was observed in 52.9% of the tumors [112]. To conclude, this retrospective study shows that radiation is safe and effective and should be considered as a treatment modality in GISTs.
8 Radiologic Response Evaluation
Assessing the treatment response in GISTs is very challenging. RECIST criteria, which define the treatment response by measuring the change in tumour size, have been used for a long time. However, GIST lesions experience different morphological changes on systemic treatment. Not only a change in tumour size but a change in tumour density can occur during the treatment course. Even an increase in size as a consequence of intratumoral haemorrhage or myxoid degeneration could be an early clinical marker of antitumor activity. Therefore, an alternative method to evaluate radiographic response was established in recent years. These criteria, called Choi criteria (see below), include both tumour size and density in the radiographic response evaluation. Choi criteria have been shown to correlate significantly better with either disease-specific survival or time to tumour progression than RECIST. The authors concluded that the tumour response for GISTs should preferentially be categorised by Choi criteria than by RECIST. Choi criteria are based on regular follow-up with CT, MRI or contrast-enhanced ultrasound [113, 114] (Table 37.5).
PET/CT is a very useful tool to visualise GIST lesions because of its high glucose metabolism [115]. Nevertheless, the routine use of PET as a staging procedure or for surveillance after resection is not yet recommended. However, PET is highly sensitive in the early assessment of tumour response, and a decrease in the FDG uptake can be observed as early as 24 h after treatment is initiated [116]. In the neoadjuvant treatment setting of borderline resectable GIST, close monitoring is essential. Hence, in this clinical scenario, baseline and follow-up PET are widely accepted to document treatment efficacy.
9 Follow-Up
There are no published data on what constitutes the optimal routine follow-up after completely resected GISTs, and there is no consensus for this issue. Time to recurrence is mostly dependent on the different prognostic factors such as the mitotic index, tumor site and size. Therefore, risk assessment should guide the choice of the optimal follow-up schedule. High-risk patients generally tend to recur within the first 2 years from the end of adjuvant therapy, whereas low-risk patients may relapse subsequently. For example, the ESMO guidelines recommend CT or MRI every 3–6 months for 3 years during adjuvant therapy for high-risk patients. After cessation of adjuvant imatinib treatment, regular follow-up is suggested to be every 3 months in the first 2 years, every 6 months until 5 years and annually for an additional 5 years from the discontinuation of adjuvant drug treatment. The value of regular follow-up in the low-risk setting remains unclear; however, if carried out, follow-up is suggested to occur every 6–12 months for approximately 5 years. As relapses mainly present with liver and/or peritoneal metastasis, abdominal imaging should be performed with CT or MRI, considering the harmful cumulative X-ray exposure [44].
Multiple-Choice Questions
-
1.
What is the most common patient age at GIST diagnosis?
-
(a)
Up to 20 years
-
(b)
From 20 to 30 years old
-
(c)
From 30 to 40 years old
-
(d)
From 60 to 70 years
-
(e)
Over 70 years
-
(a)
-
The median age at diagnosis is 63 years.
-
2.
Which portion of the gastrointestinal tract is most commonly affected in GIST?
-
(a)
Duodenum
-
(b)
Colon
-
(c)
Straight
-
(d)
Appendix
-
(e)
Stomach
-
(a)
-
60–70% of cases occur in the stomach.
-
3.
Which of the following is positive in GIST?
-
(a)
CD20
-
(b)
CD117
-
(c)
S-100
-
(d)
CD45
-
(e)
OCT4
-
(a)
-
CD117 or KIT is characteristic of GIST.
-
4.
What is the most frequent KIT mutation in GIST?
-
(a)
Exon 9
-
(b)
Exon 8
-
(c)
Exon 11
-
(d)
Exon 13
-
(e)
Exon 17
-
(a)
-
About three-quarters of cases have a mutation in Exon 11.
-
5.
What is the second most common receptor affected in GIST, after KIT?
-
(a)
VEGFR
-
(b)
PDGFRa
-
(c)
EGFR
-
(d)
IGFR
-
(e)
HER2
-
(a)
-
The second most common mutation is in the PDGFRα gene.
-
6.
Which mutation is associated with the best prognosis?
-
(a)
Exon 9
-
(b)
Exon 8
-
(c)
Exon 11
-
(d)
Exon 13
-
(e)
Exon 17
-
(a)
-
Mutation in Exon 11 is associated with an increased overall and progression-free survival.
