Abstract
Comparative studies of diverse species provide a wealth of information about active touch and corresponding brain specializations in the somatosensory system. Here the results of numerous studies of brain and behavior in shrews and moles are reviewed and discussed. Water shrews have elaborate whiskers and can detect prey based on both texture and movement. In contrast to rodents, shrew whiskers are not reflected by barrels in the cortex, but are reflected in the brainstem by prominent barrelettes. Although shrews have a simpler cortical anatomy than rodents, star-nosed mole’s cortices are more complex, with three histologically visible and interconnected cortical maps that reflect the nasal rays on the contralateral star. One ray of the star is used as the tactile fovea, and is greatly over-represented in the neocortex. This finding highlights similarities between specialized somatosensory, visual, and auditory systems—each of which may have a sensory fovea for high resolution sensory processing. Both water shrews and star-nosed moles exhibit the remarkable ability to sniff underwater by exhaling and reinhaling air bubbles as they forage. This allows visualization of sniffing during natural behaviors and provides a unique window into the behavioral integration of touch and smell . Finally, eastern moles have the least specialized set of mechanoreceptors but exhibit remarkable olfactory abilities using stereo nasal cues—in conjunction with touch—to efficiently locate prey. These results highlight the many insights that may be derived from specialized model animals.
Supported by NSF grant 1456472 to KCC
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Tactile
- Touch
- Olfaction
- Smell
- Stereo
- Mechanosensory
- Barrel
- Barrelette
- Whisker
- Brain Evolution
- Shrew
- Mole
- Neocortex
- Trigeminal
- Behavior
Introduction
Investigations of sensory and motor specialists have provided many key insights into brain organization, function, and evolution . Perhaps the best-known example of this strategy is Hodgkin and Huxley’s landmark studies of the giant axon that mediates escape responses in squid, which revealed the ionic basis of action potential conduction [1, 2]. Some other well know examples include studies of barn owls for understanding the neural basis of auditory localization based on coincidence detection [3–5], the use of electric fish for determining the neural basis of rhythmic signaling, jamming avoidance, and animal communication [6, 7], and the study of songbirds for determining the plasticity of networks mediating social learning [8–10]. In a similar way, the specialized whisker-barrel system of rodents has been particularly useful for understanding the neural basis of touch because rodents have an elaborate somatosensory system and at the same time they share many features in common with other mammals. Most importantly, they have a neocortex with somatosensory areas that are homologous to the somatosensory areas found in nearly all other mammals including humans [11]. This homology from mouse to man allows inferences about basic cortical circuitry to be more confidently extended to a wide range of other mammal species. But the key technical advantage of the rodent system was the discovery of histologically visible units, or barrels, in the primary somatosensory system of mice [12] and subsequently rats [13]. The later discovery of similar barrel-like subdivisions at the thalamic [14] and brainstem [15] level (barreloids and barrelettes, respectively) added another dimension to the system, providing the advantages of “visible” whisker maps in the entire pathway from mechanoreceptors to primary somatosensory cortex. These findings greatly facilitated subsequent investigations of neuronal electrophysiology, connectivity, development, and plasticity. More recently these studies have been integrated with detailed behavioral and biomechanical studies, providing one of the most comprehensive views of brain and behavior for any mammalian species.
At the same time that our understanding of rodents’ somatosensory systems have been expanding, advances in the technique of flattening cortex by carefully removing underlying white matter before compressing the cortical hemispheres have provided ever more clear views of the histological patterns in layer 4 cortex in diverse species. This includes the discovery of whisker related barrels in numerous rodents, marsupials, and insectivores. Modules representing alternating electrosensory and mechanosensory inputs have been described in the cortex of the duck-billed platypus [16] and stripes corresponding to nasal appendages have been identified in both S1 and S2 of the star-nosed mole [17, 18]. In the case of primates, myelin-dark modules representing individual fingers have been described in the hand area of area 3B [19, 20]. The latter finding suggests that similar mechanisms may segregate cortical (and subcortical) inputs from discontinuous sensory surfaces into modules during development in diverse species, ranging from rodents to primates.
Clearly there is a rich source of diversity for revealing general principles of brain organization and development by examining a range of different mammalian somatosensory systems. In this chapter we will provide an overview of the brains and behavior of the water shrew, the star-nosed mole, and the eastern mole. Each of these species is differently specialized in a manner that illuminates a particular facet of sensory biology. Like rats and mice, water shrews have an exquisitely specialized whisker system used to explore their environment. Yet, despite sharing similar mechanoreceptors (whiskers) the central representation of those receptors is strikingly different in the neocortex. Moles on the other hand are also touch specialists, but instead of whiskers they rely on specialized skin surfaces to explore their environment. As in the barrel system , the nasal appendages of star-nosed moles are reflected at cortical and subcortical levels by modules isomorphic with the sensory surface. But in this case, they appear as stripes rather than traditional columns and their sizes reflect the differential behavioral importance of different sensory appendages. This species provides an additional example of modular, visible brain maps and illustrates parallels between high-resolution somatosensory systems, visual systems, and auditory systems. Finally, eastern moles have recently been shown to integrate their somatosensory exploration with the use of bilateral comparisons of olfactory cues (stereo smell) for food localization. Together these insectivores demonstrate a wide range of peripheral mechanoreceptors, diverse cortical representations, and interesting behaviors.
Water Shrews—Variations on a Theme
Figure 2.1 shows a predatory grasshopper mouse (Onychimys leucogaster) alongside of a water shrew (Sorex palustris). These two species nicely illustrate some of the commonalities and differences in anatomy and brain organization found among mammals. First, we should point out that water shrews are not rodents, they are part of the historical order Insectivora that includes moles, shrews , hedgehogs, and solenodons. Thus, despite appearances, shrews are only very distantly related to rodents. Like all other shrew species, the water shrew is a predator. The grasshopper mouse, on the other hand, is a rodent, albeit it has the distinction of being one of the few predatory rodent species. Both species use their elaborate whiskers in active touch as they identify prey and guide attacks on fast moving and sometimes dangerous invertebrates (grasshopper mice feed on scorpions). Yet despite this similarity in form and function, the cortical representation of the whiskers is very different between these two small mammals.
