Abstract
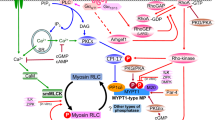
Cyclic AMP-dependent protein kinase phosphorylates myosin light chain kinase purified from gizzard smooth muscle (Adelstein et al., 1978). In the absence of Ca2+/calmodulin, 2 sites (sites A and B) are phosphorylated and the concentration of Ca2+/calmodulin required for half-maximal activation (KcaM) increases 10 fold (Conti and Adelstein, 1981). In the presence of Ca2+/calmodulin, phosphate is incorporated into site B with no effect on myosin light chain kinase activity. As shown in Figure 1, site A is near the calmodulin binding domain whereas site B is more toward the c-terminus. Similar observations on kinase phosphorylation have been made with myosin light chain kinases purified from mammalian smooth muscles including trachea (Miller et al., 1983), stomach (Walsh et al., 1982), myometrium (Higashi et al., 1983), aorta (Vallet et al., 1981), and carotid artery (Bhalla et al., 1982). It has been proposed that phosphorylation of site A by cyclic AMP-dependent protein kinase could decrease the extent of myosin light chain kinase activation with a resultant decrease in myosin light chain phosphorylation and inhibition of contraction (Conti and Adelstein, 1981).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Adelstein, R. S., Conti, M. A., Hathaway, D. R., and Klee, C. B., 1978, Phosphorylation of smooth muscle myosin light chain kinase by the catalytic subunit of adenosine 3′: 5′-monophosphate-dependent protein kinase, J. Biol. Chem., 253: 8347.
Bhalla, R. C., Sharma, R. V., and Gupta, R. C., 1982, Isolation of two myosin light-chain kinases from bovine carotid artery and their regulation by phosphorylation mediated by cyclic AMP-dependent protein kinase, Biochem. J., 203: 583.
Cohen, P., 1989, The structure and regulation of protein phosphatases, Ann. Rev. Biochem., 58: 453.
Conti, M. A. and Adelstein, R. S., 1981, The relationship between calmodulin binding and phosphorylation of smooth muscle myosin kinase by the catalytic subunit of 3′: 5′ cAMP-dependent protein kinase, J. Biol. Chem., 256: 3178.
de Lanerolle, P., Nishikawa, M., Yost, D. A., and Adelstein, R. S., 1984, Increased phosphorylation of myosin light chain kinase after an increase in cyclic AMP in intact smooth muscle, Science, 223: 1415.
Hashimoto, Y. and Soderling, T. R., 1990, Phosphorylation of smooth muscle myosin light chain kinase by Ca2+/calmodulin-dependent protein kinase-II. Comparative study of the phosphorylation sites, Arch. Biochem. Biophys., 278: 41.
Higashi, K., Fukunaga, K., Matsui, K., Maeyama, M., and Miyamoto, E., 1983, Purification and characterization of myosin light-chain kinase from porcine myometrium and its phosphorylation and modulation by cyclic AMP-dependent protein kinase, Biochim. Biophys. Acta, 747: 232.
Himpens, B., Matthijs, G., and Somlyo, A. P., 1989, Desensitization to cytoplasmic Ca2+ and Ca2+ sensitivities of guinea-pig ileum and rabbit pulmonary artery smooth muscle, J. Physiol, 413: 489.
Ikebe, M., Inagaki, M., Kanamaru, K., and Hidaka, H., 1985, Phosphorylation of smooth muscle myosin light chain kinase by Ca2+-activated, phospho-lipid-dependent protein kinase, J. Biol. Chem., 260: 4547.
Kamm, K. E. and Stull, J. T., 1985, The function of myosin and myosin light chain kinase phosphorylation in smooth muscle, Ann. Rev. Pharmacol. Toxicol., 25: 593.
Kamm, K. E., Hsu, L.-C., Kubota, Y., and Stull, J. T., 1989, Phosphorylation of smooth muscle myosin heavy and light chains: Effects of phorbol dibutyrate and agonists, J. Biol Chem., 264: 21223.