-
7.
Which mutation benefits from imatinib 800 mg daily in the metastatic setting?
-
(a)
Exon 9
-
(b)
Exon 8
-
(c)
Exon 11
-
(d)
Exon 13
-
(e)
Exon 17
-
(a)
-
Mutation in exon 9 is more sensitive to imatinib in higher doses.
-
8.
Which mutation is resistant to sunitinib?
-
(a)
Exon 9
-
(b)
Exon 8
-
(c)
Exon 11
-
(d)
Exon 13
-
(e)
Exon 17
-
(a)
-
Although its better prognosis, mutation in Exon 11 is more resistant to sunitinib.
-
9.
A 60-year-old patient with a palpable mass in the abdomen underwent biopsy that showed poorly differentiated carcinoma with peritoneal metastasis. He started treatment with FOLFOX. However, a clinical and radiological progression could be documented during chemotherapy. Which immunohistochemistry test could help?
-
(a)
PSA
-
(b)
KIT
-
(c)
S-100
-
(d)
CA 19–9
-
(e)
PLAP
-
(a)
-
Positive KIT confirms diagnosis in virtually all GIST cases and allows treatment changes.
-
10.
Patient with a gastric GIST having the following characteristics: mutation in exon 9 of KIT) 7 cm in size, 8mitosis/50 HPF. What is the recommended adjuvant treatment?
-
(a)
Follow-up
-
(b)
Imatinib 400 mg adjuvant for 6 months
-
(c)
Imatinib 400 mg adjuvant for 3 years
-
(d)
Imatinib adjuvant 800 mg for 3 years
-
(e)
Imatinib 800 mg for 6 months
-
(a)
-
The standard adjuvant treatment is imatinib 400 mg for 3 years. Based on the literature the impact of a higher imatinib dose is unclear.
-
11.
Patient with a small intestine GIST received an emergency surgical treatmentdue to intestinal occlusion. The tumor measured 9 cm and had 3 mitosis/50 HPF. What is the best adjuvant treatment?
-
(a)
Follow-up
-
(b)
Imatinib 400 mg adjuvant for 6 months
-
(c)
Imatinib 400 mg adjuvant for 3 years
-
(d)
Imatinib adjuvant 800 mg for 3 years
-
(e)
Imatinib 800 mg for 6 months
-
(a)
-
Adjuvant treatment is defined by primary site, tumor size and presence of mitosis. Urgent surgery is not a risk factor for recurrence.
Clinical Case
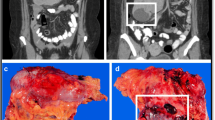
A 65 years old woman presented with a 3 months history of gastric pain. On examination mass in the upper abdomen could be palpated. CT scan reveiled a 15 cm width gastric mass and several peritoneal metastasis. A CT-guided biopsy was performed and the diagnosed a GIST could be made. Histologically, 15 mitosis per HPF could be documented.
Imatinib treatment 400 mg daily was started and tolerated very well. The first response assessment showed stable disease. She was treated with imatinib for 12 months. After 12 months of treatment, the patient presentedwith worsening performance status, abdominal pain and anemia and. A CT scan confirmed disease progression (20 cm on the largest axis).
Consequently, imatinib dose was increased to 800 mg. A short-term disease stabilization could be achieved. After anew GIST progression treatment was modified to sunitinib 50 mg 2 weeks-on and 1 week-off. Two months later, a new CT scan showed a slight increase in the main lesion (to 22 cm); however, the patient was not tolerating the treatment.
Therefore, the treatment was changed to pazopanib 800 mg daily. This clinical case illustrates that GIST therapy senquencing.
