Abstract
Cardiac risk assessment has traditionally been based on conventional risk factors; the shortcomings of this approach are all too often highlighted by major cardiac events occurring in presumably low-risk people. The annual presentation of 650,000 previously asymptomatic patients with an acute coronary event as the initial manifestation of coronary artery disease (CAD) [1] is a testimony to the failure of our current risk assessment model. Consequently, there has been a focus on markers of subclinical atherosclerosis that may be utilized for risk assessment of individuals, rather than extrapolating from risk factors that reflect trends in large groups of patients in epidemiologic studies. The most powerful of these subclinical markers is coronary artery calcium (CAC).
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Coronary Artery Calcium
- Coronary Compute Tomographic Angiography
- Coronary Artery Calcium Score
- Calcium Score
- National Cholesterol Education Program
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Cardiac risk assessment has traditionally been based on conventional risk factors; the shortcomings of this approach are all too often highlighted by major cardiac events occurring in presumably low-risk people. The annual presentation of 650,000 previously asymptomatic patients with an acute coronary event as the initial manifestation of coronary artery disease (CAD) [1] is a testimony to the failure of our current risk assessment model. Consequently, there has been a focus on markers of subclinical atherosclerosis that may be utilized for risk assessment of individuals, rather than extrapolating from risk factors that reflect trends in large groups of patients in epidemiologic studies. The most powerful of these subclinical markers is coronary artery calcium (CAC).
1 Background
CAC is pathognomonic for atherosclerosis [2–4]. Mönckeberg’s calcific medial sclerosis does not occur in the coronary arteries [5]; atherosclerosis is the only vascular disease known to be associated with coronary calcification. Calcium phosphate (in the hydroxyapatite form) and cholesterol accumulate in atherosclerotic lesions. Circulating proteins that are normally associated with bone remodeling play an important role in coronary calcification, and arterial calcium in atherosclerosis is a regulated active process similar to bone formation, rather than a passive precipitation of calcium phosphate crystals [6–9]. Rumberger et al. [10] demonstrated that the total area of coronary artery calcification is highly correlated (r= 0.9) in a linear fashion with the total area of coronary artery plaque on a segmental, individual, and whole coronary artery system basis (Figure 4.1), and the areas of coronary calcification comprise approximately one-fifth that of the associated coronary plaque. Additionally, there were plaque areas without associated coronary calcium, suggesting that there may be a coronary plaque size most commonly associated with coronary calcium but, in the smaller plaques, the calcium is either not present or is undetectable.
Correlation between calcified and total plaque burden in histopathologic coronary artery specimens (reproduced with permission of Wolters Kluwer from Rumberger et al. [10])
Intravascular ultrasound [11, 12] measures of combined calcified and noncalcified plaque confirm the strong relationship (Figure 4.2).
2 Methodology
2.1 Technical
Until recently, the data substantiating the importance of CAC have been derived almost exclusively through the use of electron beam tomography (EBT), utilizing a rotating electron beam to acquire prospectively triggered, tomographic 100-ms X-ray images at 3 mm intervals in the space of a 30- to 40-s breathhold. The multidetector computed tomography (MDCT) technology is a more recent development and employs a rotating gantry with a special X-ray tube and variable number of detectors (from 4 to 64), with 165–375-ms images at 0.5, 1.5, 2.0, or 3.0 mm intervals, depending on the protocol and manufacturer.
2.2 Scoring
The presence of coronary calcium is sequentially quantified through the entire epicardial coronary system. Coronary calcium is defined as a lesion above a threshold of 130 Hounsfield units (which range from −1,000 (air), through 0 (water), and up to +1,000 (dense cortical bone)), with an area of three or more adjacent pixels (at least 1 mm2). The original calcium score developed by Agatston et al [13] is determined by the product of the calcified plaque area and maximum calcium lesion density (from 1 to 4 based upon Hounsfield units). Standardized categories for the calcium score have been developed with scores of 1–10 considered minimal, 11–100 mild, 101–400 moderate, and >400 severe. Examples are shown in Figure 4.1. The calcium volume score [14] is a more reproducible parameter that is independent of calcium density and is considered to be the parameter of choice for serial studies to track progression or regression of atherosclerosis. Phantom-based calcium mass scores are being developed that will be applicable to any CT scanner [15], but have yet to be validated. Examples of CAC scans are shown in Figure 4.3.
2.3 Epidemiology
By comparing a person’s calcium score to others of the same age and gender through the use of large databases of asymptomatic subjects, a calcium percentile is generated [16]. This is an index of the prematurity of atherosclerosis; for example, a 50-year-old man in the 76th percentile has more plaque than 75% but less plaque than 24% of asymptomatic 50-year-old men. Although there is an increasing incidence of coronary calcification with increasing age (Figure 4.4), this simply parallels the development of coronary atherosclerosis.
Prevalence of CAC in men and women by decades (reproduced with permission of Elsevier from Janowitz et al [16])
Table 4.1 shows coronary calcification incidence by EBT in an unselected patient population of men and women [17]. The amount of CAC in women is similar to that in men a decade younger, paralleling the 10-year lag in women of the development of clinical atherosclerosis.
Useful though these current nomograms are, variations according to ethnicity have been described, and data regarding these variations are still being collected and separated. In earlier studies, Blacks were noted to have either lower [18, 19] or similar [20, 21] amounts of CAC as Caucasians of the same age; Hispanics had less CAC than Caucasians [18]. In the more recent Multi-Ethnic Study of Atherosclerosis (MESA) of 6,110 asymptomatic patients with 53% female and an average age of 62 years, men had greater calcium levels than women, and calcium amount and prevalence continually increased with increasing age [22]. In men, Caucasians and Hispanics were the first and second highest respectively; Blacks were lowest at the younger ages, and Chinese were lowest at the older ages. In women, whites were highest, Chinese and Black were intermediate, and Hispanics were the lowest except for Chinese in the oldest age group. Thus, predictive indices should be extrapolated to non-whites with caution. However, MESA demonstrated very strong CAC predictive for all groups [23].
Younger patients with a family history of premature CAD have significantly higher CAC scores than similar aged individuals without this risk factor, particularly if there is a sibling history of premature CAD [24]. In MESA, the odds ratios for the presence of CAC independent of all risk factors in those with compared to those without a family history of premature CAD were 2.74 with premature CAD in both a parent and a sibling, 2.06 in a sibling alone, and 1.52 in a parent alone [25].
2.4 Radiation
The vast majority of CAC scanning is being performed on MDCT scanners; in the future it will be the exclusive technology as electron beam computed tomography (EBCT) scanners disappear. The radiation exposure is 1.0 mSv [26]: appropriate perspective is obtained by comparing this exposure to the 0.75 mSv of the annual mammographic examination recommended for women 45 years and older.
3 Coronary Artery Calcium and Obstructive Disease
The relationship of CAC to obstructive disease has been extensively investigated, and was misunderstood by the 2000 ACC/AHA Consensus Document on EBT [27], which focused on the low specificity as a critical flaw. While the presence of CAC is nearly 100% specific for atherosclerosis, it is not specific for obstructive disease since both obstructive and nonobstructive lesions have calcification present in the intima. Comparisons with pathology specimens have shown that the degree of luminal narrowing is weakly correlated with the amount of calcification on a site-by-site basis [28–30], whereas the likelihood of significant obstruction increases with the total CAC score [4, 31, 32]. Shavelle et al. [33] reported a 96% sensitivity and 47% specificity for a calcium score >0, with a relative risk for obstructive disease of 4.5, compared to a 76% sensitivity and 60% specificity for treadmill testing, with a relative risk of 1.7. Bielak et al [34] noted a sensitivity and specificity of 99.1 and 38.6% for a calcium score >0. However, when corrected for verification bias, the specificity improved to 72.4%, without loss of sensitivity (97%). The likelihood ratio for obstruction ranged from 0.03 to 0.07 in men and women ≥50 years of age for 0 scores to 12.85 for scores >200. In the <50 years cohort, the likelihood ratios ranged from 0.1 to 0.29 for 0 scores to 54–189 for scores >100.