Comparison of a rodent and an insectivore. Although the grasshopper mouse (a) and the water shrew (b) are both predatory and locate prey using whiskers, they have very different sensory cortices. (c) The flattened cortex of the grasshopper mouse shows very prominent cortical barrels (dark circles labeled with the serotonin transporter antibody) and large primary visual and auditory areas. (d) The flattened cortex of the water shrew shows a large somatosensory cortex with two large whisker representations, but there are no visible barrels. Note also the very small areas of sensory cortex devoted to vision and olfaction in (V1 and Aud, respectively). Data in (b) from [61]. Data in (d) from [26]. Photo in (a) by Jan Decher. (Abbreviations: Aud auditory, V1 primary visual cortex, Oral oral). (Published with kind permission of © Kenneth Catania and Jan Dreher 2014)
The flattened juvenile neocortex of the grasshopper mouse (Fig. 2.1c), labeled in this case with the serotonin transporter antibody, appears much like that of other rodent species similarly prepared. It has a patently visible primary somatosensory cortex (S1) containing subdivisions that can be very easily recognized as representing the same body parts that are visible in cortex of laboratory rats and mice. This includes a barrel pattern that clearly reflects the prominent facial whiskers . In contrast, the juvenile water shrew neocortex (in this case processed for the metabolic enzyme cytochrome oxidase (CO)) contains a prominent whisker representation (see [21] for physiological recording data), but no obvious barrels representing the large facial whiskers (Fig. 2.1d). Why this striking difference in brain organization between physically similar animals with otherwise similar peripheral anatomical features? The answer is not clear, but additional aspects of water shrew behavior may provide some clues.
Water Shrew Senses
Water shrews are adept predators that forage primarily at night along the sides of streams and ponds in North America. It seems remarkable that these animals, the world’s smallest mammalian divers, can make a living and avoid predators using this foraging strategy, and it is natural to wonder about the relative contribution of their different senses to this activity. Figure 2.1 (c and d) provides an important and obvious clue to the sensory priorities of this species. In contrast to the grasshopper mouse, water shrews have tiny eyes. This anatomical feature is in turn reflected in their neocortex. Water shrews have a very small primary visual area (V1) compressed to the far caudal and dorsal aspect of the hemisphere. Somatosensory cortex appears to have “taken over” much of the cortical territory. Though this last interpretation is almost certainly backwards. Because shrews resemble ancestral mammals in many respects [22], it is more likely that visual cortex in rodents has “taken over” territory that was once somatosensory during the course of evolution. In any case, visual cortex is very small in water shrews, and the same is true for auditory cortex at the more caudal and lateral extreme of the hemisphere (see [21] for shrew electrophysiology). The latter observation is of interest because it has been suggested that some shrews may echolocate [23, 24]. This would be surprising in the case of water shrews, as auditory cortex is very small. Indeed, experiments show water shrews do not use echolocation [25]. In concordance with these observations, counts of cranial nerve number in water shrews reveal a tiny optic nerve (6000 fibers) and an equally small auditory nerve (7000 fibers). In contrast, the trigeminal nerve carrying information from the whiskers contains 27,500 fibers—similar in size to that of laboratory mice [26].
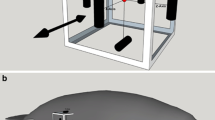
To investigate water shrew behavior and the possible contribution of vision in foraging, shrews were offered live fish in a small chamber under either full spectrum lighting or infrared lighting (Fig. 2.2a). Shrews were very efficient and equally fast at capturing fish under both conditions, demonstrating that vision was not required for this behavior. Many fish were captured in less than one second from the time the shrew entered the water [25]. Slow motion analysis of water shrews capturing fish suggested that water motion generated by fish escape responses might be an important cue used to identify the location of prey. To further investigate this possibility, water shrews were presented with very brief, periodic pulses of water in the absence of prey and filmed with high speed video. This paradigm was designed to simulate the brief water disturbance caused by an escaping fish. The results clearly showed that water shrew attacks were triggered by brief water movements (Fig. 2.2b, c). In addition to illustrating that water shrews may use prey escape responses for localization, the experiments further highlight their reliance on somatosensation, rather than vision, as the water movements were not visible [25]. Finally, water shrews were incredibly fast, attacking the stimulus with a latency of only 20 milliseconds (from stimulus to initiation of attack).
Water shrews detect motion and can capture prey in water without the use of vision. a Schematic illustration of the chamber used to examine the foraging efficiency of water shrews capturing live fish under either full spectrum lighting or infrared lighting. Shrews were filmed with a high-speed camera. Shrews were equally efficient under both lighting conditions. b Schematic illustration of the chamber used to test responses to brief water pulses simulating escaping fish. Shrews attacked the water motion with a short latency. c Frame captured from high-speed video showing a shrew attacking the water motion in the absence of prey. (Published with kind permission of © Kenneth Catania 2014
The experiments described above highlight the strategy shrews use to locate active prey, but shrews also feed on many immobile invertebrates. To investigate their responses to shapes and textures , rather than just movement, water shrews were presented with simulated, highly detailed caste silicone fish, along with a series of rectangular and spherical shapes as distractors (Fig. 2.3a). Even in the absence of visual or olfactory information the shrews were dramatically successful at choosing the silicone fish over the similarly sized silicone shape models (Fig. 2.3c). This demonstrated that water shrews cannot only detect movements, but they can also use their whiskers to identify objects via shape and texture. As might be expected, with no reward for retrieving inedible silicone fish, the shrews soon stopped capturing these imposters. But if caste fish were made to move (by placing a small piece of metal in them and moving them with a magnet) the water shrews’ responses were resurrected and they again attacked the silicone fish in preference to the other objects. Together these results show the value of both movement and shape in eliciting attacks (Fig. 2.3d). This seems appropriate, given that small prey hidden in the shallow water along streams and ponds would be expected to exhibit distinctive shapes and textures and some would also be likely to move (e.g. escape responses of fish and crayfish, for example). Other shrews have also been shown to use prey shape as an important criterion for predatory attack [27].