Abbreviations
- GIST::
-
Gastrointestinal Stromal Tumor
- PDGFRA::
-
Platelet-derived Growth Factor Receptor Alpha
- NF-1::
-
Neurofibromatosis type I
- SDHB/C/D::
-
Succinate Dehydrogenase Complex Subunit B, C or D
- RTK::
-
Receptor Tyrosine Kinase
- SMA::
-
Smooth Muscle Actin
- SCF::
-
Stem Cell Factor
- DOG1::
-
Discovered on GIST
- SDH::
-
Succinate Dehydrogenase
- ESMO::
-
European Society for Medical Oncology
- CT::
-
Computed Tomography
- MRI::
-
Magnetic Resonance Image
- PET::
-
Positron Emission Tomography
- AFIP::
-
Armed Forced Institute of Pathology
- NIH::
-
National Institutes of Health
- HPF::
-
High Power Fields
- NA::
-
Not Available
- EUS::
-
Endoscopic Ultrasound
- RFS::
-
Relapse-free Survival
- SSG::
-
Scandinavian Sarcoma Group
- CI::
-
Confidence Internal
- ATP::
-
Adenosine Triphosphate
- NCCN::
-
National Cancer Comprehensive Network
- TSH::
-
Thyroid-Stimulating Hormone
- BSC::
-
Best Supportive Care
- TAM::
-
Tumor-associated macrophages
- CTLA-4::
-
Cytotoxic T-lymphocyte Associated Protein 4
- PD-1::
-
Programmed-death Receptor 1
- PD-L1::
-
Programmed-death Receptor Ligand 1
- ITT::
-
Intention-to-treat
- RECIST::
-
Response Evaluation Criteria In Solid Tumors
References
Miettinen M, Lasota J (2001) Gastrointestinal stromal tumors – definition, clinical, histological, immunohistochemical, and molecular genetic features and differential diagnosis. Virchows Arch 438(1):1–12
Thomas RM, Sobin LH (1995) Gastrointestinal cancer. Cancer 75(1 Suppl):154–170
Gatta G et al (2011) Rare cancers are not so rare: the rare cancer burden in Europe. Eur J Cancer 47(17):2493–2511
Steigen SE, Eide TJ (2006) Trends in incidence and survival of mesenchymal neoplasm of the digestive tract within a defined population of northern Norway. APMIS 114(3):192–200
Agaimy A et al (2007) Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. Am J Surg Pathol 31(1):113–120
Tran T, Davila JA, El-Serag HB (2005) The epidemiology of malignant gastrointestinal stromal tumors: an analysis of 1,458 cases from 1992 to 2000. Am J Gastroenterol 100(1):162–168
Tryggvason G et al (2005) Gastrointestinal stromal tumors in Iceland, 1990–2003: the icelandic GIST study, a population-based incidence and pathologic risk stratification study. Int J Cancer 117(2):289–293
Pappo AS, Janeway KA (2009) Pediatric gastrointestinal stromal tumors. Hematol Oncol Clin North Am 23(1):15–34. vii
Maeyama H et al (2001) Familial gastrointestinal stromal tumor with hyperpigmentation: association with a germline mutation of the c-kit gene. Gastroenterology 120(1):210–215
Miettinen M et al (2006) Gastrointestinal stromal tumors in patients with neurofibromatosis 1: a clinicopathologic and molecular genetic study of 45 cases. Am J Surg Pathol 30(1):90–96
Stratakis CA, Carney JA (2009) The triad of paragangliomas, gastric stromal tumours and pulmonary chondromas (Carney triad), and the dyad of paragangliomas and gastric stromal sarcomas (Carney-Stratakis syndrome): molecular genetics and clinical implications. J Intern Med 266(1):43–52
Agarwal R, Robson M (2009) Inherited predisposition to gastrointestinal stromal tumor. Hematol Oncol Clin North Am 23(1):1–13. vii
Miettinen M, Lasota J (2006) Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol 23(2):70–83
Miettinen M, Lasota J (2006) Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med 130(10):1466–1478
Chabot B et al (1988) The proto-oncogene c-kit encoding a transmembrane tyrosine kinase receptor maps to the mouse W locus. Nature 335(6185):88–89
Huizinga JD et al (1995) W/kit gene required for interstitial cells of Cajal and for intestinal pacemaker activity. Nature 373(6512):347–349
Miettinen M, Sobin LH, Sarlomo-Rikala M (2000) Immunohistochemical spectrum of GISTs at different sites and their differential diagnosis with a reference to CD117 (KIT). Mod Pathol 13(10):1134–1142
Sircar K et al (1999) Interstitial cells of Cajal as precursors of gastrointestinal stromal tumors. Am J Surg Pathol 23(4):377–389
Fletcher C, Bridge JA, Hogendoorn PCW, Mertens F (2013) In: Fred T, Bosman ESJ, Lakhani SR, Ohgaki H (eds) WHO classification of tumours of soft tissue and bone, 4th edn. International Agency for Research in Cancer, Lyon
