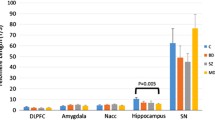
Abstract
Patients with major psychiatric disorders mainly schizophrenia (SZ) and major depression (MD) and with bipolar disorder (BD) are prone to diseases associated with aging. They reveal tendency to have shorter natural life span than the general population. Telomeres, composed of a repetitive DNA sequence and associated proteins, are located at both ends of each chromosome and play an important role in preserving information of cell genome. In somatic cells, telomeres progressively shorten with each cell division which is an indicator of aging and reduction of body’s ability to regenerate in response to various damaging factors. Chronic oxidative stress that accompanies metabolic changes in the course of SZ, MD, and BD can accelerate telomere shortening. Cross-sectional studies on telomere length in blood leukocytes proved their shortening in patients with these major psychiatric disorders versus matched controls. One study described the inverse association of leukocyte telomere length with circulating markers of oxidative stress in MD patients. No studies on this topic that involved SZ and BD groups have been executed so far. Postmortem brain samples revealed distinct features of oxidative stress while no shortening of telomere length was found in SZ, MD, and BD. No results of prospective longitudinal studies devoted to analyze the rate of telomere shortening and intensity of oxidative stress in the course of SZ, MD, and BD have been published so far. Therefore, whether these major psychiatric disorders can accelerate telomere erosion (via mechanisms dependent on oxidative stress) in blood leukocytes and other somatic cells remains to be investigated.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Bipolar Disorder
- Major Depression
- Telomere Length
- Chronic Fatigue Syndrome
- Coronary Heart Disease Patient
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Telomeres are located at both ends of each chromosome and are composed of a repetitive DNA sequence (TTAGGG repeats) and associated proteins (shelterin – protein complex). They have a very important vital function for cell biology by preserving the information in their genome. In particular, this consists in protection of the genome from nucleolytic degradation, unnecessary recombination, and interchromosomal fusion (assurance of proper positioning of chromosomes during replication), as well as repair (protection of the chromosome end from being recognized as DNA double-strand breaks by the DNA damage response mechanisms).
During each cell division, a small segment of telomeric DNA is lost leading to the reduction of telomere length (TL). Although this is a normal physiological process, it can result in cell senescence and/or apoptosis when TL reaches a critical limit (Donate and Blasco 2011; Shammas 2011; Martínez and Blasco 2010).
TL can be restored by the enzyme telomerase (ribonucleoprotein reverse transcriptase); however, the majority of cells and tissues (except of stem cells, germ cells, and regenerating tissues) has very low telomerase activities. TL could be maintained by the other alternative mechanism (ALT – alternative lengthening of telomeres) where telomeres use other telomeric DNA as a template for DNA synthesis (Neumann et al. 2013; Muntoni et al. 2009). However, these mechanisms are not efficient at maintaining TL, and consequently telomere erosion occurs with age in numerous somatic tissues. Thus, TL or its shortening could be recognized as a predictive factor of the life span of cells and organisms and the age-related reduction of body’s ability to regenerate in response to action of damaging factors (Donate and Blasco 2011; Shammas 2011) and even individual death (Cawthon et al. 2003).
Numerous factors associated with lifestyle, nutrition, and coexistent diseases can predispose to accelerate TL shortening and be responsible for negative effect on individual subject life span and state of health. Among them are lack of exercise, obesity, cigarette smoking, alcohol abuse, exposure to air pollutants, cardiovascular diseases, and diabetes, and these issues have recently been largely reviewed and discussed elsewhere (Donate and Blasco 2011; Paul 2011; Price et al. 2013). On the other hand, plasma levels of vitamin A and D and habitual tea drinking (ingestion of tea polyphenols) were positively associated with TL (Paul 2011). Since venous blood is more easily accessible than most other tissues, majority of these data were derived from cross-sectional studies focused on TL of peripheral blood leukocytes.
Chronic oxidative stress and inflammation are implicated in speeding up the aging process. Numerous studies with cell cultures and animal models proved faster TL shortening under conditions of oxidative stress. Due to its high content of guanines and inefficient repair of single-strand breaks, telomeric DNA is highly sensitive to damage by reactive oxygen species (Petersen et al. 1998; Oikawa et al. 2001; Oikawa and Kawanishi 1999). Moreover, increased levels of pro-inflammatory cytokines, especially tumor necrosis factor alpha (TNF-α), that are frequently accompanied with oxidative stress can decrease the telomerase activity (Beyne-Rauzy et al. 2004, 2005) and oxidative damage to the bases of telomeric DNA accumulated over the life span of cells (Kawanishi and Oikawa 2004). Therefore, TL could be a biomarker of chronic oxidative stress (Houben et al. 2008).
