Abstract
Cerebral aging is associated with characteristic changes of main EEG patterns recorded during wakefulness and sleep. They include a slowing of posterior dominant rhythm, the reduction of beta activity, and the appearance of theta-delta activities particularly over left and temporal regions. With the exception of frontotemporal dementia, there is usually a good correlation between the degree of cognitive impairment in dementia syndromes and the EEG changes. In multi-infarct dementia, the presence of focal and/or epileptiform discharges depends on the location and size of infarcts. In Parkinson’s disease the EEG changes are usually nonspecific.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 EEG Changes in the Elderly
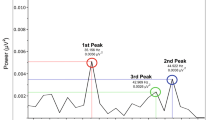
Cerebral aging is associated with characteristic changes of main EEG patterns recorded during wakefulness and sleep [1]. In wakefulness the amplitude of background alpha rhythm is lower in adults and elderly people compared to the adolescent, particularly in the occipital region, and sometimes an instability of alpha can be seen due to a scarce tendency to synchronization. However, these changes are sometimes the mere expression of anxiety and are therefore less evident during and after hyperventilation. When the alpha rhythm is well synchronized, such as during quite wakefulness with closed eyes, the EEG recorded in the elderly may show a tendency toward an anterior diffusion of the background activity; the typical posterior dominance is therefore less evident (Fig. 41.1). Over the years, a progressive slowing of alpha rhythm of approximately 0.08 Hz/year occurs after the age of 60. In our experience, patients in their 80s have a mean alpha rhythm of 8.5–9 Hz. Individuals who are over 100 years old have a physiological reduction of alpha frequency to 7.5–8 Hz [2]. Figures 41.2 and 41.3 show examples of changes in background activities at different ages, assessed both visually and quantitatively. After the age of 50, subharmonics of background activity at 4–5 Hz (slow alpha variant) and an asynchronous and asymmetrical background rhythm can be seen. The mu rhythm is less visible in elderly compared to younger subjects, and the photic drive during intermittent photic stimulation is less evident.
With increasing age, the beta activity is less represented and has lower amplitude, particularly over precentral regions, especially for 25–30 Hz frequencies. Compared to males with the same age, females have greater amount of beta activity [3, 4].
After the age of 60, slow focal activities (theta and/or delta) are more represented and prevalent over left anterior temporal region. Very small amount of theta and delta sequences are nevertheless considered normal for the age. Hyperventilation can lead to a slow widespread theta-delta activity and an increase in a focal temporal slowing already present in baseline EEG recording.
During the first phases of NREM sleep, a reduction in amplitude and frequency of sleep spindles and K complexes can be seen, with a reduced voltage of vertex waves and positive occipital sharp transients of sleep (POSTS). A shorter duration of phases 3 and 4 of NREM sleep with a lower density of delta waves can occur, together with a reduction in REM (rapid eye movements) latency, duration, and density. Overall, there is a reduction in the total number of hours spent to sleep, which is more superficial and characterized by frequent interruptions due to nocturnal awakenings.
In conclusion, different criteria should be used when assessing the EEG of elderly subjects: if the same criteria valid for young and adult individuals were adopted, more than 50% of EEG recordings in subjects older than 60 years would be classified abnormal. The most relevant changes include the alpha rhythm (of lower amplitude and lower frequency and with tendency to anteriorization), theta-delta activities (more evident, particularly over left and temporal regions), and beta activity (less evident and of lower voltage, particularly among men) [1].
2 Use and Advantages of the EEG Recording in the Elderly
EEG recordings are usually required in the elderly for syncopal episodes, confusional states, epileptic seizures with motor phenomena, and episodes of transient loss of consciousness [5]. Based on the available literature (mostly unselected and homogeneous case series), approximately 50% of the EEGs are abnormal, particularly for the presence of focal and/or widespread slowing, which are encountered more frequently in patients older than 80 years. Epileptiform abnormalities are rarely seen.
