Abstract
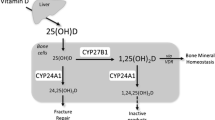
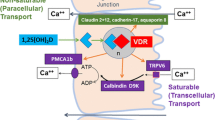
The skeleton provides rigid mechanical support to the body, protects vital organs, and serves as a reservoir of ions, especially for calcium and phosphate required for serum homeostasis. The integrity of the skeleton is maintained by continuous remodeling of bone tissues throughout life in response to a broad spectrum of physiologic signals. As described in Chapter 9, the active hormone 1,25-dihydroxyvitamin D3 (vitamin D) plays a key role in the maintenance of calcium and phosphate blood levels. In response to reduced serum calcium levels, calcium transport is stimulated across the gut and from the renal tubular lumen into the bloodstream. At the same time, calcium is mobilized from bone. Vitamin D actively promotes the release of bone mineral into the circulation by direct effects on the several cellular populations that reside in bone. The hormone influences differentiation and activity of cells of the osteoblast lineage, which form the mineralized bone matrix and cells of the osteoclast lineage, which resorb the mineralized bone (Fig. 1). Vitamin D exerts its effects on these cells by modulating the transcription of a broad spectrum of genes related to these bone cell phenotypes (1). How vitamin D mediates resorption of the bone matrix and subsequent bone formation through complex interactions between different populations of bone cells and at the level of regulation of gene expression is the primary subject of this chapter. The molecular mechanisms contributing to vitamin D-dependent transcription of the bone-specific osteocalcin gene have provided new insights for understanding steroid hormone responses in relation to a broad spectrum of physiologic conditions and phenotypic properties of a cell.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Hannah SS, Norman AW. 1-Alpha,25(OH)2 vitamin D3-regulated expression of the eukaryotic genome. Nutr Rev 1994; 52: 376–382.
Anderson HC. Molecular biology of matrix vesicles. Clin Orthop 1995; 314: 266–280.
Boskey AL. Mineral-matrix interactions in bone and cartilage. Clin Orthop 1992; 281: 244–274.
Gehron Robey P. Bone matrix proteoglycans and glycoproteins. In: Principles of Bone Biology. Bilezikian JP, Raisz LG, Rodan GA, eds. San Diego: Academic Press, 1996; 155–165.
Hui M, Li SQ, Holmyard D, Cheng P. Stable transfection of nonosteogenic cell lines with tissue nonspecific alkaline phosphatase enhances mineral deposition both in the presence and absence of beta-glycerophosphate: possible role for alkaline phosphatase in pathological mineralization. Calcif Tissue Int 1997; 60: 467–472.
Lian JB, Stein GS. Vitamin D regulation of osteoblast growth and differentiation. In: Nutrition and Gene Expression. Berdanier CD, Hargrove JL, eds. Boca Raton, FL: CRC Press, 1993; 391–429.
Boyan BD, Schwartz Z, Swain LD. In vitro studies on the regulation of endochondral ossification by vitamin D. Crit Rev Oral Biol Med 1992; 3: 15–30.
Wittenberg JB, Stein JL. Hemoglobin in the symbiont-harboring gill of the marine gastropod Alviniconcha hessleri. Biol Bull 1995; 188: 5–7.
Marie PJ, Hott M, Garba MT. Contrasting effects of 1,25-dihydroxyvitamin D3 on bone matrix and mineral appositional rates in the mouse. Metabolism 1985; 34: 777–783.
Hock JM, Gunness-Hey M, Poser J, Olson H, Bell NH, Raisz LG. Stimulation of undermineralized matrix formation by 1,25 dihydroxyvitamin D3 in long bones of rats. Calcif Tissue Int 1986; 38: 79–86.
Wronski TJ, Halloran BP, Bikle DD, Globus RK, Morey-Holton ER. Chronic administration of 1,25-dihydroxyvitamin D3: increased bone but impaired mineralization. Endocrinology 1986; 119: 2580–2585.