Systemic and cerebral oxidative stress has been demonstrated in the major psychiatric disorders like schizophrenia (SZ), major depression (MD), and bipolar disorder (BD). Although these issues are discussed elsewhere in this book, it is necessary to mention that patients suffering from SZ, MD, and BD had elevated circulating levels of products deriving from oxidative damage to lipids, proteins, and DNA and decreased plasma antioxidant defense (Kim and Andreazza 2012; Ng et al. 2008; Maes et al. 2011; Yao and Reddy 2011; Bitanihirwe and Woo 2011), and analysis of postmortem obtained brain specimens revealed increased content of oxidative stress markers (Gawryluk et al. 2011a; Michel et al. 2011a; Che et al. 2010; Andreazza et al. 2010; Wang et al. 2009).
Moreover, these groups of patients are prone to diseases associated with aging (e.g., diabetes, cardiovascular diseases) and reveal tendency to have shorter natural life span than the general population. Therefore, researches executed a dozen or so clinical studies (in the vast majority cross-sectional) on blood leukocyte TL in these major psychiatric disorders during the last few years. This chapter will review the results of these studies with special attention to intensity of systemic and brain oxidative stress.
2 Telomere Length in Blood Leukocytes and Brain Samples of Patients with Schizophrenia
Table 1 summarizes the results of cross-sectional studies on TL in patients with SZ. These studies mostly compared TL in DNA isolated from blood leukocytes between SZ patients and matched controls (Kao et al. 2008; Yu et al. 2008; Fernandez-Egea et al. 2009; Mansour et al. 2011). In one of them the additional group of patients with type I bipolar disorder (BD I) was investigated (Mansour et al. 2011). Only one report was based on postmortem brain samples (gray matter of cerebellum) obtained from patients suffering from SZ, BD, MD, and controls where TL was analyzed by quantitative PCR (Zhang et al. 2010). Concerning the TL in blood leukocytes, two reports revealed telomere shortening in SZ subjects (Kao et al. 2008; Fernandez-Egea et al. 2009). One study found this difference limited to SZ patients who were poor responders to the treatment versus healthy matched controls. However, good responders did not differ from controls (Yu et al. 2008).
On the other hand, Mansour et al. on the basis of investigation of relatively large group of subjects (Table 1) did not find any significant differences in the leukocyte TL between SZ group, BD I patients, and controls (Mansour et al. 2011). In agreement with this report, no difference of TL in the gray matter of cerebellum was noted in SZ, BD, and MD patients compared to controls. Similarly, no differences in brain TL were found between these three groups of psychiatric disorders (Zhang et al. 2010).
In conclusion, studies on the TL in blood leukocytes of SZ patients are not fully conclusive. More studies involving larger patients groups and matched controls are required. Longitudinal studies on telomere shortening in the course of SZ along with the effect of antipsychotic treatment are necessary.
2.1 Discordance Between Telomere Length and Oxidative Stress in Brain Tissue of Patients with Schizophrenia and Other Major Psychiatric Disorders (Bipolar Disorder, Major Depression)
The aforementioned results on TL in brain samples are more than somewhat surprising since numerous studies proved occurrence of oxidative stress in some regions of brain tissue (postmortem samples) in patients with SZ, BD, and MD:
-
(a)
Samples of prefrontal cortex from patients with SZ and BD revealed downregulation of uncoupling protein 2 (involved in controlling the mitochondrial production of ROS) mRNA levels (Gigante et al. 2011).
-
(b)
Samples of prefrontal cortex of patients with SZ, MD, and BD had decreased concentrations of reduced, oxidized, and total glutathione (Gawryluk et al. 2011a). Moreover, the levels of glutathione peroxidase and the mu isoenzyme of glutathione S-transferase (GST mu) were decreased in MD and SZ brain samples evaluated with immunoblotting technique (Gawryluk et al. 2011a, b).
-
(c)
There was an increased activity of prooxidant enzyme xanthine oxidase in the brain tissue (samples of thalamus and putamen) of patients with MD (Michel et al. 2011a). However, decreased activity was noted in SZ brain specimens (Michel et al. 2011b).
-
(d)
Samples of hippocampus of patients with SZ, BD, and MD had increased content of 8-hydroxy-guanosine, a marker of RNA oxidative damage (Che et al. 2010).
