Abstract
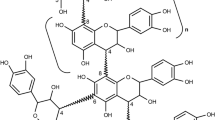
The condensed tannins are oligomers and polymers of polyhydroxyflavan-3-ol monomer units linked by acid-labile 4 → 6 or 4 → 8 bonds. The polymers are polydisperse, and the chains are irregular, sometimes branched, and often contain more than one type of configurational base unit. Condensed tannins are relatively unstable in aqueous solution and disproportionate in mild acids or bases. The monomer units extensively rearrange in stronger acidic or basic solutions. The reactions are largely radical-mediated in basic solutions to form highly rearranged and oxidatively coupled products. Condensed tannins complex strongly with metal ions, carbohydrates, and proteins.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Haslam, E. Plant polyphenols. Cambridge University Press, Cambridge, p 11 (1989)
Ishimaru, K.; Ishimatsu, M.; Nonaka, G.; Mihashi, K.; Iwase, Y.; Nishioka, I. Tannins and related compounds. LXXII. Isolation and characterisation of mongolicanin (procyanidino-ellagitannin), mongolinin A, acutissimin C and vescalagin carboxylic acid, novel tannins from Quercus mongolica var. grosseterrata. Chem. Pharm. Bull. 36:3319 (1988).
Nonaka, G.; Nishioka, L; Nagasawa, T.; Oura, H. Tannins and related compounds. I. Rhubarb (1). Chem. Pharm. Bull. 29:2862 (1981).
Porter, L.J. Flavans and proanthocyanidins. In: Harborne, J.B. (ed.). The flavonoids. Chapman and Hall, London, pp 21–62 (1988).
Roux, D.G.; Ferreira, D. Structure and function in the biomimetic synthesis of linear, angular, and branched condensed tannins. Pure Applied Chem. 54:2465 (1982).
Porter, L.J. Number-and weight-average molecular weights for some proanthocyanidin polymers (condensed tannins). Aust. J. Chem. 39:557 (1986).
Williams, V.M.; Porter, L.J.; Hemingway, R.W. Molecular weight profiles of proanthocyanidin polymers. Phytochemistry 22:569 (1983).
Mattice, W.L.; Porter, L.J. Molecular weight averages and 13C NMR intensities provide evidence for branching in proanthocyanidin polymers. Phytochemistry 23:1309 (1984).
Roux, D.G.; Ferreira, D.; Hundt, H.K.L.; Malan, E. Structure, stereochemistry, and reactivity of natural condensed tannins as a basis for their extended industrial application. Applied Polymer Symp. 28:335 (1975).
Kennedy, J.A.; Powell, H.K.J. Polyphenol interactions with aluminium (III) and iron (III): their possible involvement in the podzolization process. Aust. J. Chem. 38:879 (1985).
Powell, H.K.J.; Rate, A.W. Aluminium-tannin equilibria: a potentiometric study. Aust. J. Chem. 40:2015 (1987).
Matsuo, T.; Itoo, S. A simple and rapid purification method of condensed tannins from several young fruits. Agric. Biol. Chem. 45:1885 (1981).
Rahman, M.D.; Richards, G.N. Interaction of starch and other polysaccharides with condensed tannins in hot water extracts of ponderosa pine bark. J. Wood Chem. Technol. 8:111 (1988).
Sears, K.D.; Casebier, R.L. The reaction of thioglycollic acid with polyflavanoid bark fractions of Tsuga heterophylla. Phyto chemistry 9:1589 (1970).
Brieskorn, C.H.; Betz, R. Proanthocyanidin polymers critical to the structure of almond seed coat. Z. Lebensm-Unters Forsch 187:347 (1988).
Porter, L.J.; Hrstich, L.N.; Chan, B.G. The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 25:223 (1986).
Kennedy, J.A.; Munro, M.H.G.; Powell, H.K.J.; Porter, L.J.; Foo, L.Y. The protonation reactions of catechin, epicatechin, and related compounds. Aust. J. Chem. 37:885 (1984).
Nonaka, G.; Morimoto, S.; Kinjo, J.; Nohara, T.; Nishioka, I. Tannins and related compounds. L. Structures of proanthocyanidin A-l and related compounds. Chem. Pharm Bull 35:149 (1987).
Markham, K.R.; Porter, L.J. Extractives of Pinus radiata bark. 1. Phenolic components. N.Z. J. Science. 16:751 (1973).
Jacques, D.; Haslam, E.; Bedford, G.R.; Greatbanks, D. Plant proanthocyanidins, Part II. Proanthocyanidin-A2 and its derivatives. J. Chem. Soc, Perkin Trans. 1:2663 (1974).