Rumberger et al [35] demonstrated that higher calcium scores are associated with a greater specificity for obstructive disease at the expense of sensitivity; for example, a threshold score of 368 was 95% specific for the presence of obstructive CAD. In 1,764 persons undergoing angiography, the sensitivity and negative predictive value in men and women were >99% [36]; a score of 0 virtually excluded patients with obstructive CAD. In a separate study of 1,851 patients undergoing CAC scanning and angiography [37], CAC scanning by EBT in conjunction with pretest probability of disease derived by a combination of age, gender, and risk factors, facilitated prediction of the severity and extent of angiographically significant CAD in symptomatic patients.
In a recent meta-analysis of 10,355 symptomatic patients who underwent cardiac catheterization and CAC, 0 CAC was noted in 1941. Significant obstructive disease, defined as >50% diameter stenosis, was noted in 5,805 (56%). For CAC > 0 and the presence of >50% diameter stenosis, the following were reported: sensitivity 98%, specificity 40%, positive predictive value 68%, and negative predictive value 93% [38].
3.1 Prognostic Studies in Symptomatic Patients
The prognostic value of extensive coronary calcium (>1,000) in symptomatic males with established advanced CAD was demonstrated in a 5-year follow-up study of 150 patients [39]. More recently, in a meta-analysis of 3,924 symptomatic patients with a 3.5 year follow-up, the cardiac event rate was 2.6% per year in those with CAC > 0 and 0.5% per year in 0 CAC patients [38]. However, with the development of coronary computed tomographic angiography (CCTA), CAC alone is not justified in the symptomatic population; CCTA will identify the noncalcified plaque and even obstructive disease that may be noted in these patients, even with 0 CAC.
3.2 Clarification
Despite the apparently reasonable specificities, which are similar to those of stress testing, it must be understood that the purpose of CAC scanning is not to detect obstructive disease and, therefore, it is inappropriate to even use “specificity” in the context of obstruction. Rather, its purpose is to detect subclinical atherosclerosis in its early stages, for which it is virtually 100% specific.
4 Key Prognostic Studies in Primary Prevention
The utility of CAC for risk evaluation in the asymptomatic primary prevention population is dependent on prognostic studies documenting the relative risk conferred by calcified plaque quantitation compared to conventional risk factors. Raggi et al [40] demonstrated, in 632 asymptomatic patients followed for 32 months, an annualized event rate of 0.1% per year in patients with 0 scores, compared to 2.1% per year with scores of 1–99, 4.1% per year with scores of 100–400, and 4.8% per year with scores >400. Thus, the annualized event rates associated with coronary calcium were in the range considered to warrant secondary prevention classification by the Framingham Risk Score (Figure 4.5).
The odds ratio conferred by a calcium percentile >75% was 21.5 times greater than for the lowest 25%, compared to an odds ratio of 7 for the highest vs. lowest quartiles of National Cholesterol Education Program (NCEP) risk factors (Figure 4.6).
Wong et al [41], in 926 asymptomatic patients followed for 3.3 years, noted a relative risk of 8 for scores >270, after adjusting for age, gender, hypertension, high cholesterol, smoking, and diabetes. Arad et al [42], in 1,132 subjects followed for 3.6 years, reported odds ratios of 14.3–20.2 for scores ranging from >80 to >600; these were three to seven times greater than for the NCEP risk factors. In a retrospective analysis of 5,635 asymptomatic, predominantly low to moderate risk, largely middle-aged patients followed for 37 ± 12 months, Kondos et al. [43] found that the presence of any CAC by EBT was associated with a relative risk for events of 10.5, compared to 1.98 and 1.4 for diabetes and smoking, respectively. In women, only CAC was linked to events, with a relative risk of 2.6; risk factors were not related. The presence of CAC provided prognostic information incremental to age and other risk factors.
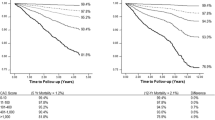
Shaw et al [44] retrospectively analyzed 10,377 asymptomatic patients with a 5-year follow-up after an initial EBT evaluation. All-cause mortality increased proportional to CAC (Figure 4.7), which was an independent predictor of risk after adjusting for all of the Framingham risk factors (p< 0.001).
Cumulative survival of 10,377 patients from the National Death Registry according to CAC categories (reproduced with permission of the RSNA from Shaw et al [45], p. 831).
Superiority of CAC to conventional Framingham risk factor assessment was demonstrated by a significantly greater area under the ROC curves (0.73 vs. 0.67, p< 0.001). Incremental value of CAC to Framingham risk was also established by a significant increase of the area under the ROC curves, from 0.72 for Framingham risk to 0.78 with the addition of CAC (p< 0.001). Risk stratification was present in each risk group, and was particularly strong in the 10–20% 10-year Framingham risk group. Stratification of mortality risk by CAC score was as effective in women as in men.
Greenland et al [45] analyzed a population-based study of 1,461 prospectively followed, asymptomatic subjects who were predominantly moderate to high risk, and found that CAC scores >300 significantly added prognostic information to Framingham risk analysis in the 10–20% Framingham risk category. The results of the St Francis Heart Study by Arad et al [46] in a prospective, population-based study of 5,585 asymptomatic, predominantly moderate- to moderately-high-risk men and women, mirrored previous retrospective studies [7, 18–20], and confirmed the higher event rates associated with increasing CAC scores. CAC scores >100 were associated with relative risks of from 12 to 32, and were secondary prevention equivalent, with event rates>2% per year (Figure 4.8).
Incremental information over Framingham scores was documented with areas under the ROC curves of 0.81 for CAC and 0.71 for Framingham (p< 0.01). Importantly, classification by CAC tertiles changed the risk group of approximately 67% of patients classified in the Framingham 10–20% 10-year event rate group to either lower or higher risk. The appropriateness of this change was confirmed by outcome measures of cardiac events. Furthermore, in the Framingham high-risk category (>20% 10-year event rate), 45% were correctly moved to lower-risk categories by CAC tertile reclassification. Finally, in the Framingham <10% 10-year risk group, 29% had scores >100 with an associated 1.7% per year event rate (Figure 4.9).
The prognostic significance of very high calcium scores was provided in a study of 98 asymptomatic patients with a CAC score >1,000 who were followed for 17 months [47] during which 35 patients (36%) suffered a hard cardiac event (myocardial infarction (MI) or cardiac death). The annualized event rate of 25% refuted the erroneous concept that extensive calcified plaque may confer protection against plaque rupture and events.
In a younger cohort of 2,000 asymptomatic Army personnel, Taylor et al [48] demonstrated the powerful predictive value of CAC. There was a relative risk of 11.8 in patients with CAC > 44 compared to those with 0 CAC, after correcting for the Framingham Risk Score. In a much more elderly population (71 years), Vliegenthart et al [49] found a hazard ratio of 4.6 for CAC 400–1,000 compared to <100 after 3.3 years of follow-up.
Subsequently, even more powerful data have emerged. Budoff et al [50] in another all-cause mortality study, with retrospective analysis of 25,203 asymptomatic patients after 6.8 years, found that CAC > 400 was associated with a hazard ratio of 9.2. In the largest study using coronary calcium percentile rather than absolute scores, Becker et al. [51] in 1,724 patients followed prospectively for 3.4 years, reported hazard ratios for CAC percentile >75% vs. 0% of 6.8 for men and 7.9 for women. The area under the ROC curve for CAC percentile (0.81) was significantly superior to the Framingham (0.66), European Society of Cardiology (0.65), and PROCAM risk scores (0.63). Eighty-two percent of patients who developed MI or cardiac death were correctly classified as high risk by CAC percentile, compared to only 30% by Framingham, 36% by the European Society of Cardiology, and 32% by PROCAM.
Perhaps the most important study is the MESA, an NHLBI sponsored prospective evaluation of 6,814 patients followed for 3.8 years [23]. Compared to patients with 0 CAC, the hazard ratios for a coronary event were 7.73 for those with CAC 101–300, and 9.67 among participants for CAC > 300 (p < 0.001) (Table 4.2; Figure 4.10).
Among the four racial and ethnic groups (Caucasian, Chinese, Hispanic, Black), doubling the CAC increased risk of any coronary event by 18–39%. The ROC curve areas were significantly higher (p < 0.001) with the addition of CAC to standard risk factors. CAC was more predictive of coronary disease than carotid intima-media thickness; the hazard ratios per 1-SD increment increased 2.5-fold (95% CI, 2.1–3.1) for CAC and 1.2-fold (95% CI, 1.0–1.4) for IMT [52].
