Abstract
Previous studies have reported significant region-dependent differences in the fiber-type composition of human skeletal muscle. It is therefore hypothesized that there is a difference between the deep and superficial parts of muscle energy metabolism during exercise. We hypothesized that the inorganic phosphate (Pi)/ phosphocreatine (PCr) ratio of the superficial parts would be higher, compared with the deep parts, as the work rate increases, because the muscle fiber-type composition of the fast-type may be greater in the superficial parts compared with the deep parts. This study used two-dimensional 31Phosphorus Chemical Shift Imaging (31P-CSI) to detect differences between the deep and superficial parts of the human leg muscles during dynamic knee extension exercise. Six healthy men participated in this study (age 27 ± 1 year, height 169.4 ± 4.1 cm, weight 65.9 ± 8.4 kg). The experiments were carried out with a 1.5-T superconducting magnet with a 5-in. diameter circular surface coil. The subjects performed dynamic one-legged knee extension exercise in the prone position, with the transmit-receive coil placed under the right quadriceps muscles in the magnet. The subjects pulled down an elastic rubber band attached to the ankle at a frequency of 0.25, 0.5 and 1 Hz for 320 s each. The intracellular pH (pHi) was calculated from the median chemical shift of the Pi peak relative to PCr. No significant difference in Pi/PCr was observed between the deep and the superficial parts of the quadriceps muscles at rest. The Pi/PCr of the superficial parts was not significantly increased with increasing work rate. Compared with the superficial areas, the Pi/PCr of the deep parts was significantly higher (p < 0.05) at 1 Hz. The pHi showed no significant difference between the two parts. These results suggest that muscle oxidative metabolism is different between deep and superficial parts of quadriceps muscles during dynamic exercise.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Previous study has reported that there are significant region-dependent differences in the fiber-type composition of human skeletal muscle [1, 2]. It is therefore hypothesized that there is a difference between the deep and superficial parts of muscle energy metabolism during exercise. Chance et al. [3] have reported that high oxidative capacity muscle demonstrated lower inorganic phosphate (Pi)/ phosphocreatine (PCr) at the same relative work rate compared with low oxidative capacity muscle. Recently, creating a localized metabolic map during exercise has been possible using 31Phosphorus Chemical Shift Imaging (31P-CSI) [4–6]. Therefore, the purpose of this study was to detect metabolic disturbances between the deep and superficial parts of the human leg muscles during dynamic knee extension exercise using two-dimensional 31P-CSI. We hypothesized that the Pi/PCr of the superficial parts would be higher, compared with the deep parts, as the work rate increases, because the muscle fiber-type composition of the fast-type may be higher in the superficial parts compared with the deep parts.
2 Methods
2.1 Subjects
Six healthy men (age: 28 ± 1 year, height: 170.4 ± 4.1 cm, weight: 66.8 ± 7.4 kg) participated in this study. All subjects were briefed about the experimental protocol, and written informed consent was obtained before the experiment. The institutional review board of the Tokyo Medical University approved the research protocol.
2.2 Experimental Design
After receiving written informed consent from the subjects, the experiments were carried out with a 1.5-T superconducting magnet (GE Healthcare, Milwaukee, WI, USA) with a circular surface coil (GE Healthcare, Milwaukee, WI, USA) double-tuned to 1H at 63.5 MHz and 31P at 25.8 MHz. The subjects lay prone in the bore to obtain T2-weighted 1H images. After completion, regional differences in phosphorus signals were obtained by 31P-CSI. Before the 31P-CSI acquisitions, the magnetic field homogeneity was optimized using the localized water signal from a quadriceps muscles.
2.3 Measurements
A one-pulse 31P-MRS acquisition was carried out with a 5-in. transmit/receive surface coil placed under the knee extensors [rectus femoris (RF), vastus medialis (VM), vastus intermedius (VI), and vastus lateralis (VL)] of the right leg. Spatially resolved acquisition relied on 31P-CSI with 3-cm slice thickness, and a 24-cm2 field of view. The volume of each voxel was 3 × 3 × 3 cm, or 27 cm3. A TR of 1000 ms was used, and a two-dimensional 31P metabolite map was generated every 388 s. This acquisition time was determined from pilot measurements to ensure good spectral data from the quadriceps and represents an optimal compromise between signal-to-noise, temporal resolution, and spatial resolution.