Medeiros F et al (2004) KIT-negative gastrointestinal stromal tumors: proof of concept and therapeutic implications. Am J Surg Pathol 28(7):889–894
Miettinen M, Sobin LH, Lasota J (2005) Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol 29(1):52–68
Hirota S et al (1998) Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science 279(5350):577–580
Rubin BP, Fletcher JA, Fletcher CD (2000) Molecular insights into the histogenesis and pathogenesis of gastrointestinal stromal tumors. Int J Surg Pathol 8(1):5–10
Miettinen M, Lasota J (2005) KIT (CD117): a review on expression in normal and neoplastic tissues, and mutations and their clinicopathologic correlation. Appl Immunohistochem Mol Morphol 13(3):205–220
Fletcher CD et al (2002) Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol 33(5):459–465
Demetri GD et al (2010) NCCN task force report: update on the management of patients with gastrointestinal stromal tumors. J Natl Compr Cancer Netw 8(Suppl 2):S1–S41; quiz S42-4
Debiec-Rychter M et al (2004) Gastrointestinal stromal tumours (GISTs) negative for KIT (CD117 antigen) immunoreactivity. J Pathol 202(4):430–438
Corless CL et al (2005) PDGFRA mutations in gastrointestinal stromal tumors: frequency, spectrum and in vitro sensitivity to imatinib. J Clin Oncol 23(23):5357–5364
Miselli F et al (2008) PDGFRA immunostaining can help in the diagnosis of gastrointestinal stromal tumors. Am J Surg Pathol 32(5):738–743
Peterson MR et al (2006) Strong PDGFRA positivity is seen in GISTs but not in other intra-abdominal mesenchymal tumors: immunohistochemical and mutational analyses. Appl Immunohistochem Mol Morphol 14(4):390–396
Espinosa I et al (2008) A novel monoclonal antibody against DOG1 is a sensitive and specific marker for gastrointestinal stromal tumors. Am J Surg Pathol 32(2):210–218
Pantaleo MA et al (2011) SDHA loss-of-function mutations in KIT-PDGFRA wild-type gastrointestinal stromal tumors identified by massively parallel sequencing. J Natl Cancer Inst 103(12):983–987
Gaal J et al (2011) SDHB immunohistochemistry: a useful tool in the diagnosis of carney-stratakis and carney triad gastrointestinal stromal tumors. Mod Pathol 24(1):147–151
Emile JF et al (2004) Clinicopathologic, phenotypic, and genotypic characteristics of gastrointestinal mesenchymal tumors. Clin Gastroenterol Hepatol 2(7):597–605
Singer S et al (2002) Prognostic value of KIT mutation type, mitotic activity, and histologic subtype in gastrointestinal stromal tumors. J Clin Oncol 20(18):3898–3905
Martin J et al (2005) Deletions affecting codons 557-558 of the c-KIT gene indicate a poor prognosis in patients with completely resected gastrointestinal stromal tumors: a study by the Spanish Group for Sarcoma Research (GEIS). J Clin Oncol 23(25):6190–6198
Heinrich MC et al (2003) Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol 21(23):4342–4349
Heinrich MC et al (2003) PDGFRA activating mutations in gastrointestinal stromal tumors. Science 299(5607):708–710
Janeway KA et al (2011) Defects in succinate dehydrogenase in gastrointestinal stromal tumors lacking KIT and PDGFRA mutations. Proc Natl Acad Sci U S A 108(1):314–318
Agaimy A et al (2009) V600E BRAF mutations are alternative early molecular events in a subset of KIT/PDGFRA wild-type gastrointestinal stromal tumours. J Clin Pathol 62(7):613–616
Falchook GS et al (2013) BRAF mutant gastrointestinal stromal tumor: first report of regression with BRAF inhibitor dabrafenib (GSK2118436) and whole exomic sequencing for analysis of acquired resistance. Oncotarget 4(2):310–315
Pink D et al (2005) Severe hypoglycemia caused by paraneoplastic production of IGF-II in patients with advanced gastrointestinal stromal tumors: a report of two cases. J Clin Oncol 23(27):6809–6811
DeMatteo RP et al (2000) Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg 231(1):51–58
ESMO / European Sarcoma Network Working Group (2012) Gastrointestinal stromal tumors: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 23(Suppl 7):vii49–vii55
Choi YR et al (2014) Differentiation of large (>/= 5 cm) gastrointestinal stromal tumors from benign subepithelial tumors in the stomach: radiologists’ performance using CT. Eur J Radiol 83(2):250–260