Water shrews use their whiskers to detect texture/shape of objects. a Schematic illustration of the chamber used to test water shrews’ ability to detect an object without olfactory or visual cues. Three silicone rectangles and three silicone cylinders were placed in the chamber, along with a silicone model fish. b A water shrew attacks and grabs a silicone fish under infrared lighting. Shrews often took the model fish back to their home cages. c Graph showing the average number of times over 4 trials that each of 4 shrews bit either a distractor object (1–6) or the model fish “F”. d Graph showing the average number of attacks (retrieving, biting or lunging with open mouth) for each moving object for 3 shrews over 4 trials. Objects were moved with a magnet under the chamber. (Published with kind permission of © Kenneth Catania 2014)
Underwater Sniffing
As suggested by their anatomy, behavioral experiments indicate that water shrews depend heavily on their whiskers to locate prey while foraging . Yet their speed and efficiency raise the possibility that other senses might be involved. As described previously, there was no evidence for the use of echolocation or sonar. In addition, we tested for the ability to detect electric fields, both in terms of behavioral responses and by surveying the skin surface of the head to detect potential electroreceptive organs. There was no evidence for electroreception in terms of behavior or peripheral anatomy. However, water shrews were able to use olfaction in a very unique way. When searching for prey while submerged, they emitted air bubbles from their nostrils that spread over objects they were exploring and then re-inhaled the same air (Fig. 2.4).
Ten frames taken from high-speed video showing a single underwater sniff by a water shrew. In this case the shrew is sniffing a small piece of wax. The animal has paused during its movements and expires air (upper row) that comes in direct contact with the object. This air is then re-inhaled (lower row). Using this strategy, water shrews can follow a submerged scent trail. (Published with kind permission of © Kenneth Catania 2014)
This behavior was remarkable, because it had all the characteristics of sniffing , but occurred underwater (the behavior was first observed in semi-aquatic star-nosed moles, see later section). To investigate this further, shrews were trained to follow a scent trail underwater in a two choice test. They were very proficient at following the trail as long as the emitted air bubbles could make direct contact with the scent trail they were following [28]. When the air bubbles were blocked with a stainless steel grid, the shrews’ performances dropped to chance, despite the close proximity of the scent trail. This form of underwater sniffing seems to require direct contact of the air with odorants to provide relevant information.
Water Shrew Brainstem—Barrelettes without Barrels
When first investigating the neocortex of shrews [21] one of our interests was determining whether this lineage of mammals exhibited cortical barrels. Five different shrew species (including water shrews) were examined using eletrophysiological mapping with dense microelectrode penetrations combined with subsequent analysis of flattened cortical sections processed for CO. The primary (S1) and secondary (S2) somatosensory areas were both identified. S2 was larger in shrews compared to most other mammal species that have been investigated, taking up roughly the same amount of neocortex as S1 and being characterized by neurons with relatively small receptive fields. As expected from shrew behavior and the cranial nerve counts described above, both S1 and S2 were dominated by large representations of the whiskers from the contralateral side of the face. The S1 representation of the whiskers was visible by a CO dark wedge of tissue in most species. The S1 whisker representation was most obvious in the smallest shrew species (the masked shrew, Sorex cinereus) [21]. But in no case, for any species, were cortical barrels apparent.
As is familiar to most investigators of small mammal cortical histology, cyto- and chemoarchitectural borders and modules such as barrels are usually more apparent in juvenile animals than in adults. When water shrews fortuitously gave birth in the lab, we once again examined somatosensory cortex, this time in juveniles [26]. The goal was to specify borders between areas in greater detail for these unique species and to search once more for cortical barrels that might be evident at early stages of development but later obscured. We were successful at more clearly delineating borders of sensory areas and even numerous subdivisions representing body parts, especially the large, S1 whisker representation marked by the wedge of CO dark tissue. But, once again, we concluded there were no cortical barrels apparent even at juvenile stages of cortical development [26].
With these previous investigations in mind, we were surprised to later discover in the juvenile water shrew brainstem [29] perhaps the clearest and most prominent barrelettes yet observed in a mammal (Fig. 2.5b, c, d). Barrelettes were apparent in the principle nucleus (PrV), the interpolar spinal trigeminal nucleus (SpI), and the caudal spinal trigeminal nucleus (SpC). Barrelettes were apparent in adult water shrew brainstem as well, though (as is the case for rodents) they were slightly less clear than in juveniles. Injection of anatomical tracers into the adult water shrew whisker pad indicated that barrelettes in shrews, as in rodents, reflect the selective aggregation of afferent terminals from the whiskerpad [29].
Water shrews have barrelettes without barrels. (a) Flattened juvenile water shrew cortex processed for cytochrome oxidase and showing the large whisker representation devoid of barrels. (b–d) Prominent barrelettes are visible in trigeminal sensory nuclei: (b) the principle trigeminal nuclei (PrV), (c) the interpolar spinal trigeminal nucleus (SpI) and (c) the caudal spinal trigeminal nucleus (SpC) of juvenile water shrews. Scale = 0.5 mm. (Published with kind permission of © Kenneth Catania 2014)
These findings highlight the different ways that whiskers can be represented in diverse mammals. In rodents, cortical barrels representing the whiskers are the most obvious, whereas trigeminal barrelettes and thalamic barreloids are much less clear. In contrast, trigeminal barrelettes in water shrews are strikingly clear despite the absence of barrels at the cortical level.
Star-Nosed Moles
Olfaction might be the first thing that comes to mind when one considers a star-nosed mole (Condylura cristata). In fact, recent studies show that star-nosed moles and their relatives have impressive olfactory abilities, but the star is a tactile organ , not a chemoreceptor. It consists of 22 epidermal appendages that ring the nostrils in 11 symmetric pairs (Fig. 2.6a). Each appendage, or “ray” is covered with many hundreds of small epidermal domes called Eimer’s organs (Fig. 2.6b). Together they are innervated by over 100,000 myelinated nerve fibers, giving this skin surface, which is only about a centimeter across, the highest innervation density of any known skin surface. Eimer’s organs are a characteristic feature of mole nasal epidermis and are found on the skin of almost all of the nearly 30 different mole species [30]. But only the star-nosed mole has evolved nasal rays that increase the surface area of the sensory epithelium providing room for 25,000 Eimer’s organs. Evolution of this delicate structure could probably only occur in the star-nosed mole’s wetland environment, a unique habitat for moles, and this at least partially explains why no other mole has such an elaborate and fragile snout. Because the star is essentially made of Eimer’s organs, knowing the function of these structures is fundamental for understanding the star.