-
(e)
Samples of prefrontal cortex of BD patients had increased levels of oxidized proteins (carbonylated proteins) and 3-nitrotyrozine. Elevated 3-nitrotyrosine was also observed in SZ brain tissue (Andreazza et al. 2010).
-
(f)
Anterior cingulate brain sections from BD and SZ subjects (but not from MD patients) had elevated content of 4-hydroxynonenal, a major product of lipid peroxidation (Wang et al. 2009).
Since oxidative stress predisposes to increased telomere erosion, one may expect the shortening of TL in brain specimens of patients with SZ and other major psychiatric disorders (MD, BD). However, brain specimens from patients with SZ, BD, and MD did not reveal telomere shortening in comparison to control samples (Zhang et al. 2010). Similar results (no significant difference in TL) were noted by other studies with postmortem brain specimens (dorsolateral prefrontal cortex) obtained from depressive patients and matched controls (Teyssier et al. 2011). Moreover, there was no difference in expression of genes involved in the oxidative-stress response and repair between brain samples from both studied groups (Teyssier et al. 2011) (Table 3). These findings suggest that telomere erosion did not occur in brain cortex despite the presence of distinct features of brain oxidative stress.
On the other hand, it cannot be excluded that telomere damage and oxidative stress occur in the brain; simply these phenomena could be limited to other brain regions than those studied so far.
Therefore, further studies analyzing additional brain regions in respect of telomere dysfunction, DNA damage, and intensity of oxidative stress in the course of SZ as well as other major psychiatric disorders are necessary.
3 Telomere Length in Blood Leukocytes and Brain Samples of Patients with Major Depression and Bipolar Disorder
Much more cross-sectional studies have been executed on the telomere shortening in MD and BD patients. These involved larger patients groups and were not only focused on comparison of the TL in DNA extracted from blood leukocytes between patients with mood disorders and matched controls (Tables 2 and 3) but also looked for an association between TL and various clinical variables such us disease severity and duration, treatment effect, intensity of oxidative stress, inflammatory response and perceived stress, and the monoamine oxidase A (MAOA) promoter and apolipoprotein E2 (ApoE2) polymorphism (Lung et al. 2007; Hartmann et al. 2010; Wikgren et al. 2012; Wolkowitz et al. 2011). Out of 7 reports that compared the leukocyte TL in patients with mood disorders (MD and BD) and matched controls, two described telomere shortening in BD patients (Simon et al. 2006; Elvsashagen et al. 2011) and six in MD patients (Lung et al. 2007; Hartmann et al. 2010; Wikgren et al. 2012; Simon et al. 2006; Garcia-Rizo et al. 2012), respectively. Only one study did not report significant shortening of TL in patients with MD (Wolkowitz et al. 2011). However, patient subgroups with cumulative duration of depression ≥9.2 years had significantly shorter telomeres than the control subjects (Wolkowitz et al. 2011). TL was inversely correlated with the ratio of plasma F2-isoprostanes (biomarkers of lipid peroxidation) to vitamin C and positively with the circulating vitamin C levels in MD patients (Wolkowitz et al. 2011). TL was also inversely associated with the stress measured with the self-report questionnaire (Wikgren et al. 2012).
However, TL in MD did not correlate with MAOA promoter and ApoE2 polymorphism (Lung et al. 2007) and neither with disease severity, duration of illness, and number of hospital stays (Hartmann et al. 2010) nor with lymphocyte count (Garcia-Rizo et al. 2012). On the other hand, the load of short telomeres in DNA isolated from the blood mononuclear cells positively correlated with the high number of previous depressive episodes in bipolar disorder type II (BD II) patients (Elvsashagen et al. 2011).
These results obtained with well-defined MD and BD patients groups clearly show the occurrence of some processes that are responsible for the telomere shortening in blood DNA of these patients.
Oxidative stress could be responsible for enhanced telomere erosion in the course of MD. However, only one study was devoted to the analysis of the association between TL and markers of oxidative stress in MD (Wolkowitz et al. 2011). Although this study concluded to a positive correlation between telomere erosion and intensity of oxidative stress, the sample size (n = 18) was too low to solve this issue conclusively (Table 3).
Surprisingly, two large cross-sectional studies did not confirm negative effect of depression on TL (Shaffer et al. 2012; Huzen et al. 2010). In a population-based survey that involved 2,225 apparently healthy participants, no association between the leukocyte TL and depressive symptoms as well as TL and probable depressive disorder was found (Shaffer et al. 2012). Similarly, analysis of TL and psychological well-being in 890 patients with chronic heart failure did not reveal any significant association with the depressive symptoms (Huzen et al. 2010). However, no subject had syndromal MD in these studies. Therefore, the intensity and cumulative duration of depressive symptoms could be too low to exert negative effect on the leukocyte TL.