Kamm, K. E. and Stull, J. T., 1989, Regulation of smooth muscle contractile elements by second messengers, Ann. Rev. Physiol, 51: 299.
Karaki, H., 1989, Ca2+ localization and sensitivity in vascular smooth muscle, Trends Pharmacol Sci., 10: 320.
Lukas, T. J., Burgess, W. H., Prendergast, F. G., Lau, W., and Watterson, D. M, 1986, Calmodulin binding domains: Characterization of a phosphorylation and calmodulin binding site from myosin light chain kinase, Biochemistry, 25: 1458.
Miller, J. R., Silver, P. J., and Stull, J. T., 1983, The role of myosin light chain kinase phosphorylation in β-adrenergic relaxation of tracheal smooth muscle, Mol Pharmacol., 24: 235.
Morgan, J. P. and Morgan, K. G., 1984, Stimulus-specific patterns of intracellular calcium levels in smooth muscle of ferret portal vein, J. Physiol, 351: 155.
Nishikawa, M., Shirakawa, S., and Adelstein, R. S., 1985, Phosphorylation of smooth muscle myosin light chain kinase by protein kinase C. Comparative study of the phosphorylated sites, J. Biol. Chem., 260: 8978.
Pato, M. D. and Adelstein, R. S., 1983, Characterization of a Mg2+-dependent phosphatase from turkey gizzard smooth muscle, J. Biol Chem., 258: 7055.
Ratz, P. H. and Murphy, R. A., 1987, Contributions of intracellular and extracellular Ca2+ pools to activation of myosin phosphorylation and stress in swine carotid media, Circ. Res., 60: 410.
Rembold, C. M. and Murphy, R. A., 1988, Myoplasmic [Ca2+] determines myosin phosphorylation in agonist-stimulated swine arterial smooth muscle, Circ. Res., 63: 593.
Stull, J. T., Nunnally, M. H., and Michnoff, C. H., 1986, Calmodulin-dependent protein kinases, in: “The Enzymes”, E. G. Krebs, P. D. Boyer, eds., Academic Press, Orlando, p. 113.
Stull, J. T., Hsu, L.-C., Tansey, M. G., and Kamm, K. E., 1990, Myosin light chain kinase phosphorylation in tracheal smooth muscle, J. Biol. Chem., 265: 16683.
Sutton, T. A. and Haeberle, J. R., 1990, Phosphorylation by protein kinase C of the 20,000-dalton light chain of myosin in intact and chemically skinned vascular smooth muscle, J. Biol. Chem., 265: 2749.
Vallet, B., Molla, A., and Demaille, J. G., 1981, Cyclic adenosine 3′,5′-monophosphate-dependent regulation of purified bovine aortic calcium/calmodulin-dependent myosin light chain kinase, Biochim. Biophys. Acta, 674: 256.
Walsh, M. P., Hinkins, S., Flink, I. L., and Hartshorne, D. J., 1982, Bovine stomach myosin light chain kinase: Purification, characterization, and comparison with the turkey gizzard enzyme, Biochemistry, 21: 6890.
Yagi, S., Becker, P. L., and Fay, F. S., 1988, Relationship between force and Ca2+ concentration in smooth muscle as revealed by measurements on single cells, Proc. Nat’l Acad. Sci. U.S.A., 85: 4109.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1991 Plenum Press, New York
About this chapter
Cite this chapter
Stull, J.T., Tansey, M.G., Word, R.A., Kubota, Y., Kamm, K.E. (1991). Myosin Light Chain Kinase Phosphorylation: Regulation of the Ca2+ Sensitivity of Contractile Elements. In: Moreland, R.S. (eds) Regulation of Smooth Muscle Contraction. Advances in Experimental Medicine and Biology, vol 304. Springer, Boston, MA. https://doi.org/10.1007/978-1-4684-6003-2_12
Download citation
DOI: https://doi.org/10.1007/978-1-4684-6003-2_12
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4684-6005-6
Online ISBN: 978-1-4684-6003-2
eBook Packages: Springer Book Archive