Burger EH, Klein-Nulend J, van der Plas A, Nijweide PJ. Function of osteocytes in bone-their role in mechanotransduction. J Nutr 1995; 125: 2020S - 20235.
Mundy GR, Boyce B, Hughes D, Wright K, Bonewald L, Dallas S, Harris S, Ghosh-Choudhury N, Chen D, Dunstan C, et al. The effects of cytokines and growth factors on osteoblastic cells. Bone 1995; 17: 71S - 715S.
Gerstenfeld LC, Zurakowski D, Schaffer JL, Nichols DP, Toma CD, Broess M, Bruder SP, Caplan AI. Variable hormone responsiveness of osteoblast populations isolated at different stages of embryo-genesis and its relationship to the osteogenic lineage. Endocrinology 1996; 137: 3957–3968.
Owen TA, Aronow MS, Barone LM, Bettencourt B, Stein GS, Lian JB. Pleiotropic effects of vitamin D on osteoblast gene expression are related to the proliferative and differentiated state of the bone cell phenotype: dependency upon basal levels of gene expression, duration of exposure, and bone matrix competency in normal rat osteoblast cultures. Endocrinology 1991; 128: 1496–1504.
Suda T, Takahashi N, Etsuko A. Role of vitamin D in bone resorption. J Cell Biochem 1992; 49: 53–58.
Roodman GD. Advances in bone biology: the osteoclast. Endocr Rev 1996; 17: 308–332.
Baron R, Neff L, Louvard D, Courtoy PJ. Cell-mediated extracellular acidification and bone resorption: evidence for a low pH in resorbing lacunae and localization of a 100-kD lysosomal membrane protein at the osteoclast ruffled border. J Cell Biol 1985; 101: 2210–2222.
Noda M, Vogel R, L., Craig AM, Prahl J, DeLuca HF, Denhardt D. Identification of a DNA sequence responsible for binding of the 1,25-dihydroxyvitamin D3 receptor and 1,25-dihydroxyvitamin D3 enhancement of mouse secreted phosphoprotein 1 (Spp-1 or osteopontin) gene expression. Proc Natl Acad Sci USA 1990; 87: 9995–9999.
Cao X, Ross FP, Zhang L, MacDonald PN, Chappel J, Teitelbaum SL. Cloning of the promoter for the avian integrin beta 3 subunit gene and its regulation by 1,25-dihydroxyvitamin D3. J Biol Chem 1993; 268:27, 371–27, 380.
Reinholt FP, Hultenby K, Oldberg A, Heinegard D. Osteopontin-a possible anchor of osteoclasts to bone. Proc Natl Acad Sci USA 1990; 87: 4473–4475.
Grano M, Zigrino P, Colucci S, Zambonin G, Trusolino L, Serra M, Baldini N, Teti A, Marchisio PC, Zallone AZ. Adhesion properties and integrin expression of cultured human osteoclast-like cells. Exp Cell Res 1994; 212: 209–218.
Oursler MJ. Osteoclast synthesis and secretion and activation of latent transforming growth factor beta. J Bone Miner Res 1994; 9: 443–252.
Lian JB, Stein GS. Osteoblast biology. In: Osteoporosis. Marcus R, Feldman D, Kelsey J, eds. San Diego: Academic Press, 1996; 23–60.
Aubin JE, Liu F. The osteoblast lineage. In: Principles of Bone Biology. Bilezikian JP, Raisz LG, Rodan GA, eds. San Diego: Academic Press, 1996; 51–68.
McCabe LR, Kockx M, Lian J, Stein J, Stein G. Selective expression of fos-and jun-related genes during osteoblast proliferation and differentiation. Exp Cell Res 1995; 218: 255–262.