Only one study was executed with postmortem brain samples (dorsolateral prefrontal cortex) of MD patients and matched controls. Neither TL nor the expression of genes involved in the antioxidant defense and repair differed between MD group and controls (Teyssier et al. 2011) (Table 3). These results are analogous to those obtained with postmortem brain samples of patients with SZ discussed in previous subsection.
4 Prospective Studies on Telomere Length in Patients with Mood Disorders
Scanty data exist on association between the TL and the further development of mood disorders and on effect of the current mood disorders on the TL shortening over subsequent time. Moreover, studies reporting these associations involved observation of subjects that did not suffer from MD (and other mood disorders) as the underlying disease (Table 4).
In one study patients suffering from coronary heart disease (CHD) were screened for the presence of coexisting MD, and the TL in their blood leukocytes was measured at baseline and after 5 years (Hoen et al. 2011). Although at baseline CHD patients with current MD had shorter TL than those without MD, the MD group did not reveal a higher rate of telomere shortening over 5-year follow-up. These facts suggest that presence of MD cannot be used as a predictive factor of telomere shortening in patients with CHD (Hoen et al. 2011). As underlined by the authors, this study had some limitations, and the two most important, in my opinion, are the following: groups may differ in the CHD severity, and leukocyte telomerase activity may affect telomere shortening and be responsible for negative results of this study (Hoen et al. 2011). Therefore, it is open to question whether the coexistent MD can accelerate the telomere erosion in CHD patients.
A second study on association between the rate of telomere shortening and the poor mental well-being and poor self-rated health in community-dwelling elderly men also revealed negative results (Rius-Ottenheim et al. 2012) (Table 4). However, a third study presented partially positive results. This study involved a group of female rape survivors investigated within 2 weeks from the rape incident and after 3-month follow-up. TL in blood DNA measured at baseline was associated with the development of posttraumatic stress disorder. Victims presenting with posttraumatic stress disorder had shorter TL than those free of this abnormality. On the other hand, TL did not associate with the presence of trauma-induced MD either at baseline or after 3-month follow-up (Malan et al. 2011).
Description of these studies and their results clearly shows that there is a great need of prospective longitudinal studies involving patients suffering from MD, SZ, or BD and matched controls to evaluate the effect of major psychiatric disorders on the rate of telomere shortening.
5 Telomerase Activity and Expression of Genes Involved in DNA Repair in Patients with Schizophrenia and Major Depression
Since the TL in blood leukocytes of patients with SZ and MD was shorter in comparison to the matched healthy controls, one may assume that this may be related to decreased telomerase activity in these cells. Moreover, it cannot be excluded that the rise in telomerase activity may counteract this negative process, perhaps, related to the systemic oxidative stress.
This was investigated in the group of patients with MD and SZ and resulted in opposite outcomes (Table 5). Telomerase activity in subjects with SZ did not differ significantly from that found in the group of their unaffected relatives (Porton et al. 2008) and even was lower when compared to the reference group composed of unaffected relatives and unrelated controls (Porton et al. 2008). This suggests the reduction of telomerase activity in blood lymphocytes in SZ. On the other hand, no correlation between the TL and telomerase activity in lymphocytes of SZ patients was noted (Porton et al. 2008). Similarly, TL did not correlate with the telomerase activity in other immune cells (T lymphocytes, blood mononuclear cells) isolated from healthy subjects (Pan et al. 1997; Iwama et al. 1998). Therefore, it seems that suppression of telomerase activity cannot be a culprit of decreased TL in leukocytes of SZ patients.
In contrast to these results, medication-free MD patients had elevated telomerase activity in blood mononuclear cells in comparison to the unaffected controls (Wolkowitz et al. 2012). No significant correlation was noted between the TL and telomerase activity in these patients likewise to SZ group. Moreover, telomerase activity in mononuclear cells did not correlate with various markers of oxidative stress (plasma concentrations of F2-isoprostanes, 8-hydroxydeoxyguanosine, and vitamin C) and inflammation (interleukin-6 and C-reactive protein levels) both in MD subjects and controls (Wolkowitz et al. 2012). Therefore, it is difficult to judge whether the rise of telomerase activity is a defensive mechanism against the oxidative stress observed in the course of MD.