In the elderly subjects, it is important that the recordings include an ECG monitoring and last long enough to detect fluctuations in vigilance. Whenever possible a video-EEG recording should also be obtained for differential diagnosis [6], since approximately 50% of the paroxysmal episodes requiring EEG are nonepileptic events. In the elderly, the EEG should be recorded not for detecting focal and/or widespread slowing but to correlate the cognitive deficit or the consciousness impairment with nonconvulsive status epilepticus/seizures and toxic-dismetabolic or infective encephalopathies. In these conditions, the EEG can be more sensitive than neuroimaging, particularly in the initial stages [7]. For instance, in elderly individuals taking several drugs, the EEG can reveal signs compatible with drug toxicity, such as a slowing of the background activity incongruous with the age of the subjects.
3 Dementia Syndromes
Dementia is a brain disorder characterized by a progressive deficit of cognitive functions with subsequent impairment of social and working activities. Primary degenerative dementias (Alzheimer’s disease, frontotemporal dementia, dementia with Lewy bodies) represent approximately 60–70% of cases, the vascular dementias the 20–30%, and other etiologies (e.g., dementia associated with Parkinson’s disease, Huntington chorea, progressive supranuclear palsy) the remaining 10–20%.
The electroencephalogram (EEG) is noninvasive, inexpensive, highly available, and sensitive to changes in the functional state of the human brain. These features explain why EES has been used for many years in dementia research, with the aim of differentiating subjects with various types and severity of cognitive impairment from healthy individuals. Visual EEG analysis is routinely used in clinical practice. A more accurate differential diagnosis between the various subtypes of dementia relies on quantitative EEG (qEEG), which requires extensive technical knowledge in the field of digital signal processing.
The EEG may prove useful in the diagnosis of dementia, because there is usually a good correlation between the degree of cognitive impairment and the EEG changes [1]. In dementia syndromes EEG changes include a progressive slowing of posterior dominant alpha rhythm, a progressive increase in theta-delta activity, and a reduction in beta activity. A highest amount of slowing is related with a more rapid course of disease.
The EEG may also indicate whether the dementia is associated with a focal or a diffuse process or if the cognitive impairment is due to a continuous epileptic activity (i.e., nonconvulsive status epilepticus [7]). Finally, a normal or near-normal EEG in a patient with cognitive deficits suggests the presence of depressive pseudodementia or frontotemporal dementia [1].
3.1 Alzheimer’s Disease
Usually, in the initial stages of the disease, the EEG shows a nonspecific slowing of alpha rhythm, later evolving into a widespread theta-delta activity and a decrease in beta activity (Fig. 41.4a). In patients with mild cognitive impairment, the EEG can be normal. In some patients the dominant posterior rhythm can still be within the alpha frequency; however, if previous EEGs are available, a slowing of the frequency can be observed. With disease progression bursts of bilateral slow potentials appear, prevalently over anterior regions (Fig. 41.4b). Epileptiform abnormalities are rare and may have a triphasic morphology. Sometimes these graphoelements are periodic and more evident over middle posterior head regions.
EEG recordings obtained in the same patient with Alzheimer’s disease. (a) In the initial stage, the EEG recording shows a modest slowing of background activity. (b) In a later stage, the tracing is diffusely slowed, particularly over middle anterior regions. The eye opening leads to a background activity which is better synchronized and more rapid (paradoxical alpha)
In Alzheimer’s disease the slowing of the EEG tracing is directly correlated with the degree of cognitive impairment. In mild cognitive impairment, the EEG is normal or may show only a modest increase of theta activity associated with reduced beta activity. Patients with Alzheimer’s disease typically show a reduced reactivity of alpha rhythm to eye opening (partial alpha blockade) (Fig. 41.5) and during memory testing, perhaps related to a reduced dopaminergic tone.
The polysomnography shows a progressive reduction in total sleep time, affecting both NREM and REM. Compared to nondemented subjects with the same age, patients with Alzheimer’s disease have a higher sleep fragmentation and a reduction of sleep spindles and K complexes.
Studies with quantitative EEG have provided promising results to identify EEG biomarkers of the progressive cognitive impairment, such as increased power of theta-delta frequency bands, a corresponding reduced power of fast alpha-beta frequencies [8], and the reduced reactivity of alpha rhythm to eye opening and during memory testing [9, 10]. EEG coherence is reduced in Alzheimer’s disease, suggesting a reduced interaction between different cerebral areas [11].