In the 2,684 patients in the female component of MESA [53], Lagoski et al. reported a 6.5 hazard ratio for the 32% with a CAC > 0 vs. the 68% with 0 CAC, even though 90% were low risk by Framingham. In an analysis of all-cause mortality in 44,052 asymptomatic patients followed for 5.6 years [54], the deaths/1,000 patient years were 7.48 for CAC > 10, compared to 1.92 for CAC 1–10, and 0.87 for 0 CAC. Finally, in a meta-analysis of 64,873 patients followed for 4.2 years, the coronary event rate was 1% per year for the 42,283 with CAC > 0, compared to 0.13% per year in the 25,903 patients with 0 CAC [38].
Finally, in the Heinz Nixdorf Recall Study [55], 4,487 subjects without CAD were followed for 5 years. Low ATP III risk was noted in 51.5%, while 28.8 and 19.7% were at intermediate and high risk, respectively. The prevalence of low (<100), intermediate (100–399) and high (≥400) CAC scores was 72.9, 16.8, and 10.3%, respectively (p < 0.0001). The relative risk of CAC >75th vs. ≤25th percentile was 11.1 (p < 0.0001) for men and 3.2 (p = 0.006) for women. Adding CAC to the ATP III categories improved the AUC from 0.602 to 0.727 in men and from 0.660 to 0.723 in women, and led to a reclassification of 77.1% of intermediate risk individuals (62.9% into low risk, and 14.1% into high risk group). The relative risk associated with doubling of the CAC score was 1.32 (95% CI: 1.20–1.45, p < 0.001) in men and 1.25 (95% CI: 1.11–1.42, p < 0.0001) in women.
In all of these studies, receiver operator characteristic curves for CAC were superior to the Framingham Risk Score and the annual event rate for CAC > 100–400 exceeded the CAD equivalent of >2% per year. Table 4.3 summarizes the relative risk results of the largest published outcome studies.
5 Zero Coronary Artery Calcium Scores
Individuals with zero CAC scores have not yet developed detectable, calcified coronary plaque but they may have fatty streaking and early stages of plaque. Noncalcified plaques are present in many young adults. Nonetheless, the event rate in patients with CAC score 0 is very low [40, 45, 46]. Raggi et al. [40] demonstrated an annual event rate of 0.11% in asymptomatic subjects with 0 scores (amounting to a 10-year risk of only 1.1%), and in the St Francis Heart Study [46], scores of 0 were associated with a 0.12% annual event rate over the ensuing 4.3 years. Greenland et al. [45], in a higher-risk asymptomatic cohort, noted a higher annual event rate (0.62%) with 0 CAC scores; a less sensitive CAC detection technique and marked ethnic heterogeneity may have contributed to their findings [56]. In the definitive MESA study [23], 0 CAC was associated with a 0.11% annual event rate. In a meta-analysis of 64,873 patients followed for 4.2 years [54], the coronary event rate was 0.13% per year in the 25,903 patients with 0 CAC compared to 1% per year for the 42,283 with CAC > 0. In an analysis of all-cause mortality in 44,052 asymptomatic patients followed for 5.6 years [54], the deaths/1,000 patient years for the 19,898 with 0 CAC was 0.87, compared to 1.92 for CAC 1–10, and 7.48 for CAC > 10.
While noncalcified, potentially “vulnerable” plaque is by definition not detected by CAC testing, CAC can identify the pool of higher-risk asymptomatic patients out of which will emerge approximately 95% of the patients presenting each year with sudden death or an acute MI. While the culprit lesion contains calcified plaque in only 80% of the acute events [57], of greater importance is the observation that exclusively soft, noncalcified plaque has been seen in only 5% of acute ischemic syndromes in both younger and older populations [12, 58]. In a more recent meta-analysis [38], only 2 of 183 (1.1%) 0 CAC patients were ultimately diagnosed with an acute coronary syndrome after presenting with acute chest pain, normal troponin, and equivocal EKG findings. CAC > 0 had 99% sensitivity, 57% specificity, 24% positive predictive value, and 99% negative predictive value for ACS. Thus, while it is uncommon that a patient with an imminent acute ischemic syndrome would have had a 0 CAC score, further evaluation, particularly with CCTA, is recommended.
6 Compliance with Therapeutic Interventions
With the exception of a single study flawed by insufficient power [59], CAC has been shown to have a positive effect on compliance and initiation of and adherence to medication and life style changes. In 505 asymptomatic patients, statin adherence 3.6 years after visualizing their CAC scan was 90% in those with CAC > 400 compared to 75% for 100–399, 63% for 1–99, and 44% for 0 CAC (p < 0.0001) [60]. Similarly, in 980 asymptomatic subjects followed for 3 years, ASA initiation, dietary changes, and exercise increased significantly from those with 0 CAC (29, 33, 44%, respectively) and was lowest (29%) in those with CAC > 400 (61, 67, 56%, respectively [61]. Finally, after a 6 year follow-up in 1,640 asymptomatic subjects, the odds ratios for those with CAC > 0 compared to 0 CAC for usage of statins, ASA, and statin + ASA were 3.53, 3.05 and 6.97, respectively [62].
7 Coronary Artery Calcium and Guidelines
Guidelines have been increasingly positive regarding the value of CAC scanning. The American College of Cardiology/American Heart Association expert consensus document (2000) concluded that:
-
1.
A negative EBCT test makes the presence of atherosclerotic plaque, including unstable plaque, very unlikely.
-
2.
A negative test is highly unlikely in the presence of significant luminal obstructive disease.
-
3.
Negative tests occur in the majority of patients who have angiographically normal coronary arteries.
-
4.
A negative test may be consistent with a low risk of a cardiovascular event in the next 2–5 years.
-
5.
A positive EBCT confirms the presence of a coronary atherosclerotic plaque.
-
6.
The greater the amount of calcium, the greater the likelihood of occlusive CAD, but there is not a 1-to-1 relationship, and findings may not be site specific.
-
7.
The total amount of calcium correlates best with the total amount of atherosclerotic plaque, although the true “plaque burden” is underestimated.
-
8.
A high calcium score may be consistent with moderate to high risk of a cardiovascular event within the next 2–5 years [27].
The American Heart Association Prevention V Update (2000) suggested that CAC be considered for risk assessment in the 6–20% Framingham 10-year risk category [63]. The final report of the NCEP guidelines [64] made the following recommendation on the basis of existing data at the time of publication (2002):
Therefore, measurement of coronary calcium is an option for advanced risk assessment in appropriately selected persons. In persons with multiple risk factors, high coronary calcium scores (e.g., >75th percentile for age and sex) denotes advanced coronary atherosclerosis and provides a rationale for intensified LDL-lowering therapy.
The European Guidelines on Cardiovascular Disease Prevention in Clinical Practice (2003) state “Coronary calcium scanning is thus especially suited for patients at medium risk,” and use CAC to qualify conventional risk analysis [65]. The American Heart Association Guidelines for Cardiovascular Disease Prevention in Women (2004) listed coronary calcification as an example of subclinical cardiovascular disease (CVD) placing patients in the 10–20% Framingham 10-year risk category and acknowledged that “some patients with subclinical CVD will have >20% 10-year CHD risk and should be elevated to the high-risk category” [66].
In 2006, the SHAPE guidelines (Figure 4.11) recommended CAC or carotid intima-media thickening for all but the lowest risk asymptomatic men >45 and women >55 years, with subsequent treatment based upon the amount of CAC [67].
Based upon the accumulated evidence at the time, which did not yet include the MESA [23] and Becker [51] data, the ACCF/AHA 2007 Clinical Expert Consensus Document [68] judged that in the intermediate risk population “it may be reasonable to consider use of CAC measurement in such patients based on available evidence that demonstrates incremental risk prediction information in this selected (intermediate risk) patient group.” A more robust endorsement is anticipated in future recommendations.
