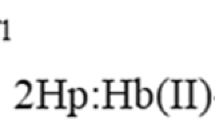Abstract
In 1969 a model was proposed (BARNIKOL et al.) to explain the effect of the Hb- concentration on the O2- Hb- binding curve (GROTE, 1967). The existence of at least one low molecular substance (called Z) besides H and CO2 was postulated, which is equimolar with Hb (≈ 5 mM) and which is bound preferentially to deoxy- Hb. From a simple stoichiometric consideration it follows, that a substance can only have a measurable influence on the O2- Hb- binding curve, if its molar concentration is not to low compared with the molar Hb- concentration. For example, 2,3- diphosphogly-cerate (= DPG), glutathione and Mg++ have intracellular concentrations of the necessary magnitude. Only a small part of Magnesium is bound to the membrane of the erythrocytes (HARRISON et al.). Principally all these substances are candidates for Z. In 1967 it was shown by BENESCH et al. and by CHANUTIN et al., that ATP and DPG influence the O2-Hb- binding curve as was postulated for Z, and it was shown (BENESCH et al., 1968), that the organic phosphates are bound preferentially to deoxy-Hb. In our speculation on the chemical nature of Z first we prefered Mg++ because of its complex forming properties. It is known for a long time, that Mg++ forms a complex with ATP (MARTELL et al.,1956) and in 1970 it was found, that DPG forms also a complex with Mg++ (COLLIER et al.).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Literature
G. Amiconi, E. Antonini, M. Brunori, J. Wyman in “Hemoglobin and Myoglobin in thin Reactions with ligands”, authors: E. Antonini and M. Brunori, North- Holland Publishing Company, Amsterdam — London, 1971, p.246
W.K.R. Barnikol, G. Thews Zur Interpretation der O2- Bindungskurve des Human- Hämoglobins. Pflügers Arch. 309, 232–249 (1969)
W.K.R. Barnikol, W. Wahler On the Accuracy of an Improved Method for the Measurement of O2- Dissociation Curves According to NIESEL and THEWS, 1961. Adv. in Exp. Medicine and Biology 37A, 325–331 (1973) (Editors: D.F. Bruley, H.I. Bicher), Plenum Press, New York- London
R. Benesch, R.E. Benesch The Effect of Organic Phosphates from the Human Erythrocyte on the Allosteric Properties of Hemoglobin. Biochem. Biophys. Res. Comm. 26, 162–167 (1967)
R. Benesch, R.E. Benesch, C.I. Yu Reciprocal Binding of Oxygen and Diphosphoglycerate by Human Hemoglobin. Proc. Nat. Acad. Sci. (Wash.) 59, 526–532 (1968)
H. Berger, G.R. Jänig, G. Gerber, K. Ruckpaul, S.M. Rapoport Interaction of Hemoglobin with Ions. Interactions among Magnesium, Adenosine 5- Triphosphate, 2,3- Biphosphoglycerate, and Oxygenated and Deoxygenated Human Hemoglobin under Simulated Intracellular Conditions. Eur. J. Biochem. 38, 553–562 (1973)
H.F. Bunn, B.J. Ransil, A. Chao The Interaction between Erythrocyte Organic Phosphates, Magnesium Ion, and Hemoglobin. J. Biol Chem. 246, 5273–5279 (1971)
A. Chanutin, R. Curnish Effect of Organic and Inorganic Phosphates on the Oxygen Equilibrium of Human Erythrocytes. Arch. Biochem. 121, 96–102 (1967)
H.B. Collier A. Lam Binding of Ca2+ and Mg2+ by 2,3 Diphosphoglycerate. Biochim. Biophys. Acta 222, 299–306 (1970)
L. Garby, G. Gerber, C.H. de Verdier Binding of 2,3 — Diphosphoglycerate and Adenosine Triphosphate to Human Hemoglobin A. Eur. J. Biochem. 10, 110–115 (1969)
L. Garby, C.H. de Verdier Affinity of Human Hemoglobin to 2,3- Diphosphoglycerate. Effect of Hemoglobin Concentration and pH. Scand. J. Clin. Lab. Invest. 27, 345–350 (1971)
J. Grote Die Bestimmung der Sauerstoff- Bindungskurve von hochverdünnten Hämoglobinlösungen. Pflügers Arch. 296, 202–211 (1967)
D.G. Harrison, C. Long The Calcium Content of Human Erythrocytes. J. Physiol. 199, 367–381 (1968)
A.E. Martel I, G. Schwarzenbach Adenosinphosphate und Triphosphat als Komplexbildner für Calcium und Magnesium. Helv. Chim. Acta 39, 653–661 (1956)
J.R. Murphy The Influence of pH on Physical Properties of the Erythrocyte. in “Stoffwechsel- und Membranpermeabilitäten von Erythrocyten und Thrombocyten”, Thieme, Stuttgart 1968, p. 452
O. Siggaard- Andersen Oxygen- Linked Hydrogen Ion Binding of Human Hemoglobin. Effects of Carbon- Dioxide and 2,3- Diphosphoglycerate. I. Studies on Erythrolysate. Scand. J. Clin. Lab. Invest. 27, 351–360 (1971)
O. Warburg, W. Christian Isolierung und Kristallisation des Gärungsfermentes Enolase. Biochem. Zeitschr. 310, 384–425 (1942)
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1976 Plenum Press, New York
About this chapter
Cite this chapter
Barnikol, W. (1976). The Influence of MgCl2 on the O2-Hb-Binding Curve of Human Hemoglobin Under Intracellular Conditions. In: Grote, J., Reneau, D., Thews, G. (eds) Oxygen Transport to Tissue — II. Advances in Experimental Medicine and Biology, vol 75. Springer, Boston, MA. https://doi.org/10.1007/978-1-4684-3273-2_13
Download citation
DOI: https://doi.org/10.1007/978-1-4684-3273-2_13
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4684-3275-6
Online ISBN: 978-1-4684-3273-2
eBook Packages: Springer Book Archive