Another study that involved female MD patients and matched controls studied the expression of the set of gene encoding products that are implicated in and being markers of processes of the telomere dysfunction and repair (STMN1, encoding stathmin; TERT, encoding the catalytic subunit of the telomerase), the aging and senescence (p16ink4a encoded by the CDKN2A locus), the oxidative stress and DNA repair (OGG1 – encoding 8-oxoguanine-DNA glycosylase1), the response to anxiety and psychogenic stress (FOS gene, DUSP-1 gene), and the inflammatory response (Il-6 gene) in blood leukocytes. Although TL did not differ between MD women and controls, there was significant overexpression of OGG1, P16ink4a, and STMN1 genes in the MD group. These results suggest the occurrence of DNA damage and the telomere dysfunction probably due to oxidative stress in leukocytes of female MD patients (51).
While the results seem interesting, the major limitation of these studies is the low number of analyzed patients. Therefore, they are not conclusive and require confirmation in further studies involving larger groups of patients.
6 Concluding Remarks
Patients with MD and BD revealed shortened TL in blood leukocytes as evaluated in cross-sectional studies. Results obtained with the groups of SZ patients are not fully conclusive but also suggest telomere shortening in this disease. Numerous dietary (intake of plant polyphenols and vitamins), demographic, socioeconomic factors, and pathological conditions can affect the rate of telomere erosion in leukocytes. Therefore, it is very difficult to select homogeneous patient group (with respect to comorbidities and lifestyle) and precisely matched controls. Although the statistical analyses of these studies included the adjustment for some confounding factors, it is not easy to completely eliminate the risk of bias especially when the size of patient group is low.
In the case of blood leukocytes, measurement of the TL reflects their replicative history.
Thus, any factor (e.g., infection, inflammation) that can enhance these cells’ turnover will lead to the TL shortening and induce bias. This implicates that apart from current confounding factors, also past confounding factors can affect the results of the TL determination in blood leukocytes.
In light of this, previous reports showing prenatal exposure to influenza virus as a risk factor for adult SZ (Limosin et al. 2003; Izumoto et al. 1999), and the positive association of psychological stress with the number of upper respiratory tract infections in the subjects with chronic fatigue syndrome (Faulkner and Smith 2008), seem interesting.
It should be pointed out that early life stress (childhood adversity) is negatively associated with the TL in adult life (Price et al. 2013; Kiecolt-Glaser et al. 2011; Kananen et al. 2010), which also complicates interpretation of the results of cross-sectional studies.
There are no conclusive data on the association between intensity of systemic oxidative stress and the telomere shortening in blood leukocytes of patients with these three major psychiatric disorders. Moreover, postmortem brain samples of these patients (SZ, MD, BD) did not reveal any TL shortening, although they had elevated markers of oxidative stress. This dissonance between the TL of blood leukocytes and brain tissue is somewhat surprising since in other diseases (e.g., diabetes, autoimmune diseases, cardiovascular diseases, stroke) the reduced leukocyte TL correlated with the telomere shortening in target organs and tissues (Price et al. 2013).
Bearing this in mind, only well-planned longitudinal studies with monitoring of the blood leukocyte TL and the intensity of systemic oxidative stress will definitely solve the question whether SZ, MD, and BD are associated with accelerated telomere shortening and whether the oxidative stress belongs to the main factors responsible for this process.
Abbreviations
- ALT:
-
Alternative lengthening of telomeres
- ApoE2 :
-
Apolipoprotein E2
- BD:
-
Bipolar disorder
- BD II:
-
Bipolar disorder type II
- BD I:
-
Bipolar disorder type I
- BMI:
-
Body mass index
- CHD:
-
Coronary heart disease
- COPD:
-
