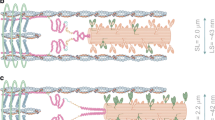
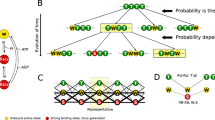
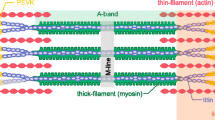
Abstract
Energy consumption in the cardiac muscle is characterized by two basic phenomena: 1) The well known linear relationship between energy consumption by the sarcomere and the mechanical energy it generates, and 2) the ability to modulate the generated mechanical energy and energy consumption to the various loading conditions, as is manifested by the Frank-Starling Law and the Fenn effect. These basic phenomena are analyzed here based on coupling calcium kinetics with crossbridge (Xb) cycling. Our previous studies established the existence of two feedback mechanism: 1) a positive feedback mechanism, the cooperativity, whereby the affinity of the troponin for calcium, and hence Xb and actomyosin-ATPase recruitment, depends on the number of force generating Xbs, and 2) a mechanical feedback, whereby the filaments shortening velocity, or the Xb strain rate, determines the rate of Xb turnover from the strong to the weak conformation. The cooperativity mechanism determines the force-length relationship (FLR) and the related FrankStarling Law. It also provides the basis for the regulation of energy consumption and the ability of the muscle to adapt its energy consumption to the loading conditions. The mechanical feedback regulates the shortening velocity and provides the analytical solution for the experimentally derived Hill’s equation for the force-velocity relationship (FVR). The mechanical feedback regulates the generated power and provides the linear relationship between energy consumption and the generated mechanical energy, i.e., the external work done and the liberated heat. Thus, the two feedback mechanisms that regulate sarcomere dynamics, and determine the FLR and FVR, also regulate the energy consumption and the mechanical energy generated by the muscle.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Suga Y. Ventricular energy. Physiological Rev 1990;70:247–277.
Hisano G, Cooper IV. Correlation of force-length area with oxygen consumption in ferret papillary muscle. Circ Res 1987;61:318–328.
Mommaerts WFHM, Seraydarian I, Marechal G. Work and mechanical change in isotonic muscular contractions. Biochem Biophys Acta 1962;57:1–12.
Fenn OW. A quantitative comparison between the energy liberation and the work performed by the isolated sartorious muscle of the frog. J Physiol London 1923;58:175–203.
Rall JA. Sense and nonsense about the Fenn effect. Am J Physiol 1982;242:H1–H6.
Landesberg A. End systolic pressure-volume relation based on the intracellular control of contraction. Am J Physiol 1996;270:H338–H349.
Landesberg A, Sideman S. Coupling calcium binding to Troponin-C and Xb cycling kinetics in skinned cardiac cells. Am J Physiol 1994;266(Heart Circ Physiol 35):H1261–H1271.
Landesberg A, Sideman S. Mechanical regulation in the cardiac muscle by coupling calcium binding to troponin-C and Xb cycling. A dynamic model. Am J Physiol 1994;267(Heart Circ Physiol 36): H779–H795.
Landesberg A, Sideman S. Regulation of energy consumption in the cardiac muscle; analysis of isometric contractions. Am J Physiol 1997; submitted.
Landesberg A, Burkhoff D, Sideman S. Calcium binding affinity based on stress-strain and free-calcium data from isolated ferret heart. Am J Physiol 1997; submitted.
Alpert NR, Mulieri LA, Hasenfuss G, Holubarsch C. Optimization of myocardial function. In: Burkhoff D, Schaefer J, Schaffner K, Yue DT, editors, Myocardial Optimization and Efficiency: Evolutionary Aspects and Philosophy of Science Considerations. Springer-Verlag: New York, (Basic Research in Cardiology, vol. 88, suppl. 2) 1993, pp. 29–41.
De Tombe PP, ter Keurs HEDJ. An internal viscous element limit unloaded velocity of sarcomere shortening in rat myocardium. J Physiol Lond 1992;454:619–642.
Daniels M, Nobel MIM, ter Kerus HEDJ, Wohlfart B. Velocity of sarcomere shortening in rat cardiac muscle: relationship to force, sarcomere length, calcium and time. J Physiol 1984;355;367–381.
Brenner B, Eisenberg E. Rate of force generation in muscle: correlation with actomyosin ATPase activity in solution. Proc Natl Acad Sci 1986;83:3542–3546.
Eisenberg E, Hill TL. Muscle contraction and free energy transduction in biological system. Science 1985;227:999–1006.
Chalovich JM, Eisenberg E. The effect of troponin — tropomyosin on the binding of heavy meromyosin to actin in the presence of ATP. J Biol Chem 1986;261:5088–5093.
Ford EL. Mechanical manifestations of activation in cardiac muscle. Circ Res 1991;68:621–637.
Campbell KB, Sharff SG, Kirkpatrick RD, Short time scale left ventricle systolic dynamics. Circ Res 1991;68:1532–1548.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1997 Springer Science+Business Media New York
About this chapter
Cite this chapter
Landesberg, A. (1997). Molecular Control of Myocardial Mechanics and Energetics: The Chemo-Mechanical Conversion. In: Sideman, S., Beyar, R. (eds) Analytical and Quantitative Cardiology. Advances in Experimental Medicine and Biology, vol 430. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-5959-7_7
Download citation
DOI: https://doi.org/10.1007/978-1-4615-5959-7_7
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-7731-3
Online ISBN: 978-1-4615-5959-7
eBook Packages: Springer Book Archive