Beham AW et al (2012) Gastrointestinal stromal tumors. Int J Color Dis 27(6):689–700
Young H et al (1999) Measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. European Organization for Research and Treatment of Cancer (EORTC) PET Study Group. Eur J Cancer 35(13):1773–1782
Akahoshi K et al (2007) Preoperative diagnosis of gastrointestinal stromal tumor by endoscopic ultrasound-guided fine needle aspiration. World J Gastroenterol 13(14):2077–2082
Sepe PS, Brugge WR (2009) A guide for the diagnosis and management of gastrointestinal stromal cell tumors. Nat Rev Gastroenterol Hepatol 6(6):363–371
Miettinen M et al (2006) Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol 30(4):477–489
Emory TS et al (1999) Prognosis of gastrointestinal smooth-muscle (stromal) tumors: dependence on anatomic site. Am J Surg Pathol 23(1):82–87
Gold JS et al (2009) Development and validation of a prognostic nomogram for recurrence-free survival after complete surgical resection of localised primary gastrointestinal stromal tumour: a retrospective analysis. Lancet Oncol 10(11):1045–1052
Hohenberger P et al (2010) Pattern of recurrence in patients with ruptured primary gastrointestinal stromal tumour. Br J Surg 97(12):1854–1859
Joensuu H et al (2012) Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol 13(3):265–274
Dematteo RP et al (2008) Tumor mitotic rate, size, and location independently predict recurrence after resection of primary gastrointestinal stromal tumor (GIST). Cancer 112(3):608–615
Edge S (2010) AJCC cancer staging manual, 7th edn. Springer, New York
Casali PG et al (2009) Gastrointestinal stromal tumours: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 20(Suppl 4):64–67
Pidhorecky I et al (2000) Gastrointestinal stromal tumors: current diagnosis, biologic behavior, and management. Ann Surg Oncol 7(9):705–712
Huguet KL et al (2008) Laparoscopic gastric gastrointestinal stromal tumor resection: the mayo clinic experience. Arch Surg 143(6):587–590; discussion 591
Everett M, Gutman H (2008) Surgical management of gastrointestinal stromal tumors: analysis of outcome with respect to surgical margins and technique. J Surg Oncol 98(8):588–593
Eisenberg BL et al (2009) Phase II trial of neoadjuvant/adjuvant imatinib mesylate (IM) for advanced primary and metastatic/recurrent operable gastrointestinal stromal tumor (GIST): early results of RTOG 0132/ACRIN 6665. J Surg Oncol 99(1):42–47
Blay JY et al (2007) Prospective multicentric randomized phase III study of imatinib in patients with advanced gastrointestinal stromal tumors comparing interruption versus continuation of treatment beyond 1 year: the French Sarcoma Group. J Clin Oncol 25(9):1107–1113
Wang D et al (2012) Phase II trial of neoadjuvant/adjuvant imatinib mesylate for advanced primary and metastatic/recurrent operable gastrointestinal stromal tumors: long-term follow-up results of Radiation Therapy Oncology Group 0132. Ann Surg Oncol 19(4):1074–1080
Doyon C et al (2012) Prolonged therapy with imatinib mesylate before surgery for advanced gastrointestinal stromal tumor results of a phase II trial. Int J Surg Oncol 2012:761576
Dematteo RP et al (2009) Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet 373(9669):1097–1104
Joensuu H et al (2012) One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA 307(12):1265–1272
Casali PG, Le Cesne A, Velasco AP et al (2013) Imatinib failure-free survival (IFS) in patients with localized gastrointestinal stromal tumors (GIST) treated with adjuvant imatinib (IM): the EORTC/AGITG/FSG/GEIS/ISG randomized controlled phase III trial (suppl;abstract 10500). J Clin Oncol:31
Debiec-Rychter M et al (2006) KIT mutations and dose selection for imatinib in patients with advanced gastrointestinal stromal tumours. Eur J Cancer 42(8):1093–1103
Ryan DP et al (2002) A phase II and pharmacokinetic study of ecteinascidin 743 in patients with gastrointestinal stromal tumors. Oncologist 7(6):531–538
Edmonson JH et al (2002) Contrast of response to dacarbazine, mitomycin, doxorubicin, and cisplatin (DMAP) plus GM-CSF between patients with advanced malignant gastrointestinal stromal tumors and patients with other advanced leiomyosarcomas. Cancer Investig 20(5–6):605–612