8 Correlation with Risk Factors
8.1 Correlation in Individual Patients
Conventional risk factors do correlate with CAC [69–71], even though CAC is superior to conventional risk factors in predicting outcomes. There is a clear association of CAC with a premature family history of CAD, diabetes, and lipid values in large groups of patients. However, the difficulty equating risk factors with CAC in individual patients has been highlighted by the work of Hecht et al in 930 consecutive primary prevention subjects undergoing EBT [70]. They found increasing likelihoods of CAC with increasing levels of low-density lipoprotein cholesterol (LDL-C) and decreasing levels of high-density lipoprotein cholesterol (HDL-C) in the population as a whole, but found no differences in the amount of plaque between groups and demonstrated a total lack of correlation in individual patients between the EBT calcium percentile and the levels of total, LDL- and HDL-cholesterol, total/HDL-cholesterol, triglycerides, lipoprotein(a) (Lp(a)), homocysteine, and LDL particle size (Figure 4.12).
Correlation between CAC percentile and plasma levels of multiple lipid parameters in 930 asymptomatic patients. Not all correlations are significant (reproduced with permission of Elsevier from Hecht [104]).
Postmenopausal women presented a striking example of the inability of conventional risk analysis to predict the presence or absence of subclinical atherosclerosis [72]. There were no differences in any lipid parameters or in the Framingham Risk Scores between postmenopausal women with and without calcified plaque, rendering therapeutic decisions that are not plaque- imaging-based extremely problematic. Further support for the poor correlation of conventional risk factors with subclinical atherosclerosis was provided by Taylor et al in 630 active duty US Army personnel aged 39–45 years, undergoing EBT [73]. The area under the ROC curve was only 0.62 for the Framingham Risk Score and 0.61 for LDL-C alone. The authors conclude: “In this age homogeneous, low-risk screening cohort, conventional coronary risk factors significantly underestimated the presence of premature, subclinical calcified coronary atherosclerosis.” These discrepancies underscore the difficulties inherent in applying population based guidelines derived from statistical analyzes to decision-making in the real world of individual patient care.
8.2 Advanced Metabolic Testing
The discovery of significant CAC in patients with ostensibly unremarkable lipid values has led to more extensive metabolic analysis in these patients in the search for treatable disorders. In 296 asymptomatic patients with CAC, Superko and Hecht [74] reported a 66% incidence of small, dense LDL; 27% had elevated Lp(a) and 7% had elevated homocysteine. While there is no clear-cut evidence for event reduction by treatment of these abnormalities, consideration should be given to the administration of niacin for small, dense LDL and Lp(a) elevations. The combination of tomographic plaque imaging with metabolic testing and aggressive treatment of identifiable abnormalities has been termed “interventional lipidology” [75].
8.3 Inflammation and Coronary Artery Calcium
8.3.1 hs-CRP
Although still controversial, high sensitivity C-reactive protein (hs-CRP) has been clearly linked to the inflammatory process inherent in CAD. Extensive investigations have led to the suggestion that it be used to screen large segments of the asymptomatic population [76] despite the absence of data demonstrating that hs-CRP is additive to standard risk factors in predicting events. There is, however, evidence that it is significantly inferior to CAC in this capacity. Park et al, in 967 asymptomatic patients, demonstrated that the relative risk of an MI or cardiac death, after adjustment for conventional risk factors, increased four to fivefold from low to high calcium scores at any hs-CRP level, and only 0.25–0.7-fold from low to high hs-CRP at any calcified plaque level (Figure 4.13).
Relative risks (RR) of non-fatal myocardial infarction (MI) or coronary death associated with high (≥75th percentile = 4.05 mg/L) and low (<4.05 mg/L) levels of C-reactive protein (CRP) and high (>142.1), medium (3.7–142.1), and low (<3.7) tertiles of calcium scores (reproduced with permission of Wolters Kluwer from Park et al [78], p. 2076).
In multivariate analysis after adjustment for risk factors, CAC was significantly predictive of events (p< 0.005); hs-CRP was not significantly predictive (p= 0.09) before or after adjustment for CAC [77]. In 323 Framingham Heart Study participants, there was a weak relationship between hs-CRP and CAC in men (r = 0.19, p < 0.05) in multrivariate analysis, and no significant correlation in women [78]. There was no significant relationship between hs-CRP and CAC after multivariate analysis in 914 asymptomatic subjects in the Study of Inherited Risk of Coronary Atherosclerosis [79]. In 1,005 asymptomatic patients randomized to treatment with atorvastatin or placebo in the St Francis Heart Study [80], only the calcium score was significantly associated with disease events (p< 0.0001) in a multivariate analysis including standard coronary disease risk factors, hs-CRP, and baseline coronary calcium score. Hs-CRP did not predict events independently of the calcium score (p= 0.47). There were no correlations between CAC and hs-CRP in either study. In the Dallas Heart Study in 3,373 asymptomatic subjects, there was no significant relationship between hs-CRP and CAC by multivariate analysis [81].
The absence of additional predictive value of hs-CRP and its lack of correlation with CAC do not challenge the inflammatory aspects of the disease process. Rather, it emphasizes the greater value of evidence of the disease itself, namely CAC, compared to a risk marker, such as hs-CRP.
8.3.2 Lipoprotein-Associated Phospholipase A2 (Lp-PLA2)
There is much less data regarding Lp-PLA2. In a nested case-control study among 266 CARDIA participants [82], Lp-PLA2 mass was significantly higher in subjects with CAC compared to those without CAC (OR 1.28). The numbers are too small to provide meaningful conclusions.
8.3.3 Unknown Factors
The lack of clear relationship between lipid levels and subclinical plaque in individual patients does not negate the atherogenic effect of these metabolic disorders. Rather, it highlights the variations in individual susceptibility to the atherogenic effects at a given plasma level, very likely mediated by as yet undetermined genetic factors. O’Donnell et al [83], in an analysis of abdominal aortic calcium in 2,151 patients in 1,159 families in the Framingham Study, noted a heritability component accounting for up to 49% of the variability in calcified plaque, and concluded that “AAC deposits are heritable atherosclerotic traits. A substantial portion of the variation is due to the additive effects of genes, which have yet to be characterized.” Peyser et al. [84], analyzing coronary calcium in 698 patients in 302 families, found a variance of up to 48% associated with additive polygenes after adjustment for covariates. They concluded that there is a:
substantial genetic component for subclinical CAD variation … even after accounting for effects of genes acting through measured risk factors. These genes may act through other measurable risk factors or through novel pathways that have not or cannot be measured in vivo. Identification of such genes will provide a better basis for prevention and treatment of subclinical CAD.
The inevitable conclusion of the consistent lack of relationship between risk factors and disease and the superiority of CAC in individual patients was summarized by Hecht [85]: “The most important role of risk factors may be to identify the modifiable targets of risk reduction in patients with risk already established by clinical events or significant CAC.”
9 Clinical Applications
9.1 Patient Selection
9.1.1 Moderately High Risk
Hecht et al. [86] have proposed recommendations for the application of CAC scanning. (Table 4.4). The Framingham Risk Score [87], incorporating both age and gender, is recommended as the initial step in selecting the appropriate test populations. Asymptomatic patients in the National Cholesterol Education Adult Treatment Program III [88] classified 10–20% Framingham 10-year risk category (moderately high risk) comprise the group that presents the greatest challenge to the treating physician, and are those in whom the application of CAC scoring is most appropriate; the CAC score can assist the physician in decisions regarding recommendations for the use of medications and the degree of emphasis to be placed on lifestyle modifications.
9.1.2 Lower Risk
Patients with less than moderately high Framingham risk may also benefit from CAC scoring to guide management decisions. For instance, most young patients with a family history of premature CAD will not have sufficient risk factors to even warrant Framingham scoring (lower NCEP risk) or will be in the moderate (1–10% 10-year Framingham risk group), since family history, while an NCEP risk factor, does not contribute points to the Framingham score. In 222 young patients presenting with an MI as the first sign of CAD (mean age 50 years), Akosah et al [89] demonstrated that 70% were in these lesser risk categories and would not have been started on a statin using NCEP guidelines. Data from Schmermund et al. [12]. and Pohle et al [58] indicate that 95% of acute MI patients would have been identified by EBT plaque imaging irrespective of age. On the basis of these observations, the use of CAC scoring should be considered in patients with a family history of premature CAD, even when their Framingham risk is moderate or even low.