31P-CSI post processing was done using Mnova software (Mestrelab Research, Spain). Baseline correction was performed semi-automatically by setting the peak ranges of Pi and PCr as references, and phase correction was applied semi-automatically using Pi and PCr as reference peaks. Supplementary manual phase or baseline correction was performed, if necessary. The peak areas and peak positions of PCr and Pi were fitted in the frequency domain. The intracellular pH (pHi) was calculated from the median chemical shift of the Pi peak relative to PCr [7].
At rest, T2-weighted 1H images were obtained with the whole body imaging coil with TR = 3200 ms and TE = 95 ms. Scan time for T2 image was 39 s.
2.4 Exercise Protocol
The subjects performed dynamic one-legged knee extension exercise in the prone position. A broad nonelastic strap over the hips served to stabilize the subject during exercise. A rubber band was attached to the ankle and the subjects exercised by expanding the rubber band at 0.25, 0.5 and 1 Hz for 380 s each. As limited by the scanner bore, a range of motion of knee extension exercise was 0–30°.
2.5 Statistics
The changes in Pi/PCr and pHi during the experiments were analyzed by two-way ANOVA for repeated measurements. The significance level was set to 0.05. Statistics were completed using the Statistical Package for the Social Sciences (SPSS) Statics (IBM, Chicago, IL).
3 Results
Typical examples of 31P-CSI spectra are shown in Fig. 6.1. To compare the metabolic differences between superficial and deep parts, three voxels of superficial parts and three of voxels of deep parts were selected, as all the data from the voxel could be monitored by all subjects. No significant difference in Pi/ PCr was observed between the deep and the superficial parts of the quadriceps muscles at rest (superficial parts; 0.21 ± 0.04, deep parts; 0.18 ± 0.03). At the superficial parts, the Pi/PCr were not significantly changed with increasing work rate. Compared with the superficial parts, the Pi/PCr of the deep parts were significantly higher (p < 0.05) at 1 Hz, and the highest peak of Pi/PCr was 0.85 ± 0.08 at D1. The averaged resting pHi was 7.08 ± 0.02, and the spatial differences of the resting pHi were not observed between every measurement part. Also, the pHi during exercise showed no significant difference between the two parts at all exercise frequencies (superficial parts; 7.02 ± 0.04, deep parts; 6.99 ± 0.03) (Fig. 6.2).
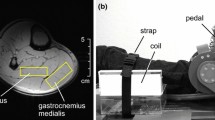
Typical example of 31P-CSI spectra and two-dimensional 1H magnetic resonance image in thigh muscles. Each was 3 × 3 × 3 cm, or 27 cm3. To compare the metabolic differences between superficial and deep parts, three voxels of superficial parts and three of voxels of deep parts were selected, as all the data from the voxel could be monitored by all subjects. The voxel was numbered from the medial to lateral parts at both superficial and deep parts
The upper figures show the Pi/PCr of deep parts at rest and during dynamic exercise at various work rates. The bottom figures show the Pi/PCr of superficial parts at rest and during exercise at various work rates. Double asterisks denote significant differences compared with rest below the 0.01 level. Double hash symbols denote significant differences compared with superficial parts below the 0.01 level
4 Discussion
The study demonstrated that the Pi/PCr of the superficial parts were not significantly increased with increasing work rate. In comparison, the Pi/PCr of the deep parts were significantly higher at 1 Hz. These results suggest that muscleoxidative metabolism is different between deep and superficial parts of quadriceps muscles during dynamic exercise. Pesta et al. [4] recently reported that PCr changes during exercise were uniform across the quadriceps muscles within sprint-trained, endurance-trained and untrained groups, and the results in the present study are different from the reported data. The reason for the discrepancy is uncertain but may be due to differences in both the type of exercise and exercise time. In almost all previous studies, work rate was increased by altering workload while keeping the rate of contraction steady. Therefore, more fast-twitch fibers may be recruited at higher workloads, and the type of exercise can lead to physiological maximal effort. In contrast, the work rate in the present study was increased by altering the rate of contraction while keeping the workload steady. As this type of exercise reduces the relaxation period of the duty cycle, muscle recruitment may be lower at increasing rate of contraction protocol than increasing workload protocol. In addition, the exercise time was long (388 s) for acquisition of spatially-resolved spectra in each voxel examined, due to limitations in our technique. In the future, we need to shorten the acquisition time by increasing slice thickness. Also, we need to localize the specific muscle signals of the thigh muscles such as rectus femoris and vastus intermedius.