Trent JC et al (2003) A two-arm phase II study of temozolomide in patients with advanced gastrointestinal stromal tumors and other soft tissue sarcomas. Cancer 98(12):2693–2699
Druker BJ et al (2001) Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 344(14):1031–1037
Heinrich MC et al (2000) Inhibition of c-kit receptor tyrosine kinase activity by STI 571, a selective tyrosine kinase inhibitor. Blood 96(3):925–932
Joensuu H et al (2001) Effect of the tyrosine kinase inhibitor STI571 in a patient with a metastatic gastrointestinal stromal tumor. N Engl J Med 344(14):1052–1056
van Oosterom AT et al (2002) Update of phase I study of imatinib (STI571) in advanced soft tissue sarcomas and gastrointestinal stromal tumors: a report of the EORTC soft tissue and bone sarcoma group. Eur J Cancer 38(Suppl 5):S83–S87
Demetri GD et al (2002) Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med 347(7):472–480
Verweij J et al (2004) Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomised trial. Lancet 364(9440):1127–1134
Blanke CD et al (2008) Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J Clin Oncol 26(4):626–632
Demetri GD et al (2009) Imatinib plasma levels are correlated with clinical benefit in patients with unresectable/metastatic gastrointestinal stromal tumors. J Clin Oncol 27(19):3141–3147
Yoo C et al (2010) Cross-sectional study of imatinib plasma trough levels in patients with advanced gastrointestinal stromal tumors: impact of gastrointestinal resection on exposure to imatinib. J Clin Oncol 28(9):1554–1559
Heinrich MC et al (2008) Correlation of kinase genotype and clinical outcome in the North American intergroup phase III trial of imatinib mesylate for treatment of advanced gastrointestinal stromal tumor: CALGB 150105 study by cancer and leukemia group B and Southwest oncology group. J Clin Oncol 26(33):5360–5367
Cassier PA et al (2012) Outcome of patients with platelet-derived growth factor receptor alpha-mutated gastrointestinal stromal tumors in the tyrosine kinase inhibitor era. Clin Cancer Res 18(16):4458–4464
Kerkela R et al (2006) Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nat Med 12(8):908–916
Noens L et al (2009) Prevalence, determinants, and outcomes of nonadherence to imatinib therapy in patients with chronic myeloid leukemia: the ADAGIO study. Blood 113(22):5401–5411
Zalcberg JR et al (2005) Outcome of patients with advanced gastro-intestinal stromal tumours crossing over to a daily imatinib dose of 800 mg after progression on 400 mg. Eur J Cancer 41(12):1751–1757
Demetri GD et al (2006) Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomised controlled trial. Lancet 368(9544):1329–1338
Demetri GD et al (2012) Complete longitudinal analyses of the randomized, placebo-controlled, phase III trial of sunitinib in patients with gastrointestinal stromal tumor following imatinib failure. Clin Cancer Res 18(11):3170–3179
Desai J et al (2006) Hypothyroidism after sunitinib treatment for patients with gastrointestinal stromal tumors. Ann Intern Med 145(9):660–664
Antonescu CR et al (2005) Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin Cancer Res 11(11):4182–4190
Wardelmann E et al (2006) Polyclonal evolution of multiple secondary KIT mutations in gastrointestinal stromal tumors under treatment with imatinib mesylate. Clin Cancer Res 12(6):1743–1749
Desai J et al (2007) Clonal evolution of resistance to imatinib in patients with metastatic gastrointestinal stromal tumors. Clin Cancer Res 13(18 Pt 1):5398–5405
Liegl B et al (2008) Heterogeneity of kinase inhibitor resistance mechanisms in GIST. J Pathol 216(1):64–74
Debiec-Rychter M et al (2005) Mechanisms of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of the PKC412 inhibitor against imatinib-resistant mutants. Gastroenterology 128(2):270–279
Miselli FC et al (2007) c-Kit/PDGFRA gene status alterations possibly related to primary imatinib resistance in gastrointestinal stromal tumors. Clin Cancer Res 13(8):2369–2377
Eechoute K et al (2011) Environmental and genetic factors affecting transport of imatinib by OATP1A2. Clin Pharmacol Ther 89(6):816–820
Guo T et al (2009) Mechanisms of sunitinib resistance in gastrointestinal stromal tumors harboring KITAY502-3ins mutation: an in vitro mutagenesis screen for drug resistance. Clin Cancer Res 15(22):6862–6870