Hoffmann HM, Catron KM, van Wijnen AJ, McCabe LR, Lian JB, Stein GS, Stein JL. Transcriptional control of the tissue-specific, developmentally regulated osteocalcin gene requires a binding motif for the Msx family of homeodomain proteins. Proc Natl Acad Sci USA 1994; 91:12, 887–12, 891.
Ryoo H-M, Hoffmann HM, Beumer TL, et al. Stage specific expression of Dlx-5 during osteoblast differentiation: involvement in regulation of osteocalcin gene expression. Mol Endocrinol 1997; 11: 1681–1694.
Komori T, Yagi H, Nomura S, Yamaguchi A, Sasaki K, Deguchi K, Shimizu Y, Bronson RT, Gao Y-H, Inada M, Sato M, Okamoto R, Kitamura Y, Yoshiki S, Kishimoto T. Targeted disruption of Cbfal results in a complete lack of bone formation owing to maturational arrest of osteoblasts. Cell 1997; 89: 755–764.
Banerjee C, McCabe LR, Choi J-Y, Hiebert SW, Stein JL, Stein GS, Lian JB. Runt homology domain proteins in osteoblast differentiation: AML-3/CBFA1 is a major component of a bone specific complex. J Cell Biochem 1997; 66: 1–8.
Ducy P, Zhang R, Geoffroy V, Ridall AL, Karsenty G. Osf2/Cbfal: a transcriptional activator of osteoblast differentiation. Cell 1997; 89: 747–754.
Pockwinse SM, Stein JL, Lian JB, Stein GS. Developmental stage-specific cellular responses to vitamin D and glucocorticoids during differentiation of the osteoblast phenotype: interrelationship of morphology and gene expression by in situ hybridization. Exp Cell Res 1995; 216: 244–260.
Glowacki J, Rey C, Glimcher MJ, Cox KA, Lian J. A role for osteocalcin in osteoclast differentiation. J Cell Biochem 1991; 45: 292–302.
Ishida H, Bellows CG, Aubin JE, Heersche JN. Characterization of the 1,25-(OH)2D3-induced inhibition of bone nodule formation in long-term cultures of fetal rat calvaria cells. Endocrinology 1993; 132: 61–66.
Lian JB, Stein GS. The developmental stages of osteoblast growth and differentiation exhibit selective responses of genes to growth factors (TGF beta 1) and hormones (vitamin D and glucocorticoids). J Oral Implantol 1993; 19: 95–105.
Candeliere GA, Prud’homme J, St-Arnaud R. Differential stimulation of fos and jun family members by calcitrol in osteoblastic cells. Mol Endocrinol 1991; 5: 1780–1788.
Wong M-M, Rao LG, Ly H, Hamilton L, Tong J, Sturtridge W, McBroom R, Aubin JE, Murray TM. Long-term effects of physiologic concentrations of dexamethasone on human bone-derived cells. J Bone Miner Res 1990; 5: 803–813.
Matsumoto T, Igarashi C, Takeuchi Y, Harada S, Kikuchi T, Yamato H, Ogata E. Stimulation by 1,25-dihydroxyvitamin D3 of in vitro mineralization induced by osteoblast-like MC3T3–E1 cells. Bone 1991; 12: 27–32.
Stein GS, Lian JB. Molecular mechanisms mediating proliferation-differentiation interrelationships during progressive development of the osteoblast phenotype: update. 1995. Endocr Rev 1995; 4: 290–297.
Merriman HL, van Wijnen AJ, Hiebert S, Bidwell JP, Fey E, Lian J, Stein J, Stein GS. The tissue-specific nuclear matrix protein, NMP-2, is a member of the AML/CBF/PEBP2/runt domain transcription factor family: interactions with the osteocalcin gene promoter. Biochemistry 1995; 34:13, 125–13, 132.
Banerjee C, Hiebert SW, Stein JL, Lian JB, Stein GS. An AML-1 consensus sequence binds an osteoblast-specific complex and transcriptionally activates the osteocalcin gene. Proc Natl Acad Sci USA 1996; 93: 4968–4973.