9.1.3 Higher Risk
Since the Framingham Risk Score is not very accurate in the high risk population, as demonstrated by the St. Francis Heart Study [46], application of CAC scanning to this group is also warranted. In addition, some Framingham high-risk patients may be intolerant of statins or may strongly prefer alternative medicine approaches. In these patients, CAC evidence of high risk may be used to reinforce the necessity for finding a statin that can be tolerated and for persuading the refractory patient of the need for aggressive treatment. Conversely, the absence of significant CAC may permit relaxation of the treatment goals.
9.2 Initiation and Goals of Drug Therapy
9.2.1 New Paradigm
The presence or absence and the amount of CAC can be useful for clinical decision-making, as previously recommended in the AHA Prevention V Update [63]. As an extension of this report, based on recent data, Table 4.4 provides simple, easily implemented treatment paradigms for combining the risks of varying CAC scores with the most recent NCEP recommendations [88]. The SHAPE guidelines [67] go one step further and recommend CAC scanning irrespective of the Framingham Risk Score.
9.2.2 Conversion to Higher or Lower Risk
Patients in the moderately high (10–20% 10-year risk) category who are then identified to be at higher risk by CAC become candidates for secondary prevention lipid goals [87] irrespective of their baseline lipid level. This would apply even for patients with LDL-C <100 mg/dL, as implied by the Heart Protection Study [90] and stated in the most recent NCEP report [88]. Based on prognostic data, CAC >100 or >75th percentile defines a CAD equivalent. In the St Francis Heart Study [46], the CAC cutpoint for initiating secondary prevention therapy was a score >100, and the >75th percentile was suggested by the NCEP guidelines [44]. In this regard, CAC scores >400 [35] or >90th percentile [68] are associated with a very high annual risk (4.8 and 6.5% respectively) and are candidates for the most aggressive approach. These recommendations also apply to initiation of the NCEP guided therapeutic life changes [91] that are an essential component of aggressive prevention.
The transformation of a moderately high-risk to a high-risk patient is shown in Figure 4.14. A 57-year-old man with hypertension, total cholesterol 235 mg/dL, LDL-C 150 mg/dL, HDL-C 75 mg/dL, and a 10-year Framingham risk of 12%, was referred for CAC scanning. The CAC score was 1,872, in the >99th % for his age, placing him in the highest risk category with LDL-C treatment goal of <70 mg/dL.
A 57-year-old man with hypertension, total cholesterol 235 mg/dL, low-density lipoprotein cholesterol (LDL-C) 150 mg/dL, high-density lipoprotein cholesterol (HDL-C) 75 mg/dL, and a 10-year Framingham risk of 12% referred for CAC scanning; CAC score was 1,872, in the >99th percentile. Slices from base (a) through apex (d) reveal significant CAC in all coronary arteries and the ascending aorta. Ao aorta; LAD left anterior descending coronary artery; LADD diagonal branch of left anterior descending coronary artery; LCx left circumflex coronary artery; PDA posterior descending branch of right coronary artery; RCA right coronary artery.
In the Framingham 10–20% 10-year risk population, patients with CAC scores >100 and >75th percentile remain in the same risk group or are transformed to lower-risk categories depending on the score, and are treated accordingly. CAC scores from 10 to 100 and <75th percentile maintain the patient in the moderately high-risk group (10–20% 10-year risk). Patients with CAC scores from 1 to 10 and <75th percentile are reclassified as moderate risk (<10% 10-year risk), and CAC scores of 0 reclassify the patient to the lower-risk category. As noted above, some patients in the lower-risk groups based on Framingham scores, such as younger patients (35–45 years of age) with a strong family history of premature coronary heart disease, are appropriate candidates for CAC scanning. In such patients, the recommendations in Table 4.3 would also apply.
Figure 4.15a displays the CAC scan of a 41-year-old woman whose mother experienced a MI at age 55. The total cholesterol was 188 mg/dL, LDL-C 112 mg/dL, HDL-C 50 mg/dL and triglycerides 132 mg/dL. She was in the 0–1 risk factor group in which a Framingham Risk Score need not be calculated. The CAC score was 110, in the left anterior descending (LAD) and diagonal branch, in the >99th percentile for her age, placing her in a high-risk category. She underwent dual isotope nuclear stress testing (Figure 4.15b), which revealed severe anteroseptal ischemia, followed by angiography and placement of a stent to treat a 95% ostial LAD stenosis (Figure 4.15c). Statin therapy was implemented to reduce the LDL-C to <70 mg/dL.
A 41-year-old woman with a premature family history of coronary artery disease (CAD), total cholesterol 188 mg/dL, LDL-C 112 mg/dL, HDL-C 50 mg/dL, and triglycerides 132 mg/dL, in the lowest Framingham risk group. (a) CAC score of 110, in the LAD and diagonal branch, in the >99th percentile. (b) Dual isotope nuclear stress testing revealing severe anteroseptal ischemia. (c) Angiography demonstrating 95% ostial LAD stenosis and severe LADD disease. LAD left anterior descending coronary artery; LADD diagonal branch of left anterior descending coronary artery.
Patients in the high-risk category (10-year Framingham risk >20%) may be downgraded if the CAC scores do not warrant the highest risk category. CAC scores <100, and, in particular, <10, imply a lower than expected risk and should reduce the intensity of therapy. For instance, a 65-year-old male hypertensive smoker, with an LDL-C of 140 mg/dL and a 10-year Framingham risk of 25%, was very reluctant to take a statin prescribed for his LDL-C. A CAC scan was performed (Figure 4.16), which demonstrated total absence of calcified plaque, despite the presumed high risk. Therapeutic life changes, rather than statins, were recommended.
10 Other Applications
10.1 Diabetes
Diabetic patients deserve special consideration. The NCEP ATP-III guidelines characterize diabetes as a CAD risk equivalent. Raggi et al [92] (Figure 4.17), however, have demonstrated that diabetic patients with 0 CAC scores have the same excellent prognosis as patients without diabetes; it is reasonable to treat those with 0 CAC scores less aggressively than would be dictated as a CAD risk equivalent.
Cumulative survival in nondiabetic (left) and diabetics (center) in relation to CAC score, and in diabetics and nondiabetics with 0 CAC scores (right) (reproduced with permission of Elsevier from Raggi et al [93], p. 1663).
At the same time, diabetic patients have CAC scores corresponding to older people [93, 94] and have a worse prognosis than those without diabetes and similar CAC scores [92], and should be treated more aggressively.
10.2 Evaluation of Therapy
The use of serial CAC scanning to evaluate the progression of disease and the effects of therapy will be covered in great detail in Chap. 5. For this purpose, patients with established CAD who would not ordinarily be candidates for CAC scanning may undergo evaluation as a baseline for future examinations. This may include patients who have had stent placement; the stented area must be excluded from the scoring. In Figure 4.18, the patient underwent stent placement in the right coronary artery. Calcified plaque was noted in the left main and LAD coronary arteries. The non-stented areas are suitable for tracking plaque progression.
Patients with coronary artery bypass grafting are not good candidates for CAC scanning; the profusion of surgical clips makes scoring difficult, and the importance of plaque progression in bypassed areas is unknown.
10.3 Stress Testing
The importance of the relationship of CAC scanning to stress testing has decreased with the growth of CCTA, which has developed as an alternative, and even as a preferential choice to stress testing [95]. While CAC scanning is almost always reserved for patients without symptoms, it has been employed following stress tests in equivocal situations to determine the need for invasive evaluation, irrespective of the symptomatic status. In a series of 118 patients, the absence of coronary calcium accurately identified those with a false-positive treadmill test with a negative predictive value of 90% [96], suggesting that CAC may be useful to enhance the accuracy of abnormal stress tests in patients with a low clinical suspicion of obstructive disease, prior to recommending angiography. In 323 primary prevention patients referred for angiography who underwent electrocardiographic stress testing and calcified plaque imaging, Schmermund et al reported that CAC significantly improved angiographic classification of patients with an equivocal or normal stress evaluation, but not of those with abnormal tests [97]. Figure 4.19 displays the CAC scan of a 51-year-old male smoker with atypical chest pain, obtained after an equivocal nuclear stress test. Extensive plaque and aneurysmal dilatation were demonstrated in both the LAD and right coronary arteries. Subsequent coronary arteriography confirmed the coronary aneurysms and revealed critical LAD stenosis and thrombus.