Anatomy of the star. a Star-nosed moles have an impressive epidermal specialization on their nose consisting of 22 appendages (rays) that surround their nostrils. b Each ray is covered with small domes called Eimer’s organs that are densely innervated. c Top view of nerve endings in a single Eimer’s organ visualized with DiI. The star has the highest innervation density of any known skin surface. d Each Eimer’s organ contains a Merkel cell-neurite complex, a lamellated corpuscle, and free nerve endings. (Published with kind permission of © Kenneth Catania 2014)
Function of Eimer’s Organs
Eimer’s organs were first described in the 1800s by Theodor Eimer in the European mole [31] and they were subsequently found on each mole species that was investigated with the exception of the eastern mole (Scalopus aquaticus) [32]. Most investigators concluded that Eimer’s organs must have a mechanoreceptive function based on their anatomy and mole behavior . Each organ is associated with Merkel cell-neurite complexes, lamellated corpuscles, and free nerve endings (Fig. 2.6c, d) [33–35]. In addition, moles repeatedly touch the skin surface containing Eimer’s organs to objects or prey as they explore their environment and search for food. More direct evidence for a mechanosensory function comes from electrophysiological recordings from the somatosensory cortex [17, 18], from afferents supplying Eimer’s organs [36], and from findings in the principle trigeminal sensory nucleus (PrV) [37].
The first direct evidence of Eimer’s organ responses came from electrophysiology recordings in the somatosensory cortex of star-nosed moles [17, 38]. Multi-unit receptive fields were extremely small and often had to be defined with the aid of a microscope. Even so, the lower limit of receptive field size was probably not determined given the limitations of manual stimulation of the skin surface. Nevertheless receptive fields on the star were well under a millimeter in diameter in some areas. Even at this early stage of investigations, there was an evident trend in relative receptive field size with the smallest receptive fields located on the midline and ventral parts of the star and larger receptive fields found for the more lateral parts of the star (see next section for correlations with behavior). Single unit analysis revealed that roughly half of cortical neurons were inhibited when areas just outside their excitatory receptive fields were stimulated—i.e. they demonstrated surround inhibition.
Later recordings from primary afferents in both star-nosed moles and coast moles provide additional evidence for Eimer’s organ function [36]. Three different response classes were evident in both species using either a dedicated Chubbuck mechanosensory stimulator [39] or a piezo bending element stimulator. These responses consisted of a Merkel-like response with sustained volleys of action potentials having variable interspike intervals, a Pacinian like response that was evident only at the onset and offset of skin depression (stimulation), and a rapidly adapting response that was directionally sensitive to a sweeping motion across the skin surface [36]. These responses were consistent with the three receptor classes associated with each Eimer’s organ. The most interesting response was the directionally sensitive afferents that suggest a roll for Eimer’s organs in detecting minute surface features on objects and prey items in the moles’ environments [36].
Stars and Stripes in the Brain
When considered in light of the whisker-barrel system of rodents, the anatomy of the star -with its separate appendages, dense innervation, and high concentration of mechanoreceptors—raised the possibility of corresponding cortical modules that separately represent each appendage. To investigate this possibility, star-nosed mole neocortex was mapped using dense microelectrode penetrations followed by anatomical analysis of layer 4 cortex processed for CO [18]. In addition to providing the initial evidence for Eimer’s organ function described above, the results of electrophysiological recordings revealed the layout of the star appendages and other body parts in neocortical maps. Three separate representations of the star were identified in lateral cortex, corresponding to the expected location of the face representation in mammals generally (Fig. 2.7a). When the neocortex was flattened and sectioned tangentially, in the same manner that reveals barrels in rodents, each of the three maps of the star was visible as a pinwheel of CO dark stripes (Fig. 2.7c). Not only did this result represent an additional example of distinctive modules in primary somatosensory cortex reflecting the distribution of mechanoreceptors on the face, but it was also the first (and only) demonstration of multiple visible maps representing the same sensory surface. Subsequent investigation of these areas using neuroanatomical tracers [40] revealed that the maps are topographically interconnected to form a cortical processing network.
Cortical organization and behavior in the star-nosed mole. a Three maps of the contralateral star exist in somatosensory cortex (S1, S2, and S3). The S2 map of the star-nosed mole is comparatively large compared to most other mammals. The S3 representation is not found in other moles or shrews, and thus arose independently. b Half of the star under a scanning electron microscope with the 11 rays labeled. c The star representation can be seen in flattened cortex processed for cytochrome oxidase. Although ray 11 is small compared to the other rays (b) it has the largest representation in S1. This reflects its use as the somatosensory fovea. d Schematic of a star-nosed mole saccade used to move the 11th appendage over an object being explored. e Frames from high-speed video showing a star saccade relative to a small prey item (red circle). (Published with kind permission of © Kenneth Catania 2014)
Several features of this processing network differ substantially from the condition in rodents. For example, in addition to containing modules representing the individual rays, the secondary somatosensory cortex is much larger than would be predicted based on studies in rodents and most other mammals. S2 is usually much smaller than S1 and is characterized by large receptive fields. In contrast star-nosed mole S2 has proportions similar to S1 and is characterized by small receptive fields on both the star and other body parts. Interestingly, a large S2 is found in shrews as well (see previous section on water shrews) and may be a general feature of shrews and moles rather than a specialization in star-nosed moles .
Despite sharing some features in common with other moles and shrews (a large S2) comparisons across species indicate that the extra, third map of the nose in lateral and caudal cortex is unique to star-nosed moles. This means that it arose independently in star-nosed moles and was most likely not in the common ancestor to shrews and moles. This is a very interesting finding because the addition of cortical areas is often hypothesized to be one of the substrates for more complex sensory processing and behavioral abilities. In most cases, such comparisons involve distantly related species that differ substantially in brain size. But moles are closely related species of similar brain and body size. The obvious difference between star-nosed moles and other mole species is the elaboration of the sensory surface and corresponding behaviors (see next section). This suggests that star-nosed moles added a cortical area to handle large amounts of complex sensory information from the star, perhaps depending on parallel processing of some aspects of touch.