Chronic obstructive pulmonary disease
- DST:
-
Dexamethasone
- ECT:
-
Electroconvulsive therapy
- ELISA:
-
Enzyme-linked immunosorbent assay
- GST mu:
-
Glutathione S-transferase
- HAM-A:
-
Hamilton anxiety score
- HAM-D:
-
Hamilton depression score
- MAOA:
-
Monoamine oxidase A
- MD:
-
Major depression
- NYHA:
-
New York Heart Association
- PCR:
-
Polymerase chain reaction
- PCR-RFLP:
-
PCR – restriction fragment length polymorphism
- PTSD:
-
Posttraumatic stress disorder
- ROS:
-
Reactive oxygen species
- STMN1:
-
Encoding stathmin
- SZ:
-
Schizophrenia
- TERT:
-
Encoding the catalytic subunit of the telomerase
- TL:
-
Telomere length
References
Andreazza AC, Shao L, Wang JF, Young LT (2010) Mitochondrial complex I activity and oxidative damage to mitochondrial proteins in the prefrontal cortex of patients with bipolar disorder. Arch Gen Psychiatry 67:360–368
Beyne-Rauzy O, Recher C, Dastugue N, Demur C, Pottier G, Laurent G, Sabatier L, Mansat-De Mas V (2004) Tumor necrosis factor alpha induces senescence and chromosomal instability in human leukemic cells. Oncogene 23:7507–7516
Beyne-Rauzy O, Prade-Houdellier N, Demur C, Recher C, Ayel J, Laurent G, Mansat-De Mas V (2005) Tumor necrosis factor-alpha inhibits hTERT gene expression in human myeloid normal and leukemic cells. Blood 106:3200–3205
Bitanihirwe BK, Woo TU (2011) Oxidative stress in schizophrenia: an integrated approach. Neurosci Biobehav Rev 35:878–893
Cawthon RM, Smith KR, O’Brien E, Sivatchenko A, Kerber RA (2003) Association between telomere length in blood and mortality in people aged 60 years or older. Lancet 361:393–395
Che Y, Wang JF, Shao L, Young T (2010) Oxidative damage to RNA but not DNA in the hippocampus of patients with major mental illness. J Psychiatry Neurosci 35:296–302
Donate LE, Blasco MA (2011) Telomeres in cancer and ageing. Philos Trans R Soc Lond B Biol Sci 366:76–84
Elvsashagen T, Vera E, Boen E, Bratlie J, Andreassen OA, Josefsen D, Malt UF, Blasco MA, Boye B (2011) The load of short telomeres is increased and associated with lifetime number of depressive episodes in bipolar II disorder. J Affect Disord 135:43–50
Faulkner S, Smith A (2008) A longitudinal study of the relationship between psychological distress and recurrence of upper respiratory tract infections in chronic fatigue syndrome. Br J Health Psychol 13:177–186
Fernandez-Egea E, Bernardo M, Heaphy CM, Griffith JK, Parellada E, Esmatjes E, Conget I, Nguyen L, George V, Stöppler H, Kirkpatrick B (2009) Telomere length and pulse pressure in newly diagnosed, antipsychotic-naïve patients with nonaffective psychosis. Schizophr Bull 35:437–442
Garcia-Rizo C, Fernandez-Egea E, Miller BJ, Oliveira C, Justicia A, Griffith JK, Heaphy CM, Bernardo M, Kirkpatrick B (2012) Abnormal glucose tolerance, white blood cell count, and telomere length in newly diagnosed, antidepressant-naïve patients with depression. Brain Behav Immun. doi:10.1016/j.bbi.2012.11.009
Gawryluk JW, Wang JF, Andreazza AC, Shao L, Young LT (2011a) Decreased levels of glutathione, the major brain antioxidant, in post-mortem prefrontal cortex from patients with psychiatric disorders. Int J Neuropsychopharmacol 14:123–130
Gawryluk JW, Wang JF, Andreazza AC, Shao L, Yatham LN, Young LT (2011b) Prefrontal cortex glutathione S-transferase levels in patients with bipolar disorder, major depression and schizophrenia. Int J Neuropsychopharmacol 14:1069–1074
Gigante AD, Andreazza AC, Lafer B, Yatham LN, Beasley CL, Young LT (2011) Decreased mRNA expression of uncoupling protein 2, a mitochondrial proton transporter, in post-mortem prefrontal cortex from patients with bipolar disorder and schizophrenia. Neurosci Lett 505:47–51
Hartmann N, Boehner M, Groenen F, Kalb R (2010) Telomere length of patients with major depression is shortened but independent from therapy and severity of the disease. Depress Anxiety 27:1111–1116
Hoen PW, de Jonge P, Na BY, Farzaneh-Far R, Epel E, Lin J, Blackburn E, Whooley MA (2011) Depression and leukocyte telomere length in patients with coronary heart disease: data from the heart and soul study. Psychosom Med 73:541–547