EEG network analysis methods are emerging as potential biomarkers for the diagnosis of AD and promising tools for studying disease progression and for early recognition of mild cognitive impairment, particularly if combined with spectral methods [12]; their place in clinical practice still needs to be further assessed.
Results of studies assessing the role of EEG in the diagnosis of dementia should however be interpreted with caution, paying attention to the presence of age-matched controls, which is necessary to avoid overestimation of EEG slowing, and to the sample size [13].
3.2 Dementia with Lewy Bodies
Dementia with Lewy bodies is the second most common neurodegenerative cause of dementia after Alzheimer’s disease. In this dementia syndrome, EEG changes in the initial phases include slowing of background activity and bursts of theta-delta activity, prevalent over frontal regions. Overall, EEG changes in dementia with Lewy bodies are usually more severe than those detected in Alzheimer’s disease, particularly for the prominent and precocious theta slowing over temporal regions [14]. Bilateral frontal intermittent rhythmic delta activity (FIRDA) seems to occur more frequently than in patients clinically diagnosed with Alzheimer’s disease and has therefore been proposed as a supportive diagnostic feature for this condition [15].
3.3 Frontotemporal Dementia
The visual inspection of EEG recordings in patients with frontotemporal dementia does not usually show relevant differences with those of healthy subjects [16]. However, a quantitative analysis may inconstantly reveal theta activity over frontoparietal regions or a reduction of beta activity [17].
4 Multi-infarct Dementia
In this type of dementia, the EEG shows focal or multifocal slowing, even in the presence of a preserved background rhythm (Fig. 41.6). The presence of an alpha rhythm, even in later stages of the disease, suggests a multi-infarct dementia, whereas in primary dementia a global slowing of background activity is usually seen in early stages. The EEG changes (slowing of background activity) correlate with cortical atrophy and leukoencephalopathy seen on neuroimaging [18]. In multi-infarct dementia, epileptiform abnormalities may also occur and are more common than in Alzheimer’s disease; they are usually sharp waves, sometimes with a quasiperiodic pattern of recurrence, and are frequently associated with large and superficial infarcts.
4.1 Subcortical Dementia
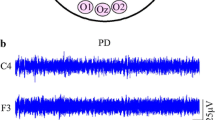
In Parkinson’s disease without dementia, the EEG shows no relevant changes, apart from those related to aging and concomitant diseases. Theta rhythmic artifactual activities due to tremor are frequently seen, particularly over posterior regions (Fig. 41.7). Parkinson’s dementia (i.e., dementia associated with Parkinson’s disease) is a clinical syndrome characterized by slowness of mental processing, impaired cognition, apathy, and depression. It is often associated with an early nonspecific slowing of background activity. During the course of the disease, there is a progressive slowing of the background activity, with a widespread monomorphic theta activity, sometimes admixed with slower (delta) sequences over frontotemporal regions, mostly on the left side (Fig. 41.8). The polysomnography is characterized by an increased sleep latency and a reduced total sleep time, with increased amount of first NREM stages and reduced slow sleep stages and REM. There are also frequent nocturnal awakenings. Quantitative EEG studies have shown that spectral analysis (increase of spectral powers in delta and theta frequency bands, with decrease in alpha and beta bands) can discriminate between dementia associated with Parkinson’s disease and Parkinson’s disease without dementia and differentiate mild cognitive impairment from normal cognition in patients with Parkinson’s disease [19].
In dementia associated with Huntington’s disease, the EEG recorded in most advanced stages shows a slowing and destructuration of background activity, with increased slowing predominant anteriorly. In initial stages there is often a low-voltage activity. In patients with myoclonic seizures, the EEG shows polyspike and polyspike-wave complexes.
In dementia associated with progressive supranuclear palsy, the EEG shows a progressive slowing of the alpha rhythm and the progressive appearance of slowing, also manifesting as frontal intermittent rhythmic delta activity or bitemporal (temporal intermittent rhythmic delta activity). In initial stages of disease, the EEG is mostly normal.
References
Babiloni C, Del Percio C, Buján A. EEG in dementing disorders. In: Schomer DL, Lopes da Silva F, editors. Niedermeyer’s electroencephalography: basic principles, clinical applications, and related fields. 7th ed. New York, NY: Oxford University Press; 2018. p. 413–32.