Morimoto, S.; Nonaka, G.; Nishioka, I. Tannins and related compounds. LX. Isolation and characterisation of proanthocyanidins with a doubly-linked unit from Vaccinium vitis-idaca L. Chem. Pharm. Bull. 36:33 (1988).
Jensen, O.N.; Pedersen, J.A. The oxidative transformations of (+)catechin and (-)epicatechin as studied by esr. Tetrahedron 39:1609 (1983).
Hashimoto, F.; Nonaka, G.; Nishioka, I. (1988). Tannins and related compounds. LXIX. Isolation and structure elucidation of B, B/-linked bisflavonoids, theasinensins D-G and oo-longtheanin from oolong tea. (2) Chem. Pharm. Bull. 36:1676 (1988).
Young, D.A.; Young, E.; Roux, D.G.; Brandt, E.V.; Ferreira, D. Synthesis of condensed tannins. Part 19. Phenol oxidative coupling of (+)-catechin and (+)-mesquitol. Conformation of bis-(+)-catechins. J. Chem. Soc., Perkin Trans. 1:2345 (1987).
Ahn, B.; Gstirner, F. Uber catechin-dimere der eichenrinde. Arch. Pharm. 304:666 (1971).
Zhang, B.; Nonaka, G.; Nishioka, I. Potentillanin, a biflavanoid and a procyanidin glycoside from Potentilla viscosa. Phytochemistry 27:3277 (1988).
Coggon, P.; Moss, G.A.; Sanderson, G.W. Tea catechol oxidase: isolation, purification and kinetic characterisation. Phytochemistry 12:1947 (1973).
Laks, P.E.; Hemingway, R.W.; Conner, A.H. Condensed tannins. Base-catalysed reactions of polymeric procyanidins with phloroglucinol:intramolecular rearrangements. J. Chem. Soc., Perkin Trans. I:1875 (1987).
Laks, P.E.; Hemingway, R.W. Condensed tannins. Structure of the ‘phenolic acids’. Holz-forschung 41:287 (1987).
Laks, P.E.; Hemingway, R.W. Condensed tannins. Base-catalysed reactions of polymeric procyanidins with toluene-α-thiol. Lability of the interflavanoid bond and pyran ring. J. Chem. Soc., Perkin Trans. 1:465 (1987).
Waters, W.A. Mechanisms of oxidation of organic compounds. Methuen, London, pp 145–146 (1964).
Steynberg, J.P.; Burger, J.F.W.; Young, D.A.; Brandt, E.V.; Steenkamp, J.A.; Ferreira, D. Oligomeric flavonoids. Part 3. Structure and synthesis of phlobatannins related to (-)-fisetinidol-(4α,6)-and (4α,8)-(+)-catechin profisetinidins. J. Chem. Soc, Perkin Trans. I:3323 (1988).
Steynberg, J.P.; Burger, J.F.W.; Young, D.A.; Brandt, E.V.; Steenkamp, J.A.; Ferreira, D. Oligomeric flavanoids, Part 4. Base-catalysed conversions of (-)-fisetinidol-(+)-catechin profisetinidins with 2,3-trans-3,4-cis-flavan-3-ol constituent units. J. Chem. Soc, Perkin Trans. 1:3331 (1988).
Burger, J.F.W.; Steynberg, J.P.; Young, D.A.; Brandt, E.V.; Ferreira, D. Oligomeric flavonoids. Part 5. Base-catalyzed C-ring isomerization of (+)-fisetinidol-(+)-catechin profisetinidins. J. Chem. Soc., Perkin Trans. 7:671 (1989).
Porter, L.J.; Ma, Z.; Chan, B.G. Cacao procyanidins: major flavanoids and identification of some minor metabolites. Phytochemistry: 30:1657 (1991).
Morgan, K.R.; Newman, R.H. Estimation of the tannin content of eucalypts and other hardwoods by carbon-13 nuclear magnetic resonance. Appita 40:450 (1987).
Wilson, M.A.; Hatcher, P.G. Detection of tannins in modern and fossil barks and in plant residues by high-resolution solid-state 13C nuclear magnetic resonance. Org. Geochem. 12:539 (1988).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1992 Springer Science+Business Media New York
About this chapter
Cite this chapter
Porter, L.J. (1992). Structure and Chemical Properties of the Condensed Tannins. In: Hemingway, R.W., Laks, P.E. (eds) Plant Polyphenols. Basic Life Sciences, vol 59. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-3476-1_14
Download citation
DOI: https://doi.org/10.1007/978-1-4615-3476-1_14
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-6540-2
Online ISBN: 978-1-4615-3476-1
eBook Packages: Springer Book Archive