An additional interesting and obvious characteristic of the star-nosed mole’s somatosensory cortex is the overrepresentation of the 11th appendage. Despite the small size of this nasal ray and the relatively few Eimer’s organs on its surface, its representation takes up 25 % of the S1 star map (Fig. 2.7b, c). In addition, although the 11th appendage is more densely innervated then the rest of the star, only approximately 10 % of the afferents supplying the star serve this appendage. Its greater innervation density stems from its small size and few sensory organs compared to the number of innervating afferents, rather than the number of afferents in total. Put another way, the innervation density of ray 11 is high as a ratio of nerve fibers to sensory organs (or skin surface).
When afferent numbers supplying the star are compared to their representations in primary somatosensory cortex, the sizes of the ray representations are not proportional to the number of nerve fibers supplying each ray [41, 42]. This can be contrasted to the situation in rodents, where the size of each cortical barrel has been found to be proportional to the number of nerve fibers supplying each whisker on the face [43]. Investigation of star-nosed mole behavior provides an explanation for the dramatic mismatch between the anatomy of the star and its representation in cortex.
Somatosensory Fovea
Star-nosed moles use the star to explore their environment with a series of high-speed touches. They may touch 10–13 different places every second as they search for food and navigate their tunnels. As was the case for water shrews, detailed investigations required the use of high-speed video recordings [44]. These revealed the explanation for the differential magnification of nasal appendages in the cortical representation; star-nosed moles have a somatosensory fovea at the center of the star. The 11th, midline pair of appendages is used for detailed investigations of objects of interest (usually food). Most objects encountered as the mole searches its environment are first contacted by the large array of Eimer’s organs that cover rays 1–10, as these make up most of the surface area of the star. For detailed investigation, moles make sudden movements of the star to reposition the 11th rays on an object for multiple touches (Fig. 2.7d, e). These nose movements are remarkably similar in their form and time-course to saccadic eye movements in primates [44].
Underwater Sniffing
Star-nosed moles are semi-aquatic and occasionally dive for food, much like water shrews. This raised the question of whether tactile cues used for detecting prey with the star would be degraded in water as a result of its greater viscosity than air. It seemed possible, for example, that movements would be slower underwater. There was no obvious indication of different use of the star underwater for mechanosensory investigation, but a different and unanticipated behavior was observed. This was under-water sniffing —as already described for water shrews, but first discovered in star-nosed moles [28]. Star-nosed moles exhaled air bubbles over objects of interest and then re-inhaled the same air. As was the case for water shrews, they could follow a scent trail laid underwater. In the case of star-nosed moles, a stainless steel grid with large openings was placed over the scent trail at all times. This prevented contact of the star to the scent trail, but allowed for air to be exhaled through the grid and then re-inhaled with each sniff. When the coarse grid was replaced by a fine grid that did not admit air bubbles the moles’ performances deteriorated to chance levels.
Measurement of the timing of sniffs and the volume of air expired and re-inhaled showed that underwater sniffing is very similar to sniffing behavior exhibited on land by other small mammals. It is important to keep in mind that small mammal sniffing consists of repeated cycles of small expirations of air paired with small inspirations of air. In contrast to human sniffing, which generally consists of repeated short inspirations, small mammal sniffing on land is essentially the same as underwater sniffing in star-nosed moles and water shrews. That is, expiring air as a part of the sniffing process is not an innovation restricted to the aquatic medium. It is worth noting in this regard, that the terrestrial small mammals (e.g. short-tailed shrews) tested did not exhibit underwater sniffing when trained to retrieve food from a shallow enclosure [45]. Despite the close similarity between terrestrial sniffing and underwater sniffing, this does not appear to be a general feature of small mammal behavior , but rather a specialization of semiaquatic mammals.
That underwater sniffing happens at all is perhaps the most surprising conclusion from these studies. But this behavior also provides an obvious and very informative window into sniffing behavior; you can see the sniffs. Because each sniff is visible as an air bubble that emerges from the nostrils and is then re-inhaled, it is possible to clearly note the timing of sniffs relative to other behaviors using high-speed video. The conclusion from such observations is that sniffing is coordinated with touching. Underwater sniffs occur as the animal decelerates to make a touch and gather tactile information with the star (mole) or whiskers (water shrew) . This in turn is consistent with classical [46] and more recent [47–49] studies of sensory integration in rodents that suggest sniffing and touching are coordinated. The other obvious conclusion from this somewhat esoteric behavior of semi-aquatic moles and shrews is that coordinated sniffing and touching likely also occur as these animals explore their terrestrial environment; we just can’t see the sniffs. This possibility was confirmed by examining a related mole species (eastern moles, Scalopus aquaticus) that have a much less developed sense of touch compared to star-nosed moles.
Stereo Sniffing in Eastern Moles
The eastern mole (Scalopus aquaticus) does not have a star and is one of the only mole species that does not have Eimer’s organs in the epidermis of its snout. This is likely the result of foraging in a drier and more abrasive environment compared to most other moles. The outer epidermal layer (stratum corneum) on the snout of eastern moles is very thick compared to other species and in this condition could not support functional Eimer’s organs [32]. Because eastern moles are among the least specialized in this regard, they were chosen as subjects in preliminary studies of foraging efficiency in comparison to star-nosed moles. Star-nosed moles are among the fastest foragers [50] and it was suspected, based on their less elaborate somatosensory system, that eastern moles would be far less efficient.
The results were surprising and defied expectations. When presented with numerous small prey items (small earthworm segments), eastern moles moved almost directly from one to the next in rapid succession. This ability was so marked, that despite their tiny eyes hidden below the fur and an optic nerve so small that we have not been able to locate it upon dissection, experiments were repeated (with the same result) under infrared lighting to exclude all possibility that vision played a role [51].