Houben JM, Moonen HJ, van Schooten FJ, Hageman GJ (2008) Telomere length assessment: biomarker of chronic oxidative stress? Free Radic Biol Med 44:235–246
Huzen J, van der Harst P, de Boer RA, Lesman-Leegte I, Voors AA, van Gilst WH, Samani NJ, Jaarsma T, van Veldhuisen DJ (2010) Telomere length and psychological well-being in patients with chronic heart failure. Age Ageing 39:223–227
Iwama H, Ohyashiki K, Ohyashiki JH, Hayashi S, Yahata N, Ando K, Toyama K, Hoshika A, Takasaki M, Mori M, Shay JW (1998) Telomeric length and telomerase activity vary with age in peripheral blood cells obtained from normal individuals. Hum Genet 102:397–402
Izumoto Y, Inoue S, Yasuda N (1999) Schizophrenia and the influenza epidemics of 1957 in Japan. Biol Psychiatry 46:119–124
Kananen L, Surakka I, Pirkola S, Suvisaari J, Lönnqvist J, Peltonen L, Ripatti S, Hovatta I (2010) Childhood adversities are associated with shorter telomere length at adult age both in individuals with an anxiety disorder and controls. PLoS One 5:e10826
Kao HT, Cawthon RM, Delisi LE, Bertisch HC, Ji F, Gordon D, Li P, Benedict MM, Greenberg WM, Porton B (2008) Rapid telomere erosion in schizophrenia. Mol Psychiatry 13:118–119
Kawanishi S, Oikawa S (2004) Mechanism of telomere shortening by oxidative stress. Ann N Y Acad Sci 1019:278–284
Kiecolt-Glaser JK, Gouin JP, Weng NP, Malarkey WB, Beversdorf DQ, Glaser R (2011) Childhood adversity heightens the impact of later-life caregiving stress on telomere length and inflammation. Psychosom Med 73:16–22
Kim HK, Andreazza AC (2012) The relationship between oxidative stress and post-translational modification of the dopamine transporter in bipolar disorder. Expert Rev Neurother 12:849–859
Limosin F, Rouillon F, Payan C, Cohen JM, Strub N (2003) Prenatal exposure to influenza as a risk factor for adult schizophrenia. Acta Psychiatr Scand 107:331–335
Lung FW, Chen NC, Shu BC (2007) Genetic pathway of major depressive disorder in shortening telomeric length. Psychiatr Genet 17:195–199
Maes M, Galecki P, Chang YS, Berk M (2011) A review on the oxidative and nitrosative stress (O&NS) pathways in major depression and their possible contribution to the (neuro)degenerative processes in that illness. Prog Neuropsychopharmacol Biol Psychiatry 35:676–692
Malan S, Hemmings S, Kidd M, Martin L, Seedat S (2011) Investigation of telomere length and psychological stress in rape victims. Depress Anxiety 28:1081–1085
Mansour H, Chowdari K, Fathi W, Elassy M, Ibrahim I, Wood J, Bamne M, Tobar S, Yassin A, Salah H, Elsayed H, Eissa A, El-Boraie H, Ibrahim NE, Elsayed M, El-Bahaei W, Gomaa Z, El-Chennawi F, Nimgaonkar VL (2011) Does telomere length mediate associations between inbreeding and increased risk for bipolar I disorder and schizophrenia? Psychiatry Res 188:129–132
Martínez P, Blasco MA (2010) Role of shelterin in cancer and aging. Aging Cell 9:653–666
Michel TM, Camara S, Tatschner T, Frangou S, Sheldrick AJ, Riederer P, Grünblatt E (2011a) Increased xanthine oxidase in the thalamus and putamen in depression. World J Biol Psychiatry 11:314–320
Michel TM, Sheldrick AJ, Camara S, Grünblatt E, Schneider F, Riederer P (2011b) Alteration of the pro-oxidant xanthine oxidase (XO) in the thalamus and occipital cortex of patients with schizophrenia. World J Biol Psychiatry 12:588–597
Muntoni A, Neumann AA, Hills M, Reddel RR (2009) Telomere elongation involves intra-molecular DNA replication in cells utilizing alternative lengthening of telomeres. Hum Mol Genet 18:1017–1027
Neumann AA, Watson CM, Noble JR, Pickett HA, Tam PP, Reddel RR (2013) Alternative lengthening of telomeres in normal mammalian somatic cells. Genes Dev 27:18–23
Ng F, Berk M, Dean O, Bush AI (2008) Oxidative stress in psychiatric disorders: evidence base and therapeutic implications. Int J Neuropsychopharmacol 11:851–876
Oikawa S, Kawanishi S (1999) Site-specific DNA damage at GGG sequence by oxidative stress may accelerate telomere shortening. FEBS Lett 453:365–368
Oikawa S, Tada-Oikawa S, Kawanishi S (2001) Site-specific DNA damage at the GGG sequence by UVA involves acceleration of telomere shortening. Biochemistry 40:4763–4768
Pan C, Xue BH, Ellis TM, Peace DJ, Diaz MO (1997) Changes in telomerase activity and telomere length during human T lymphocyte senescence. Exp Cell Res 231:346–353