Demetri GD et al (2013) Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381(9863):295–302
Reichardt P et al (2012) Phase III study of nilotinib versus best supportive care with or without a TKI in patients with gastrointestinal stromal tumors resistant to or intolerant of imatinib and sunitinib. Ann Oncol 23(7):1680–1687
Campbell NP, Wroblewski K, Maki RG, et al (2011) Final results of a University of Chicago phase II consortium trial of sorafenib (SOR) in patients (pts) with imatinib (IM)- and sunitinib (SU)-resistant (RES) gastrointestinal stromal tumors (GIST) (abstract). ASCO GI cancers symposium, San Francisco, CA, 20–22 January
Montemurro M et al (2013) Sorafenib as third- or fourth-line treatment of advanced gastrointestinal stromal tumour and pretreatment including both imatinib and sunitinib, and nilotinib: a retrospective analysis. Eur J Cancer 49(5):1027–1031
Dewaele B et al (2008) Activity of dasatinib, a dual SRC/ABL kinase inhibitor, and IPI-504, a heat shock protein 90 inhibitor, against gastrointestinal stromal tumor-associated PDGFRAD842V mutation. Clin Cancer Res 14(18):5749–5758
Kang YK et al (2013) Resumption of imatinib to control metastatic or unresectable gastrointestinal stromal tumours after failure of imatinib and sunitinib (RIGHT): a randomised, placebo-controlled, phase 3 trial. Lancet Oncol 14(12):1175–1182
Italiano A et al (2012) Patterns of care, prognosis, and survival in patients with metastatic gastrointestinal stromal tumors (GIST) refractory to first-line imatinib and second-line sunitinib. Ann Surg Oncol 19(5):1551–1559
Yeh CN et al (2010) Surgical management in metastatic gastrointestinal stromal tumor (GIST) patients after imatinib mesylate treatment. J Surg Oncol 102(6):599–603
Mussi C et al (2010) Post-imatinib surgery in advanced/metastatic GIST: is it worthwhile in all patients? Ann Oncol 21(2):403–408
Al-Batran SE et al (2007) Focal progression in patients with gastrointestinal stromal tumors after initial response to imatinib mesylate: a three-center-based study of 38 patients. Gastric Cancer 10(3):145–152
Kobayashi K et al (2006) Hepatic artery chemoembolization for 110 gastrointestinal stromal tumors: response, survival, and prognostic factors. Cancer 107(12):2833–2841
Pawlik TM et al (2006) Results of a single-center experience with resection and ablation for sarcoma metastatic to the liver. Arch Surg 141(6):537–543. discussion 543-4
Raut CP et al (2006) Surgical management of advanced gastrointestinal stromal tumors after treatment with targeted systemic therapy using kinase inhibitors. J Clin Oncol 24(15):2325–2331
DeMatteo RP et al (2007) Results of tyrosine kinase inhibitor therapy followed by surgical resection for metastatic gastrointestinal stromal tumor. Ann Surg 245(3):347–352
Knowlton CA, Brady LW, Heintzelman RC (2011) Radiotherapy in the treatment of gastrointestinal stromal tumor. Rare Tumors 3(4):e35
Cuaron JJ et al (2013) External beam radiation therapy for locally advanced and metastatic gastrointestinal stromal tumors. Radiat Oncol 8(1):274
Choi H et al (2007) Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol 25(13):1753–1759
Benjamin RS et al (2007) We should desist using RECIST, at least in GIST. J Clin Oncol 25(13):1760–1764
Kamiyama Y et al (2005) 18F-fluorodeoxyglucose positron emission tomography: useful technique for predicting malignant potential of gastrointestinal stromal tumors. World J Surg 29(11):1429–1435
Gayed I et al (2004) The role of 18F-FDG PET in staging and early prediction of response to therapy of recurrent gastrointestinal stromal tumors. J Nucl Med 45(1):17–21
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kollár, A., Aguiar, P.N., Forones, N.M., De Mello, R.A. (2019). Gastrointestinal Stromal Tumor (GIST): Diagnosis and Treatment. In: De Mello, R., Mountzios, G., Tavares, Á. (eds) International Manual of Oncology Practice. Springer, Cham. https://doi.org/10.1007/978-3-030-16245-0_37
Download citation
DOI: https://doi.org/10.1007/978-3-030-16245-0_37
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-16244-3
Online ISBN: 978-3-030-16245-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)