High-speed video suggested that eastern moles were using a serial sniffing behavior to home in on earthworms. To test this possibility, an experimental chamber was designed that allowed for non-invasive sniff monitoring, using a pressure gauge. In this way, the sniff cycle could be correlated with video frames. These experiments showed that eastern moles sniff in coordination with nose movements (Fig. 2.8), as suggested by the behavior observed for star-nosed moles and water shrews foraging while submerged. However, the extremely accurate and rapid localization of olfactory stimuli by eastern moles (see [51] for movies) raised the possibility that bilateral (stereo) olfactory cues might be aiding in the localization process. Recent investigations in rats suggest that rodents also use this strategy [52] .
Eastern moles coordinate sniffs with touches as they explore their environment. (a) A schematic of the experimental chamber used to measure sniffing. The chamber was sealed so that a pressure gauge could monitor each sniff and be compared to simultaneous high-speed video recordings. (b) Example of one mole’s sniffing behavior in the chamber. The mole moved and then sniffed repeatedly. (c–e) Frames from high-speed video as a mole moves directly toward an olfactory stimulus (earthworm segment). Note that eastern moles are blind. From [51]. (Published with kind permission of © Kenneth Catania 2014)
To investigate this possibility in moles, a single nostril was blocked with a small silicone tube and moles were given the challenge of localizing a prey item under a number of different circumstances [51]. These experiments were inspired by similar investigations of auditory localization in barn owls, in which ear plugs were used to attenuate sound in one ear [3]. The presumption in moles was that use of stereo cues based on intensity differences between the two nostrils would be revealed by a search bias toward the side of the open nostril, as occurred for sound localization in barn owls based on intensity cues. For controls, moles were allowed to search with no tube or with an open tube that did not block the airflow through the nostril.
The results of these experiments were definitive (for example experiment see Fig. 2.9). In every paradigm, moles headed in the general direction of the food item but showed a bias in the search pattern away from the food and towards the open nostril as compared to moles that had no block or an open tube (Fig. 2.9a vs. b). This suggested an important influence of comparative intensity cues across the nostrils. It is perhaps not surprising that moles headed in the general direction of the food item, given that serial sampling cues were not disrupted by this manipulation and stereo cues from the nostrils would only be expected to provide information close to the stimulus where olfactory gradients are relatively steep. Despite their bias toward the open nostrils compared to controls when approaching the food item, (Fig. 2.9c, d) they were ultimately able to locate the food (earthworm segments). This suggests that serial sniffing cues, derived from the sequential nose movements and sniffs (typically considered to be the mechanism of olfactory localization) overcame the seemingly smaller effect of nostril block.
Eastern moles used stereo sniffing to located food items. (a) Under normal conditions, with both nostrils open, moles take a relatively straight path to the food item and search the food well first. (b) When one nostril was blocked moles erred by moving in the direction of the open nostril as they searched. Moles with a left block searched to the right of the item and those with a right block searched to the left. (c–d) Summary data from multiple trials, left nostril blocks in both these examples. The arrows mark the average cross point relative to the food item for each condition. Adapted from: [51]. (Published with kind permission of © Kenneth Catania 2014)
In order to further test the use of bilateral olfactory cues by eastern moles, the nostrils were “flipped” by inserting longer, open tubes into each nostril and crossing them so that the left nostril received air from the right side and right nostril received air from the left side. In this condition the results were even more striking. Moles usually moved towards the food item until close (presumably using serial sniffing cues) but then moved back and forth in apparent confusion as they tried to localize the stimulus (earthworm segment). This greater disruption compared to the nostril block is consistent with a stereo sniffing strategy, because there is a continual mis-match between the intensity cues and the stimulus location as the animal moves back and forth in front of the stimulus. In the crossed nostril condition the moles had great difficulty locating the prey item and often missed it completely. These experiments suggest that eastern moles combine both serial sniffing cues, based on sequential olfactory samples, with instantaneous comparisons across the nostrils during each sniff. At the same time, moles are making nose movements that provide somatosensory information for each location. The combined use of touch, serial olfactory sampling, and stereo nasal cues provides and impressive sensory armament and helps to explain the success of moles in exploiting diverse soil environments.
Conclusions
The results described above raise a number of interesting questions about the organization and evolution of mammalian brains and behavior related to active touch. Water shrews most certainly use active touch as they search—at high speed—for (often) elusive prey in the shallow waters of streams and ponds. They are primed to attack water movements that result from escaping prey and can respond in as little as 20 milliseconds. They can also use their whiskers to discriminate the details of immobile objects. These two abilities are impressive and in line with the expected characteristics of the water shrews’ prey, which include stationary insect larvae but also mobile animals such as fish or crayfish. Given their heavy dependence on whiskers, it was somewhat surprising that water shrews do not exhibit the cortical barrels that characterize the whisker representation in rodents and a number of other small mammals. It is even more surprising that water shrew brainstem trigeminal nuclei exhibit what appear to be the most prominent barrelettes described in mammals. At the same time, there is to date no evidence of thalamic barreloids in water shrews. Together these results raise the possibility that water shrews emphasize sub-cortical processing of touch to a comparatively greater extent than do rodents. It is possible that such an emphasis allows for faster responses by not requiring the longer path lengths to and from the neocortex. The implications of this possibility are interesting in light of shrews’ many similarities to ancestral, stem mammals, based on fossil evidence [22]. It is tempting to conjecture that the ancestral mammalian plan had a greater sub-cortical emphasis and that enhanced cortical processing was key to mammalian diversification. Of course an alternative possibility is that there are many ways to efficiently represent mechanoreceptors in cortex—as has been proposed for ocular dominance columns in primates [53]. Shrews and rodents might simply have different but equally efficient cortical circuitries for processing whisker inputs. In support of the latter possibility, many species with whiskers do not exhibit barrels (cats and dogs) and do not resemble ancestral mammals.