Paul L (2011) Diet, nutrition and telomere length. J Nutr Biochem 22:895–901
Petersen S, Saretzki G, von Zglinicki T (1998) Preferential accumulation of single-stranded regions in telomeres of human fibroblasts. Exp Cell Res 239:152–160
Porton B, Delisi LE, Bertisch HC, Ji F, Gordon D, Li P, Benedict MM, Greenberg WM, Kao HT (2008) Telomerase levels in schizophrenia: a preliminary study. Schizophr Res 106:242–247
Price LH, Kao HT, Burgers DE, Carpenter LL, Tyrka AR (2013) Telomeres and early-life stress: an overview. Biol Psychiatry 73:15–23
Rius-Ottenheim N, Houben JM, Kromhout D, Kafatos A, van der Mast RC, Zitman FG, Geleijnse JM, Hageman GJ, Giltay EJ (2012) Telomere length and mental well-being in elderly men from the Netherlands and Greece. Behav Genet 42:278–286
Shaffer JA, Epel E, Kang MS, Ye S, Schwartz JE, Davidson KW, Kirkland S, Honig LS, Shimbo D (2012) Depressive symptoms are not associated with leukocyte telomere length: findings from the Nova Scotia Health Survey (NSHS95), a population-based study. PLoS One 7:e48318
Shammas MA (2011) Telomeres, lifestyle, cancer, and aging. Curr Opin Clin Nutr Metab Care 14:28–34
Simon NM, Smoller JW, McNamara KL, Maser RS, Zalta AK, Pollack MH, Nierenberg AA, Fava M, Wong KK (2006) Telomere shortening and mood disorders: preliminary support for a chronic stress model of accelerated aging. Biol Psychiatry 60:432–435
Teyssier JR, Ragot S, Chauvet-Gelinier JC, Trojak B, Bonin B (2011) Expression of oxidative stress-response genes is not activated in the prefrontal cortex of patients with depressive disorder. Psychiatry Res 186:244–247
Teyssier JR, Chauvet-Gelinier JC, Ragot S, Bonin B (2012) Up-regulation of leucocytes genes implicated in telomere dysfunction and cellular senescence correlates with depression and anxiety severity scores. PLoS One 7:e49677
Wang JF, Shao L, Sun X, Young LT (2009) Increased oxidative stress in the anterior cingulate cortex of subjects with bipolar disorder and schizophrenia. Bipolar Disord 11:523–529
Wikgren M, Maripuu M, Karlsson T, Nordfjall K, Bergdahl J, Hultdin J, Del-Favero J, Roos G, Nilsson LG, Adolfsson R, Norrback KF (2012) Short telomeres in depression and the general population are associated with a hypocortisolemic state. Biol Psychiatry 71:294–300
Wolkowitz OM, Mellon SH, Epel ES, Lin J, Dhabhar FS, Su Y, Reus VI, Rosser R, Burke HM, Kupferman E, Compagnone M, Nelson JC, Blackburn EH (2011) Leukocyte telomere length in major depression: correlations with chronicity, inflammation and oxidative stress–preliminary findings. PLoS One 6:e17837
Wolkowitz OM, Mellon SH, Epel ES, Lin J, Reus VI, Rosser R, Burke H, Compagnone M, Nelson JC, Dhabhar FS, Blackburn EH (2012) Resting leukocyte telomerase activity is elevated in major depression and predicts treatment response. Mol Psychiatry 17:164–172
Yao JK, Reddy R (2011) Oxidative stress in schizophrenia: pathogenetic and therapeutic implications. Antioxid Redox Signal 15:1999–2002
Yu WY, Chang HW, Lin CH, Cho CL (2008) Short telomeres in patients with chronic schizophrenia who show a poor response to treatment. J Psychiatry Neurosci 33:244–247
Zhang D, Cheng L, Craig DW, Redman M, Liu C (2010) Cerebellar telomere length and psychiatric disorders. Behav Genet 40:250–254
Acknowledgment
This work was partially supported by a grant from the EU Regional Development Fund through the Polish Innovation Economy Operational Program, contract N. UDAPOIG. 01.03.01-10-109/08-00.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this chapter
Cite this chapter
Nowak, D. (2015). Telomere Length in Major Psychiatric Disorders: Is There Any Relationship Between Telomere Length and Oxidative Stress?. In: Dietrich-Muszalska, A., Chauhan, V., Grignon, S. (eds) Studies on Psychiatric Disorders. Oxidative Stress in Applied Basic Research and Clinical Practice. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-0440-2_21
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0440-2_21
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-0439-6
Online ISBN: 978-1-4939-0440-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)