Ducy P, Karsenty G. Two distinct osteoblast-specific cis-acting elements control expression of a mouse osteocalcin gene. Mol Cell Biol 1995; 15: 1858–1869.
Bidwell JP, van Wijnen AJ, Fey EG, Dworetzky S, Penman S, Stein JL, Lian JB, Stein GS. Osteocalcin gene promoter-binding factors are tissue-specific nuclear matrix components. Proc Natl Acad Sci USA 1993; 90: 3162–3166.
Lian JB, Stein GS, Stein JL, van Wijnen AJ, McCabe L, Banerjee C, Hoffmann H. The osteocalcin gene promoter provides a molecular blueprint for regulatory mechanisms controlling bone tissue formation: role of transcription factors involved in development. Conn Tissue Res 1996; 35: 15–21.
Hoffmann HM, Beumer TL, Rahman S, McCabe LR, Banerjee C, Aslam F, Tiro JA, van Wijnen AJ, Stein JL, Stein GS, Lian JB. Bone tissue-specific transcription of the osteocalcin gene: role of an activator osteoblast-specific complex and suppressor hox proteins that bind the OC box. J Cell Biochem 1996; 61: 310–324.
Hodgkinson JE, Davidson CL, Beresford J, Sharpe PT. Expression of a human homeobox-containing gene is regulated by 1,25(OH)2D3 in bone cells. Biochim Biophys Acta 1993; 1174: 11–116.
Zhang R, Ducy P, Karsenty G. 1,25-dihydroxyvitamin D3 inhibits osteocalcin expression in mouse through an indirect mechanism. J Biol Chem 1997; 272: 110–116.
Banerjee C, Stein JL, van Wijnen AJ, Frenkel B, Lian JB, Stein GS. Transforming growth factor-beta 1 responsiveness of the rat osteocalcin gene is mediated by an activator protein-1 binding site. Endocrinology 1996; 137: 1991–2000.
Newberry EP, Boudreaux JM, Towler DA. The rat osteocalcin fibroblast growth factor (FGF)-responsive element: an okadaic acid-sensitive, FGF-selective transcriptional response motif. Mol Endocrinol 1996; 10: 1029–1040.
Rhodes SJ, Chen R, DiMattia GE, Scully KM, Kalla KA, Lin SC, Yu VC, Rosenfeld MG. A tissue-specific enhancer confers Pit- 1-dependent morphogen inducibility and autoregulation on the pit-1 gene. Genes Dev 1993; 7: 913–932.
Candeliere GA, Jurutka PW, Haussler MR, St-Arnaud R. A composite element binding the vitamin D receptor, retinoid X receptor alpha, and a member of the CTF/NF-1 family of transcription factors mediates the vitamin D responsiveness of the c-fos promoter. Mol Cell Biol 1996; 16: 584–592.
Pavlin D, Bedalov A, Kronenberg MS, Kream BE, Rowe DW, Smith CL, Pike JW, Lichtler AC. Analysis of regulatory regions in the COL1A1 gene responsible for 1,25-dihydroxyvitamin D3-mediated transcriptional repression in osteoblastic cells. J Cell Biochem 1994; 56: 490–501.
Ohyama Y, Ozono K, Uchida M, Shinki T, Kato S, Suda T, Yamamoto O, Noshiro M, Kato Y. Identification of a vitamin D-responsive element in the 5’-flanking region of the rat 25-hydroxyvitamin D3 24-hydroxylase gene. J Biol Chem 1994; 269:10, 545–10, 550.
Hahn CN, Kerry DM, Omdahl JL, May BK. Identification of a vitamin D responsive element in the promoter of the rat cytochrome P450(24) gene. Nucleic Acids Res 1994; 22: 2410–2416.