Star-nosed moles can be contrasted with water shrews by their complex set of interconnected somatosensory cortical modules. The star is represented in three different maps each characterized by a set of stripes that represent the nasal appendages. These results highlight the flexibility of module form in the mammalian neocortex. For example, it is often suggested that cortical barrels are a reflection of universal cortical subdivisions, the classical columns [54]. But, as Woolsey and Van der Loos pointed out in their original paper, the shape of a barrel reflects the distribution of mechanoreceptors around a whisker [12]. Therefore, topographic mapping could explain the cylindrical form of a barrel, rather than a fundamental constraint of cortical circuitry. In support of this latter possibility, the receptors of the star are laid out in elongated strips of tissue on each appendage. The cortex representing the rays is not organized in circular columns but rather mirrors the topography of the sensory sheet. Findings in primate somatosensory cortex show a similar reflection of fingers in the form of myelin dense modules in the area 3b finger representation (3b is the homologue of S1—[11]).
Another significant finding from star-nosed moles is the existence of a somatosensory fovea [41, 55]. The central 11th pair of rays are used for detailed investigations and moles make constant saccadic nose movements to position this area on objects of interest. The parallels with visual systems are obvious and, in addition to the behavior , include the preferential expansion of the fovea representation in cortex. The expanded cortical territory representing the tactile fovea is greater than would be predicted from afferent number alone and this too parallels the organization of (primate) visual systems [56]. The result emphasizes that common solutions arise in mammals for processing high-resolution sensory systems. We are intimately familiar with primate visual systems not only from the impressive literature resulting from years of study but also from personal, daily experience scrutinizing visual scenes with our foveas. However, it may come as some surprise that some bats have an auditory fovea. The mustached bat analyzes the 60 kHz frequency range and devotes a large part of its cochlea and cortex to analyzing this behaviorally important frequency. Most surprising is the parallel to saccades that can be drawn from bats’ Doppler shift compensation behavior. Bats constantly change outgoing call frequency to “move” the returning echoes into the range of the auditory fovea [57].
Finally, the discovery of underwater sniffing behavior in semi-aquatic water shrews and star-nosed moles stands as one of the more surprising findings in mammal sensory biology. It was thought impossible for mammals to use olfaction underwater [58–60] but moles and shrews have found a work-around. By exhaling and re-inhaling the same air as they sniff while submerged, these species can detect odorants. This behavior is fascinating by itself, but it also provided unexpected insights into the coordination of touch and smell in these species. This stems from the convenience of seeing sniffs, revealing that moles and shrews gather tactile and olfactory information in unison—supporting the generality of similar findings from laboratory rodents [47–49]. These revelations about olfactory abilities in semiaquatic moles and shrews suggested the solution to the impressive prey localization ability in terrestrial, eastern moles. This species coordinates its touches and sniffs in air, as is the case for underwater sniffing. But in addition to this serial sampling strategy, eastern moles add stereo olfactory cues to the analysis allowing for remarkably rapid and efficient movement toward olfactory stimuli.
References
Hodgkin AL, Huxley AF (1946) Potassium leakage from an active nerve fibre. Nature 158:376
Hodgkin AL, Huxley AF (1952) Movement of sodium and potassium ions during nervous activity. Cold Spring Harb Symp Quant Biol 17:43–52
Knudsen EI, Konishi M (1979) Mechanisms of sound localization in the Barn Owl (Tyto-Alba). J Comp Physiol 133(1):13–21
Knudsen EI, Konishi M (1980) Monaural occlusion shifts receptive-field locations of auditory midbrain units in the owl. J Neurophysiol 44(4):687–695
Carr CE, Boudreau RE (1991) Central projections of auditory nerve fibers in the barn owl. J Comp Neurol 314(2):306–318
Moller P (2002) Multimodal sensory integration in weakly electric fish: a behavioral account. J Physiol Paris 96(5–6):547–556
Heiligenberg W (1990) Electrosensory systems in fish. Synapse 6(2):196–206
Nottebohm F (2005) The neural basis of birdsong. PLoS Biol 3(5):e164
Nottebohm F, Liu WC (2010) The origins of vocal learning: new sounds, new circuits, new cells. Brain Lang 115(1):3–17
Doupe AJ (1993) A neural circuit specialized for vocal learning. Curr Opin Neurobiol 3(1):104–111
Kaas JH, What, If Anything (1983) Is Si - organization of 1st somatosensory area of cortex. Physiol Rev 63(1):206–231
Woolsey TA, Vanderlo H (1970) Structural organization of layer-Iv in somatosensory region (Si) of mouse cerebral cortex. Description of a cortical field composed of discrete cytoarchitectonic units. Brain Res 17(2):205
Feldman ML, Peters A (1974) A study of barrels and pyramidal dendritic clusters in the cerebral cortex. Brain Res 77(1):55–76
Van Der Loos H (1976) Barreloids in mouse somatosensory thalamus. Neurosci Lett 2(1):1–6
Ma PM, The barrelettes–architectonic vibrissal representations in the brainstem trigeminal complex of the mouse. I (1991) Normal structural organization. J Comp Neurol 309(2):161–199
Krubitzer L, Manger P, Pettigrew J, Calford M (1995) Organization of somatosensory cortex in monotremes: in search of the prototypical plan. J Comp Neurol 351(2):261–306
Catania KC, Northcutt RG, Kaas JH, Beck PD (1993) Nose stars and brain stripes. Nature 364(6437):493
Catania KC, Kaas JH (1995) Organization of the somatosensory cortex of the star-nosed mole. J Comp Neurol 351(4):549–567
Jain N, Catania KC, Kaas JH (1998) A histologically visible representation of the fingers and palm in primate area 3b and its immutability following long-term deafferentations. Cereb Cortex 8(3):227–236