CAC imaging in a 51-year-old male smoker with atypical chest pain, post equivocal nuclear stress test. Extensive plaque and aneurysmal dilatation were demonstrated in both the LAD (a) and RCA (b). Subsequent coronary arteriography confirmed the coronary aneurysms (c and d) and revealed critical LAD stenosis and thrombus. LAD left anterior descending coronary artery; LADD diagonal branch of left anterior descending coronary artery; RCA right coronary artery.
10.4 Cardiomyopathy
CAC may be used to differentiate ischemic from nonischemic cardiomyopathies. Budoff et al. [98] demonstrated in 120 patients with heart failure of unknown etiology that the presence of CAC was associated with a 99% sensitivity for ischemic cardiomyopathy.
10.5 Emergency Department Chest Pain Evaluation
Emergency department triage of chest pain patients by CAC has been totally supplanted by CCTA. Several early studies demonstrated potential application of CAC to the ED. Laudon et al [99] reported on 105 patients. Of the 46 with positive scores (>0), 14 had abnormal follow-up inpatient testing. Of the 59 with 0 calcium scores, stress evaluation and/or coronary arteriography were normal in the 54 who underwent further testing and all were free of cardiac events 4 months later (100% negative predictive value). Georgiou et al. [100] noted 41 cardiac events in 192 emergency room patients followed for 37 months; all but four were associated with calcium scores ≥4. However, CCTA data have clearly demonstrated a small (5%) but finite incidence of obstructive disease in 0 CAC patients with chest pain [101], mandating performance of CCTA rather than CAC alone in this setting.
11 Conclusions
The validation of CAC scanning as a risk assessment tool may well represent one of the most significant advances in the history of preventive medicine. It offers the possibility of accurately identifying the vast majority of patients destined to suffer acute cardiac events, and, in so doing, should allow for substantial reduction of cardiovascular mortality and morbidity by increasingly effective pharmacologic and lifestyle therapy of the underlying disease process. The sole remaining obstacle to widespread implementation:
… is a double standard that demands randomized controlled (outcome) trials for CAC screening while ignoring their necessity for every other technology…. It is incumbent on the cardiology community to temper the inflexible need for randomized trials with the reality of 565,000 patients presenting with myocardial infarctions annually as their first symptoms, 95% of whom could be identified as at high risk by CAC screening and aggressively treated to significantly reduce events [102].
It is appropriate to conclude by quoting Dr. Scott Grundy [103]:
The power of imaging for detecting subclinical atherosclerosis to predict future ASCVD events is increasingly being recognized. Imaging has at least three virtues. It individualizes risk assessment beyond use of age, which is a less reliable surrogate for atherosclerosis burden; it provides an integrated assessment of the lifetime exposure to risk factors; and it identifies individuals who are susceptible to developing atherosclerosis beyond established risk factors. Also of importance, in the absence of detectable atherosclerosis, short-term risk appears to be very low. Thus, for primary prevention, a recommendation could be established that detection of significant plaque burden is a preferred strategy for initiation of LDL-lowering drugs. With such a recommendation, major risk factors and emerging risk factors could be used as a guide for selecting subjects for imaging more than as a primary guide for therapy. Once subclinical atherosclerosis is detected, intensity of drug therapy could be adjusted for plaque burden. This 2-step approach to risk assessment could provide a solution to the dilemma of patient selection for cholesterol-lowering drugs in primary prevention. In addition, it could be applied to all population subgroups. It could also be useful as a guide to low-dose aspirin prophylaxis and cholesterol-lowering therapy.
References
American Heart Association. Heart and Stoke Statistical Update. Dallas, TX: American Heart Association; 2001.
Blankenhorn DH, Stern D. Calcification of the coronary arteries. Am J Roentgenol. 1959;81:772–777.
Frink RJ, Achor RWP, Brown AL, et al. Significance of calcification of the coronary arteries. Am J Cardiol. 1970;26:241–247.
Wexler L, Brundage B, Crouse J, et al. Coronary artery calcification: pathophysiology, epidemiology, image methods and clinical implications. A scientific statement from the American Heart Association. Circulation. 1996;94:1175–1192.
Faber A. Die Arteriosklerose, from Pathologische Anatomie, from Pathogenese Und Actiologie. In: G. Fischer, ed; 1912.
Bostrom K, Watson KE, Horn S, et al. Bone morphogenetic protein expression in human atherosclerotic lesions. J Clin Invest. 1993;91:1800–1809.
Ideda T, Shirasawa T, Esaki Y, et al. Osteopontin mRNA is expressed by smooth muscle-derived foam cells in human atherosclerotic lesions of the aorta. J Clin Invest. 1993;92:2814–2820.
Hirota S, Imakita M, Kohri K, et al. Expression of osteopontin messenger RNA by macrophages in atherosclerotic plaques. A possible association with calcification. Am J Pathol. 1993;143:1003–1008.
Shanahan CM, Cary NR, Metcalfe JC, Weissberg PL. High expression of genes for calcification-regulating proteins in human atherosclerotic plaque. J Clin Invest. 1994;93:2393–2402.
Rumberger JA, Simons DB, Fitzpatrick LA, et al. Coronary artery calcium areas by electron beam computed tomography and coronary atherosclerotic plaque area: a histopathologic correlative study. Circulation. 1995;92:2157–2162.
Baumgart D, Schmermund A, Goerge G, et al. Comparison of electron beam computed tomography with intracoronary ultrasound and coronary angiography for detection of coronary atherosclerosis. J Am Coll Cardiol. 1997;30:57–64.
Schmermund A, Baumgart D, Gorge G, et al. Coronary artery calcium in acute coronary syndromes: a comparative study of electron beam CT, coronary angiography, and intracoronary ultrasound in survivors of acute myocardial infarction and unstable angina. Circulation. 1997;96:1461–1469.
Agatston AS, Janowitz WR, Hildner FJ, et al. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 1990;15:827–832.
Callister TQ, Cooil B, Raya SP, et al. Coronary artery disease: improved reproducibility of calcium scoring with an electron-beam CT volumetric method. Radiology. 1998;208:807–814.
Becker CR, Kleffel T, Crispin A, et al. Coronary artery calcium measurement. Agreement of multirow detector and electron beam CT. Am J Roentgenol. 2001;176:1295–1298.
Janowitz WR, Agatston AS, Kaplan G, Viamonte M. Differences in prevalence and extent of coronary artery calcium detected by ultrafast computed tomography in asymptomatic men and women. Am J Cardiol. 1993;72:247–254.
Hoff JA, Chomka EV, Krainik AJ, et al. Age and gender distributions of coronary artery calcium detected by electron beam tomography in 35,246 adults. Am J Cardiol. 2001;87:1335–1339.
Budoff MJ, Yang TP, Shavelle RM. Ethnic differences in coronary atherosclerosis. J Am Coll Cardiol. 2002;39:408–412.
Newman AB, Naydeck BL, Whittle J, et al. Racial differences in coronary artery calcification in adults. Arterioscler Thromb Vasc Biol. 2002;22:424–430.
Khuran C, Rosenbaum CG, Howard BV, et al. Coronary artery calcification in black women and white women. Am Heart J. 2003;145:724–729.
Jain T, Peshock R, Darren K, et al. African Americans and Caucasians have a similar prevalence of coronary calcium in the Dallas Heart Study. J Am Coll Cardiol. 2004;44:1011–1017.
McClelland RL, Chung H, Detrano R, et al. Distribution of coronary artery calcium by race, gender, and age. Results from the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation. 2006;113:30–37.
Detrano R, Guerci AD, Carr JJ, et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med. 2008;358:1336–1345.
Nasir K, Michos ED, Rumberger JA, et al. Coronary artery calcification and family history of premature coronary heart disease: sibling history is more strongly associated than parental history. Circulation. 2004;110:2150–2156.
Khurram Nasir K, Budoff MJ, Wong ND, et al. Family history of premature coronary heart disease and coronary artery calcification. Multi-Ethnic Study of Atherosclerosis (MESA). Circulation. 2007;116:619–662.
Gerber TC, Carr JJ, Arai AE, et al. Ionizing radiation in cardiac imaging. A Science Advisory From the American Heart Association Committee on Cardiac Imaging of the Council on Clinical Cardiology and Committee on Cardiovascular Imaging and Intervention of the Council on Cardiovascular Radiology and Intervention. Circulation. 2009; 119:1056–1065.