Medhora MM, Teitelbaum S, Chappel J, Alvarez J, Mimura H, Ross FP, Hruska K. 1 Alpha,25-dihydroxyvitamin D3 up-regulates expression of the osteoclast integrin alpha v beta 3. J Biol Chem 1993; 268: 1456–1461.
Mimura H, Cao X, Ross FP, Chiba M, Teitelbaum SL. 1,25-Dihydroxyvitamin D3 transcriptionally activates the beta 3-integrin subunit gene in avian osteoclast precursors. Endocrinology 134: 1994; 1061–1066.
Yu VC, Delsert C, Andersen B, Holloway JM, Devary OV, Naar AM, Kim SY, Boutin JM, Glass CK, Rosenfeld MG. RXR beta: a coregulator that enhances binding of retinoic acid, thyroid hormone, and vitamin D receptors to their cognate response elements. Cell 1991; 67: 1251–1266.
Kliewer SA, Umesono K, Mangelsdorf DJ, Evans RM. Retinoid X receptor interacts with nuclear receptors in retinoic acid, thyroid hormone and vitamin D3 signalling. Nature 1992; 355: 446–449.
Kerner SA, Scott RA, Pike JW. Sequence elements in the human osteocalcin gene confer basal activation and inducible response to hormonal vitamin D3. Proc Natl Acad Sci USA 1989; 86: 4455–4459.
Demay MB, Kiernan MS, DeLuca HF, Kronenberg HM. Sequences in the human parathyroid hormone gene that bind the 1, 25-dihydroxyvitamin D3 receptor and mediate transcriptional repression in response to 1,25-dihydroxyvitamin D3. Proc Natl Acad Sci USA 1992; 89: 8097–8101.
Markose ER, Stein JL, Stein GS, Lian JB. Vitamin D-mediated modifications in protein-DNA interactions at two promoter elements of the osteocalcin gene. Proc Natl Acad Sci USA 1990; 87: 1701–1705.
Ozono K, Liao J, Kerner SA, Scott RA, Pike JW. The vitamin D-responsive element in the human osteocalcin gene. Association with a nuclear proto-oncogene enhancer. J Biol Chem 1990; 265: 21, 881–21, 888.
Mader S, Chen JY, Chen Z, White J, Chambon P, Gronemeyer H. The patterns of binding of RAR, RXR and TR homo-and heterodimers to direct repeats are dictated by the binding specificites of the DNA binding domains. EMBO J 1993; 12: 5029–5041.
Perlmann T, Rangarajan PN, Umesono K, Evans RM. Determinants for selective RAR and TR recognition of direct repeat HREs. Genes Dev 1993; 7: 1411–1422.
Kurokawa R, Yu VC, Naar A, Kyakumoto S, Han Z, Silverman S, Rosenfeld MG, Glass CK. Differential orientations of the DNA-binding domain and carboxy-terminal dimerization interface regulate binding site selection by nuclear receptor heterodimers. Genes Dev 1993; 7: 1423–1435.
Arbour NC, Prahl JM, DeLuca HF. Stabilization of the vitamin D receptor in rat osteosarcoma cells through the action of 1,25-dihydroxyvitamin D3. Mol Endocrinol 1993; 7: 1307–1312.
Breen EC, van Wijnen AJ, Lian JB, Stein GS, Stein JL. In vivo occupancy of the vitamin D responsive element in the osteocalcin gene supports vitamin D-dependent transcriptional upregulation in intact cells. Proc Natl Acad Sci USA 1994; 91:12,902–12,906.
Hsieh JC, Jurutka PW, Galligan MA, Terpening CM, Haussler CA, Samuels DS, Shimizu Y, Shimizu N, Haussler MR. Human vitamin D receptor is selectively phosphorylated by protein kinase C on serine 51, a residue crucial to its trans-activation function. Proc Natl Acad Sci USA 1991; 88: 9315–9319.
Schräder M, Muller KM, Nayeri S, Kahlen JP, Carlberg C. Vitamin D3-thyroid hormone receptor heterodimer polarity directs ligand sensitivity of transactivation. Nature 1994; 370: 382–386.