Qi HX, Kaas JH (2004) Myelin stains reveal an anatomical framework for the representation of the digits in somatosensory area 3b of macaque monkeys. J Comp Neurol 477(2):172–187
Catania KC, Lyon DC, Mock OB, Kaas JH (1999) Cortical organization in shrews: evidence from five species. J Comp Neurol 410(1):55–72
Rowe TB, Macrini TE, Luo ZX (2011) Fossil evidence on origin of the mammalian brain. Science 332(6032):955–957
Gould E, Negus NC, Novick A (1964) Evidence for echolocation in shrews. J Exp Zool 156:19–37
Siemers BM, Schauermann G, Turni H, von Merten S (2009) Why do shrews twitter? Communication or simple echo-based orientation. Biol Lett 5(5):593–596
Catania KC, Hare JF, Campbell KL (2008) Water shrews detect movement, shape, and smell to find prey underwater. Proc Natl Acad Sci U S A 105(2):571–576
Leitch DB, Gauthier D, Sarko DK, Catania KC (2011) Chemoarchitecture of layer 4 isocortex in the american water shrew (sorex palustris). Brain Behav Evol 78(4):261–271
Anjum F, Turni H, Mulder PGH, van der Burg J, Brecht M (2006) Tactile guidance of prey capture in Etruscan shrews. Proc Natl Acad Sci U S A 103(44):16544–16549
Catania KC (2006) Olfaction: underwater ‘sniffing’ by semi-aquatic mammals. Nature 444(7122):1024–1025
Catania KC, Catania EH, Sawyer EK, Leitch DB (2013) Barrelettes without barrels in the American water shrew. PLoS One 8(6):e65975
Catania KC (2000) Epidermal sensory organs of moles, shrew moles, and desmans: a study of the family talpidae with comments on the function and evolution of Eimer’s organ. Brain Behav Evol 56(3):146–174
Eimer T (1871) Die schnauze des maulwurfes als tastwerkzeug. Arch Mikr Anat 7:181–191
Catania KC (1995) A comparison of the Eimer's organs of three North American moles: the hairy-tailed mole (Parascalops breweri), the star-nosed mole (Condylura cristata), and the eastern mole (Scalopus aquaticus). J Comp Neurol 354(1):150–160
Halata Z (1972) Innervation of hairless skin of the nose of mole. I Intraepidermal nerve endings. Z Zellforsch Mikrosk Anat 125(1):108–120
Shibanai S (1988) Ultrastructure of the Eimer's organs of the Japanese shrew mole, Urotrichus talpoides (Insectivora, Mammalia) and their changes following infraorbital axotomy. Anat Anz 165(2–3):105–129
Catania KC (1996) Ultrastructure of the Eimer's organ of the star-nosed mole. J Comp Neurol 365(3):343–354
Marasco PD, Catania KC (2007) Response properties of primary afferents supplying Eimer’s organ. J Exp Biol 210(Pt 5):765–780
Catania KC, Leitch DB, Gauthier D (2011) A star in the brainstem reveals the first step of cortical magnification. PLoS One 6(7):e22406
Catania KC, Kaas JH (1995) Organization of the Somatosensory Cortex of the Star-Nosed Mole. J Comp Neurol 351(4):549–567
Chubbuck JG (1966) Small motion biological stimulator. John Hopkins APL Tech Dig 5:18–23
Catania KC, Kaas JH (2001) Areal and callosal connections in the somatosensory cortex of the star-nosed mole. Somatosens Mot Res 18(4):303–311
Catania KC (1995) Magnified cortex in star-nosed moles. Nature 375(6531):453–454
Catania KC, Kaas JH (1997) Somatosensory fovea in the star-nosed mole: behavioral use of the star in relation to innervation patterns and cortical representation. J Comp Neurol 387(2):215–233
Lee KJ, Woolsey TA (1975) A proportional relationship between peripheral innervation density and cortical neuron number in the somatosensory system of the mouse. Brain Res 99(2):349–353
Catania KC, Remple FE (2004) Tactile foveation in the star-nosed mole. Brain Behav Evol 63(1):1–12
Catania KC (2009) Symposium overview: Underwater sniffing guides olfactory localization in semiaquatic mammals. Ann N Y Acad Sci 1170:407–412
Welker WI (1964) Analysis of sniffing of the albino rat. Behaviour 22(3/4):223–244
Deschenes M, Moore J, Kleinfeld D (2012) Sniffing and whisking in rodents. Curr Opin Neurobiol 22(2):243–250
Moore JD, Deschenes M, Furuta T, Huber D, Smear MC, Demers M et al (2013) Hierarchy of orofacial rhythms revealed through whisking and breathing. Nature 497(7448):205–210
Ranade S, Hangya B, Kepecs A (2013) Multiple modes of phase locking between sniffing and whisking during active exploration. J Neurosci 33(19):8250–8256
Catania KC, Remple FE (2005) Asymptotic prey profitability drives star-nosed moles to the foraging speed limit. Nature 433(7025):519–522
Catania KC (2013) Stereo and serial sniffing guide navigation to an odour source in a mammal. Nat Commun 4:1441
Rajan R, Clement JP, Bhalla US (2006) Rats smell in stereo. Science 311(5761):666–670
Horton JC, Adams DL (2005) The cortical column: a structure without a function. Philos Trans R Soc Lond B Biol Sci 360(1456):837–862
Mountcastle VB (1957) Modality and topographic properties of single neurons of cat’s somatic sensory cortex. J Neurophysiol 20(4):408–434
Catania KC (2001) Early development of a somatosensory fovea: a head start in the cortical space race? Nat Neurosci 4(4):353–354
Azzopardi P, Cowey A (1993) Preferential representation of the fovea in the primary visual-cortex. Nature 361(6414):719–721
Schnitzler H-U (1968) Die Ultraschall-Ortungslaute der Hufeisen-Fledermäuse (Chiroptera-Rhinolophidae) in verschiedenen Orientierungssituationen. Z Vergl Physiol 57(4):376–408
Stephan H, Baron G, Fons R (1984) Brains of soricidae. 2. Volume comparison of brain components. Z Zool Syst Evol 22(4):328–342
Estes JA (1989) Adaptations for aquatic living by carnivores. Cornell University Press, Ithaca
Repenning CA (1976) Adaptive evolution of sea lions and walruses. Syst Zool 25(4):375–390
Sarko DK, Leitch DB, Girard I, Sikes RS, Catania KC (2011) Organization of somatosensory cortex in the Northern grasshopper mouse (Onychomys leucogaster), a predatory rodent. J Comp Neurol 519(1):64–74
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Catania, K., Catania, E. (2015). Comparative Studies of Somatosensory Systems and Active Sensing. In: Krieger, P., Groh, A. (eds) Sensorimotor Integration in the Whisker System. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-2975-7_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2975-7_2
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-2974-0
Online ISBN: 978-1-4939-2975-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)