O’Rourke RA, Brundage BH, Froelicher VF, et al. American College of Cardiology/American Heart Association expert consensus document on electron beam computed tomography for the diagnosis and prognosis of coronary artery disease. Circulation. 2000;102:126–140.
Simons DB, Schwartz RS, Edwards WD, et al. Noninvasive definition of anatomic coronary disease by ultrafast computed tomographic scanning: a quantitative pathologic comparison study. J Am Coll Cardiol. 1992;20:1118–1126.
Detrano R, Tang W, Kang X, et al. Accurate coronary calcium phosphate mass measurements from electron beam computed tomograms. Am J Card Imaging. 1995;9:167–173.
Mautner GC, Mautner SL, Froelich J, et al. Coronary artery calcification: assessment with electron beam CT and histomorphometric correlation. Radiology. 1994;192:619–623.
Budoff MJ, Georgiou D, Brody A, et al. Ultrafast computed tomography as a diagnostic modality in the detection of coronary artery disease-a multicenter study. Circulation. 1996;93:898–904.
Guerci AD, Spadaro LA, Popma JJ, et al. Electron beam tomography of the coronary arteries: relationship of coronary calcium score to arteriographic findings in asymptomatic and symptomatic adults. Am J Cardiol. 1997;79:128–133.
Shavelle DM, Budoff MJ, LaMont DH, et al. Exercise testing and electron beam computed tomography in the evaluation of coronary artery disease. J Am Coll Cardiol. 2000;36:32–38.
Bielak LF, Rumberger JA, Sheedy PF, et al. Probabilistic model for prediction of agiographically defined obstructive coronary artery disease using electron beam computed tomography calcium score strata. Circulation. 2000;102:380–385.
Rumberger JA, Sheedy PF, Breen FJ, et al. Electron beam CT coronary calcium score cutpoints and severity of associated angiography luminal stenosis. J Am Coll Cardiol. 1997;29:1542–1548.
Haberl R, Becker A, Leber A, et al. Correlation of coronary calcification and angiographically documented stenoses in patients with suspected coronary artery disease: results of 1,764 patients. J Am Coll Cardiol. 2001;37:451–457.
Budoff MJ, Raggi P, Berman D, et al. Continuous probabilistic prediction of angiographically significant coronary artery disease using electron beam tomography. Circulation. 2002;105(15):1791–1796.
Sarwar A, Shaw LJ, Shapiro MD, et al. Diagnostic and prognostic value of absence of coronary artery calcification. JACC Cardiovasc Imaging. 2009;2:675–688.
Mohlenkamp S, Lehmann N, Schmermund A, et al. Prognostic value of extensive coronary calcium quantities in symptomatic males – a 5-year follow-up study. Eur Heart J. 2003;24:845–854.
Raggi P, Callister TQ, Cooil B, et al. Identification of patients at increased risk of first unheralded acute myocardial infarction by electron beam computed tomography. Circulation. 2000;101:850–855.
Wong ND, Hsu JC, Detrano RC, et al. Coronary artery calcium evaluation by electron beam compute tomography and its relation to new cardiovascular events. Am J Cardiol. 2000;86:495–498.
Arad Y, Spadaro LA, Goodman K, et al. Prediction of coronary events with electron beam computed tomography. J Am Coll Cardiol. 2000; 36:1253–1260.
Kondos GT, Hoff JA, Sevrukov A, et al. Electron-beam tomography coronary artery calcium and cardiac events: a 37-month follow-up of 5,635 initially asymptomatic low to intermediate risk adults. Circulation. 2003;107:2571–2576.
Shaw LJ, Raggi P, Schisterman E, et al. Prognostic value of cardiac risk factors and coronary artery calcium screening for all-cause mortality. Radiology. 2003;28:826–833.
Greenland P, LaBree L, Azen SP, et al. Coronary artery calcium score combined with Framingham score for risk prediction in asymptomatic individuals. JAMA. 2004;291:210–215.
Arad Y, Goodman KJ, Roth M, et al. Coronary calcification, coronary risk factors, and atherosclerotic cardiovascular disease events. The St Francis Heart Study. J Am Coll Cardiol. 2005;46(1):158–165.
Wayhs R, Zelinger A, Raggi P. High coronary artery calcium scores pose an extremely elevated risk for hard events. J Am Coll Cardiol. 2002;39:225–230.
Taylor AJ, Bindeman J, Feuerstein I, et al. Coronary calcium independently predicts incident premature coronary heart disease over measured cardiovascular risk factors mean three-year outcomes in the prospective army C\coronary C\calcium (PACC) project. J Am Coll Cardiol. 2005;46:807–814.
Vliegenthart R, Oudkerk M, Song B, et al. Coronary calcification detected by electron-beam computed tomography and myocardial infarction. The Rotterdam Coronary Calcification Study. Eur Heart J. 2002;23:1596–1603.
Budoff MJ, Shaw LJ, Liu ST, et al. Long-term prognosis associated with coronary calcification. Observations from a registry of 25,253 patients. J Am Coll Cardiol. 2007;49:1860–1870.
Becker A, Leber A, Becker C, Knez A. Predictive value of coronary calcifications for future cardiac events in asymptomatic individuals. Am Heart J. 2008;155:154–160.
Folsom AR, Kronmal RA, Detrano RC, et al. Coronary artery calcification compared with carotid intima-media thickness in the prediction of cardiovascular disease incidence the multi-ethnic study of atherosclerosis (MESA). Arch Intern Med. 2008;168:1333–1339.
Lakoski SG, Greenland P, Wong ND, et al. Coronary artery calcium scores and risk for cardiovascular events in women classified as “Low Risk” based on Framingham risk score. The Multi-Ethnic Study of Atherosclerosis (MESA). Arch Intern Med. 2007;167(22):2437–2442.
Blaha M, Budoff MJ, Shaw LJ, et al. Absence of coronary artery calcification and all-cause mortality. JACC Cardiovasc Imaging. 2009;2:692–700.
Erbel R, Möhlenkamp S, Moebus S, et al. Coronary Risk Stratification, Discrimination, and Reclassification Improvement Based on Quantification of Subclinical Coronary Atherosclerosis. J Am Coll Cardiol. 2010. In press.
Budoff MJ, Ehrlich J, Hecht HS, Rumberger JR. Letter to the editor. JAMA. 2004;291:1822.
Mascola A, Ko J, Bakhsheshi H, et al. Electron beam tomography comparison of culprit and non-culprit coronary arteries in patients with acute myocardial infarction. Am J Cardiol. 2000;85:1357–1359.
Pohle K, Ropers D, Mäffert R, et al. Coronary calcifications in young patients with first, unheralded myocardial infarction: a risk factor matched analysis by electron beam tomography. Heart. 2003;89:625–628.
O’Malley PG, Feuerstein IM, Taylor AJ. Impact of electron beam tomography, with or without case management, on motivation, behavioral change, and cardiovascular risk profile: a randomized controlled trial. JAMA. 2003;289:2215–2223.
Kalia NK, Miller LG, Nasir K, et al. Visualizing coronary calcium is associated with improvements in adherence to statin therapy. Atherosclerosis. 2006;185:394–399.
Orakzai RH, Nasir K, Orakzai SH, et al. Effect of patient visualization of coronary calcium by electron beam computed tomography on changes in beneficial lifestyle behaviors. Am J Cardiol. 2008;101:999–1002.
Taylor AJ, Bindeman J, Feuerstein I, et al. Community-based provision of statin and aspirin after the detection of coronary artery calcium within a community-based screening cohort. J Am Coll Cardiol. 2008;51:1337–1341.
Smith SC, Greenland P, Grundy SM. Prevention Conference V: beyond secondary prevention: identifying the high-risk patient for primary prevention. Executive summary. Circulation. 2000;101:111–116.
Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Final Report. NIH Publication No. 02–5215; September 2002.
De Backer G, Ambrosioni E, Borch-Johnson K, et al. European guidelines on cardiovascular disease prevention in clinical practice. Third joint task force of European and other societies in cardiovascular disease prevention in clinical practice. Eur J Cardiovasc Prev Rehabil. 2003;10(suppl 1):S1–S10.
Mosca L, Appel LJ, Benjamin EJ, et al.; Expert Panel/Writing Group. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation. 2004;109:672–693.