Liao J, Ozono K, Sone T, McDonnell DP, Pike JW. Vitamin D receptor interaction with specific DNA requires a nuclear protein and 1,25-dihydroxyvitamin D3. Proc Natl Acad Sci USA 1990; 87: 9751–9755.
Liu M, Freedman LP. Transcriptional synergism between the vitamin D3 receptor and other nonreceptor transcription factors. Mol Endocrinol 1994; 8: 1593–1604.
St-Arnaud R, Prud’homme J, Leung-Hagesteijn C, Dedhar S. Constitutive expression of calreticulin in osteoblasts inhibits mineralization. J Cell Biol 1995; 131: 1351–1359.
MacDonald PN, Dowd DR, Nakajima S, Galligan MA, Reeder MC, Haussler CA, Ozato K, Haussler MR. Retinoid X receptors stimulate and 9-cis retinoic acid inhibits 1,25-dihydroxyvitamin D3-activated expression of the rat osteocalcin gene. Mol Cell Biol 1993; 13: 5907–5917.
Zhang XK, Lehmann J, Hoffmann B, Dawson MI, Cameron J, Graupner G, Hermann T, Tran P, Pfahl M. Homodimer formation of retinoid X receptor induced by 9-cis retinoic acid. Nature 1992; 358: 587–591.
Eisman JA. Vitamin D receptor gene alleles and osteoporosis: an affirmative view [editorial]. J Bone Miner Res 1995; 10: 1289–1293.
Morrison NA, Qi JC, Tokita A, Kelly PJ, Crofts L, Nguyen TV, Sambrook PN, Eisman JA. Prediction of bone density from vitamin D receptor alleles [see comments]. Nature 1994; 367: 284–287.
Uitterlinden AG, Pols HA, Burger H, Huang Q, Van Daele PL, Van Duijn CM, Hofman A, Birkenhager JC, Van Leeuwen JP. A large-scale population-based study of the association of vitamin D receptor gene polymorphisms with bone mineral density. J Bone Miner Res 1996; 11: 1241–1248.
Guo B, Aslam F, van Wijnen AJ, Roberts SGE, Frenkel B, Green M, DeLuca H, Lian JB, Stein GS, Stein JL. YY1 regulates VDR/RXR mediated transactivation of the vitamin D responsive osteocalcin gene. Proc Natl Acad Sci USA 1997; 94: 121–126.
Aslam F, McCabe LR, Frenkel B, van Wijnen AJ, Stein GS, Lian JB, and Stein JL. Convergence of AP-1 and vitamin D receptor (VDR) signalling pathways at the rat osteocalcin VDR element: requirement of the internal AP-1 site for vitamin D-mediated transactivation. Submitted.
Lian JB, Stein GS, Bortell R, Owen TA. Phenotype suppression: a postulated molecular mechanism for mediating the relationship of proliferation and differentiation by Fos/Jun interactions at AP-1 sites in steroid responsive promoter elements of tissue-specific genes. J Cell Biochem 1991; 45: 9–14.
Owen TA, Bortell R, Yocum SA, Smock SL, Zhang M, Abate C, Shalhoub V, Aronin N, Wright KL, van Wijnen AJ, Stein JL, Curran T, Lian JB, Stein GS. Coordinate occupancy of AP-1 sites in the vitamin D responsive and CCAAT box elements by Fos-Jun in the osteocalcin gene: model for phenotype suppression of transcription. Proc Natl Acad Sci USA 1990; 87: 9990–9994.
Blanco JCG, Wang I-M, Tsai SY, Tsai M-J, O’Malley BW, Jurutka PW, Haussler MR, Ozato K. Transcription factor TFIIB and the vitamin D receptor cooperatively activate ligand-dependent transcription. Proc Natl Acad Sci USA 1995; 92: 1535–1539.