Hubbard O, Sunde D, Goldensohn ES. The EEG in centenarians. Electroencephalogr Clin Neurophysiol. 1976;40:407–17.
Giaquinto S, Nolfe G. The EEG in the normal elderly: a contribution to the interpretation of aging and dementia. Electroencephalogr Clin Neurophysiol. 1986;63:540–6.
Brenner RP, Ulrich RF, Reinolds CF. EEG spectral analysis in healthy, elderly men and women: sex differences. Electroencephalogr Clin Neurophysiol. 1995;94:1–5.
Widdess-Walsh P, Sweeney BJ, Galvin R, McNamara B. Utilization and yield of EEG in the elderly population. J Clin Neurophysiol. 2005;22:253–5.
Kipervasser S, Neufeld MY. Video-EEG monitoring of paroxysmal events in the elderly. Acta Neurol Scand. 2007;116:221–5.
Bottaro FJ, Martinez OA, Fernandez Pardal MM, et al. Nonconvulsive status epilepticus in the elderly: a case-control study. Epilepsia. 2007;48:966–72.
Huang C, Wahlund L, Dierks T, Julin P, Winblad B, Jelic V. Discrimination of Alzheimer’s disease and mild cognitive impairment by equivalent EEG sources: a cross-sectional and longitudinal study. Clin Neurophysiol. 2000;111:1961–7.
Van der Hiele K, Vein AA, van der Welle A, et al. EEG and MRI correlates of mild cognitive impairment and Alzheimer’s disease. Neurobiol Aging. 2007;28:1322–9.
Van der Hiele K, Vein AA, Reijntjes RHAM, et al. EEG correlates in the spectrum of cognitive decline. Clin Neurophysiol. 2007;118:1931–9.
Jelles B, Scheltens PH, van der Flier WM, et al. Global dynamical analysis of the EEG in Alzheimer’s disease: frequency-specific changes of functional interactions. Clin Neurophysiol. 2008;119:837–41.
Horvath A, Szucs A, Csukly G, Sakovics A, Stefanics G, Kamondi A. EEG and ERP biomarkers of Alzheimer’s disease: a critical review. Front Biosci (Landmark Ed). 2018;23:183–220.
Van der Hiele K, Bollen ELEM, Vein AA, et al. EEG markers of future cognitive performance in the elderly. J Clin Neurophysiol. 2008;25:83–9.
Cromarty RA, Elder GJ, Graziadio S, et al. Neurophysiological biomarkers for Lewy body dementias. Clin Neurophysiol. 2016;127:349–59.
Calzetti S, Bortone E, Negrotti A, Zinno L, Mancia D. Frontal intermittent rhythmic delta activity (FIRDA) in patients with dementia with Lewy bodies: a diagnostic tool? Neurol Sci. 2002;23(Suppl 2):S65–6.
Yener GG, Leuchter AF, Jenden D, Read SL, Cummings JL, Miller BL. Quantitative EEG in frontotemporal dementia. Clin Electroencephalogr. 1996;27:61–8.
Malek N, Baker MR, Mann C, Greene J. Electroencephalographic markers in dementia. Acta Neurol Scand. 2017;135:388–93.
Erkinjuntti T, Larsen T, Sulkava R, Ketonen L, Laaksonen R, Palo J. EEG in the differential diagnosis between Alzheimer’s disease and vascular dementia. Acta Neurol Scand. 1988;77:36–43.
Cozac VV, Gschwandtner U, Hatz F, Hardmeier M, Rüegg S, Fuhr P. Quantitative EEG and cognitive decline in Parkinson’s disease. Parkinsons Dis. 2016;2016:9060649.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Brigo, F., Mecarelli, O. (2019). Aging and Degenerative Disorders. In: Mecarelli, O. (eds) Clinical Electroencephalography. Springer, Cham. https://doi.org/10.1007/978-3-030-04573-9_41
Download citation
DOI: https://doi.org/10.1007/978-3-030-04573-9_41
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-04572-2
Online ISBN: 978-3-030-04573-9
eBook Packages: MedicineMedicine (R0)