Although muscle recruitment patterns may affect Pi/PCr independently of fiber type composition, we were unable to measure T2-weighted imaging using f-MRI during exercise. Further investigations are needed to determine the regional differences in muscle energy metabolism and muscle recruitment patterns.
5 Conclusions
We determined the difference between the deep and superficial areas of muscle energy metabolism during dynamic knee extension exercise using 31P- chemical shift imaging. The study demonstrated that the Pi/PCr of the superficial parts were not significantly increased with increasing work rate. In contrast, the Pi/PCr of the deep parts were significantly higher at a higher work rate. These results could reflect (a) regional differences in fiber composition, which was our original hypothesis, or (b) different regional recruitment patterns for this particular form of exercise. These two explanations cannot be discriminated from the present study.
References
Johnson MA, Polgar J, Weightman D et al (1973) Data on the distribution of fibre types in thirty-six human muscles. An autopsy study. J Neurol Sci 18(1):111–129
Dahmane R, Djordjevic S, Simunic B et al (2005) Spatial fiber type distribution in normal human muscle: histochemical and tensiomyographical evaluation. J Biomech 38(12):2451–2459
Chance B, Leigh JS Jr, Clark BJ et al (1985) Control of oxidative metabolism and oxygen delivery in human skeletal muscle: a steady-state analysis of the work/energy cost transfer function. Proc Natl Acad Sci U S A 82:8384–8388
Pesta D, Paschke V, Hoppel F et al (2013) Different metabolic responses during incremental exercise assessed by localized 31P MRS in sprint and endurance athletes and untrained individuals. Int J Sports Med 34(8):669–675
Cannon DT, Howe FA, Whipp BJ et al (2013) Muscle metabolism and activation heterogeneity by combined 31P chemical shift and T2 imaging, and pulmonary O2 uptake during incremental knee-extensor exercise. J Appl Physiol 115(6):839–849
Forbes SC, Slade JM, Francis RM et al (2009) Comparison of oxidative capacity among leg muscles in humans using gated 31P 2-D chemical shift imaging. NMR Biomed 22(10):1063–1071
Petroff OA, Prichard JW, Behar KL et al (1985) Cerebral intracellular pH by 31P nuclear magnetic resonance spectroscopy. Neurology 35:781–788
Acknowledgments
The authors are grateful for revision of this manuscript by Andrea Hope. This study was supported in part by Grant-in-Aid for scientific research from the Japan Society for Promotion of Science (24500799) to R. K.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media, New York
About this paper
Cite this paper
Kime, R., Kaneko, Y., Hongo, Y., Ohno, Y., Sakamoto, A., Katsumura, T. (2016). Regional Differences in Muscle Energy Metabolism in Human Muscle by 31P-Chemical Shift Imaging. In: Elwell, C.E., Leung, T.S., Harrison, D.K. (eds) Oxygen Transport to Tissue XXXVII. Advances in Experimental Medicine and Biology, vol 876. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-3023-4_6
Download citation
DOI: https://doi.org/10.1007/978-1-4939-3023-4_6
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-3022-7
Online ISBN: 978-1-4939-3023-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)