MacDonald PN, Sherman DR, Dowd DR, Jefcoat SCJ, DeLisle K. The vitamin D receptor interacts with general transcription factor IIB. J Biol Chem 1995; 270: 4748–4752.
Usheva A, Shenk T. TATA-binding protein-independent initiation: YY1, TFIIB, and RNA polymerase II direct basal transcription on supercoiled template DNA. Cell 1994; 76: 1115–1121.
Staal A, van Wijnen AJ, Birkenhäger JC, Pols HAP, Prahl J, DeLuca H, Gaub M-P, Lian JB, Stein GS, van Leeuwen JPTM, Stein JL. Distinct conformations of VDR/RXRa heterodimers are specified by dinucleotide differences in the vitamin D responsive elements of the osteocalcin and osteopontin genes. Mol Endocrinol 1996; 10: 1444–1456.
Kesterson RA, Stanley L, DeMayo F, Finegold M, Pike JW. The human osteocalcin promoter directs bone-specific vitamin D-regulatable gene expression in transgenic mice. Mol Endocrinol 1993; 7: 462–467.
Baker AR, Hollingshead PG, Pitts-Meek S, Hansen S, Taylor R, Stewart TA. Osteoblast-specific expression of growth hormone stimulates bone growth in transgenic mice. Mol Cell Biol 1992; 12: 5541–5547.
Towler DA, Bennett CD, Rodan GA. Activity of the rat osteocalcin basal promoter in osteoblastic cells is dependent upon homeodomain and CP1 binding motifs. Mol Endocrinol 1994; 8: 614–624.
Uchida M, Ozono K, Pike JW. Activation of the human osteocalcin gene by 24R,25-dihydroxyvitamin D3 occurs through the vitamin D receptor and the vitamin D-responsive element. J Bone Miner Res 1994; 9: 1981–1987.
Lian JB, Shalhoub V, Aslam F, Frenkel B, Green J, Hamrah M, Stein GS, Stein JL. Species-specific glucocorticoid and 1,25-dihydroxyvitamin D responsiveness in mouse MC3T3–E1 osteoblasts: dexamethasone inhibits osteoblast differentiation and vitamin D downregulates osteocalcin gene expression. Endocrinology 1997; 138: 2117–2127.
Montecino M, Pockwinse S, Lian J, Stein G, Stein J. DNase I hypersensitive sites in promoter elements associated with basal and vitamin D dependent transcription of the bone-specific osteocalcin gene. Biochemistry 1994; 33: 348–353.
Montecino M, Lian J, Stein G, Stein J. Changes in chromatin structure support constitutive and develomentally regulated transcription of the bone-specific osteocalcin gene in osteoblastic cells. Biochemistry 1996; 35: 5093–5102.
Dworetzky SI, Fey EG, Penman S, Lian JB, Stein JL, Stein GS. Progressive changes in the protein composition of the nuclear matrix during rat osteoblast differentiation. Proc Natl Acad Sci USA 1990; 87: 4605–4609.
Zeng C, van Wijnen Ai, Stein JL, Meyers S, Sun W, Shopland L, Lawrence JB, Penman S, Lian JB, Stein GS, Hiebert SW. Identification of a nuclear matrix targeting signal in the leukemia and bone-related AML/CBFa transcription factors. Proc Natl Acad Sci USA 1997; 94: 6746–6751.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1999 Springer Science+Business Media New York
About this chapter
Cite this chapter
Lian, J.B., Staal, A., van Wijnen, A., Stein, J.L., Stein, G.S. (1999). Biologic and Molecular Effects of Vitamin D on Bone. In: Holick, M.F. (eds) Vitamin D. Nutrition and Health. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-4757-2861-3_11
Download citation
DOI: https://doi.org/10.1007/978-1-4757-2861-3_11
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-4757-2863-7
Online ISBN: 978-1-4757-2861-3
eBook Packages: Springer Book Archive