Naghavi M, Falk E, Hecht HS, et al. From vulnerable plaque to vulnerable patient – part III: executive summary of the Screening for Heart Attack Prevention and Education (SHAPE) Task Force Report. Am J Cardiol. 2006;98(suppl):2H–15H.
Greenland P, Bonow RO, Brundage BH, et al. ACCF/AHA 2007 clinical expert consensus document on coronary artery calcium scoring by computed tomography in global cardiovascular risk assessment and in evaluation of patients with chest pain: a report of the American College of Cardiology Clinical Expert Consensus Task Force (ACCF/AHA Writing Committee to Update the 2000 Expert Consensus Document on Electron Beam Computed Tomography). Circulation. 2007;115:402–426.
Kuller LH, Matthews KA, Sutton-Tyrrell K, et al. Coronary and aortic calcification among women 8 years after menopause and their premenopausal risk factors: The Healthy Women Study. Arterioscler Thromb Vasc Biol. 1999;19:2189–2198.
Hecht HS, Superko HR, Smith LK, et al. Relation of coronary artery calcium identified by electron beam tomography to serum lipoprotein levels and implications for treatment. Am J Cardiol. 2001;87:406–412.
Daviglus ML, Pirzada A, Liu K, et al. Comparison of low risk and higher risk profiles in middle age to frequency and quantity of coronary artery calcium years later. Am J Cardiol. 2004;94:367–369.
Hecht HS, Superko HR. Electron beam tomography and national cholesterol education program guidelines in asymptomatic women. J Am Coll Cardiol. 2001;37:1506–1511.
Taylor AJ, Feuerstein I, Wong H, et al. Do conventional risk factors predict subclinical coronary artery disease? Results from the Prospective Army Coronary Calcium Project. Am Heart J. 2001;141:463–468.
Superko HR, Hecht HS. Metabolic disorders contribute to subclinical coronary atherosclerosis in patients with coronary calcification. Am J Cardiol. 2001;88:260–264.
Hecht HS. “Interventional lipidology”: tomographic plaque imaging and aggressive treatment of metabolic disorders. Am J Cardiol. 2002;90:268–270.
Ridker PM, Wilson PW, Grundy SM. Should C-reactive protein be added to metabolic syndrome and to assessment of global cardiovascular risk? Circulation. 2004;109:2818–2825.
Park R, Detrano R, Xiang M, et al. Combined use of computed tomography coronary calcium scores and C-reactive protein levels in predicting cardiovascular events in nondiabetic individuals. Circulation. 2002;106:2073–2077.
Wang TJ, Larson MG, Levy D, et al. Epicardial coronary calcification in men and women. The Framingham Heart Study. Circulation. 2002;106:1189–1191.
Reilly MP, Wolfe ML, Localio AR, Rader DJ. C-reactive protein and coronary artery calcification: The Study of Inherited Risk of Coronary Atherosclerosis (SIRCA). Arterioscler Thromb Vasc Biol. 2003; 23:1851–1856.
Arad Y, Spadaro LA, Roth M, et al. Treatment of asymptomatic adults with elevated coronary calcium scores with atorvastatin, vitamin C, and vitamin E: The St Francis Heart Study randomized clinical trial. J Am Coll Cardiol. 2005;46(1):166–172.
Khera A, de Lemos JA, Peshock RM, et al. Relationship between C-reactive protein and subclinical atherosclerosis. The Dallas Heart Study. Circulation. 2006;113:38–43.
Iribarren C, Gross MD, Darbinian JA, et al. Association of lipoprotein-associated phospholipase A2 mass and activity with calcified coronary plaque in young adults. The CARDIA study. Arterioscler Thromb Vasc Biol. 2005;25:216–221.
O’Donnell CJ, Chazaro I, Wilson PWF, et al. Evidence for heritability of abdominal aortic calcific deposits in the Framingham Heart Study. Circulation. 2002;106:337–341.
Peyser PA, Bielak LF, Chu J, et al. Heritability of coronary artery calcium quantity measured by electron beam computed tomography in asymptomatic adults. Circulation. 2002;106:304–308.
Hecht HS. Risk factors revisited. Am J Cardiol. 2003;93:73–75.
Hecht HS, Budoff M, Ehrlich J, Rumberger J. Coronary artery calcium scanning: clinical recommendations for cardiac risk assessment and treatment. Am Heart J. 2006;151:1139–1146.
Wilson PWF, D’Agostino B, Levy D, et al. Prediction of coronary heart disease using risk factor categories. Circulation. 1998;97:1837–1847.
Grundy SM, Cleeman JI, Merz CNB, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation. 2004;110:227–239.
Akosah K, Schaper A, Cogbill C, Schoenfeld P. Preventing myocardial infarction in the young adult in the first place: how do the National Cholesterol Education Panel III guidelines perform? J Am Coll Cardiol. 2003;41:1475–1479.
Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20 536 high-risk individuals: a randomised placebo-controlled trial. Lancet. 2002;360:7–22.
Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of The National Cholesterol Education Program (NCEP) (Adult Treatment Panel III). JAMA. 2001;285:2486–2497.
Raggi P, Shaw LJ, Berman DS, Callister TQ. Prognostic value of coronary artery calcium screening in subjects with and without diabetes. J Am Coll Cardiol. 2004;43:1663–1669.
Kuller LH, Velentgas P, Barzilay J, et al. Diabetes mellitus, subclinical cardiovascular disease and risk of incident cardiovascular disease and all-cause mortality. Arterioscler Thromb Vasc Biol. 2000;20:823–829.
Hoff JA, Quinn L, Sevrukov A, et al. The prevalence of coronary artery calcium among diabetic individuals without known coronary artery disease. J Am Coll Cardiol. 2003;41:1008–1012.
Hecht HS. A paradigm shift: coronary computed tomographic angiography before stress testing. Am J Cardiol. 2009;104(4):613–618.
LaMont DH, Budoff MJ, Shavelle DM, et al. Coronary calcium screening identifies patients with false positive stress tests [abstract]. Circulation. 1997;96:306–I.
Schmermund A, Baumgart D, Sack S, et al. Assessment of coronary calcification by electron-beam computed tomography in symptomatic patients with normal, abnormal or equivocal exercise stress test. Eur Heart J. 2000;21:1674–1682.
Budoff MJ, Shavelle DM, Lamont DH, et al. Usefulness of electron beam computed tomography scanning for distinguishing ischemic from non-ischemic cardiomyopathy. J Am Coll Cardiol. 1998;32:1173–1178.
Laudon DA, Vukov LF, Breen JF, et al. Use of electron-beam computed tomography in the evaluation of chest pain patients in the emergency department. Ann Emerg Med. 1999;33:15–21.
Georgiou D, Budoff MJ, Kaufer E, et al. Screening patients with chest pain in the emergency department using electron beam tomography: a follow-up study. J Am Coll Cardiol. 2001;38:105–110.
Rosen BD, Fernandes V, McClelland RL, et al. The prevalence of flow limiting stenoses in coronary arteries with previously documented zero calcium score: The Multi-Ethnic Study of Atherosclerosis (MESA). J Am Coll Cardiol Img. 2009;2:1175–1183.
Hecht HS. The deadly double standard: the saga of screening for subclinical atherosclerosis. Am J Cardiol. 2008;101:1085–1087.
Grundy SM. Is lowering low-density lipoprotein an effective strategy to reduce cardiac risk? Promise of low-density lipoprotein–lowering therapy for primary and secondary prevention. Circulation. 2008;117:569–573.
Hecht HS. Translating tomographic plaque imaging into treatment: interventional lipidology. Prog Cardiovasc Dis. 2003;46(2):149–170.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer-Verlag London Limited
About this chapter
Cite this chapter
Hecht, H.S. (2010). Assessment of Cardiovascular Calcium: Interpretation, Prognostic Value, and Relationship to Lipids and Other Cardiovascular Risk Factors. In: Budoff, M., Shinbane, J. (eds) Cardiac CT Imaging. Springer, London. https://doi.org/10.1007/978-1-84882-650-2_4
Download citation
DOI: https://doi.org/10.1007/978-1-84882-650-2_4
Published:
Publisher Name: Springer, London
Print ISBN: 978-1-84882-649-6
Online ISBN: 978-1-84882-650-2
eBook Packages: MedicineMedicine (R0)